Submitted:
03 November 2023
Posted:
06 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Project Participants
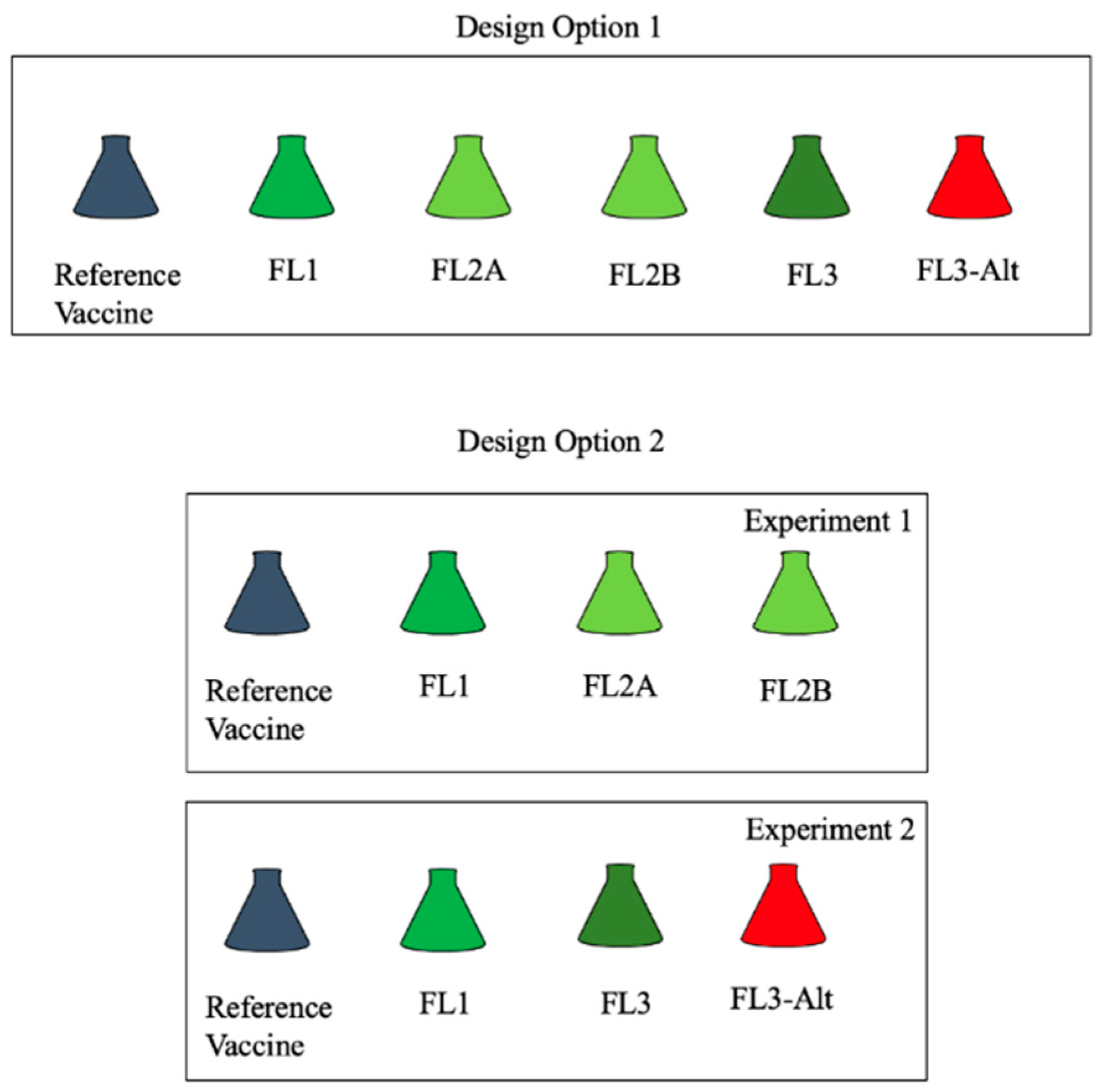
2.2. Project Design
2.3. Test and Reference Vaccines
2.4. Mice and Their Evaluation for Absence of Bordetella spp.
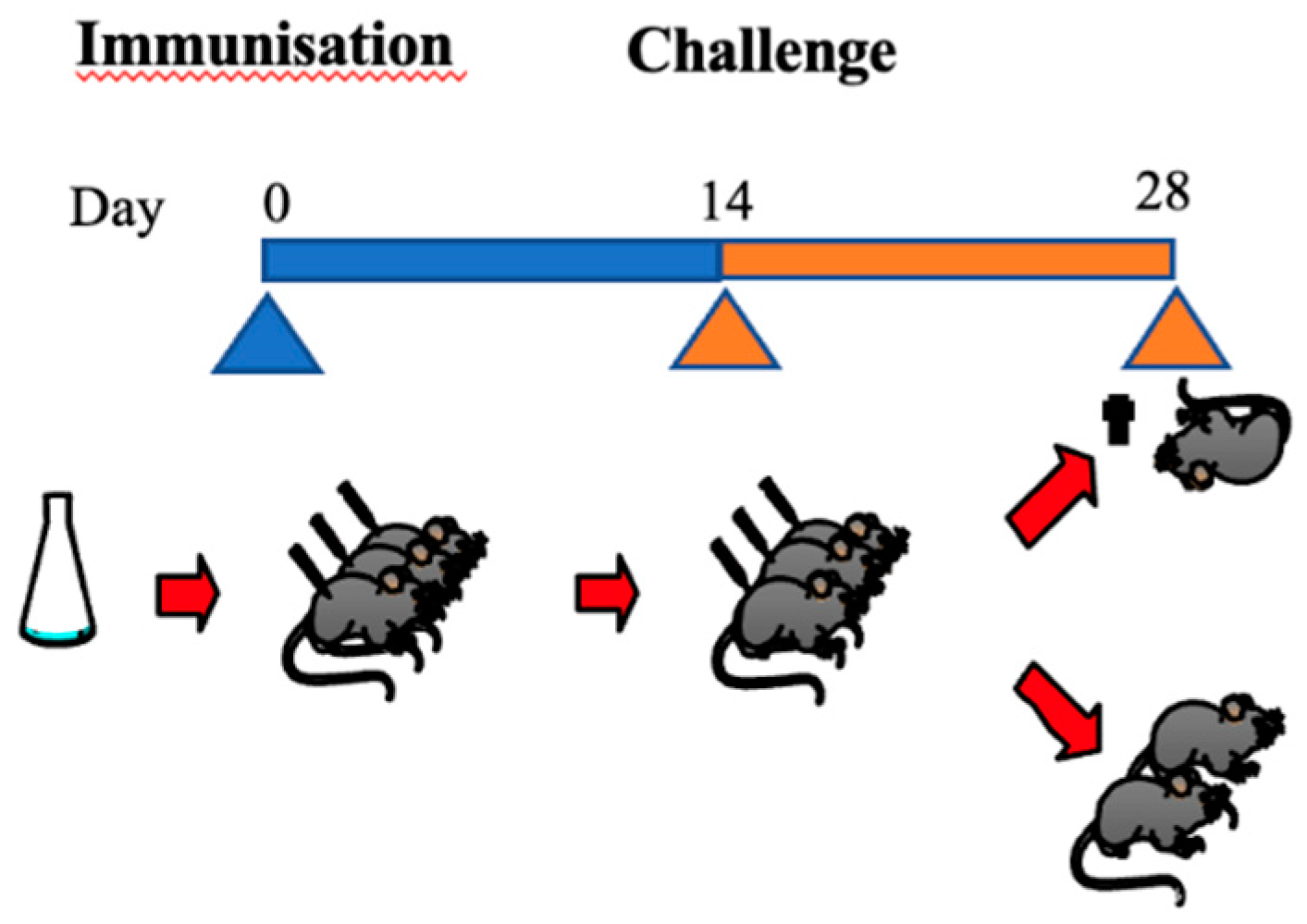
2.5. Intracerebral Mouse Protection Test (MPT)
2.6. Pertussis Serological Potency Test (PSPT)
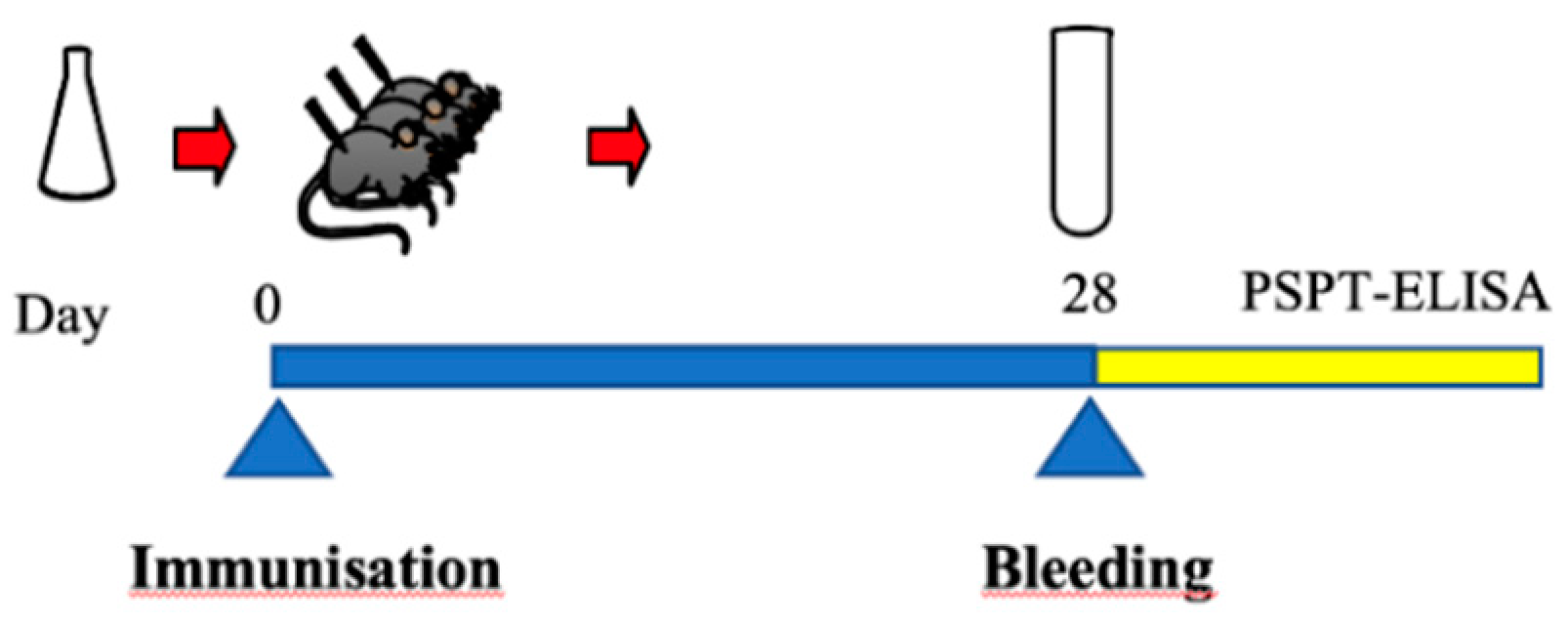
2.6.1. Immunization of Mice and Preparation of Serum Samples
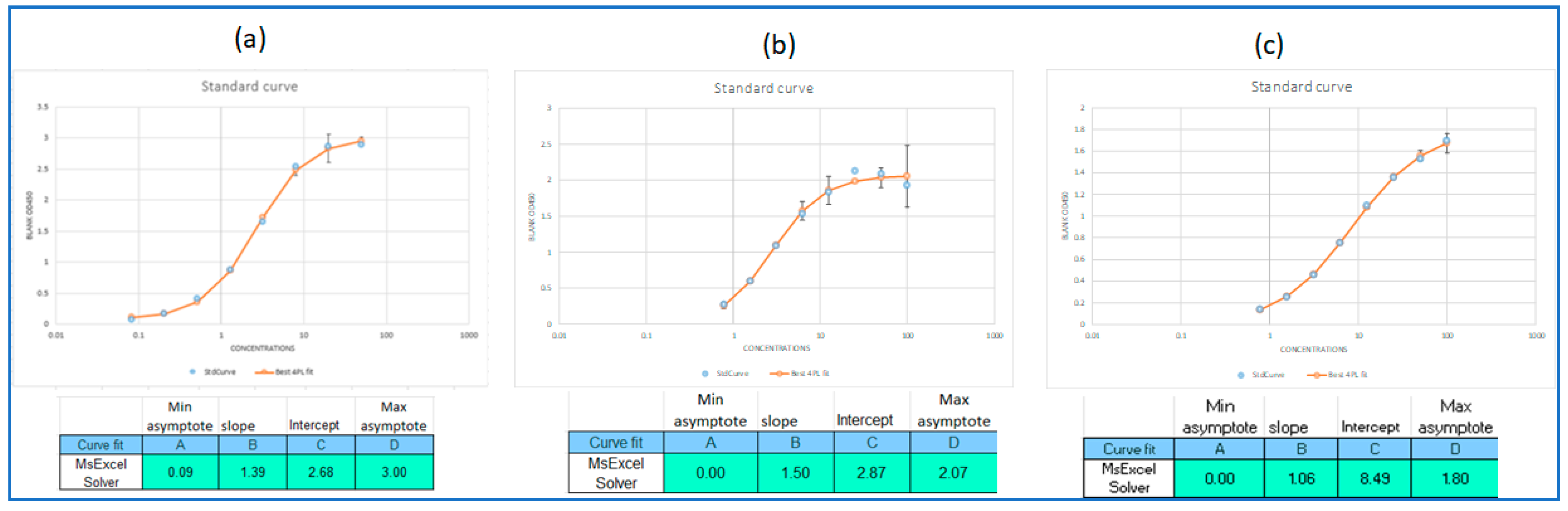
2.6.2. PSPT Whole-Cell ELISA (WCE)
2.7. WCE Coating Antigen
2.8. Preparation of Pertussis Positive and Negative Control Sera for the WCE
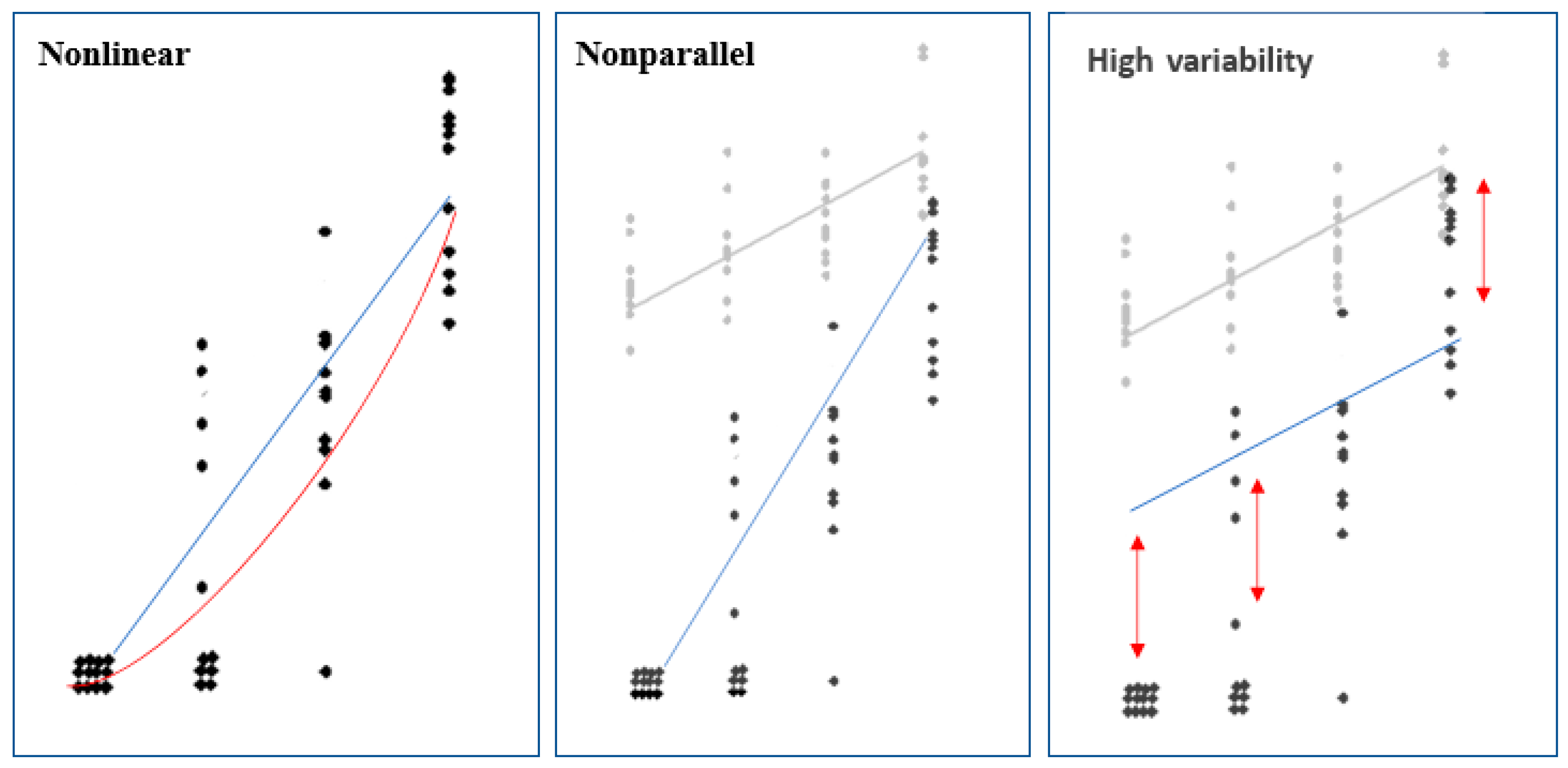
2.9. Statistical Methods
3. Results
3.1. Determination of wP Vaccine Induced IgG Responses by WCE
3.4. Determination of Relative Potencies of wP Vaccine Batches by PLA
3.5. PSPT Project Results
3.5.1. Assay and Batch Variabilities
3.5.2. Detection of the Altered Batches by MPT and PSPT
4. Discussion
5. Conclusions
Funding
Disclaimer
Declaration of interest
Acknowledgements
Appendix A
| Participant | Type of organization | Country | Names |
|---|---|---|---|
| Bharat Biotech | Manufacturer | India | Gopal Singh; Brunda Ganneru; Ganesh Dubey; Tarun Neha |
| Biological E | Manufacturer | India | Venugopal Bandameedi; Venkatarakesh Deevi |
| BB-NCIPD | Manufacturer | Bulgaria | Viktor Denev; Elena Nikolova; Valentina Borisova |
| Central Drugs Standard Control Organization. Central Drugs Laboratory, Kasauli |
National Control Laboratory | India | Sushil Sahu |
| Institute of Biological Products (IBP), Department of Medical Sciences | National Control Laboratory | Thailand | Apichai Supasansatorn; Wereyarmarst Jaroenkunathum |
| National Quality Control Laboratory of Drug and Food (NQCLDF) | National Control Laboratory | Indonesia | Anissa Wari Murti; Zulfa Noerhidayati; Ratih Pujilestari; Keti Yuliani |
| Panacea Biotec | Manufacturer | India | Rashi Saini; Yashpal Kaushik; Deepak Mahajan; Bonny Sharma; Maya Ramdas |
| PT Bio Farma (Persero) | Manufacturer | Indonesia | Dewi Dahlia Yuliarsih; Aini Qolbiyah Afgani; Chairunnisaa Jabal Rahmah |
| Sanofi Healthcare India Private Limited | Manufacturer | India | Surender Reddy Battula; Sunil Reddy Dadhireddy |
| Serum Institute of India | Manufacturer | India | Ghule Arvindkumar; Karegaonkar Ritesh; M. Anadkumar |
References
- Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. http://data.europa.eu/eli/dir/2010/63/oj.
- Good manufacturing practice | European Medicines Agency (europa.eu).
- Hendriksen, C.; Arciniega, J.L; Bruckner, L.; Chevalier, M.; Coppens, E.; Descamps, J.; Duchene, M.; Dusek, D.M.; Halder, M.; Kreeftenberg, H.; Maes, A.; Redhead, K.; Ravetkar, S.D., Spieser, J.M.; Swam, H. The consistency approach for the quality control of vaccines. Biologicals 2008, 36, 73-77.
- Dierick, J.F.; Halder, M.; Jungbäck, C.; Lorenz J.; Préaud, J.M.; Riou, P.; Tesolin, L.; Uhlrich, S.; van der Molle, W.; Vandeputte, J. The consistency approach for the substitution of in vivo testing for the quality control of established vaccines: practical considerations and progressive vision. Open Research Europe 2022, 2:116. [CrossRef]
- Kumar, S.; Singh, M.P.; Bharti V.K.; Pandey R.P. Quality control of vaccines- A journey from classical approach to 3Rs. Microbiol Curr. Res. 2018, 2, 45-71.
- Zhang, X.; Wu, X.; He, Q.; Wang, J.; Mao, Q.; Liang, Z.; Xu, M. Research progress on substitution of in vivo method(s) by in vitro method(s) for human vaccine potency assays. Expert Rev Vaccines 2023, 22(1), 270-277. [CrossRef]
- Home | Vac2Vac (europevaccine.wixsite.com).
- Assay of diphtheria vaccine, Monograph 20707. European Pharmacopoeia, 11 Edition, 2023, Council of Europe, Strasbourg, France.
- Assay of tetanus vaccine, Monograph 20708. European Pharmacopoeia, 11 Edition, 2023, Council of Europe, Strasbourg, France.
- Testing for diphtheria/tetanus vaccine. World Health Organization Manual for Quality Control of Diphtheria, Tetanus and Pertussis Vaccines, Chapter II and III. 2013. WHO/IVB/11.11. Manual for quality control of diphtheria, tetanus, pertussis and combined vaccines (who.int) .
- The Periscope Consortium. Periscope: road towards effective control of pertussis 2018. Lancet Infect. Dis. 2018. [CrossRef]
- Esposito, S.; Stefanelli, P.; Fry, NK; Fedele, G.; He, Q.; Paterson, P.;Tan, T.; Knuf, M.; Rodrigo, C.; Weil Olivier,C; Flanagan, KL.; Hung, I.; Lutsar, I.; Edwards, K.; O’Ryan, M.; Principi, N. Pertussis prevention: reasons for resurgence, and differences in the current acellular pertussis vaccines. Front Immunol 2019, 10:1344. [CrossRef]
- Zerbo, O.; Bartlett, J.; Goddard, K.; Fireman, B.; Lewis, E.; Klein, NP. Acellular pertussis vaccine effectiveness over time. Pediatrics 2019, 144 (1) e20183466. [CrossRef]
- Guiso, N.; Meade, B.D.; Wirsing von König, K.H.; Pertussis vaccines: The first hundred years. Vaccine 2020, 38, 1271-1276. [CrossRef]
- World Health Organization: Pertussis Vaccines: WHO position paper – August 2015Pertussis vaccines : WHO position paper — August 2015 = Note de synthèse : Position de l’OMS concernant les vaccins anticoquelucheux — août 2015 (accessed October 2023).
- Testing for whole cell pertussis vaccine. In Manual for Quality Control of Diphtheria, Tetanus and Pertussis Vaccines, Chapter IV. 2013. WHO/IVB/11.11. Manual for quality control of diphtheria, tetanus, pertussis and combined vaccines (who.int) .
- Canthaboo, C,; Xing, D.; Douglas, A.; Corbel, M. Investigation of an aerosol challenge model as alternative to the intracerebral mouse protection test for potency assay of whole cell pertussis vaccines. Biologicals 2000, 28, 241-6. [CrossRef] [PubMed]
- Canthaboo, C.; Xing, D.; Corbel, M. Development of a nitric oxide induction assay as a potential replacement for the intracerebral mouse protection test for potency assay of pertussis whole cell vaccines. Dev Biol Stand 1999, 101,95-103.
- van der Ark, A.; van Straaten-van de Kappelle, I.; Akkermans, A.; Hendriksen, C.; van de Donk, H. Development of Pertussis Serological Potency Test: serological assessment of response induced by whole cell vaccines as an alternative to mouse protection in an intracerebral challenge model. Biologicals 1994, 22, 233–242. [CrossRef]
- van der Ark, A.; Straaten-van de Kappelle, I.; Ölander, R.M.; Enssle, K.H.; Jadhav, S.; van de Donk, H.; Hendriksen, C. The Pertussis Serological Potency Test: Collaborative study to evaluate replacement of the mouse protection test. Biologicals 2000, 28,105-118. [CrossRef]
- von Hunolstein, C.; Gomez Miguel, M.J.; Pezzella, C.; Scopetti, F.; Behr-Gross, M.E.; Halder, M.; Hoffman, S.; Levels, L.; van der Gun, J.; Hendriksen, C. Evaluation of the serological methods for potency testing of whole cell pertussis vaccines. Pharmeuropa Bio 2008 (1),7-18.
- Hendriksen C, von Hunolstein C, van der Gun J, Gomez Miguel MJ, Daas A, Terao E. International collaborative study for the validation of an alternative serological assay for potency testing of whole cell pertussis vaccines – BSP104-Phase 1. PA/PH/BIO (13) 20, funded by EDQM, Biological Standardisation Programme (Unpublished work).
- Developing Countries Vaccine Manufacturers Network. List of Vaccines – DCVMN (accessed October 2023).
- NIIMBL, the National Institute for Innovation in Manufacturing Biopharmaceuticals NIIMBL Launches Global Health Fund (force.com) (accessed October 2023).
- Temperature sensitivity of vaccines. World Health Organization. 2006. WHO/IVB/06.10, https://apps.who.int/iris/handle/10665/69387.
- Heat stability of pertussis vaccine. Wkly Epidem rec 1985, 39,300 Expanded Programme on Immunization.
- Statistical analysis of results of biological assays and tests. European Pharmacopeia 11 edition, 2023, Council of Europe, Strasbourg, France.
- General Chapter 1033, Biological assay validation. United States Pharmacopeia 2020. [CrossRef]
- General Chapter 1032, Design and Development of Biological Assays. United States Pharmacopeia, 2022. [CrossRef]
- van der Biggelaar, R.G.A; Hoefnagel, M.H.N.; Vanderbriel, R.J; Sloot, A.;Hendriksen, C.F.M; van Eden, W.; Rutten, V.P.M.G.; Jansen, C.A. Overcoming scientific barriers in the transition from in vivo to non-animal batch testing of human and veterinary vaccines. Expert Review Vaccine, 2021, 20,1221-1223. [CrossRef]
- Recommendations to assure the quality, safety and efficacy of acellular pertussis vaccines. Annex 4 of WHO Technical Report Series, No. 979, 2013. Recommendations to assure the quality, safety and efficacy of acellular pertussis vaccines, Annex 4, TRS No 979 (who.int).
- National Control Laboratories National Control Laboratories | WHO - Prequalification of Medical Products (IVDs, Medicines, Vaccines and Immunization Devices, Vector Control) (accessed October 2023).
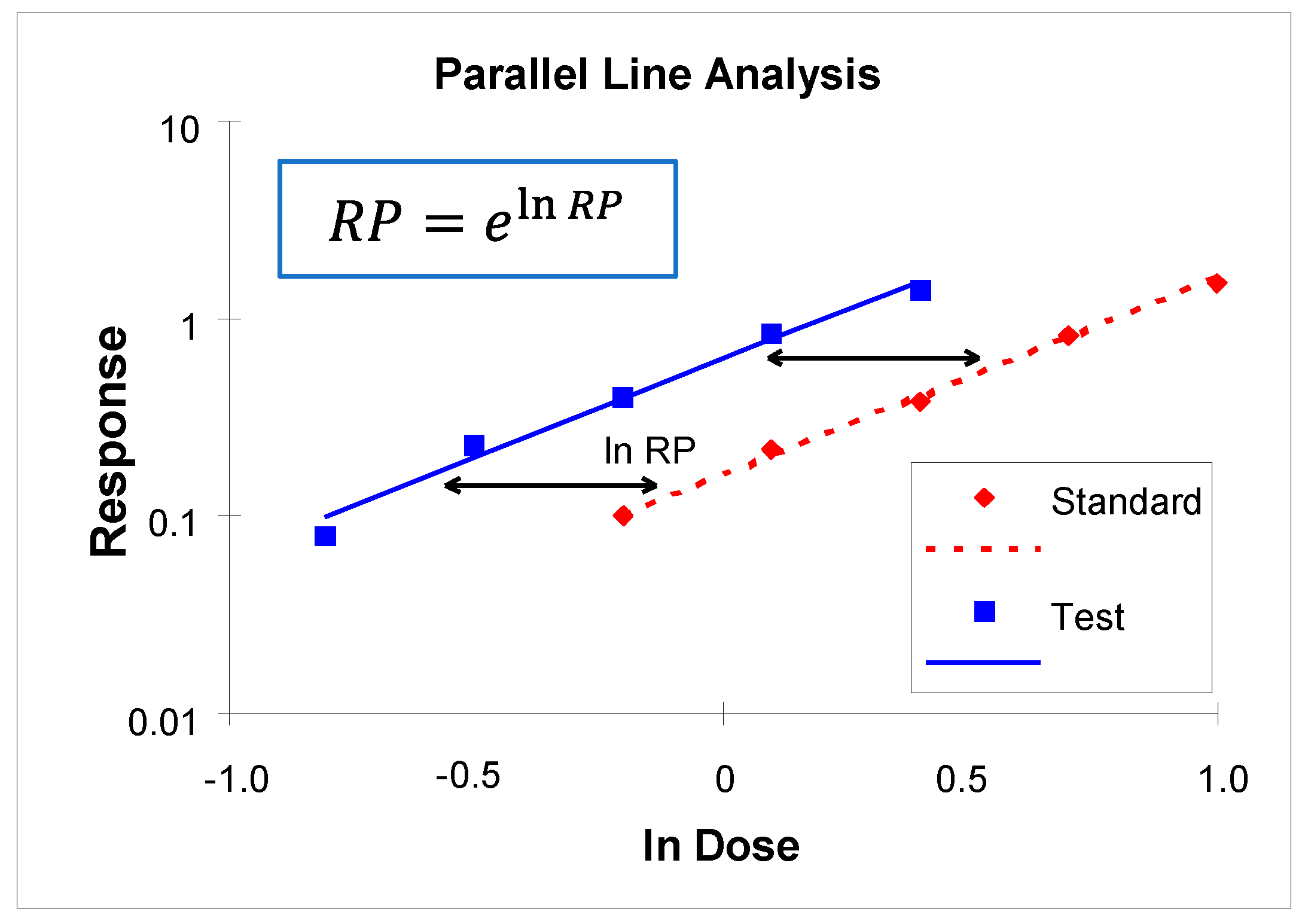
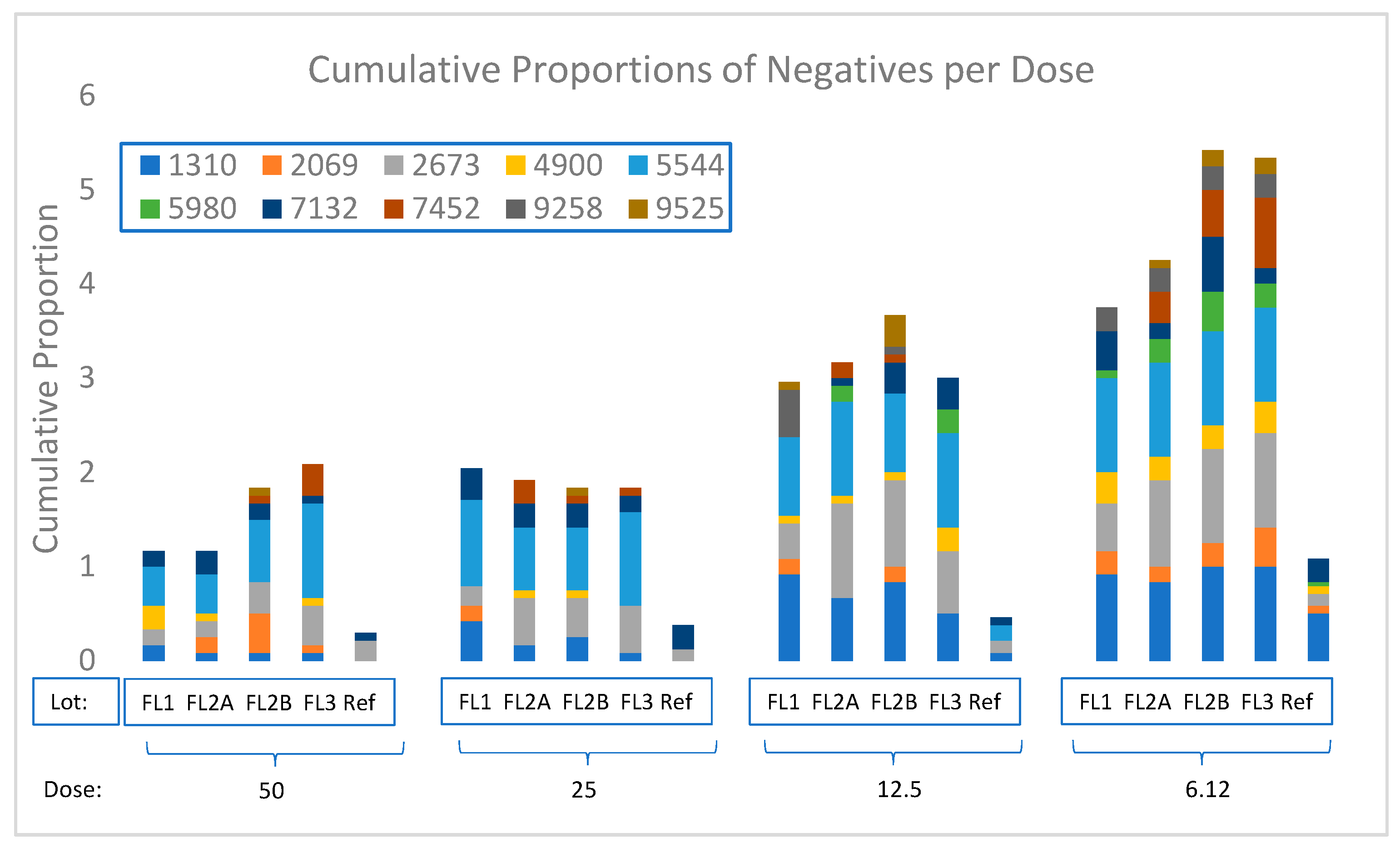
| Laboratory Code | Test Vaccine | ERS ** | ||||
|---|---|---|---|---|---|---|
| Components | OU*/mL | Origin | Batch n. /code | IU/vial § | ||
| 1310 | DTwP-HepB-Hib | 24 | National | RWRS 01/11 | 63 | |
| 2069 | DTwP-HepB-Hib | 24 | National | IPRS/20/PERT | 75 | |
| 2673 | DTwP-HepB-Hib | 24 | In-house | STBF 2015-1 | 62 | |
| 4990 | DTwP-HepB-Hib-IPV | 30 | National | IPRS/20/PERT | 75 | |
| 5544 | DTwP | 30 | WHO I.S. | NIBSC 94/532 | 40 | |
| 5980 | DTwP-HepB-Hib | 28 | National | IPRS/20/PERT | 75 | |
| 7132 | DTwP-HepB-Hib | 24,28,32 | National | IPRS/20/PERT | 75 | |
| 7452 | DTwP-HepB-Hib | 24,28,32 | National | RWRS 01/11 | 63 | |
| 9258 | DTwP-HepB-Hib | 32 | National | IPRS/20/PERT | 75 | |
| 9525 | DTwP-HepB-Hib | 16 | National | IPRS/20/PERT | 75 | |
| Laboratory Code | 1310 | 2069 | 2673 | 4900 | 5980 | 7132 | 7452 | 9258 | 5544 | 9525 |
|---|---|---|---|---|---|---|---|---|---|---|
| Strain of mouse, sex, and breeding | ddY (Male) | Swiss Albino (Male) | ddYbf (Male) | ICR (50/50) | NIH (Ola Hsd) (50/50) | Swiss Albino (Male) | ICR (Female) | Swiss Webster (50/50) | ICR (50/50) | Swiss Albino (Male) |
| Outbred | Outbred | Outbred | Outbred | Inbred | Outbred | Outbred | Outbred | Outbred | Outbred |
| Laboratory | Parallelism | Linearity | Confidence Interval of Relative Potency |
|---|---|---|---|
| 1310† | * | * | |
| 2069 | **** | ||
| 2673†† | ***** | ||
| 4900 | **** | ||
| 5544 | ** | **** | |
| 5980†† | * | **** | |
| 7132† | **** | ||
| 7452† | *** | ||
| 9258 | ** | ||
| 9525 | **** |
| Laboratory | Parallelism | Linearity | Confidence Interval of Relative Potency |
|---|---|---|---|
| 1310† | * | ||
| 2069 | ** | ||
| 2673†† | * | * | |
| 4900 | |||
| 5544 | ** | ||
| 5980†† | |||
| 7132† | * | ||
| 7452† | * | * | |
| 9258 | |||
| 9525 |
| Reference | Lab | GM† |
(Assay) |
(Total) |
|||
| External | 1310†† | 2.22 | 0.0025 | 0.0936 | 0.0962 | 5.2% | 36.4% |
| 2069 | 1.72 | 0.0005 | 0.0355 | 0.0360 | 2.3% | 20.9% | |
| 2673 | 0.06 | 1.3432 | 0.3595 | 1.7027 | 218.7% | 268.7% | |
| 4900 | 0.90 | 0.0000 | 0.0046 | 0.0046 | 0.3% | 7.0% | |
| 5544 | 0.14 | 1.5780 | 0.0000 | 1.5780 | 251.2% | 251.2% | |
| 5980 | 0.57 | 0.0834 | 0.0000 | 0.0834 | 33.5% | 33.5% | |
| 7132†† | 0.76 | 0.0013 | NA | NA | 3.7% | NA | |
| 7452†† | 1.12 | 0.0313 | NA | NA | 19.4% | NA | |
| 9258 | 1.38 | 0.0025 | 0.0129 | 0.0154 | 5.1% | 13.2% | |
| 9525 | 2.17 | 0.0245 | 0.0177 | 0.0422 | 16.9% | 22.8% | |
| Internal | 1310†† | 1.48 | 0.0600 | 0.0381 | 0.0981 | 27.8% | 36.8% |
| 2069 | 0.69 | 0.0007 | 0.0037 | 0.0044 | 2.7% | 6.9% | |
| 2673 | 0.62 | 0.0385 | 0.0598 | 0.0983 | 21.7% | 36.8% | |
| 4900 | 1.06 | 0.0001 | 0.0164 | 0.0165 | 0.8% | 13.7% | |
| 5544 | 1.11 | 0.2184 | 0.0000 | 0.2184 | 59.6% | 59.6% | |
| 5980 | 0.86 | 0.0004 | 0.0227 | 0.0231 | 2.1% | 16.4% | |
| 7132†† | 1.12 | 0.0127 | NA | NA | 11.9% | NA | |
| 7452†† | NA | NA | NA | NA | NA | NA | |
| 9258 | 1.37 | 0.0041 | 0.1080 | 0.1121 | 6.6% | 39.8% | |
| 9525 | 1.22 | 0.0057 | 0.0364 | 0.0421 | 7.9% | 22.8% |
| MPT Potency |
PSPT potency vs ERS |
PSPT Potency vs IRS –FL1 |
|||||||
| Lab | FL3 | FL3-Alt | % Red. | FL3 | FL3-Alt | % Red. | FL3 | FL3-Alt | % Red. |
| 1310 | 6.29 (2.12 - 19.75) |
2.93 (1.03 - 8.31) |
53% | 3.0802 (1.804-4.589) |
1.8998 (1.1556-2.7563) |
38% | 1.9380 (1.1932-3.6533) |
0.9312 (0.6408-1.3150) |
52% |
| 2069 | 5.03 (2.34 - 10.96) |
4.06 (1.78 - 9.32) |
19% | 1.6492 (0.4429-3.3560) |
0.7195 (0.0502-2.0298) |
56% | 0.6456 (0.3918-0.9827) |
0.3738 (0.1540-0.6675) |
42% |
| 2673† | 7.07 (2.80 - 9.22) |
4.88 (2.20 –10.80) |
31% | 1.1070 (0.7561-1.6469) |
0.7897 (0.5541-1.1035) |
29% | 1.1703 (0.7384-1.8917) |
0.7890 (0.5071-1.1724) |
33% |
| 4900 | 7.60 (4.40 -13.6) |
5.18 (2.10-12.5) |
32% | 0.9246 (0.2047-1.944) |
0.6916 (0.0452-1.8851) |
25% | 0.9410 (0.6104-1.426) |
0.4774 (0.1395-1.1969) |
49% |
| 5544 | 7.41 (2.71-3.38) |
2.01 (0.46 - 6.72) |
73% | 0.0301 (0.000-0.3280) |
0.0196 (0.000-0.2507) |
35% | 0.8790 (0.1123-2.3366) |
0.4023 (0.0569-1.0258) |
54% |
| 5980 | 5.18 (2.94 -9.16) |
3.12 (1.71-5.65) |
40% | 0.5424 (0.1662-1.113) |
0.4938 (0.1503-1.0230) |
9% | 0.9900 (0.7308-1.3333) |
0.7535 (0.5476-1.0150) |
24% |
| 7132 | 8.10 (4.56-13.94) |
0.69 (0.18 - 1.71) |
91% | 0.9294 (0.1007-2.4381) |
0.5131 (0.0633-1.3739) |
45% | 1.4376 (0.7584-3.0630) |
0.6549 (0.3428-1.1190) |
54% |
|
7452 |
5.95 (2.59-14.12) |
2.04 (0.87 - 4.59) |
66% |
0.0533 (0.0025-0.2206) |
0.0053 (0.0000-0.0367) |
90% |
0.22 (0.1069-0.3585) |
0.071 (0.0239-0.1353) |
68% |
| 9258 | 7.41 (3.17 -16.15) |
3.13 (1.12 - 6.87) |
58% | 1.4932 (0.6621-2.5552) |
1.3631 (0.6518-2.2591) |
9% | 1.8775 (1.3169-2.8592) |
1.2291 (0.8874-1.7322) |
35% |
| 9525 | 5.90 (3.40 - 10.6) |
1.85 (0.80 - 3.70) |
69% | 1.8450 (0.0094-5.834) |
0.3086 (0.0000-2.1434) |
83% | 1.4753 (0.8719-2.6497) |
0.7025 (0.3099-1.2679) |
52% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




