Submitted:
31 October 2023
Posted:
31 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
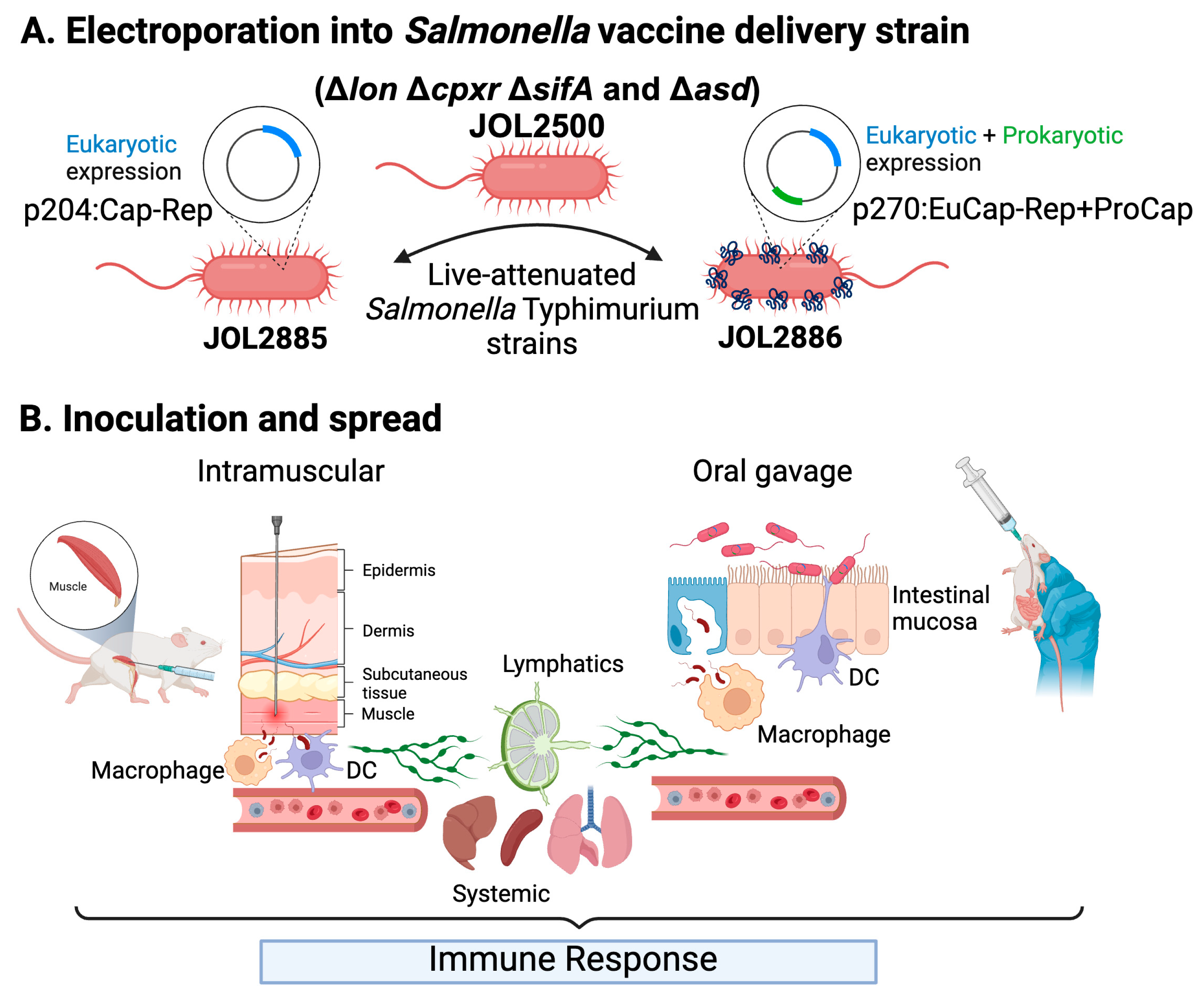
2.1. Bacterial strains, plasmids, viruses and cell lines
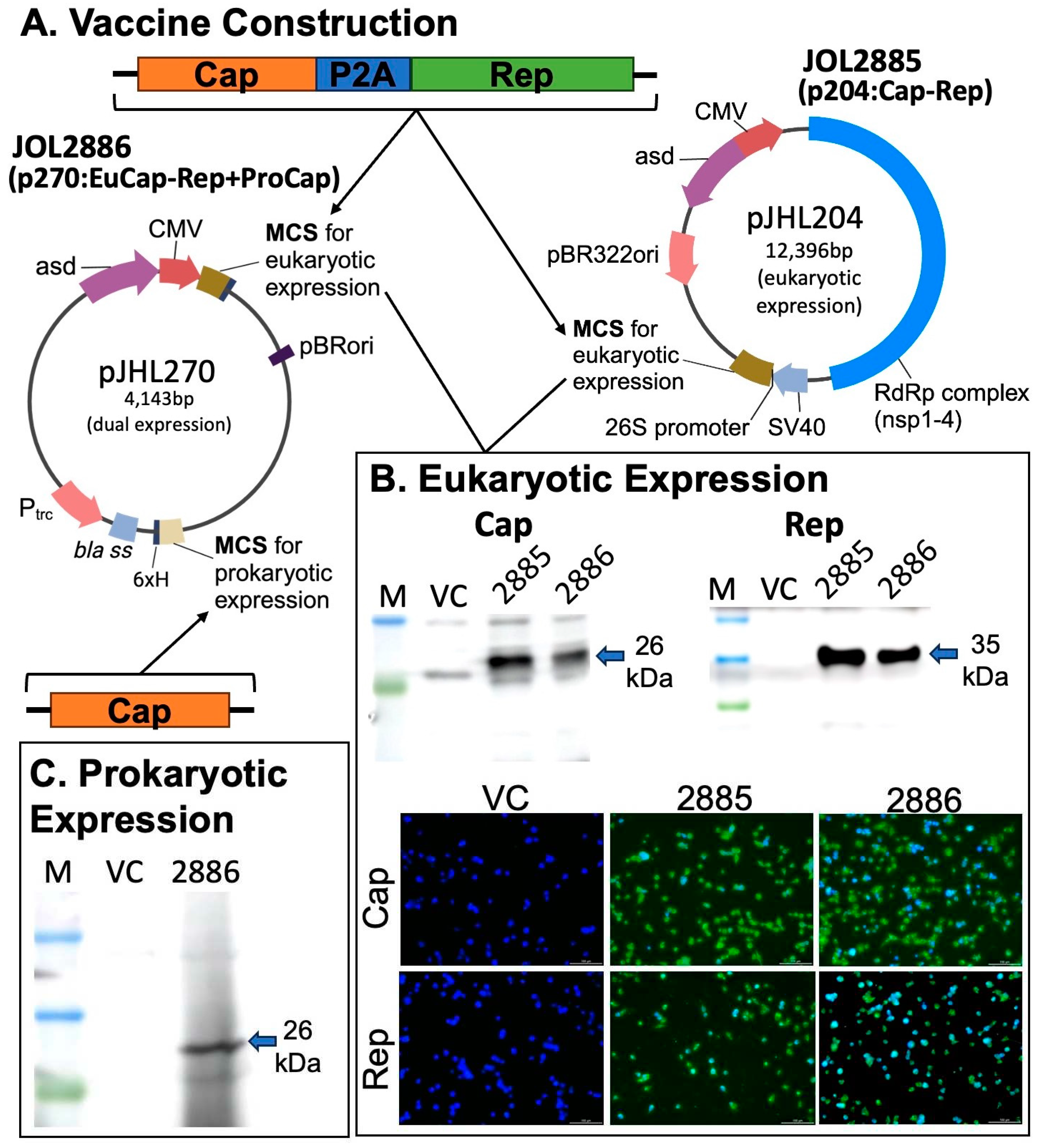
2.2. Design and development of the vaccine candidates
2.3. Confirming the expression by RT-PCR, immunofluorescence and western blot
2.4. Mice immunization, PCV2d virus challenge and sampling
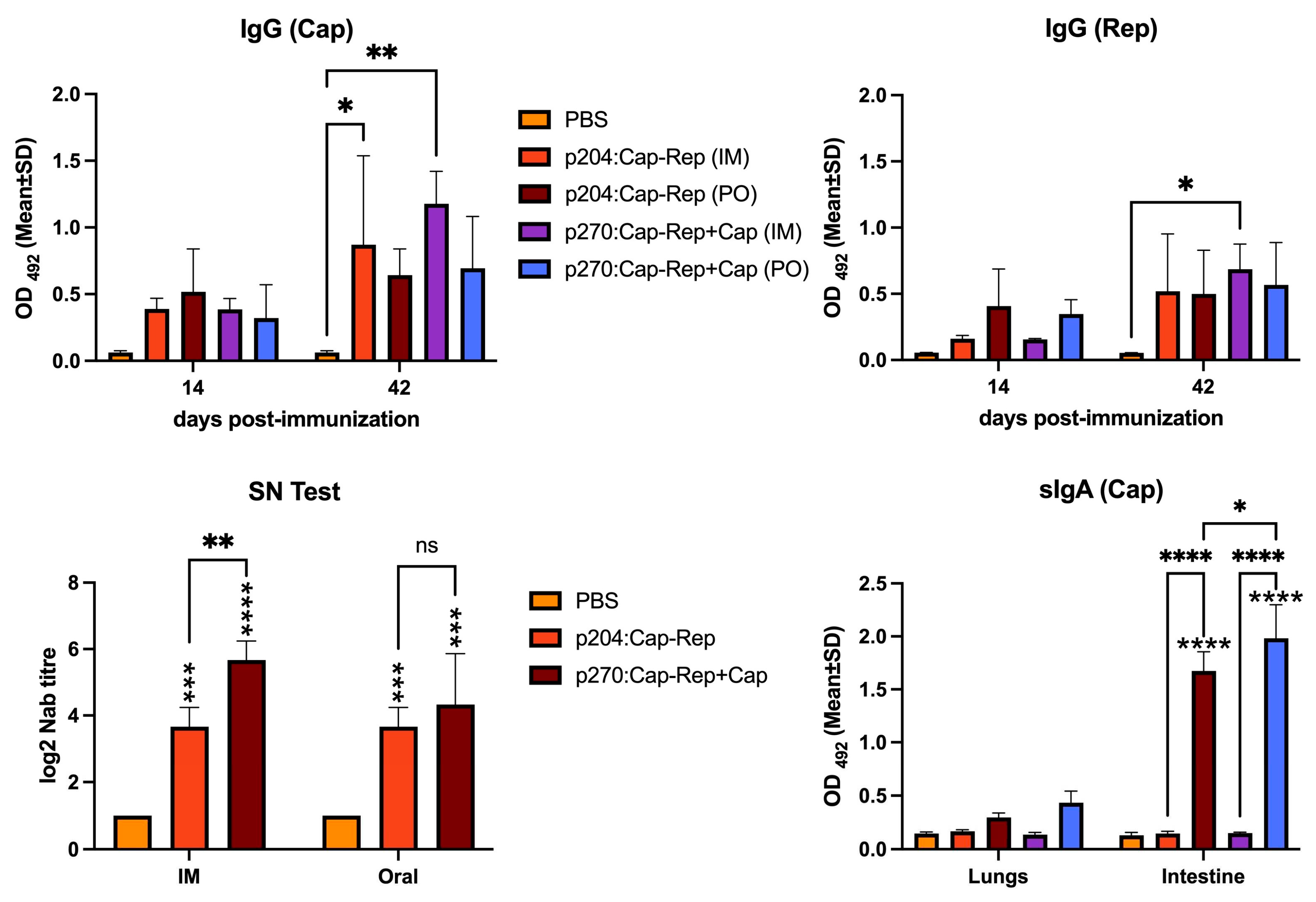
2.5. Enzyme-linked immunosorbent assay (ELISA)
2.6. Virus neutralization assay
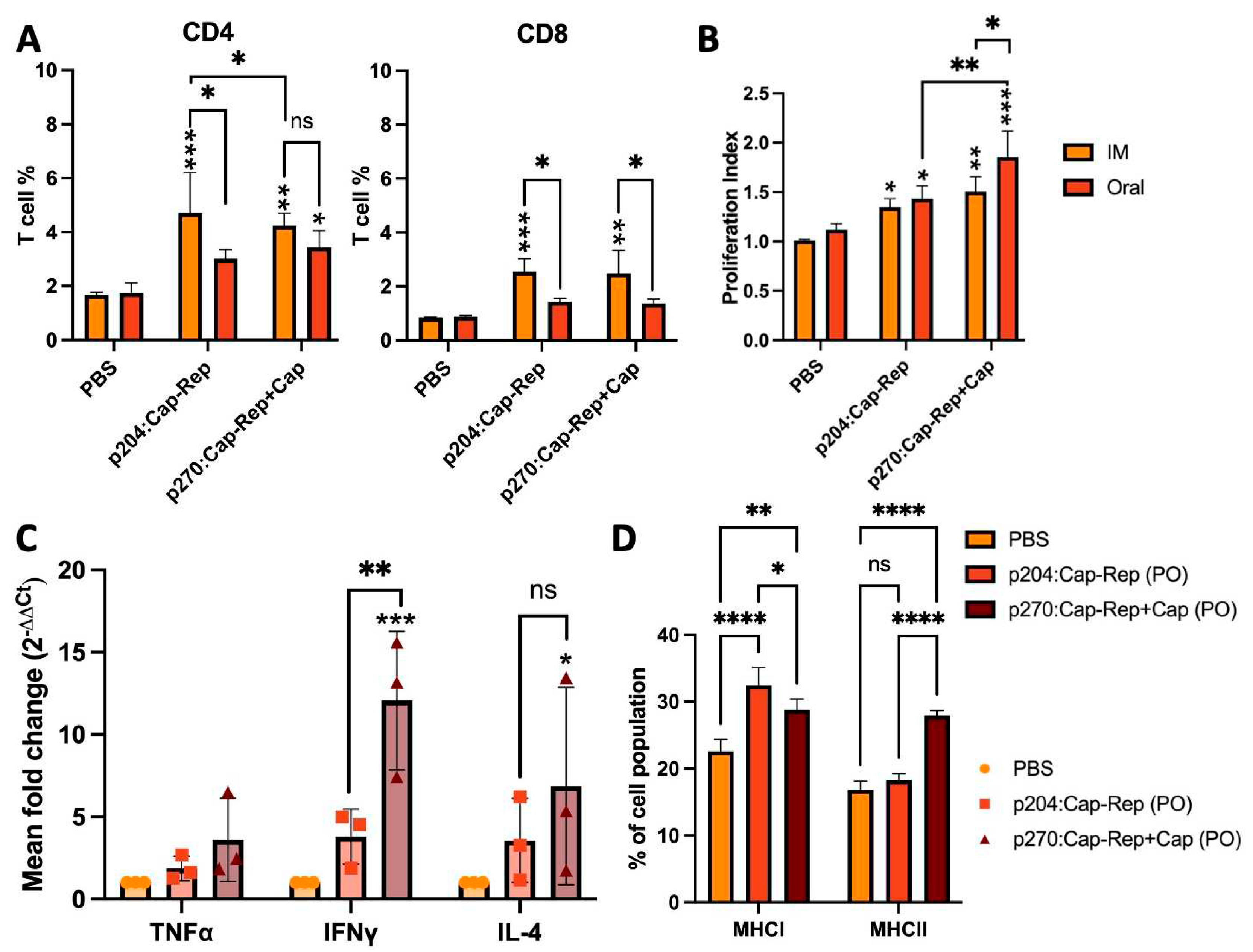
2.7. Lymphocyte proliferation assay
2.8. Flow cytometric analysis and cytokine expression
2.9. Detection of PCV2 in mice tissues by qPCR and immunohistochemistry
2.10. Statistical analysis
3. Results
3.1. Vaccine construction and confirmation of eukaryotic and prokaryotic expression of target antigens
3.2. Humoral and mucosal immune responses induced by the Salmonella-delivered vaccines
3.3. CD4+ and CD8+ T cell differentiation and lymphocyte proliferative response
3.4. Cytokine expression profile and MHC class I and MHC class II molecules stimulation
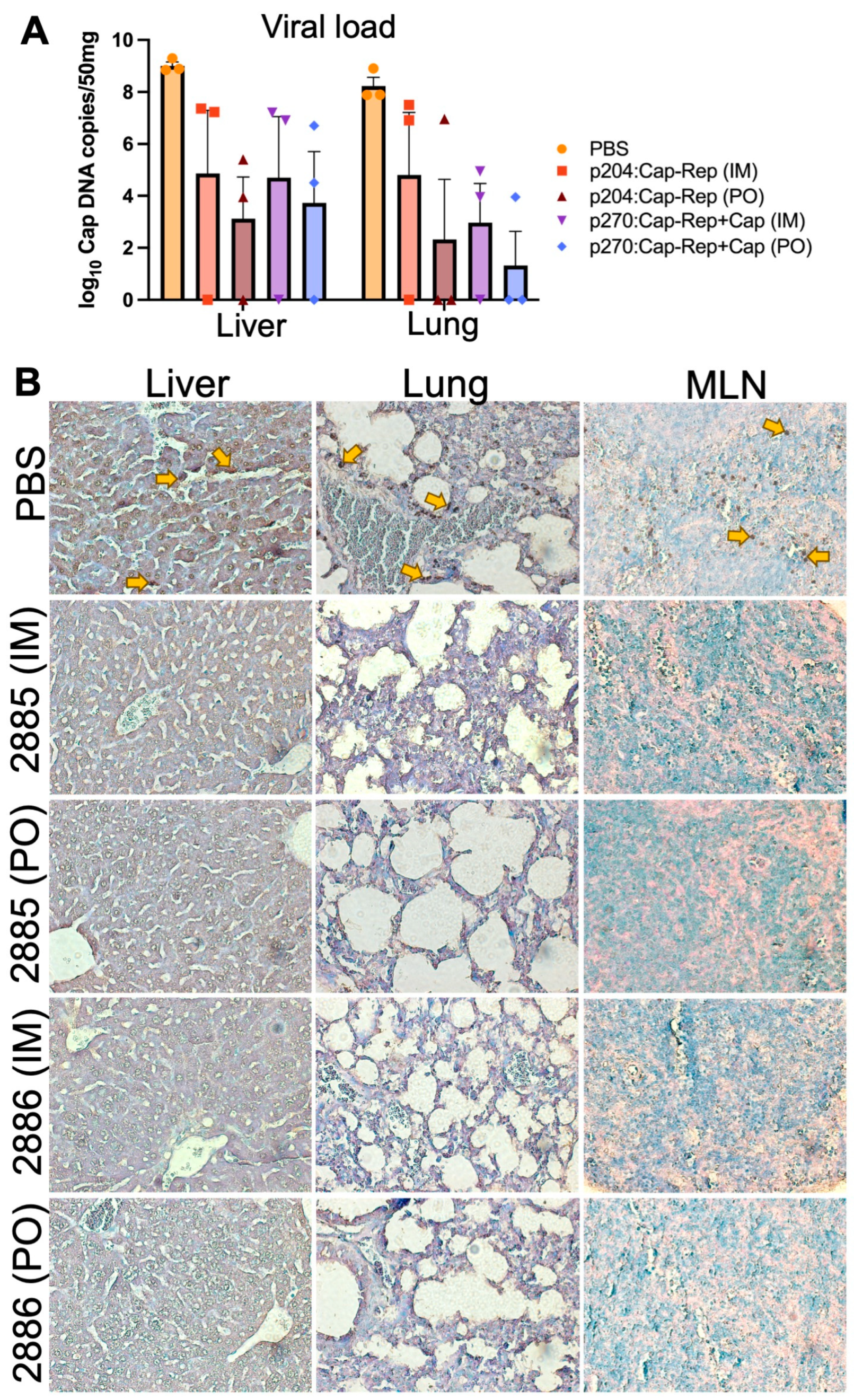
3.5. Detection of PCV2 in virus-challenged mice
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gillespie, J.; Opriessnig, T.; Meng, X.J.; Pelzer, K.; Buechner-Maxwell, V. Porcine Circovirus Type 2 and Porcine Circovirus-Associated Disease. J Vet Intern Med 2009, 23. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, A.; Miłek, D.; Bąska, P.; Stadejek, T. Does Porcine Circovirus Type 3 (PCV3) Interfere with Porcine Circovirus Type 2 (PCV2) Vaccine Efficacy? Transbound Emerg Dis 2019, 66. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.T.; Halbur, P.G.; Opriessnig, T. Global Molecular Genetic Analysis of Porcine Circovirus Type 2 (PCV2) Sequences Confirms the Presence of Four Main PCV2 Genotypes and Reveals a Rapid Increase of PCV2d. Journal of General Virology 2015, 96. [Google Scholar] [CrossRef] [PubMed]
- Karuppannan, A.K.; Opriessnig, T. Porcine Circovirus Type 2 (PCV2) Vaccines in the Context of Current Molecular Epidemiology. Viruses 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Xiao, S.; Tong, T.; Wang, S.; Xie, L.; Jiang, Y.; Chen, H.; Fang, L. Immunogenicity of Porcine Circovirus Type 2 Capsid Protein Targeting to Different Subcellular Compartments. Mol Immunol 2008, 45. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, P.; Mahé, D.; Cariolet, R.; Keranflec’h, A.; Baudouard, M.A.; Cordioli, P.; Albina, E.; Jestin, A. Protection of Swine against Post-Weaning Multisystemic Wasting Syndrome (PMWS) by Porcine Circovirus Type 2 (PCV2) Proteins. Vaccine 2003, 21. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Yu, W.; Cai, X.; Lei, X.; Lei, H.; Wang, A.; Sun, Y.; Wang, N.; Deng, Z.; Yang, Y. Correction for Zhan et al., “The Carboxyl Terminus of the Porcine Circovirus Type 2 Capsid Protein Is Critical to Virus-Like Particle Assembly, Cell Entry, and Propagation.”. J Virol 2021, 95. [Google Scholar] [CrossRef] [PubMed]
- Rose, N.; Opriessnig, T.; Grasland, B.; Jestin, A. Epidemiology and Transmission of Porcine Circovirus Type 2 (PCV2). Virus Res 2012, 164. [Google Scholar] [CrossRef] [PubMed]
- Patterson, A.R.; Opriessnig, T. Epidemiology and Horizontal Transmission of Porcine Circovirus Type 2 (PCV2). Animal health research reviews / Conference of Research Workers in Animal Diseases 2010, 11. [Google Scholar] [CrossRef]
- Alarcon, P.; Rushton, J.; Nathues, H.; Wieland, B. Economic Efficiency Analysis of Different Strategies to Control Post-Weaning Multi-Systemic Wasting Syndrome and Porcine Circovirus Type 2 Subclinical Infection in 3-Weekly Batch System Farms. Prev Vet Med 2013, 110. [Google Scholar] [CrossRef]
- Chae, C. Commercial Porcine Circovirus Type 2 Vaccines: Efficacy and Clinical Application. Veterinary Journal 2012, 194. [Google Scholar] [CrossRef] [PubMed]
- Rose, N.; Andraud, M.; Bigault, L.; Jestin, A.; Grasland, B. A Commercial PCV2a-Based Vaccine Significantly Reduces PCV2b Transmission in Experimental Conditions. Vaccine 2016, 34. [Google Scholar] [CrossRef] [PubMed]
- Lycke, N. Recent Progress in Mucosal Vaccine Development: Potential and Limitations. Nat Rev Immunol 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, D.; Sun, B.; Yu, M.; Wang, Y.; Ru, Y.; Jiang, Y.; Qiao, X.; Cui, W.; Zhou, H.; et al. Oral Vaccination with the Porcine Circovirus Type 2 (PCV-2) Capsid Protein Expressed by Lactococcus Lactis Induces a Specific Immune Response against PCV-2 in Mice. J Appl Microbiol 2020, 128. [Google Scholar] [CrossRef]
- Li, W.; Li, J.; Dai, X.; Liu, M.; Khalique, A.; Wang, Z.; Zeng, Y.; Zhang, D.; Ni, X.; Zeng, D.; et al. Surface Display of Porcine Circovirus Type 2 Antigen Protein Cap on the Spores of Bacillus Subtilis 168: An Effective Mucosal Vaccine Candidate. Front Immunol 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Becker, P.D.; Noerder, M.; Guzmán, C.A. Genetic Immunization: Bacteria as DNA Vaccine Delivery Vehicles. Hum Vaccin 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- Dieye, Y. Bacterial Delivery Vehicles for Mucosal Vaccines: A Promising Tool for Mass Vaccination. Am J Biomed Sci Res 2022, 17. [Google Scholar] [CrossRef]
- Cazorla, S.I.; Becker, P.D.; Frank, F.M.; Ebensen, T.; Sartori, M.J.; Corral, R.S.; Malchiodi, E.L.; Guzmán, C.A. Oral Vaccination with Salmonella Enterica as a Cruzipain-DNA Delivery System Confers Protective Immunity against Trypanosoma Cruzi. Infect Immun 2008, 76. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Mengyue, M.; Qimuge, A.; Bilige, B.; Baiyin, T.; Temuqile, T.; Chen, S.; Borjigen, S.; Baigude, H.; Yang, D. Oral Delivery of SARS-CoV-2 DNA Vaccines Using Attenuated Salmonella Typhimurium as a Carrier in Rat. Molecular Genetics, Microbiology and Virology 2022, 37. [Google Scholar] [CrossRef]
- Weiss, S. Transfer of Eukaryotic Expression Plasmids to Mammalian Hosts by Attenuated Salmonella Spp. International Journal of Medical Microbiology 2003, 293. [Google Scholar] [CrossRef]
- Lee, J.S.; Shin, K.S.; Pan, J.G.; Kim, C.J. Surface-Displayed Viral Antigens on Salmonella Carrier Vaccine. Nat Biotechnol 2000, 18. [Google Scholar] [CrossRef] [PubMed]
- Yoon, W.; Park, Y.; Kim, S.; Bang, I.S. Development of an Oral Salmonella-Based Vaccine Platform against SARS-CoV-2. Vaccines (Basel) 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Sivasankar, C.; Hewawaduge, C.; Lee, J.H. Novel Pro-and Eukaryotic Expression Plasmid Expressing Omicron Antigens Delivered via Salmonella Elicited MHC Class I and II Based Protective Immunity. Journal of Controlled Release 2023, 357. [Google Scholar] [CrossRef] [PubMed]
- Miquel-Clopés, A.; Bentley, E.G.; Stewart, J.P.; Carding, S.R. Mucosal Vaccines and Technology. Clin Exp Immunol 2019, 196. [Google Scholar] [CrossRef] [PubMed]
- Senevirathne, A.; Park, J.Y.; Hewawaduge, C.; Perumalraja, K.; Lee, J.H. Eukaryotic Expression System Complemented with Expressivity of Semliki Forest Virus’s RdRp and Invasiveness of Engineered Salmonella Demonstrate Promising Potential for Bacteria Mediated Gene Therapy. Biomaterials 2021, 279. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Zhang, H.; Sun, P.; Khan, A.; Guo, J.; Zheng, X.; Sun, Y.; Fan, K.; Yin, W.; Li, H. Matrine Exhibits Antiviral Activity in a PRRSV/PCV2 Co-Infected Mouse Model. Phytomedicine 2020, 77. [Google Scholar] [CrossRef]
- Giulietti, A.; Overbergh, L.; Valckx, D.; Decallonne, B.; Bouillon, R.; Mathieu, C. An Overview of Real-Time Quantitative PCR: Applications to Quantify Cytokine Gene Expression. Methods 2001, 25. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, S.R.; Li, L.H.; Park, H.J.; Park, J.H.; Lee, K.Y.; Kim, M.K.; Shin, B.A.; Choi, S.Y. High Cleavage Efficiency of a 2A Peptide Derived from Porcine Teschovirus-1 in Human Cell Lines, Zebrafish and Mice. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Jawalagatti, V.; Kirthika, P.; Hewawaduge, C.; Yang, M. sik; Park, J.Y.; Oh, B.; Lee, J.H. Bacteria-Enabled Oral Delivery of a Replicon-Based MRNA Vaccine Candidate Protects against Ancestral and Delta Variant SARS-CoV-2. Molecular Therapy 2022, 30. [Google Scholar] [CrossRef]
- Cságola, A.; Cadar, D.; Tuboly, T. Replication and Transmission of Porcine Circovirus Type 2 in Mice. Acta Vet Hung 2008, 56. [Google Scholar] [CrossRef]
- Zhang, C.; Zhu, S.; Wei, L.; Yan, X.; Wang, J.; Quan, R.; She, R.; Hu, F.; Liu, J. Recombinant Flagellin-Porcine Circovirus Type 2 Cap Fusion Protein Promotes Protective Immune Responses in Mice. PLoS One 2015, 10. [Google Scholar] [CrossRef]
- Aravindaram, K.; Kuo, T.Y.; Lan, C.W.; Yu, H.H.; Wang, P.H.; Chen, Y.S.; Chen, G.H.C.; Yang, N.S. Protective Immunity against Porcine Circovirus 2 in Mice Induced by a Gene-Based Combination Vaccination. Journal of Gene Medicine 2009, 11. [Google Scholar] [CrossRef]
- Li, S.; Wang, B.; Jiang, S.; Lan, X.; Qiao, Y.; Nie, J.; Yin, Y.; Shi, Y.; Kong, W.; Shan, Y. Expression and Evaluation of Porcine Circovirus Type 2 Capsid Protein Mediated by Recombinant Adeno-Associated Virus 8. J Vet Sci 2021, 22. [Google Scholar] [CrossRef]
- Sylla, S.; Cong, Y.L.; Sun, Y.X.; Yang, G.L.; Ding, X.M.; Yang, Z.Q.; Zhou, Y.L.; Yang, M.; Wang, C.F.; Ding, Z. Protective Immunity Conferred by Porcine Circovirus 2 ORF2-Based DNA Vaccine in Mice. Microbiol Immunol 2014, 58. [Google Scholar] [CrossRef]
- Hegazy, W.A.H.; Hensel, M. Salmonella Enterica as a Vaccine Carrier. Future Microbiol 2012, 7. [Google Scholar] [CrossRef]
- Galen, J.E.; Wahid, R.; Buskirk, A.D. Strategies for Enhancement of Live-Attenuated Salmonella-Based Carrier Vaccine Immunogenicity. Vaccines (Basel) 2021, 9. [Google Scholar] [CrossRef]
- Bai, Y.; Gong, H.; Li, H.; Vu, G.P.; Lu, S.; Liu, F. Oral Delivery of RNase P Ribozymes by Salmonella Inhibits Viral Infection in Mice. Proc Natl Acad Sci U S A 2011, 108. [Google Scholar] [CrossRef]
- Jawalagatti, V.; Kirthika, P.; Hewawaduge, C.; Park, J.Y.; Yang, M.S.; Oh, B.; So, M.Y.; Kim, B.; Lee, J.H. A Simplified SARS-CoV-2 Mouse Model Demonstrates Protection by an Oral Replicon-Based MRNA Vaccine. Front Immunol 2022, 13. [Google Scholar] [CrossRef]
- Park, J.Y.; Hewawaduge, C.; Sivasankar, C.; Lloren, K.K.S.; Oh, B.; So, M.Y.; Lee, J.H. An MRNA-Based Multiple Antigenic Gene Expression System Delivered by Engineered Salmonella for Severe Fever with Thrombocytopenia Syndrome and Assessment of Its Immunogenicity and Protection Using a Human DC-SIGN-Transduced Mouse Model. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Plotkin, S.; Robinson, J.M.; Cunningham, G.; Iqbal, R.; Larsen, S. The Complexity and Cost of Vaccine Manufacturing – An Overview. Vaccine 2017, 35. [Google Scholar]
- Kang, S.H.; Hong, S.J.; Lee, Y.K.; Cho, S. Oral Vaccine Delivery for Intestinal Immunity-Biological Basis, Barriers, Delivery System, and M Cell Targeting. Polymers (Basel) 2018, 10. [Google Scholar] [CrossRef]
- Baker, P.J. Advantages of an Oral Vaccine to Control the COVID-19 Pandemic. American Journal of Medicine 2022, 135. [Google Scholar] [CrossRef]
- Vela Ramirez, J.E.; Sharpe, L.A.; Peppas, N.A. Current State and Challenges in Developing Oral Vaccines. Adv Drug Deliv Rev 2017, 114. [Google Scholar] [CrossRef]
- Wang, S.; Liu, H.; Zhang, X.; Qian, F. Intranasal and Oral Vaccination with Protein-Based Antigens: Advantages, Challenges and Formulation Strategies. Protein Cell 2015, 6. [Google Scholar] [CrossRef]
- Cárdenas, L.; Clements, J.D. Oral Immunization Using Live Attenuated Salmonella Spp. as Carriers of Foreign Antigens. Clin Microbiol Rev 1992, 5. [Google Scholar] [CrossRef]
- Kiupel, M.; Stevenson, G.W.; Choi, J.; Latimer, K.S.; Kanitz, C.L.; Mittal, S.K. Viral Replication and Lesions in BALB/c Mice Experimentally Inoculated with Porcine Circovirus Isolated from a Pig with Postweaning Multisystemic Wasting Disease. Vet Pathol 2001, 38. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.; Feng, H.; Zhao, B.; Chen, Y.; Huang, H.; Wang, P.; Deng, R.; Zhang, G. PCV Cap Proteins Fused with Calreticulin Expressed into Polymers in Escherichia Coli with High Immunogenicity in Mice. BMC Vet Res 2020, 16. [Google Scholar] [CrossRef]
- Ouyang, T.; Liu, X. hui; Ouyang, H. sheng; Ren, L. zhu Mouse Models of Porcine Circovirus 2 Infection. Animal Model Exp Med 2018, 1. [Google Scholar]
- Mantis, N.J.; Rol, N.; Corthésy, B. Secretory IgA’s Complex Roles in Immunity and Mucosal Homeostasis in the Gut. Mucosal Immunol 2011, 4. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Function of Mucosa-Associated Lymphoid Tissue in Antibody Formation. Immunol Invest 2010, 39. [Google Scholar] [CrossRef]
- Meerts, P.; Misinzo, G.; Lefebvre, D.; Nielsen, J.; Bøtner, A.; Kristensen, C.S.; Nauwynck, H.J. Correlation between the Presence of Neutralizing Antibodies against Porcine Circovirus 2 (PCV2) and Protection against Replication of the Virus and Development of PCV2-Associated Disease. BMC Vet Res 2006, 2. [Google Scholar] [CrossRef]
- Germain, R.N. T-Cell Development and the CD4-CD8 Lineage Decision. Nat Rev Immunol 2002, 2. [Google Scholar] [CrossRef] [PubMed]
- Dave, V. Control of CD4 Helper and CD8 Cytotoxic T Cell Differentiation. J Cell Immunol 2020, 2. [Google Scholar] [CrossRef]
- Nawagitgul, P.; Morozov, I.; Bolin, S.R.; Harms, P.A.; Sorden, S.D.; Paul, P.S. Open Reading Frame 2 of Porcine Circovirus Type 2 Encodes a Major Capsid Protein. Journal of General Virology 2000, 81. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.K. Transcriptional Analysis of Porcine Circovirus Type 2. Virology 2003, 305. [Google Scholar] [CrossRef] [PubMed]
| Plasmid/Bacteria/Primer | Description | Reference |
|---|---|---|
| Plasmids | ||
| pET28a(+) | IPTG-inducible expression vector; Kanamycin resistance | Novagen, USA |
| pJHL204 | asd+, CMV promoter, RdRp complex, SV40 promoter, pBR322 ori | [25] |
| pJHL270 | asd+, CMV eukaryotic promoter, Ptrc prokaryotic promoter, pBR322 ori | [23] |
| pJHL204:Cap-Rep | asd+, CMV promoter, RdRp complex, SV40 promoter, pBR322 ori, Cap-P2A-Rep | This study |
| pJHL270:EuCap-Rep+ProCap | asd+, CMV eukaryotic promoter, Cap-P2A-Rep, Ptrc prokaryotic promoter, Cap, pBR322 ori | This study |
| S. Typhimurium | ||
| JOL2500 | Salmonella Typhimurium with genotype ∆lon ∆cpxr ∆sifA ∆asd | Lab stock |
| JOL2885 | JOL2500 carrying pJHL204:Cap-Rep | This study |
| JOL2886 | JOL2500 carrying pJHL270:EuCap-Rep+ProCap | This study |
| E. coli | ||
| DH5α | E. coli F-Φ80dlacZ∆M15∆ (lacZYA-argF) U169recA1 endA1 hsdR17(rk-, mk+) phoA supE44 thi1 gyr A96 relA1λ- | Lab stock |
| JOL2873 | DH5α carrying pET28(a) + Cap | Lab stock |
| JOL2869 | DH5α carrying pET28(a) + Rep | Lab stock |
| JOL2874 | DE3 carrying pET28(a) + Cap | Lab stock |
| JOL2870 | DE3 carrying pET28(a) + Rep | Lab stock |
| E. coli 232 | F – λ – φ80 ∆(lacZYA-argF) endA1 recA1 hadR17 deoR thi-1 glnV44 gyrA96 relA1 ∆asdA4 | Lab stock |
| JOL2881 | E. coli 232 carrying pJHL204:Cap-Rep | This study |
| JOL2879 | E. coli 232 carrying pJHL270:EuCap-Rep+ProCap | This study |
| Primers | ||
| CAP P2A FW | GGGCCCATGACGTATCCAAGGAGGCGTTTC | This study |
| CAP-P2A33 RV | CTTCAGCAGGCTGAAGTTAGTAGCTCCGCTTCCCTTAGGGTTAAGTGGGG | This study |
| P2A33-ORF1 FW | CAGGCTGGAGACGTGGAGGAGAACCCTGGACCTATGCCCAGCAAGAAGAG | This study |
| ORF1 P2A RV | GGCGCGCCTCAGTAATTTATTTCATATGGAAATTCAGGG | This study |
| CAP OEP RV | CTCCAGCCTGCTTCAGCAGGCTGAAGTT | This study |
| ORF1 OEP FW | CCTGCTGAAGCAGGCTGGAGACGTGGAG | This study |
| qPCR-Cap FW | GTCTACATTTCCAGTAGTTTG | [26] |
| qPCR-Cap RV | CTCCCGCCATACCATAA | [26] |
| IFN-γ FW | TCAAGTGGCATAGATGTGGAAGAA | [27] |
| IFN-γ RV | TGGCTCTGCAGGATTTTCATG | [27] |
| TNF-α FW | CATCTTCTCAAAATTCGAGTGACAA | [27] |
| TNF-α RV | TGGGAGTAGACAAGGTACAACCC | [27] |
| IL-4 FW | ACAGGAGAAGGGACGCCAT | [27] |
| IL-4 RV | GAAGCCCTACAGACGAGCTCA | [27] |
| β-actin FW | AGAGGGAAATCGTGCGTGAC | [27] |
| β-actin RV | CAATAGTGATGACCTGGCCGT | [27] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





