Submitted:
26 October 2023
Posted:
27 October 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Developing pups
2.2.1. Protein isolation and immunoblotting
2.2.2. Immunofluorescent-immunohistochemistry to examine synaptogenesis and astrocyte phagocytosis in developing pup brains
2.2.3. Confocal imaging and analysis
2.3. Organotypic Brain Slice Cultures (OBSCs)
2.3.1. Organotypic Brain Slice preparation
2.3.2. Organotypic Brain Slice Culture
2.3.3. Cell viability assay
2.3.4. Protein isolation and immunoblotting
2.3.5. Immunofluorescent-immunohistochemistry to examine astrocyte phagocytosis in OBSCs
2.3.6. Confocal imaging and analysis
2.4. Statistical Analysis
3. Results
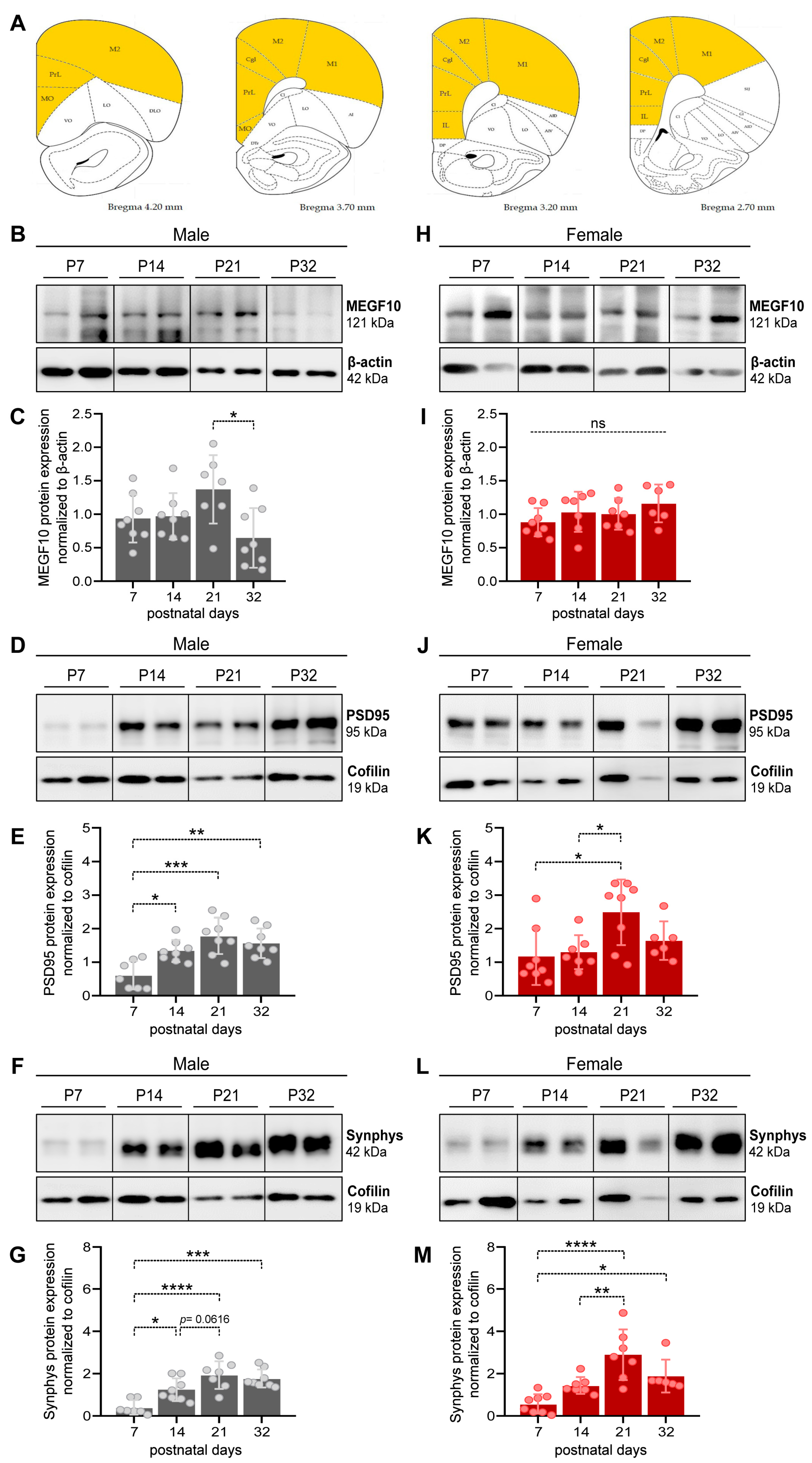
3.1. MEGF10, PSD95 and synaptophysin expression in the PFC of male and female developing rat pups
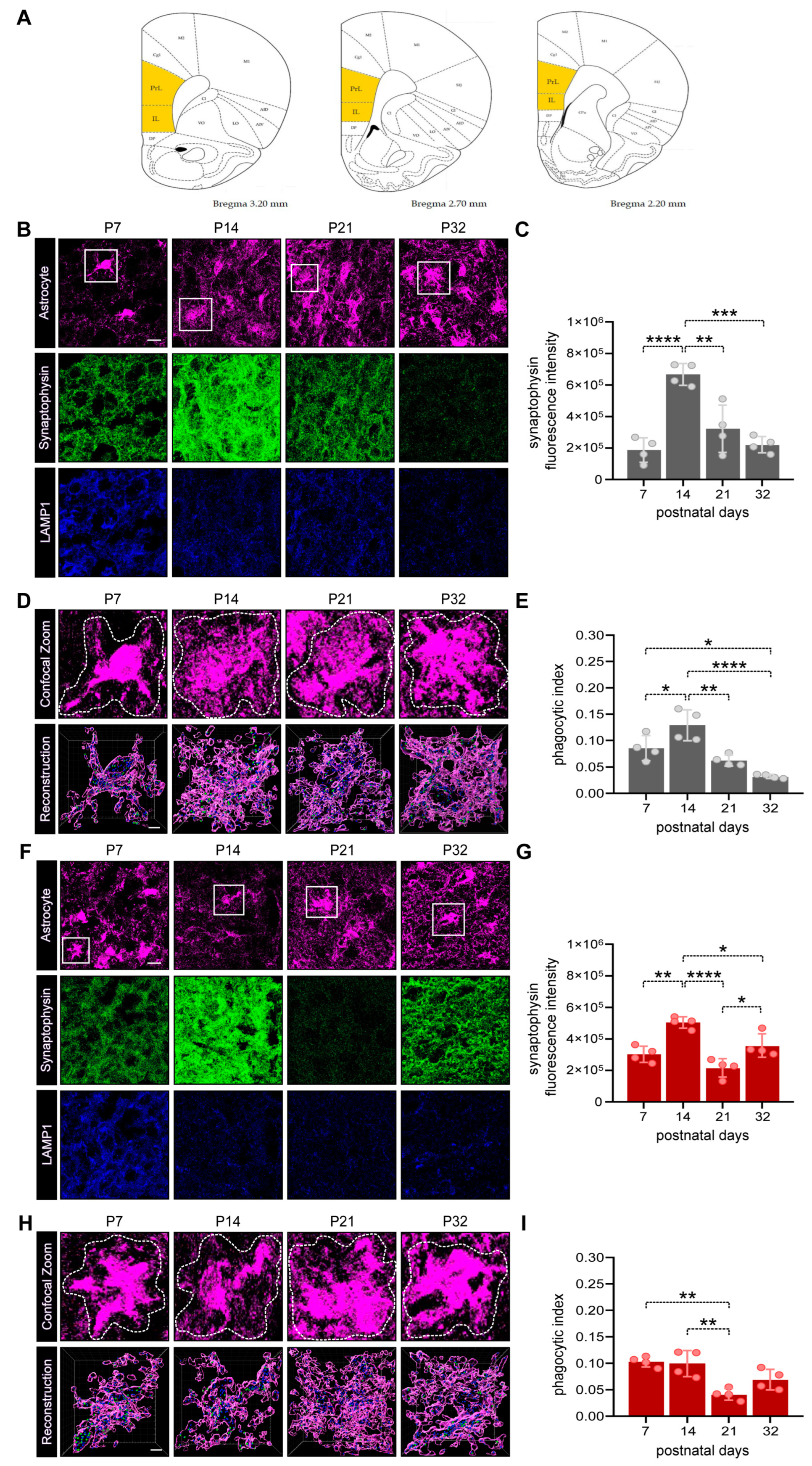
3.2. Astrocyte-dependent synapse elimination in the PFC of male and female developing rat pups
3.3. Establishing organotypic brain slice cultures (OBSCs) to examine synapse elimination in the PFC during early postnatal developmental stages
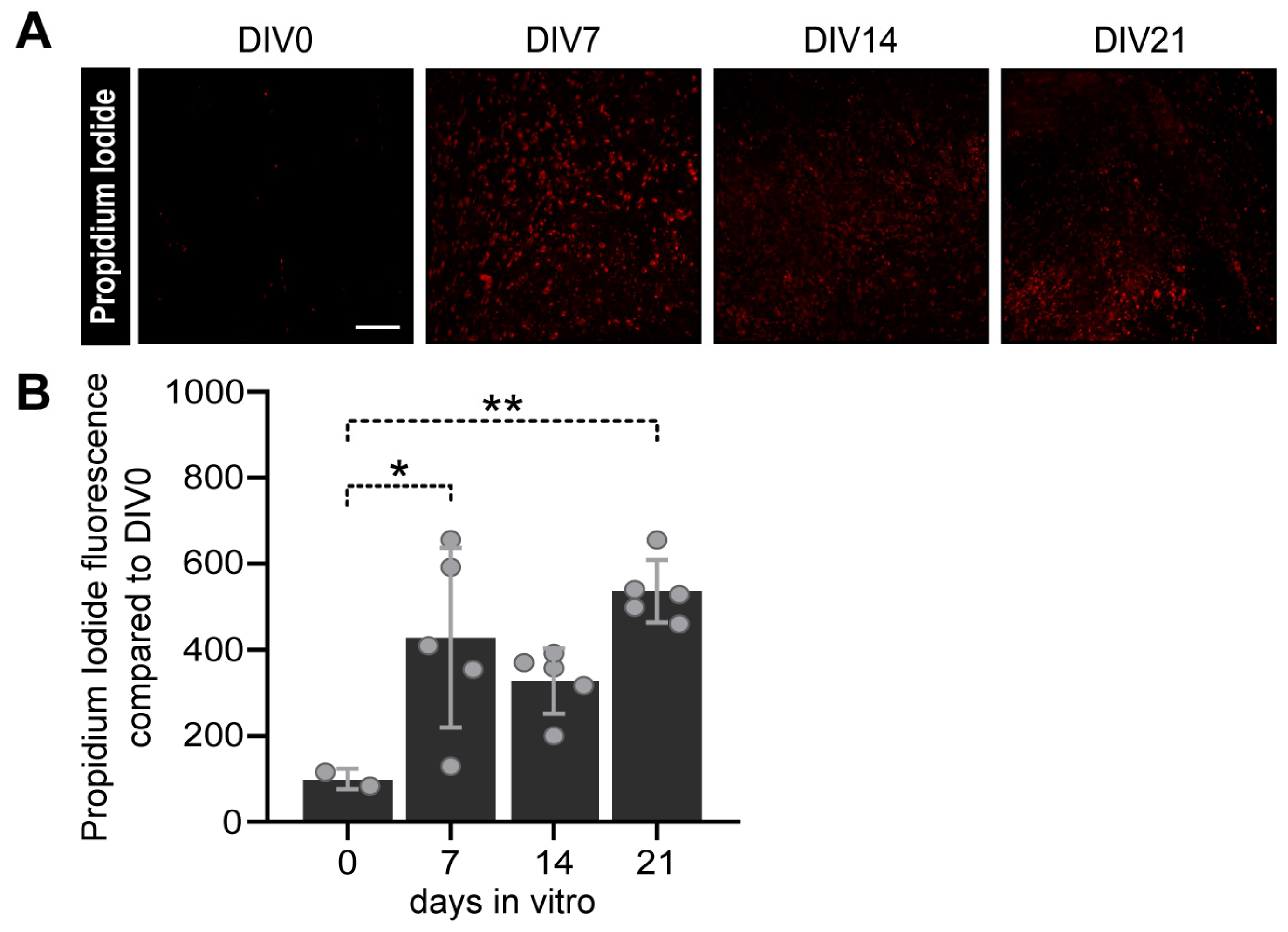
3.3.1. Viability assay
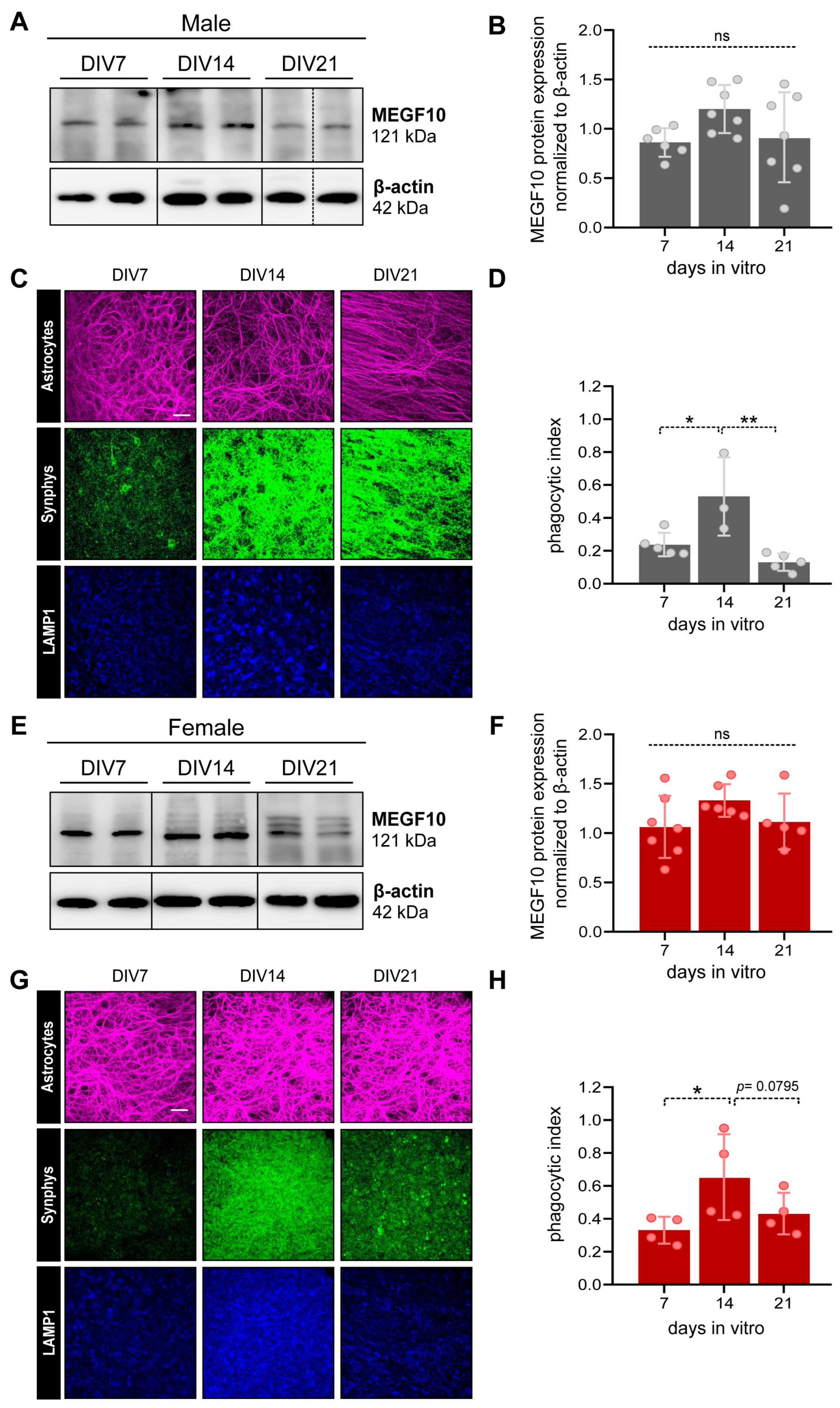
3.3.2. MEGF10 expression and astrocyte-dependent synapse elimination in the PFC of OBSCs derived from male and female rat pups
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Magistretti, P.J.; Allaman, I. Lactate in the Brain: From Metabolic End-Product to Signalling Molecule. Nat Rev Neurosci 2018, 19, 235–249. [CrossRef]
- Hashimoto, Y.; Greene, C.; Munnich, A.; Campbell, M. The CLDN5 Gene at the Blood-Brain Barrier in Health and Disease. Fluids Barriers CNS 2023, 20, 22. [CrossRef]
- Bosworth, A.P.; Allen, N.J. The Diverse Actions of Astrocytes during Synaptic Development. Current Opinion in Neurobiology 2017, 47, 38–43. [CrossRef]
- Verkhratsky, A.; Untiet, V.; Rose, C.R. Ionic Signalling in Astroglia beyond Calcium. J Physiol 2020, 598, 1655–1670. [CrossRef]
- Stogsdill, J.A.; Eroglu, C. The Interplay between Neurons and Glia in Synapse Development and Plasticity. Current Opinion in Neurobiology 2017, 42, 1–8. [CrossRef]
- Akdemir, E.S.; Huang, A.Y.-S.; Deneen, B. Astrocytogenesis: Where, When, and How. F1000Res 2020, 9, 233. [CrossRef]
- Raponi, E.; Agenes, F.; Delphin, C.; Assard, N.; Baudier, J.; Legraverend, C.; Deloulme, J.-C. S100B Expression Defines a State in Which GFAP-Expressing Cells Lose Their Neural Stem Cell Potential and Acquire a More Mature Developmental Stage. Glia 2007, 55, 165–177. [CrossRef]
- Bandeira, F.; Lent, R.; Herculano-Houzel, S. Changing Numbers of Neuronal and Non-Neuronal Cells Underlie Postnatal Brain Growth in the Rat. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 14108–14113. [CrossRef]
- Milbocker, K.A.; Campbell, T.S.; Collins, N.; Kim, S.; Smith, I.F.; Roth, T.L.; Klintsova, A.Y. Glia-Driven Brain Circuit Refinement Is Altered by Early-Life Adversity: Behavioral Outcomes. Front. Behav. Neurosci. 2021, 15, 786234. [CrossRef]
- Chugani, H.T. A Critical Period of Brain Development: Studies of Cerebral Glucose Utilization with PET. Preventive Medicine 1998, 27, 184–188. [CrossRef]
- Juraska, J.M.; Willing, J. Pubertal Onset as a Critical Transition for Neural Development and Cognition. Brain Research 2017, 1654, 87–94. [CrossRef]
- Bushong, E.A.; Martone, M.E.; Ellisman, M.H. Maturation of Astrocyte Morphology and the Establishment of Astrocyte Domains during Postnatal Hippocampal Development. Int. j. dev. neurosci. 2004, 22, 73–86. [CrossRef]
- Allen, N.J.; Eroglu, C. Cell Biology of Astrocyte-Synapse Interactions. Neuron 2017, 96, 697–708. [CrossRef]
- Perez-Catalan, N.A.; Doe, C.Q.; Ackerman, S.D. The Role of Astrocyte-Mediated Plasticity in Neural Circuit Development and Function. Neural Dev 2021, 16, 1. [CrossRef]
- Cahoy, J.D.; Emery, B.; Kaushal, A.; Foo, L.C.; Zamanian, J.L.; Christopherson, K.S.; Xing, Y.; Lubischer, J.L.; Krieg, P.A.; Krupenko, S.A.; et al. A Transcriptome Database for Astrocytes, Neurons, and Oligodendrocytes: A New Resource for Understanding Brain Development and Function. J. Neurosci. 2008, 28, 264–278. [CrossRef]
- Chung, W.-S.; Clarke, L.E.; Wang, G.X.; Stafford, B.K.; Sher, A.; Chakraborty, C.; Joung, J.; Foo, L.C.; Thompson, A.; Chen, C.; et al. Astrocytes Mediate Synapse Elimination through MEGF10 and MERTK Pathways. Nature 2013, 504, 394–400. [CrossRef]
- Lee, J.-H.; Kim, J.; Noh, S.; Lee, H.; Lee, S.Y.; Mun, J.Y.; Park, H.; Chung, W.-S. Astrocytes Phagocytose Adult Hippocampal Synapses for Circuit Homeostasis. Nature 2021, 590, 612–617. [CrossRef]
- Roeper, J. Closing Gaps in Brain Disease — from Overlapping Genetic Architecture to Common Motifs of Synapse Dysfunction. Current Opinion in Neurobiology 2018, 48, 45–51. [CrossRef]
- Blokland, G.A.M.; Grove, J.; Chen, C.-Y.; Cotsapas, C.; Tobet, S.; Handa, R.; St Clair, D.; Lencz, T.; Mowry, B.J.; Periyasamy, S.; et al. Sex-Dependent Shared and Nonshared Genetic Architecture Across Mood and Psychotic Disorders. Biological Psychiatry 2022, 91, 102–117. [CrossRef]
- Seedat, S.; Scott, K.M.; Angermeyer, M.C.; Berglund, P.; Bromet, E.J.; Brugha, T.S.; Demyttenaere, K.; de Girolamo, G.; Haro, J.M.; Jin, R.; et al. Cross-National Associations Between Gender and Mental Disorders in the World Health Organization World Mental Health Surveys. Arch Gen Psychiatry 2009, 66, 785. [CrossRef]
- Drzewiecki, C.M.; Juraska, J.M. The Structural Reorganization of the Prefrontal Cortex during Adolescence as a Framework for Vulnerability to the Environment. Pharmacology Biochemistry and Behavior 2020, 199, 173044. [CrossRef]
- Glantz, L.A.; Gilmore, J.H.; Hamer, R.M.; Lieberman, J.A.; Jarskog, L.F. Synaptophysin and Postsynaptic Density Protein 95 in the Human Prefrontal Cortex from Mid-Gestation into Early Adulthood. Neuroscience 2007, 149, 582–591. [CrossRef]
- Markham, J.A.; Morris, J.R.; Juraska, J.M. Neuron Number Decreases in the Rat Ventral, but Not Dorsal, Medial Prefrontal Cortex between Adolescence and Adulthood. Neuroscience 2007, 144, 961–968. [CrossRef]
- Stoppini, L.; Buchs, P.-A.; Muller, D. A Simple Method for Organotypic Cultures of Nervous Tissue. Journal of Neuroscience Methods 1991, 37, 173–182. [CrossRef]
- Stoppini, L.; Buchs, P.-A.; Brun, R.; Muller, D.; Duport, S.; Parisi, L.; Seebeck, T. Infection of Organotypic Slice Cultures from Rat Central Nervous Tissue with Trypanosoma Brucei Brucei. International Journal of Medical Microbiology 2000, 290, 105–113. [CrossRef]
- Muller, D.; Buchs, P.-A.; Stoppini, L. Time Course of Synaptic Development in Hippocampal Organotypic Cultures. Developmental Brain Research 1993, 71, 93–100. [CrossRef]
- Lancaster, M.A.; Knoblich, J.A. Generation of Cerebral Organoids from Human Pluripotent Stem Cells. Nat Protoc 2014, 9, 2329–2340. [CrossRef]
- Kyrousi, C.; Cappello, S. Using Brain Organoids to Study Human Neurodevelopment, Evolution and Disease. WIREs Dev Biol 2020, 9. [CrossRef]
- Humpel, C. Organotypic Brain Slice Cultures: A Review. Neuroscience 2015, 305, 86–98. [CrossRef]
- Kamikubo, Y.; Jin, H.; Zhou, Y.; Niisato, K.; Hashimoto, Y.; Takasugi, N.; Sakurai, T. Ex Vivo Analysis Platforms for Monitoring Amyloid Precursor Protein Cleavage. Front. Mol. Neurosci. 2023, 15, 1068990. [CrossRef]
- Humpel, C. Organotypic Brain Slice Cultures. Current Protocols in Immunology 2018, 123, e59. [CrossRef]
- Hezel, M.; Ebrahimi, F.; Koch, M.; Dehghani, F. Propidium Iodide Staining: A New Application in Fluorescence Microscopy for Analysis of Cytoarchitecture in Adult and Developing Rodent Brain. Micron 2012, 43, 1031–1038. [CrossRef]
- Staal, J.A.; Alexander, S.R.; Liu, Y.; Dickson, T.D.; Vickers, J.C. Characterization of Cortical Neuronal and Glial Alterations during Culture of Organotypic Whole Brain Slices from Neonatal and Mature Mice. PLoS ONE 2011, 6, e22040. [CrossRef]
- Huttenlocher, P.R.; Dabholkar, A.S. Regional Differences in Synaptogenesis in Human Cerebral Cortex. J. Comp. Neurol. 1997, 387, 167–178. [CrossRef]
- Petanjek, Z.; Judaš, M.; Šimić, G.; Rašin, M.R.; Uylings, H.B.M.; Rakic, P.; Kostović, I. Extraordinary Neoteny of Synaptic Spines in the Human Prefrontal Cortex. Proc. Natl. Acad. Sci. U.S.A. 2011, 108, 13281–13286. [CrossRef]
- Peter R., H. Synaptic Density in Human Frontal Cortex — Developmental Changes and Effects of Aging. Brain Research 1979, 163, 195–205. [CrossRef]
- Knickmeyer, R.C.; Styner, M.; Short, S.J.; Lubach, G.R.; Kang, C.; Hamer, R.; Coe, C.L.; Gilmore, J.H. Maturational Trajectories of Cortical Brain Development through the Pubertal Transition: Unique Species and Sex Differences in the Monkey Revealed through Structural Magnetic Resonance Imaging. Cerebral Cortex 2010, 20, 1053–1063. [CrossRef]
- Whitford, T.J.; Rennie, C.J.; Grieve, S.M.; Clark, C.R.; Gordon, E.; Williams, L.M. Brain Maturation in Adolescence: Concurrent Changes in Neuroanatomy and Neurophysiology. Hum. Brain Mapp. 2007, 28, 228–237. [CrossRef]
- Dosenbach, N.U.F.; Nardos, B.; Cohen, A.L.; Fair, D.A.; Power, J.D.; Church, J.A.; Nelson, S.M.; Wig, G.S.; Vogel, A.C.; Lessov-Schlaggar, C.N.; et al. Prediction of Individual Brain Maturity Using fMRI. Science 2010, 329, 1358–1361. [CrossRef]
- Michaelson, S.D.; Müller, T.M.; Bompolaki, M.; Miranda Tapia, A.P.; Villarroel, H.S.; Mackay, J.P.; Balogun, P.J.; Urban, J.H.; Colmers, W.F. Long-Lived Organotypic Slice Culture Model of the Rat Basolateral Amygdala. Current Protocols 2021, 1. [CrossRef]
- Sengpiel, F. The Critical Period. Current Biology 2007, 17, R742–R743. [CrossRef]
- Guirado, R.; Perez-Rando, M.; Ferragud, A.; Gutierrez-Castellanos, N.; Umemori, J.; Carceller, H.; Nacher, J.; Castillo-Gómez, E. A Critical Period for Prefrontal Network Configurations Underlying Psychiatric Disorders and Addiction. Front. Behav. Neurosci. 2020, 14, 51. [CrossRef]
- Benediktsson, A.M.; Schachtele, S.J.; Green, S.H.; Dailey, M.E. Ballistic Labeling and Dynamic Imaging of Astrocytes in Organotypic Hippocampal Slice Cultures. Journal of Neuroscience Methods 2005, 141, 41–53. [CrossRef]
- Rooney, B.; Leng, K.; McCarthy, F.; Rose, I.V.L.; Herrington, K.A.; Bax, S.; Chin, M.Y.; Fathi, S.; Leonetti, M.; Kao, A.W.; et al. mTOR Controls Neurotoxic Lysosome Exocytosis in Inflammatory Reactive Astrocytes; Cell Biology, 2021; [CrossRef]
- Arora, T.; Mehta, A.K.; Joshi, V.; Mehta, K.D.; Rathor, N.; Mediratta, P.K.; Sharma, K.K. Substitute of Animals in Drug Research: An Approach Towards Fulfillment of 4R’s. Indian J Pharm Sci 2011, 73, 1–6. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



