Submitted:
10 March 2025
Posted:
12 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- 1
- reaction between elements
- 2
- Composition of elements
- 3
- intensity of the specific physical concept
2. Reactions Between Elements
- A.
- example of acid rain
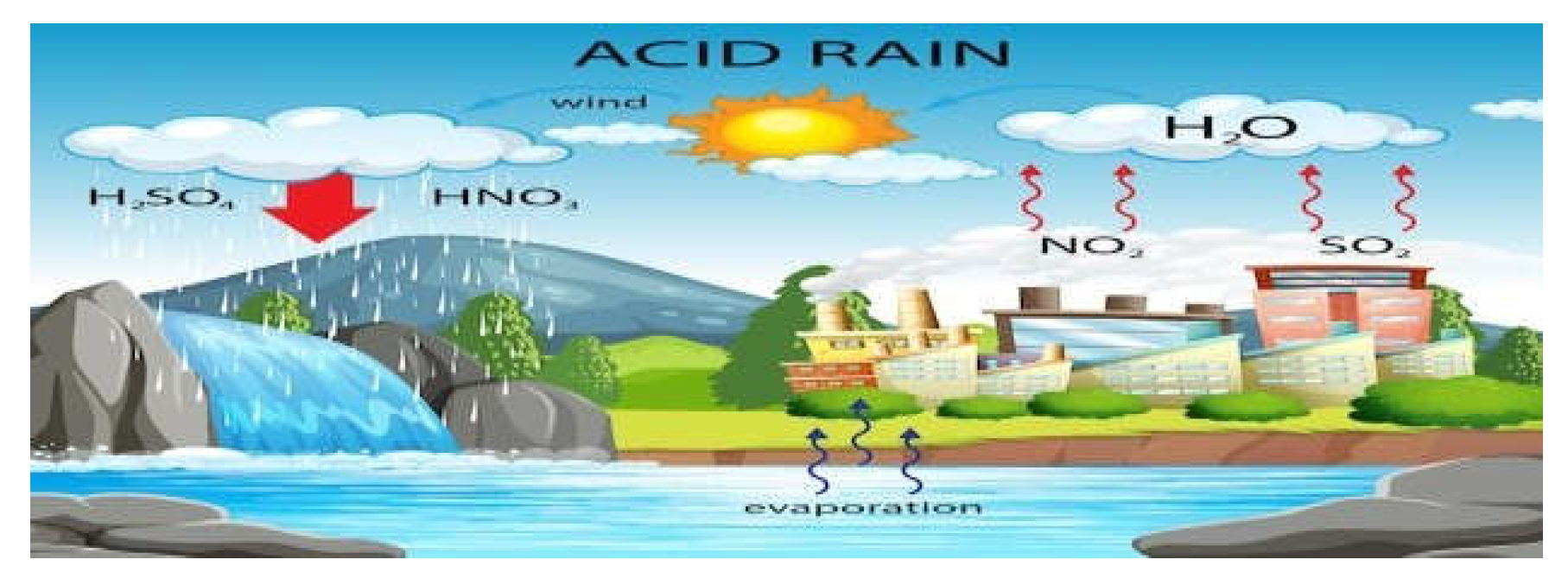
- B.
- example of rust
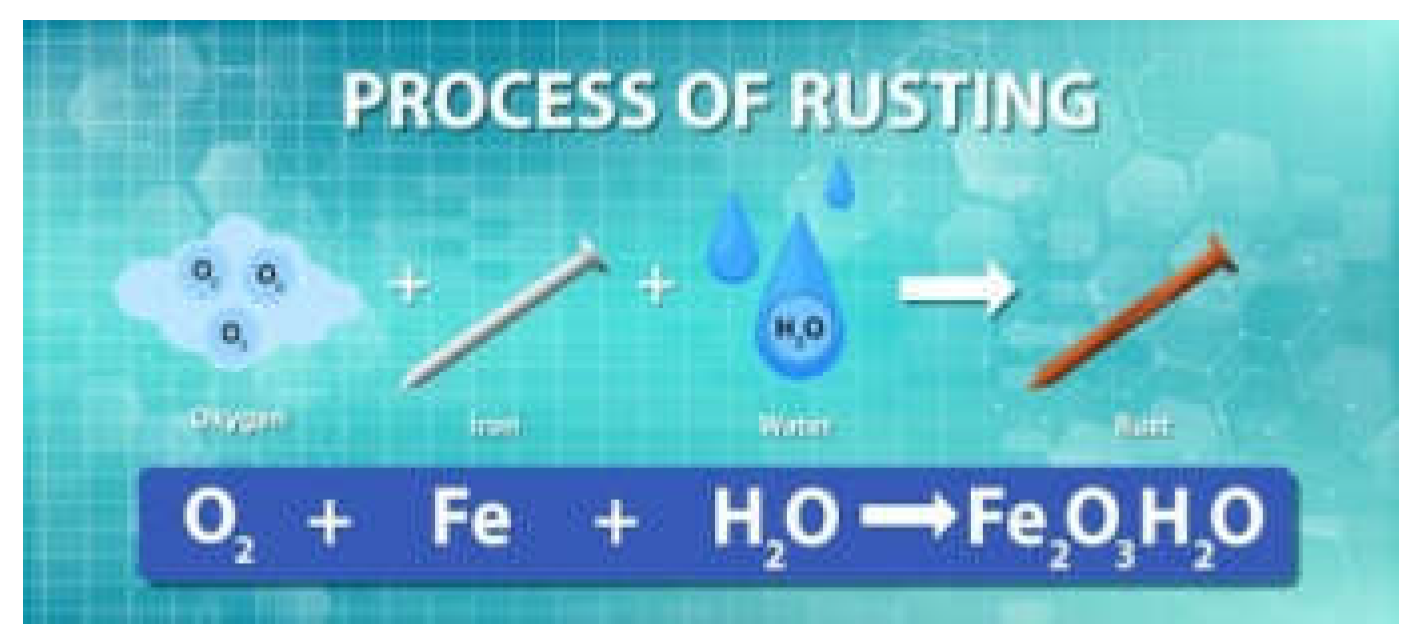
- c.
- example of water
- 1)
- synthesis reaction A + B → AB is when the reactants combine to form the single element
- 2)
- analysis reaction AB → A + B is when the reagents decompose giving rise to simple elements
- 3)
- The single exchange reaction A + BC → AC + B is when a simple substance reacts with a compound forming a new simple substance and another compound.
- 4)
-
double substitution reaction AB + CD → AD + CB is when compound substances react with another compound substance giving rise to two compound substances.The conditions for chemical reaction are contact between reactants and chemical affinity.In this sense, chemical reactions are capable of altering physical concepts by comparing the before and after of the chemical reaction. It is worth mentioning that it is possible to observe this pattern within chemistry.
3. Intensity of the Specific Physical Concept
4. Composition of Elements
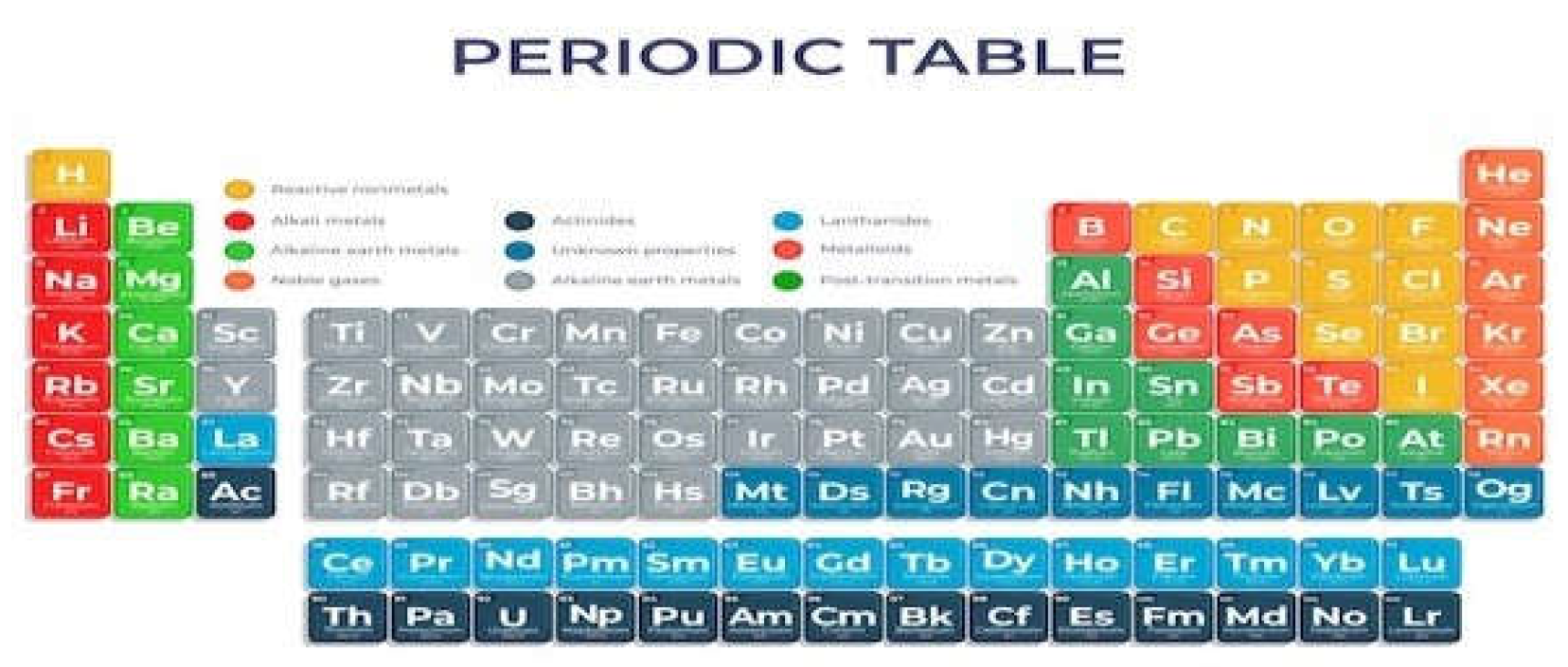
-
Metals: Metals make up most of the elements on the Periodic Table. Some examples are gold, silver, copper, zinc, iron, platinum, aluminum, sodium, potassium, among others. Elements belonging to this group have the following main properties:
- -
- - to have shine
- -
- They are solid
- -
- -conducts electric current
- -
- conducts heat
- -
- They are malleable
- -
- They are ductile
-
Non-metals: They are composed of 11 elements carbon, nitrogen, phosphorus, oxygen, sulfur, selenium, fluorine, chlorine, bromine, iodine and astatine that have different properties than metals:
- -
- don't shine
- -
- Does not conduct electricity
- -
- Does not conduct heat
- -
- fragmentation occurs
-
Semimetals: They are composed of 7 elements boron, silicon, germanium, arsenic, antimony, tellurium and polonium that have intermediate properties to metals and non-metals:
- -
- They have shine
- -
- poor conduction of electricity
- -
-
Separation occurs.
- Noble gases: They are the elements of family 18 of the Periodic Table. They are helium, neon, argon, krypton, xenon and radon.
- hydrogen: Hydrogen is different from any other chemical element, as it does not fit into any of the groups presented.
- A)
- Mercury (hg) and bromine (br): both are liquid at room temperature, but have different characteristics. Mercury has characteristics that allow its use in the manufacture of mirrors and thermometers. Bromine has characteristics that allow its use in firefighting.
- B)
- Carbon (c), phosphorus (p), sulfur (s)...: both are solid, but have different characteristics. Carbon has characteristics that allow its use in the production of energy and in the manufacture of jewelry. Phosphorus has characteristics that make it used in the manufacture of matchboxes. Sulfur has characteristics that allow its use in the production of fertilizers and paper.
- C)
- Oxygen (o), nitrogen (n) both gases, but with different characteristics. Oxygen is used in the respiration of many living things. Nitrogen has characteristics that make it used in dyes and explosives.
5. Relationship of Uses of Elements with Physical Concepts
6. The Physics Between Different Elements
- I.
- LEVELS OF ORGANIZATION AND THEIR COMPARISONS
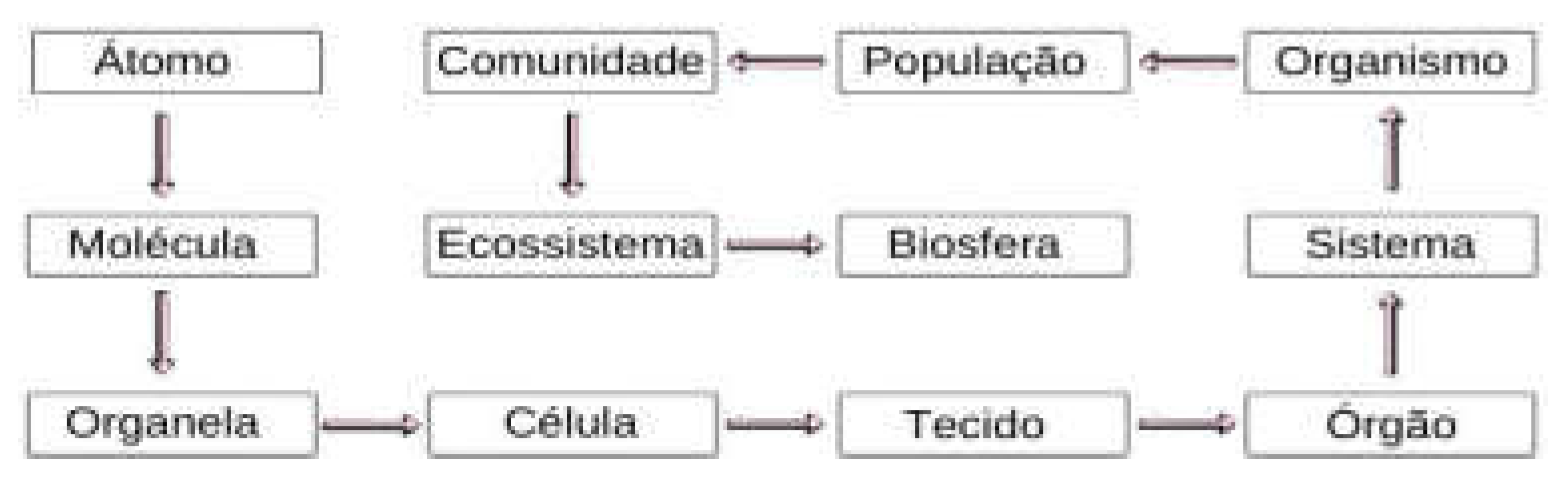
7. Examples that Reinforce the Theory
- A)
- Balls of different elemental compositions with the same shape tend to move differently.
- B)
- Styrofoam and iron of the same shape and size have different masses.
- C)
- an increase in the same chemical element causes changes in physical concepts, such as the amount of carbon. an example is the differences between methane (CH4) and pentane (C5H12), which are the same chemical elements in different quantities. Methane can be found in its physical gaseous state, while pentane can be found in its liquid state. Furthermore, the melting and boiling points are different between methane and pentane.
8. Knowing Planet Earth
9. Quantum Physics
10. General Theory of Relativity
11. Quantum Physics and General Theory of Relativity
12. Theory of Everything
13. Representation and Its Relations
- RE X → EF X ∨ IC X → EF X ∨ CO X → EFX ∨ RE X + IC X + CO X → EF X ≠ RE Y → EF Y ∨ IC Y → EF Y ∨ CO Y → EF Y ∨ RE Y + IC Y + CO Y → EF Y ∴ EF X ≠ EF Y
- RE X = REACTION OF THE X
- ELEMENTS IC X = INTENSITY OF THE
- SPECIFIC PHYSICAL CONCEPT X
- CO X= COMPOSITION OF
- ELEMENT X EF X = ELEMENT OR
- FACT X
- RE Y = REACTION OF Y
- ELEMENTS IC Y = INTENSITY OF
- THE SPECIFIC PHYSICAL
- CONCEPT Y
- CO F = COMPOSITION OF
- ELEMENT Y EF Y = ELEMENT OR
- FACT Y
14. Observing the specific elemental composition
15. Observing the Specific Reaction
16. Observing the Intensity of the Specific Physical Concept
17. The Case of the Electron
18. Using the Example of Reaction, Composition and intensity of the specific physical concept
19. Elements in the Universe and Their Physical Concepts
20. Conclusions
References
- Stephen Hawking. A Brief History of Time. Intrinsic, rio de janeiro, 256 p. January 2015.
- Hawking, Stephen. The Universe in a nutshell. Arx, Sao Paulo, 216 p. April 2004.
- LAZKOZ, Ruth.How much time has passed since the Big Bang and how is it measured.Science - BBC News Brazil.June 20, 2022.
- String Theory: How to Understand the Universe with the Mathematics of Pythagoras' Music.Science - BBC News Brazil.October 26, 2021.
- SANTOS, Vanessa Sardinha dos. "Levels of Organization in Biology"; Brazil School. Available at: https://brasilescola.uol.com.br/biologia/niveis-organizacao-biologia.htm. Accessed on February 9, 2023.
- What is quantum physics and what is it for?Science - BBC News Brazil.June 30, 2020.
- Costa, Camilla, Pais, Ana. What is Einstein's Theory of general relativity?.Science - BBC News Brazil.May 24, 2019.
- Wilson, Alastair.What was the Universe like before the Big Bang?Science - BBC News Brazil.January 6, 2022.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



