Submitted:
20 October 2023
Posted:
24 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals, Cellular Lines, and Viruses
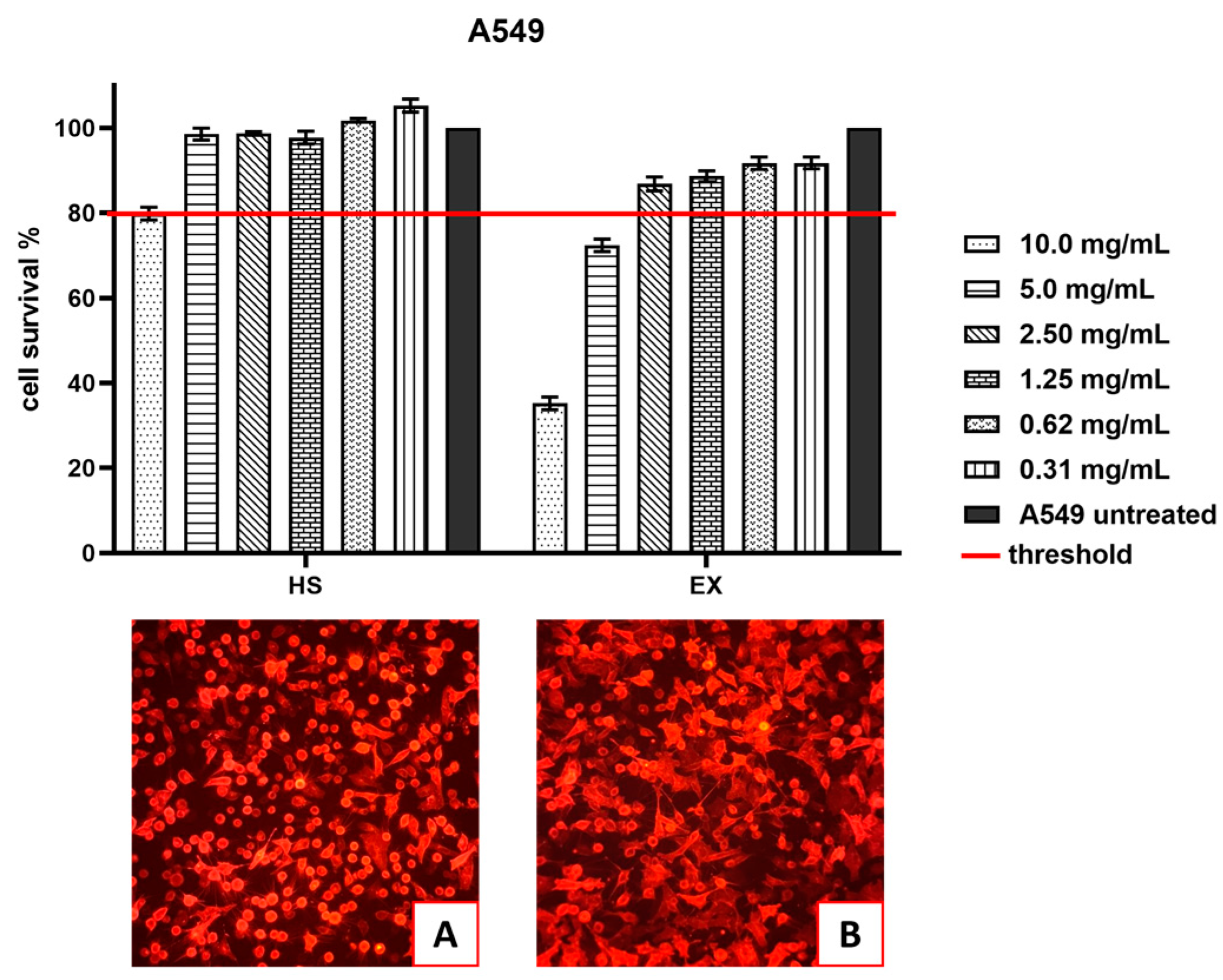
2.2. Cell Viability Assay by MTT
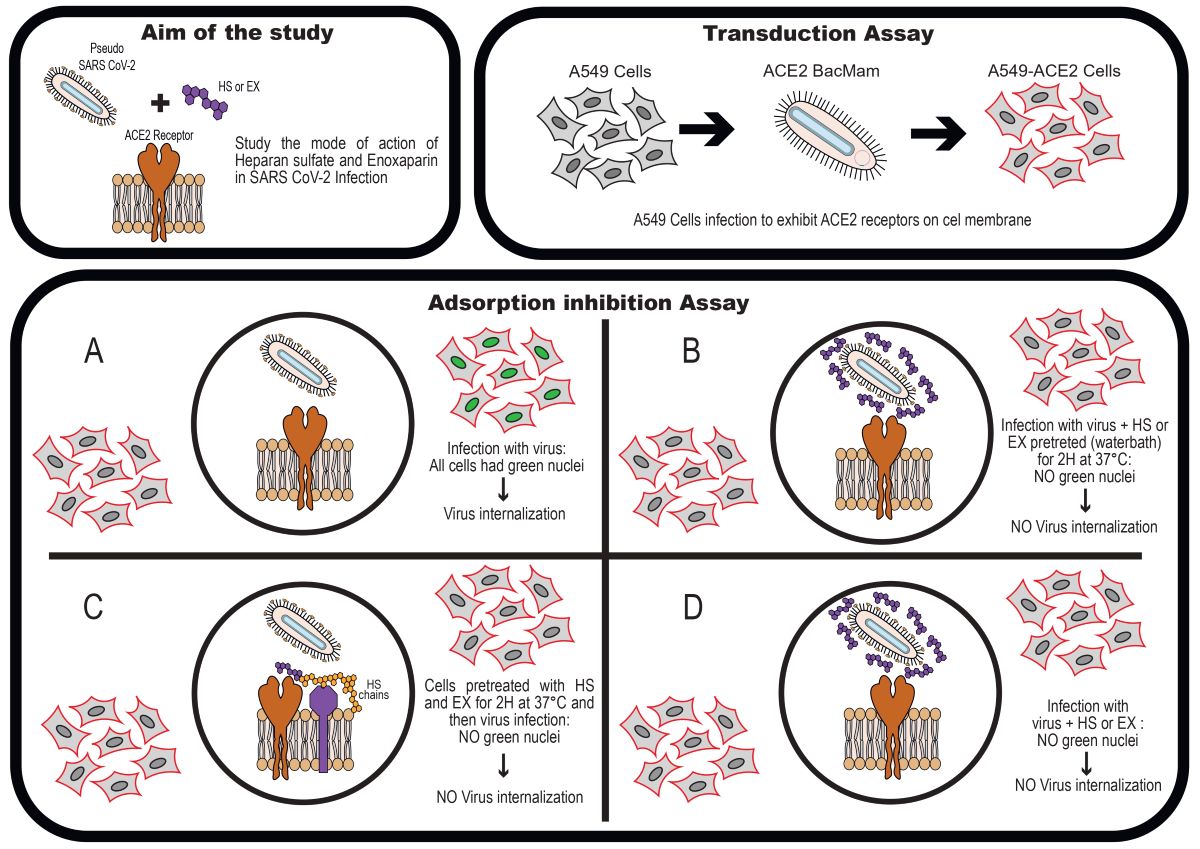
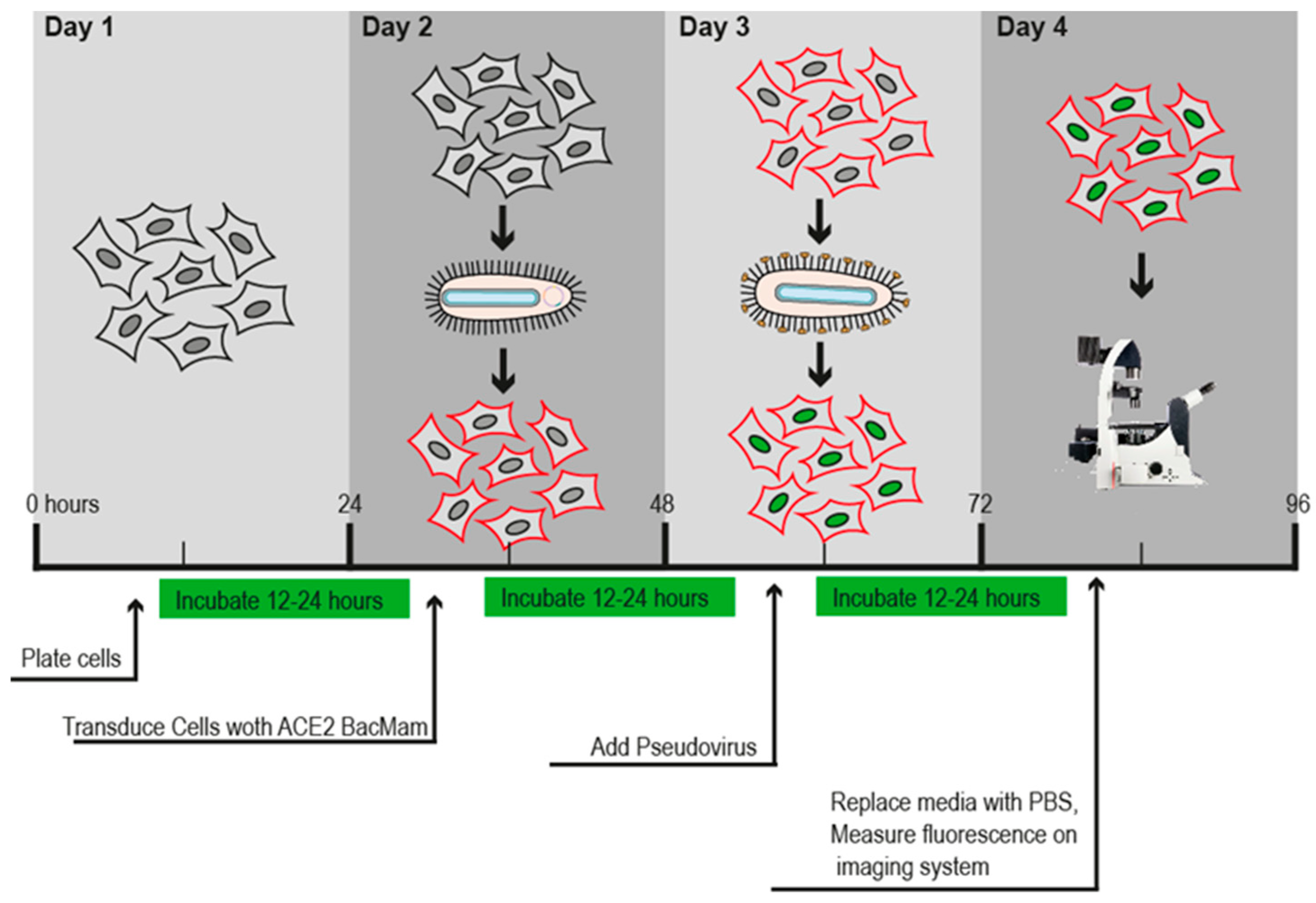
2.3. Transduction assay
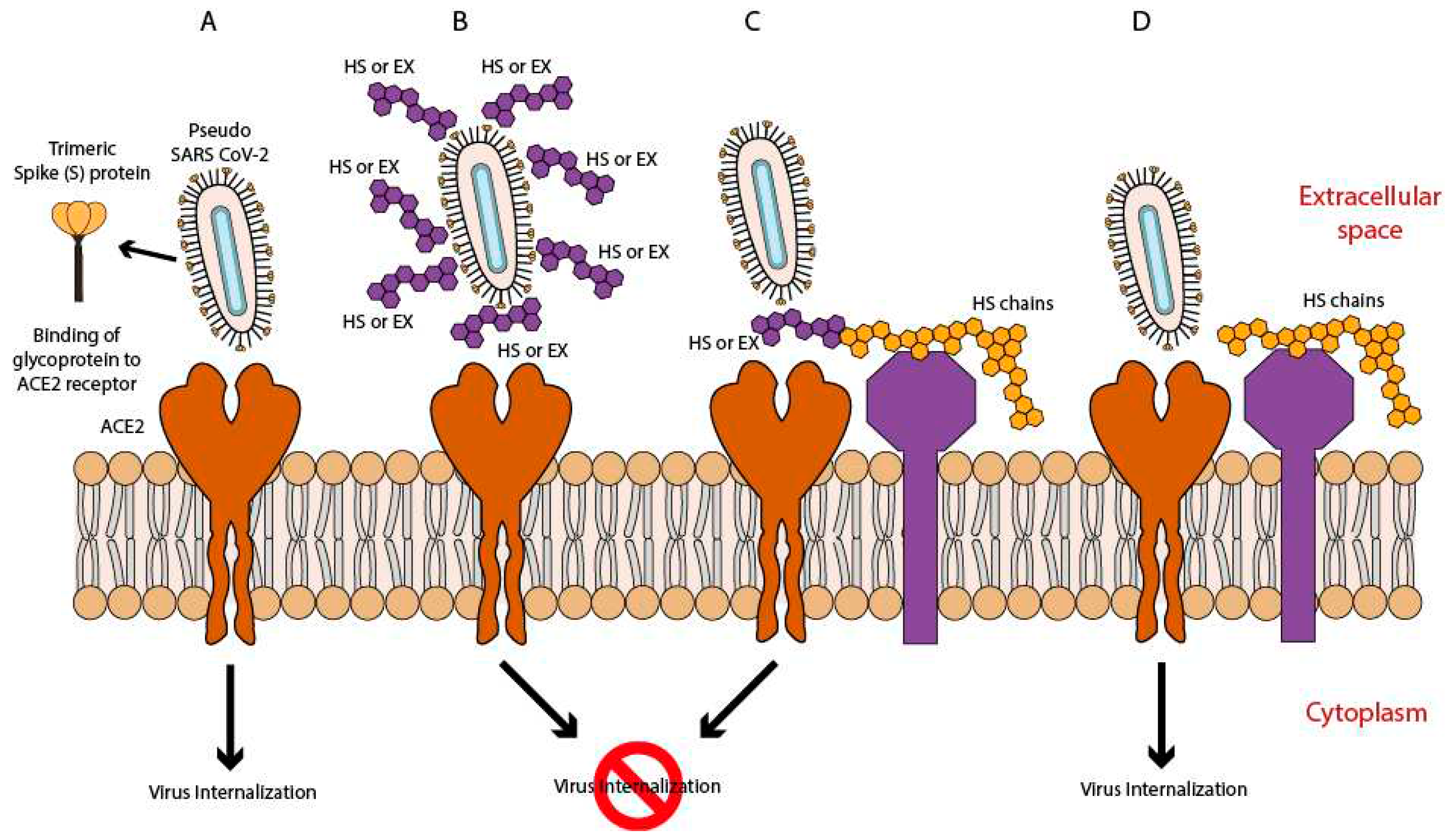
2.4. Adsorption inhibition assay
2.5. Statistical analyses
2.6. Molecular modeling
3. Results and Discussion
3.1. Cell Viability
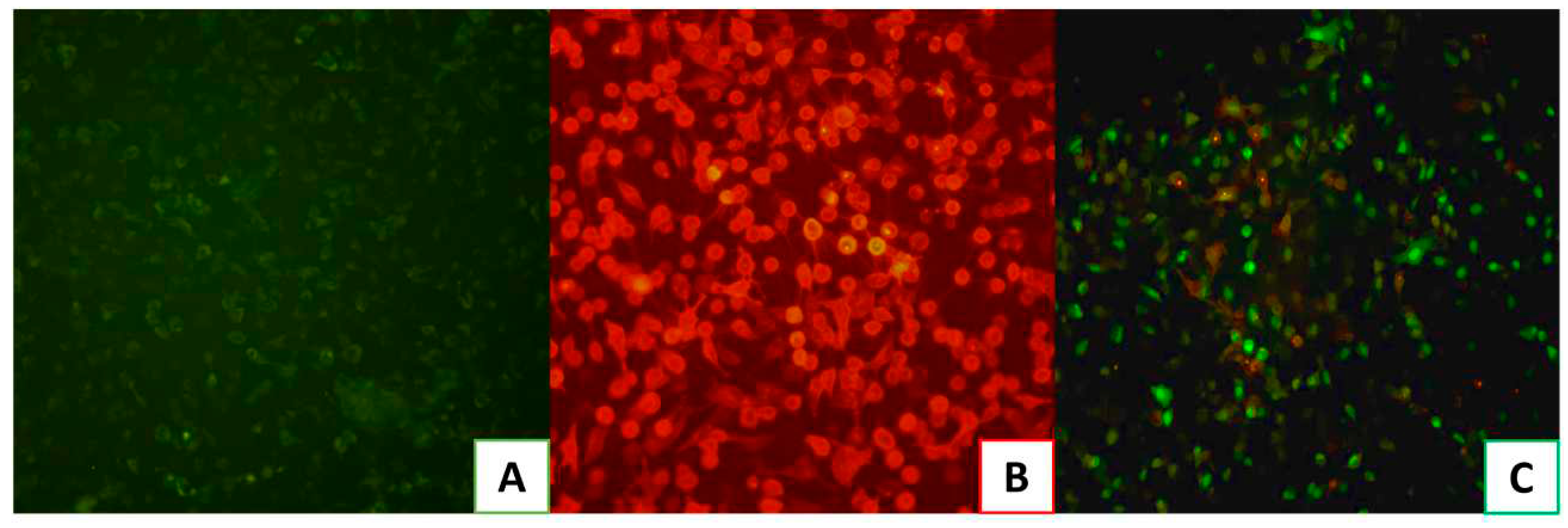
3.2. Pseudovirus SARS-CoV-2 affinity for ACE2 validation.
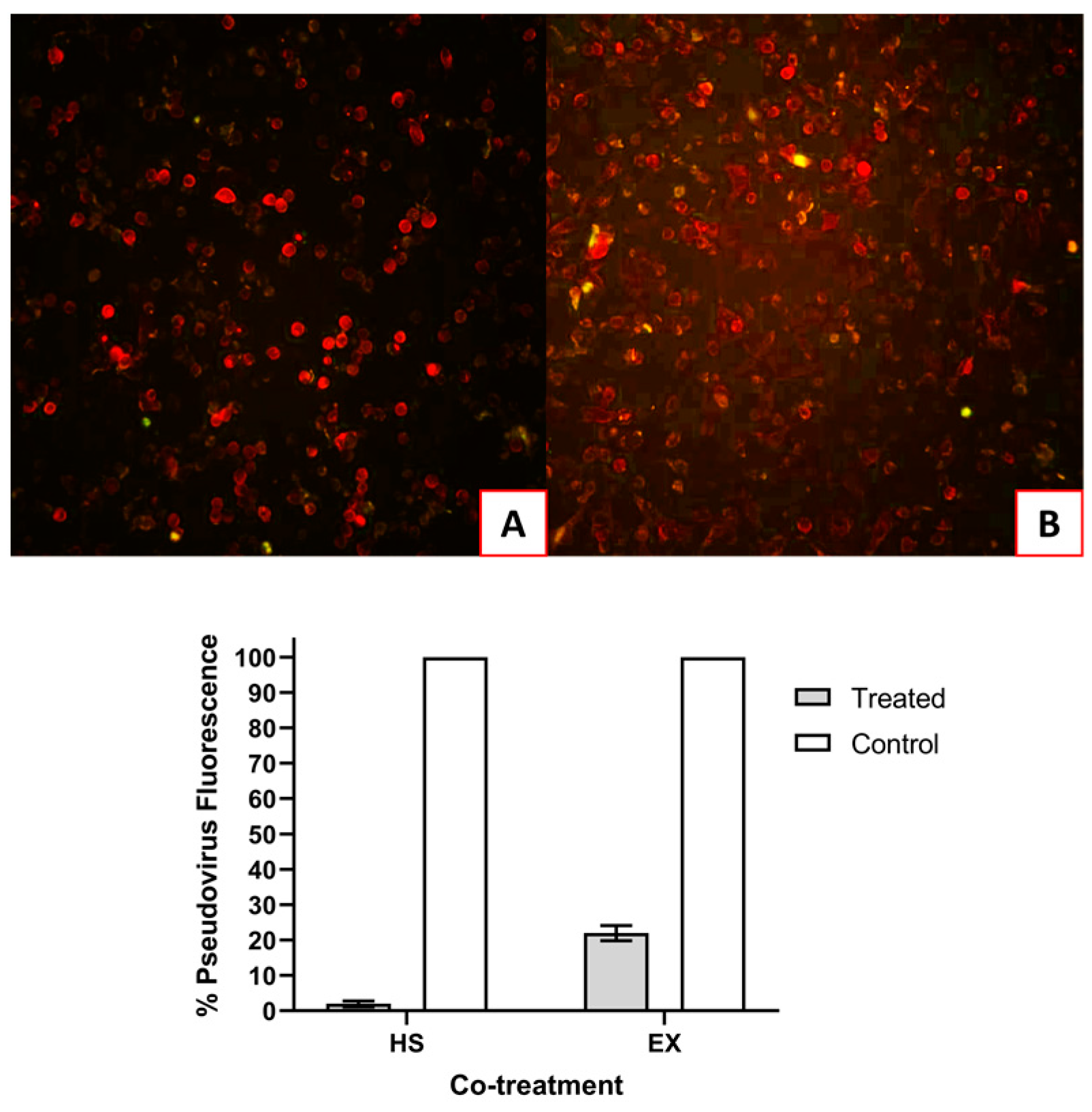
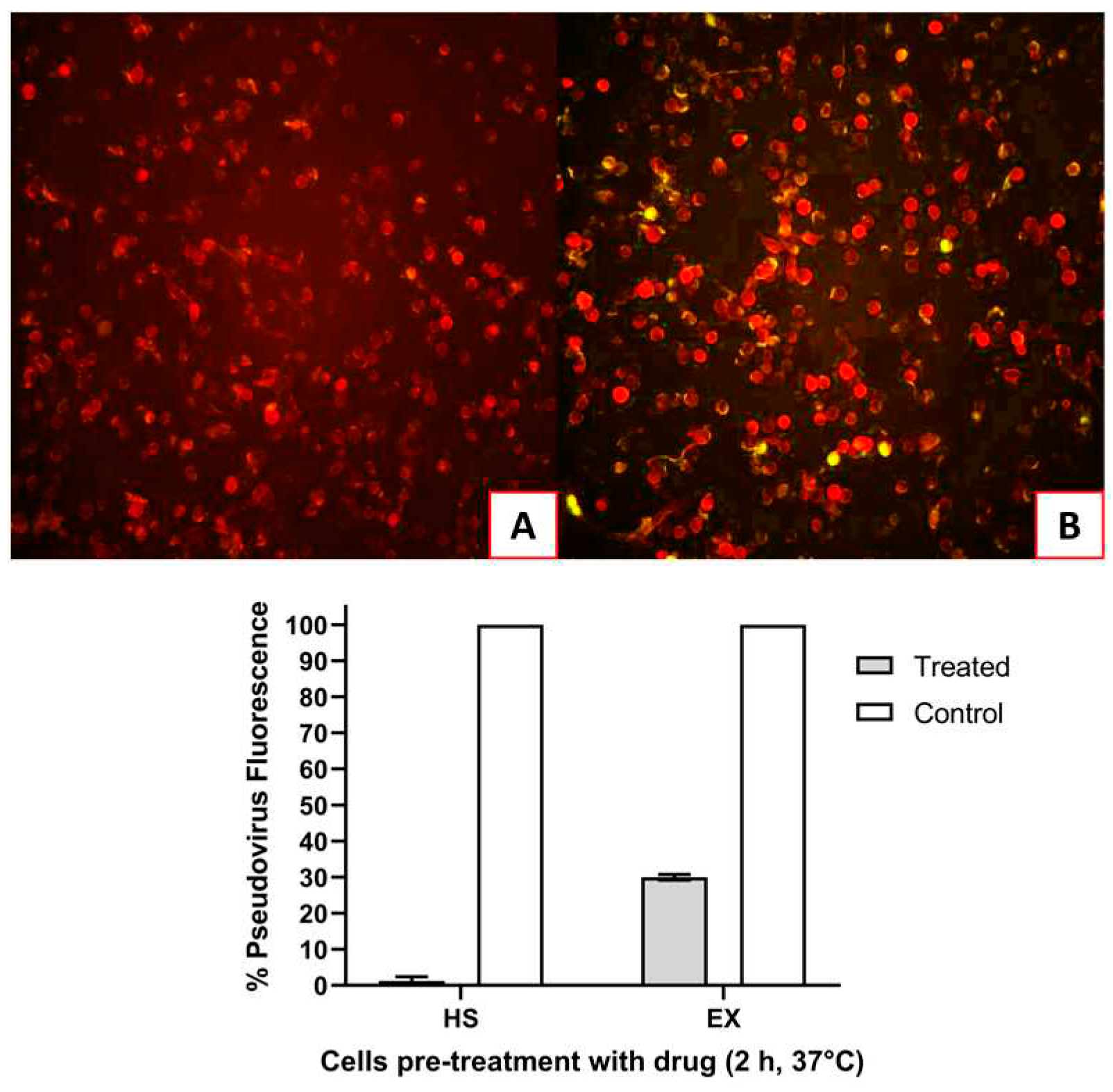
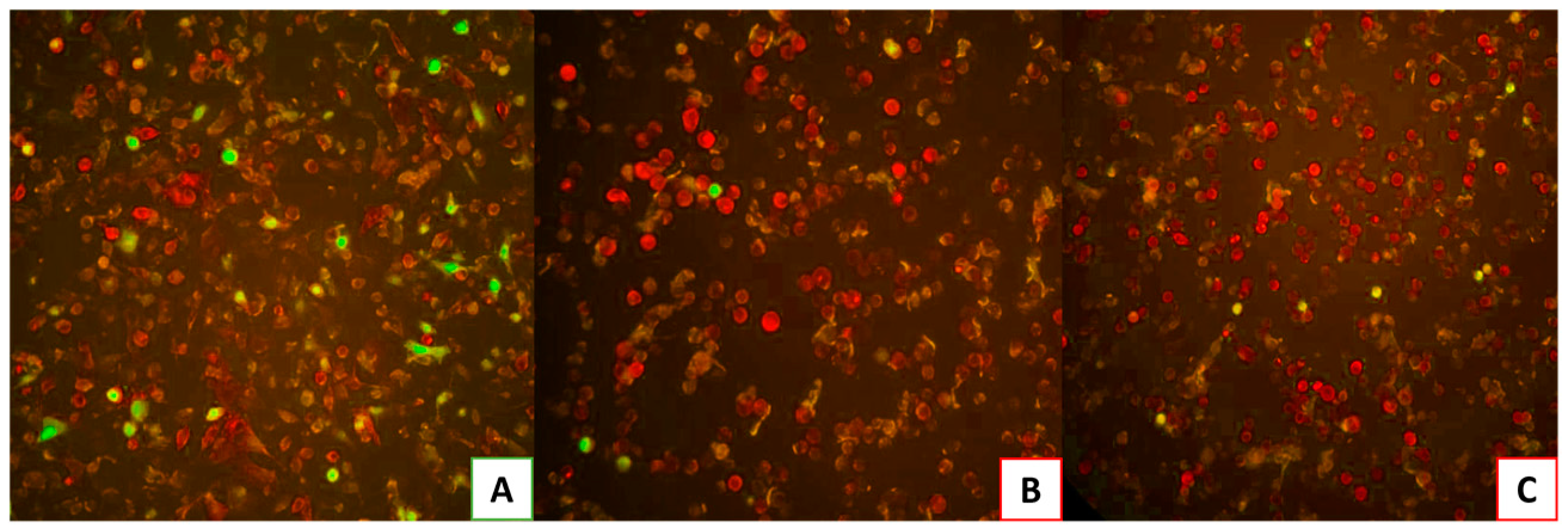
3.3. Antiviral activity of HS and EX
3.4. Molecular modeling
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ullah, H.; Ullah, A.; Gul, A.; Mousavi, T.; Khan, M. W. , Novel coronavirus 2019 (COVID-19) pandemic outbreak: A comprehensive review of the current literature. Vacunas 2021, 22, 106–113. [Google Scholar] [CrossRef]
- Hidalgo, P.; Valdés, M.; González, R.A. Molecular biology of coronaviruses: an overview of virus-host interactions and pathogenesis. 78, 58. [CrossRef]
- Fuochi, V.; Floresta, G.; Emma, R.; Patamia, V.; Caruso, M.; Zagni, C.; Ronchi, F.; Ronchi, C.; Drago, F.; Rescifina, A.; et al. Heparan Sulfate and Enoxaparin Interact at the Interface of the Spike Protein of HCoV-229E but Not with HCoV-OC43. Viruses 2023, 15, 663. [Google Scholar] [CrossRef] [PubMed]
- Hulswit, R.J.G.; Lang, Y.; Bakkers, M.J.G.; Li, W.; Li, Z.; Schouten, A.; Ophorst, B.; van Kuppeveld, F.J.M.; Boons, G.-J.; Bosch, B.-J.; et al. Human coronaviruses OC43 and HKU1 bind to 9- O -acetylated sialic acids via a conserved receptor-binding site in spike protein domain A. Proc. Natl. Acad. Sci. 2019, 116, 2681–2690. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.; Gautam, R.K.; Chopra, H.; Dubey, A.K.; Singla, R.K.; A Rayan, R.; Kamal, M.A. COMPARATIVE HIGHLIGHTS ON MERS-COV, SARS-COV-1, SARS-COV-2, AND NEO-COV. 21, 1245. [Google Scholar] [CrossRef]
- Rawat, P.; Jemimah, S.; Ponnuswamy, P. K.; Gromiha, M. M. , Why are ACE2 binding coronavirus strains SARS-CoV/SARS-CoV-2 wild and NL63 mild? Proteins 2021, 89, 389–398. [Google Scholar] [CrossRef]
- Tandon, R.; Sharp, J. S.; Zhang, F.; Pomin, V. H.; Ashpole, N. M.; Mitra, D.; McCandless, M. G.; Jin, W.; Liu, H.; Sharma, P.; Linhardt, R. J. , Effective Inhibition of SARS-CoV-2 Entry by Heparin and Enoxaparin Derivatives. J. Virol. 2021, 95. [Google Scholar] [CrossRef] [PubMed]
- Gentile, D.; Patamia, V.; Fuochi, V.; Furneri, P. M.; Rescifina, A. , Natural Substances in the Fight of SARS-CoV-2: A Critical Evaluation Resulting from the Cross-Fertilization of Molecular Modeling Data with the Pharmacological Aspects. Current Medicinal Chemistry 2021, 28, 8333–8383. [Google Scholar] [CrossRef] [PubMed]
- Merry, C. L. R.; Lindahl, U.; Couchman, J.; Esko, J. D. , Proteoglycans and Sulfated Glycosaminoglycans. In Essentials of Glycobiology, Varki, A.; Cummings, R. D.; Esko, J. D.; Stanley, P.; Hart, G. W.; Aebi, M.; Mohnen, D.; Kinoshita, T.; Packer, N. H.; Prestegard, J. H.; Schnaar, R. L.; Seeberger, P. H., Eds. 2022; 217–232. [Google Scholar] [CrossRef]
- Gupta, R.; Ponnusamy, M.P. Analysis of sulfates on low molecular weight heparin using mass spectrometry: structural characterization of enoxaparin. Expert Rev. Proteom. 2018, 15, 503–513. [Google Scholar] [CrossRef]
- Florian, J. A.; Kosky, J. R.; Ainslie, K.; Pang, Z.; Dull, R. O.; Tarbell, J. M. , Heparan sulfate proteoglycan is a mechanosensor on endothelial cells. Circ Res 2003, 93, e136–142. [Google Scholar] [CrossRef]
- Möckl, L. The Emerging Role of the Mammalian Glycocalyx in Functional Membrane Organization and Immune System Regulation. Front. Cell Dev. Biol. 2020, 8, 253. [Google Scholar] [CrossRef]
- Constantinou, P. E.; Morgado, M.; Carson, D. D. , Transmembrane Mucin Expression and Function in Embryo Implantation and Placentation. Adv Anat Embryol Cell Biol 2015, 216, 51–68. [Google Scholar]
- Lipowsky, H.H. The Endothelial Glycocalyx as a Barrier to Leukocyte Adhesion and Its Mediation by Extracellular Proteases. Ann. Biomed. Eng. 2011, 40, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Weber, T.; Chandrasekaran, V.; Stamer, I.; Thygesen, M.B.; Terfort, A.; Lindhorst, T.K. Switching of Bacterial Adhesion to a Glycosylated Surface by Reversible Reorientation of the Carbohydrate Ligand. Angew. Chem. Int. Ed. 2014, 53, 14583–14586. [Google Scholar] [CrossRef] [PubMed]
- Wadowski, P.P.; Jilma, B.; Kopp, C.W.; Ertl, S.; Gremmel, T.; Koppensteiner, R. Glycocalyx as Possible Limiting Factor in COVID-19. Front. Immunol. 2021, 12, 607306. [Google Scholar] [CrossRef] [PubMed]
- Reitsma, S.; Slaaf, D.W.; Vink, H.; van Zandvoort, M.A.M.J.; Oude Egbrink, M.G. The endothelial glycocalyx: composition, functions, and visualization. Pflug. Arch. 2007, 454, 345–359. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Snyder, N.L.; Hartmann, L. Polymers Inspired by Heparin and Heparan Sulfate for Viral Targeting. Macromolecules 2022, 55, 7957–7973. [Google Scholar] [CrossRef] [PubMed]
- Shukla, D.; Liu, J.; Blaiklock, P.; Shworak, N.W.; Bai, X.; Esko, J.D.; Cohen, G.H.; Eisenberg, R.J.; Rosenberg, R.D.; Spear, P.G. A Novel Role for 3-O-Sulfated Heparan Sulfate in Herpes Simplex Virus 1 Entry. Cell 1999, 99, 13–22. [Google Scholar] [CrossRef]
- Artpradit, C.; Robinson, L.N.; Gavrilov, B.K.; Rurak, T.T.; Ruchirawat, M.; Sasisekharan, R. Recognition of heparan sulfate by clinical strains of dengue virus serotype 1 using recombinant subviral particles. Virus Res. 2013, 176, 69–77. [Google Scholar] [CrossRef]
- Israelsson, S.; Gullberg, M.; Jonsson, N.; Roivainen, M.; Edman, K.; Lindberg, A.M. Studies of Echovirus 5 interactions with the cell surface: Heparan sulfate mediates attachment to the host cell. Virus Res. 2010, 151, 170–176. [Google Scholar] [CrossRef]
- Goodfellow, I.G.; Sioofy, A.B.; Powell, R.M.; Evans, D.J. Echoviruses Bind Heparan Sulfate at the Cell Surface. J. Virol. 2001, 75, 4918–4921. [Google Scholar] [CrossRef]
- Gardner, C.L.; Ebel, G.D.; Ryman, K.D.; Klimstra, W.B. Heparan sulfate binding by natural eastern equine encephalitis viruses promotes neurovirulence. Proc. Natl. Acad. Sci. 2011, 108, 16026–16031. [Google Scholar] [CrossRef]
- Compton, T.; Nowlin, D.M.; Cooper, N.R. Initiation of Human Cytomegalovirus Infection Requires Initial Interaction with Cell Surface Heparan Sulfate. Virology 1993, 193, 834–841. [Google Scholar] [CrossRef]
- Trybala, E.; Bergström, T.; Spillmann, D.; Svennerholm, B.; Flynn, S. J.; Ryan, P. , Interaction between pseudorabies virus and heparin/heparan sulfate. Pseudorabies virus mutants differ in their interaction with heparin/heparan sulfate when altered for specific glycoprotein C heparin-binding domain. J Biol Chem 1998, 273, 5047–5052. [Google Scholar] [CrossRef]
- Schowalter, R.M.; Pastrana, D.V.; Buck, C.B. Glycosaminoglycans and Sialylated Glycans Sequentially Facilitate Merkel Cell Polyomavirus Infectious Entry. PLOS Pathog. 2011, 7, e1002161. [Google Scholar] [CrossRef] [PubMed]
- Leistner, C.M.; Gruen-Bernhard, S.; Glebe, D. Role of glycosaminoglycans for binding and infection of hepatitis B virus. Cell. Microbiol. 2007, 10, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Longarela, O.L.; Schmidt, T.T.; Schöneweis, K.; Romeo, R.; Wedemeyer, H.; Urban, S.; Schulze, A. Proteoglycans Act as Cellular Hepatitis Delta Virus Attachment Receptors. PLoS ONE 2013, 8, e58340. [Google Scholar] [CrossRef] [PubMed]
- Gentile, D.; Floresta, G.; Patamia, V.; Chiaramonte, R.; Mauro, G.L.; Rescifina, A.; Vecchio, M. An Integrated Pharmacophore/Docking/3D-QSAR Approach to Screening a Large Library of Products in Search of Future Botulinum Neurotoxin A Inhibitors. Int. J. Mol. Sci. 2020, 21, 9470. [Google Scholar] [CrossRef]
- Cheng, A.; A Best, S.; Merz, K.M.; Reynolds, C.H. GB/SA water model for the Merck molecular force field (MMFF). J. Mol. Graph. Model. 2000, 18, 273–282. [Google Scholar] [CrossRef]
- Stewart, J.J.P. Optimization of parameters for semiempirical methods IV: extension of MNDO, AM1, and PM3 to more main group elements. J. Mol. Model. 2004, 10, 155–164. [Google Scholar] [CrossRef]
- Patamia, V.; Floresta, G.; Zagni, C.; Pistarà, V.; Punzo, F.; Rescifina, A. 1,2-Dibenzoylhydrazine as a Multi-Inhibitor Compound: A Morphological and Docking Study. Int. J. Mol. Sci. 2023, 24, 1425. [Google Scholar] [CrossRef]
- Stewart, J.J.P. MOPAC: A semiempirical molecular orbital program. J. Comput. Mol. Des. 1990, 4, 1–103. [Google Scholar] [CrossRef]
- Ielo, L.; Patamia, V.; Citarella, A.; Efferth, T.; Shahhamzehei, N.; Schirmeister, T.; Stagno, C.; Langer, T.; Rescifina, A.; Micale, N.; et al. Novel Class of Proteasome Inhibitors: In Silico and In Vitro Evaluation of Diverse Chloro(trifluoromethyl)aziridines. Int. J. Mol. Sci. 2022, 23, 12363. [Google Scholar] [CrossRef]
- Wallbrecher, R.; Verdurmen, W.P.R.; Schmidt, S.; Bovee-Geurts, P.H.; Broecker, F.; Reinhardt, A.; van Kuppevelt, T.H.; Seeberger, P.H.; Brock, R. The stoichiometry of peptide-heparan sulfate binding as a determinant of uptake efficiency of cell-penetrating peptides. Cell. Mol. Life Sci. 2013, 71, 2717–2729. [Google Scholar] [CrossRef]
- Krieger, E.; Vriend, G. , YASARA View—molecular graphics for all devices—from smartphones to workstations. Bioinformatics 2014, 30, 2981–2982. [Google Scholar] [CrossRef]
- Krieger, E.; Koraimann, G.; Vriend, G. Increasing the precision of comparative models with YASARA NOVA-a self-parameterizing force field. Proteins 2002, 47, 393–402. [Google Scholar] [CrossRef]
- Cardullo, N.; Catinella, G.; Floresta, G.; Muccilli, V.; Rosselli, S.; Rescifina, A.; Bruno, M.; Tringali, C. Synthesis of Rosmarinic Acid Amides as Antioxidative and Hypoglycemic Agents. J. Nat. Prod. 2019, 82, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Floresta, G.; Amata, E.; Gentile, D.; Romeo, G.; Marrazzo, A.; Pittalà, V.; Salerno, L.; Rescifina, A. Fourfold Filtered Statistical/Computational Approach for the Identification of Imidazole Compounds as HO-1 Inhibitors from Natural Products. Mar. Drugs 2019, 17, 113. [Google Scholar] [CrossRef] [PubMed]
- Ciaffaglione, V.; Intagliata, S.; Pittalà, V.; Marrazzo, A.; Sorrenti, V.; Vanella, L.; Rescifina, A.; Floresta, G.; Sultan, A.; Greish, K.; et al. New Arylethanolimidazole Derivatives as HO-1 Inhibitors with Cytotoxicity against MCF-7 Breast Cancer Cells. Int. J. Mol. Sci. 2020, 21, 1923. [Google Scholar] [CrossRef] [PubMed]
- Zagni, C.; Pistarà, V.; Oliveira, L.A.; Castilho, R.M.; Romeo, G.; Chiacchio, U.; Rescifina, A. Serendipitous discovery of potent human head and neck squamous cell carcinoma anti-cancer molecules: A fortunate failure of a rational molecular design. Eur. J. Med. Chem. 2017, 141, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Floresta, G.; Patamia, V.; Zagni, C.; Rescifina, A. Adipocyte fatty acid binding protein 4 (FABP4) inhibitors. An update from 2017 to early 2022. Eur. J. Med. Chem. 2022, 240, 114604. [Google Scholar] [CrossRef] [PubMed]
- Floresta, G.; Patamia, V.; Gentile, D.; Molteni, F.; Santamato, A.; Rescifina, A.; Vecchio, M. , Repurposing of FDA-Approved Drugs for Treating Iatrogenic Botulism: A Paired 3D-QSAR/Docking Approach(dagger). ChemMedChem 2020, 15, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zeng, W.; Kabubei, K. M.; Rasanathan, J. J. K.; Kazungu, J.; Ginindza, S.; Mtshali, S.; Salinas, L. E.; McClelland, A.; Buissonniere, M.; Lee, C. T.; Chuma, J.; Veillard, J.; Matsebula, T.; Chopra, M. , Modelling the economic burden of SARS-CoV-2 infection in health care workers in four countries. Nat Commun 2023, 14, 2791. [Google Scholar] [CrossRef] [PubMed]
- Komarova, N.L.; Azizi, A.; Wodarz, D. Network models and the interpretation of prolonged infection plateaus in the COVID19 pandemic. Epidemics 2021, 35, 100463–100463. [Google Scholar] [CrossRef] [PubMed]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; Xiang, Z.; Mu, Z.; Chen, X.; Chen, J.; Hu, K.; Jin, Q.; Wang, J.; Qian, Z. , Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef] [PubMed]
- Walls, A. C.; Park, Y. J.; Tortorici, M. A.; Wall, A.; McGuire, A. T.; Veesler, D. , Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 183, 1735. [Google Scholar] [CrossRef]
- Ennemoser, M.; Rieger, J.; Muttenthaler, E.; Gerlza, T.; Zatloukal, K.; Kungl, A. J. , Enoxaparin and Pentosan Polysulfate Bind to the SARS-CoV-2 Spike Protein and Human ACE2 Receptor, Inhibiting Vero Cell Infection. Biomedicines 2021, 10. [Google Scholar] [CrossRef]
| Amount/ Well | Final Concentration | |
|---|---|---|
| ACE2 BacMam | 5 μL | 6.6 × 108 VG/mL |
| Sodium butyrate | 0.6 μL | 2 mM |
| Complete media | 44.4 μL |
| Amount/ Well | Final Concentration | |
|---|---|---|
| SARS-CoV-2 Pseudovirus | 2.5 μL | 3.3 × 108 VG/mL |
| Sodium butyrate | 0.6 μL | 2 mM |
| Compounds | 5.0 mg/mL (HS); 1.25 mg/mL (EX) | |
| Complete media | adjust |
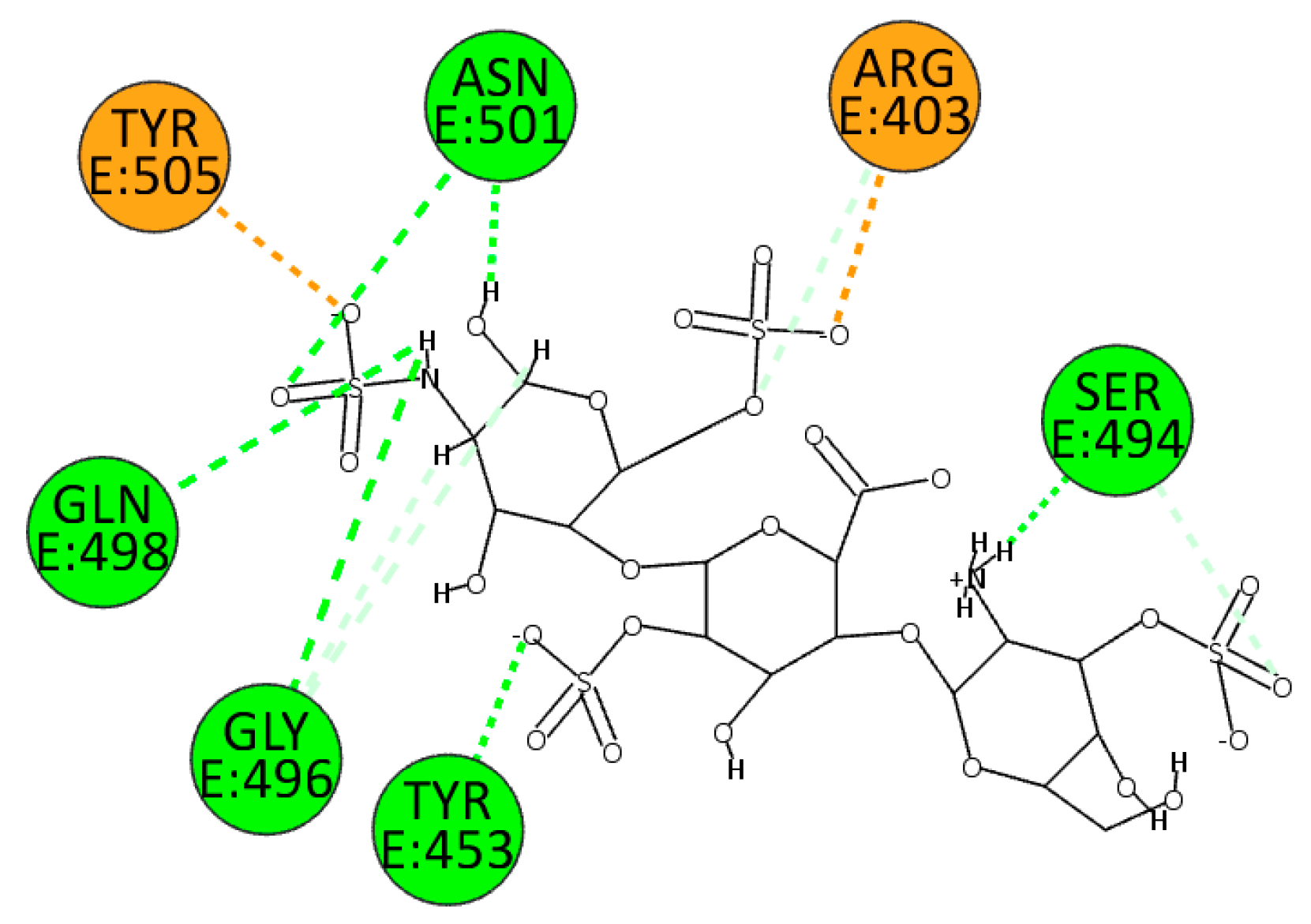
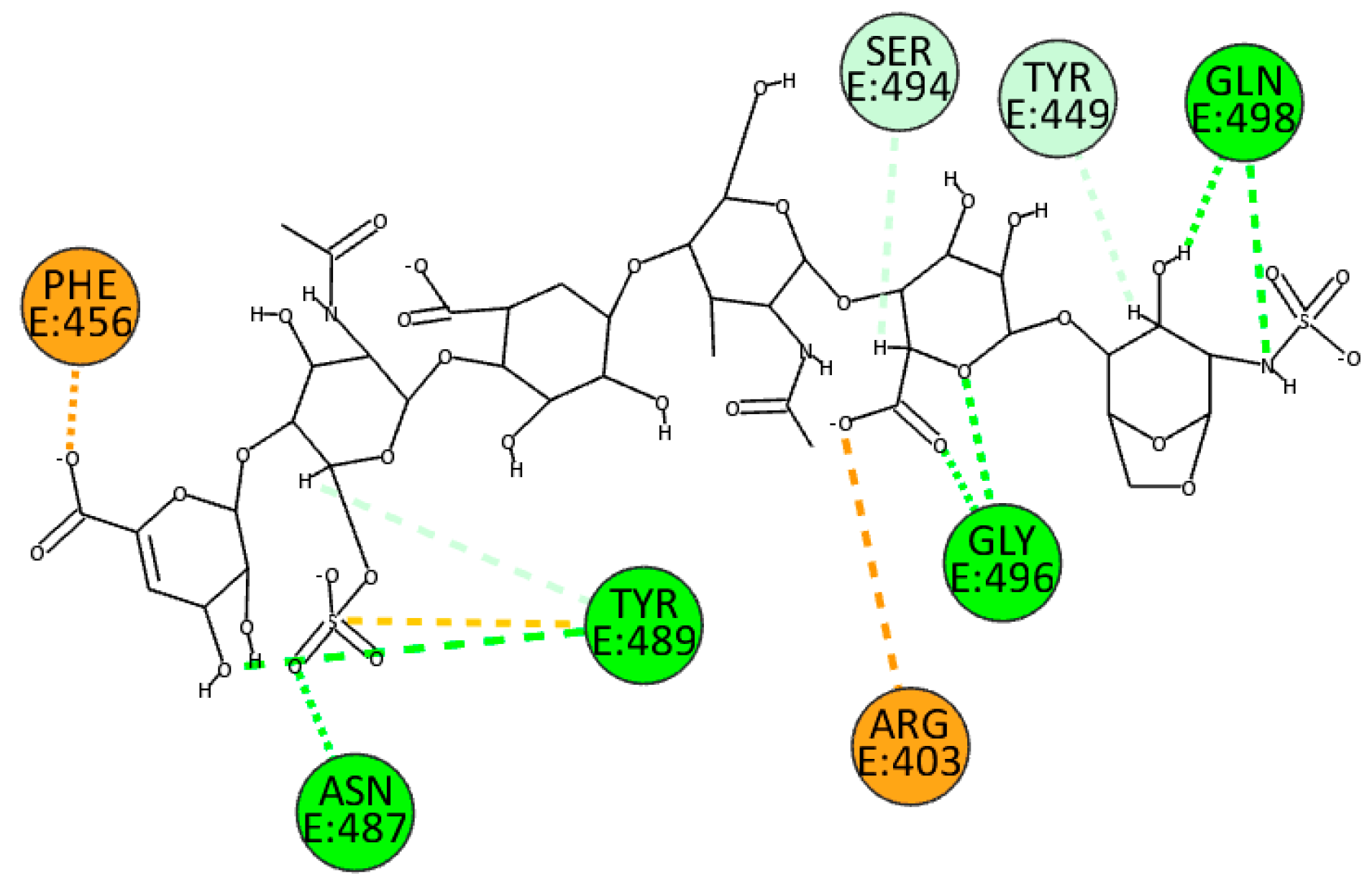
| PDB ID: 6M0J | ||
| Attractive Charge | Hydrogen Bond | |
| HS | Arg403, Tyr505 | Tyr453, Ser494, Gly496, Gln498, Asn501 |
| EX | Arg403, Phe456 | Asn487, Tyr489, Gly496, Gln498 |
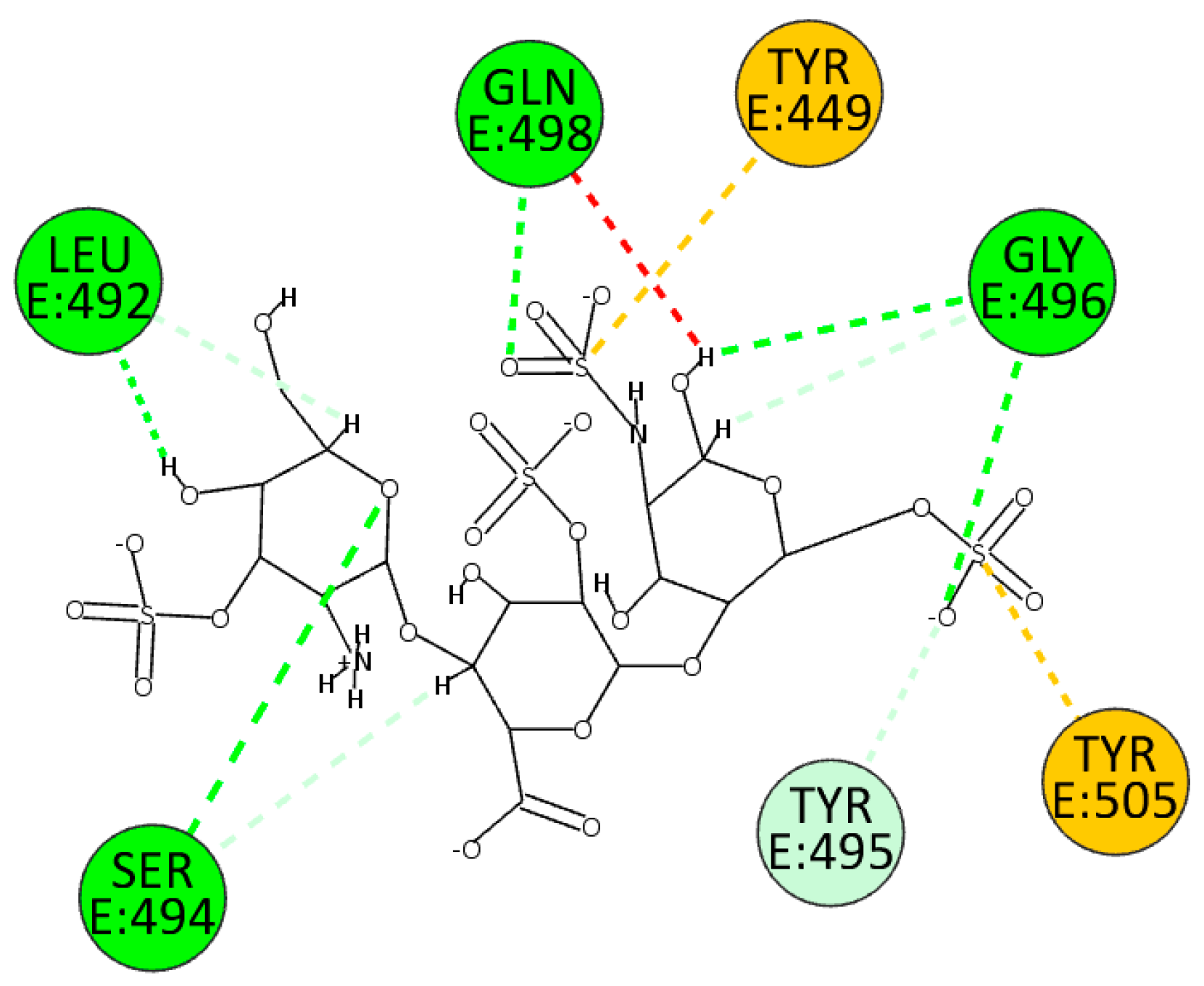
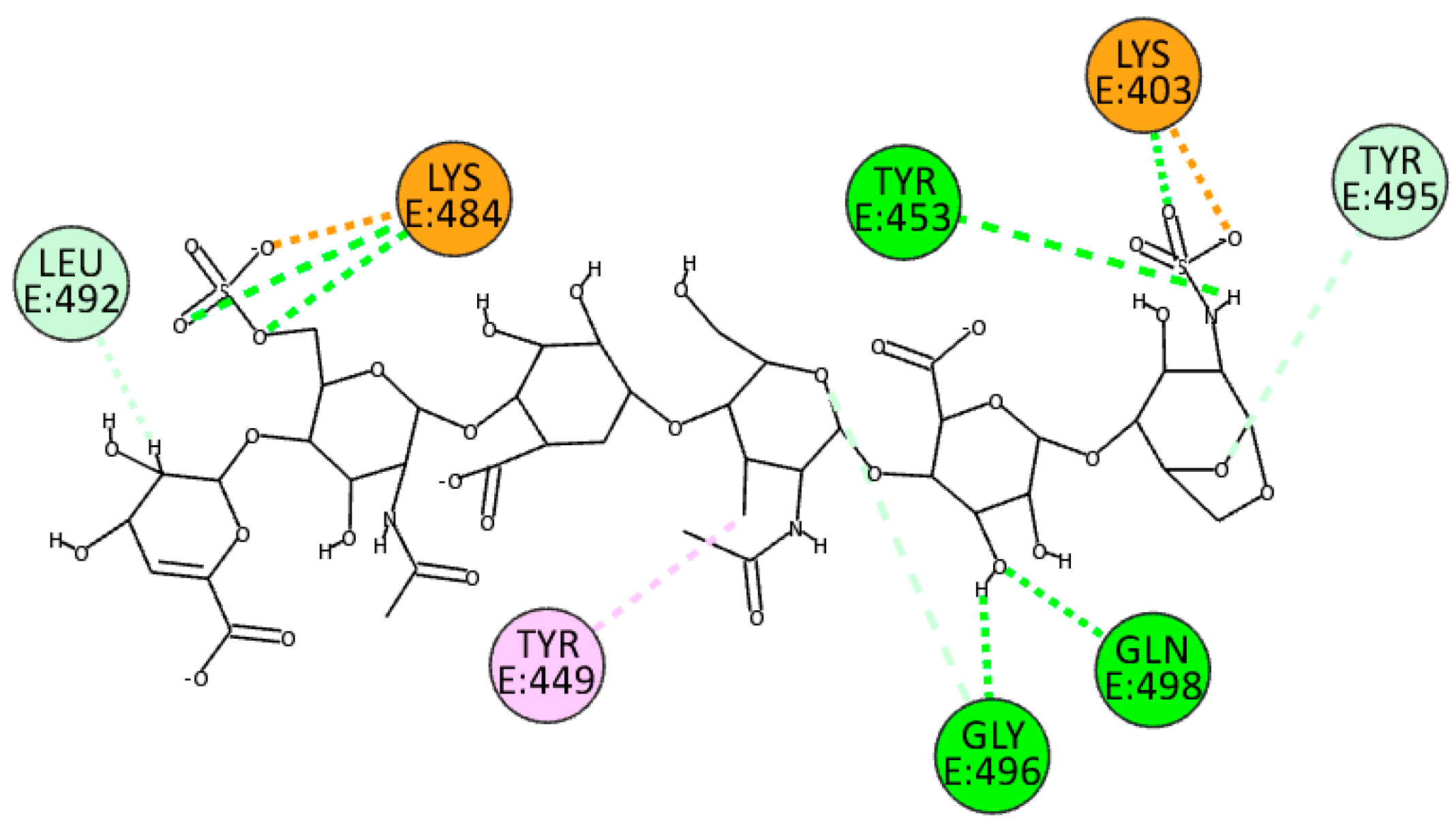
| BA.2.86 variant | ||
| Attractive Charge | Hydrogen Bond | |
| HS | Tyr449, Tyr505 | Leu492, Ser494, Gly496, Gln498 |
| EX | Lys403, Lys484 | Tyr453, Gly496, Gln498 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




