Submitted:
22 October 2023
Posted:
23 October 2023
Read the latest preprint version here
Abstract
Keywords:
Antagonistic pleiotropy “has so far failed to explain what is actually at the background of the ‘switching-over’ of the developmental effect of genes to the damaging one and at what stage” Vladimir Dilman(Dilman, 1986)
Introduction
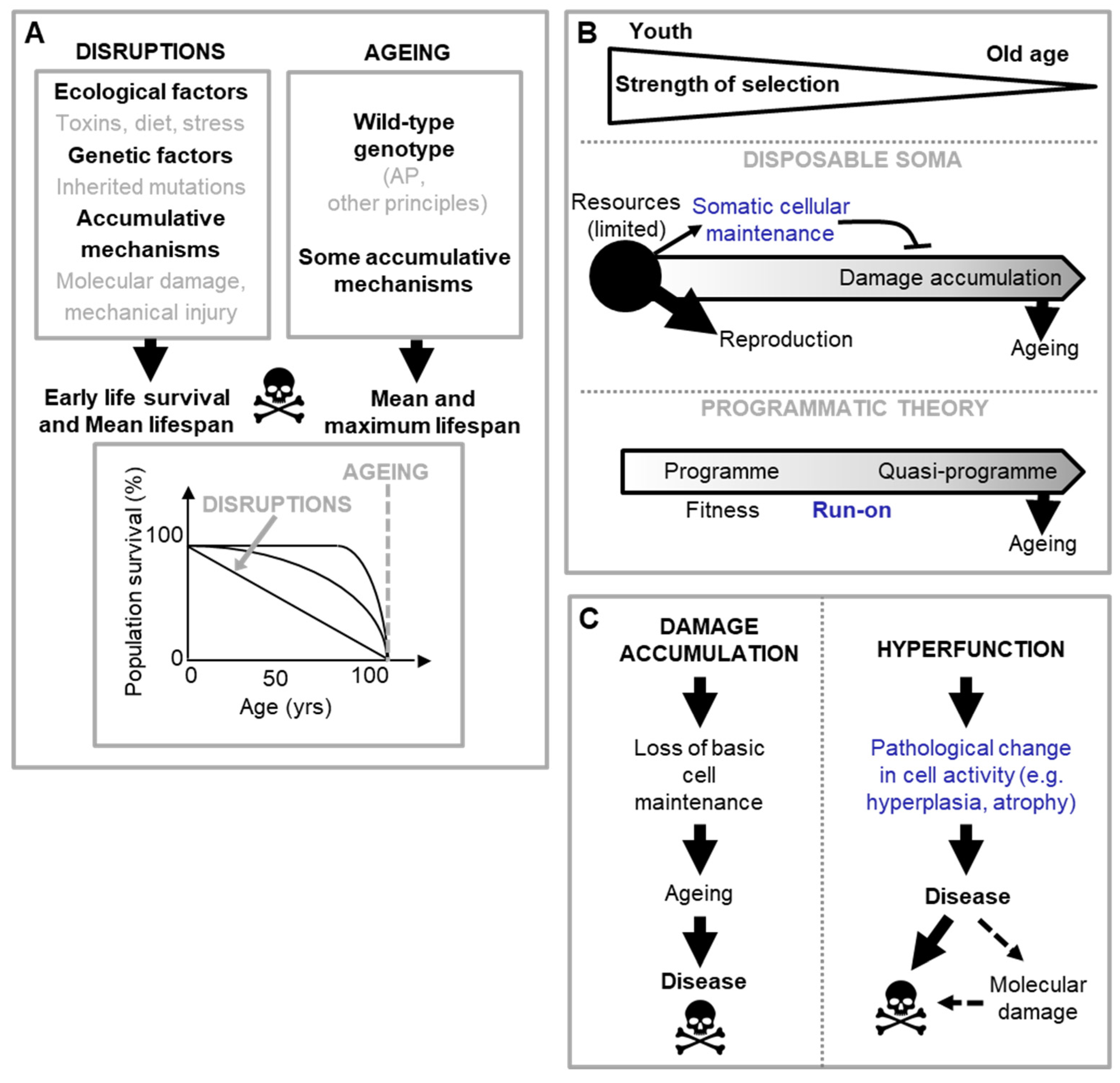
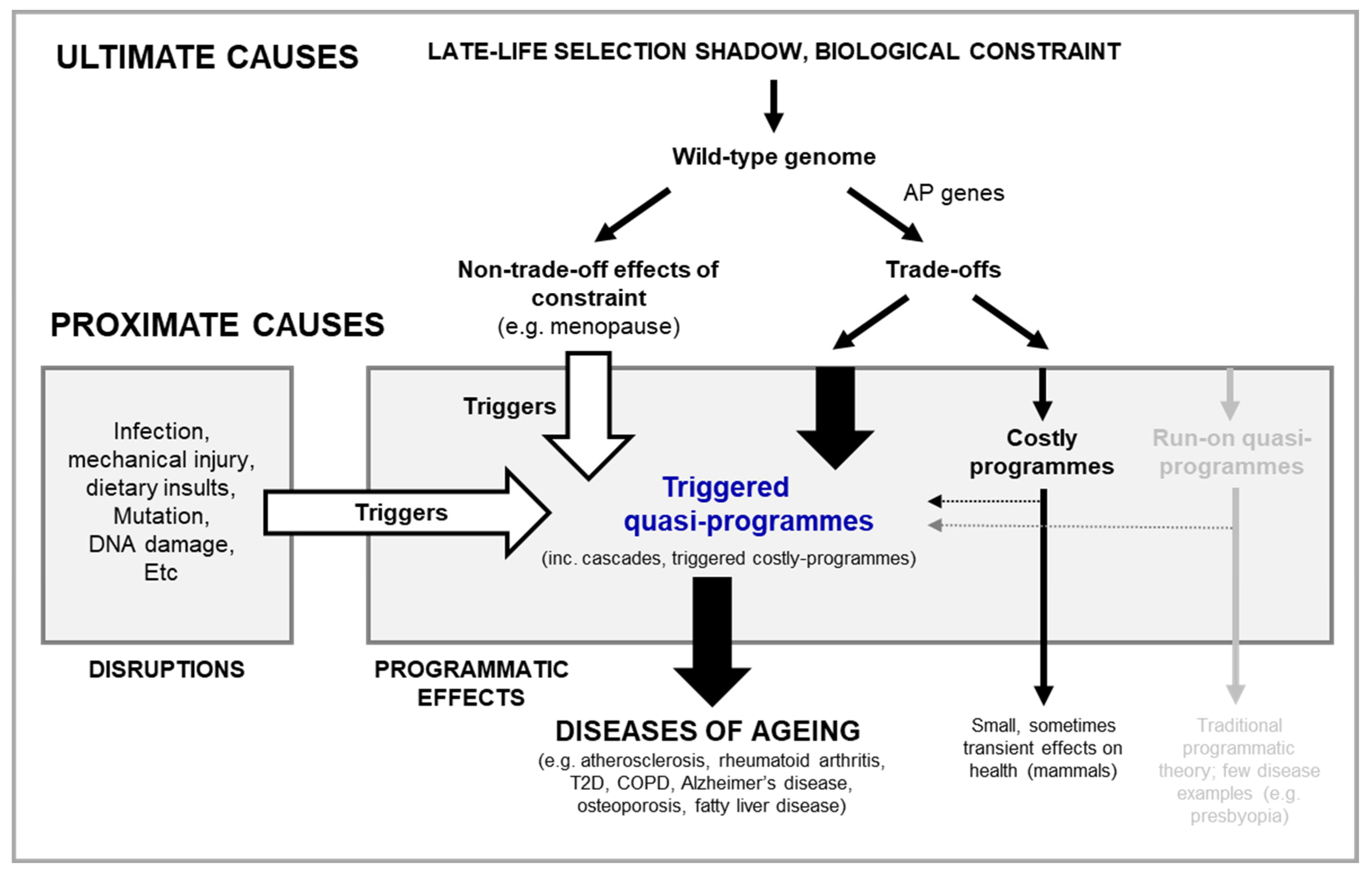
The programmatic theory as proximate mechanism
Limitations of the programmatic theory
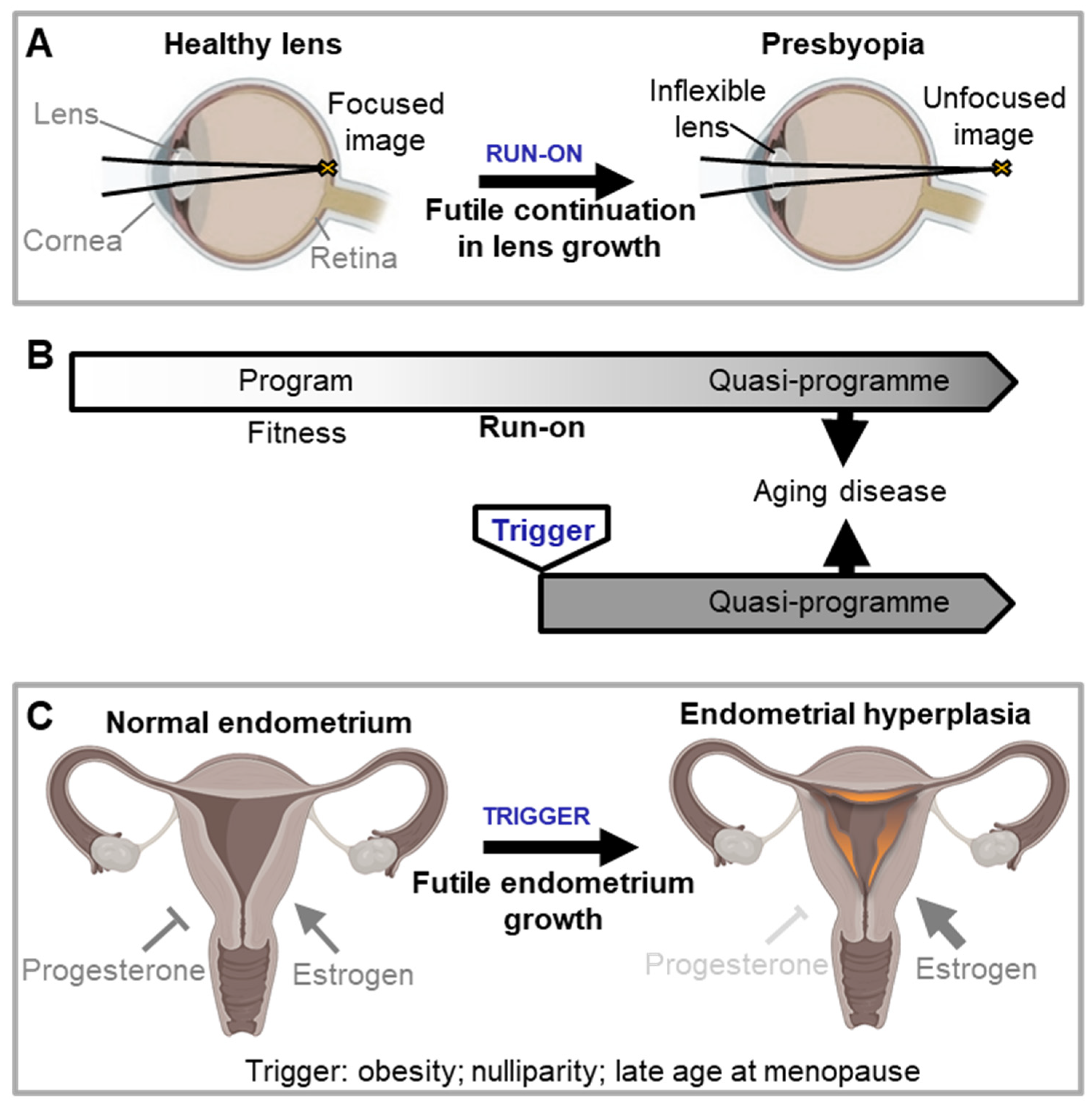
Triggered quasi-programmes
Costly programmes may be triggered later in life as quasi-programmes
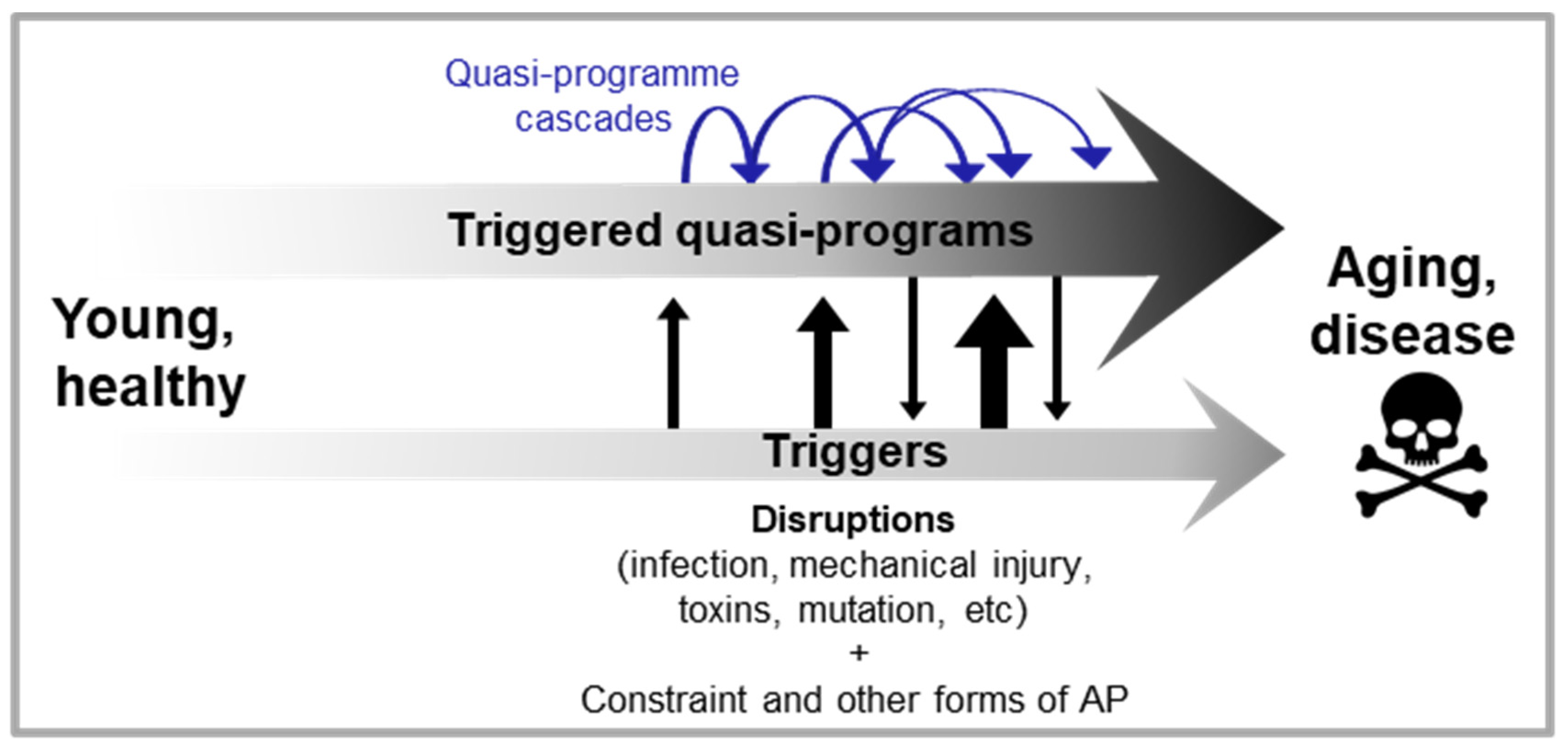
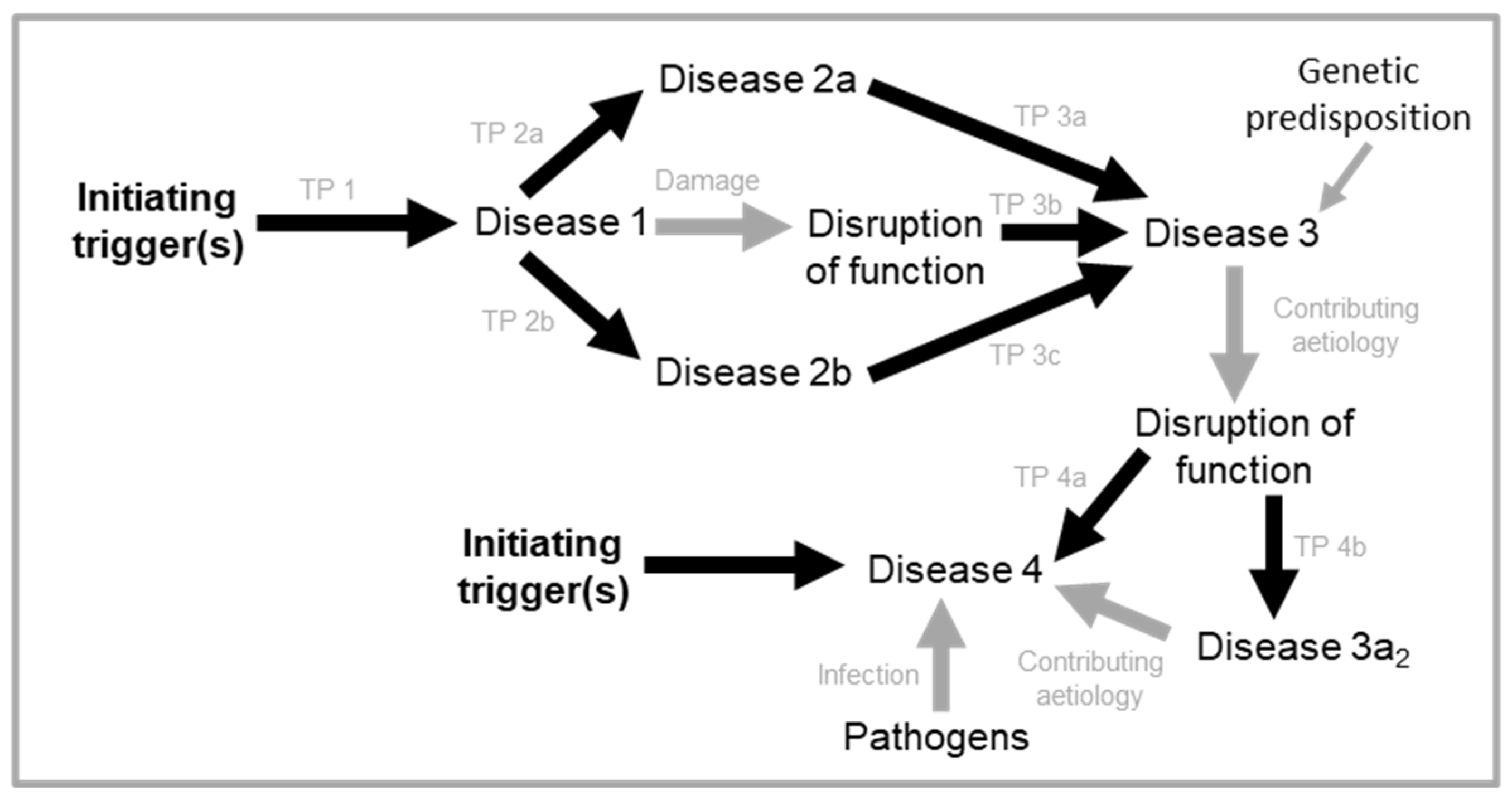
Cascades of triggered quasi-programmes
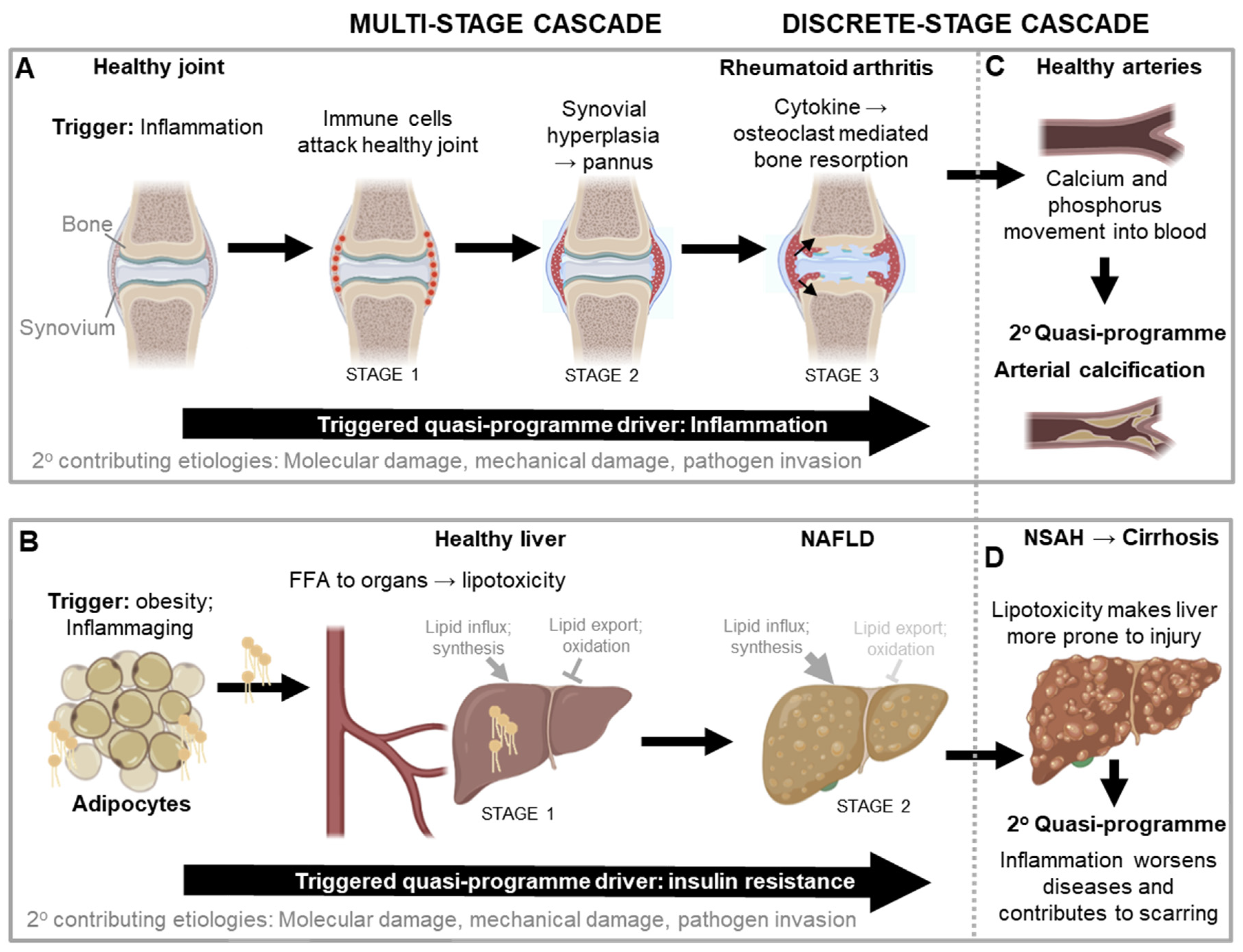
Multi-stage quasi-programme cascades
Discrete stage quasi-programme cascades
Moving away from a mTOR-centric model
Diseases prior to aging: triggered quasi-programmes in infectious disease
AP is a feature of all wild-type genes, and constraint determines risk of disease
Syndromes of senescence: triggered quasi-programmes can act within wider aetiological webs
Triggered quasi-programme cascades are amenable to medical intervention
Rapamycin as an anti-aging drug
Predictions of the new theory
Conclusions
Glossary
| Antagonistic pleiotropy (AP) | Where action of a given gene is both beneficial and detrimental to fitness. If the latter occurs later in life and is therefore subject to weaker selection, such a gene may be favoured by natural selection, and promote aging (Williams, 1957). |
| Biological constraint | A property of organisms and/or their ecology that prevents the evolution of traits that would increase fitness. |
| Damage/maintenance paradigm | Theory that aging is largely caused by accumulation of molecular damage, which can be prevented by somatic maintenance functions. Various theories of aging are based on this broad assumption. |
| Discrete stagequasi-programme cascade (New term) | A series of quasi-programmes triggered in a causal chain where quasi-programme are temporally or spatially separated from one another (c.f. multi-stage quasi-programme cascade). |
| Disposable soma theory | Theory proposing that natural selection favors investment of limited resources into reproduction rather than somatic maintenance, accelerating damage accumulation and, therefore, senescence (Kirkwood, 1977). |
| Hyperfunction | Where wild-type gene function actively leads to senescent pathology, as opposed to passive random damage or wear and tear (Blagosklonny, 2006). |
| Multi-stage quasi-programmecascade (New term) | Where a single quasi-programme driving pathology progression involves a cascade of distinguishable stages (c.f. discrete stage quasi-programme cascade). |
| Programmed aging | Where complex biological processes contributes to senescence, but not necessarily to fitness (cf. quasi-programmes and triggered quasi-programmes). |
| Quasi-programmed aging | Senescence caused by futile gene action. This can arise from a futile run-on of wild-type programmes that promote fitness earlier in life (Blagosklonny, 2006), or (new addition), as argued in this essay, may be triggered by other factors (programmatic or non-programmatic) like infection or injury. Tissue may undergoe changes with age that prime it and make it more susceptible to triggers. |
| Run on | Futile continuation of gene function or processes in later life, leading to pathology (cf. quasi-programme). |
| Signaling constraint (new term) | Where a given signaling molecule (e.g. hormone or growth factor, receptor, signaling kinase, transcription factor) acts in diverse contexts (cell types, tissues, organs), such that optimization of function in all contexts is not possible. |
| Selection shadow | Decrease in selection with increasing age, leading to weaker selection against genes with deleterious effects on fitness and health the later in life those effects are expressed. Environmental factors that increase mortality rate (e.g. predators, infectious pathogens, starvation) can deepen the selection shadow. |
| Senescence | The overall process of deterioration with age or the resulting pathological condition (not to be confused with cellular senescence (sensu Hayflick), which is a particular form of cell cycle arrest affecting some vertebrate cell types). Although aging has several meanings, in the biological context it is usually synonymous with senescence. |
| Quasi-programme cascade (New term) | A causal chain of quasi-programmes, in which each triggers the next in the chain (c.f. multi-stage quasi-programme cascade and discrete quasi-programme cascade). |
| Ultimate-proximate theories of aging | These combine explanations of the evolutionary (ultimate) and mechanistic (proximate) causes of aging into a single integrated account. |
| Wild-type | Genes maintained in the population, i.e. non atypical mutant. |
References
- Abedin, M.; Tintut, Y.; Demer, L.L. Vascular calcification: mechanisms and clinical ramifications. Arterioscler Thromb Vasc Biol. 2004, 24, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A.; Angulo, P.; Lindor, K.D. Nonalcoholic fatty liver disease. Canad. Med. Assoc. J. 2005, 172, 899–905. [Google Scholar] [CrossRef]
- Adinolfi, L.E.; Rinaldi, L.; Guerrera, B.; Restivo, L.; Marrone, A.; Giordano, M.; Zampino, R. NAFLD and NASH in HCV Infection: Prevalence and Significance in Hepatic and Extrahepatic Manifestations. Int. J. Mol. Sci. 2016, 17, 803. [Google Scholar] [CrossRef]
- Admasu, T.D.; Rae, M.J.; Stolzing, A. Dissecting primary and secondary senescence to enable new senotherapeutic strategies. Ageing Res. Rev. 2021, 70, 101412. [Google Scholar] [CrossRef]
- Al-Maskari, A.Y.; Al-Maskari, M.Y.; Al-Sudairy, S. Oral Manifestations and Complications of Diabetes Mellitus: A review. Sultan Qaboos University Medical Journal 2011, 11, 179–186. [Google Scholar]
- Al Anouti, F.; Taha, Z.; Shamim, S.; Khalaf, K.; Al Kaabi, L.; Alsafar, H. An insight into the paradigms of osteoporosis: From genetics to biomechanics. Bone Rep. 2019, 11, 100216. [Google Scholar] [CrossRef]
- Angeles-Albores, D.; Lee, R.Y.N.; Chan, J.; Sternberg, P.W. Tissue enrichment analysis for C. elegans genomics. BMC Bioinform. 2016, 17, 1–10. [Google Scholar] [CrossRef]
- Armstrong, R.A. What causes Alzheimer's disease? Folia Neuropathol. 2013, 51, 169–188. [Google Scholar] [CrossRef]
- Austad, S.; Hoffman, J. Is antagonistic pleiotropy ubiquitous in aging biology? Evol Med Public Health 2018, 2018, 287–294. [Google Scholar] [CrossRef]
- Austad, S.N. Menopause: An evolutionary perspective. Exp. Gerontol. 1994, 29, 255–263. [Google Scholar] [CrossRef]
- Berbudi, A.; Rahmadika, N.; Tjahjadi, A.I.; Ruslami, R. Type 2 Diabetes and its Impact on the Immune System. Curr. Diabetes Rev. 2020, 16, 442–449. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Bhardwaj, S.V. Effect of menopause on women′s periodontium. J. Mid-life Heal. 2012, 3, 5–9. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. Aging and Immortality: Quasi-Programmed Senescence and Its Pharmacologic Inhibition. Cell Cycle 2006, 5, 2087–2102. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. Aging: ROS or TOR. Cell Cycle 2008, 7, 3344–3354. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. Answering the ultimate question "what is the proximal cause of aging?". Aging (Albany NY) 2012, 4, 861–877. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. Rapamycin for longevity: opinion article. Aging 2019, 11, 8048–8067. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. The hyperfunction theory of aging: three common misconceptions. Oncoscience 2021, 8, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Calvo, E.; Castañeda, S.; Largo, R.; Fernández-Valle, M.; Rodríguez-Salvanés, F.; Herrero-Beaumont, G. Osteoporosis increases the severity of cartilage damage in an experimental model of osteoarthritis in rabbits. Osteoarthr. Cartil. 2007, 15, 69–77. [Google Scholar] [CrossRef]
- Chen, N.X.; Moe, S.M. Vascular Calcification: Pathophysiology and Risk Factors. Curr. Hypertens. Rep. 2012, 14, 228–237. [Google Scholar] [CrossRef]
- Chen, Z.; Yu, R.; Xiong, Y.; Du, F.; Zhu, S. A vicious circle between insulin resistance and inflammation in nonalcoholic fatty liver disease. Lipids Health Dis. 2017, 16, 203. [Google Scholar] [CrossRef]
- Chughtai, B.; Forde, J.C.; Thomas, D.D.; Laor, L.; Hossack, T.; Woo, H.H.; Te, A.E.; Kaplan, S.A. Benign prostatic hyperplasia. Nat Rev Dis Primers 2016, 2, 16031. [Google Scholar] [CrossRef]
- Çinar, Y.; Şenyol, A.M.; Duman, K. Blood viscosity and blood pressure: role of temperature and hyperglycemia. Am. J. Hypertens. 2001, 14, 433–438. [Google Scholar] [CrossRef]
- Cooper, G.S.; Bynum, M.L.; Somers, E.C. Recent insights in the epidemiology of autoimmune diseases: Improved prevalence estimates and understanding of clustering of diseases. J. Autoimmun. 2009, 33, 197–207. [Google Scholar] [CrossRef] [PubMed]
- de Magalhaes, J.; Church, G. Genomes optimize reproduction: aging as a consequence of the developmental program. Physiology 2005, 20, 252–259. [Google Scholar] [CrossRef] [PubMed]
- de Magalhães, J.P. Open-minded scepticism: inferring the causal mechanisms of human ageing from genetic perturbations. Ageing Res. Rev. 2005, 4, 1–22. [Google Scholar] [CrossRef]
- de Magalhães, J.P.; Church, G.M. Genomes optimize reproduction: aging as a consequence of the developmental program. Physiology 2005, 20, 252–259. [Google Scholar] [CrossRef]
- Deepa, D.; Jain, G. Assessment of periodontal health status in postmenopausal women visiting dental hospital from in and around Meerut city: Cross-sectional observational study. J. Mid-life Heal. 2016, 7, 175–179. [Google Scholar] [CrossRef]
- Demaria, M.; Ohtani, N.; Youssef, S.A.; Rodier, F.; Toussaint, W.; Mitchell, J.R.; Laberge, R.-M.; Vijg, J.; Van Steeg, H.; Dollé, M.E.; et al. An Essential Role for Senescent Cells in Optimal Wound Healing through Secretion of PDGF-AA. Dev. Cell 2014, 31, 722–733. [Google Scholar] [CrossRef]
- Demer, L.L. Vascular calcification and osteoporosis: inflammatory responses to oxidized lipids. Leuk. Res. 2002, 31, 737–741. [Google Scholar] [CrossRef]
- Dilman, V. Ontogenetic model of ageing and disease formation and mechanisms of natural selection. J. Theor. Biol. 1986, 118, 73–81. [Google Scholar] [CrossRef]
- Dilman, V.M. Development, Aging and Disease: A New Rationale for an Intervention Strategy; Harwood Academic Publishers: 1994.
- Distler, J.H.W.; Wenger, R.H.; Gassmann, M.; Kurowska, M.; Hirth, A.; Gay, S.; Distler, O. Physiologic responses to hypoxia and implications for hypoxia-inducible factors in the pathogenesis of rheumatoid arthritis. Arthritis Rheum. 2004, 50, 10–23. [Google Scholar] [CrossRef]
- Epplein, M.; Reed, S.D.; Voigt, L.F.; Newton, K.M.; Holt, V.L.; Weiss, N.S. Risk of Complex and Atypical Endometrial Hyperplasia in Relation to Anthropometric Measures and Reproductive History. Am. J. Epidemiology 2008, 168, 563–570. [Google Scholar] [CrossRef]
- Feil, S.; Fehrenbacher, B.; Lukowski, R.; Essmann, F.; Schulze-Osthoff, K.; Schaller, M.; Feil, R. Transdifferentiation of Vascular Smooth Muscle Cells to Macrophage-Like Cells During Atherogenesis. Circ. Res. 2014, 115, 662–667. [Google Scholar] [CrossRef]
- Gems, D. Understanding hyperfunction: an emerging paradigm for the biology of aging. Preprints 2021. [Google Scholar] [CrossRef]
- Gems, D. The hyperfunction theory: An emerging paradigm for the biology of aging. Ageing Res. Rev. 2022, 74, 101557–101557. [Google Scholar] [CrossRef]
- Gems, D.; de la Guardia, Y. Alternative perspectives on aging in C. elegans: reactive oxygen species or hyperfunction? Antioxid Redox Signal. 2013, 19, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Gems, D.; Doonan, R. Antioxidant defense and aging in C. elegans: is the oxidative damage theory of aging wrong? Cell Cycle 2009, 8, 1681–1687. [Google Scholar] [CrossRef]
- Gems, D.; Kern, C.C. Biological constraint as a cause of aging. 2022, In preparation.
- Gems, D.; Kern, C.C.; Nour, J.; Ezcurra, M. Semelparity and reproductive death in Caenorhabditis elegans. Preprints 2020, 2020110019. [Google Scholar]
- Glocker, M.O.; Guthke, R.; Kekow, J.; Thiesen, H.-J. Rheumatoid arthritis, a complex multifactorial disease: On the way toward individualized medicine. Med. Res. Rev. 2005, 26, 63–87. [Google Scholar] [CrossRef]
- Glyn-Jones, S.; Palmer, A.J.; Agricola, R.; Price, A.J.; Vincent, T.L.; Weinans, H.; Carr, A.J. Osteoarthritis. Lancet 2015, 386, 376–387. [Google Scholar] [CrossRef] [PubMed]
- Hadi, H.A.R.; Suwaidi, J.A. Endothelial dysfunction in diabetes mellitus. Vasc. Health Risk Manag. 2007, 3, 853–876. [Google Scholar]
- Haldane, J.B.S. New Paths in Genetics; Allen and Unwin: London, 1941. [Google Scholar]
- Hamilton, W. The moulding of senescence by natural selection. J. Theor. Biol. 1966, 12, 12–45. [Google Scholar] [CrossRef] [PubMed]
- Hannemann, M.M.; Alexander, H.M.; Cope, N.J.; Acheson, N. Endometrial hyperplasia: a clinician's review. Obst. Gyn. & Reproduct. Med. 2007, 17, 169–172. [Google Scholar]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; Pahor, M.; Javors, M.A.; Fernandez, E.; Miller, R.A. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Hu, Q.; Xu, X.; Niu, Y.; Chen, Y.; Lu, Y.; Su, Q.; Qin, L. Advanced glycation end products enhance M1 macrophage polarization by activating the MAPK pathway. Biochem. Biophys. Res. Commun. 2020, 525, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Heppner, F.L.; Ransohoff, R.M.; Becher, B. Immune attack: the role of inflammation in Alzheimer disease. Nat. Rev. Neurosci. 2015, 16, 358–372. [Google Scholar] [CrossRef]
- Hibler, E.A.; Kauderer, J.M.; Greene, M.H.; Rodriguez, G.C.; Alberts, D.S. Bone loss after oophorectomy among high-risk women: an NRG oncology/gynecologic oncology group study. Menopause 2016, 23, 1228–1232. [Google Scholar] [CrossRef] [PubMed]
- Higashi, Y.; Gautam, S.; Delafontaine, P.; Sukhanov, S. IGF-1 and cardiovascular disease. Growth Horm. IGF Res. 2019, 45, 6–16. [Google Scholar] [CrossRef]
- Holstein, J.H.; Klein, M.; Garcia, P.; Histing, T.; Culemann, U.; Pizanis, A.; Laschke, M.W.; Scheuer, C.; Meier, C.; Schorr, H.; et al. Rapamycin affects early fracture healing in mice. Br. J. Pharmacol. 2008, 154, 1055–1062. [Google Scholar] [CrossRef]
- Horikawa, A.; Miyakoshi, N.; Shimada, Y.; Kodama, H. The Relationship between Osteoporosis and Osteoarthritis of the Knee: A Report of 2 Cases with Suspected Osteonecrosis. Case Rep. Orthop. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Horvath, S.; Erhart, W.; Brosch, M.; Ammerpohl, O.; von Schoenfels, W.; Ahrens, M.; Heits, N.; Bell, J.T.; Tsai, P.C.; Spector, T.D.; Deloukas, P.; Siebert, R.; Sipos, B.; Becker, T.; Roecken, C.; Schafmayer, C.; Hampe, J. Obesity accelerates epigenetic aging of human liver. Proc Natl Acad Sci U S A 2014, 111, 15538–15543. [Google Scholar] [CrossRef] [PubMed]
- Huertas, A.; Palange, P. COPD: a multifactorial systemic disease. Ther. Adv. Respir. Dis. 2011, 5, 217–224. [Google Scholar] [CrossRef]
- Hughes, K.A. Mutation and the evolution of ageing: from biometrics to system genetics. Philos. Trans. R. Soc. B: Biol. Sci. 2010, 365, 1273–1279. [Google Scholar] [CrossRef]
- Ising, C.; Venegas, C.; Zhang, S.; Scheiblich, H.; Schmidt, S.V.; Vieira-Saecker, A.; Schwartz, S.; Albasset, S.; McManus, R.M.; Tejera, D.; et al. NLRP3 inflammasome activation drives tau pathology. Nature 2019, 575, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Iwabuchi, H.; Fujibayashi, T.; Yamane, G.-Y.; Imai, H.; Nakao, H. Relationship between Hyposalivation and Acute Respiratory Infection in Dental Outpatients. Gerontology 2012, 58, 205–211. [Google Scholar] [CrossRef]
- Karmakar, S.; Kay, J.; Gravallese, E.M. Bone Damage in Rheumatoid Arthritis: Mechanistic Insights and Approaches to Prevention. Rheum. Dis. Clin. North Am. 2010, 36, 385–404. [Google Scholar] [CrossRef]
- Kern, C.; Gems, D. How biological mechanisms shape population dynamics and lifespan through ageing. 2023a, In preparation.
- Kern, C.C. Ageing through reproductive death in Caenorhabditis elegans. (UCL) University College London, 2020.
- Kern, C.C.; Gems, D. Semelparous Death as one Element of Iteroparous Aging Gone Large. Front. Genet. 2022, 13, 880343. [Google Scholar] [CrossRef]
- Kern, C.C.; Gems, D. Unifying theory: Life history strategy, trade-offs and constraints shape ageing mechanisms and pathology. 2023b, In preparation.
- Kern, C.C.; Morley, H.; Gems, D. Lactation reactivation syndrome. 2023a, In preparation.
- Kern, C.C.; Srivastava, S.; Ezcurra, M.; Hsiung, K.C.; Hui, N.; Townsend, S.; Maczik, D.; Zhang, B.; Tse, V.; Konstantellos, V.; et al. elegans ageing is accelerated by a self-destructive reproductive programme. Nat. Commun. 2023, 14, 1–12. [Google Scholar] [CrossRef]
- Kern, C.C.; Townsend, S.; Salzmann, A.; Rendell, N.B.; Taylor, G.W.; Comisel, R.M.; Foukas, L.C.; Bähler, J.; Gems, D. C. C. elegans feed yolk to their young in a form of primitive lactation. Nat. Commun. 2021, 12, 1–11. [Google Scholar] [CrossRef]
- Kirkwood, T.B.L. Understanding the Odd Science of Aging. Cell 2005, 120, 437–447. [Google Scholar] [CrossRef]
- Kirkwood, T.B.L. Evolution of ageing. Nature 1977, 270, 301–304. [Google Scholar] [CrossRef]
- Kovacs, C.S. Maternal Mineral and Bone Metabolism During Pregnancy, Lactation, and Post-Weaning Recovery. Physiol. Rev. 2016, 96, 449–547. [Google Scholar] [CrossRef]
- Liu, H.-F.; Zhang, H.-J.; Hu, Q.-X.; Liu, X.-Y.; Wang, Z.-Q.; Fan, J.-Y.; Zhan, M.; Chen, F.-L. Altered Polarization, Morphology, and Impaired Innate Immunity Germane to Resident Peritoneal Macrophages in Mice with Long-Term Type 2 Diabetes. J. Biomed. Biotechnol. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Madersbacher, S.; Sampson, N.; Culig, Z. Pathophysiology of Benign Prostatic Hyperplasia and Benign Prostatic Enlargement: A Mini-Review. Gerontology 2019, 65, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Maklakov, A.A.; Chapman, T. Evolution of ageing as a tangle of trade-offs: energy versus function. Proc. R. Soc. B: Biol. Sci. 2019, 286, 20191604. [Google Scholar] [CrossRef]
- Mancuso, P.; Bouchard, B. The impact of aging on adipose function and adipokine synthesis. Front. Endocrinol. 2019, 10, 137–137. [Google Scholar] [CrossRef] [PubMed]
- Marlowe, F. The patriarch hypothesis: An alternative explanation of menopause. Human Nature 2000, 11, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.M. Genetic syndromes in man with potential relevance to the pathobiology of aging. Birth Defects 1978, 14, 5–39. [Google Scholar]
- Mauvais-Jarvis, F.; Manson, J.E.; Stevenson, J.C.; Fonseca, V.A. Menopausal Hormone Therapy and Type 2 Diabetes Prevention: Evidence, Mechanisms, and Clinical Implications. Endocr. Rev. 2017, 38, 173–188. [Google Scholar] [CrossRef]
- McNulty, P.; Pilcher, R.; Ramesh, R.; Necuiniate, R.; Hughes, A.; Farewell, D.; Holmans, P.; Jones, L.; REGISTRY Investigators of the European Huntington's Disease Network. Reduced cancer incidence in Huntington's disease: analysis in the registry study. J. Huntingtons Dis. 2018, 7, 209–222. [Google Scholar]
- Medawar, P.B. An Unsolved Problem Of Biology, H.K. Lewis, London, 1952.
- More, C.; Bettembuk, P.; Bhattoa, H.P.; Balogh, A. The Effects of Pregnancy and Lactation on Bone Mineral Density. Osteoporos. Int. 2001, 12, 732–737. [Google Scholar] [CrossRef]
- Nacusi, L.P.; Tindall, D.J. Targeting 5α-reductase for prostate cancer prevention and treatment. Nat. Rev. Urol. 2011, 8, 378–384. [Google Scholar] [CrossRef]
- NCI, 2023. Surveillance, Epidemiology, and End Results Program http://seer.cancer.gov/.
- Nussey, D.H.; Froy, H.; Lemaitre, J.F.; Gaillard, J.M.; Austad, S.N. Senescence in natural populations of animals: widespread evidence and its implications for bio-gerontology. Ageing Res Rev. 2013, 12, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Paschou, S.A.; Papanas, N. Type 2 diabetes mellitus and menopausal hormone therapy: an update. Diabetes Ther. 2019, 10, 2313–2320. [Google Scholar] [CrossRef] [PubMed]
- Pavlou, S.; Lindsay, J.; Ingram, R.; Xu, H.; Chen, M. Sustained high glucose exposure sensitizes macrophage responses to cytokine stimuli but reduces their phagocytic activity. BMC Immunol. 2018, 19, 24. [Google Scholar] [CrossRef]
- Pérez-Ros, P.; Navarro-Flores, E.; Julián-Rochina, I.; Martínez-Arnau, F.M.; Cauli, O. Changes in Salivary Amylase and Glucose in Diabetes: A Scoping Review. Diagnostics (Basel) 2021, 11. [Google Scholar] [CrossRef]
- Pollreisz, A.; Schmidt-Erfurth, U. Diabetic cataract-pathogenesis, epidemiology and treatment. J Ophthalmol. 2010, 2010, 608751. [Google Scholar] [CrossRef]
- Prata, L.G.P.L.; Ovsyannikova, I.G.; Tchkonia, T.; Kirkland, J.L. Senescent cell clearance by the immune system: Emerging therapeutic opportunities. Seminars in immunology 2018, 40, 101275–101275. [Google Scholar] [CrossRef]
- Robert, G.; Descazeaud, A.; Nicolaïew, N.; Terry, S.; Sirab, N.; Vacherot, F.; Maillé, P.; Allory, Y.; de la Taille, A. Inflammation in benign prostatic hyperplasia: a 282 patients' immunohistochemical analysis. Prostate 2009, 69, 1774–1780. [Google Scholar] [CrossRef]
- Robertson, O.H. Prolongation of the life span of kokanee salmon (Oncorhynchus nerka kennerlyi) by castration before the beginning of gonad development. Proc. Natl. Acad. Sci. USA 1961, 47, 609–621. [Google Scholar] [CrossRef]
- Rong, J.X.; Shapiro, M.; Trogan, E.; Fisher, E.A. Transdifferentiation of mouse aortic smooth muscle cells to a macrophage-like state after cholesterol loading. Proc Natl Acad Sci U S A 2003, 100, 13531–13536. [Google Scholar] [CrossRef]
- Rose, M.; Charlesworth, B. A test of evolutionary theories of senescence. Nature 1980, 287, 141–142. [Google Scholar] [CrossRef] [PubMed]
- Sage, A.P.; Tintut, Y.; Demer, L.L. Regulatory mechanisms in vascular calcification. Nat Rev Cardiol. 2010, 7, 528–536. [Google Scholar] [CrossRef]
- Sala Frigerio, C.; Wolfs, L.; Fattorelli, N.; Thrupp, N.; Voytyuk, I.; Schmidt, I.; Mancuso, R.; Chen, W.; Woodbury, M.; Srivastava, G. The major risk factors for Alzheimer’s disease: age, sex, and genes modulate the microglia response to Aβ plaques. Cell Rep 2019, 27, 1293–1306. [Google Scholar] [CrossRef] [PubMed]
- Salvestrini, V.; Sell, C.; Lorenzini, A. Obesity may accelerate the aging process. Front. Endocrinol. 2019, 10, 266. [Google Scholar] [CrossRef] [PubMed]
- Sapey, E.; Greenwood, H.; Walton, G.; Mann, E.; Love, A.; Aaronson, N.; Insall, R.H.; Stockley, R.A.; Lord, J.M. Phosphoinositide 3-kinase inhibition restores neutrophil accuracy in the elderly: toward targeted treatments for immunosenescence. Blood 2014, 123, 239–248. [Google Scholar] [CrossRef]
- Sears, B.; Perry, M. The role of fatty acids in insulin resistance. Lipids Health Dis. 2015, 14, 121–121. [Google Scholar] [CrossRef]
- Shields, H.J.; Traa, A.; Van Raamsdonk, J.M. Beneficial and detrimental effects of reactive oxygen species on lifespan: a comprehensive review of comparative and experimental studies. Front. Cell Dev. Biol. 2021, 9, 628157. [Google Scholar] [CrossRef]
- Shokeir, M. Investigation on Huntington’s disease in the Canadian Prairies. II. Fecundity and fitness. Clin. Genet. 1975, 7, 349–353. [Google Scholar] [CrossRef]
- Sohal, R.S.; Weindruch, R. Oxidative stress, caloric restriction, and aging. Science 1996, 273, 59–63. [Google Scholar] [CrossRef]
- Sorenson, S.A.; Fenger, K.; Olsen, J. Significantly lower incidence of cancer among patients with Huntington’s disease. Cancer 1999, 6, 1342–1346. [Google Scholar] [CrossRef]
- Strenk, S.A.; Strenk, L.M.; Koretz, J.F. The mechanism of presbyopia. Prog. Retin. Eye Res. 2005, 24, 379–393. [Google Scholar] [CrossRef]
- Unger, R.H. Minireview: weapons of lean body mass destruction: the role of ectopic lipids in the metabolic syndrome. Endocrinology 2003, 144, 5159–5165. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, G.; Madersbacher, S.; Berger, P. Benign prostatic hyperplasia: age-related tissue-remodeling. Exp Gerontol. 2005, 40, 121–128. [Google Scholar] [CrossRef] [PubMed]
- van Deursen, J.M. The role of senescent cells in ageing. Nature 2014, 509, 439–446. [Google Scholar] [CrossRef]
- Virchow, R. Cellular Pathology, as Based upon Physiological and Pathological Histology; Dover Publications, Inc: New York, NY, 1863. [Google Scholar]
- Voynow, J.A.; Shinbashi, M. Neutrophil Elastase and Chronic Lung Disease. Biomolecules 2021, 11. [Google Scholar] [CrossRef]
- Walker, D.A.; Harper, P.S.; Newcombe, R.G.; Davies, K. Huntington's chorea in South Wales: mutation, fertility, and genetic fitness. J. Med. Genet. 1983, 20, 12–17. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, Y.; Zhou, J. Neuroinflammation in Parkinson's disease and its potential as therapeutic target. Transl Neurodegener. 2015, 4, 19. [Google Scholar] [CrossRef]
- Waters, D.J.; Shen, S.; Glickman, L.T. Life expectancy, antagonistic pleiotropy, and the testis of dogs and men. The Prostate 2000, 43, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Whiteside, S.K.; Snook, J.P.; Williams, M.A.; Weis, J.J. Bystander T Cells: A Balancing Act of Friends and Foes. Trends Immunol. 2018, 39, 1021–1035. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.E.; Burmeister, L.; Brooks, S.V.; Chan, C.C.; Friedline, S.; Harrison, D.E.; Hejtmancik, J.F.; Nadon, N.; Strong, R.; Wood, L.K.; Woodward, M.A.; Miller, R.A. Rapamycin slows aging in mice. Aging Cell. 2012, 11, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.C. Pleiotropy, natural selection and the evolution of senescence. Evolution 1957, 11, 398–411. [Google Scholar] [CrossRef]
- Xie, J.; Van Hoecke, L.; Vandenbroucke, R.E. The Impact of Systemic Inflammation on Alzheimer's Disease Pathology. Front Immunol. 2021, 12, 796867. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



