Submitted:
20 October 2023
Posted:
23 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Bioavailability, Biotransformation and Stability of GPS
4. Pharmacological Properties of GPS In Vitro
4.1. Anti-Inflammatory Activity of GPS and Its Semi-Synthetic Analogues
4.2. Wound Healing Effect of GPS
4.3. Anti-Rheumatic Activity of GPS
4.4. Promotion of the Osteogenic Effect by GPS in Bone Mesenchymal Stem Cells
4.5. Neurogenic and Neuroprotective Activitity of GPS
4.6. Antiaging Properties of GPS
4.7. Anti-Diabetic, Hypoglycemic and Hypolipidemic Effect of GPS
4.8. Hepatoprotective Effect of GPS
4.9. Miorelaxant Activity of GPS
4.10. Antibacterial Potential of GPS
4.10.1. Antibacterial Activity of Nanoparticles with GPS
4.11. In Vitro Antiviral Activity of Semi-Synthetic GPS Derivatives
4.12. Oxidative Stress Reducing Potential of GPS
4.13. Anti-Cancer Properties
4.13.1. Ovarian Cancer
4.14.2. Cervical Cancer
4.14.3. Gastric Cancer
4.14.4. Hepatocellular Carcinoma
5.1. Possible Long-Term Adverse Effects of GPS as a Single Compound
6. Pharmacological Properties of Gentiopicroside In Vivo
6. Pharmacological Properties of Gentiopicroside, Clinical Studies
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Raka, F.; Farr, S.; Kelly, J.; Stoianov, A.; Adeli, K. Metabolic control via nutrient-sensing mechanisms: role of taste receptors and the gut-brain neuroendocrine axis. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E559–E572. [Google Scholar] [CrossRef]
- Lu, P.; Zhang, C.-H.; Lifshitz, L.M.; ZhuGe, R. Extraoral Bitter Taste Receptors in Health and Disease. J. Gen. Physiol. 2017, 149, 181–197. [Google Scholar] [CrossRef]
- Harmon, C.P.; Deng, D.; Breslin, P.A.S. Bitter Taste Receptors (T2Rs) are Sentinels that Coordinate Metabolic and Immunological Defense Responses. Curr. Opin. Physiol. 2021, 20, 70–76. [Google Scholar] [CrossRef]
- Pallante, L.; Malavolta, M.; Grasso, G.; Korfiati, A.; Mavroudi, S.; Mavkov, B.; Kalogeras, A.; Alexakos, C.; Martos, V.; Amoroso, D.; Di Benedetto, G.; Piga, D.; Theofilatos, K.; Deriu, M.A. On the human taste perception: Molecular-level understanding empowered by computational methods. Trends. Food. Sci. Tech. 2021, 116, 445–459. [Google Scholar] [CrossRef]
- Jensen, S. R., & Schripsema, J. (2002). Chemotaxonomy and pharmacology of Gentianaceae. In L. Struwe, & V. Albert (Eds.), Gentianaceae - Systematics and Natural History (Vol. Chapter 6, pp. 573-631). Cambridge University Press.
- 5Changzeng, W.; Dequan, Y. Diterpenoid, Sesquiterpenoid and Secoiridoid Glucosides from Aster Auriculatus. Phytochemistry 1997, 45, 1483–1487. [Google Scholar] [CrossRef]
- 6Daud, M.N.H.; Wibowo, A.; Abdullah, N.; Ahmad, R. Bioassay-Guided Fractionation of Artocarpus Heterophyllus L. J33 Variety Fruit Waste Extract and Identification of Its Antioxidant Constituents by TOF-LCMS. Food Chem. 2018, 266, 200–214. [Google Scholar] [CrossRef]
- Mustafayeva, K.; Di Giorgio, C.; Elias, R.; Kerimov, Y.; Ollivier, E.; De Méo, M. DNA-Damaging, Mutagenic, and Clastogenic Activities of Gentiopicroside Isolated from Cephalaria Kotschyi Roots. J. Nat. Prod. 2010, 73, 99–103. [Google Scholar] [CrossRef] [PubMed]
- 3Jiang, M.; Cui, B.-W.; Wu, Y.-L.; Nan, J.-X.; Lian, L.-H. Genus Gentiana: A Review on Phytochemistry, Pharmacology and Molecular Mechanism. J. Ethnopharmacol. 2021, 264, 113391. [Google Scholar] [CrossRef]
- European Pharmacopoeia, 10th Edition.
- Zeng, W.; Han, H.; Tao, Y.; Yang, L.; Wang, Z.; Chen, K. Identification of Bio-Active Metabolites of Gentiopicroside by UPLC/Q-TOF MS and NMR. Biomed. Chromatogr. 2013, 27, 1129–1136. [Google Scholar] [CrossRef]
- el-Sedawy, A.I.; Hattori, M.; Kobashi, K.; Namba, T. Metabolism of Gentiopicroside (Gentiopicrin) by Human Intestinal Bacteria. Chem. Pharm. Bull. (Tokyo). 1989, 37, 2435–2437. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Xiao, H.-Q.; Li, Y.; Chen, Y.; Chen, S.-Y.; Li, Y.-T.; Huang, Y.; Zheng, L.; Wang, Y.-L.; Gong, Z.-P. Uptake and Transport of Laportea Bulbifera Extract in Caco-2 Cell Model. Zhongguo Zhong Yao Za Zhi 2022, 47, 5617–5626. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Gao, D.; Zhang, W.; Wang, J. Stability of Gentiopicrin in Biological Fluids in Vitro. Fudan Xuebao Yixueban 2008, 35, 220–223. [Google Scholar]
- Zhang, Q.; Zhang, J.; Xia, P.; Peng, X.; Li, H.; Jin, H.; Li, Y.; Yang, J.; Zhao, L. Anti-Inflammatory Activities of Gentiopicroside against INOS and COX-2 Targets. Chinese Herb. Med. 2019, 11, 108–112. [Google Scholar] [CrossRef]
- De Vita, S.; Chini, M.G.; Saviano, G.; Finamore, C.; Festa, C.; Lauro, G.; De Marino, S.; Russo, R.; Avagliano, C.; Casapullo, A.; et al. Metabolites Using Computational Approaches and in Vitro Tests. Biomolecules 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Tian, Y.; Zhou, D.; Yang, L.; Liu, T.-M.; Liu, Z.-G.; Wang, S.-W. Gentiopicroside Ameliorates Ethanol-Induced Gastritis via Regulating MMP-10 and PERK1/2 Signaling. Int. Immunopharmacol. 2021, 90, 107213. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, X.; Yang, L.; Luo, M.; Han, L.; Lu, Y.; Shi, Q.; Wang, Y.; Liang, Q. Gentiopicroside (GENT) Protects against Sepsis Induced by Lipopolysaccharide (LPS) through the NF-ΚB Signaling Pathway. Ann. Transl. Med. 2019, 7, 731. [Google Scholar] [CrossRef]
- Zhang, Q.-L.; Xia, P.-F.; Peng, X.-J.; Wu, X.-Y.; Jin, H.; Zhang, J.; Zhao, L. Synthesis, and Anti-Inflammatory Activities of Gentiopicroside Derivatives. Chin. J. Nat. Med. 2022, 20, 309–320. [Google Scholar] [CrossRef]
- Öztürk, N.; Korkmaz, S.; Öztürk, Y.; Başer, K.H.C. Effects of Gentiopicroside, Sweroside and Swertiamarine, Secoiridoids from Gentian (Gentiana Lutea Ssp. Symphyandra), on Cultured Chicken Embryonic Fibroblasts. Planta Med. 2006, 72, 289–294. [Google Scholar] [CrossRef]
- Zhang, N.; Jiang, Y.; Mu, F.; Wu, H.; You, Q. Gentiopicrin Exerts Anti-Rheumatic Effect in Human Fibroblast-like Synoviocytes via Inhibition of P38MAPK/NF-ΚB Pathway. Cell. Mol. Biol. (Noisy-le-grand). 2019, 65, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, H.; Wang, Y.; Hao, Y.; Huang, Y.; Wang, X.; Lu, Y.; Du, Y.; Fu, F.; Xin, W.; et al. Anti-Rheumatic Properties of Gentiopicroside Are Associated With Suppression of ROS-NF-ΚB-NLRP3 Axis in Fibroblast-Like Synoviocytes and NF-ΚB Pathway in Adjuvant-Induced Arthritis. Front. Pharmacol. 2020, 11, 515. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhong, J.; Li, W.; Dong, J.; Xian, C.J.; Shen, Y.-K.; Yao, L.; Wu, Q.; Wang, L. Gentiopicroside Promotes the Osteogenesis of Bone Mesenchymal Stem Cells by Modulation of β-Catenin-BMP2 Signalling Pathway. J. Cell. Mol. Med. 2021, 25, 10825–10836. [Google Scholar] [CrossRef] [PubMed]
- Chiba, K.; Yamazaki, M.; Kikuchi, M.; Kakuda, R.; Kikuchi, M. New Physiological Function of Secoiridoids: Neuritogenic Activity in PC12h Cells. J. Nat. Med. 2011, 65, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-Z.; Lei, Y.-J.; Jiang, H.-Y.; Wang, F.-Q. Effects of Gentiopicroside on Caspase-3, Bax and Bcl-2 in Neonatal Rat Hippocampal Neurons Following Oxygen-Glucose Deprivation and Reperfusion Injury. Jiepou Xuebao 2016, 47, 28–33. [Google Scholar]
- Liu, Q.; Cheng, L.; Matsuura, A.; Xiang, L.; Qi, J. Gentiopicroside, a Secoiridoid Glycoside from Gentiana Rigescens Franch, Extends the Lifespan of Yeast via Inducing Mitophagy and Antioxidative Stress. Oxid. Med. Cell. Longev. 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-Q.; Chen, Y.-D.; Li, H.; Hui, X.; Gao, W.-Y. Geniposide and Gentiopicroside Suppress Hepatic Gluconeogenesis via Regulation of AKT-FOXO1 Pathway. Arch. Med. Res. 2018, 49, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Lin, Z.; Zeng, J.; Chen, R.; Li, C.; Xiao, H.; Huang, H.; Xu, S.; Lan, T. Gentiopicroside Ameliorates Glucose and Lipid Metabolism in T2DM by Activating PI3K/AKT Pathway Via FGFR1. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Xiao, H.; Sun, X.; Lin, Z.; Yang, Y.; Zhang, M.; Xu, Z.; Liu, P.; Liu, Z.; Huang, H. Gentiopicroside Targets PAQR3 to Activate the PI3K/AKT Signaling Pathway and Ameliorate Disordered Glucose and Lipid Metabolism. Acta Pharm. Sin. B 2022, 12, 2887–2904. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhang, M.; Wang, Y.; Chen, R.; Xu, S.; Sun, X.; Yang, Y.; Lin, Z.; Wang, S.; Huang, H. Gentiopicroside Ameliorates Diabetic Renal Tubulointerstitial Fibrosis via Inhibiting the AT1R/CK2/NF-ΚB Pathway. Front. Pharmacol. 2022, 13, 848915. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, L.; Yang, Y.; Sun, W.; Xie, R.; Liu, X.; Wang, Q. In Vitro Inhibition and Induction of Human Liver Cytochrome P450 Enzymes by Gentiopicroside: Potent Effect on CYP2A6. Drug Metab. Pharmacokinet. 2013, 28, 339–344. [Google Scholar] [CrossRef]
- Yang, H.-X.; Shang, Y.; Jin, Q.; Wu, Y.-L.; Liu, J.; Qiao, C.-Y.; Zhan, Z.-Y.; Ye, H.; Nan, J.-X.; Lian, L.-H. Gentiopicroside Ameliorates the Progression from Hepatic Steatosis to Fibrosis Induced by Chronic Alcohol Intake. Biomol. Ther. (Seoul). 2020, 28, 320–327. [Google Scholar] [CrossRef]
- Rojas, A.; Bah, M.; Rojas, J.I.; Gutiérrez, D.M. Smooth Muscle Relaxing Activity of Gentiopicroside Isolated from Gentiana Spathacea. Planta Med. 2000, 66, 765–767. [Google Scholar] [CrossRef]
- Kumarasamy, Y.; Nahar, L.; Sarker, S.D. Bioactivity of Gentiopicroside from the Aerial Parts of Centaurium Erythraea. Fitoterapia 2003, 74, 151–154. [Google Scholar] [CrossRef]
- Almukainzi, M.; El-Masry, A. T.; . Negm, A. W.; Elekhnawy, E.; Saleh, A.; Sayed, E. A.; Khattab, A.M.; Abdelkader, H.D. Gentiopicroside PLGA Nanospheres: fabrication, in vitro characterization, antimicrobial action, and in vivo effect for enhancing wound healing in diabetic rats. Int. J. Nanomed. 2022, 17, 1203–1225. [Google Scholar] [CrossRef]
- Wu, S.; Yang, L.; Sun, W.; Si, L.; Xiao, S.; Wang, Q.; Dechoux, L.; Thorimbert, S.; Sollogoub, M.; Zhou, D.; et al. Design, Synthesis and Biological Evaluation of Gentiopicroside Derivatives as Potential Antiviral Inhibitors. Eur. J. Med. Chem. 2017, 130, 308–319. [Google Scholar] [CrossRef]
- Jin, M.; Feng, H.; Wang, Y.; Yan, S.; Shen, B.; Li, Z.; Qin, H.; Wang, Q.; Li, J.; Liu, G. Gentiopicroside Ameliorates Oxidative Stress and Lipid Accumulation through Nuclear Factor Erythroid 2-Related Factor 2 Activation. Oxid. Med. Cell. Longev. 2020, 2020, 2940746. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Kossaï, M.; Leary, A.; Scoazec, J.-Y.; Genestie, C. Ovarian Cancer: A Heterogeneous Disease. Pathobiology 2018, 85, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.M.; Jordan, S.J. Epidemiology of Epithelial Ovarian Cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 41, 3–14. [Google Scholar] [CrossRef]
- Li, X.; Yang, C.; Shen, H. Gentiopicroside Exerts Convincing Antitumor Effects in Human Ovarian Carcinoma Cells (SKOV3) by Inducing Cell Cycle Arrest, Mitochondrial Mediated Apoptosis and Inhibition of Cell Migration. J. BUON. 2019, 24, 280–284. [Google Scholar]
- Tian, H.; Liu, L.; Yang, T.; Wang, Y. Gentiopicroside Inhibits Cancer Cell Growth in OVCAR-3 Ovary Cancer Cells through the Mediation of Apoptosis , Loss of Mitochondrial Transmembrane Potential and NF-KB Signalling Pathway. Biomedical Research 2016, 27, 413–418. [Google Scholar]
- Tsikouras, P.; Zervoudis, S.; Manav, B.; Tomara, E.; Iatrakis, G.; Romanidis, C.; Bothou, A.; Galazios, G. Cervical Cancer: Screening, Diagnosis and Staging. J. BUON. 2016, 21, 320–325. [Google Scholar] [PubMed]
- Hu, Z.; Wang, H.; Fu, Y.; Ma, K.; Ma, X.; Wang, J. Gentiopicroside Inhibits Cell Growth and Migration on Cervical Cancer via the Reciprocal MAPK/Akt Signaling Pathways. Nutr. Cancer 2021, 73, 1459–1470. [Google Scholar] [CrossRef]
- Song, Z.; Wu, Y.; Yang, J.; Yang, D.; Fang, X. Progress in the Treatment of Advanced Gastric Cancer. Tumour Biol. J. Int. Soc. Oncodevelopmental Biol. Med. 2017, 39, 1010428317714626. [Google Scholar] [CrossRef] [PubMed]
- Johnston, F.M.; Beckman, M. Updates on Management of Gastric Cancer. Curr. Oncol. Rep. 2019, 21, 67. [Google Scholar] [CrossRef]
- Huang, Y.; Lin, J.; Yi, W.; Liu, Q.; Cao, L.; Yan, Y.; Fu, A.; Huang, T.; Lyu, Y.; Huang, Q.; et al. Research on the Potential Mechanism of Gentiopicroside Against Gastric Cancer Based on Network Pharmacology. Drug Des. Devel. Ther. 2020, 14, 5109–5118. [Google Scholar] [CrossRef]
- Di Bisceglie, A.M.; Rustgi, V.K.; Hoofnagle, J.H.; Dusheiko, G.M.; Lotze, M.T. NIH Conference. Hepatocellular Carcinoma. Ann. Intern. Med. 1988, 108, 390–401. [Google Scholar] [CrossRef]
- Huang, X.; Luo, M.; Qi, H.; Wang, Z. Proliferation Effects of Gentiopicroside and Other Five Traditional Chinese Medicines on SMMC-7721 Human Hepatocarcinoma Cell Line.
- Antoniadi, L.; Wawruszak, A.; Angelis, A.; Czerwonka, A.; Jarzáb, A.; Halabalaki, M.; Kukula-Koch, W.; Skaltsounis, L.A. High-throughput isolation of gentiopicroside from Gentiana lutea and the study on its anticancer properties in vitro. Ind. Crops Prod. 2023, 195, 116448. [Google Scholar] [CrossRef]
- Li, X.; Yang, C.; Shen, H. Gentiopicroside Exerts Convincing Antitumor Effects in Human Ovarian Carcinoma Cells (SKOV3) by Inducing Cell Cycle Arrest, Mitochondrial Mediated Apoptosis and Inhibition of Cell Migration. J. BUON. 2019, 24, 280–284. [Google Scholar] [PubMed]
- Sobot, A.V.; Savic, J.; Trickovic, J.F.; Drakulic, D.; Joksic, G. Toxicity Assessment of Gentiana Lutea L. Root Extract and Its Monoterpene Compounds. Indian J. Exp. Biol. 2020, 58, 609–616. [Google Scholar] [CrossRef]
- Jia, N.; Ma, H.; Zhang, T.; Wang, L.; Cui, J.; Zha, Y.; Ding, Y.; Wang, J. Gentiopicroside Attenuates Collagen-Induced Arthritis in Mice via Modulating the CD147/P38/NF-ΚB Pathway. Int. Immunopharmacol. 2022, 108, 108854. [Google Scholar] [CrossRef]
- Lu, Y.; Yao, J.; Gong, C.; Wang, B.; Zhou, P.; Zhou, S.; Yao, X. Gentiopicroside Ameliorates Diabetic Peripheral Neuropathy by Modulating PPAR- Γ/AMPK/ACC Signaling Pathway. Cell. Physiol. Biochem. 2018, 50, 585–596. [Google Scholar] [CrossRef]
- He, M.; Hu, C.; Chen, M.; Gao, Q.; Li, L.; Tian, W. Effects of Gentiopicroside on Activation of NLRP3 Inflammasome in Acute Gouty Arthritis Mice Induced by MSU. J. Nat. Med. 2022, 76, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.-Y.; Nam, S.-J.; Lee, H.-I.; Lee, J.; Leutou, A.S.; Ri Ham, J.; Lee, M.-K. Gentiopicroside Isolated from Gentiana Scabra Bge. Inhibits Adipogenesis in 3T3-L1 Cells and Reduces Body Weight in Diet-Induced Obese Mice. Bioorg. Med. Chem. Lett. 2019, 29, 1699–1704. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, Y.; Jin, Q.; Xia, K.-L.; Jiang, M.; Cui, B.-W.; Wu, Y.-L.; Song, S.-Z.; Lian, L.-H.; Nan, J.-X. Liver Kinase B1/AMP-Activated Protein Kinase-Mediated Regulation by Gentiopicroside Ameliorates P2X7 Receptor-Dependent Alcoholic Hepatosteatosis. Br. J. Pharmacol. 2018, 175, 1451–1470. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, Y.; Wang, Y.; Cheng, M.; Yin, J.; Zhang, X.; Hong, Z. Gentiopicroside Ameliorates Bleomycin-Induced Pulmonary Fibrosis in Mice via Inhibiting Inflammatory and Fibrotic Process. Biochem. Biophys. Res. Commun. 2018, 495, 2396–2403. [Google Scholar] [CrossRef]
- Wang, Y.M.; Xu, M.; Wang, D.; Yang, C.R.; Zeng, Y.; Zhang, Y.J. Anti-inflammatory compounds of "Qin-Jiao", the roots of Gentiana dahurica (Gentianaceae). .J Ethnopharmacol. 2013, 147, 2–341. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhao, R.; Li, X.; Guo, H.; Tian, Z.; Zhang, N.; Gao, G.; Zhao, M. Attenuation of Reserpine-Induced Pain/Depression Dyad by Gentiopicroside Through Downregulation of GluN2B Receptors in the Amygdala of Mice. NeuroMolecular Med. 2014, 16, 350–359. [Google Scholar] [CrossRef]
- Yao, T.; Cui, Q.; Liu, Z.; Wang, C.; Zhang, Q.; Wang, G. Metabolomic Evidence for the Therapeutic Effect of Gentiopicroside in a Corticosterone-Induced Model of Depression. Biomed. Pharmacother. 2019, 120, 109549. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, M.; Xu, T.; Jin-Hou; Li, X. Gentiopicroside Abrogates Lipopolysaccharide-Induced Depressive-like Behavior in Mice through Tryptophan-Degrading Pathway. Metab. Brain Dis. 2018, 33, 1413–1420. [Google Scholar] [CrossRef]
- Almukainzi, M.; El-Masry, T.A.; Negm, W.A.; Elekhnawy, E.; Saleh, A.; Sayed, A.E.; Ahmed, H.M.; Abdelkader, D.H. Co-Delivery of Gentiopicroside and Thymoquinone Using Electrospun m-PEG/PVP Nanofibers: In Vitro and In Vivo Studies for Antibacterial Wound Dressing in Diabetic Rats. Int. J. Pharm. 2022, 625, 122106. [Google Scholar] [CrossRef]
- Berthon, J.Y.; Cabannes, M.; Bouton, C.; Carre, M.; Bridon, E.; Filaire, E. In vitro, ex vivo and clinical approaches to evaluate the potential effect of Gentiana lutea extract on skin. Int. J. Cosmet. Sci. 2023, 45, 5–688. [Google Scholar] [CrossRef] [PubMed]
- Koch, W.; Czop, M.; Iłowiecka, K.; Nawrocka, A.; Wiącek, D. Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products-Results of Analytical Determinations. Nutrients 2022, 14, 1626. [Google Scholar] [CrossRef] [PubMed]
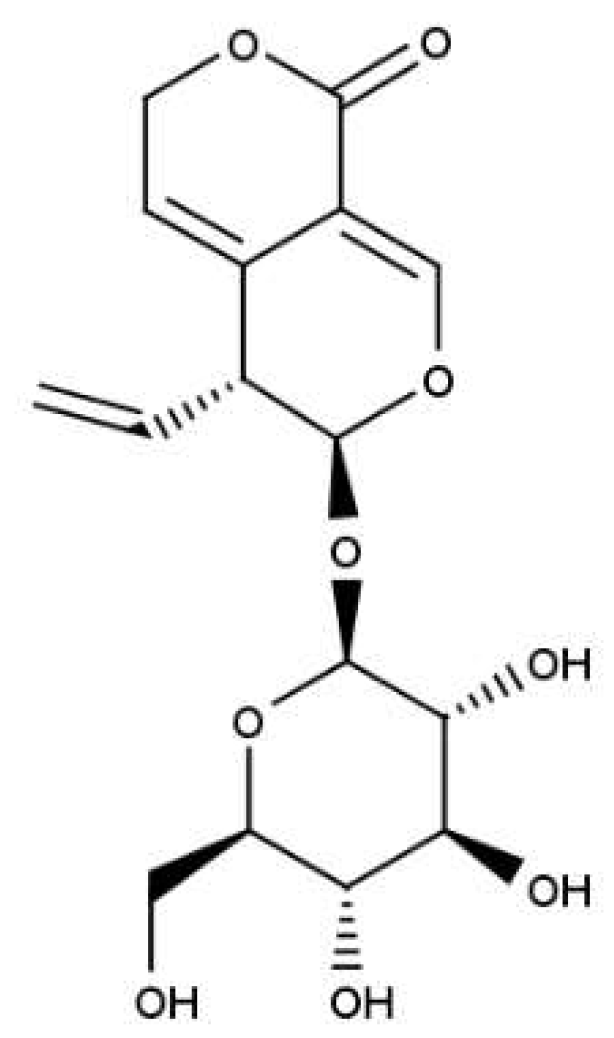
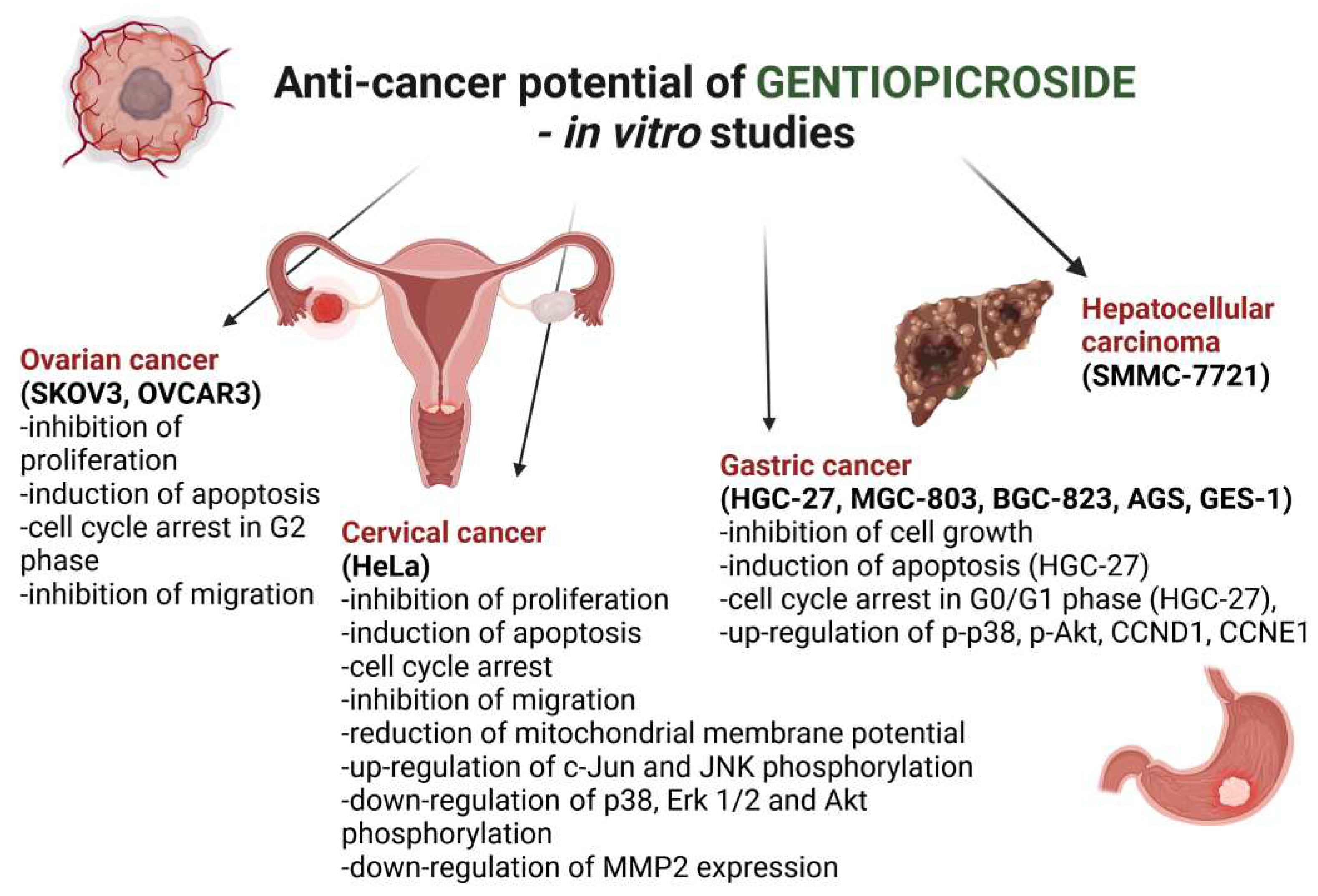
| Type of cancer | Cell lines | Mechanism of action | References |
| Ovarian | SKOV3 |
|
[51] |
| OVCAR3 |
|
[42] | |
| Cervical | HeLa |
|
[44] |
| Gastric | HGC-27 MGC-803 BGC-823 AGS GES-1 |
|
[47] |
| Hepatocellular carcinoma | SMMC-7721 | Not analyzed | [49] |
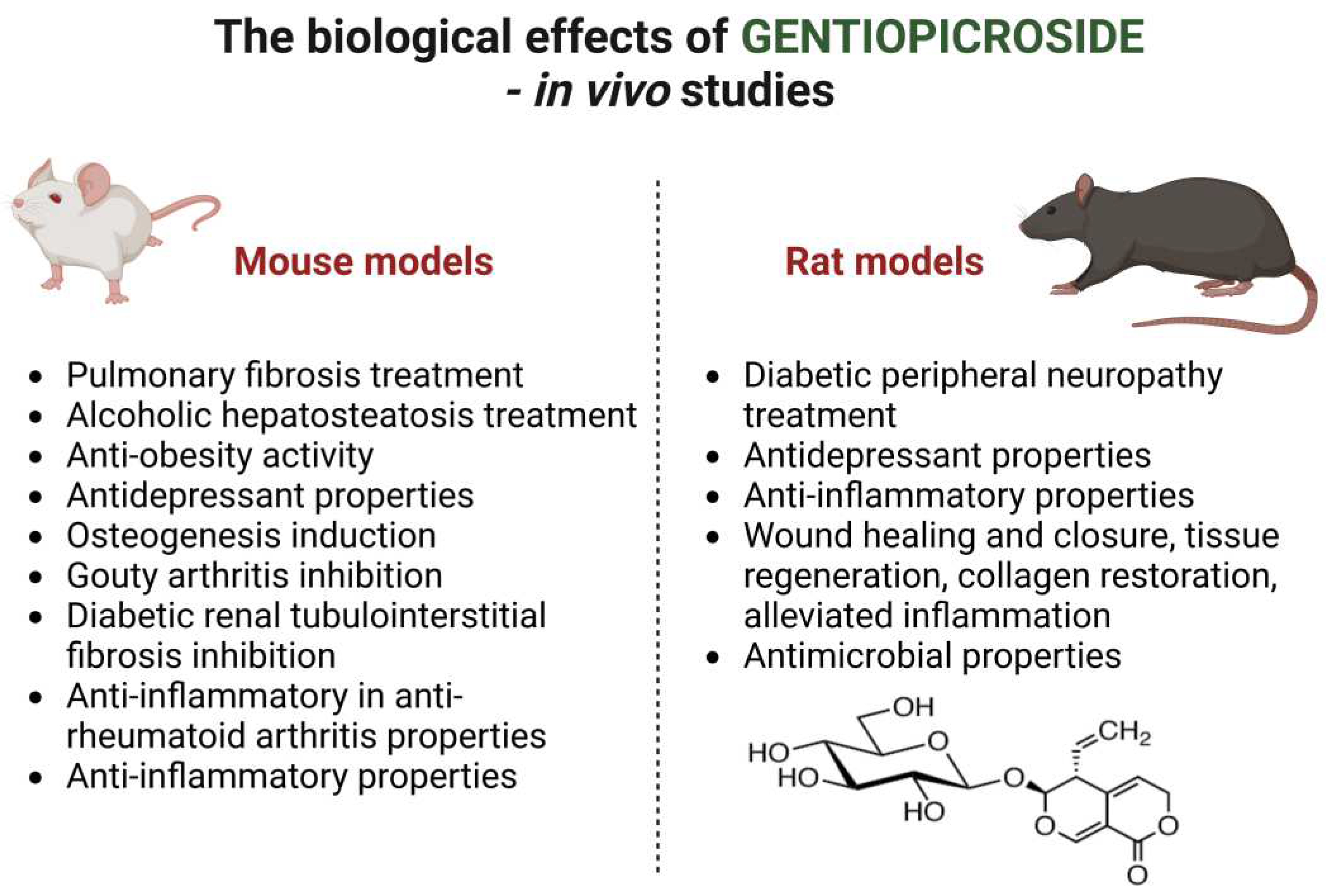
| Activity | Organism and dosage | Administration type | Duration | Action Mode | Reference |
| Pulmonary fibrosis treatment | Male SPF mice of Kunming strain 2.5 and 10 mg/ kg b.w. |
Intrapritoneal | 28 days | ↓hydroxyproline in lung tissue ↓ fibrotic and inflammatory responses. ↓ degeneration of typelalveolar epithelial cells ↓ TNF-α, IL-1β and TGF-β1 |
[58] |
| Alcoholic hepatosteatosis treatment | Male C57BL/6 mice Acute treatment: 40 and 80 mg/kg b.w. Chronic treatment: 40 mg/kg b.w. |
Intragastric | Acute treatment: 36 h, a dose every 12 h Chronic treatement: one dose daily for 10 days |
↓ ALT, AST, TG ↓ lipid droplets in the liver ↑LKB1 and AMPK phosphorylation ↓SREBP1 ↑phosphorylation of ACC ↓PPARα regulatory genes ↓lipogenesis ↓ alcohol-induced expression of NLRP3 and P2X7 |
[57] |
| Diabetic peripheral neuropathy treatment | Spraw Dawley rats 50 and 100 mg/ kg b.w. |
Intragastric | 14 days | ↑ restoring nerve blood flow, ↑moter nerve conduction velocity ↑ sensory nerve conduction velocity Regulation of dyslipidemia. |
[54] |
| Anti-obesity activity | C57BL/6N male mice 50 mg/ kg b.w. |
Oral | 12 weeks | ↓PPARγ, C/EBPα, SREBP-1c, ↓ genes related to fatty acids transport , uptake and synthesis ↓ cytokines ↓body weight ↓visceral fat mass ↓size of adipocytes |
[56] |
| Antidepressant properties | BALB/C mice 50 mg/ kg b.w. |
Intraperitoneal | 3 days | ↓ tryptophan metabolic pathways ↓ TNF-α and IL-1β ↑ expression of GluN2B subunit of NMDA receptor ↓ indoleamine 2,3-oxygenase ↓tryptophane-degrading pathway. |
[62] |
| Antidepressant properties | Sprague-Dawley rats 100 mg/ kg b.w. |
Intragastric | 21 days | ↓ apoptosis in hippocampus ↓ loss of Niss bodies ↑ serotonin ↑ BDNF and alleviate ↓ caspase 3, bax expression in brain. ↓ sphinganine, steroylethanolamide, guanosine, arachidonic acid, oxoadipic acid, L-phenlalanine and thiamine |
[61] |
| Antidepressant properties | Male adult C57BL/6 mice 50, 100 and 200 mg/kg |
Intragastric | 3 days, twice daily, once on the 4th day | ↓ mechanical allodynia ↑ traveled distance and the time in the center area ↑ norepinephrine, dopamine and serotonin ↓ intracellular levels of MDA ↑ CAT, Bcl-2 ↓ caspase 3 ↓ GluN2B expression |
[60] |
| Anti-inflammatory properties | Male Sprague-Dawley rats 100 and 200 mg/ kg |
Intragastric | 14 days | ↓paw swelling ↓ arthritis index ↓inflammation, bone destruction, synovial hyperplasia and pannus formation = body weight or spleen index, ↓reduction in thymus index ↓ IkBa degradation ↓ p-IkBa and p-p65 expression |
[22] |
| Osteogenesis induction | Female C57BL/6 mice 50 mg/ kg b.w. |
Oral gavage | 3 months | ↑alkaline phosphatase ↑osteogenic factors (Runx2, OCN, BMP2) ↑ thickness and number of trabeculae |
[23] |
| Gouty arthritis inhibition | Male C57BL/6 mice 50, 100, 200 mg/kg |
Intragastric | 24 h | ↓IL-1β, IL-6, IL-18, and TNF-α ↓ inflammatory infiltration ↓ caspase-1, NOD-like receptor protein 3, ACS ↑analgesic properties |
[55] |
| Diabetic renal tubulointerstitial fibrosis inhibition | Db/db male mice 50, 100 and 200 mg/ kg b.w. |
Intragastric | 10 weeks | ↓AT1R/CK2/NF-κB pathway ↓Scr, BUN, and 24 Up ↓FBG, of HbA1c, GSP, LDL-C, TG, body weight ↑Lumen expansion in TIF, glycogen deposition in the cortex ↓FN, α-SMA, and vimentin ↑ E-cadherin |
[30] |
| Anti-inflammatory in anti-rheumatoid arthritis | Male mice C57BL/6J with chicken type II collagen induced arthritis 20-40 mg/ kg b.w. |
Intragastric | 30 days of treatment | Modulation of CD147/p38/NF-κB pathway ↓pain ↓ p38, IκBα, p65 ↓ paw edema, mechanical allodynia ↓ inflammatory infiltrations ↓ IL-1β, IL-6, and TNF-α ↓ CD147, p-p38, p-IκBα, MMP1, MMP2, and MMP |
[53] |
| Anti-inflammatory properties | KM mice 0.28 mmol/ kg b.w. |
Oral | 7 days | ↓ edema degree by 34.17 % | [19] |
| Wound healing properties | Sprague-Dawley rats 300 mg/ kg b.w. |
Oral | 12 days | Wound healing and closure, tissue regeneration, collagen restoration, alleviated inflammation, antimicrobial properties, | [35] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




