Submitted:
19 October 2023
Posted:
19 October 2023
You are already at the latest version
Abstract
Keywords:
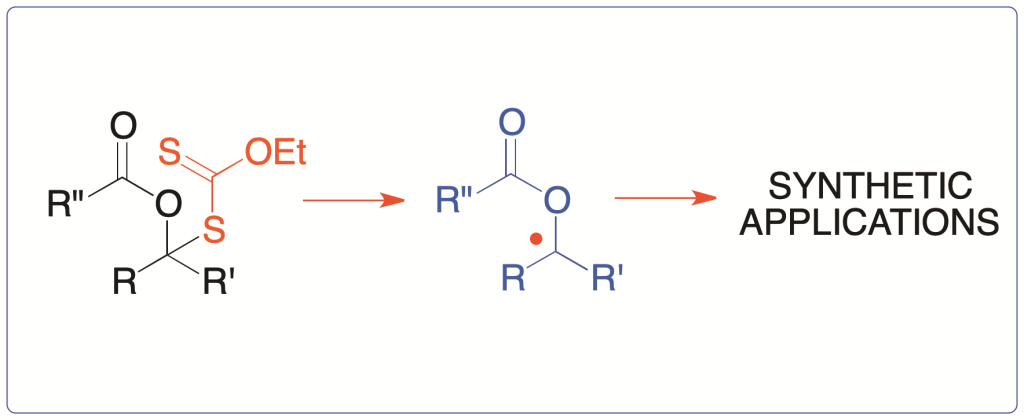
1. Introduction
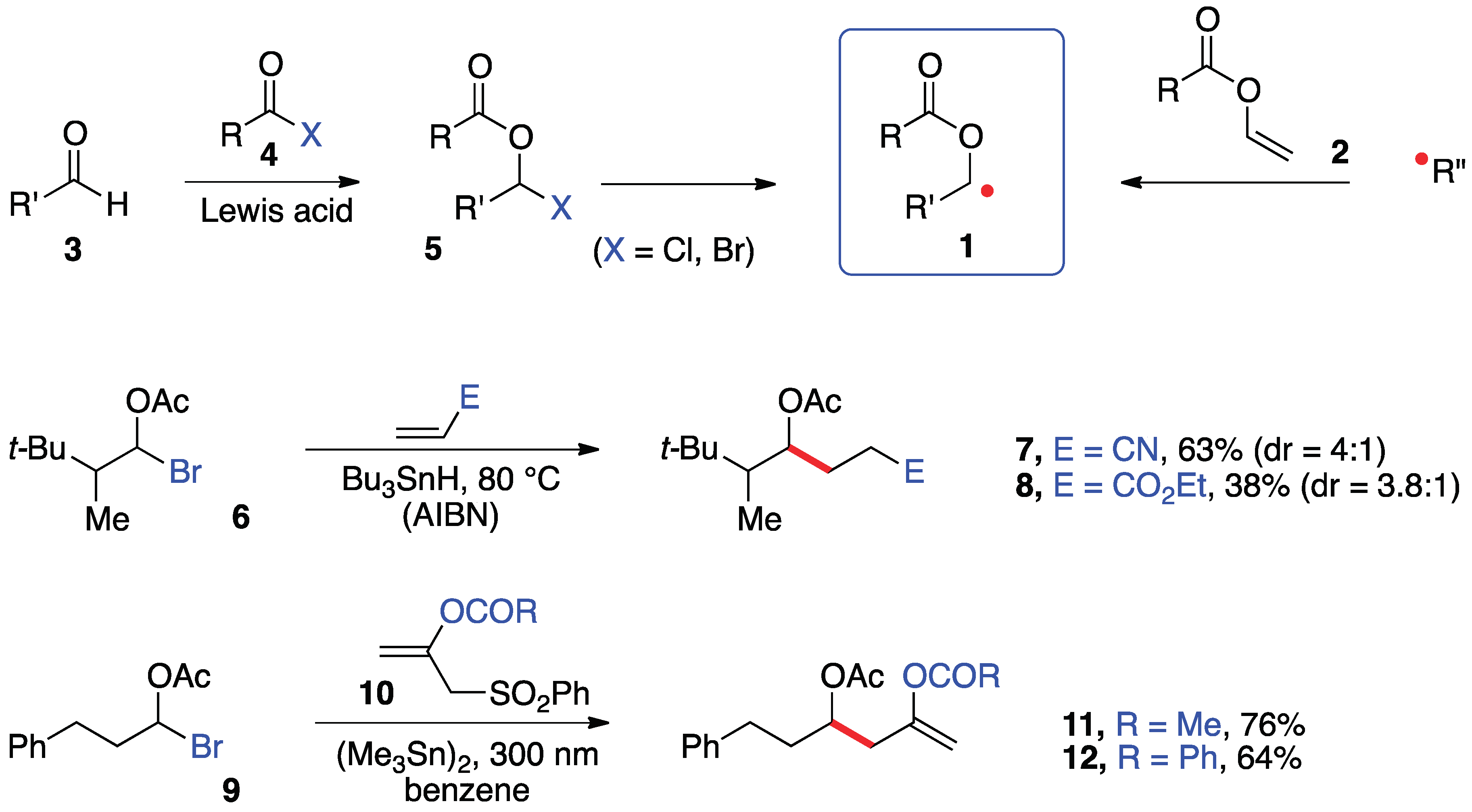
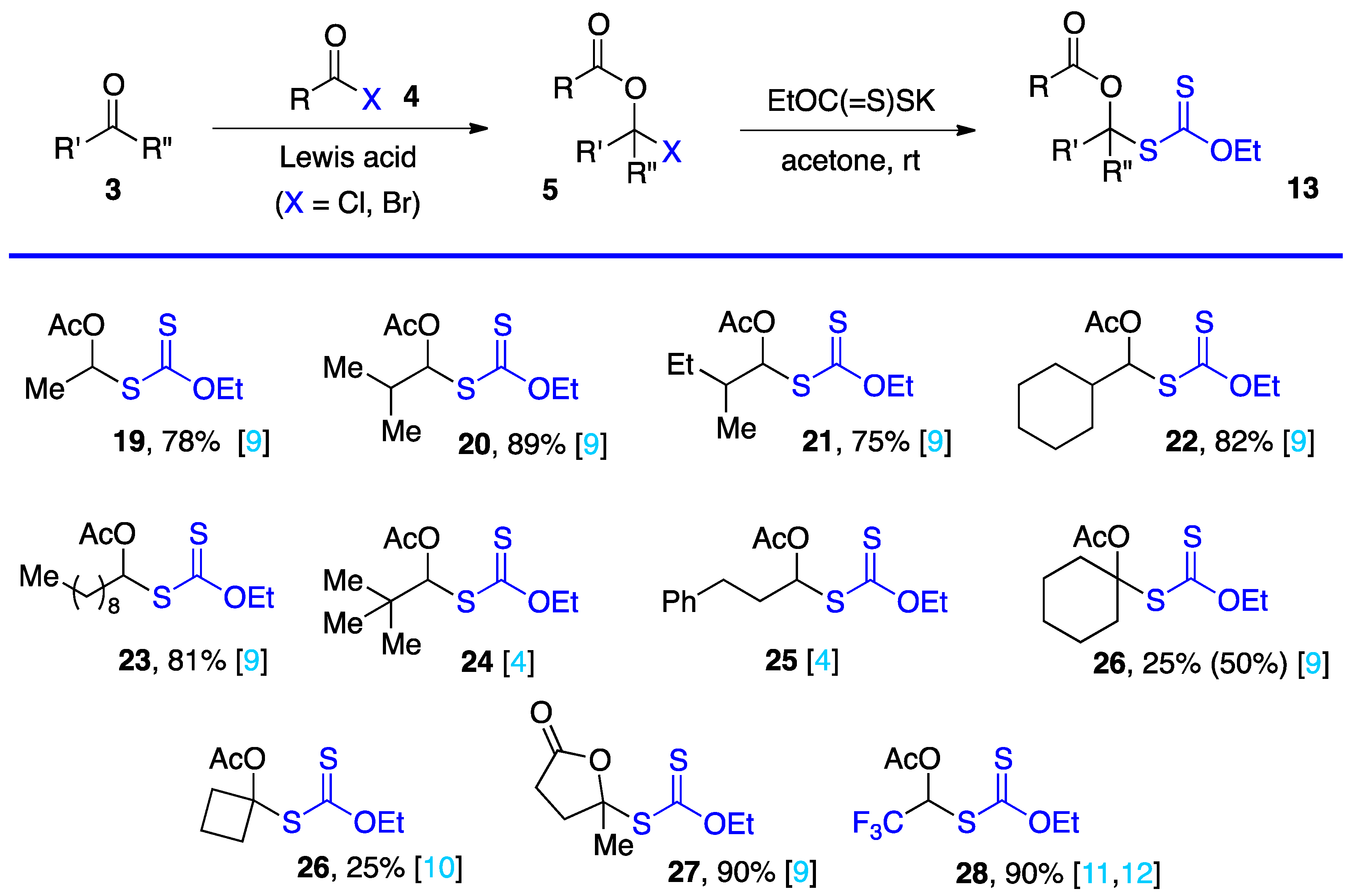
2. Synthesis of S-α-(acyloxy)alkyl xanthates
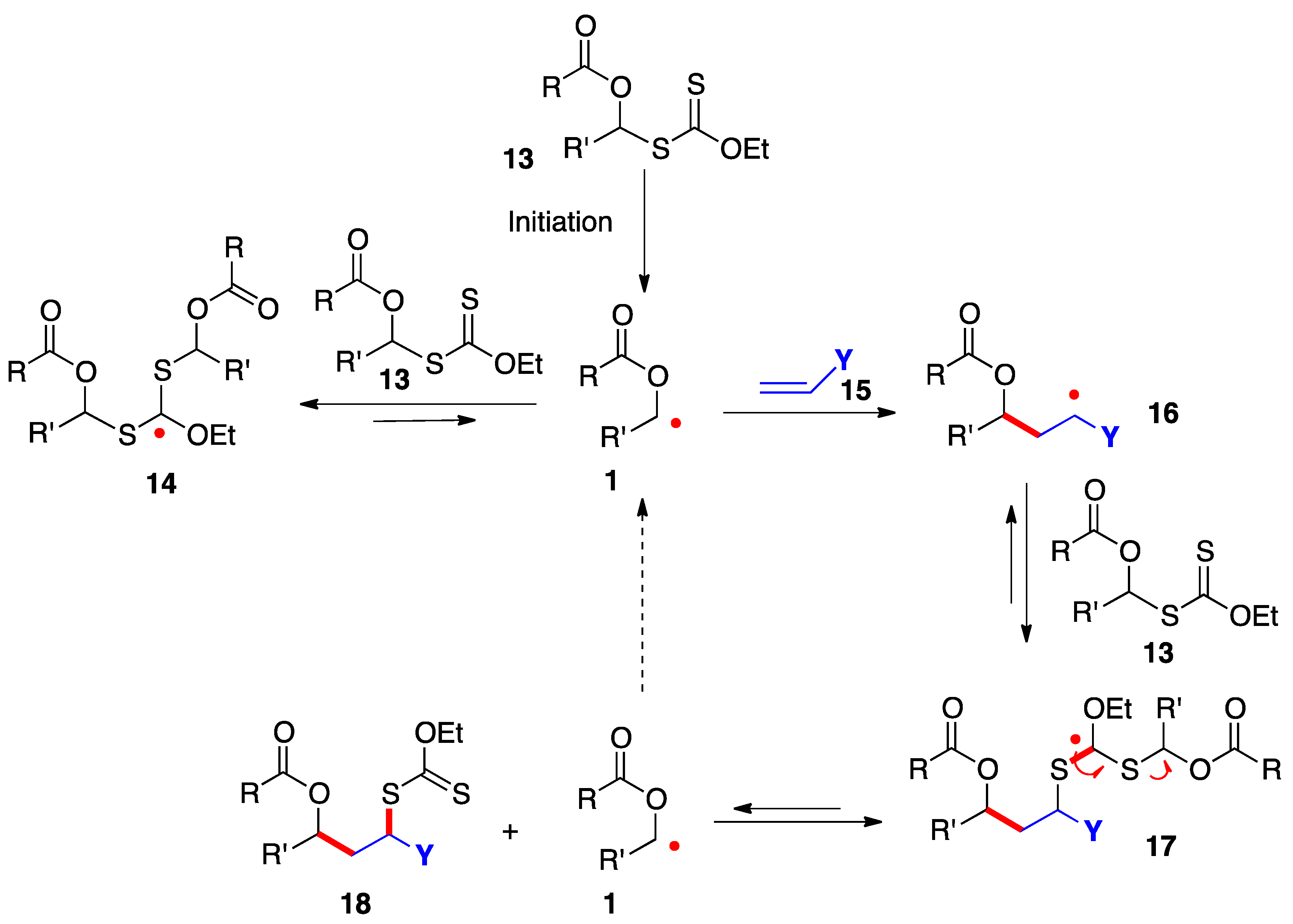
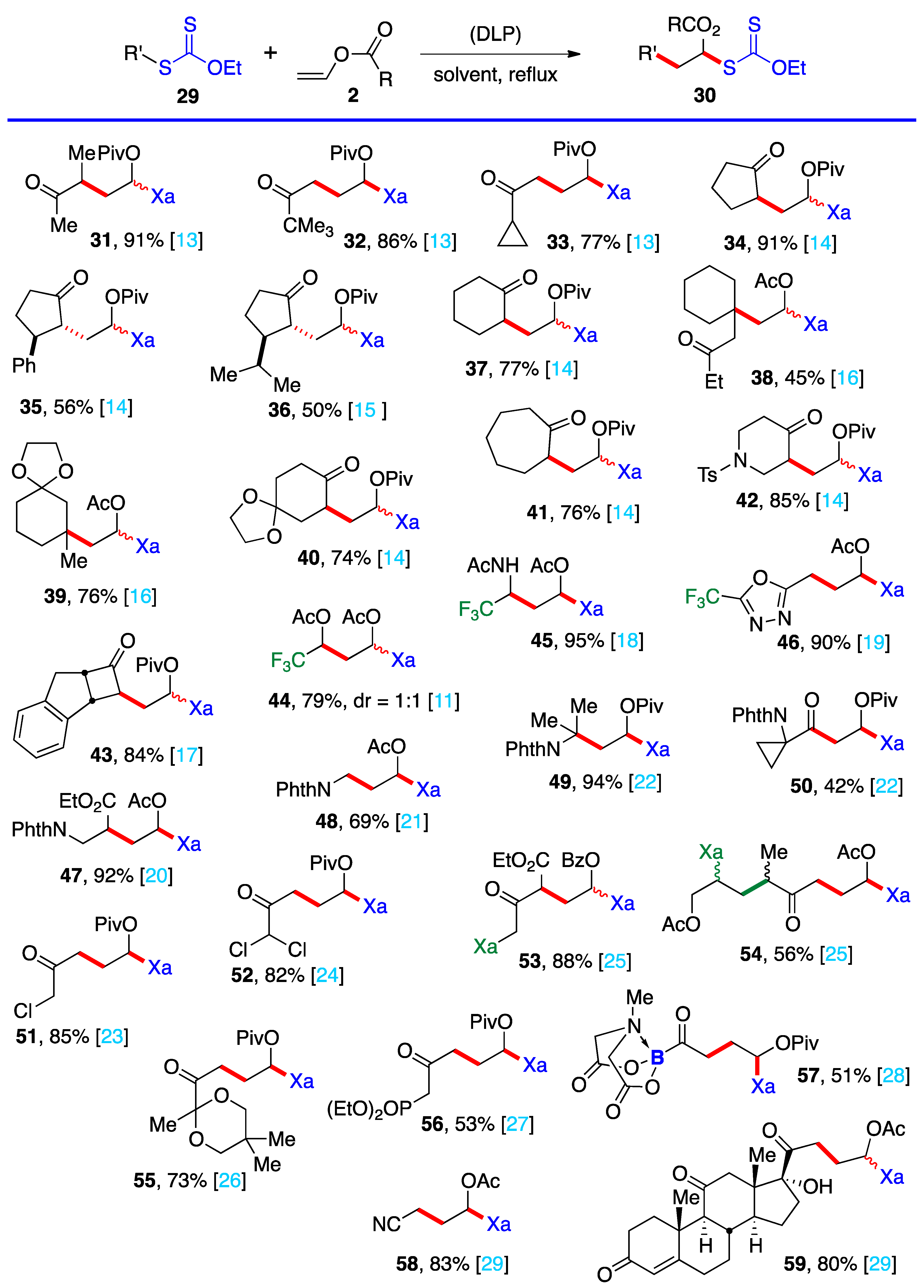
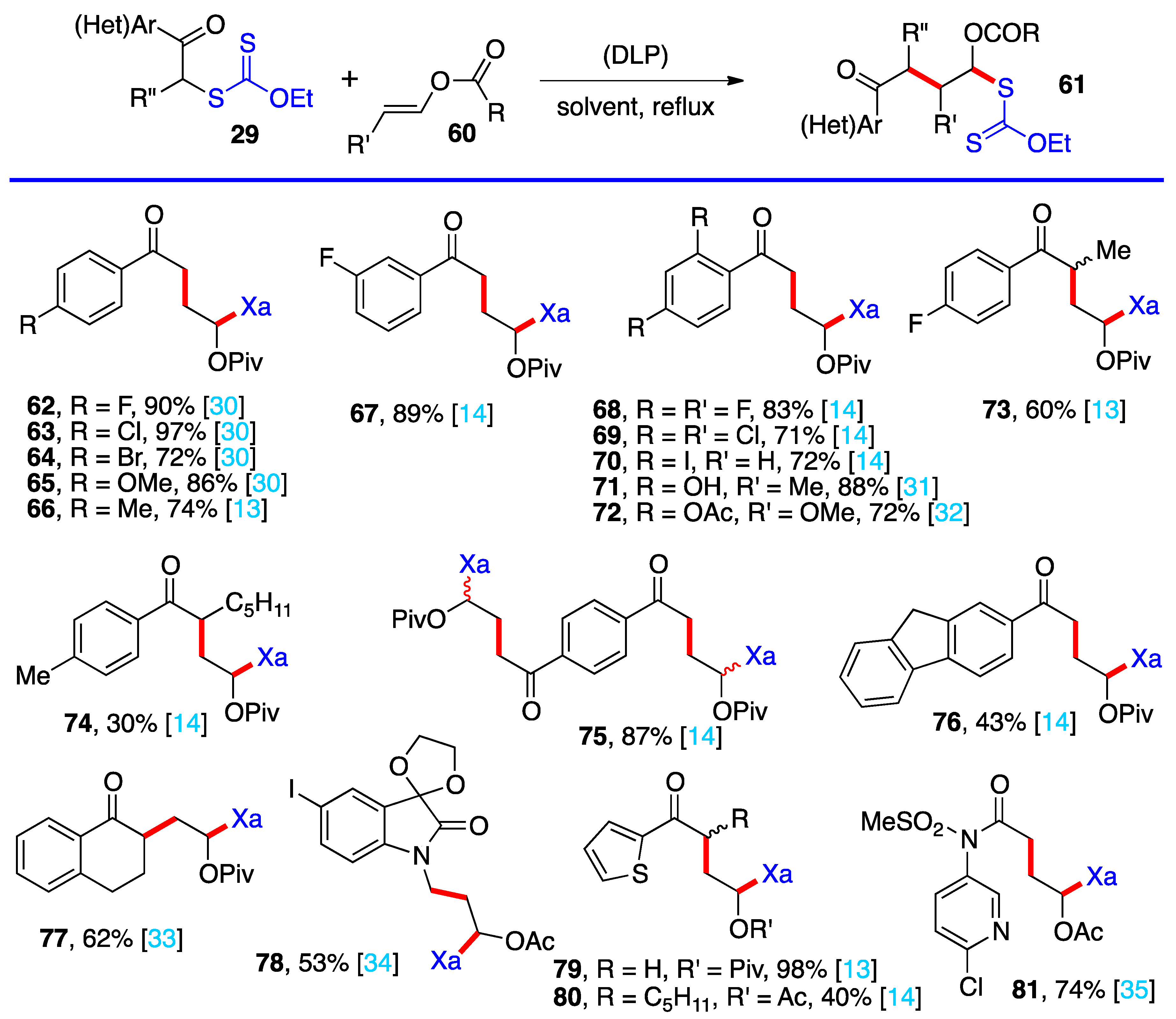
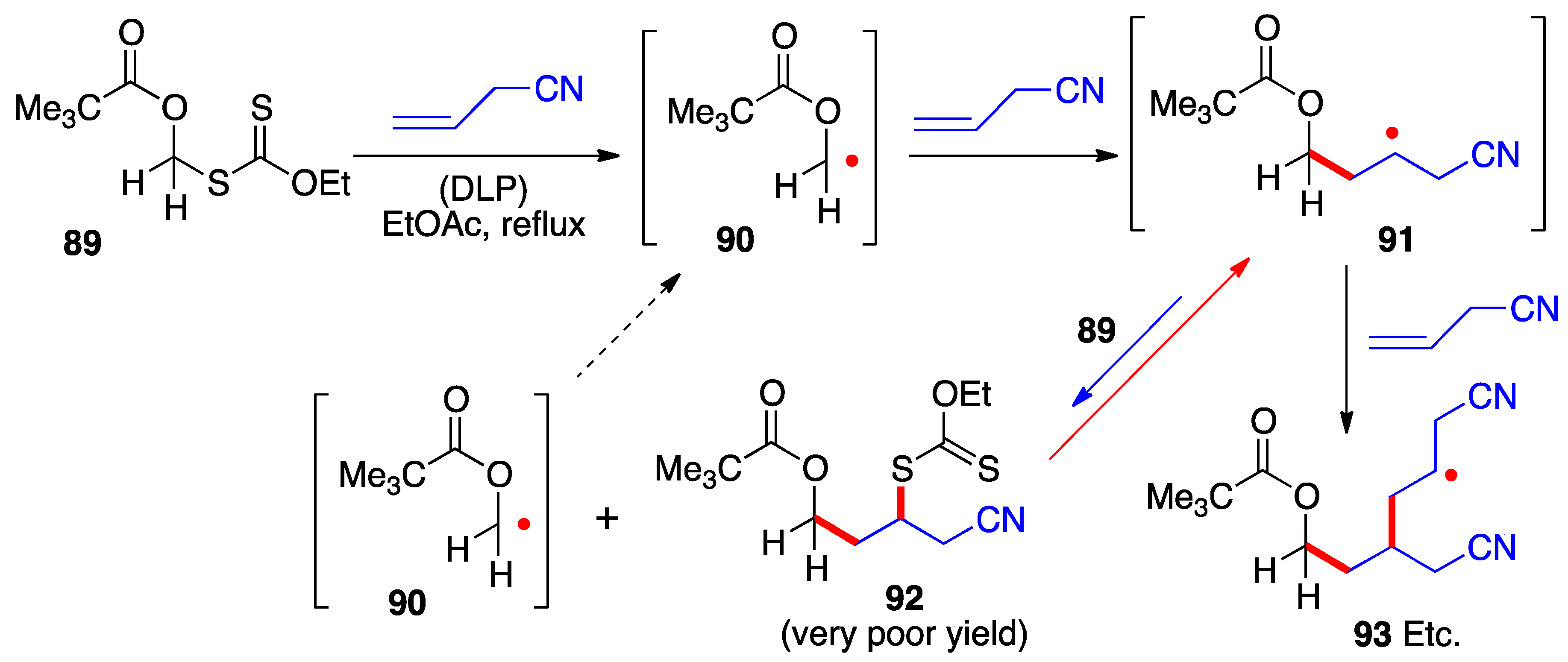
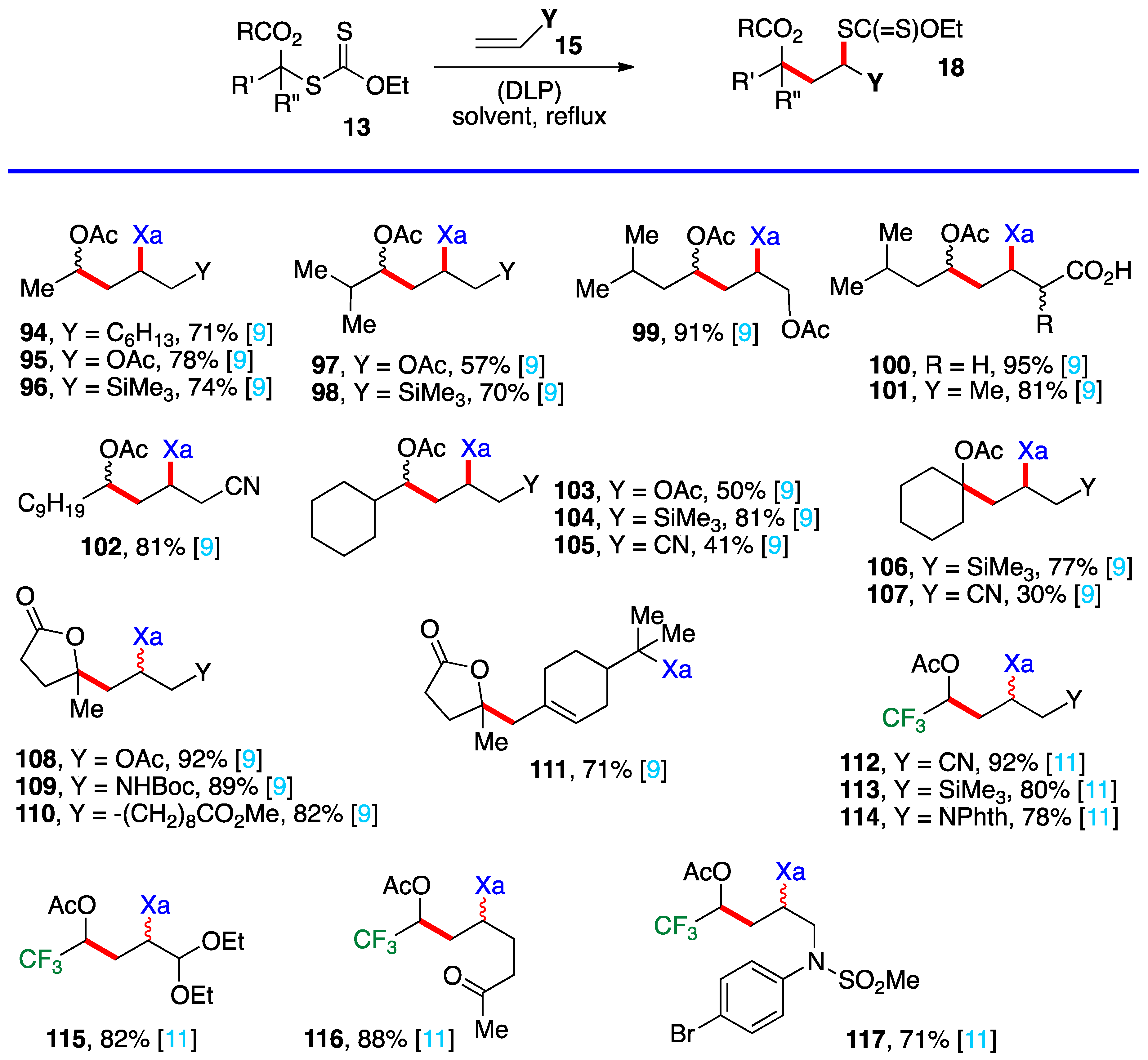
3. Radical additions of S-α-(acyloxy)alkyl xanthates
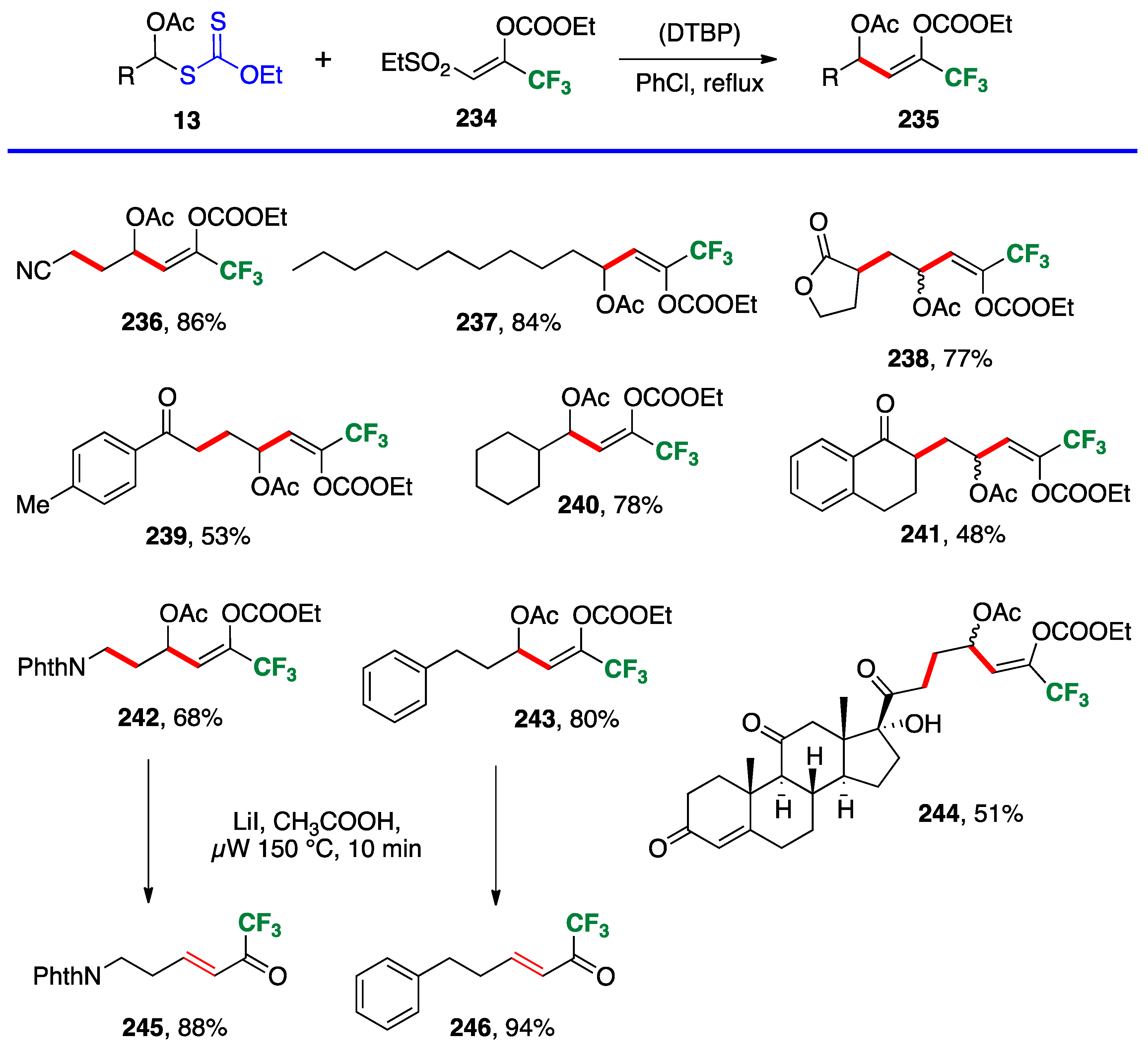
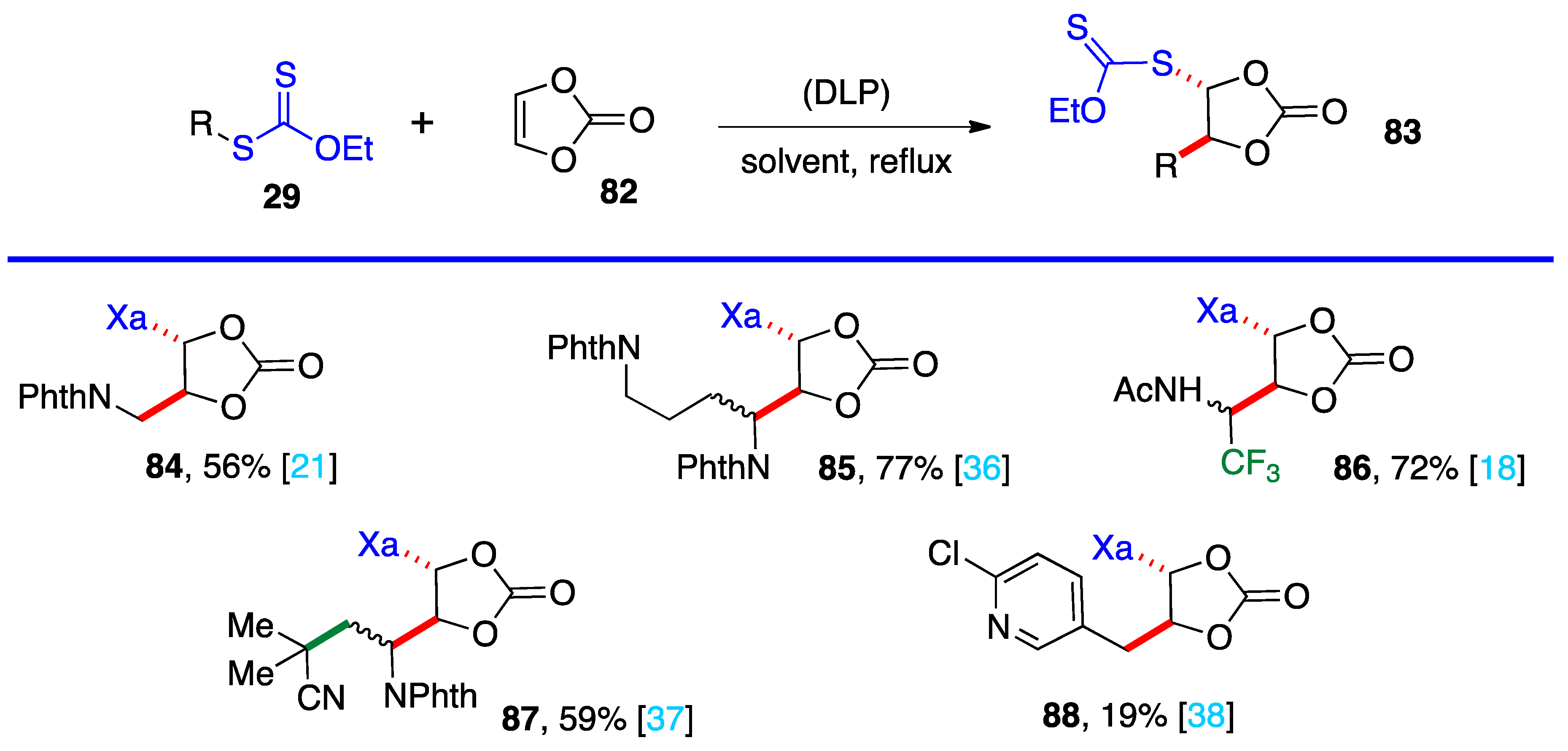
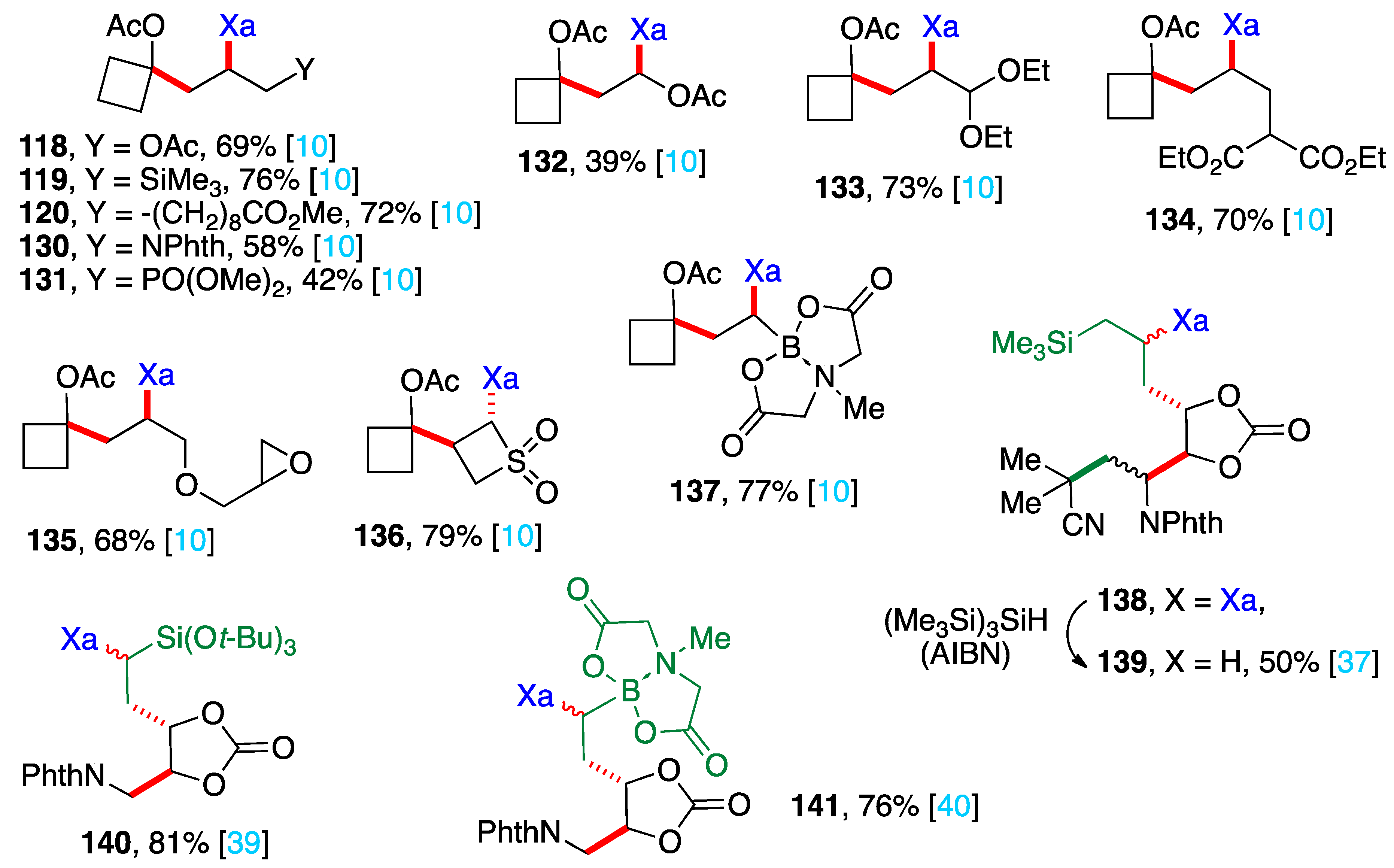
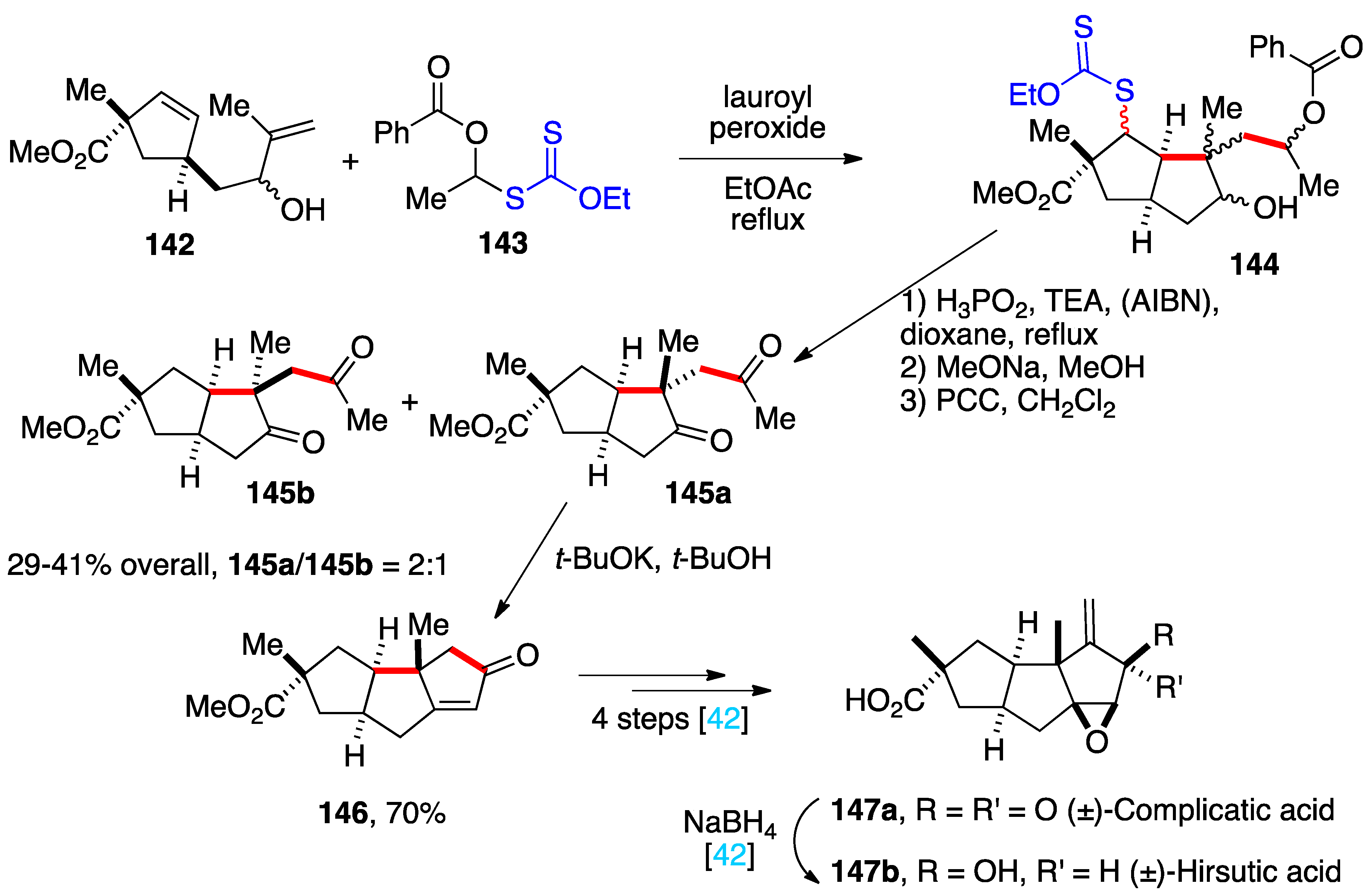
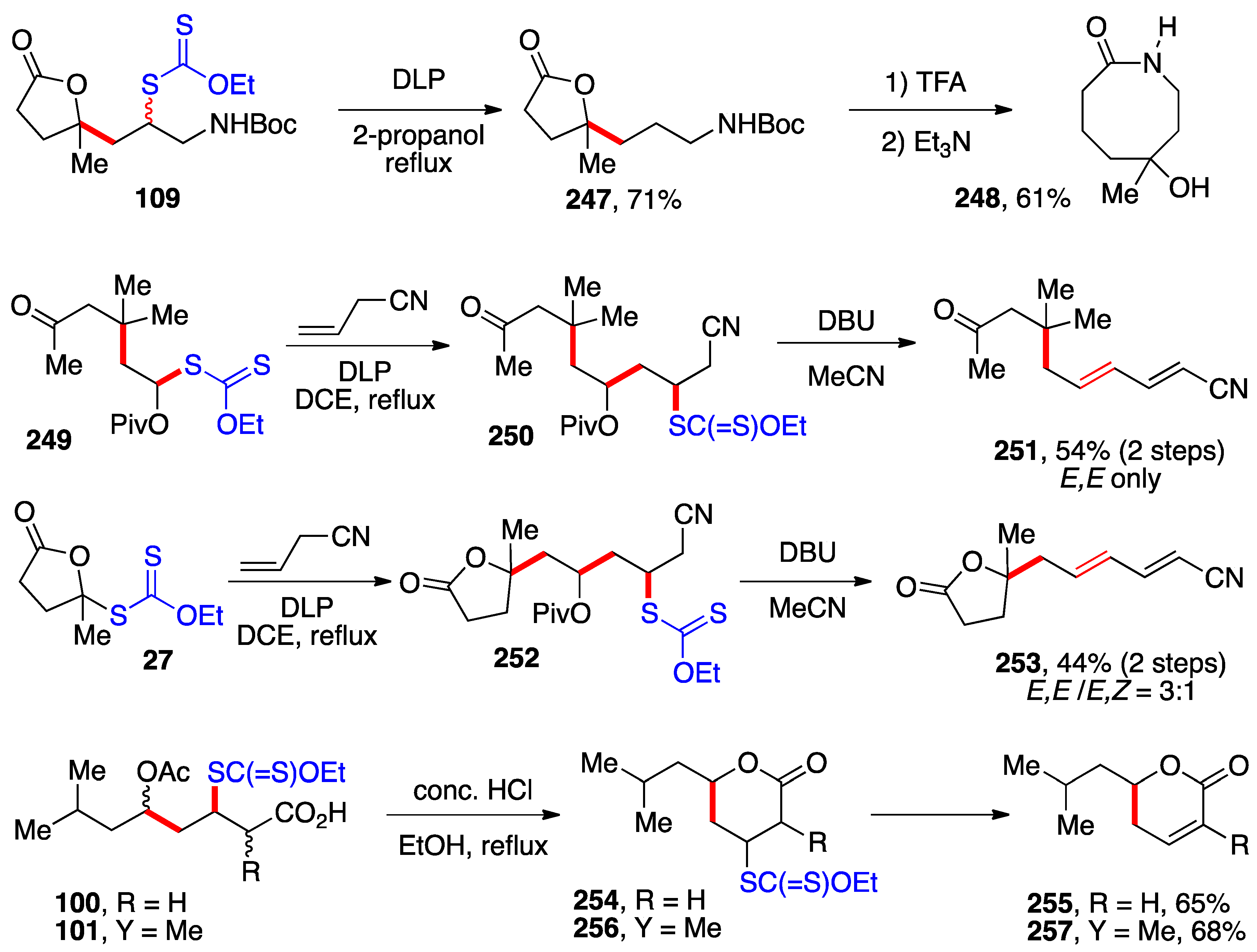
4. Further additions and applications of S-α-(acyloxy)alkyl xanthates
5. Concluding remarks
Acknowledgments
Conflicts of Interest
References
- Neuenschwander, M.; Bigler, P.; Christen, K.; Iseli, R.; Kyburz, R.; Mohle, H. ‘Chloracylierung und Bromacylierung von Carbonylverbindungen: Eine in Vergessenheit geratene Carbonylreaktion. I. Präparative Anwendungsbreite’. Helv. Chim. Acta 1978, 61, 2047–2058. [Google Scholar] [CrossRef]
- Chou, T.-S.; Knochel, P. A General Preparation of Highly Functionalized Zinc and Copper Organometallics at the a-Position to an Oxygen. J. Org. Chem. 1990, 55, 4791–4793. [Google Scholar] [CrossRef]
- Giese, B.; Damm, W.; Dickhaut, J.; Wetterich, F.; Sun, S.; Curran, D.P. Cram’s Rule for Radical Reactions. Tetrahedron Lett. 1991, 32, 6097–6100. [Google Scholar] [CrossRef]
- Lee J., Y.; Kim, S. Tin-free Radical Allylation of a-Acetoxy Alkyl Xanthates. Routes to Homoallyl Alcohols and a,b-Unsaturated Ketones. Bull. Korean Chem. Soc. 2006, 27, 189–190. [Google Scholar]
- Quiclet-Sire, B.; Zard, S.Z. Fun with Radicals: Some New Perspectives for Organic Synthesis. Pure & Appl. Chem. 2011, 83, 519–551. [Google Scholar]
- Quiclet-Sire, B.; Zard, S.Z. On the Strategic Impact of the Degenerative Transfer of Xanthates on Synthetic Planning. Isr. J. Chem. 2017, 57, 202–217. [Google Scholar] [CrossRef]
- Zard, S.Z. Radical Alliances: Solutions and Opportunities for Organic Synthesis. Helv. Chim. Acta 2019, 102, e1900134. [Google Scholar] [CrossRef]
- Zard, S.Z. Discovery of the RAFT/MADIX Process. Mechanistic Insights and Polymer Chemistry Implications. Macromolecules 2020, 53, 8144–8159. [Google Scholar] [CrossRef]
- Bagal, S.K.; Tournier, L.; Zard, S.Z. Radicals from Aldehydes. A Convergent Access to Dienes and Unsaturated δ-Lactones. Synlett 2006, 1485–1490. [Google Scholar]
- Revil-Baudard, V.L.; Zard, S.Z. A Practical Route to Cyclobutanols and to Fluorocyclobutanes. Helv. Chim. Acta 2021, 104, e2100106. [Google Scholar] [CrossRef]
- Tournier, L.; Zard, S.Z. A Direct Approach to α-Hydroxy- and α-Chloro-Trifluoromethyl Derivatives. Tetrahedron Lett. 2005, 46, 455–459. [Google Scholar] [CrossRef]
- Salomon, P.; Kosnik, W.; Zard, S.Z. A Convergent, Modular Access to α-Chloro-Trifluoromethyl Derivatives and to 1,1-Difluoroalkenes. Tetrahedron 2015, 71, 7144–7153. [Google Scholar] [CrossRef]
- Quiclet-Sire, B.; Quintero, L.; Sanchez-Jimenez, G.; Zard, S.Z. A New Practical Variant of the Paal-Knorr Synthesis of Pyrroles. Synlett 2003, 75–78. [Google Scholar]
- Jullien, H.; Quiclet-Sire, B.; Tétart, T.; Zard, S.Z. Convergent Routes to Thiophenes. Org. Lett. 2014, 16, 302–305. [Google Scholar] [CrossRef]
- Bieszczad, B.; Chen, X.; Zard, S.Z. An Ionic-Radical Approach to Vicinally Functionalized Cyclopentanones and Cyclohexanones. Org. Lett. 2022, 24, 9370–9374. [Google Scholar] [CrossRef]
- Binot, G.; Quiclet-Sire, B.; Saleh, T.; Zard, S.Z. A Convergent Construction of Quaternary Centres and Polycyclic Structures. Synlett 2003, 382–386. [Google Scholar]
- Binot, G.; Zard, S.Z. Intermolecular Additions of Cyclobutanone Derived Radicals. A Convergent, Highly Efficient Access to Polycyclic Cyclobutane Containing Structures. Tetrahedron Lett. 2003, 44, 7703–7706. [Google Scholar] [CrossRef]
- Gagosz, F.; Zard, S.Z. A Direct Approach to α-Trifluoromethylamines. Org. Lett. 2003, 5, 2655–2657. [Google Scholar] [CrossRef]
- Qin, L.; Zard, S.Z. A Radical Based Route to 2-(Trifluoromethyl)-1,3,4-oxadiazoles and to Trifluoromethyl-Substituted Polycyclic 1,2,4-Triazoles and Dihydrofuran. Org. Lett. 2015, 17, 1577–1580. [Google Scholar] [CrossRef]
- Chen, X.; Zard, S.Z. A Convergent Route to β-Amino Acids and to β-Heteroarylethylamines: An Unexpected Vinylation Reaction. Org. Lett. 2020, 22, 3628–3632. [Google Scholar] [CrossRef]
- Quiclet-Sire, B.; Zard, S.Z. A Radical Aminomethylation of Alkenes. Org. Lett. 2008, 10, 3279–3282. [Google Scholar] [CrossRef]
- Heinrich, M.; Zard, S.Z. The Generation and Intermolecular Capture of Cyclopropylacyl Radicals. Org. Lett. 2004, 6, 4969–4972. [Google Scholar] [CrossRef] [PubMed]
- Bergeot, O.; Corsi, C.; El Qacemi, M.; Zard, S.Z. S-(3-Chloro-2-oxo-propyl)-O-ethyl Xanthate: A Linchpin Radical Coupling Agent for the Synthesis of Heterocyclic and Polycyclic Compounds. Org. Biomol. Chem. 2006, 4, 278–290. [Google Scholar] [CrossRef] [PubMed]
- Anthore, L.; Li, S.; White, L.V.; Zard, S.Z. A Radical Solution to the Alkylation of the Highly Base-Sensitive 1,1-Dichloroacetone. Application to the Synthesis of Z-alkenoates and E, E-dienoates. Org. Lett. 2015, 17, 5320–5323. [Google Scholar] [CrossRef]
- Anthore-Dalion, L.; Liu, Q.; Zard, S.Z. A Radical Bidirectional Fragment Coupling Route to Unsymmetrical Ketones. J. Am. Chem. Soc. 2016, 138, 8404–8407. [Google Scholar] [CrossRef] [PubMed]
- Mougin, C.; Sançon, J.; Zard, S.Z. A Practical Route to Substituted Pyrazines, Quinoxalines, and an Unusual Synthesis of Benzimidazoles. Heterocycles 2007, 74, 211–218. [Google Scholar]
- Corbet, M.; de Greef, M.; Zard, S.Z. A Highly Conjunctive β-Keto Phosphonate: Application to the Synthesis of Pyridine Alkaloids Xestamines C, E, and H. Org. Lett. 2008, 10, 253–256. [Google Scholar] [CrossRef]
- Qiao, H.; Michalland, J.; Huang, Q.; Zard, S.Z. A Versatile Route to Acyl (MIDA)boronates. Chem. Eur. J. 2023, 29, e202302235. [Google Scholar] [CrossRef]
- Huang, Q.; Qin, L.; Zard, S.Z. Xanthate Mediated Intermolecular Alkylation of Pyrazines. Tetrahedron 2018, 74, 5804–5817. [Google Scholar] [CrossRef]
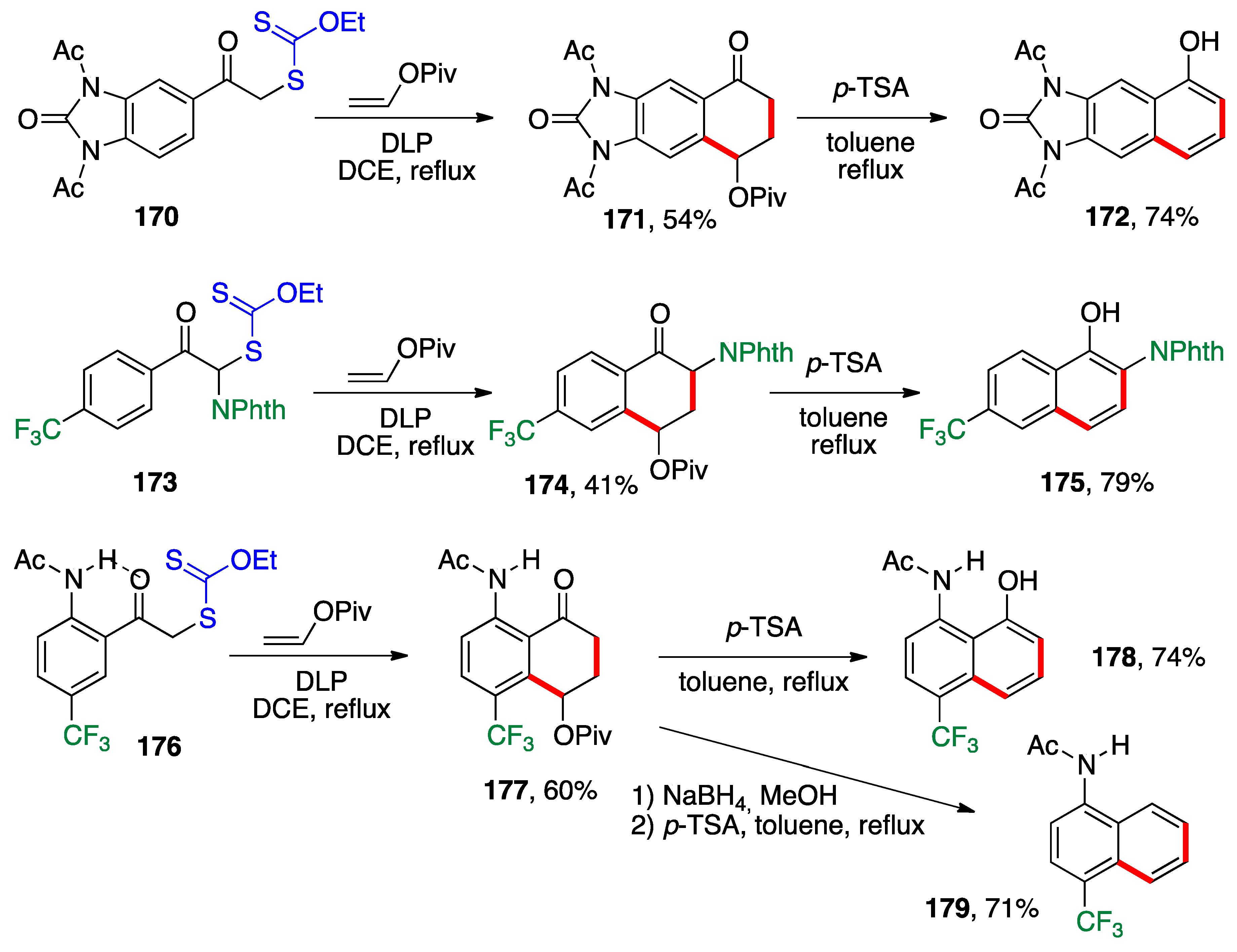
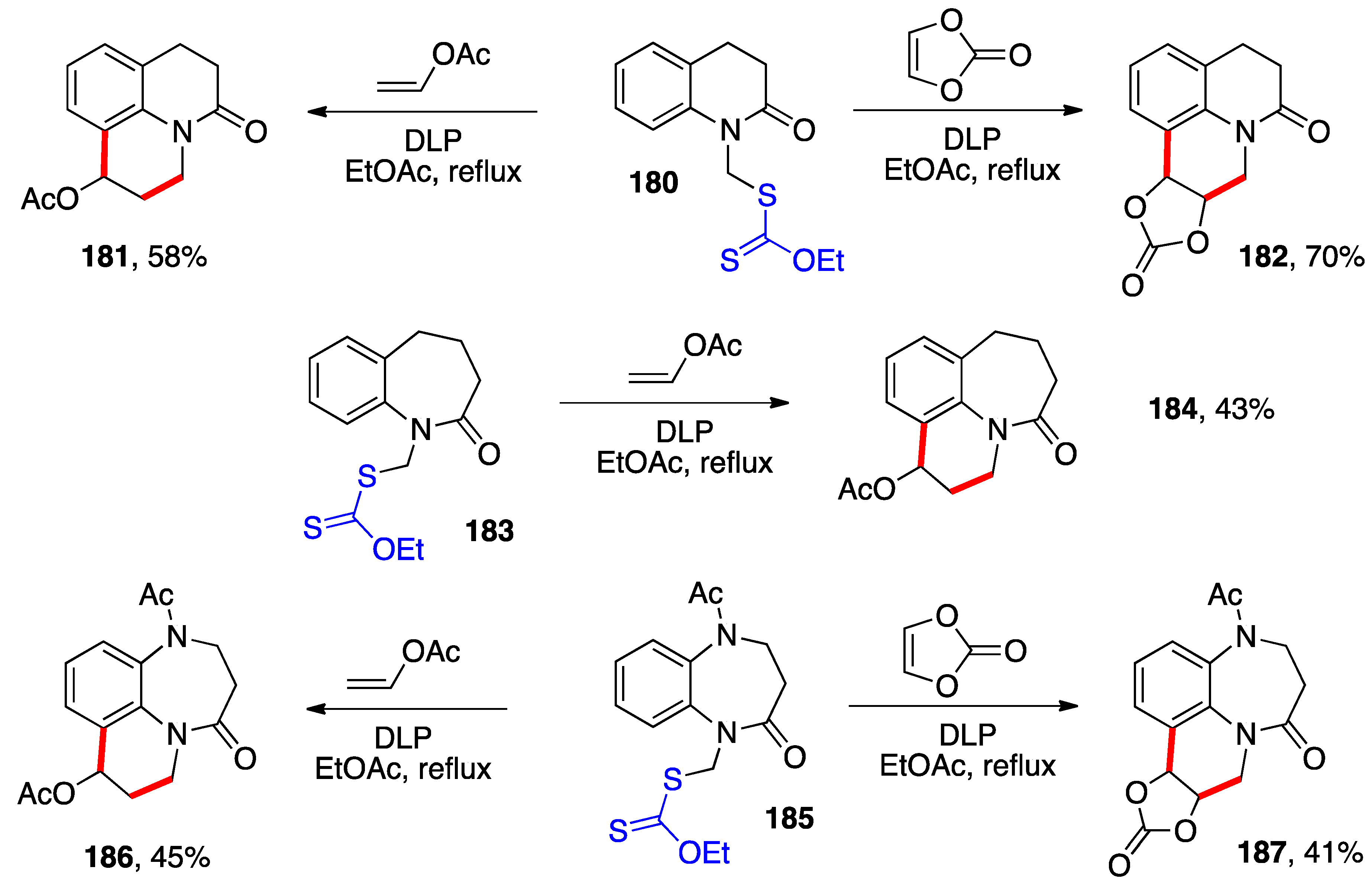
- Cordero-Vargas, A.; Quiclet-Sire, B.; Zard, S.Z. A Practical Method for the Preparation of Substituted Benzazepines: Application to the Synthesis of Tolvaptan. Bioorg. Med. Chem. 2006, 14, 6165–6173. [Google Scholar] [CrossRef]
- Petit, L.; Zard, S.Z. A Radical-Based Approach to Hydroxytetralones from Unprotected Phenols. Chem. Commun. 2010, 46, 5148–5150. [Google Scholar] [CrossRef] [PubMed]
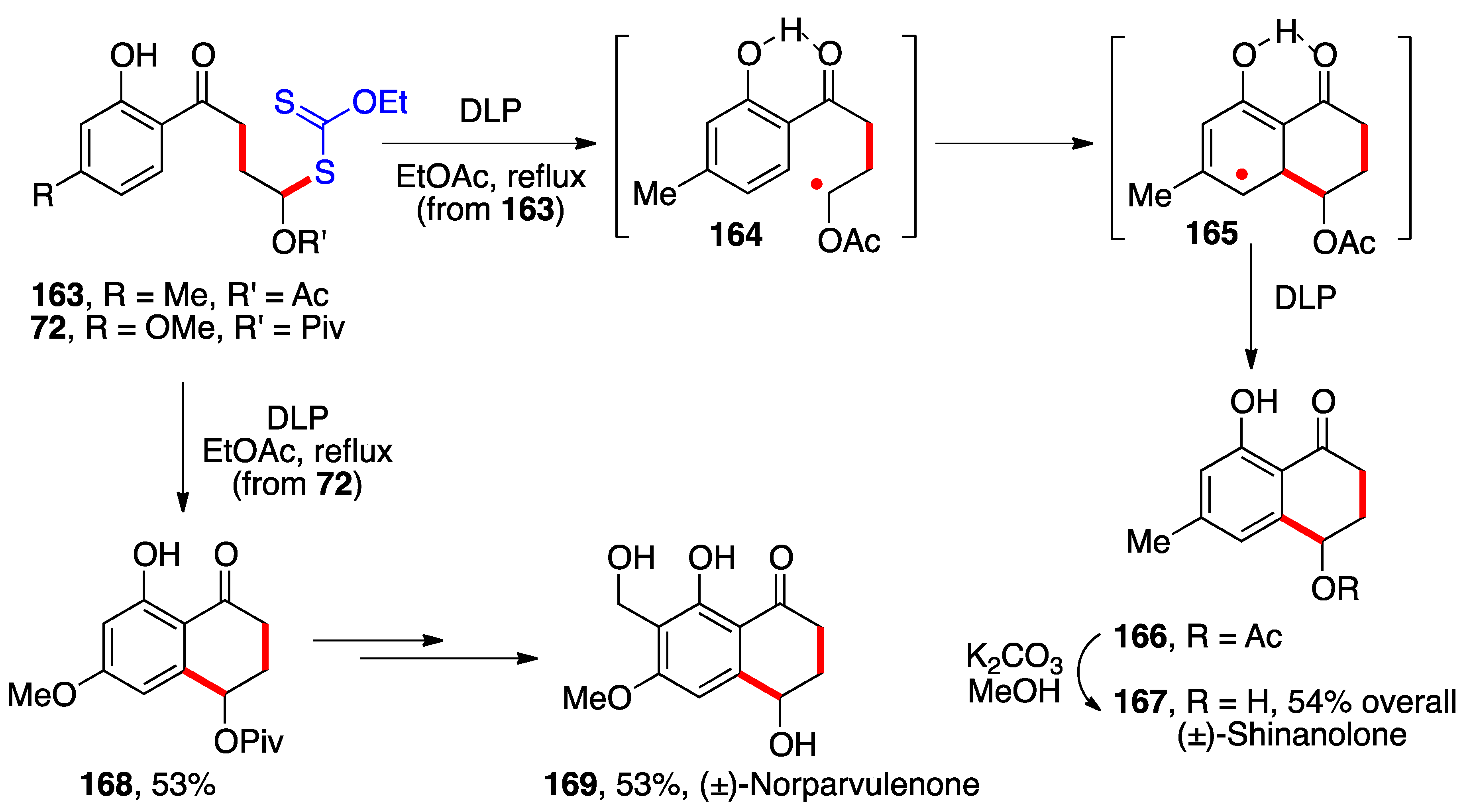
- Cordero Vargas, A.; Quiclet-Sire, B.; Zard, S.Z. Total Synthesis of 10-Norparvulenone and of O-Methylasparvenone Using a Xanthate-Mediated Free Radical Addition-Cyclisation Sequence. Org. Lett. 2003, 5, 3717–3719. [Google Scholar] [CrossRef] [PubMed]
- Quiclet-Sire, B.; Sanchez-Jimenez, G.; Zard, S.Z. A New, Unexpected Synthesis of 1,3-Dithietanones. Chem. Commun. 2003, 1408–1409. [Google Scholar] [CrossRef] [PubMed]
- Lebreux, F.; Quiclet-Sire, B.; Zard, S.Z. Radical Arylaminomethylation of Unactivated Alkenes. Org. Lett. 2009, 11, 2844–2847. [Google Scholar] [CrossRef] [PubMed]
- Dorokhov, V.S.; Zard, S.Z. Modular Approach to Pyridoazepinones. Org. Lett. 2021, 23, 2164–2168. [Google Scholar] [CrossRef]
- Quiclet-Sire, B.; Revol, G.; Zard, S.Z. Functional Primary Amines and Diamines from α-Aminoacids. A Concise Route to Substituted 2-Aminotetralins. Org. Lett. 2009, 11, 3554–3557. [Google Scholar] [CrossRef]
- Quiclet-Sire, B.; Revol, G.; Zard, S.Z. A Convergent, Modular Approach to Complex Amines. Tetrahedron 2010, 66, 6656–6666. [Google Scholar] [CrossRef]
- Huang, Q.; Zard, S.Z. A Modular Route to Azaindanes. Org. Lett. 2017, 19, 3895–3898. [Google Scholar] [CrossRef] [PubMed]
- Quiclet-Sire, B.; Yanagisawa, Y.; Zard, S.Z. A Direct, Versatile Route to Functionalized Trialkoxysilanes. Chem. Commun. 2014, 50, 2324–2326. [Google Scholar] [CrossRef]
- Quiclet-Sire, B.; Zard, S.Z. Radical Instability in Aid of Efficiency. A Powerful Route to Highly Functional MIDA Boronates. J. Am. Chem. Soc. 2015, 137, 6762–6765. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, C.; Revol, G.; Zard, S.Z. A Short Formal Total Synthesis of (±)-Hirsutic Acid. Can. J. Chem. 2012, 90, 927–931. [Google Scholar]
- Hashimoto, H.; Tsuzuki, K.; Sakan, F.; Shirahama, H.; Matsumoto, T. Total synthesis of dl-hirsutic acid. Tetrahedron Lett. 1974, 15, 3745–3748. [Google Scholar] [CrossRef]
- Heng, R.; Zard, S.Z. A flexible, Unified Approach to Polycyclic Structures. Org. Biomol. Chem. 2011, 9, 3396–3404. [Google Scholar] [CrossRef] [PubMed]
- Ingold, K.U.; Pratt, D.A. Advances in Radical-Trapping Antioxidant Chemistry in the 21st Century: A Kinetics and Mechanisms Perspective. Chem. Rev. 2014, 114, 9022–9046. [Google Scholar] [CrossRef] [PubMed]
- Avila, D.V.; Ingold, K.U.; Lusztyk, J.; Green, W.H.; Procopio, D.R. Dramatic Solvent Effects on the Absolute Rate Constants for Abstraction of the Hydroxylic Hydrogen Atom from tert-Butyl Hydroperoxide and Phenol by the Cumyloxyl Radical. The Role of Hydrogen Bonding. J. Am. Chem. Soc. 1995, 117, 2929–2930. [Google Scholar] [CrossRef]
- Cordero-Vargas, A.; Pérez-Martin, I.; Quiclet-Sire, B.; Zard, S.Z. Synthesis of Substituted Naphthalenes from α-Tetralones Generated by a Xanthate Radical Addition-Cyclisation Sequence. Org. Biomol. Chem. 2004, 2, 3018–3025. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.D.M.; Zard, S.Z. Convergent Routes to Substituted Naphthylamides. Org. Biomol. Chem. 2014, 12, 3251–3264. [Google Scholar] [CrossRef] [PubMed]
- Quiclet-Sire, B.; Zard, S.Z. The Xanthate Route to Tetralones, Tetralins, and Naphthalenes. A Brief Account. Org. Biomol. Chem. 2023, 21, 910–924. [Google Scholar] [CrossRef] [PubMed]
- Biéchy, A.; Zard, S.Z. A Flexible, Convergent Approach to Polycyclic Indole Structures: Formal Synthesis of (±)-Mersicarpine. Org. Lett. 2009, 11, 2800–2803. [Google Scholar] [CrossRef]
- Charrier, N.; Gravestock, D.; Zard, S.Z. Radical Additions of Xanthates to Vinyl Epoxides and Related derivatives: A Powerful Tool for the Modular Creation of Quaternary Centres. Angew. Chem. Int. Ed. Eng. 2006, 45, 6520–6523. [Google Scholar] [CrossRef]
- Suzuki, A.; Miyaura, N.; Itoh, M.; Brown, H.C.; Holland, G.W.; Negishi, E.-I. New four-carbon-atom homologation involving the free-radical chain reaction of 1,3-butadiene monoxide with organoboranes. Synthesis of 4-alkyl-2-buten-1-ols from olefins via hydroboration. J. Am. Chem. Soc. 1971, 93, 2792–2793. [Google Scholar]
- Ichinose, Y.; Oshima, K.; Utimoto, K. Et3B-Induced Radical Reaction of 1,3-Diene Monoxide with C6F13I, PhSH, or Ph3GeH. Chem. Lett. 1988, 17, 1437–1440. [Google Scholar] [CrossRef]
- Crich, D.; Mo, X.-S. Free Radical Chemistry of β-Lactones. Arrhenius Parameters for the Decarboxylative Cleavage and Ring Expansion of 2-Oxetanon-4-ylcarbinyl Radicals. Facilitation of Chain Propagation by Catalytic Benzeneselenol. J. Am. Chem. Soc. 1998, 120, 8298–8304. [Google Scholar] [CrossRef]
- Debien, L.; Quiclet-Sire, B.; Zard, S.Z. Allylic Alcohols: Ideal Radical Allylating Agents? Acc. Chem. Res. 2015, 48, 1237–1253. [Google Scholar] [CrossRef] [PubMed]
- Charrier, N.; Quiclet-Sire, B.; Zard, S.Z. Allylic Alcohols as Radical Allylating Agents. An Overall Olefination of Aldehydes and Ketones. J. Am. Chem. Soc. 2008, 130, 8898–8899. [Google Scholar] [CrossRef] [PubMed]
- Michalland, J.; Zard, S.Z. A Convergent, Stereoselective Route to Trisubstituted Alkenyl Boronates. Org. Lett. 2021, 23, 8018–8022. [Google Scholar] [CrossRef]
- Braun, M.-G.; Quiclet-Sire, B.; Zard, S.Z. A Highly Stereoselective, Modular Route to (E)-Vinylsulfones and to (Z)- and (E)-Alkenes. J. Am. Chem. Soc. 2011, 133, 15954–15957. [Google Scholar] [CrossRef] [PubMed]
- Debien, L.; Quiclet-Sire, B.; Zard, S.Z. A Modular Approach to Saturated and α, β-Unsaturated Ketones. Org. Lett. 2011, 13, 5676–5679. [Google Scholar] [CrossRef] [PubMed]
- Charrier, N.; Zard, S.Z. Radical Allylation with α-Branched Allyl Sulfones. Angew. Chem. Int. Ed. 2008, 47, 9443–9446. [Google Scholar] [CrossRef]
- Anthore, L.; Zard, S.Z. A Convergent Radical Based Route to Trifluoromethyl Ketones and to α, β–Unsaturated Trifluoromethyl Ketones. Org. Lett. 2015, 17, 3058–3061. [Google Scholar] [CrossRef]
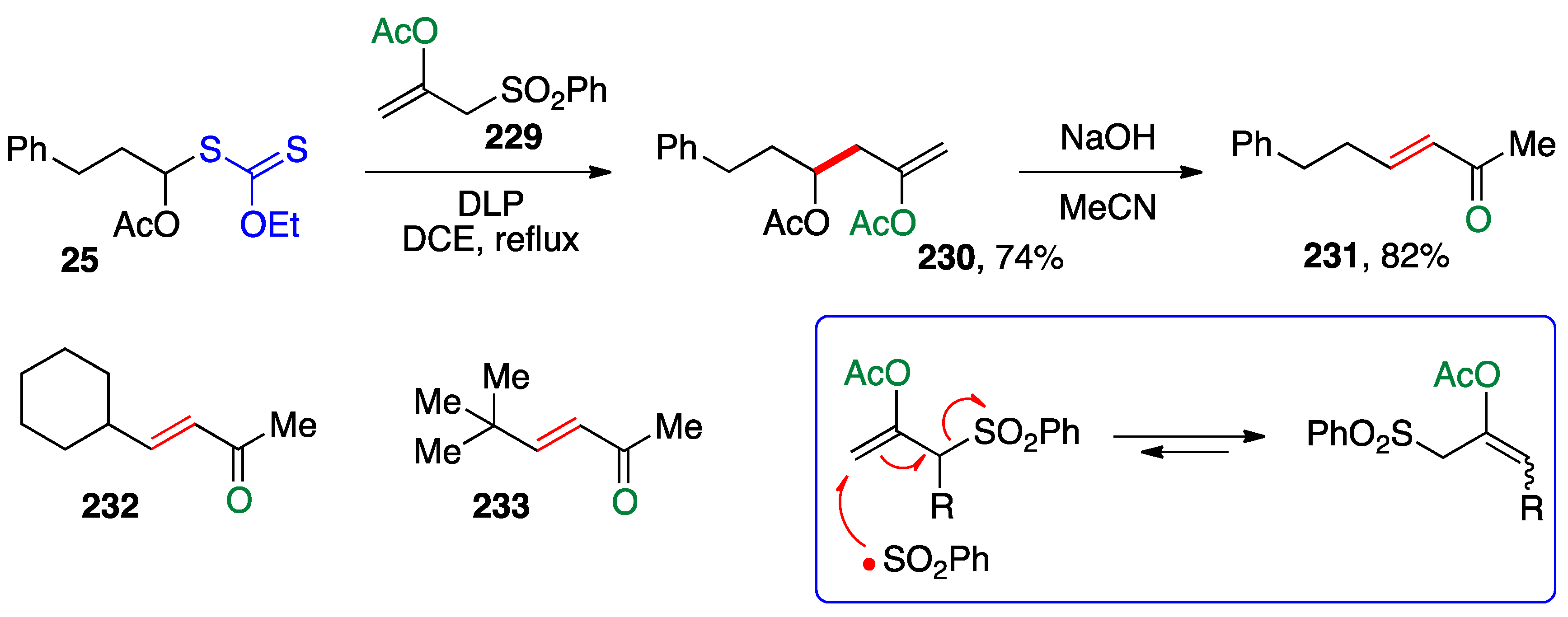
- Quiclet-Sire, B.; Zard, S.Z. Xanthates and Vinyl Esters, a Remarkably Powerful Alliance. Heterocycles 2019, 99, 742–765. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



