Submitted:
18 October 2023
Posted:
19 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Parasite and Mammalian Cell Maintenance
2.2. T7Cas9 Line Generation
2.3. Endogenous C-terminus Tagging by CRISPR/Cas9 Editing
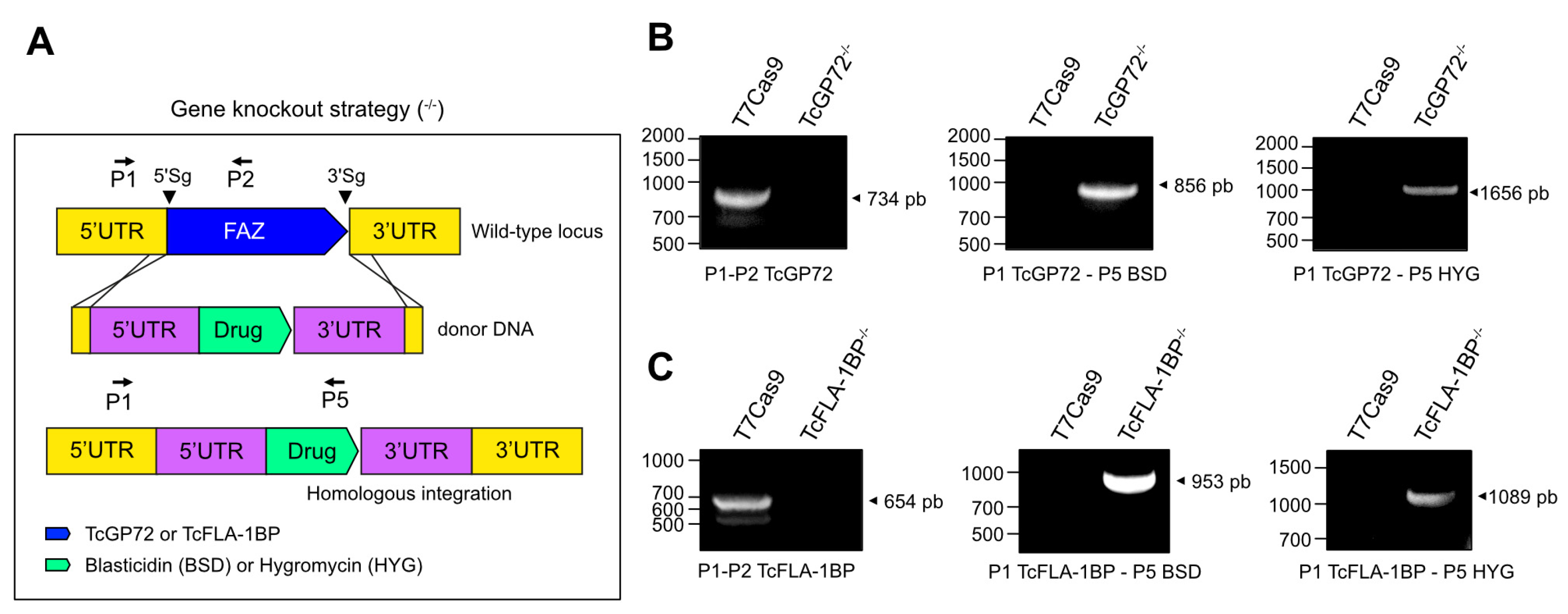
2.4. Generation of TcGP72-/- and TcFLA-1BP-/- Double-Knockout Cells via CRISPR/Cas9 Editing and Genotyping
2.5. Western Blotting
2.6. Flow Cytometry of Fluorescent Parasites
2.7. Localization of Protein Tags In Vivo and Immunofluorescence Microscopy
2.8. Cell Cycle Analysis
2.9. Ultrastructural Analysis
2.10. Growth Curve
2.11. Morphometric and Statistical Analyses
2.12. Genes IDs
3. Results
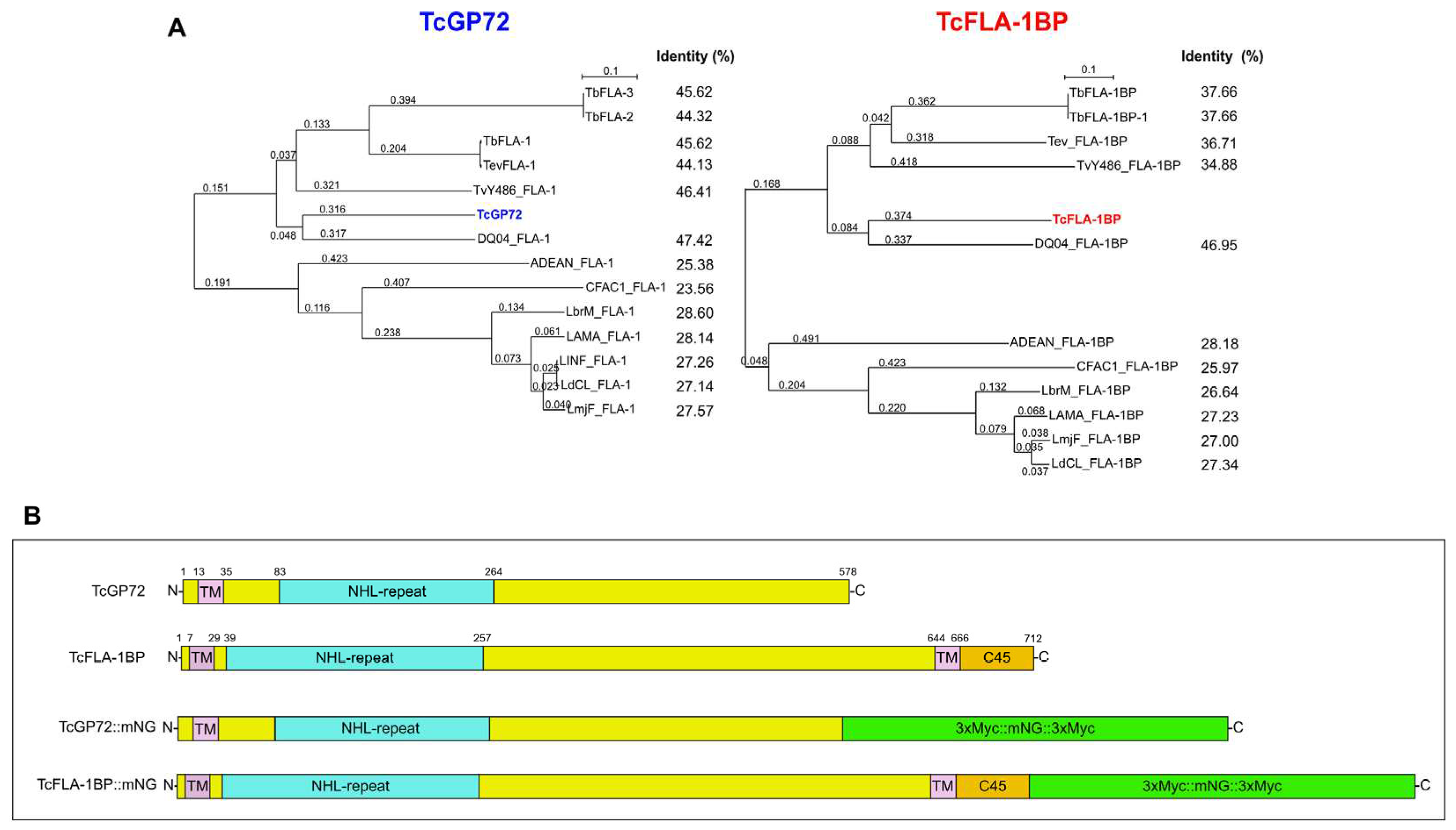
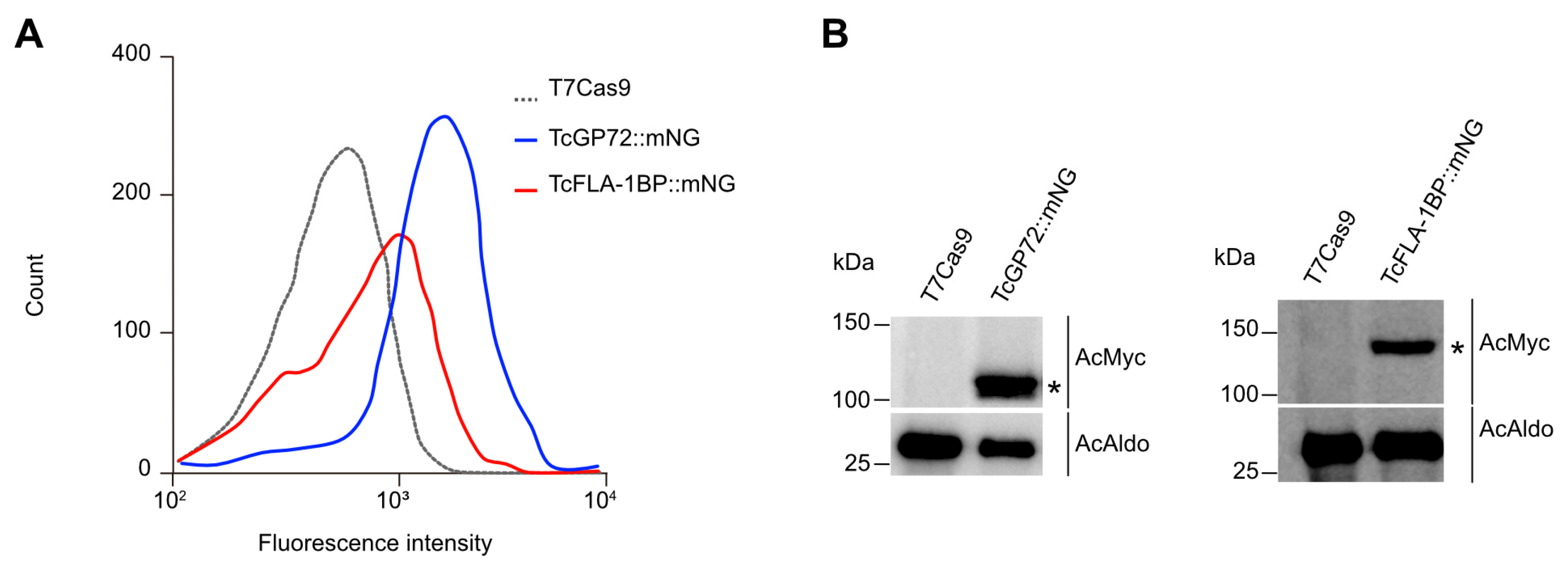
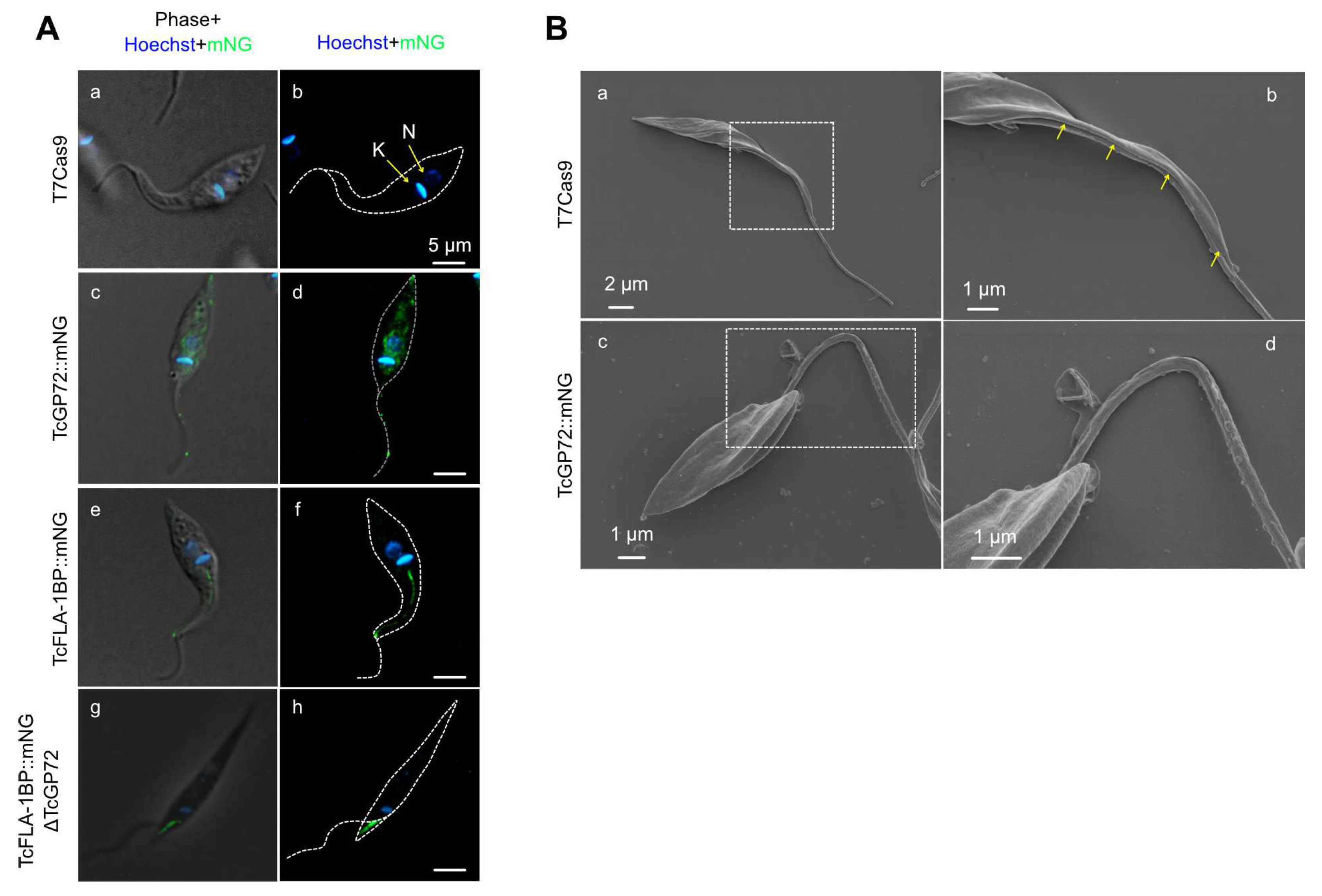
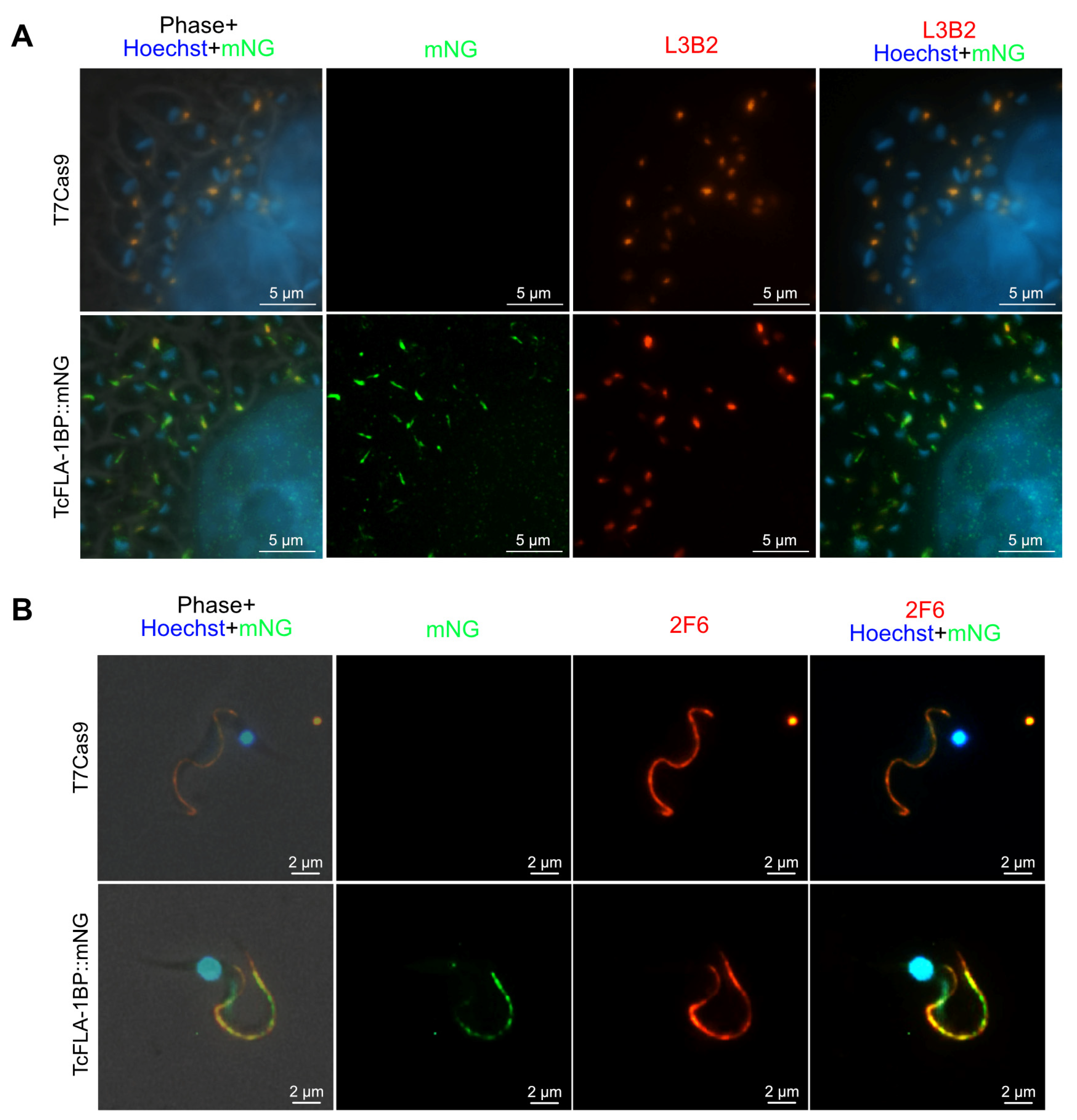
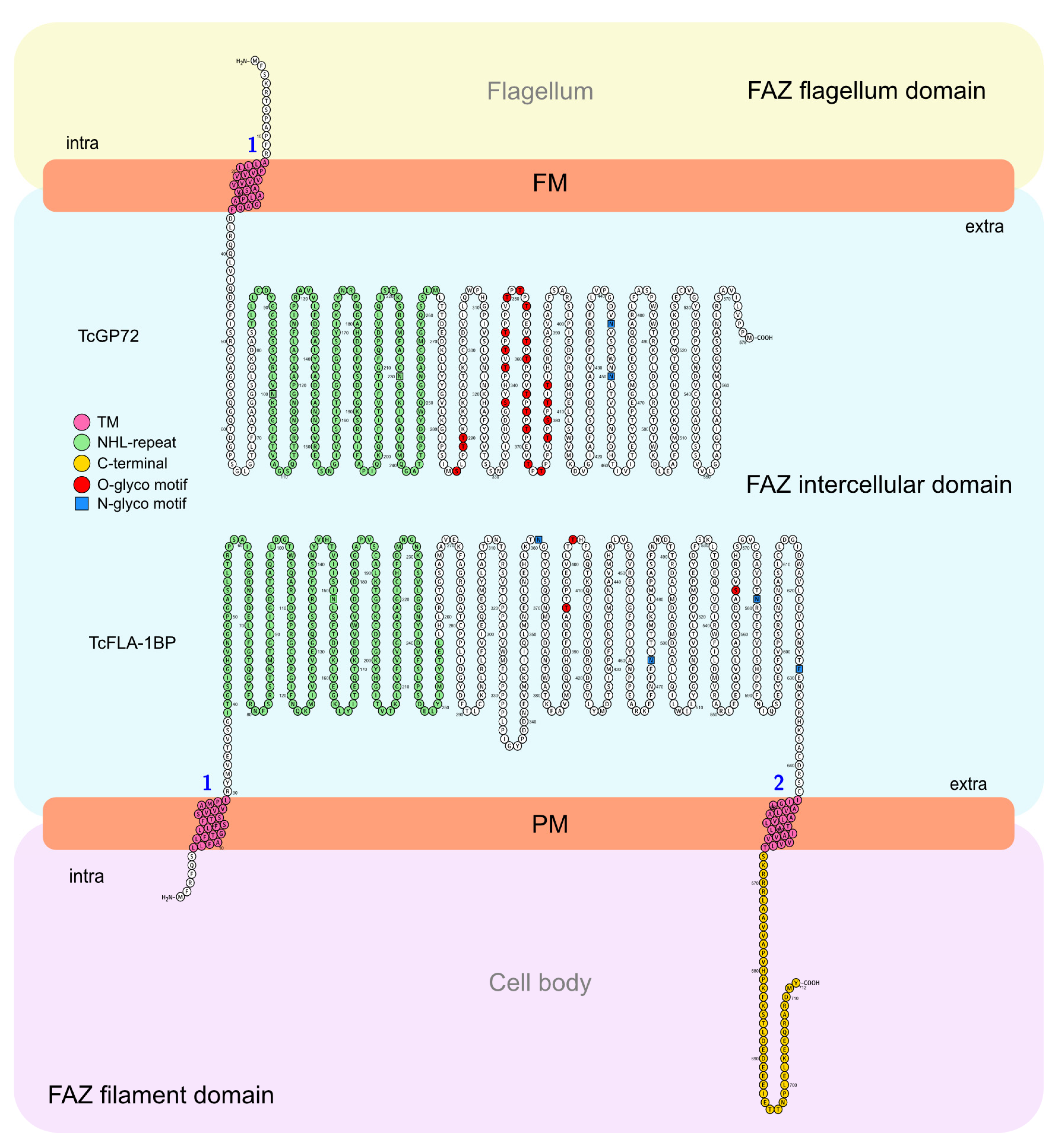
3.1. In Silico Analysis and Protein Localization of TcGP72 and TcFLA-1BP
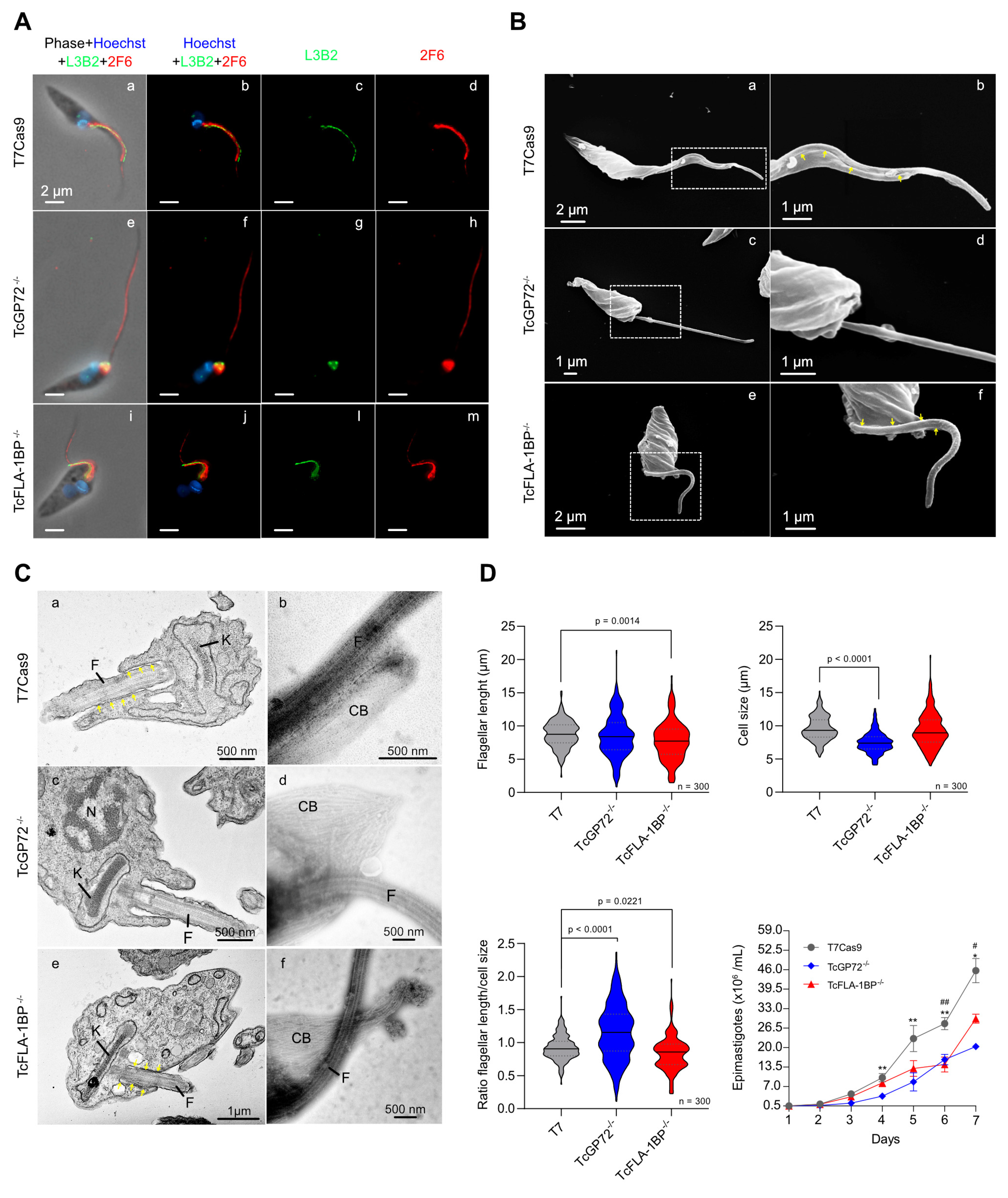
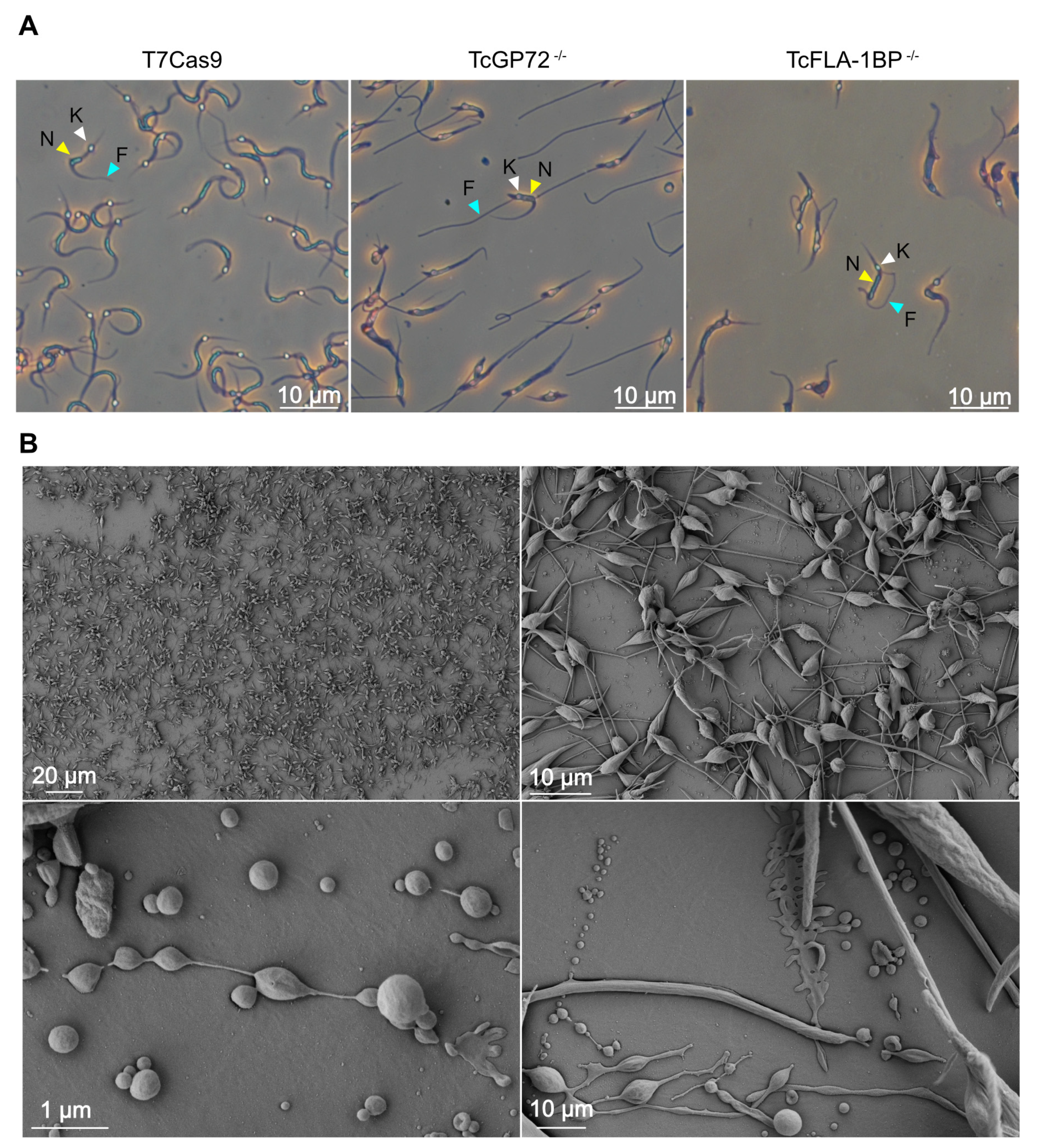
3.2. Knockout of TcGP72 and TcFLA-1BP Affects T. cruzi Cell Morphology and Cell Cycle Progression of Epimastigotes
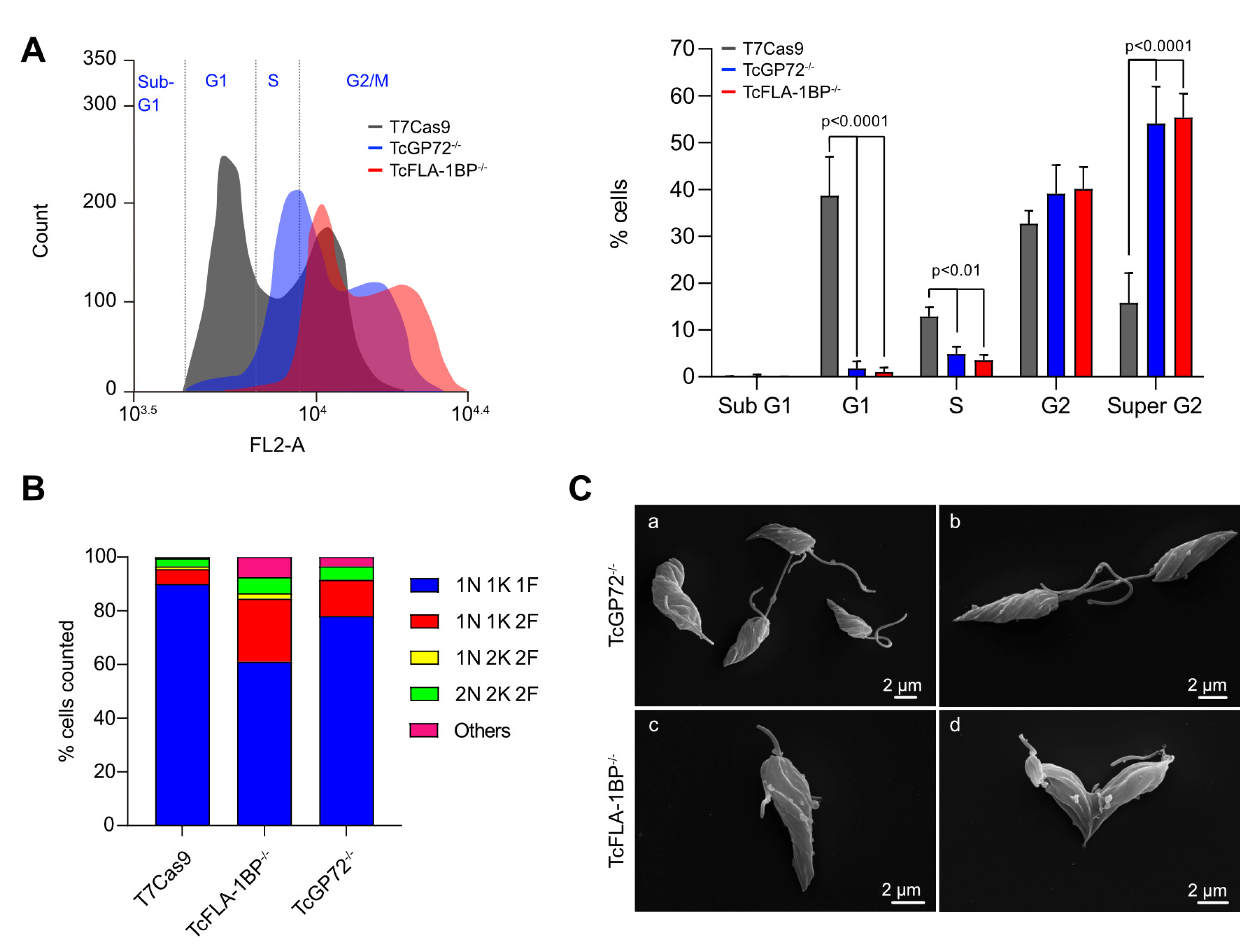
3.3. Deletion of TcGP72 and TcFLA-1BP Alters Cell Cycle Progression
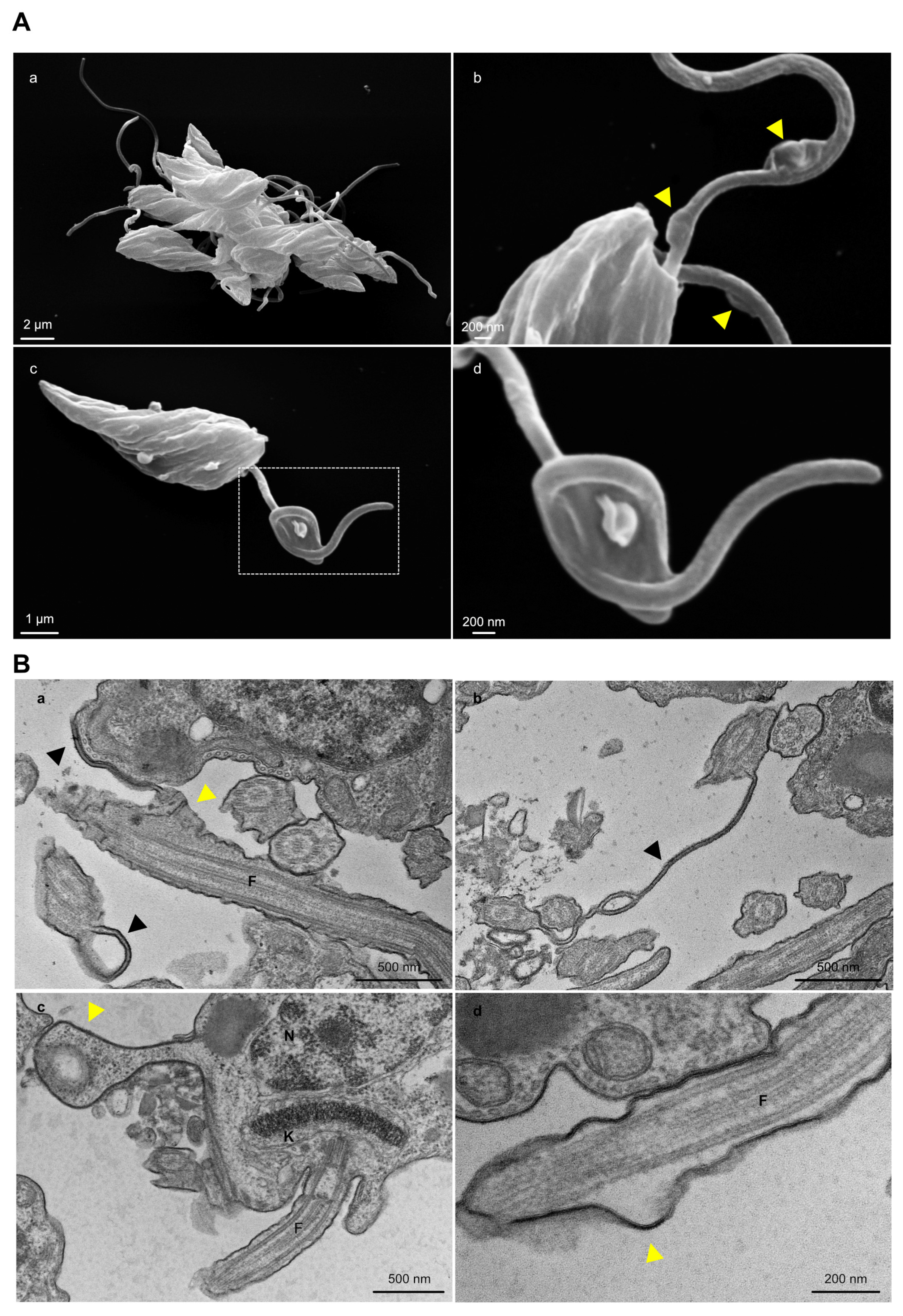
3.4. Knockout of TcGP72 and TcFLA-1BP Affects T. cruzi Cell Morphology during Metacyclogenesis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brener, Z. Biology of Trypanosoma Cruzi. Annu Rev Microbiol 1973, 27, 347–382. [Google Scholar] [CrossRef]
- Burleigh, B.A.; Andrews, N.W. The Mechanisms of Trypanosoma Cruzi Invasion of Mammalian Cells. Annu Rev Microbiol 1995, 49, 175–200. [Google Scholar] [CrossRef]
- De Souza, W. Cell Biology of Trypanosoma Cruzi. In International Review of Cytology; 1984; Vol. 86, pp. 197–283.
- Fernandes, M.C.; Andrews, N.W. Host Cell Invasion by Trypanosoma Cruzi: A Unique Strategy That Promotes Persistence. FEMS Microbiol Rev 2012, 36, 734–747. [Google Scholar] [CrossRef] [PubMed]
- Moretti, N.S.; Mortara, R.A.; Schenkman, S. Trypanosoma Cruzi. Trends Parasitol 2020, 36, 404–405. [Google Scholar] [CrossRef]
- Zuma, A.A.; dos Santos Barrias, E.; de Souza, W. Basic Biology of Trypanosoma Cruzi. Curr Pharm Des 2021, 27, 1671–1732. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, D.E.; Benchimol, M.; Crepaldi, P.H.; de Souza, W. Interactive Multimedia to Teach the Life Cycle of Trypanosoma Cruzi, the Causative Agent of Chagas Disease. PLoS Negl Trop Dis 2012, 6, e1749. [Google Scholar] [CrossRef]
- Vidal, J.C.; De Souza, W. 3D FIB-SEM Structural Insights into the Architecture of Sub-Pellicular Microtubules of Trypanosoma Cruzi Epimastigotes. Biol Cell 2022, 114, 203–210. [Google Scholar] [CrossRef]
- Ávila, A.R.; Dallagiovanna, B.; Yamada-Ogatta, S.F.; Monteiro-Góes, V.; Fragoso, S.P.; Krieger, M.A.; Goldenberg, S. Stage-Specific Gene Expression during Trypanosoma Cruzi Metacyclogenesis. Genetics and Molecular Research 2003, 2, 159–168. [Google Scholar]
- Li, Y.; Shah-Simpson, S.; Okrah, K.; Belew, A.T.; Choi, J.; Caradonna, K.L.; Padmanabhan, P.; Ndegwa, D.M.; Temanni, M.R.; Corrada Bravo, H.; et al. Transcriptome Remodeling in Trypanosoma Cruzi and Human Cells during Intracellular Infection. PLoS Pathog 2016, 12, e1005511. [Google Scholar] [CrossRef] [PubMed]
- Minning, T.A.; Weatherly, D.B.; Atwood, J.; Orlando, R.; Tarleton, R.L. The Steady-State Transcriptome of the Four Major Life-Cycle Stages of Trypanosoma Cruzi. BMC Genomics 2009, 10, 370. [Google Scholar] [CrossRef]
- Ballesteros-Rodea, G.; Santillán, M.; Martínez-Calvillo, S.; Manning-Cela, R. Flagellar Motility of Trypanosoma Cruzi Epimastigotes. J Biomed Biotechnol 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Bastin, P.; Sherwin, T.; Gull, K. Paraflagellar Rod Is Vital for Trypanosome Motility. Nature 1998, 391, 548–548. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.L. Biology and Mechanism of Trypanosome Cell Motility. Eukaryot Cell 2003, 2, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Rocha, G.M.; Brandão, B.A.; Mortara, R.A.; Attias, M.; de Souza, W.; Carvalho, T.M.U. The Flagellar Attachment Zone of Trypanosoma Cruzi Epimastigote Forms. J Struct Biol 2006, 154, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Bastin, P.; Pullen, T.J.; Moreira-Leite, F.F.; Gull, K. Inside and Outside of the Trypanosome Flagellum:A Multifunctional Organelle. Microbes Infect 2000, 2, 1865–1874. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, R.J.; Sunter, J.D.; Gull, K. Flagellar Pocket Restructuring through the Leishmania Life Cycle Involves a Discrete Flagellum Attachment Zone. J Cell Sci 2016, 854–867. [Google Scholar] [CrossRef]
- Alcantara, C. de L.; de Souza, W.; da Cunha e Silva, N.L. Tridimensional Electron Microscopy Analysis of the Early Endosomes and Endocytic Traffic in Trypanosoma Cruzi Epimastigotes. Protist 2018, 169, 887–910. [Google Scholar] [CrossRef]
- De Souza, W.; Martinez-Palomo, A.; Gonzalez-Robles, A. The Cell Surface of Trypanosoma Cruzi: Cytochemistry and Freeze-Fracture. J Cell Sci 1978, 33, 285–299. [Google Scholar] [CrossRef]
- Martínez-Palomo, A.; DeSouza, W.; Gonzalez-Robles, A. Topographical Differences in the Distribution of Surface Coat Components and Intramembrane Particles. A Cytochemical and Freeze-Fracture Study in Culture Forms of Trypanosoma Cruzi. Journal of Cell Biology 1976, 69, 507–513. [Google Scholar] [CrossRef]
- Pimenta, P.F.; de Souza, W.; Souto-Padrón, T.; Pinto da Silva, P. The Cell Surface of Trypanosoma Cruzi: A Fracture-Flip, Replica-Staining Label-Fracture Survey. Eur J Cell Biol 1989, 50, 263–271. [Google Scholar]
- Vickerman, K. On The Surface Coat and Flagellar Adhesion in Trypanosomes. J Cell Sci 1969, 5, 163–193. [Google Scholar] [CrossRef]
- Kohl, L.; Sherwin, T.; Gull, K. Assembly of the Paraflagellar Rod and the Flagellum Attachment Zone Complex During the Trypanosoma Brucei Cell Cycle. J Eukaryot Microbiol 1999, 46, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Rotureau, B.; Blisnick, T.; Subota, I.; Julkowska, D.; Cayet, N.; Perrot, S.; Bastin, P. Flagellar Adhesion in Trypanosoma Brucei Relies on Interactions between Different Skeletal Structures in the Flagellum and Cell Body. J Cell Sci 2013, 204–215. [Google Scholar] [CrossRef]
- Sunter, J.D.; Gull, K. The Flagellum Attachment Zone: ‘The Cellular Ruler’ of Trypanosome Morphology. Trends Parasitol 2016, 32, 309–324. [Google Scholar] [CrossRef]
- Woodward, R.; Carden, M.J.; Gull, K. Immunological Characterization of Cytoskeletal Proteins Associated with the Basal Body, Axoneme and Flagellum Attachment Zone of Trypanosoma Brucei. Parasitology 1995, 111, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Y.; Wang, C.; Yuan, Y.A.; He, C.Y. An Intracellular Membrane Junction Consisting of Flagellum Adhesion Glycoproteins Links Flagellum Biogenesis to Cell Morphogenesis in Trypanosoma Brucei. J Cell Sci 2013, 126, 520–531. [Google Scholar] [CrossRef]
- Sunter, J.D.; Varga, V.; Dean, S.; Gull, K. A Dynamic Coordination of Flagellum and Cytoplasmic Cytoskeleton Assembly Specifies Cell Morphogenesis in Trypanosomes. J Cell Sci 2015, 128, 1580–1594. [Google Scholar] [CrossRef]
- de Jesus, A.R.; Cooper, R.; Espinosa, M.; Gomes, J.E.; Garcia, E.S.; Paul, S.; Cross, G.A. Gene Deletion Suggests a Role for Trypanosoma Cruzi Surface Glycoprotein GP72 in the Insect and Mammalian Stages of the Life Cycle. J Cell Sci 1993, 106, 1023–1033. [Google Scholar] [CrossRef]
- Basombrío, M.A.; Segura, M.A.; Nasser, J.R. Relationship between Long-Term Resistance to Trypanosoma Cruzi and Latent Infection, Examined by Antibody Production and Polymerase Chain Reaction in Mice. J Parasitol 2002, 88, 1107–1112. [Google Scholar] [CrossRef]
- LaCount, D.J.; Barrett, B.; Donelson, J.E. Trypanosoma Brucei FLA1 Is Required for Flagellum Attachment and Cytokinesis. Journal of Biological Chemistry 2002, 277, 17580–17588. [Google Scholar] [CrossRef]
- CAMARGO, E.P. Growth and Differentiation in Trypanosoma Cruzi. I. Origin of Metacyclic Trypanosomes in Liquid Media. Rev Inst Med Trop Sao Paulo 1964, 6, 93–100. [Google Scholar]
- Contreras, V.T.; Salles, J.M.; Thomas, N.; Morel, C.M.; Goldenberg, S. In Vitro Differentiation of Trypanosoma Cruzi under Chemically Defined Conditions. Mol Biochem Parasitol 1985, 16, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.M.G.; Yoshida, N. Stage-Specific Surface Antigens of Metacyclic Trypomastigotes of Trypanosoma Cruzi Identified by Monoclonal Antibodies. Mol Biochem Parasitol 1986, 18, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Lugo, L.; Díaz-Olmos, Y.; Sáenz-García, J.; Probst, C.M.; DaRocha, W.D. Effective Gene Delivery to Trypanosoma Cruzi Epimastigotes through Nucleofection. Parasitol Int 2017, 66, 236–239. [Google Scholar] [CrossRef]
- Alves, A.A.; Alcantara, C.L.; Dantas-Jr, M.V.A.; Sunter, J.D.; De Souza, W.; Cunha-e-Silva, N.L. Dynamics of the Orphan Myosin MyoF over Trypanosoma Cruzi Life Cycle and along the Endocytic Pathway. Parasitol Int 2022, 86, 102444. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.C.; Francisco, A.F.; Jayawardhana, S.; Calderano, S.G.; Lewis, M.D.; Olmo, F.; Beneke, T.; Gluenz, E.; Sunter, J.; Dean, S.; et al. Expanding the Toolbox for Trypanosoma Cruzi: A Parasite Line Incorporating a Bioluminescence-Fluorescence Dual Reporter and Streamlined CRISPR/Cas9 Functionality for Rapid in Vivo Localisation and Phenotyping. PLoS Negl Trop Dis 2018, 12, e0006388. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.A.; Alcantara, C.L.; Dantas-Jr, M.V.A.; Sunter, J.D.; De Souza, W.; Cunha-e-Silva, N.L. Dynamics of the Orphan Myosin MyoF over Trypanosoma Cruzi Life Cycle and along the Endocytic Pathway. Parasitol Int 2022, 86. [Google Scholar] [CrossRef] [PubMed]
- Beneke, T.; Gluenz, E. LeishGEdit: A Method for Rapid Gene Knockout and Tagging Using CRISPR-Cas9. In Methods in Molecular Biology; Humana Press Inc., 2019; Vol. 1971, pp. 189–210.
- Barbosa Leite, A.; Severo Gomes, A.A.; de Castro Nascimento Sousa, A.C.; Roberto de Mattos Fontes, M.; Schenkman, S.; Silvio Moretti, N. Effect of Lysine Acetylation on the Regulation of Trypanosoma Brucei Glycosomal Aldolase Activity. Biochemical Journal 2020, 477, 1733–1744. [Google Scholar] [CrossRef] [PubMed]
- Castro Machado, F.; Bittencourt-Cunha, P.; Malvezzi, A.M.; Arico, M.; Radio, S.; Smircich, P.; Zoltner, M.; Field, M.C.; Schenkman, S. <scp>EIF2α</Scp> Phosphorylation Is Regulated in Intracellular Amastigotes for the Generation of Infective Trypanosoma Cruzi Trypomastigote Forms. Cell Microbiol 2020, 22. [Google Scholar] [CrossRef]
- Vaughan, S.; Kohl, L.; Ngai, I.; Wheeler, R.J.; Gull, K. A Repetitive Protein Essential for the Flagellum Attachment Zone Filament Structure and Function in Trypanosoma Brucei. Protist 2008, 159, 127–136. [Google Scholar] [CrossRef]
- Willingham, M.C.; Rutherford, A. V The Use of Osmium-Thiocarbohydrazide-Osmium (OTO) and Ferrocyanide-Reduced Osmium Methods to Enhance Membrane Contrast and Preservation in Cultured Cells. Journal of Histochemistry & Cytochemistry 1984, 32, 455–460. [Google Scholar] [CrossRef]
- Haynes, P.A.; Russell, D.G.; Cross, G.A. Subcellular Localization of Trypanosoma Cruzi Glycoprotein Gp72. J Cell Sci 1996, 109 (Pt 13) Pt 13, 2979–2988. [Google Scholar] [CrossRef]
- Wheeler, R.J.; Gull, K.; Sunter, J.D. Coordination of the Cell Cycle in Trypanosomes. Annu Rev Microbiol 2019, 73, 133–154. [Google Scholar] [CrossRef] [PubMed]
- Meyer, H.; De Souza, W. Electron Microscopic Study of Trypanosoma Cruzi Periplast in Tissue Cultures. I. Number and Arrangement of the Peripheral Microtubules in the Various Forms of the Parasite’s Life Cycle. J Protozool 1976, 23, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.R.; Sherwin, T.; Ploubidou, A.; Byard, E.H.; Gull, K. Microtubule Polarity and Dynamics in the Control of Organelle Positioning, Segregation, and Cytokinesis in the Trypanosome Cell Cycle. Journal of Cell Biology 1995, 128, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Barrias, E.; Reignault, L.; de Carvalho, T.M.U.; de Souza, W. Clathrin Coated Pit Dependent Pathway for Trypanosoma Cruzi Internalization into Host Cells. Acta Trop 2019, 199, 105057. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, C.S.; Ávila, A.R.; de Souza, W.; Motta, M.C.M.; Cavalcanti, D.P. Revisiting the Trypanosoma Cruzi Metacyclogenesis: Morphological and Ultrastructural Analyses during Cell Differentiation. Parasit Vectors 2018, 11, 83. [Google Scholar] [CrossRef]
- Engstler, M.; Pfohl, T.; Herminghaus, S.; Boshart, M.; Wiegertjes, G.; Heddergott, N.; Overath, P. Hydrodynamic Flow-Mediated Protein Sorting on the Cell Surface of Trypanosomes. Cell 2007, 131, 505–515. [Google Scholar] [CrossRef]
- Lander, N.; Cruz-Bustos, T.; Docampo, R. A CRISPR/Cas9-Riboswitch-Based Method for Downregulation of Gene Expression in Trypanosoma Cruzi. Front Cell Infect Microbiol 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Cooper, R.; Inverso, J.A.; Espinosa, M.; Nogueira, N.; Cross, G.A.M. Characterization of a Candidate Gene for GP72, an Insect Stage-Specific Antigen OfTrypanosoma Cruzi. Mol Biochem Parasitol 1991, 49, 45–59. [Google Scholar] [CrossRef]
- Snary, D.; Ferguson, M.A.J.; Scott, M.T.; Allen, A.K. Cell Surface Antigens of Trypanosoma Cruzi: Use of Monoclonal Antibodies to Identify and Isolate an Epimastigote Specific Glycoprotein. Mol Biochem Parasitol 1981, 3, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Absalon, S.; Blisnick, T.; Kohl, L.; Toutirais, G.; Doré, G.; Julkowska, D.; Tavenet, A.; Bastin, P. Intraflagellar Transport and Functional Analysis of Genes Required for Flagellum Formation in Trypanosomes. Mol Biol Cell 2008, 19, 929–944. [Google Scholar] [CrossRef] [PubMed]
- Davidge, J.A.; Chambers, E.; Dickinson, H.A.; Towers, K.; Ginger, M.L.; McKean, P.G.; Gull, K. Trypanosome IFT Mutants Provide Insight into the Motor Location for Mobility of the Flagella Connector and Flagellar Membrane Formation. J Cell Sci 2006, 119, 3935–3943. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



