Submitted:
12 October 2023
Posted:
13 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- S.Z. Rogovina, C. V Alexanyan, E. V Prut, Biodegradable blends based on chitin and chitosan: Production, structure, and properties, J. Appl. Polym. Sci. (2011). [CrossRef]
- K. Guillén-Carvajal, B. Valdez-Salas, E. Beltrán-Partida, J. Salomón-Carlos, N. Cheng, Chitosan, Gelatin, and Collagen Hydrogels for Bone Regeneration., Polymers (Basel). 15 (2023) 2762. [CrossRef]
- C. Chen, D. Li, H. Yano, K. Abe, Insect Cuticle-Mimetic Hydrogels with High Mechanical Properties Achieved via the Combination of Chitin Nanofiber and Gelatin, J. Agric. Food Chem. (2019). [CrossRef]
- S. Ifuku, M. Nogi, K. Abe, K. Abe, M. Yoshioka, M. Morimoto, H. Saimoto, H. Yano, Preparation of Chitin Nanofibers with a Uniform Width as α-Chitin from Crab Shells, Biomacromolecules. (2009). [CrossRef]
- H. Merzendorfer, The cellular basis of chitin synthesis in fungi and insects: Common principles and differences, Eur. J. Cell Biol. 90 (2011) 759–769. [CrossRef]
- C.K.S. Pillai, W. Paul, C.P. Sharma, Chitin and chitosan polymers: Chemistry, solubility and fiber formation, Prog. Polym. Sci. 34 (2009) 641–678. [CrossRef]
- R. Jayakumar, M. Prabaharan, S. V. Nair, H. Tamura, Novel chitin and chitosan nanofibers in biomedical applications, Biotechnol. Adv. 28 (2010) 142–150. [CrossRef]
- M.N.V.R. Kumar, A review of chitin and chitosan applications, React. Funct. Polym. (2000). [CrossRef]
- D. Elieh-Ali-Komi, M.R. Hamblin, Chitin and Chitosan: Production and Application of Versatile Biomedical Nanomaterials., Int. J. Adv. Res. 4 (2016) 411–427.
- B.-M. Min, S.W. Lee, S.W. Lee, J.N. Lim, Y. You, T.S. Lee, P.H. Kang, W.H. Park, W.H. Park, W.H. Park, Chitin and chitosan nanofibers: electrospinning of chitin and deacetylation of chitin nanofibers, Polymer (Guildf). (2004). [CrossRef]
- X. Xu, Chitin Nanocomposite Scaffolds for Advanced Medications, (2019). https://www.semanticscholar.org/paper/e9e771646911ba6c93e4231b8cddb33cebe1447b.
- S.K. Arya, M. Manohar, G. Singh, W.A. Siddiqui, Chitin and Chitosan-Complexes and Their Applications, Chitosan. (2017) 151–165. [CrossRef]
- S. Kumar, R. Dhiman, C.R. Prudencio, A.C. da Costa, A. Vibhuti, E. Leal, C.-M. Chang, V.S. Raj, R.P. Pandey, Chitosan: Applications in Drug Delivery System, Mini-Reviews Med. Chem. 23 (2022) 187–191. [CrossRef]
- D. Gheorghiță, H. Moldovan, A. Robu, A.-I. Bița, E. Grosu, A. Antoniac, I. Corneschi, I. Antoniac, A.D. Bodog, C.I. Băcilă, Chitosan-Based Biomaterials for Hemostatic Applications: A Review of Recent Advances., Int. J. Mol. Sci. 24 (2023) 10540. [CrossRef]
- N. Isobe, Y. Kaku, S. Okada, S. Kawada, K. Tanaka, Y. Fujiwara, R. Nakajima, D. Bissessur, C. Chen, Identification of Chitin Allomorphs in Poorly Crystalline Samples Based on the Complexation with Ethylenediamine, Biomacromolecules. 23 (2022) 4220–4229. [CrossRef]
- Z. Yu, Z.P. Xu, D. Lau, Effect of Acidity on Chitin–Protein Interface: A Molecular Dynamics Study, J. Bionanoscience. (2014). [CrossRef]
- K. Jin, X.-Q. Feng, Z.P. Xu, Mechanical Properties of Chitin–Protein Interfaces: A Molecular Dynamics Study, J. Bionanoscience. (2013). [CrossRef]
- M. Hudek, K. Kubiak-Ossowska, K. Johnston, V. Ferro, P. Mulheran, Chitin and Chitosan Binding to the α-Chitin Crystal: A Molecular Dynamics Study, ACS Omega. (2023). [CrossRef]
- M. Petrov, L. Lymperakis, M. Friák, J. Neugebauer, Ab Initio Based conformational study of the crystalline α-chitin, Biopolymers. (2013). [CrossRef]
- M. Haneef, L. Ceseracciu, C. Canale, I.S. Bayer, J.A. Heredia-Guerrero, A. Athanassiou, Advanced Materials From Fungal Mycelium: Fabrication and Tuning of Physical Properties., Sci. Rep. (2017). [CrossRef]
- D. Raabe, C. Sachs, P. Romano, The crustacean exoskeleton as an example of a structurally and mechanically graded biological nanocomposite material, Acta Mater. (2005). [CrossRef]
- M.M. Abo Elsoud, E.M. El Kady, Current trends in fungal biosynthesis of chitin and chitosan, Bull. Natl. Res. Cent. 2019 431. 43 (2019) 1–12. [CrossRef]
- L. Hartl, S. Zach, V. Seidl-Seiboth, Fungal chitinases: diversity, mechanistic properties and biotechnological potential, Appl. Microbiol. Biotechnol. 93 (2012) 533. [CrossRef]
- Z. Xu, X. Lv, J. Chen, L. Jiang, Y. Lai, J. Li, DFT investigation of capacious, ultrafast and highly conductive hexagonal Cr 2 C and V 2 C monolayers as anode materials for high-performance lithium-ion batteries, Phys. Chem. Chem. Phys. 19 (2017) 7807–7819. [CrossRef]
- Wei, A. Wei, J. Fu, F. Guo, F. Guo, F. Guo, Z. Střelcová, P. Kulhánek, M. Friák, H.-O. Fabritius, M. Petrov, J. Neugebauer, J. Koča, Z. Yu, D. Lau, S.Z. Rogovina, C. V Alexanyan, E. V Prut, J. Cui, Z. Yu, D. Lau, F. Ding, H. Deng, Y. Du, X. Shi, Q. Wang, T. Nishino, R. Matsui, K. Nakamae, M. Hudek, K. Kubiak-Ossowska, K. Johnston, V. Ferro, P. Mulheran, M. Ofem, P.A. Ubi, A. Christian, G. Tishchenko, P. Morganti, M. Stoller, J. Mikešov, D. Kubies, Effect of Acetyl Group on Mechanical Properties of Chitin/Chitosan Nanocrystal: A Molecular Dynamics Study., J. Mater. Sci. (2016). [CrossRef]
- K. Piekarska, M. Sikora, M. Owczarek, J. Jóźwik-Pruska, M. Wiśniewska-Wrona, Chitin and Chitosan as Polymers of the Future—Obtaining, Modification, Life Cycle Assessment and Main Directions of Application, Polym. 2023, Vol. 15, Page 793. 15 (2023) 793. [CrossRef]
- B. Mortazavi, G. Cuniberti, Atomistic modeling of mechanical properties of polycrystalline graphene, Nanotechnology. 25 (2014). [CrossRef]
- B. Mortazavi, A Theoretical Investigation of the Structural, Electronic and Mechanical Properties of Pristine and Nitrogen-Terminated Carbon Nanoribbons Composed of 4–5–6–8-Membered Rings, J. Compos. Sci. 7 (2023) 269. [CrossRef]
- M.-B. Coltelli, M.B. Coltelli, P. Cinelli, V. Gigante, L. Aliotta, P. Morganti, L. Panariello, A. Lazzeri, Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties, Int. J. Mol. Sci. (2019). [CrossRef]
- S. Nikolov, M. Petrov, L. Lymperakis, M. Friák, C. Sachs, H.-O. Fabritius, D. Raabe, J. Neugebauer, Revealing the Design Principles of High-Performance Biological Composites Using Ab initio and Multiscale Simulations: The Example of Lobster Cuticle, Adv. Mater. 22 (2010) 519–526. [CrossRef]
- K.J. Narayana, R.G. Burela, Multi-scale modeling and simulation of natural fiber reinforced composites (Bio-composites), J. Phys. Conf. Ser. 1240 (2019) 012103. [CrossRef]
- A. Wei, A. Wei, J. Fu, F. Guo, F. Guo, F. Guo, Mechanical properties of chitin polymorphs: A computational study, J. Mater. Sci. (2021). [CrossRef]
- R.R. Faria, R.F. Guerra, L.R. De Sousa Neto, L.F. Motta, E.D.F. Franca, Computational study of polymorphic structures of α- and β- chitin and chitosan in aqueous solution, J. Mol. Graph. Model. 63 (2016) 78–84. [CrossRef]
- S. Plimpton, Fast parallel algorithms for short-range molecular dynamics, J. Comput. Phys. 117 (1995) 1–19. [CrossRef]
- O. Guvench, S.S. Mallajosyula, E.P. Raman, E. Hatcher, K. Vanommeslaeghe, T.J. Foster, F.W. Jamison, A.D. MacKerell, CHARMM additive all-atom force field for carbohydrate derivatives and its utility in polysaccharide and carbohydrate-protein modeling, J. Chem. Theory Comput. 7 (2011) 3162–3180. [CrossRef]
- A.D. MacKerell, E.P. Raman, O. Guvench, CHARMM additive all-atom force field for glycosidic linkages in carbohydrates involving furanoses, J. Phys. Chem. B. 114 (2010) 12981–12994. [CrossRef]
- O. Guvench, E. Hatcher, R.M. Venable, R.W. Pastor, A.D. MacKerell, CHARMM additive all-atom force field for glycosidic linkages between hexopyranoses, J. Chem. Theory Comput. 5 (2009) 2353–2370. [CrossRef]
- H.B. Lee, W. Cai, Ewald Summation for Coulomb Interactions in a Periodic Supercell, (2009).
- G. Marc, W.G. McMillan, The Virial Theorem, (2007) 209–361. [CrossRef]
- A. Stukowski, Visualization and analysis of atomistic simulation data with OVITO-the Open Visualization Tool, Model. Simul. Mater. Sci. Eng. 18 (2010) 015012. [CrossRef]
- P. Sikorski, R. Hori, M. Wada, Revisit of α-chitin crystal structure using high resolution X-ray diffraction data, Biomacromolecules. 10 (2009) 1100–1105. [CrossRef]
- V.L. Deringer, U. Englert, R. Dronskowski, Nature, Strength, and Cooperativity of the Hydrogen-Bonding Network in α-Chitin, Biomacromolecules. 17 (2016) 996–1003. [CrossRef]
- N. V. Smirnova, K.A. Kolbe, E.N. Dresvyanina, S.F. Grebennikov, I.P. Dobrovolskaya, V.E. Yudin, T. Luxbacher, P. Morganti, Effect of Chitin Nanofibrils on Biocompatibility and Bioactivity of the Chitosan-Based Composite Film Matrix Intended for Tissue Engineering, Mater. (Basel, Switzerland). 12 (2019). [CrossRef]
- Y. Liu, R. Liu, J. Shi, R. Zhang, H. Tang, C. Xie, F. Wang, J. Han, L. Jiang, Chitosan/esterified chitin nanofibers nanocomposite films incorporated with rose essential oil: Structure, physicochemical characterization, antioxidant and antibacterial properties, Food Chem. X. 18 (2023). [CrossRef]
- S. Sundar, A.A. Sandilya, M.H. Priya, Unraveling the Influence of Osmolytes on Water Hydrogen-Bond Network: From Local Structure to Graph Theory Analysis, J. Chem. Inf. Model. 61 (2021) 3927–3944. [CrossRef]
- J. Cui, Z. Yu, D. Lau, Effect of Acetyl Group on Mechanical Properties of Chitin/Chitosan Nanocrystal: A Molecular Dynamics Study., Int. J. Mol. Sci. (2016). [CrossRef]
- R. Sulthan, A. Reghunadhan, S. Sambhudevan, A new era of chitin synthesis and dissolution using deep eutectic solvents- comparison with ionic liquids, J. Mol. Liq. 380 (2023) 121794. [CrossRef]
- M.A. P, 4 - Chitin-based nanomaterials, in: S. Kanwar, A. Kumar, T.A. Nguyen, S. Sharma, Y.B.T.-B.N. Slimani (Eds.), Micro Nano Technol., Elsevier, 2021: pp. 61–99. [CrossRef]
- N.E. Mushi, S. Utsel, L.A. Berglund, Nanostructured biocomposite films of high toughness based on native chitin nanofibers and chitosan, Front. Chem. 2 (2014). [CrossRef]
- K. Azuma, S. Ifuku, T. Osaki, Y. Okamoto, S. Minami, Preparation and biomedical applications of chitin and chitosan nanofibers, J. Biomed. Nanotechnol. 10 (2014) 2891–2920. [CrossRef]
- M.A. Jansen, J. Williams, N. Chawla, N.M. Franz, Avoidance of Catastrophic Structural Failure as an Evolutionary Constraint: Biomechanics of the Acorn Weevil Rostrum, Adv. Mater. 31 (2019) 1903526. [CrossRef]
- F. Hou, Z. Gong, F. Jia, W. Cui, S. Song, J. Zhang, Y. Wang, W. Wang, Insights into the relationships of modifying methods, structure, functional properties and applications of chitin: A review, Food Chem. 409 (2023). [CrossRef]
- T.H.E. Kings, “Advanced mechanics of materials” 5th edition, A.P. Boresi, R.J. Schmidt and O.M. Sidebottom, Strain. 29 (1993) 141–142. [CrossRef]
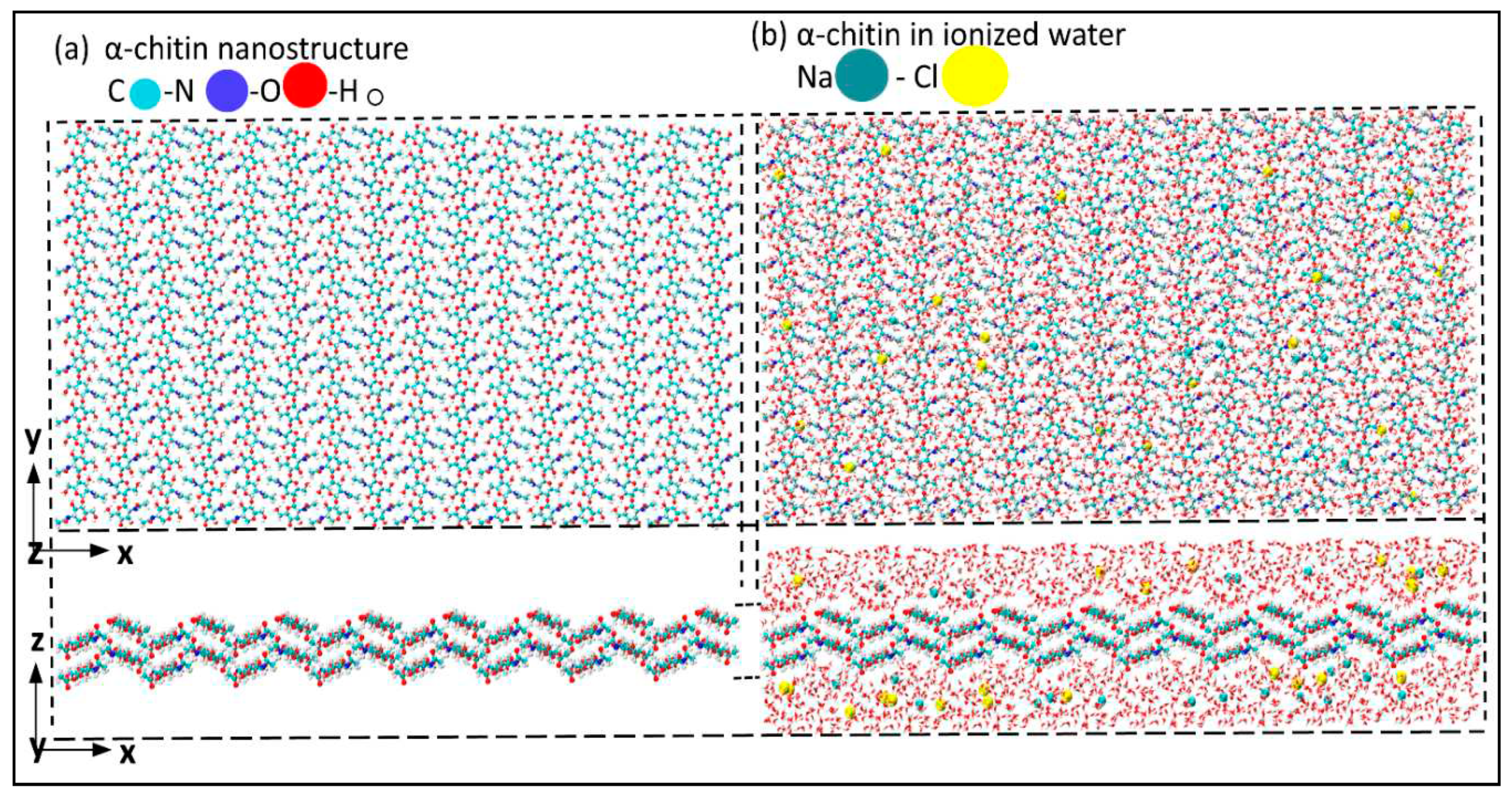
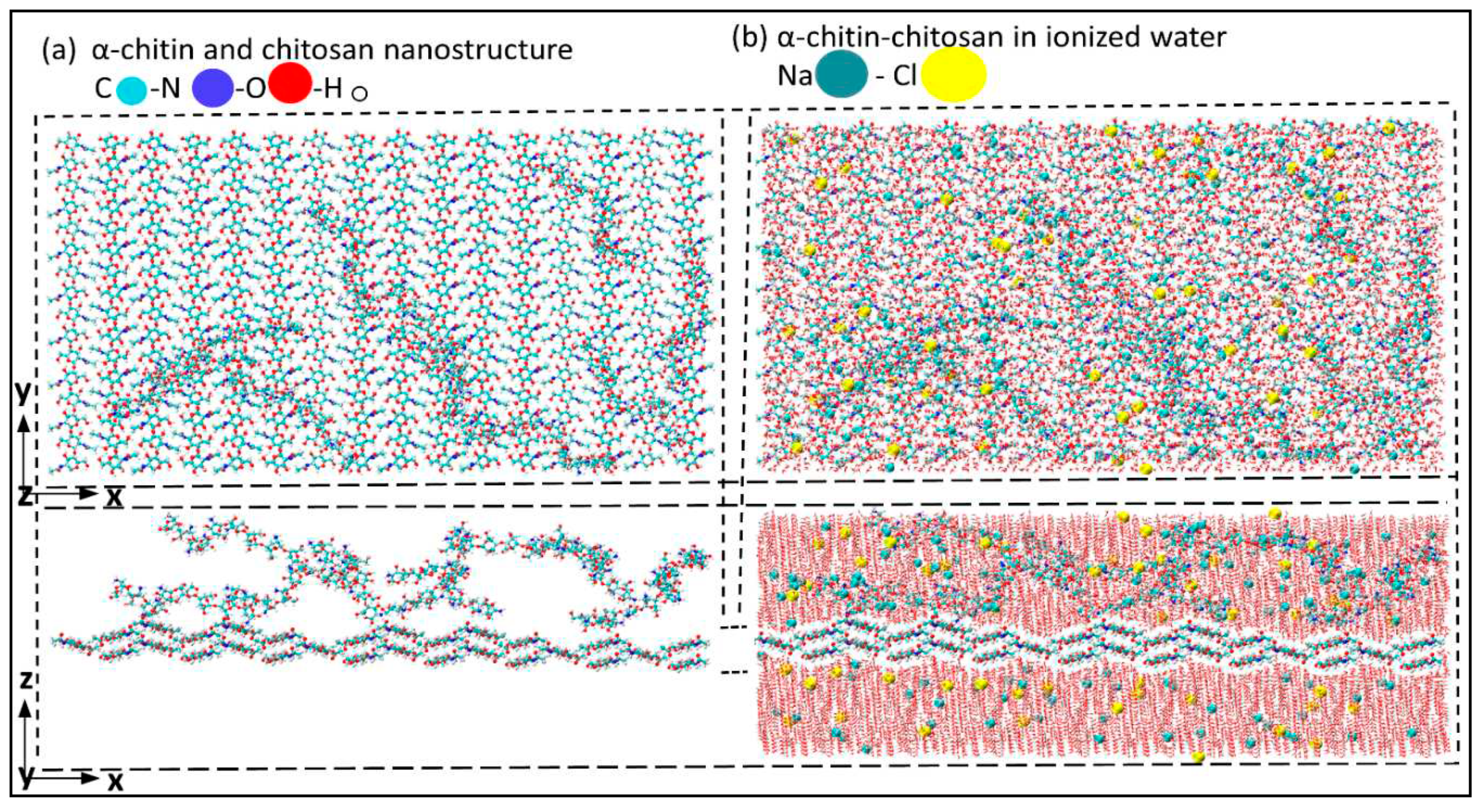
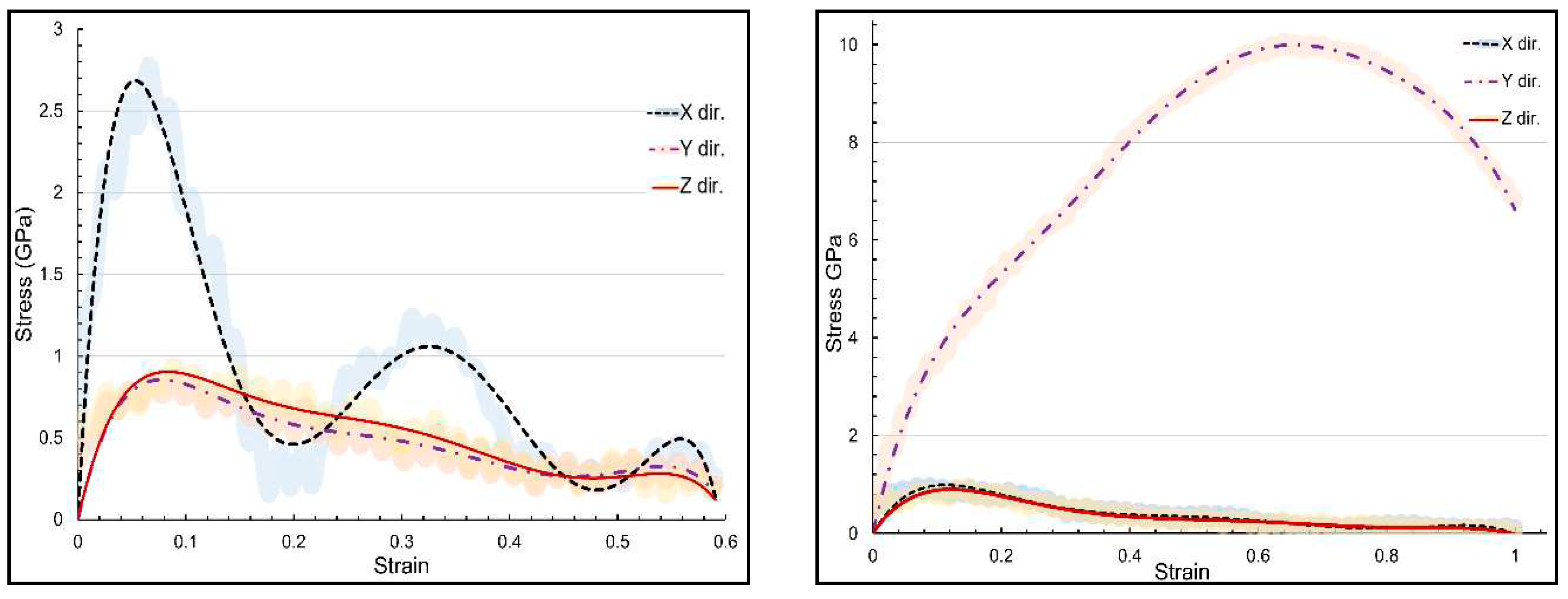
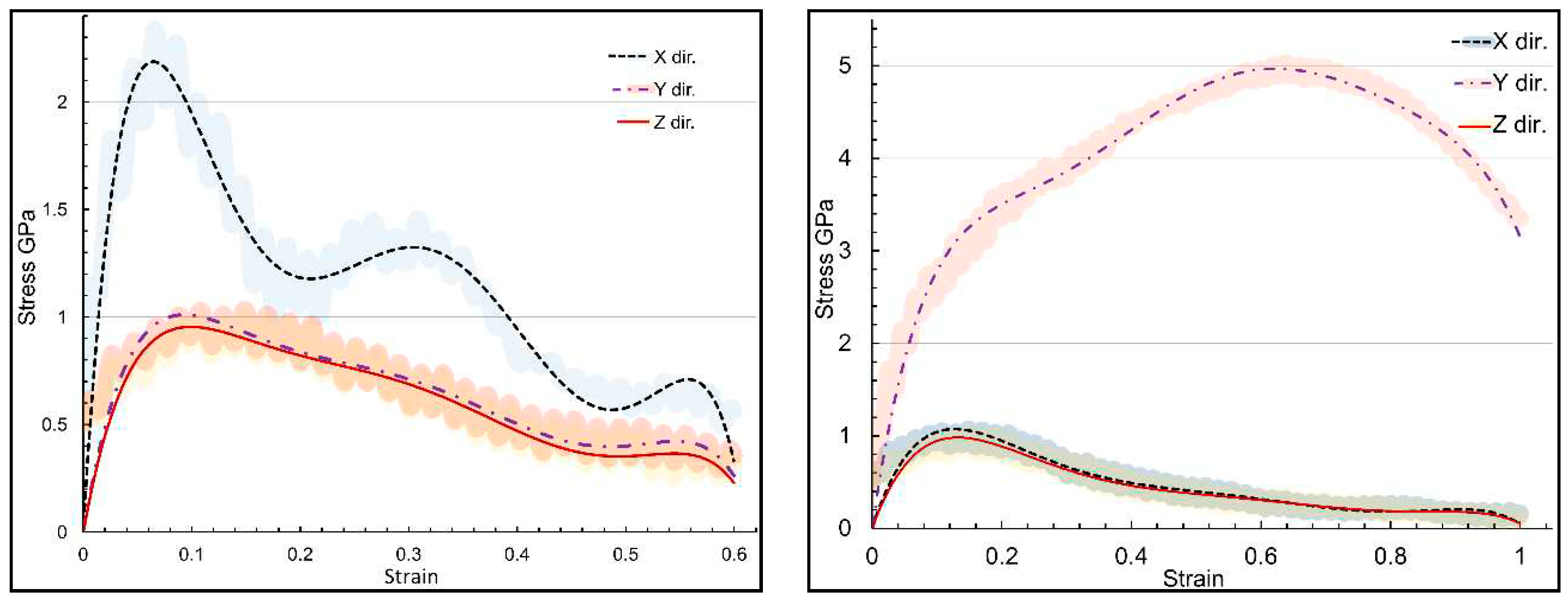
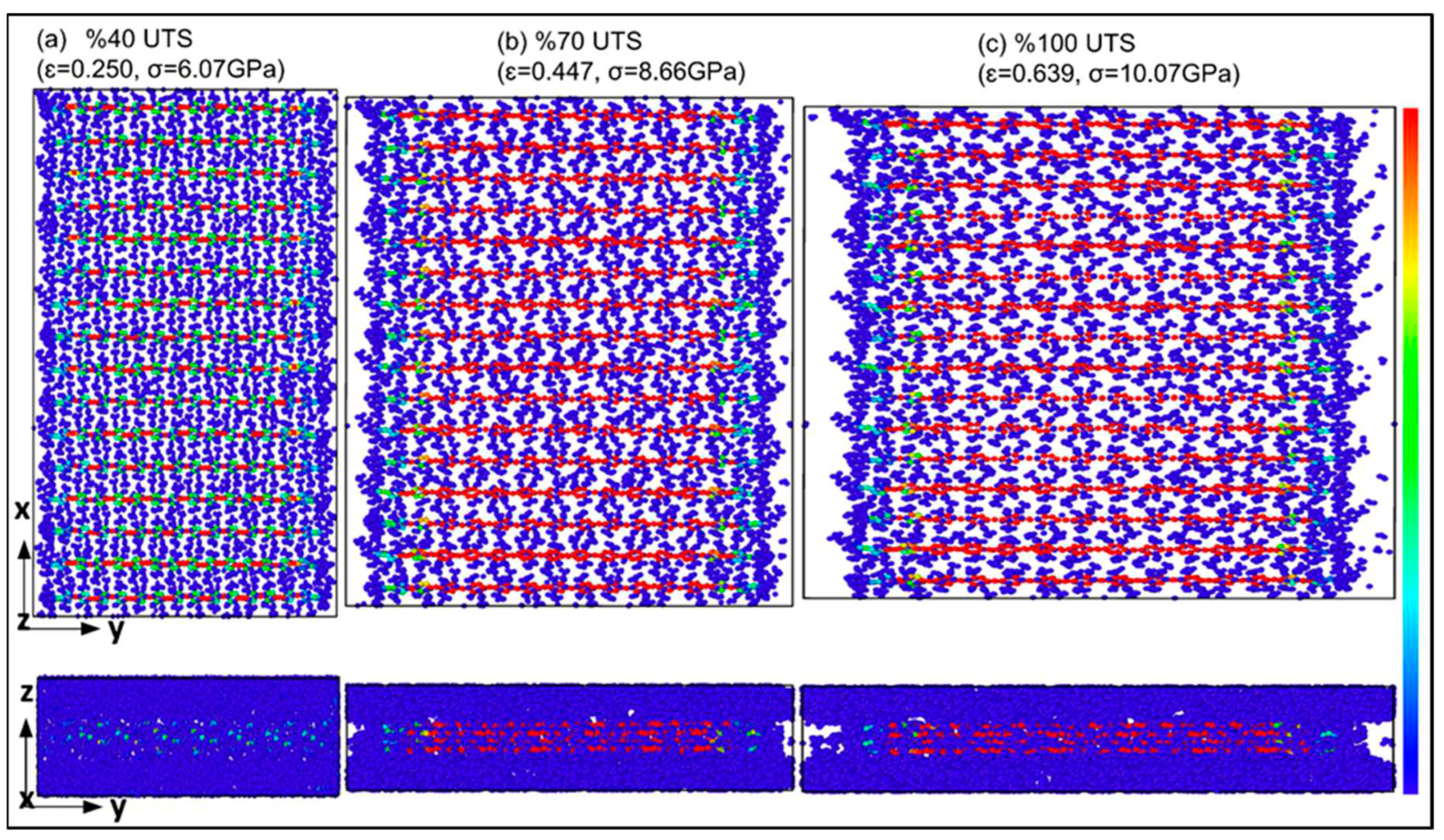
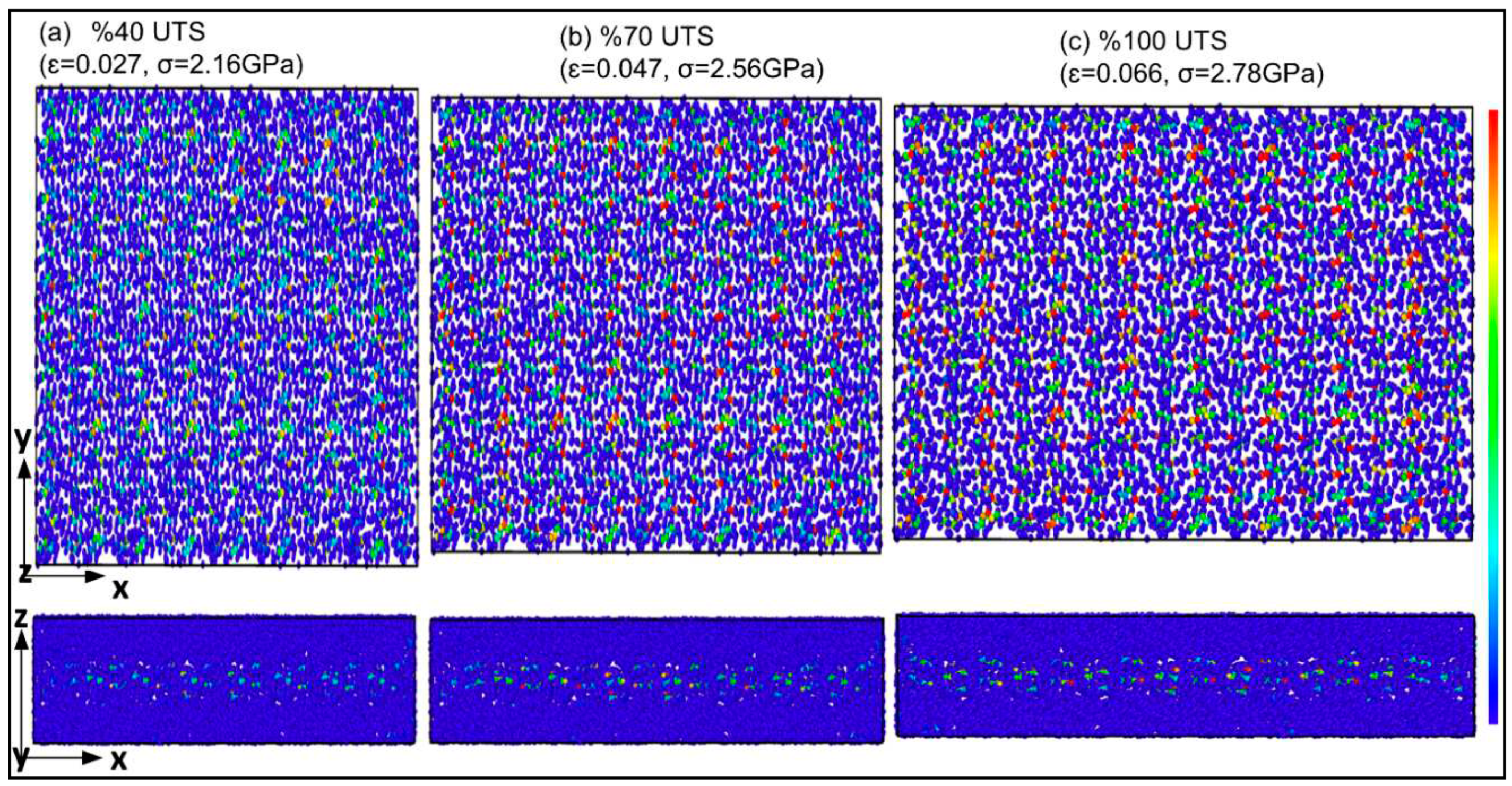
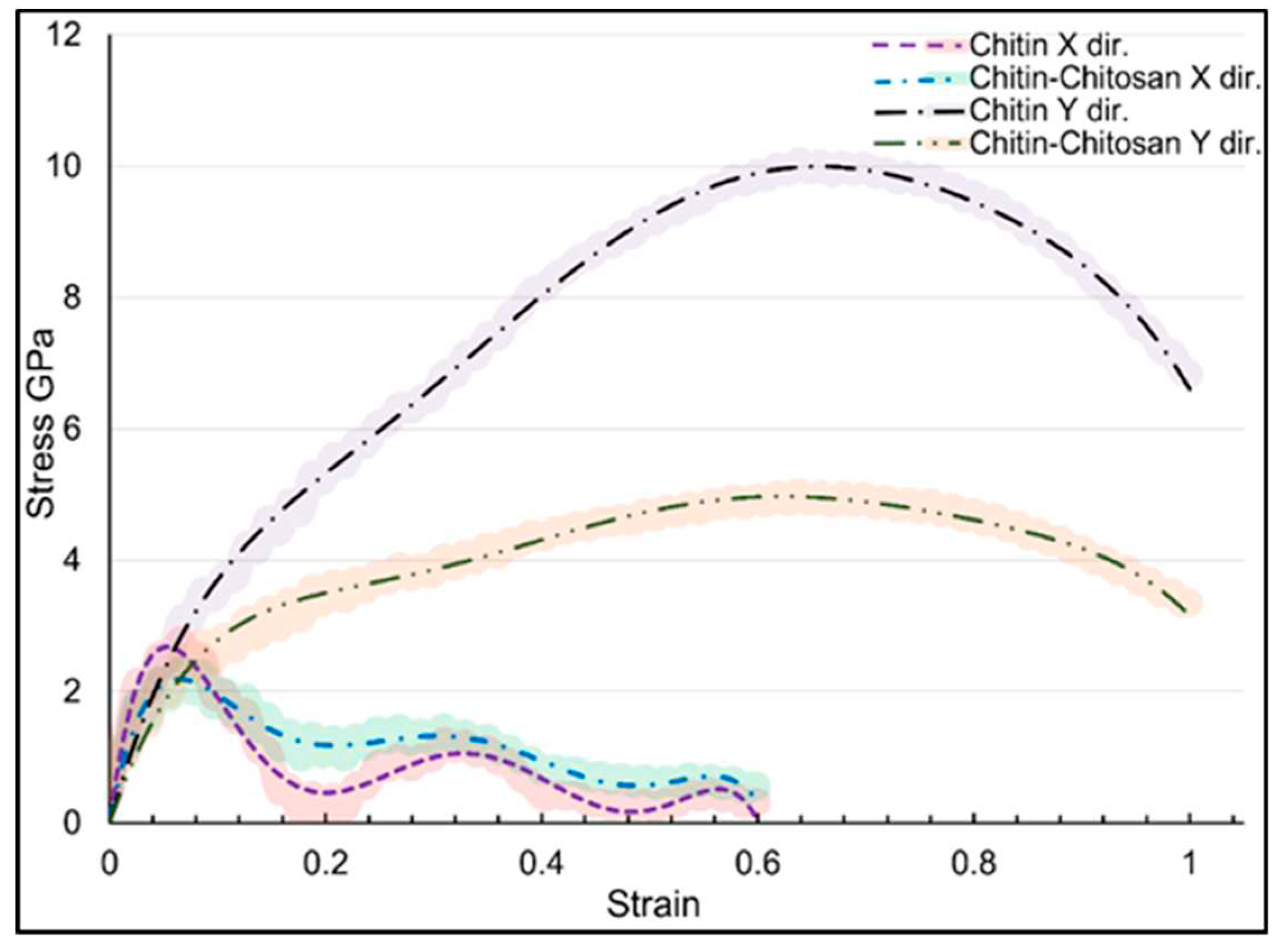
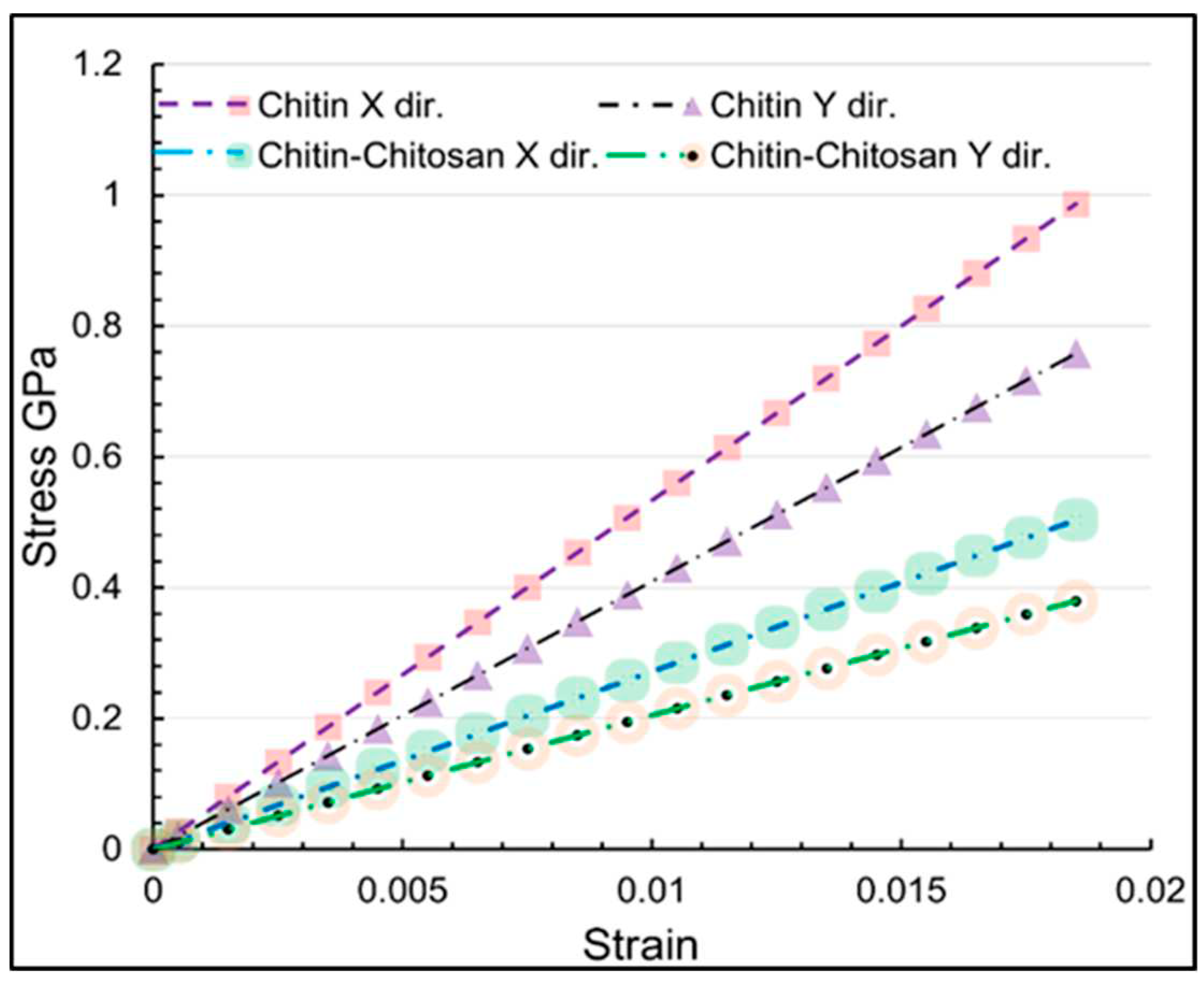
| Structure | axis | Gradient at 0.02 strain |
|---|---|---|
| α-chitin | x | 53.31 |
| α-chitin | y | 40.95 |
| α-chitin-chitosan | x | 27.19 |
| α-chitin-chitosan | y | 20.50 |
| Structure | axis | E | ν | ɛu | UTS |
|---|---|---|---|---|---|
| α-chitin | x | 51.76 | 0.151 | 0.066 | 2.78 |
| α-chitin | y | 39.76 | 0.193 | 0.639 | 10.07 |
| α-chitin-chitosan | x | 31.66 | 0.153 | 0.066 | 2.34 |
| α-chitin-chitosan | y | 26.00 | 0.185 | 0.639 | 5.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




