Submitted:
10 October 2023
Posted:
11 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusion
Author Contributions
Data Availability
Acknowledgments
Conflicts of Interest
- Register Name: PROSPERO
- Registration Number: 471034
Amendments
Abbreviations
| AR: | Androgen receptor |
| ARSI | Androgen Receptor Signaling Inhibitor |
| CRPC: | Castration-Resistant Prostate Cancer |
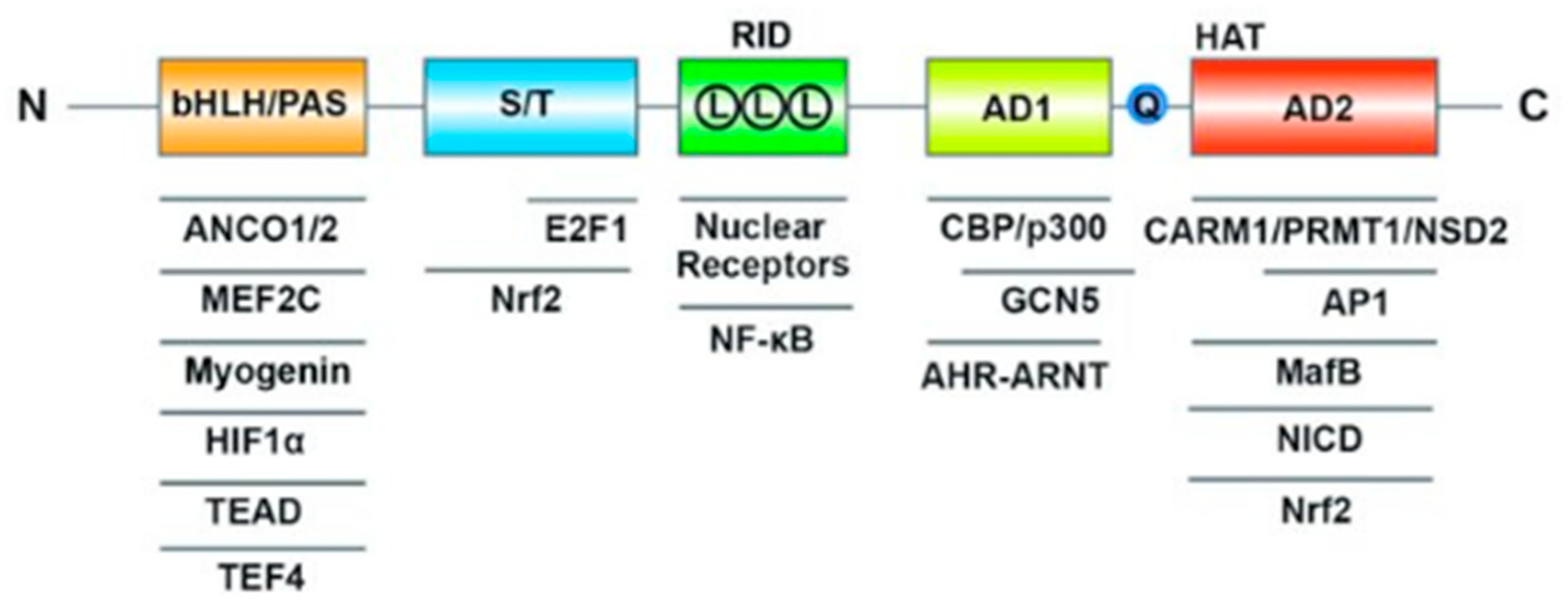
| HAT: | Histone Acetyltransferase |
| NCO3: | Nuclear Receptor Coactivator 3 |
| RID: | Receptor-Interaction Domain |
| SRC-3: | Steroid receptor coactivator 3 |
| AD: | Transcriptional Activation Domains |
| PTMs: | Post-translational Modifications |
References
- ACS, “Key Statistics for Prostate Cancer | Prostate Cancer Facts.” Accessed: Aug. 29, 2023. [Online]. Available: https://www.cancer.org/cancer/types/prostate-cancer/about/key-statistics.html.
- Barnard, M.; Mostaghel, E.A.; Auchus, R.J.; Storbeck, K.-H. The role of adrenal derived androgens in castration resistant prostate cancer. J. Steroid Biochem. Mol. Biol. 2020, 197, 105506. [Google Scholar] [CrossRef]
- Crona, D.J.; Whang, Y.E. Androgen Receptor-Dependent and -Independent Mechanisms Involved in Prostate Cancer Therapy Resistance. Cancers 2017, 9, 67. [Google Scholar] [CrossRef]
- Bernasocchi, T.; Theurillat, J.-P.P. SPOP-mutant prostate cancer: Translating fundamental biology into patient care. Cancer Lett. 2021, 529, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Su, P.; Wang, Y.; Chang, K.; Su, Y.; Chang, P.; Kuan, F.; Hsieh, C.; Kuo, Y.; Sheng, T.; et al. Long-term use and risk of major adverse cardiac events: Comparing enzalutamide and abiraterone in chemotherapy-naïve patients with metastatic castration-resistant prostate cancer. Int. J. Cancer 2023, 152, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Szwarc, M.M.; Kommagani, R.; Lessey, B.A.; Lydon, J.P. The p160/Steroid Receptor Coactivator Family: Potent Arbiters of Uterine Physiology and Dysfunction1. Biol. Reprod. 2014, 91, 122. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, Q. Review of the in Vivo Functions of the p160 Steroid Receptor Coactivator Family. Mol. Endocrinol. 2003, 17, 1681–1692. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Deng, C.-X.; Chen, Q. SRC-3, a Steroid Receptor Coactivator: Implication in Cancer. Int. J. Mol. Sci. 2021, 22, 4760. [Google Scholar] [CrossRef]
- Maurya, V.K.; Szwarc, M.M.; Lonard, D.M.; Gibbons, W.E.; Wu, S.-P.; O’malley, B.W.; DeMayo, F.J.; Lydon, J.P. Decidualization of human endometrial stromal cells requires steroid receptor coactivator-3. Front. Reprod. Heal. 2022, 4, 1033581. [Google Scholar] [CrossRef] [PubMed]
- Mohler, M.L.; Sikdar, A.; Ponnusamy, S.; Hwang, D.-J.; He, Y.; Miller, D.D.; Narayanan, R. An Overview of Next-Generation Androgen Receptor-Targeted Therapeutics in Development for the Treatment of Prostate Cancer. Int. J. Mol. Sci. 2021, 22, 2124. [Google Scholar] [CrossRef]
- Li, L.; Deng, C.-X.; Chen, Q. SRC-3, a Steroid Receptor Coactivator: Implication in Cancer. Int. J. Mol. Sci. 2021, 22, 4760. [Google Scholar] [CrossRef]
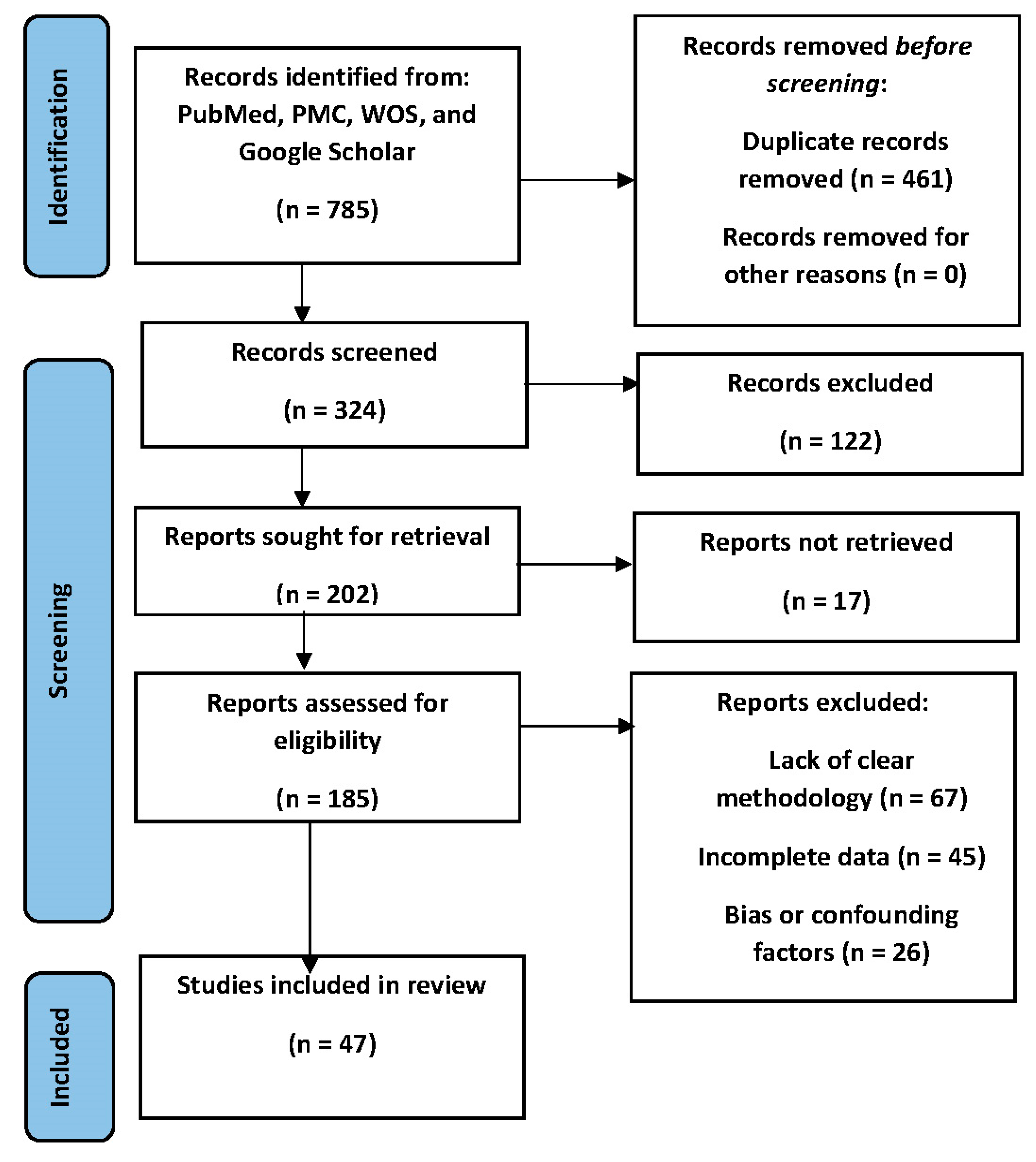
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; et al. he PRISMA 2020 statement : an updated guideline for reporting systematic reviews Systematic reviews and Meta-Analyses. BMJ 2021, 372, 71. [Google Scholar] [CrossRef] [PubMed]
- Dahiya, U.R.; Heemers, H.V. Analyzing the Androgen Receptor Interactome in Prostate Cancer: Implications for Therapeutic Intervention. Cells 2022, 11, 936. [Google Scholar] [CrossRef] [PubMed]
- Tien, J.C.-Y.; Liu, Z.; Liao, L.; Wang, F.; Xu, Y.; Wu, Y.-L.; Zhou, N.; Ittmann, M.; Xu, J. The Steroid Receptor Coactivator-3 Is Required for the Development of Castration-Resistant Prostate Cancer. Cancer Res 2013, 73, 3997–4008. [Google Scholar] [CrossRef] [PubMed]
- L. Qin et al., “Hypoxia-reoxygenation couples 3βHSD1 enzyme and cofactor upregulation to facilitate androgen biosynthesis and hormone therapy resistance in prostate cancer,” Cancer research, vol. 82, no. 13, pp. 2417–2430, 2022. [CrossRef]
- Dasgupta, S.; Putluri, N.; Long, W.; Zhang, B.; Wang, J.; Kaushik, A.K.; Arnold, J.M.; Bhowmik, S.K.; Stashi, E.; Brennan, C.A.; et al. Coactivator SRC-2–dependent metabolic reprogramming mediates prostate cancer survival and metastasis. J. Clin. Investig. 2015, 125, 1174–1188. [Google Scholar] [CrossRef] [PubMed]
- Geng, C.; He, B.; Xu, L.; Barbieri, C.E.; Eedunuri, V.K.; Chew, S.A.; Zimmermann, M.; Bond, R.; Shou, J.; Li, C.; et al. Prostate cancer-associated mutations in speckle-type POZ protein (SPOP) regulate steroid receptor coactivator 3 protein turnover. Proc. Natl. Acad. Sci. USA 2013, 110, 6997–7002. [Google Scholar] [CrossRef]
- Gong, J.; Zhu, J.; Goodman, O.B.; Pestell, R.G.; Schlegel, P.N.; Nanus, D.M.; Shen, R. Activation of p300 histone acetyltransferase activity and acetylation of the androgen receptor by bombesin in prostate cancer cells. Oncogene 2006, 25, 2011–2021. [Google Scholar] [CrossRef]
- Debes, J.D.; Sebo, T.J.; Lohse, C.M.; Murphy, L.M.; Haugen, D.A.L.; Tindall, D.J. p300 in prostate cancer proliferation and progression. Cancer Res. 2003, 63, 7638–7640. [Google Scholar]
- Zhou, H.-J.; Yan, J.; Luo, W.; Ayala, G.; Lin, S.-H.; Erdem, H.; Ittmann, M.; Tsai, S.Y.; Tsai, M.-J. SRC-3 Is Required for Prostate Cancer Cell Proliferation and Survival. Cancer Res 2005, 65, 7976–7983. [Google Scholar] [CrossRef]
- Fujita, K.; Nonomura, N. Role of Androgen Receptor in Prostate Cancer: A Review. World J. Men's Heal. 2019, 37, 288–295. [Google Scholar] [CrossRef]
- Gnanapragasam, V.J.; Leung, H.Y.; Pulimood, A.S.; E Neal, D.; Robson, C.N. Expression of RAC 3, a steroid hormone receptor co-activator in prostate cancer. Br. J. Cancer 2001, 85, 1928–1936. [Google Scholar] [CrossRef]
- Kiliti, A.J.; Sharif, G.M.; Martin, M.B.; Wellstein, A.; Riegel, A.T. AIB1/SRC-3/NCOA3 function in estrogen receptor alpha positive breast cancer. Frontiers in Endocrinology 2023, 14, 1250218. [Google Scholar] [CrossRef]
- Yao, L.; Wang, L.; Cao, Z.-G.; Hu, X.; Shao, Z.-M. High expression of metabolic enzyme PFKFB4 is associated with poor prognosis of operable breast cancer. Cancer Cell Int. 2019, 19, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Watters, R.J.; Verdelis, K.; Lucas, P.C.; Jiang, S.; Chen, Y.; Lu, F.; Martin, B.M.; Lukashova, L.; Pecar, G.; Morales-Restrepo, A.; et al. A Novel Mouse Model for SNP in Steroid Receptor Co-Activator-1 Reveals Role in Bone Density and Breast Cancer Metastasis. Endocrinology 2021, 162, bqab094. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Shan, N.; Xu, P.; Ge, H.; Yuan, Y.; Liu, Y.; Zhang, P.; Wen, L.; Zhang, F.; Xiong, L.; et al. Hypoxia-induced Downregulation of SRC-3 Suppresses Trophoblastic Invasion and Migration Through Inhibition of the AKT/mTOR Pathway: Implications for the Pathogenesis of Preeclampsia. Sci. Rep. 2019, 9, 10349. [Google Scholar] [CrossRef] [PubMed]
- Tanizaki, Y.; Bao, L.; Shi, B.; Shi, Y.-B. A Role of Endogenous Histone Acetyltransferase Steroid Hormone Receptor Coactivator 3 in Thyroid Hormone Signaling DuringXenopusIntestinal Metamorphosis. Thyroid® 2021, 31, 692–702. [Google Scholar] [CrossRef]
- K. Takayama, “The Biological Role of Androgen Receptor in Prostate Cancer Progression,” in Advances in Testosterone Action, M. Estrada, Ed., IntechOpen, 2018. [CrossRef]
- A. Shrestha, “The Regulation of Steroid Receptor Co-activator-3 Activity by p38MAPK-MK2 Signaling Pathway,” 2022.
- Iheagwam, F.; Iheagwam, O.; Odiba, J.; Ogunlana, O.; Chinedu, S. Cancer and Glucose Metabolism: A Review on Warburg Mechanisms. Tropical Journal of Natural Product Research 2022, 6. [Google Scholar] [CrossRef]
- Xu, X.D.; Shao, S.X.; Jiang, H.P.; Cao, Y.W.; Wang, Y.H.; Yang, X.C.; Wang, Y.L.; Wang, X.S.; Niu, H.T. Warburg Effect or Reverse Warburg Effect? A Review of Cancer Metabolism. Oncol. Res. Treat. 2015, 38, 117–122. [Google Scholar] [CrossRef]
- Lu, J.; Tan, M.; Cai, Q. The Warburg effect in tumor progression: Mitochondrial oxidative metabolism as an anti-metastasis mechanism. Cancer Lett. 2015, 356, 156–164. [Google Scholar] [CrossRef]
- Yoo, H.C.; Yu, Y.C.; Sung, Y.; Han, J.M. Glutamine reliance in cell metabolism. Exp. Mol. Med. 2020, 52, 1496–1516. [Google Scholar] [CrossRef]
- Dessai, A.S.; Kalhotra, P.; Novickis, A.T.; Dasgupta, S. Regulation of tumor metabolism by post translational modifications on metabolic enzymes. Cancer Gene Ther. 2022, 30, 548–558. [Google Scholar] [CrossRef]
- Kishore, C.; Zi, X. Wnt Signaling and Therapeutic Resistance in Castration-Resistant Prostate Cancer. Curr. Pharmacol. Rep. 2023, 1–14. [Google Scholar] [CrossRef]
- A Blundon, M.; Dasgupta, S. Metabolic Dysregulation Controls Endocrine Therapy–Resistant Cancer Recurrence and Metastasis. Endocrinology 2019, 160, 1811–1820. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Xu, Z.; Zhu, X.; Zhang, J. New insights into molecules and pathways of cancer metabolism and therapeutic implications. Cancer Commun. 2020, 41, 16–36. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, Q.; Dong, C. Metabolic reprogramming in triple-negative breast cancer. Cancer Biol. Med. 2020, 17, 44. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Chen, J.; Lu, D.; Jain, P.; Yu, Y.; Cardenas, D.; Peng, X.; Yu, X.; Xu, J.; Wang, J.; et al. Development of improved SRC-3 inhibitors as breast cancer therapeutic agents. Endocrine-Related Cancer 2021, 28, 657. [Google Scholar] [CrossRef]
- Gilad, Y.; Eliaz, Y.; Yu, Y.; Dean, A.M.; Han, S.J.; Qin, L.; O’malley, B.W.; Lonard, D.M. A genome-scale CRISPR Cas9 dropout screen identifies synthetically lethal targets in SRC-3 inhibited cancer cells. Commun. Biol. 2021, 4, 399. [Google Scholar] [CrossRef] [PubMed]
- Asangani, I.; Blair, I.A.; Van Duyne, G.; Hilser, V.J.; Moiseenkova-Bell, V.; Plymate, S.; Sprenger, C.; Wand, A.J.; Penning, T.M. Using biochemistry and biophysics to extinguish androgen receptor signaling in prostate cancer. J. Biol. Chem. 2021, 296, 100240. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, H.; Jia, L.; He, J.; Li, Y.; Liu, H.; Wu, R.; Qiu, Y.; Zhan, Y.; Yuan, Z.; et al. Bufalin targets the SRC-3/MIF pathway in chemoresistant cells to regulate M2 macrophage polarization in colorectal cancer. Cancer Lett. 2021, 513, 63–74. [Google Scholar] [CrossRef]
- Wang, Y.; Luo, X.; Wu, N.; Liao, Q.; Wang, J. SRC-3/TRAF4 facilitates ovarian cancer development by activating the PI3K/AKT signaling pathway. Med Oncol. 2023, 40, 76. [Google Scholar] [CrossRef]
- Osborne, C.K.; et al. Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. Journal of the National Cancer Institute 2003, 95, 353–361. [Google Scholar] [CrossRef]
- Rotimi, S.O.; Rotimi, O.A.; Salhia, B. A Review of Cancer Genetics and Genomics Studies in Africa. Front. Oncol. 2021, 10, 606400. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Chen, J.; Zhao, M.; Zhang, C.; Yu, Y.; Lonard, D.M.; Chow, D.-C.; Palzkill, T.; Xu, J.; O’malley, B.W.; et al. Development of potent small-molecule inhibitors to drug the undruggable steroid receptor coactivator-3. Proc. Natl. Acad. Sci. 2016, 113, 4970–4975. [Google Scholar] [CrossRef] [PubMed]
- Axlund, S.D.; Lambert, J.R.; Nordeen, S.K. HOXC8 Inhibits Androgen Receptor Signaling in Human Prostate Cancer Cells by Inhibiting SRC-3 Recruitment to Direct Androgen Target Genes. Mol. Cancer Res. 2010, 8, 1643–1655. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



