Submitted:
26 September 2023
Posted:
03 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
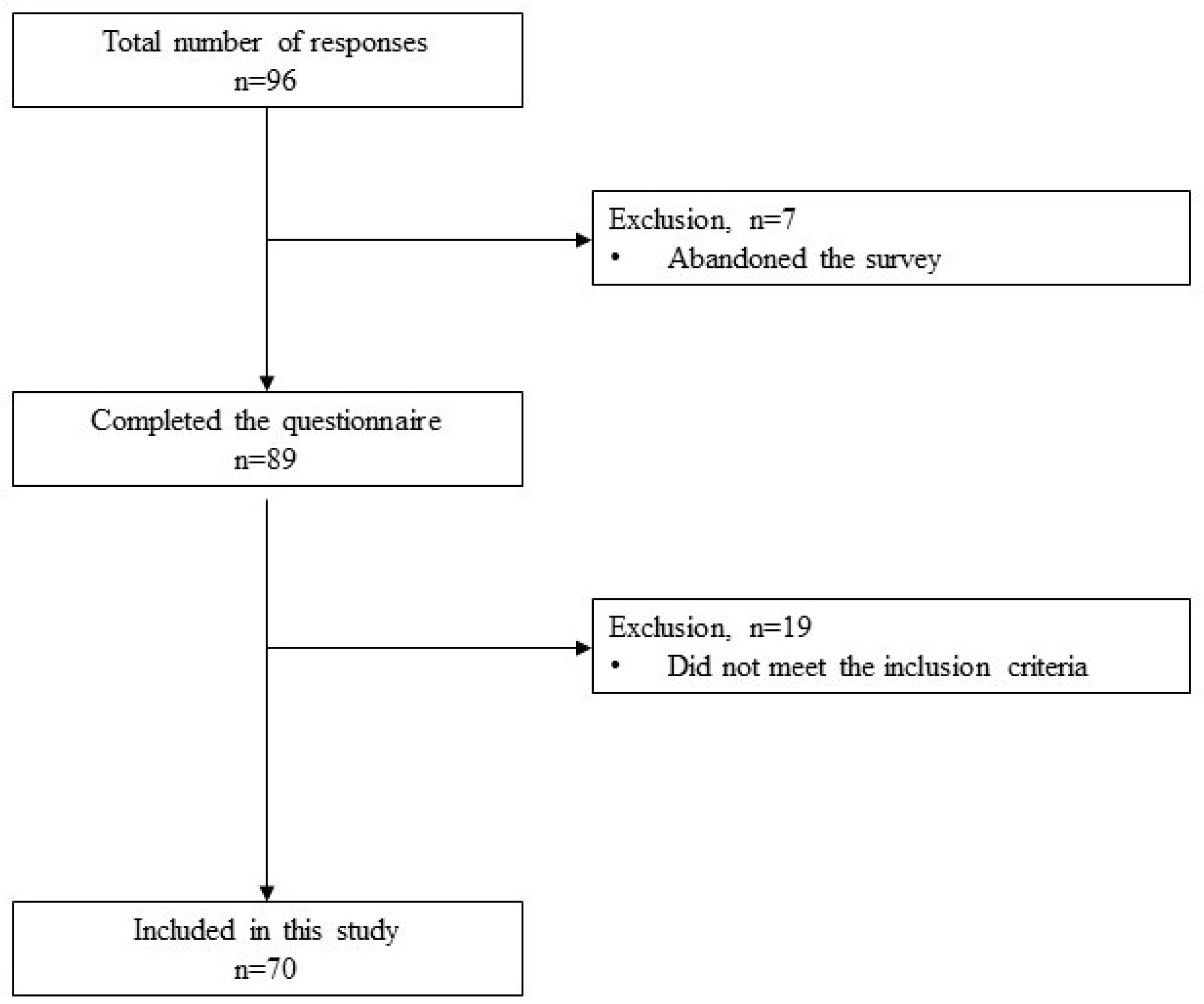
Study design and data collection
Inclusion criteria and definition of groups
- Have you already worked with patients treated with cannabinoid drugs?
- Were any of them of pediatric age (0-18y)?
- Were any of these children/adolescents diagnosed with cerebral palsy?
- Did you ever experience a situation in which the prescription of cannabinoids in this type of patient was debated, but for some reason not prescribed?
Statistical analysis
Ethical statement
3. Results
3.1. Study participants
3.1. Indications
3.1. Prescribed formulas
3.1. Perceived effect of the treatment and side effects
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosenbaum, P.; Paneth, N.; Leviton, A.; Goldstein, M.; Bax, M.; Damiano, D.; Dan, B.; Jacobsson, B. A report: The definition and classification of cerebral palsy april 2006. Dev Med Child Neurol Suppl 2007, 109, 8–14. [Google Scholar] [PubMed]
- Surveillance of Cerebral Palsy in Europe. Surveillance of cerebral palsy in europe: A collaboration of cerebral palsy surveys and registers. Surveillance of cerebral palsy in europe (scpe). Dev Med Child Neurol 2000, 42, 816–824. [Google Scholar] [CrossRef]
- Horwood, L.; Li, P.; Mok, E.; Oskoui, M.; Shevell, M.; Constantin, E. Behavioral difficulties, sleep problems, and nighttime pain in children with cerebral palsy. Res Dev Disabil 2019, 95, 103500. [Google Scholar] [CrossRef] [PubMed]
- Horwood, L.; Li, P.; Mok, E.; Shevell, M.; Constantin, E. A systematic review and meta-analysis of the prevalence of sleep problems in children with cerebral palsy: How do children with cerebral palsy differ from each other and from typically developing children? Sleep Health 2019, 5, 555–571. [Google Scholar] [CrossRef]
- Gururaj, A.K.; Sztriha, L.; Bener, A.; Dawodu, A.; Eapen, V. Epilepsy in children with cerebral palsy. Seizure 2003, 12, 110–114. [Google Scholar] [CrossRef]
- Tsubouchi, Y.; Tanabe, A.; Saito, Y.; Noma, H.; Maegaki, Y. Long-term prognosis of epilepsy in patients with cerebral palsy. Dev Med Child Neurol 2019, 61, 1067–1073. [Google Scholar] [CrossRef]
- Singhi, P.; Jagirdar, S.; Khandelwal, N.; Malhi, P. Epilepsy in children with cerebral palsy. J Child Neurol 2003, 18, 174–179. [Google Scholar] [CrossRef]
- Fluss, J.; Lidzba, K. Cognitive and academic profiles in children with cerebral palsy: A narrative review. Ann Phys Rehabil Med 2020, 63, 447–456. [Google Scholar] [CrossRef]
- Novak, I.; McIntyre, S.; Morgan, C.; Campbell, L.; Dark, L.; Morton, N.; Stumbles, E.; Wilson, S.A.; Goldsmith, S. A systematic review of interventions for children with cerebral palsy: State of the evidence. Dev Med Child Neurol 2013, 55, 885–910. [Google Scholar] [CrossRef]
- Harvey, A.R.; Baker, L.B.; Reddihough, D.S.; Scheinberg, A.; Williams, K. Trihexyphenidyl for dystonia in cerebral palsy. Cochrane Database Syst Rev 2018, 5, CD012430. [Google Scholar] [CrossRef]
- Hasnat, M.J.; Rice, J.E. Intrathecal baclofen for treating spasticity in children with cerebral palsy. Cochrane Database Syst Rev 2015, CD004552. [Google Scholar] [CrossRef] [PubMed]
- Hoare, B.J.; Wallen, M.A.; Imms, C.; Villanueva, E.; Rawicki, H.B.; Carey, L. Botulinum toxin a as an adjunct to treatment in the management of the upper limb in children with spastic cerebral palsy (update). Cochrane Database Syst Rev 2010, CD003469. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, M.M.; Queiroz, R.H.; Zuardi, A.W.; Crippa, J.A. Safety and side effects of cannabidiol, a cannabis sativa constituent. Curr Drug Saf 2011, 6, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.S.; Wilens, T.E. Medical cannabinoids in children and adolescents: A systematic review. Pediatrics 2017, 140. [Google Scholar] [CrossRef]
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S.; Cannabidiol in Dravet Syndrome Study Group. Trial of cannabidiol for drug-resistant seizures in the dravet syndrome. N Engl J Med 2017, 376, 2011–2020. [Google Scholar] [CrossRef]
- Devinsky, O.; Patel, A.D.; Cross, J.H.; Villanueva, V.; Wirrell, E.C.; Privitera, M.; Greenwood, S.M.; Roberts, C.; Checketts, D.; VanLandingham, K.E.; et al. Effect of cannabidiol on drop seizures in the lennox-gastaut syndrome. N Engl J Med 2018, 378, 1888–1897. [Google Scholar] [CrossRef]
- Thiele, E.A.; Marsh, E.D.; French, J.A.; Mazurkiewicz-Beldzinska, M.; Benbadis, S.R.; Joshi, C.; Lyons, P.D.; Taylor, A.; Roberts, C.; Sommerville, K.; et al. Cannabidiol in patients with seizures associated with lennox-gastaut syndrome (gwpcare4): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018, 391, 1085–1096. [Google Scholar] [CrossRef]
- Aran, A.; Cayam-Rand, D. Medical cannabis in children. Rambam Maimonides Med J 2020, 11. [Google Scholar] [CrossRef]
- Ali, S.; Scheffer, I.E.; Sadleir, L.G. Efficacy of cannabinoids in paediatric epilepsy. Dev Med Child Neurol 2019, 61, 13–18. [Google Scholar] [CrossRef]
- Erridge, S.; Holvey, C.; Coomber, R.; Hoare, J.; Khan, S.; Platt, M.W.; Rucker, J.J.; Weatherall, M.W.; Beri, S.; Sodergren, M.H. Clinical outcome data of children treated with cannabis-based medicinal products for treatment resistant epilepsy-analysis from the uk medical cannabis registry. Neuropediatrics 2023, 54, 174–181. [Google Scholar] [CrossRef]
- Kilcher, G.; Zwahlen, M.; Ritter, C.; Fenner, L.; Egger, M. Medical use of cannabis in switzerland: Analysis of approved exceptional licences. Swiss Med Wkly 2017, 147, w14463. [Google Scholar]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for medical use: A systematic review and meta-analysis. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Germanos, R.; Weier, M.; Pollard, J.; Degenhardt, L.; Hall, W.; Buckley, N.; Farrell, M. The use of cannabis and cannabinoids in treating symptoms of multiple sclerosis: A systematic review of reviews. Curr Neurol Neurosci Rep 2018, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.S.; Friend, A.J.; Gibson, F.; Houghton, E.; Gopaul, S.; Craig, J.V.; Pizer, B. Antiemetic medication for prevention and treatment of chemotherapy-induced nausea and vomiting in childhood. Cochrane Database Syst Rev 2016, 2, CD007786. [Google Scholar] [CrossRef] [PubMed]
- Kuhlen, M.; Hoell, J.I.; Gagnon, G.; Balzer, S.; Oommen, P.T.; Borkhardt, A.; Janssen, G. Effective treatment of spasticity using dronabinol in pediatric palliative care. Eur J Paediatr Neurol 2016, 20, 898–903. [Google Scholar] [CrossRef]
- Efron, D.; Taylor, K. Medicinal cannabis for paediatric developmental, behavioural and mental health disorders. Int J Environ Res Public Health 2023, 20. [Google Scholar] [CrossRef] [PubMed]
- Bohn, E.; Goren, K.; Switzer, L.; Falck-Ytter, Y.; Fehlings, D. Pharmacological and neurosurgical interventions for individuals with cerebral palsy and dystonia: A systematic review update and meta-analysis. Dev Med Child Neurol 2021, 63, 1038–1050. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Murnion, B.; Campbell, G.; Young, H.; Hall, W. Cannabinoids for the treatment of spasticity. Dev Med Child Neurol 2019, 61, 631–638. [Google Scholar] [CrossRef]
- Fairhurst, C.; Kumar, R.; Checketts, D.; Tayo, B.; Turner, S. Efficacy and safety of nabiximols cannabinoid medicine for paediatric spasticity in cerebral palsy or traumatic brain injury: A randomized controlled trial. Dev Med Child Neurol 2020, 62, 1031–1039. [Google Scholar] [CrossRef]
- Pawliuk, C.; Chau, B.; Rassekh, S.R.; McKellar, T.; Siden, H.H. Efficacy and safety of paediatric medicinal cannabis use: A scoping review. Paediatr Child Health 2021, 26, 228–233. [Google Scholar] [CrossRef]
- Zürcher, K.; Dupont, C.; Weber, P.; Grunt, S.; Wilhelm, I.; Eigenmann, D.E.; Reichumth, M.L.; Fankhauser, M.; Egger, M.; Lukas, F. Medical cannabis in children and adolescents in switzerland: A retrospective study. Eur J Pediatr 2021. [Google Scholar]
- da Rovare, V.P.; Magalhaes, G.P.A.; Jardini, G.D.A.; Beraldo, M.L.; Gameiro, M.O.; Agarwal, A.; Luvizutto, G.J.; Paula-Ramos, L.; Camargo, S.E.A.; de Oliveira, L.D.; et al. Cannabinoids for spasticity due to multiple sclerosis or paraplegia: A systematic review and meta-analysis of randomized clinical trials. Complement Ther Med 2017, 34, 170–185. [Google Scholar] [CrossRef]
- Nielsen, S.; Hall, W. Efficacy of cannabinoids for treating paediatric spasticity in cerebral palsy or traumatic brain injury: What is the evidence? Dev Med Child Neurol 2020, 62, 1007. [Google Scholar] [CrossRef] [PubMed]
- Stockings, E.; Campbell, G.; Hall, W.D.; Nielsen, S.; Zagic, D.; Rahman, R.; Murnion, B.; Farrell, M.; Weier, M.; Degenhardt, L. Cannabis and cannabinoids for the treatment of people with chronic noncancer pain conditions: A systematic review and meta-analysis of controlled and observational studies. Pain 2018, 159, 1932–1954. [Google Scholar] [CrossRef]
- Aviram, J.; Samuelly-Leichtag, G. Efficacy of cannabis-based medicines for pain management: A systematic review and meta-analysis of randomized controlled trials. Pain Physician 2017, 20, E755–E796. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, C.T.; Meehan, E.M.; Harvey, A.R.; Antolovich, G.C.; Morgan, P.E. Prevalence and characteristics of pain in children and young adults with cerebral palsy: A systematic review. Dev Med Child Neurol 2019, 61, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Mascia, M.M.; Carmagnini, D.; Defazio, G. Cannabinoids and dystonia: An issue yet to be defined. Neurol Sci 2020, 41, 783–787. [Google Scholar] [CrossRef]
- Libzon, S.; Schleider, L.B.; Saban, N.; Levit, L.; Tamari, Y.; Linder, I.; Lerman-Sagie, T.; Blumkin, L. Medical cannabis for pediatric moderate to severe complex motor disorders. J Child Neurol 2018, 33, 565–571. [Google Scholar] [CrossRef]
- Chayasirisobhon, S. The role of cannabidiol in neurological disorders. Perm J 2021, 25. [Google Scholar] [CrossRef]
- Izzo, A.A.; Borrelli, F.; Capasso, R.; Di Marzo, V.; Mechoulam, R. Non-psychotropic plant cannabinoids: New therapeutic opportunities from an ancient herb. Trends Pharmacol Sci 2009, 30, 515–527. [Google Scholar] [CrossRef]
- Amin, M.R.; Ali, D.W. Pharmacology of medical cannabis. Adv Exp Med Biol 2019, 1162, 151–165. [Google Scholar] [PubMed]
| Total n=70 (%) |
Experience n=47 (%) |
No experience n=23 (%) |
p-value1 | ||
|---|---|---|---|---|---|
| Sex | 0.96 | ||||
| Male | 25 (36) | 17 (36) | 8 (35) | ||
| Female | 43 (61) | 29 (62) | 14 (61) | ||
| Unknown | 2 (3) | 1 (2) | 1 (4) | ||
| Age in years (interquartile range) | 48 (42-57) | 47.5 (41-56) | 50 (42-60) | 0.92 | |
| Specialisation1 | |||||
| Pediatrics | 45 (64) | 33 (70) | 12 (52) | 0.14 | |
| Physical Medicine and Rehabilitation | 28 (40) | 18 (38) | 10 (44) | 0.80 | |
| Neurology | 10 (14) | 8 (17) | 2 (9) | 0.35 | |
| Anesthesia | 1 (1) | 0 | 1 (4) | 0.33 | |
| Subspecialization | |||||
| Yes | 53 (76) | 39 (88) | 14 (61) | 0.04 | |
| Neuropediatric | 28 (40) | 19 (40) | 9 (39) | 0.92 | |
| Pediatric rehabilitation medicine | 21 (30) | 18 (38) | 3 (13) | 0.05 | |
| Developmental pediatrics | 8 (11) | 5 (11) | 3 (13) | 1.0 | |
| Pediatric palliative care | 5 (7) | 3 (6) | 2 (9) | 1.0 | |
| Pediatric oncology-hematology | 1 (1) | 1 (2) | 0 (0) | 1.0 | |
| Neurometabolic | 1 (1) | 0 (0) | 1 (4) | 1.0 | |
| No | 17 (24) | 8 (17) | 9 (39) | ||
| Work experience in specialty in years | 0.81 | ||||
| 0-5 | 12 (17) | 8 (17) | 4 (32) | ||
| 6-10 | 13 (19) | 8 (17) | 5 (22) | ||
| 11-15 | 10 (14) | 8 (17) | 2 (9) | ||
| >15 | 35 (50) | 23 (49) | 12 (52) | ||
| Work experience in the treatment of children with CP in years | 0.72 | ||||
| 0-5 | 15 (21) | 9 (19) | 6 (26) | ||
| 6-10 | 16 (23) | 11 (23) | 5 (22) | ||
| 11-15 | 18 (26) | 14 (30) | 4 (17) | ||
| >15 | 21 (30) | 13 (28) | 8 (35) | ||
| Workplace1 | 0.01 | ||||
| Any hospital | 56 (80) | 42 (89) | 14 (61) | ||
| University hospital | 23 (33) | 20 (43) | 3 (13) | ||
| General hospital | 27 (39) | 18 (38) | 9 (39) | ||
| University and general hospital | 6 (9) | 4 (8) | 2 (9) | ||
| Rehabilitation center | 12 (17) | 4 (8) | 8 (35) | ||
| Private/Joined practice | 2 (3) | 2 (4) | 0 (0) | ||
| Knowledge Acquisition1 | |||||
| Individual learning | 45 (64) | 33 (70) | 12 (52) | 0.14 | |
| Colleagues | 28 (40) | 19 (40) | 9 (39) | 0.92 | |
| Conferences | 17 (24) | 11 (23) | 6 (26) | 0.81 | |
| Part of education | 15 (21) | 10 (21) | 5 (22) | 0.97 | |
| Parents & families | 10 (14) | 7 (15) | 3 (13) | 0.84 | |
| Institution | 8 (11) | 6 (13) | 2 (9) | 0.62 | |
| Companies | 2 (3) | 1 (2) | 1 (4) | 0.60 | |
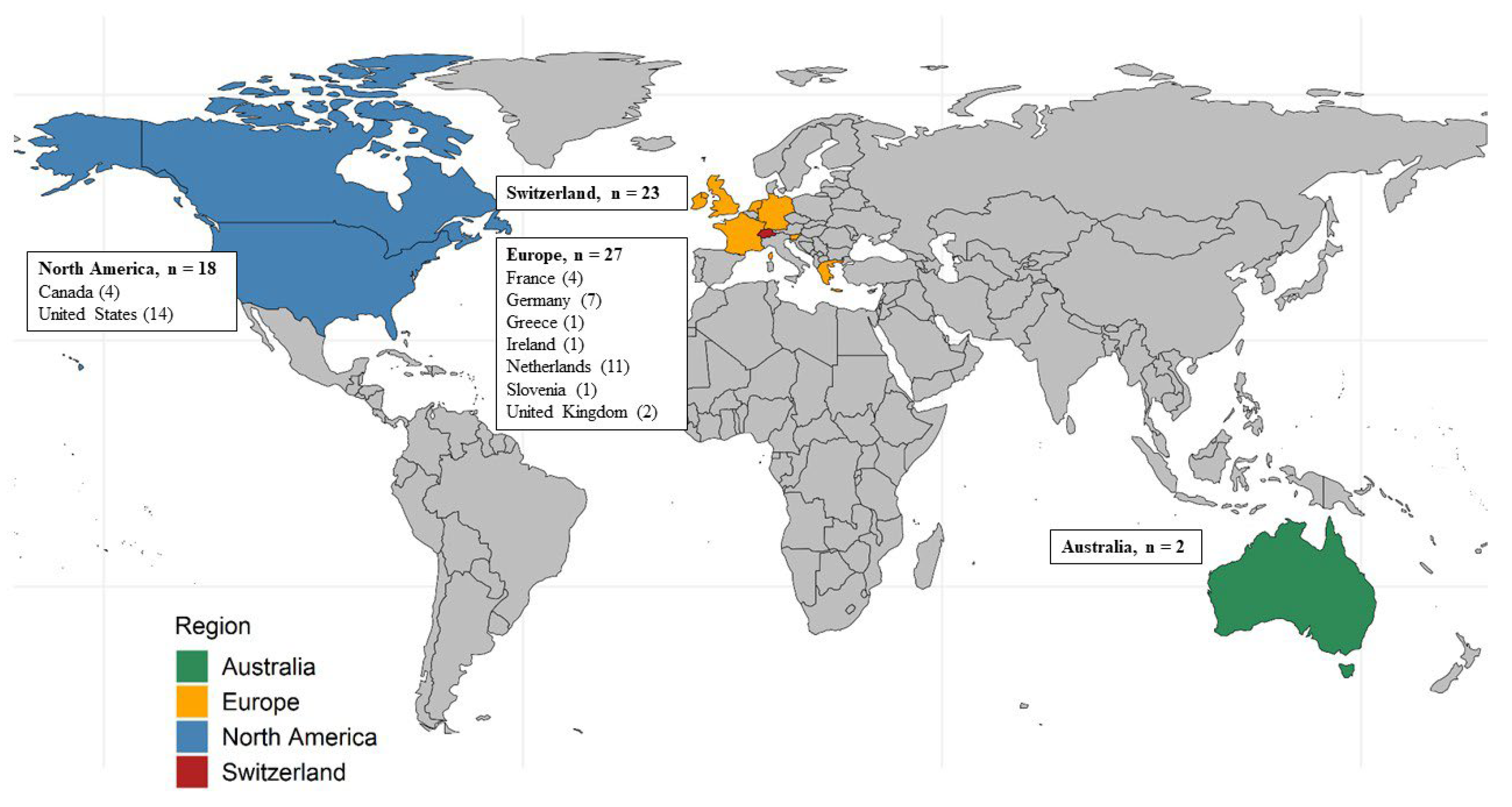
| Country of Residence (grouped) | 0.004 | ||||
| Switzerland | 23 (33) | 15 (32) | 8 (35) | ||
| Europe excluding Switzerland | 27 (39) | 13 (28) | 14 (61) | ||
| North America | 18 (26) | 18 (38) | 0 (0) | ||
| Australia | 2 (3) | 1 (2) | 1 (4) | ||
| Total n=70, (%) |
Experience n=47, (%) |
No experience n=23, (%) |
p-value3 | |
|---|---|---|---|---|
| Indication (overall)1,2 | ||||
| Epilepsy | 48 (69) | 37 (79) | 11 (48) | 0.009 |
| Spasticity | 45 (64) | 34 (72) | 11 (48) | 0.04 |
| Pain | 44 (63) | 33 (70) | 11 (48) | 0.06 |
| Behavioural problems | 12 (17) | 9 (19) | 3 (13) | 0.74 |
| Sleep disturbance | 11 (16) | 8 (17) | 3 (14) | 0.67 |
| Dystonia | 8 (11) | 7 (15) | 1 (4) | 0.26 |
| None | 5 (7) | 1 (2) | 4 (17) | 0.04 |
| Context of prescription | 0.01 | |||
| Co-medication | 28 (40) | 22 (47) | 6 (26) | |
| Second line treatment | 16 (23) | 11 (23) | 5 (22) | |
| Palliative treatment | 7 (10) | 6 (13) | 1 (4) | |
| First line treatment | 2 (3) | 2 (4) | 0 (0) | |
| Parents initiated treatment | 2 (3) | 2 (4) | 0 (0) | |
| Not applicable | 6 (9) | 2 (4) | 4 (17) | |
| No response | 9 (13) | 2 (4) | 7 (30) | |
| Reasons to not initiate cannabinoids1 | ||||
| Lack of cost coverage | 24 (34) | 19 (40) | 5 (22) | 0.12 |
| Age of the patient | 19 (27) | 15 (32) | 4 (17) | 0.26 |
| Lack of evidence on effectiveness and side effects | 15 (21) | 10 (21) | 5 (22) | 0.97 |
| Parents wish | 13 (19) | 10 (21) | 3 (13) | 0.52 |
| Drug interaction | 10 (14) | 8 (17) | 2 (9) | 0.48 |
| Other co-morbidities | 3 (4) | 3 (6) | 0 (0) | 0.55 |
| None | 8 (11) | 5 (11) | 3 (13) | 1.00 |
| Total | Total n=70, (%) |
Experience n=47, (%) |
No experience n=23, (%) |
|---|---|---|---|
| Preparation | |||
| Dronabinol solution: Δ9-THC 2.5% | 16 (23) | 14 (30) | 2 (9) |
| Self-medication: Diff. contents of Δ9-THC & CBD | 13 (19) | 12 (26) | 1 (4) |
| Cannabis oil: Δ9-THC/CBD = 11/24mg per g | 11 (16) | 8 (17) | 3 (13) |
| Cannabis tincture: Δ9-THC/CBD = 11/22mg per g | 3 (4) | 2 (4) | 1 (4) |
| Cannabis sativa spray (Sativex): Δ9-THC/CBD = 2.7/2.5 mg per spray | 4 (6) | 2 (4) | 2 (9) |
| CBD 2,5,10% | 3 (4) | 1 (2) | 2 (9) |
| Epidiolex® | 3 (4) | 2 (4) | 1 (4) |
| No information | 17 (24) | 6 (13) | 11 (48) |
| Total | Total n=70, (%) |
Experience n=47, (%) |
No experience n=23, (%) |
|---|---|---|---|
| Effectiveness (physician reported) | |||
| Insignificant | 2 (3) | 1 (2) | 1 (4) |
| Weak | 12 (17) | 10 (21) | 2 (9) |
| Moderate | 35 (50) | 28 (60) | 7 (30) |
| Strong | 6 (9) | 4 (9) | 2 (9) |
| No response | 15 (21) | 4 (9) | 11 (48) |
| Short-term side effects | |||
| Yes | 29 (41) | 25 (53) | 4 (17) |
| Drowsiness | 14 (20) | 12 (26) | 2 (9) |
| Somnolence | 10 (14) | 9 (19) | 1 (4) |
| Fatigue | 6 (9) | 6 (13) | 0 (0) |
| Diarrhoea | 6 (9) | 6 (13) | 0 (0) |
| Nausea | 5 (7) | 4 (9) | 1 (4) |
| Anxiety | 4 (6) | 4 (9) | 0 (0) |
| Confusion | 4 (6) | 4 (9) | 0 (0) |
| Disorientation | 3 (4) | 3 (6) | 0 (0) |
| Euphoria | 3 (4) | 3 (6) | 0 (0) |
| Dry mouth | 2 (3) | 2 (4) | 0 (0) |
| Hallucination | 2 (3) | 2 (4) | 0 (0) |
| Vomiting | 2 (3) | 2 (4) | 0 (0) |
| Weakness | 1 (1) | 1 (2) | 0 (0) |
| Asthenia | 1 (1) | 1 (2) | 0 (0) |
| Depression | 1 (1) | 1 (2) | 0 (0) |
| Dizziness | 1 (1) | 1 (2) | 0 (0) |
| Paranoia | 1 (1) | 1 (2) | 0 (0) |
| Psychosis | 1 (1) | 1 (2) | 0 (0) |
| Hypotonia | 1 (1) | 1 (2) | 0 (0) |
| Increased liver enzymes | 1 (1) | 1 (2) | 0 (0) |
| No | 41 (59) | 22 (47) | 19 (83) |
| Long-term side effects | |||
| Yes | 0 (0) | 0 (0) | 0 (0) |
| No | 41 (59) | 35 (75) | 6 (26) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




