Submitted:
22 September 2023
Posted:
27 September 2023
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Methods
2.1. Bacterial cultures and media
2.2. L. monocytogenes antibiotic susceptibility testing
2.3. Detection of enterocin genes
2.3. Bacteriocin assay and spectrum of activity
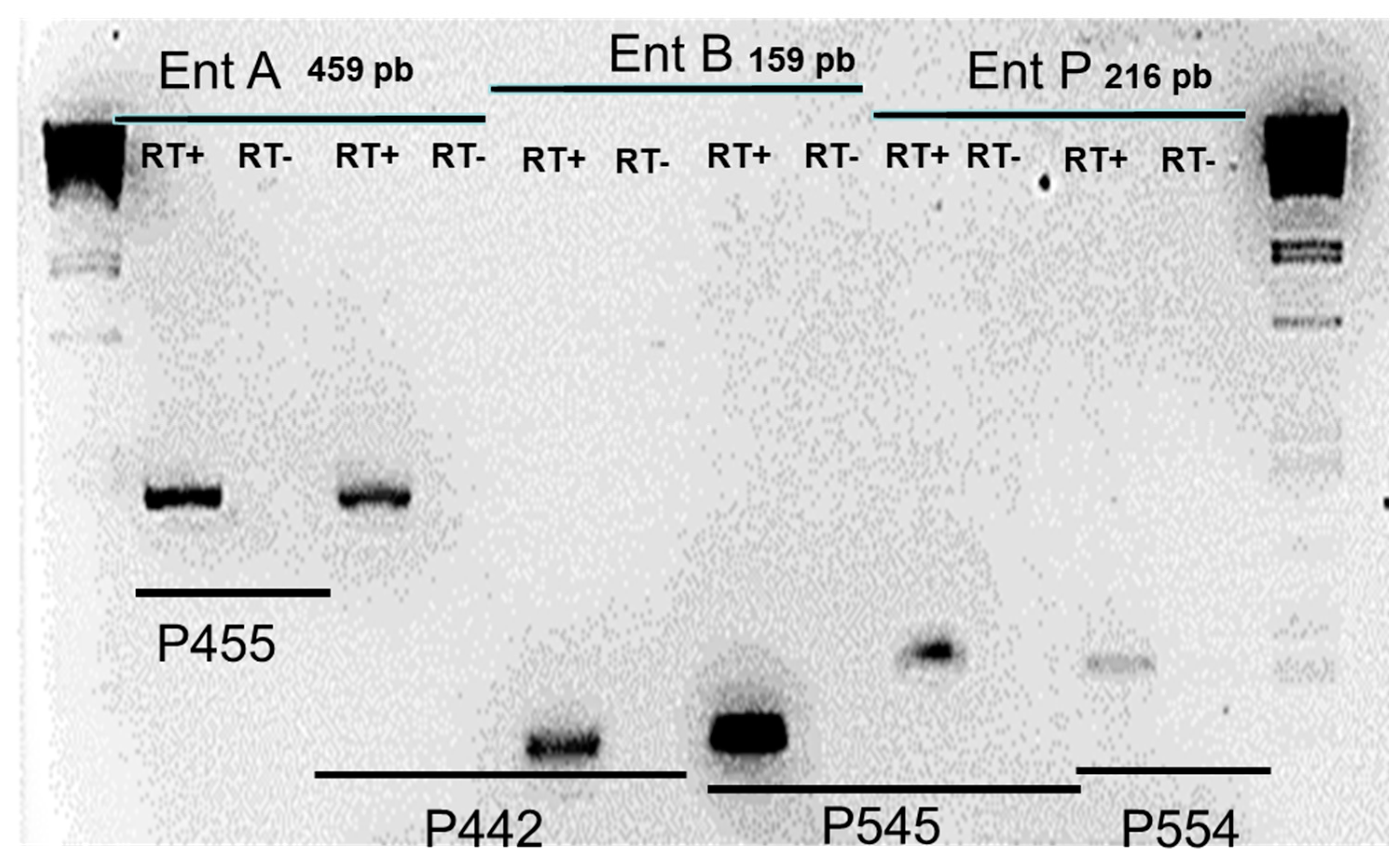
2.4. Total RNA isolation and reverse transcription (RT)-PCR analysis
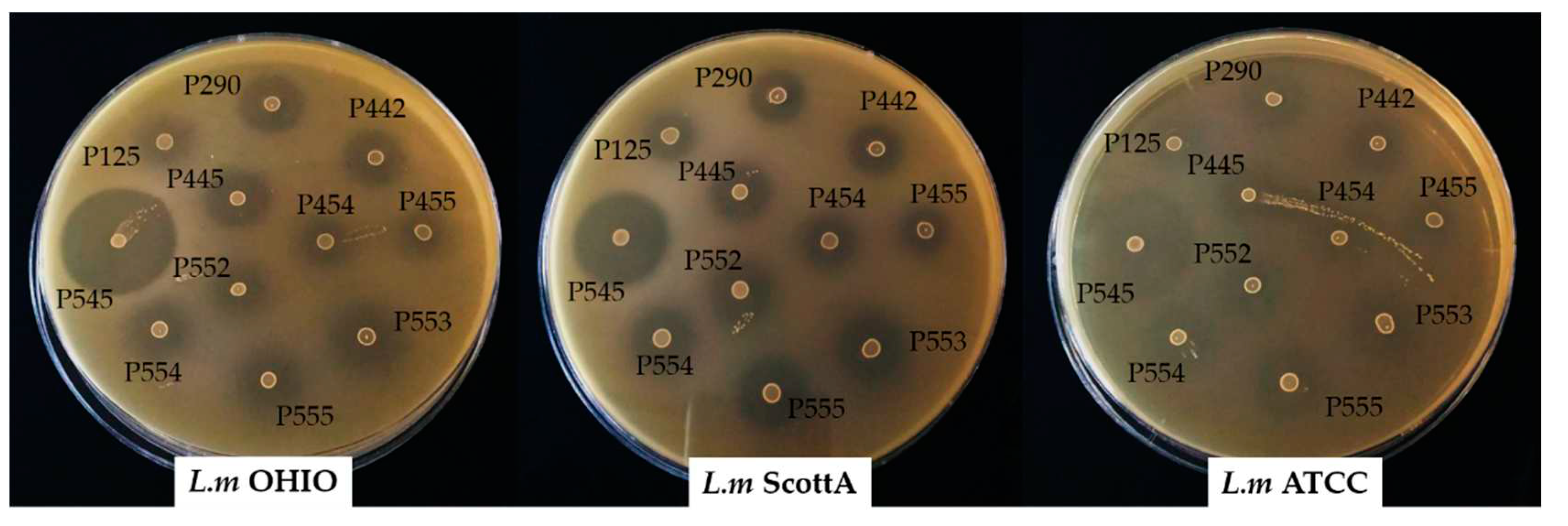
2.5. Overlay agar spot assay
3. Results
3.1. Detection of enterocin genes
3.2. Antibiotic susceptibility of L. monocytogenes
3.2. Antimicrobial activity
3.3. Transcriptional analysis of enterocins
3.4. Overlay agar spot assay
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McLauchlin, J.; Amar, C.F.L.; Grant, K.A. Neonatal cross-infection due to Listeria monocytogenes. Epidemiol Infect 2022, e77. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Al-Holy, M.A.; Shahbaz, H.M.; Al-Nabulsi, A.A.; Abu Ghoush, M.H.; Osaili, T.M.; Ayyash, M.M.; Holley, R.A. Emergence of antibiotic resistance in Listeria monocytogenes isolated from food products: a comprehensive review. Compr Rev Food Sci Food Saf 2018, 17, 1277–1292. [Google Scholar] [CrossRef]
- Wilking, H.; Lachmann, R.; Holzer, A.; Halbedel, S.; Flieger, A.; Stark, K. Ongoing high incidence and case-fatality rates for invasive listeriosis, Germany, 2010–2019. Emerg Infect Dis 2021, 27, 2485–2488. [Google Scholar] [CrossRef]
- Pohl, A.M.; Pouillot, R.; Bazaco, M.C.; Wolpert, B.J.; Healy, J.M.; Bruce, B.B.; Laughlin, M.E.; Hunter, J.C.; Dunn, J.R.; Hurd, S.; Rowlands, J.V.; Saupe, A.; Vugia, D.J.; Van Doren, J.M. Differences among incidence rates of invasive listeriosis in the U.S. FoodNet population by age, sex, race/ethnicity, and pregnancy status, 2008–2016. Foodborne Pathog Dis 2019, 16, 290–297. [Google Scholar] [CrossRef]
- Herrador, Z.; Gherasim, A.; Lopez-Velez, R.; Benito, A. Listeriosis in Spain based on hospitalisation records, 1997 to 2015: need for greater awareness. Euro Surveill 2019, 24, 1800271. [Google Scholar] [CrossRef]
- Li, W.; Bai, L.; Ma, X.; Zhang, X.; Li, X.; Yang, X.; Huang, J.Y.; Fanning, S.; Guo, Y. Sentinel listeriosis surveillance in selected hospitals, China, 2013–2017. Emerg Infect Dis 2019, 25, 2274–2277. [Google Scholar] [CrossRef]
- Zhang, X.; Niu, Y.; Liu, Y.; Lu, Z.; Wang, D.; Cui, X.; Chen, Q.; Ma, X. Isolation and characterization of clinical Listeria monocytogenes in Beijing, China, 2014–2016. Front Microbiol 2019, 10, 981. [Google Scholar] [CrossRef]
- Ke, Y.; Ye, L.; Zhu, P.; Sun, Y.; Zhu, Z. Listeriosis during pregnancy: a retrospective cohort study. BMC Pregnancy Childbirth 2022, 22, 261. [Google Scholar] [CrossRef]
- Wang, Z.; Tao, X.; Liu, S.; Zhao, Y.; Yang, X. An update review on Listeria infection in pregnancy. Infect Drug Resist 2021, 14, 1967–1978. [Google Scholar] [CrossRef]
- Layton, B.A.; Walters, S.P.; Lam, L.H.; Boehm, A.B. Enterococcus species distribution among human and animal hosts using multiplex PCR. J Appl Microbiol 2020, 109, 539–547. [Google Scholar] [CrossRef]
- Hanchi, H.; Mottawea, W.; Sebei, K.; Hammami, R. The genus Enterococcus: between probiotic potential and safety concerns—An update. Front Microbiol 2018, 9, 1791. [Google Scholar] [CrossRef] [PubMed]
- Martín, R.; Langa, S.; Reviriego, C.; Jiménez, E.; Marín, M.L.; Xaus, J.; Fernández, L.; Rodríguez, J.M. Human milk is a source of lactic acid bacteria for the infant gut. J Pediatr 2003, 143, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M.; Peirotén, A.; Medina, M.; Arqués, J.L.; Rodriguez, E. Virulence and antibiotic resistance of enterococci isolated from healthy breastfed infants. Microb Drug Resist 2018, 24, 63–69. [Google Scholar] [CrossRef]
- Francino, M.P. Antibiotics and the human gut microbiome: dysbioses and accumulation of resistances. Front Microbiol 2016, 6, 1543. [Google Scholar] [CrossRef]
- Lebreton, F.; van Schaik, W.; McGuire, M.A.; Godfrey, P.; Griggs, A.; Mazumdar, V.; Corander, J.; Cheng, L.; Saif, S.; Young, S.; Zeng, Q.; Wortman, J.; Birren, B.; Willems, R.J.L.; Earl, A.M.; Gilmore, M.S. Emergence of epidemic multidrug-resistant Enterococcus faecium from animal and commensal strains. mBio 2013, 4, e00534-13. [Google Scholar] [CrossRef] [PubMed]
- Neves, J.V. Editorial for Special Issue “Alternatives to Antibiotics: Bacteriocins and Antimicrobial Peptides”. Antibiotics 2022, 11, 860. [Google Scholar] [CrossRef] [PubMed]
- Cavera, V.L.; Arthur, T.D.; Kashtanov, D.; Chikindas, M.L. Bacteriocins and their position in the next wave of conventional antibiotics. Int J Antimicrob Agents 2015, 46, 494–501. [Google Scholar] [CrossRef]
- Cavicchioli, V.Q.; Camargo, A.C.; Todorov, S.D.; Nero, L.A. Novel bacteriocinogenic Enterococcus hirae and Pediococcus pentosaceus strains with antilisterial activity isolated from Brazilian artisanal cheese. J Dairy Sci 2017, 100, 2526–2535. [Google Scholar] [CrossRef]
- Moraes, P.M.; Perin, L.M.; Todorov, S.D.; Franco, S.D.; Silva, A.; Franco, B.D.G.M.; Nero, L.A. Bacteriocinogenic and virulence potential of Enterococcus isolates obtained from raw milk and cheese. J Appl Microbiol 2012, 113, 318–28. [Google Scholar] [CrossRef] [PubMed]
- Im, E.J.; Lee, H.H.; Kim, M.; Kim, M.K. Evaluation of enterococcal probiotic usage and review of potential health benefits, safety, and risk of antibiotic-resistant strain emergence. Antibiotics (Basel) 2023, 12, 1327. [Google Scholar] [CrossRef]
- Almeida-Santos, A.C.; Novais, C.; Peixe, L.; Freitas, A.R. Enterococcus spp. as a producer and target of bacteriocins: A double-edged sword in the antimicrobial resistance crisis context. Antibiotics 2021, 10, 1215. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, L.; Langa, S.; Martin, V.; Maldonado, A.; Jimenez, E.; Martin, R.; Rodríguez, J.M. The human milk microbiota: origin and potential roles in health and disease. Pharmacol Res 2013, 69, 1–10. [Google Scholar] [CrossRef] [PubMed]
- EUCAST Disk Diffusion Method for Antimicrobial Susceptibility Testing. Version 11.0 (January 2023). www.eucast.org.
- Martínez-Bueno, M.; Valdivia, E.; Gálvez, A.; Coyett, e J.; Maqueda, M. Analysis of the gene cluster involved in production and immunity of the peptide antibiotic AS-48 in Enterococcus faecalis. Molecular Microbiol 1998, 27, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Hidalgo, M.; Maqueda, M.; Gálvez, A.; Abriouel, H.; Valdivia, E.; Martínez-Bueno, M. The genes coding for enterocin EJ97 production by Enterococcus faecalis EJ97 are located on a conjugative plasmid. Appl Environ Microbiol 2003, 69, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Aymerich, T.; Holo, H.; Havarstein, L.S.; Hugas, M.; Garriga, M.; Nes, I.F. Biochemical and genetic characterization of enterocin A from Enterococcus faecium, a new antilisterial bacteriocin in the pediocin family of bacteriocins. Appl Environ Microbiol 1996, 62, 1676–1682. [Google Scholar] [CrossRef] [PubMed]
- Du Toit, M.; Franz, C.; Dicks, L.M.T; Holzapfel, W.H. Preliminary characterization of bacteriocins produced by Enterococ,cus faecium and Enterococcus faecalis isolated from pig faeces. J Applied Microbiol 2000, 88, 482–494. [Google Scholar] [CrossRef]
- Ruiz-Rodríguez, M.; Martínez-Bueno, M.; Martín-Vivaldi, M.; Valdivia, E.; Soler, J.J. Bacteriocins with a broader antimicrobial spectrum prevail in enterococcal symbionts isolated from the hoopoe’s uropygial gland. FEMS Microbiol Ecol 2013, 85, 495–502. [Google Scholar] [CrossRef]
- Yousif, N.M.K.; Dawyndt, P.; Abriouel, H.; Wijaya, A.; Schillinger, U.; Vancanneyt, M.; Swing, s J.; Dirar, H.A. Molecular characterization, technological properties and safety aspects of enterococci from ‘Hussuwa’, an African fermented sorghum product. J Appl Microbiol 2005, 98, 216–228. [Google Scholar] [CrossRef]
- De Vuyst, L.; Moreno, M.F.; Revets, H. Screening for enterocins and detection of hemolysin and vancomycin resistance in enterococci of different origins. Int J Food Microbiol 2003, 84, 299–318. [Google Scholar] [CrossRef]
- Pangallo, D.; Harichová, J.; Karelová, E.; Drahovská, H.; Chovanová, K.; Ferianc, P. Timko, J. Molecular investigation of enterococci isolated from different environmental sources. Biologia 2004, 59, 829–837. [Google Scholar]
- Özdemir, G.B.; Oryasm, E.; Biyik, H.H.; Özteber, M.; Bozdoğan, B. Phenotypic and genotypic characterization of bacteriocins in enterococcal isolates of different sources. Ind J Microbiol 2011, 51, 182–187. [Google Scholar] [CrossRef]
- Poeta, P.; Costa, D.; Rojo-Bezares, B.; Zarazaga, M.; Klibi, N.; Rodrigues, J.; Torres, C. Detection of antimicrobial activities and bacteriocin structural genes in faecal enterococci of wild animals. Microbiol Res 2007, 162, 257–263. [Google Scholar] [CrossRef]
- Casaus, F.; Nilsen, T.; Cintas, L.M.; Nes, L.F.; Hernández, P.E.; Holo, H. Enterocin B, a new bacteriocin from Enterococcus faecium TI36 which can act synergistically with enterocin A. Microbiology 1997, 143, 2287–2294. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Worobo, R.W.W.; Quadri, L.E.N.; Schillinger, U.; Holzapfel, W.H.; Vederas, J.C.; Stiles, M.E. Atypical genetic locus associated with constitutive production of enterocin B by Enterococcus faecium BFE 900. Appl Environ Microbiol 1999, 65, 2170–2178. [Google Scholar] [CrossRef] [PubMed]
- Foulquié Moreno, M.R.; Callewaert, R.; Devreese, B.; Van Beeumen, J.; De Vuyst, L. Isolation and biochemical characterisation of enterocins produced by enterococci from different sources. J Appl Microbiol 2003, 94, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Arqués, J.L.; Rodríguez, E.; Langa, S.; Landete, J.M.; Medina, M. Antimicrobial activity of lactic acid bacteria in dairy products and gut: effect on pathogens. Biomed Res Int 2015, 584183. [Google Scholar] [CrossRef] [PubMed]
- Ogaki, M.B.; Rocha, K.R.; Terra, M.R.; Furlaneto, M.C.; Furlaneto-Maia, L. Screening of the enterocin-encoding genes and antimicrobial activity in Enterococcus species. J Microbiol Biotechnol 2016, 26, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Darbandi, A.; Asadi, A.; Ari, M.M.; Ohadi, E.; Talebi, M.; Zadeh, M.H.; Emamie, A.D.; Ghanavati, R.; Kakanj, M. Bacteriocins: Properties and potential use as antimicrobials. J Clin Lab Anal 2022, 36, 24093. [Google Scholar] [CrossRef]
- Wu, Y.; Pang, X.; Wu, Y.; Liu, X.; Zhang, X. Enterocins: Classification, Synthesis, Antibacterial Mechanisms and Food Applications. Molecules 2022, 27, 2258. [Google Scholar] [CrossRef]
- Zdolec, N; Kiš, Marta. Antimicrobial properties of food enterococci. In Lactic Acid Bacteria in Food Biotechnology. Innovations and Functional Aspects.; Ray, R.C., Paramithiotis, P., Azevedo, V.A.C., Montet, D., Eds.; 2022; Volume 11, pp. 195–203. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Huch, M.; Abriouel, H.; Holzapfel, W.; Gálvez, A. Enterococci as probiotics and their implications in food safety. Int J Food Microbiol 2011, 151, 125–40. [Google Scholar] [CrossRef]
- Perez, R.H.; Himeno, K.; Ishibashi, N.; Masuda, Y.; Zendo, T.; Fujita, K.; Wilaipun, P.; Leelawatcharamas, V.; Nakayama, J.; Sonomoto, K. Monitoring of the multiple bacteriocin production by Enterococcus faecium NKR-5-3 through a developed liquid chromatography and mass spectrometry-based quantification system. J Biosci Bioeng 2012, 114, 490–6. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.; Guarner, F.; Bustos Fernandez, L.; Maruy, A.; Sdepanian, V.L.; Cohen, H. Antibiotics as major disruptors of gut microbiota. Front. Cell. Infect. Microbiol 2020, 10, 572912. [Google Scholar] [CrossRef]
- Gensollen, T.; Iyer, S.S.; Kasper, D.L.; Blumberg, R.S. How colonization by microbiota in early life shapes the immune system. Science 2016, 352, 539–544. [Google Scholar] [CrossRef]
- He, Y.; Yang, Q.; Tian, L.; Zhang, Z.; Qiu, L; Tao, X.; Wei, H. Protection of surface layer protein from Enterococcus faecium WEFA23 against Listeria monocytogenes CMCC54007 infection by modulating intestinal permeability and immunity. Appl Microbiol Biotechnol 2021, 105, 4269–4284. [Google Scholar] [CrossRef] [PubMed]
- Popović, N.; Djokić, J.; Brdarić, E.; Dinić, M.; Terzić-Vidojević, A.; Golić, N.; Veljović, K. The influence of heat-killed Enterococcus faecium BGPAS1-3 on the tight junction protein expression and immune function in differentiated Caco-2 cells infected with Listeria monocytogenes ATCC 19111. Front Microbiol 2019, 10, 412. [Google Scholar] [CrossRef] [PubMed]
- Rajput, K.; Dubey, R.C.; Kumar, A. Probiotic potential and immunomodulatory properties in Enterococcus faecium GMB24 and Enterococcus hirae SMB16 isolated from goat and sheep milk. Arch Microbiol 2022, 204, 619. [Google Scholar] [CrossRef]
- Salvucci, E.; Saavedra, L.; Hebert, E.M.; Haro, C.; Sesma, F. Enterocin CRL35 inhibits Listeria monocytogenes in a murine model. Foodborne Pathog Dis 2012, 9, 68–74. [Google Scholar] [CrossRef]
- Song, MW; Kim, K-T; Paik, H-D. Probiotics as a functional health supplement in infant formulas for the improvement of intestinal microflora and immunity. Food Rev Intl. 2021, 39, 858–74. [Google Scholar] [CrossRef]
| Gene | Primers | Sequence (5`to 3`) | Product size (pb) | References |
|---|---|---|---|---|
| EJ97 | E21-4 | GCAGCTAAGCTAACGACT | 279 | [24] |
| E21-9 | AGGGGAATTTGAACAGA | |||
| A | P9 | GAGATTTATCTCCATAATCT | 452 | [25] |
| P10 | GTACCACTCATAGTGGAA | |||
| B | EntB(f) | GAAAATGATCACAGAATGCCTA | 159 | [26] |
| EntB(r) | GTTGCATTTAGAGTATACATTTG | |||
| P | EntP1 | ATGAGAAAAAAATTATTTAGTTT | 216 | [27] |
| EntP2 | TTAATGTCCCATACCTGCCAAACC | |||
| MR10A | LICIJ1 | ATGGGAGCAATCGCAAAA | 135 | [28] |
| LICJ2A | TTAAATATGTTTTTTAATCCA | |||
| L50 | L50F | ATGGGAGCAATCGCAAAATTAG | 98 | [29] |
| L50R | ATTGCCCATCCTTCTCCAAT | |||
| AS-48 | As48-1 | AATAAACTACATGGGT | 377 | [24] |
| As-48-5 | CCAAGCAATAACTGCTCTTT |
| Strain | Enterocin | Gene1 | Inhibition EU/mL2 | ||
|---|---|---|---|---|---|
| expression | L.m OHIO | L.m ScottA | L.m ATCC | ||
| E. faecium INIA P125 | A | A (-) | - | - | - |
| E. faecalis INIA P290 | AS48 | AS48 (++) | 2560 | 1280 | 2560 |
| E. faecium INIA P442 | A, B | A (++), B (+++) | 5120 | 2560 | 5120 |
| E. faecium INIA P445 | A, B | A (+), B (++) | 2560 | 1280 | 2560 |
| E. faecium INIA P454 | A, B | A (+), B (-) | - | - | - |
| E. faecium INIA P455 | A | A (++) | - | - | - |
| E. faecium INIA P545 | B, P | B (++++), P (+) | 81920 | 10240 | 81920 |
| E. faecium INIA P552 | A | A (-) | - | - | - |
| E. faecium INIA P553 | P | P (++) | - | - | - |
| E. faecium INIA P554 | P | P(+) | - | - | - |
| E. faecium INIA P555 | P | P(+) | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




