Submitted:
11 September 2023
Posted:
12 September 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study site
2.2. Experimental design
2.3. Selection of treatments
2.4. Soil sampling and analysis
2.5. Soil chemical analyses
2.6. Determination of microbial biomass carbon
2.7. Deoxyribonucleic acid (DNA) extraction from soil
2.7.1. Soil DNA sequencing, bioinformatics sequence processing and taxonomic identification
2.7.2. Identification of the potential zinc solubilizing microbial species
2.8. Statistical data analysis
3. Results
3.1. Effects of management practices on soil chemical and physical characteristics in INM3 site
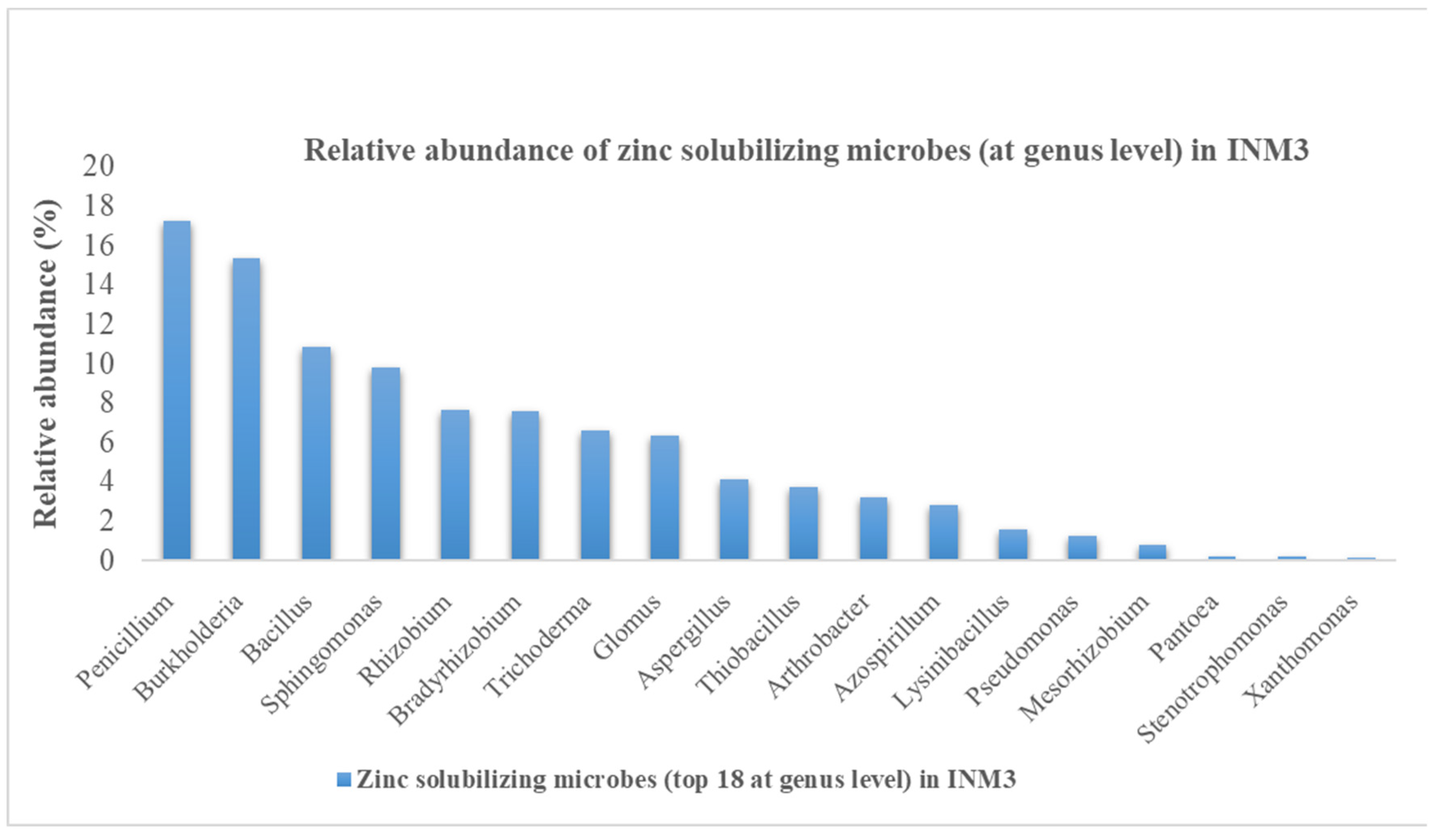
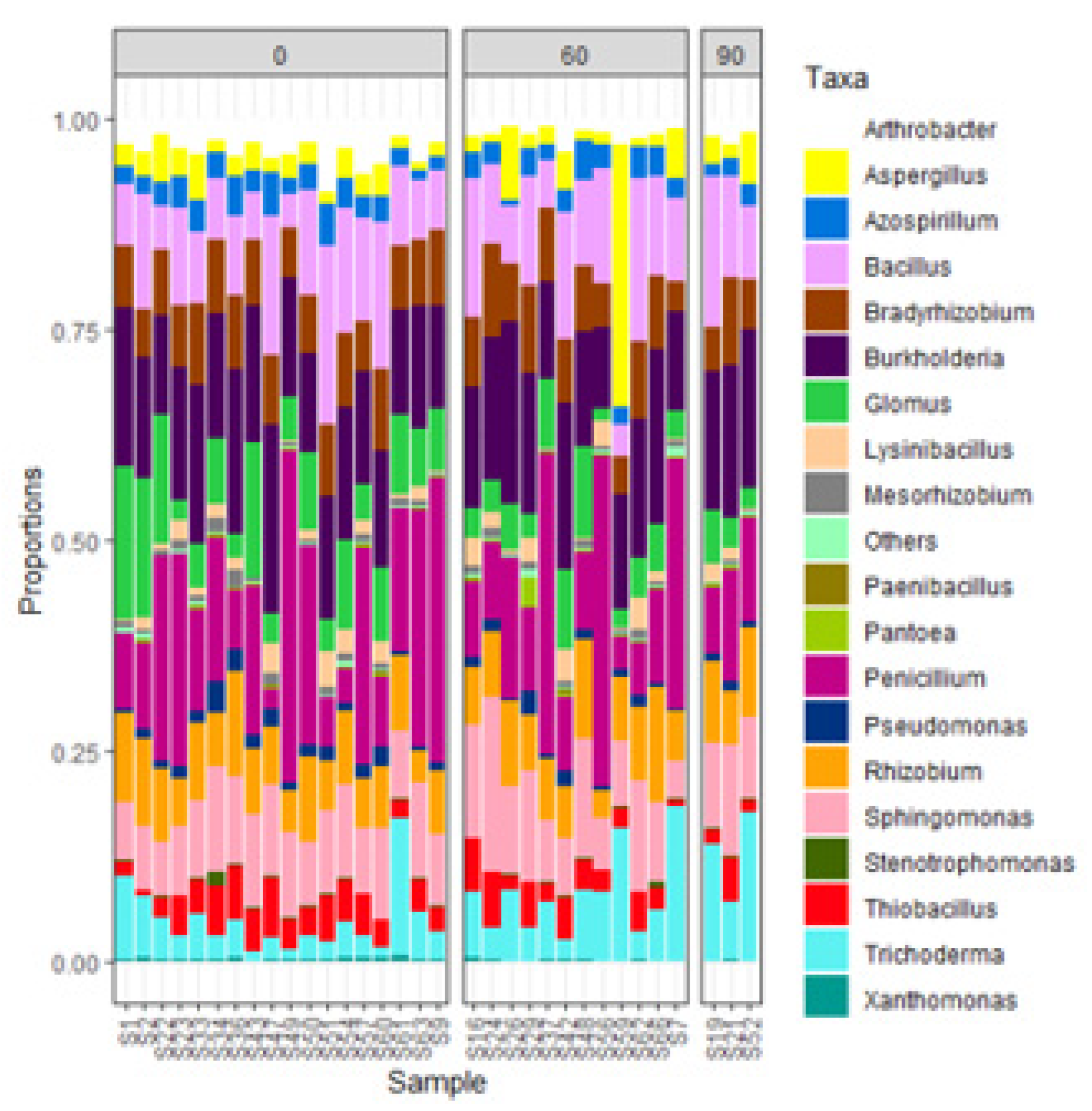
3.2. Overall microbial abundance and ZSM species abundance in INM3
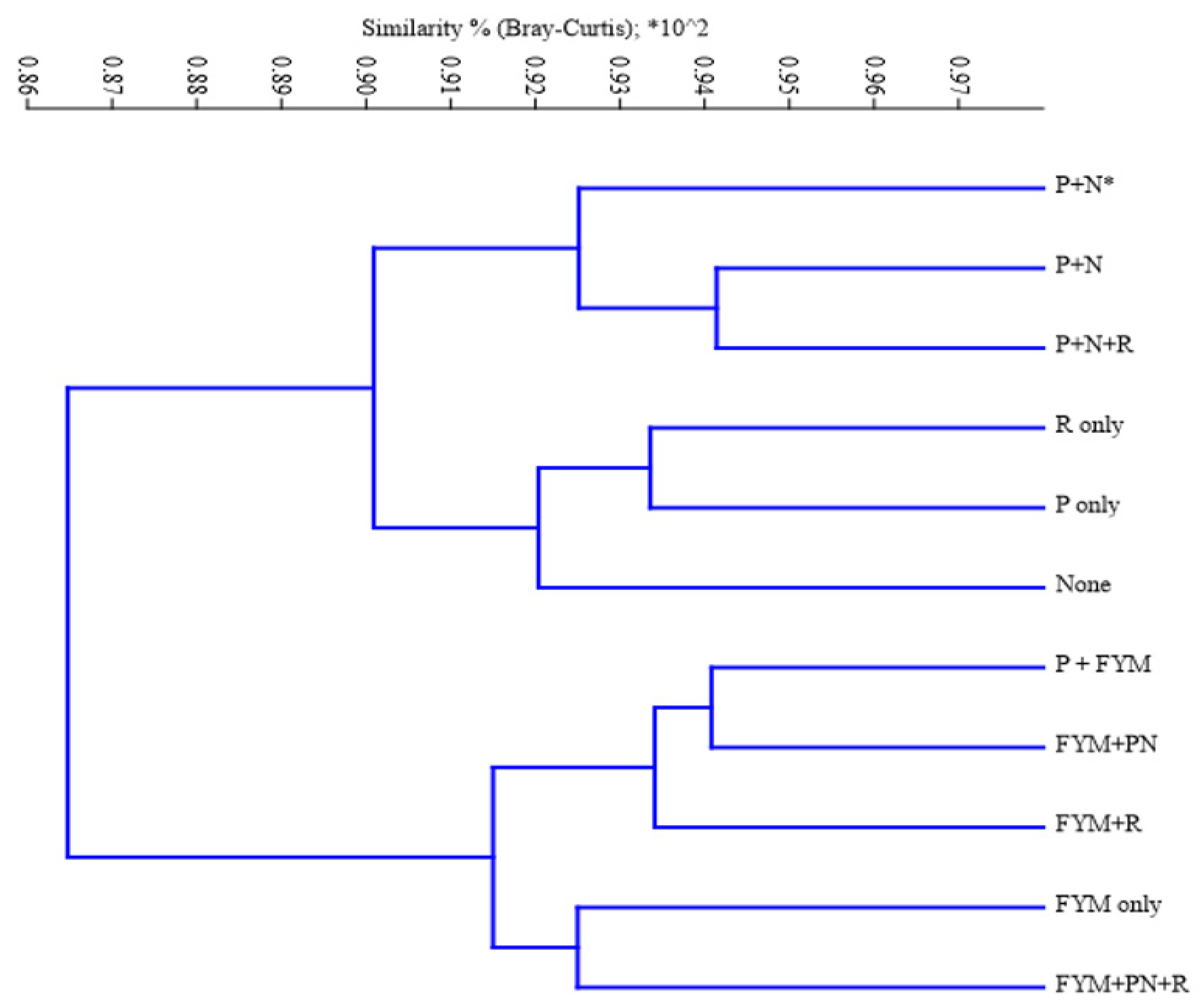
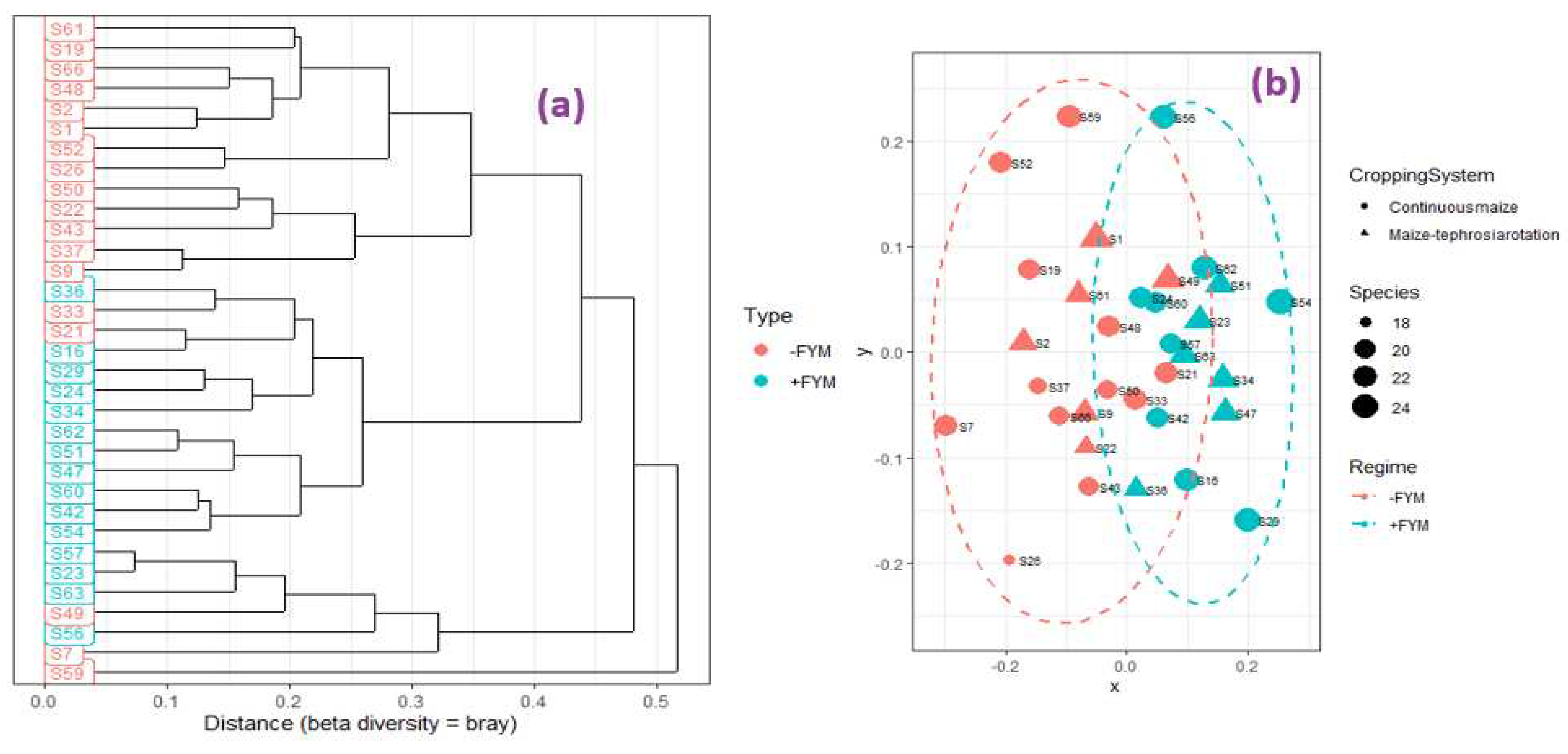
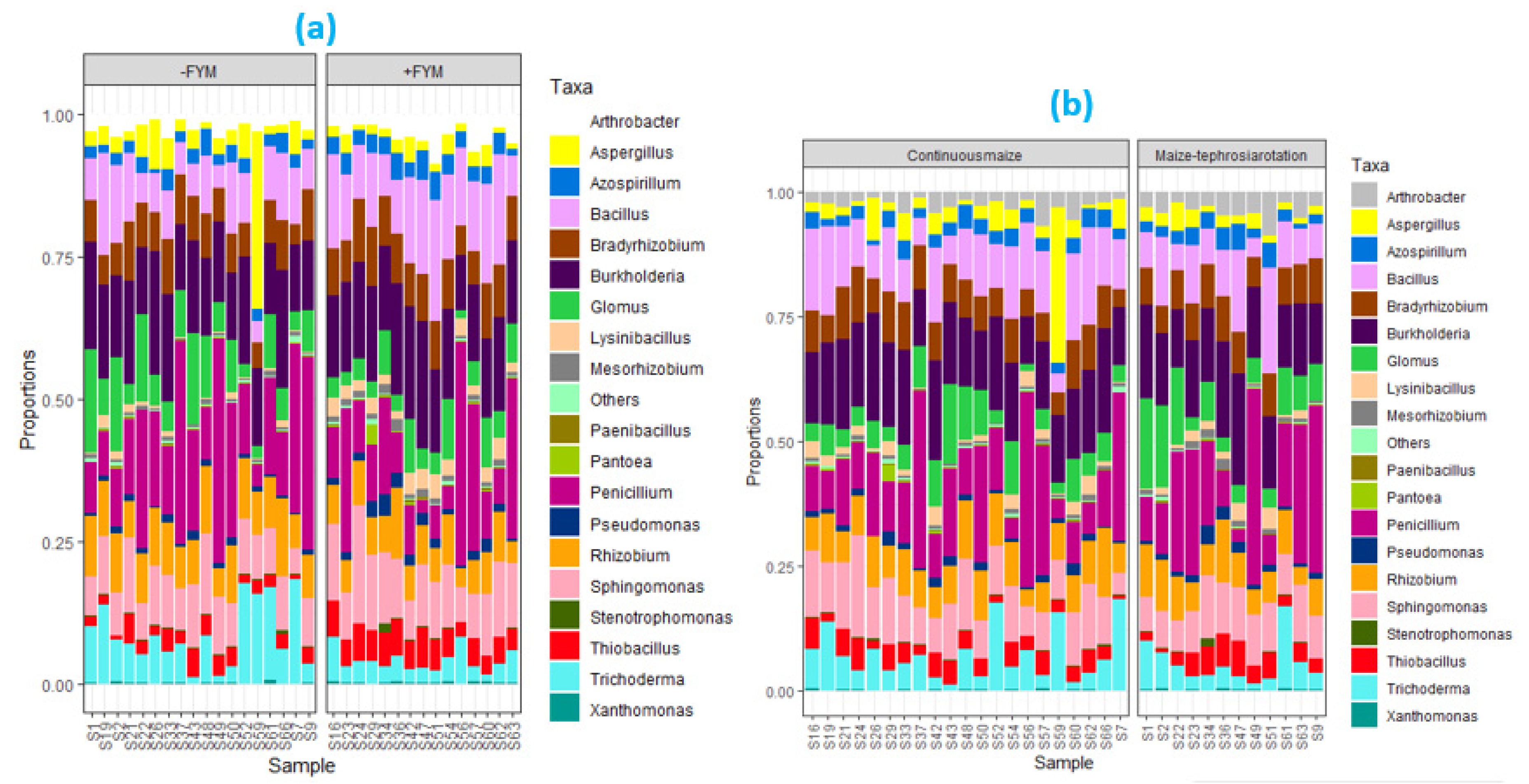
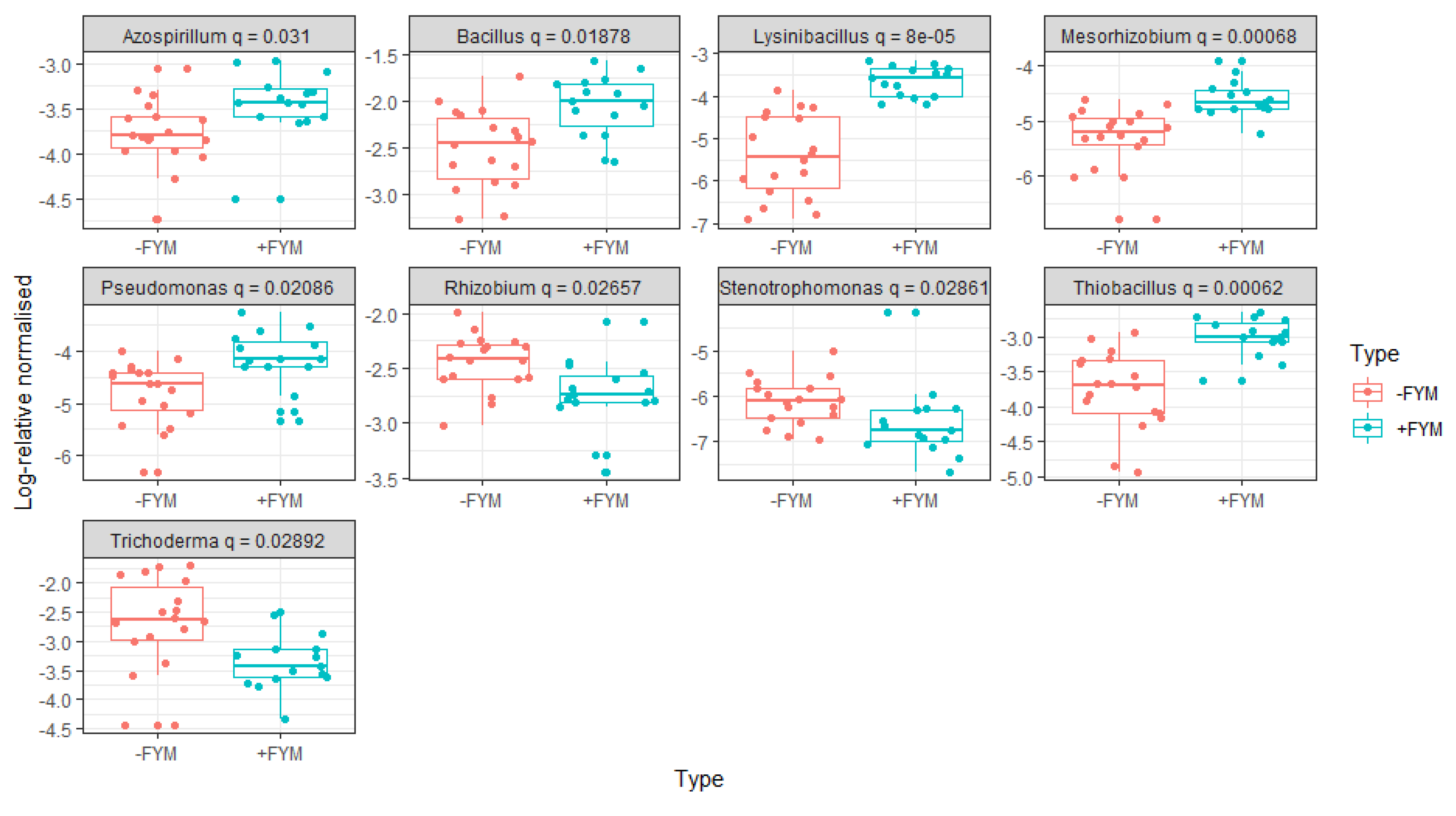
3.3. Effect of agronomic management practices on ZSM species
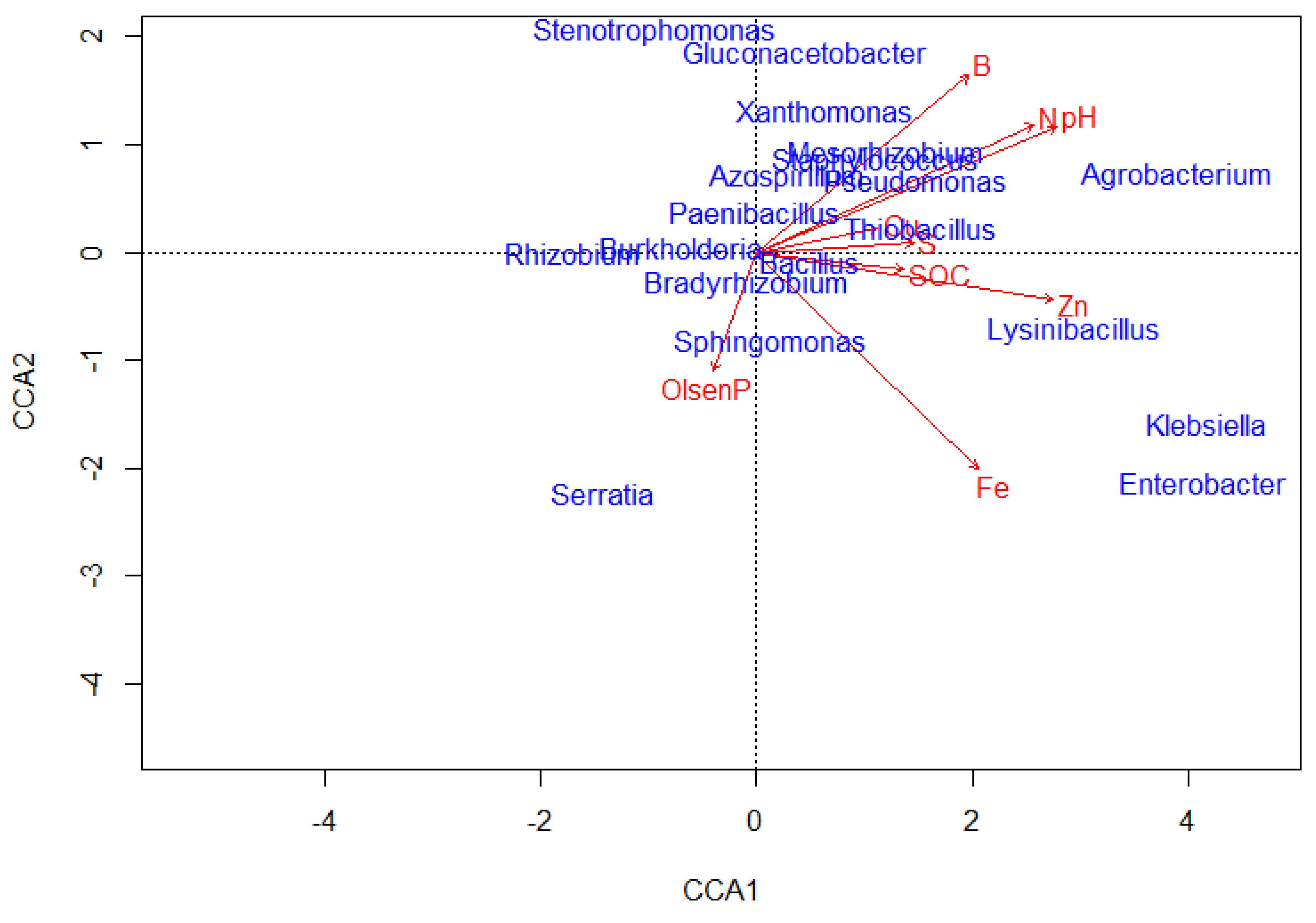
3.3. Relationship between soil chemical characteristics and ZSM abundance
4. Discussion
4.1. Influence of soil chemical properties on zinc solubilizing microbial species
4.2. Stimulation of zinc solubilizing microbial species with FYM application
5. Conclusions
Acknowledgments
References
- Alori, E. T., Glick, B. R., & Babalola, O. O. (2017). Microbial phosphorus solubilization and its potential for use in sustainable agriculture. Frontiers in microbiology, 8, 971. [CrossRef]
- Anuradha, P., Syed, I., Swati, M., & Patil, V. D. (2015). Solubilization of insoluble zinc compounds by different microbial isolates in vitro condition. International Journal of Tropical Agriculture, 33(2 (Part II)), 865-869.
- Ashworth, A. J., DeBruyn, J. M., Allen, F. L., Radosevich, M., & Owens, P. R. (2017). Microbial community structure is affected by cropping sequences and poultry litter under long-term no-tillage. Soil Biology and Biochemistry, 114, 210-219. [CrossRef]
- Ayaga, G., Todd, A., & Brookes, P. C. (2006). Enhanced biological cycling of phosphorus increases its availability to crops in low-input sub-Saharan farming systems. Soil Biology and Biochemistry, 38(1), 81-90. [CrossRef]
- Babin, D., Deubel, A., Jacquiod, S., Sørensen, S. J., Geistlinger, J., Grosch, R., & Smalla, K. (2019). Impact of long-term agricultural management practices on soil prokaryotic communities. Soil Biology and Biochemistry, 129, 17-28. [CrossRef]
- Balota, E. L., Colozzi Filho, A., Andrade, D. S., & Dick, R. P. (2004). Long-term tillage and crop rotation effects on microbial biomass and C and N mineralization in a Brazilian Oxisol. Soil and Tillage Research, 77(2), 137-145. [CrossRef]
- Bolo, P., Kihara, J., Mucheru-Muna, M., Njeru, E. M., Kinyua, M., & Sommer, R. (2021). Application of residue, inorganic fertilizer and lime affect phosphorus solubilizing microorganisms and microbial biomass under different tillage and cropping systems in a Ferralsol. Geoderma, 390, 114962. [CrossRef]
- Cai, A., Xu, M., Wang, B., Zhang, W., Liang, G., Hou, E., & Luo, Y. (2019). Manure acts as a better fertilizer for increasing crop yields than synthetic fertilizer does by improving soil fertility. Soil and Tillage Research, 189, 168-175. [CrossRef]
- Cakmak, I. (2008). Enrichment of cereal grains with zinc: agronomic or genetic biofortification?. Plant and soil, 302(1), 1-17. [CrossRef]
- Cakmak, I., McLaughlin, M. J., & White, P. (2017). Zinc for better crop production and human health. Plant and Soil, 411, 1–4. [CrossRef]
- Caporaso, J. G., Lauber, C. L., Walters, W. A., Berg-Lyons, D., Huntley, J., Fierer, N., Owens, S.M., Betley, J., Fraser, L., Bauer, M., Gormley, N, Gilbert, J.A, Smith, G., & Knight, R. (2012). Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. The ISME journal, 6(8), 1621-1624. [CrossRef]
- Dhaked, B. S., Triveni, S., Reddy, R. S., & Padmaja, G. (2017). Isolation and screening of potassium and zinc solubilizing bacteria from different rhizosphere soil. Int. J. Curr. Microbiol. App. Sci, 6(8), 1271-1281. [CrossRef]
- Fasim, F., Ahmed, N., Parsons, R., & Gadd, G. M. (2002). Solubilization of zinc salts by a bacterium isolated from the air environment of a tannery. FEMS microbiology letters, 213(1), 1-6. [CrossRef]
- Gautam, A., Sekaran, U., Guzman, J., Kovács, P., Hernandez, J. L. G., & Kumar, S. (2020). Responses of soil microbial community structure and enzymatic activities to long-term application of mineral fertilizer and beef manure. Environmental and Sustainability Indicators, 8, 100073. [CrossRef]
- Giller, K. E., Hijbeek, R., Andersson, J. A., & Sumberg, J. (2021). Regenerative Agriculture: An agronomic perspective. Outlook on Agriculture, 50(1), 13-25. [CrossRef]
- Gupta, N., Ram, H., & Kumar, B. (2016). Mechanism of Zinc absorption in plants: uptake, transport, translocation and accumulation. Reviews in Environmental Science and Bio/Technology, 15(1), 89-109. [CrossRef]
- Hacisalihoglu, G. (2020). Zinc (Zn): The last nutrient in the alphabet and shedding light on zn efficiency for the future of crop production under suboptimal zn. Plants, 9(11), 1471. [CrossRef]
- Hammer, Ø., Harper, D. A., & Ryan, P. D. (2001). PAST: Paleontological statistics software package for education and data analysis. Palaeontologia electronica, 4(1), 9.
- Hodge, A., & Fitter, A. H. (2010). Substantial nitrogen acquisition by arbuscular mycorrhizal fungi from organic material has implications for N cycling. Proceedings of the National Academy of Sciences, 107(31), 13754-13759. [CrossRef]
- Hussain, A., Zahir, Z. A., Asghar, H. N., Ahmad, M., Jamil, M., Naveed, M., & Akhtar, M. F. U. Z. (2018). Zinc solubilizing bacteria for zinc biofortification in cereals: a step toward sustainable nutritional security. In Role of rhizospheric microbes in soil (pp. 203-227). Springer, Singapore. [CrossRef]
- Hussain, A., Zahir, Z.A., Ditta, A., Tahir, M.U., Ahmad, M., Mumtaz, M.Z., Hayat, K. & Hussain, S. (2020). Production and implication of bio-activated organic fertilizer enriched with zinc-solubilizing bacteria to boost up maize (Zea mays L.) production and biofortification under two cropping seasons. Agronomy, 10(1), p.39. [CrossRef]
- Jama, B., Palm, C. A., Buresh, R. J., Niang, A., Gachengo, C., Nziguheba, G., & Amadalo, B. (2000). Tithonia diversifolia as a green manure for soil fertility improvement in western Kenya: a review. Agroforestry systems, 49(2), 201-221. [CrossRef]
- Kalayu, G. (2019). Phosphate solubilizing microorganisms: promising approach as biofertilizers. International Journal of Agronomy, 2019. [CrossRef]
- Kearney, S. G., Carwardine, J., Reside, A.E., Adams, V.M., Nelson, R., Coggan, A., Spindler, R. & Watson, J. E. (2022). Saving species beyond the protected area fence: Threats must be managed across multiple land tenure types to secure Australia’s endangered species. Conservation Science and Practice, e617. [CrossRef]
- Khande, R., Sharma, S. K., Ramesh, A., & Sharma, M. P. (2017). Zinc solubilizing Bacillus strains that modulate growth, yield and zinc biofortification of soybean and wheat. Rhizosphere, 4, 126-138. [CrossRef]
- Khanghahi, M. Y., Ricciuti, P., Allegretta, I., Terzano, R., & Crecchio, C. (2018). Solubilization of insoluble zinc compounds by zinc solubilizing bacteria (ZSB) and optimization of their growth conditions. Environmental Science and Pollution Research, 25(26), 25862-25868. [CrossRef]
- Kihanda, F. M., Warren, G. P. & Micheni, A. N. (2006). Effect of manure application on crop yield and soil chemical properties in a long-term field trial of semi-arid Kenya. Nutrient Cycling in Agroecosystems 76:341–354. [CrossRef]
- Kihara, J. M. (2009). Conservation tillage in Kenya: the biophysical processes affecting its effectiveness. Center for Development Research, PHD (2009), p. 141.
- Kihara, J., Bolo, P., Kinyua, M., Rurinda, J., & Piikki, K. (2020). Micronutrient deficiencies in African soils and the human nutritional nexus: opportunities with staple crops. Environmental geochemistry and health, 1-19. [CrossRef]
- Kihara, J., Martius, C., Bationo, A., Thuita, M., Lesueur, D., Herrmann, L., Amelung, W. & Vlek, P. L. (2012). Soil aggregation and total diversity of bacteria and fungi in various tillage systems of sub-humid and semi-arid Kenya. Applied Soil Ecology, 58, 12-20. [CrossRef]
- Klassen-Wigger, P., Geraets, M., Messier, M. C., Detzel, P., Lenoble, H. P., & Barclay, D. V. (2018). Micronutrient fortification of bouillon cubes in Central and West Africa. In Food Fortification in a Globalized World (pp. 363-372). Academic Press. [CrossRef]
- Koorem, K., Gazol, A., Öpik, M., Moora, M., Saks, Ü., Uibopuu, A., Sõber, V., & Zobel, M. (2014). Soil nutrient content influences the abundance of soil microbes but not plant biomass at the small-scale. PLoS One, 9(3), e91998. [CrossRef]
- Kushwaha, P., Kashyap, P. L., Pandiyan, K., & Bhardwaj, A. K. (2020). Zinc-Solubilizing Microbes for Sustainable Crop Production: Current Understanding, Opportunities, and Challenges. In Phytobiomes: Current Insights and Future Vistas (pp. 281-298). Springer, Singapore. [CrossRef]
- Li, H. Y., Zhu, Y. G., Smith, S. E., & Smith, F. A. (2003). Phosphorus–zinc interactions in two barley cultivars differing in phosphorus and zinc efficiencies. Journal of plant nutrition, 26(5), 1085-1099. [CrossRef]
- Lian, T., Mu, Y., Jin, J., Ma, Q., Cheng, Y., Cai, Z., & Nian, H. (2019). Impact of intercropping on the coupling between soil microbial community structure, activity, and nutrient-use efficiencies. PeerJ, 7, e6412. [CrossRef]
- Lladó S., & Baldrian P (2017) Community-level physiological profiling analyses show potential to identify the copiotrophic bacteria present in soil environments. PLoS One 12:e0171638. [CrossRef]
- Loneragan, J. F., & Webb, M. J. (1993). Interactions between zinc and other nutrients affecting the growth of plants. In Zinc in soils and plants (pp. 119-134). Springer, Dordrecht. [CrossRef]
- Margenot, A. J., Sommer, R., Mukalama, J., & Parikh, S. J. (2017). Biological P cycling is influenced by the form of P fertilizer in an Oxisol. Biology and Fertility of Soils, 53(8), 899-909. [CrossRef]
- Mucheru-Muna, M., Mugendi, D., Pypers, P., Mugwe, J., Kung’u, J., Vanlauwe, B., & Merckx, R. (2014). Enhancing maize productivity and profitability using organic inputs and mineral fertilizer in central Kenya small-hold farms. Experimental Agriculture, 50(2), 250-269. [CrossRef]
- Mugwe, J., Mugendi, D., Mucheru-Muna, M., Odee, D., & Mairura, F. (2009). Effect of selected organic materials and inorganic fertilizer on the soil fertility of a Humic Nitisol in the central highlands of Kenya. Soil Use and Management, 25(4), 434-440. [CrossRef]
- Mumtaz, M. Z., Ahmad, M., Jamil, M., & Hussain, T. (2017). Zinc solubilizing Bacillus spp. potential candidates for biofortification in maize. Microbiological research, 202, 51-60. [CrossRef]
- Niu, H., Pang, Z., Fallah, N., Zhou, Y., Zhang, C., Hu, C., Lin, W. & Yuan, Z. (2021). Diversity of microbial communities and soil nutrients in sugarcane rhizosphere soil under water soluble fertilizer. Plos one, 16(1), e0245626. [CrossRef]
- Orwa, P., Mugambi, G., Wekesa, V., & Mwirichia, R. (2020). Isolation of haloalkaliphilic fungi from Lake Magadi in Kenya. Heliyon, 6(1), e02823. [CrossRef]
- Pasley, H. R., Cairns, J. E., Camberato, J. J., & Vyn, T. J. (2019). Nitrogen fertilizer rate increases plant uptake and soil availability of essential nutrients in continuous maize production in Kenya and Zimbabwe. Nutrient cycling in agroecosystems, 115(3), 373-389. [CrossRef]
- Paul, B. K., Vanlauwe, B., Ayuke, F., Gassner, A., Hoogmoed, M., Hurisso, T.T., Koala, S., Lelei, D., Ndabamenye, T., Six, J. and Pulleman, M.M. (2013). Medium-term impact of tillage and residue management on soil aggregate stability, soil carbon and crop productivity. Agriculture, ecosystems & environment, 164, 14-22. [CrossRef]
- Prasad, R., Shivay, Y. S., & Kumar, D. (2016). Interactions of zinc with other nutrients in soils and plants-A Review. Indian Journal of Fertilisers, 12(5), 16-26.
- R Development Core Team R: A Language and Environment for Statistical Computing R Foundation for Statistical Computing, Vienna (2016).
- Rani, N., Kaur, R., & Kaur, S. (2020). Zinc solubilising bacteria to augment soil fertility–a comprehensive review. Int J AgriSci Vet Med, 8(1), 38-44.
- Rawat, N., Neelam, K., Tiwari, V. K., & Dhaliwal, H. S. (2013). Biofortification of cereals to overcome hidden hunger. Plant Breeding, 132(5), 437-445. [CrossRef]
- Rengel, Z., Batten, G. D., & Crowley, D. D. (1999). Agronomic approaches for improving the micronutrient density in edible portions of field crops. Field crops research, 60(1-2), 27-40. [CrossRef]
- Rezaeiniko, B., Enayatizamir, N., & Norouzi Masir, M. (2019). The Effect of Zinc Solubilizing Bacteria on Zinc Uptake and Some Properties of Wheat in the Greenhouse. JWSS-Isfahan University of Technology, 22(4), 249-260. [CrossRef]
- Sadeghzadeh, B. (2013). A review of zinc nutrition and plant breeding. Journal of soil science and plant nutrition, 13(4), 905-927. [CrossRef]
- Saini, P., Nagpal, S., Saini, P., Kumar, A., & Gani, M. (2021). Microbial Mediated Zinc Solubilization in Legumes for Sustainable Agriculture. Phytomicrobiome Interactions and Sustainable Agriculture, 254-276. [CrossRef]
- Saravanan, V. S., Madhaiyan, M., & Thangaraju, M. (2007). Solubilization of zinc compounds by the diazotrophic, plant growth promoting bacterium Gluconacetobacter diazotrophicus. Chemosphere, 66(9), 1794-1798. [CrossRef]
- Tang, H., Li, C., Xiao, X., Shi, L., Cheng, K., Wen, L., & Li, W. (2020). Effects of short-term manure nitrogen input on soil microbial community structure and diversity in a double-cropping paddy field of southern China. Scientific Reports, 10(1), 1-9. [CrossRef]
- Tomsone, L., Kruma, Z., Alsina, I., & Lepse, L. (2012). The application of hierarchical cluster analysis for clasifying horseradish genotypes (Armoracia rusticana L.) roots. Chemical Technology, 62(4), 52-56. [CrossRef]
- Vidyashree, D. N., Muthuraju, R., & Panneerselvam, P. (2018). Evaluation of zinc solubilizing bacterial (ZSB) strains on growth, yield and quality of tomato (Lycopersicon esculentum). Int. J. Curr. Microbiol. Appl. Sci, 7(04), 2018. [CrossRef]
- Vukicevich, E., Lowery, T., Bowen, P., Úrbez-Torres, J. R., & Hart, M. (2016). Cover crops to increase soil microbial diversity and mitigate decline in perennial agriculture. A review. Agronomy for Sustainable Development, 36(3), 1-14. [CrossRef]
- Welch, R. M. (2002). The impact of mineral nutrients in food crops on global human health. Plant and Soil, 247(1), 83-90. [CrossRef]
- White, P. J., & Broadley, M. R. (2011). Physiological limits to zinc biofortification of edible crops. Frontiers in plant science, 2, 80. [CrossRef]
- Xue, P. P., Carrillo, Y., Pino, V., Minasny, B., & McBratney, A. (2018). Soil properties drive microbial community structure in a large scale transect in South Eastern Australia. Scientific reports, 8(1), 1-11. [CrossRef]
- Zhang, Y. Q., Deng, Y., Chen, R.Y., Cui, Z.L., Chen, X.P., Yost, R., Zhang, F.S. & Zou, C. Q. (2012). The reduction in zinc concentration of wheat grain upon increased phosphorus-fertilization and its mitigation by foliar zinc application. Plant and Soil, 361(1), 143-152. [CrossRef]
- Zhi-Hui, Y. A. N. G., Stöven, K., Haneklaus, S., Singh, B. R., & Schnug, E. (2010). Elemental sulfur oxidation by Thiobacillus spp. and aerobic heterotrophic sulfur-oxidizing bacteria. Pedosphere, 20(1), 71-79. [CrossRef]
| Code | Treatment abbreviation |
Treatment description | Cropping system |
FYM | Res | N (Kg ha-1) |
P (Kg ha-1) |
|---|---|---|---|---|---|---|---|
| Inm1 | None | No input | MT | - | - | 0 | 0 |
| Inm2 | P+N | NP fertilizer only | MM | - | - | 60 | 45 |
| Inm3 | R only | Residue only | MT | - | + | 0 | 0 |
| Inm4 | FYM only | FYM only | MT | + | - | 0 | 0 |
| Inm5 | P+N+R | NP fertilizer + residue only | MM | - | + | 60 | 45 |
| Inm6 | FYM+PN+R | NP fertilizer + residue +FYM | MM | + | + | 60 | 45 |
| Inm7 | FYM+PN | NP fertilizer + FYM only | MT | + | - | 60 | 45 |
| Inm8 | FYM+R | Residue + FYM only | MM | + | + | 0 | 0 |
| Inm9 | P only | P fertilizer only | MM | - | - | 0 | 45 |
| Inm10 | P+FYM | P + FYM only | MM | + | - | 0 | 45 |
| Inm11 | P+N* | 90N+P fertilizer only | MM | - | - | 90 | 45 |
| Treatment | pH | SOC (%) | N (%) | P (mg kg-1) |
Mg (mg kg-1) |
Mn (mg kg-1) |
S (mg kg-1) |
Cu (mg kg-1) |
B (mg kg-1) |
Zn (mg kg-1) |
Fe (mg kg-1) |
EC | CEC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inm1 | 4.84de | 1.70cd | 0.13ef | 18.1e | 37.5d | 394ab | 17.1d | 5.26c | 0.13cd | 1.00c | 118c | 31.7b | 6.54d |
| Inm2 | 4.51f | 1.64d | 0.12f | 79.0a | 37.5d | 431ab | 22.1bc | 5.51bc | 0.17bcd | 1.30c | 128bc | 43.6ab | 6.84d |
| Inm3 | 4.88de | 1.83bcd | 0.14def | 13.0e | 45.3d | 400ab | 19.0cd | 5.62abc | 0.18abcd | 1.23c | 117c | 32.0b | 6.64d |
| Inm4 | 5.16abc | 2.05a | 0.17ab | 23.8de | 122.3ab | 396ab | 19.8cd | 5.92abc | 0.23abc | 2.61ab | 143b | 42.0ab | 11.8ab |
| Inm5 | 4.66ef | 1.68cd | 0.13f | 71.2ab | 36.7d | 441a | 23.3ab | 5.65abc | 0.12cd | 1.28c | 133bc | 44.0ab | 6.71d |
| Inm6 | 5.06bcd | 1.96ab | 0.16bcd | 65.6ab | 138.7a | 397ab | 21.1bc | 6.29ab | 0.19abcd | 3.57a | 169a | 52.2a | 12.9a |
| Inm7 | 4.95cd | 1.86abc | 0.15cde | 55.9bc | 126.7ab | 366b | 23.7ab | 6.12abc | 0.17bcd | 3.29a | 174a | 50.4a | 12.6a |
| Inm8 | 5.43a | 2.07a | 0.18a | 20.5e | 152.3a | 420ab | 21.8bc | 6.37a | 0.29a | 3.46a | 126bc | 42.5ab | 12.1ab |
| Inm9 | 4.98cd | 1.70cd | 0.13ef | 56.7bc | 89.8bc | 389ab | 24.0ab | 5.85abc | 0.17bcd | 1.84bc | 126bc | 42.7ab | 10.1bc |
| Inm10 | 5.32ab | 1.83bcd | 0.16bc | 59.2abc | 161.5a | 405ab | 26.2a | 6.23ab | 0.27ab | 2.58ab | 139bc | 47.8a | 13.0a |
| Inm11 | 4.54f | 1.74cd | 0.14ef | 42.5cd | 55.4cd | 425ab | 24.3ab | 5.77abc | 0.12d | 1.61bc | 129bc | 48.2a | 8.62cd |
| P† | ||||||||||||||||
| P† | 1 | N% | ||||||||||||||
| N (%) | -.43* | 1 | SOC% | |||||||||||||
| SOC (%) | -.43* | .85** | 1 | pH | ||||||||||||
| pH (1:2) ₩ | -.34 | .76** | .70** | 1 | K† | |||||||||||
| K† | -.40* | .76** | .72** | .80** | 1 | Ca† | ||||||||||
| Ca† | -.10 | .71** | .54** | .82** | .58** | 1 | Mg† | |||||||||
| Mg† | -.10 | .70** | .73** | .85** | .70** | .87** | 1 | Mn† | ||||||||
| Mn† | .07 | .15 | .07 | -.02 | -.11 | -.04 | -.07 | 1 | S† | |||||||
| S† | .45* | -.02 | -.18 | -.01 | -.16 | .32 | .25 | -.11 | 1 | Cu† | ||||||
| Cu† | -.07 | .44* | .54** | .49** | .46** | .56** | .65** | .01 | .26 | 1 | B† | |||||
| B† | -.30 | .73** | .60** | .72** | .54** | .69** | .63** | .23 | .11 | .43* | 1 | Zn† | ||||
| Zn† | -.02 | .73** | .70** | .65** | .62** | .67** | .84** | -.04 | .23 | .54** | .42* | 1 | Fe† | |||
| Fe† | .36* | .19 | .30 | .14 | .21 | .29 | .41* | -.14 | .10 | .28 | -.09 | .56** | 1 | Na† | ||
| Na† | .13 | -.01 | .24 | .21 | -.05 | .38* | .41* | -.04 | .18 | .38* | .08 | .18 | .26 | 1 | EC | |
| EC | .43* | .18 | -.05 | -.10 | -.02 | .23 | .19 | -.22 | .47** | .28 | -.04 | .40* | .47** | .09 | 1 | C.E.C |
| C.E.C | .06 | .67** | .60** | .67** | .45** | .85** | .82** | .08 | .28 | .60** | .53** | .74** | .55** | .45* | .41* | 1 |
| Inputs | Diversity (Shannon) |
Richness | pH | SOC (%) |
N (%) |
Olsen P (mg kg-1) |
K (mg kg-1) |
Zn (mg kg-1) |
Fe (mg kg-1) |
|---|---|---|---|---|---|---|---|---|---|
|
Nitrogen (kg ha-1) 0 |
2.33 ± 0.15a | 21.2 ± 1.44a | 5.10 ± 0.26a | 1.85 ± 0.17a | 0.15 ± 0.02a | 31.9 ± 22.0b | 343±123a | 2.12 ± 1.02a | 128 ± 15.5a |
| 60 | 2.27 ± 0.19a | 21.3 ± 2.10a | 4.80 ± 0.27a | 1.79 ± 0.18a | 0.14 ± 0.02a | 66.6 ± 15.8a | 251±94a | 2.36 ± 1.27a | 151 ± 25.2a |
| 90 | 2.32 ± 0.07a | 21.0 ± 1.00a | 4.54 ± 0.13a | 1.75 ± 0.10a | 0.14 ± 0.01a | 42.5 ± 16.7ab | 162±39a | 1.61 ± 0.50a | 129 ± 7.8a |
|
Phosphorus (kg ha-1) 0 |
2.30 ± 0.16a | 21.1 ± 1.24a | 5.08 ± 0.26a | 1.90 ± 0.19a | 0.16 ± 0.02a | 18.9 ± 5.65b | 381±124a | 2.07 ± 1.18a | 126 ± 14.6a |
| 45 | 2.31 ± 0.16a | 21.3 ± 1.85a | 4.86 ± 0.32a | 1.77 ± 0.15a | 0.14 ± 0.02a | 60.7 ± 18.0a | 243±89.3a | 2.21 ± 1.04a | 143± 23.1a |
|
FYM ( t ha-1) 0 |
2.25 ± 0.15b | 20.44 ± 1.42b | 4.74 ± 0.21b | 1.72 ± 0.11b | 0.13 ± 0.01b | 46.8 ± 27.0a | 212±60.5b | 1.38 ± 0.45b | 125 ± 9.8b |
| 4 | 2.38 ± 0.15a | 22.2 ± 1.37a | 5.19 ± 0.24a | 1.94 ± 0.15a | 0.16 ± 0.02a | 43.9 ± 23.5a | 390±104a | 3.10 ± 0.82a | 150 ± 24.3a |
|
Residue (t ha-1) 0 |
2.35 ± 0.12a | 21.2 ± 1.61a | 4.9 ± 0.32a | 1.79 ± 0.15a | 0.14 ± 0.02a | 47.1 ± 23.1a | 257±103a | 2.03 ± 0.84a | 137 ± 21.1a |
| 2 | 2.23 ± 0.20a | 21.3 ± 1.76a | 5.01 ± 0.31a | 1.87 ± 0.20a | 0.15 ± 0.02a | 42.6 ± 29.1a | 357±130a | 2.38 ± 1.42a | 136 ± 23.8a |
| Microbial Indices | Olsen P (mg kg-1) |
N (%) |
SOC (%) |
pH | K (mg kg-1) |
Ca (mg kg-1) |
Mg (mg kg-1) |
Zn (mg kg-1) |
Fe (mg kg-1) |
C.E.C (meq 100g-1) |
|---|---|---|---|---|---|---|---|---|---|---|
| Diversity (Shannon) | -.137 | .286 | .167 | .375* | .312 | .498** | .480** | .468** | .283 | .350* |
| Species richness | .105 | .252 | .390* | .272 | .300 | .382* | .422* | .348* | .642** | .514** |
| Variables | Burkholderia spp | Bacillus spp | Trichoderma spp | Rhizobium spp | Sphingomonas spp | Bradyrhizobium spp | Glomus spp | Aspergillus spp | Arthrobacter spp | Thiobacillus spp | Pseudomonas spp | Azospirillum spp | Lysinibacillus spp | Mesorhizobium spp | Paenibacillus spp | Pantoea spp | Xanthomonas spp | Staphylococcus spp | Mucor spp | Serratia spp | Agrobacterium spp | Micrococcus spp | Sporosarcina spp |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P (ppm) | 0.01 | 0.17 | .37* | 0.24 | 0.02 | -0.06 | -0.23 | 0.26 | -0.33 | -0.19 | -0.24 | 0.08 | 0.09 | -0.24 | 0.19 | 0.08 | -0.33 | 0.25 | 0.01 | 0.25 | -0.08 | -0.02 | 0.10 |
| N (%) | .41* | .37* | -.35* | -0.21 | .41* | .47** | -0.30 | -0.19 | .64** | .77** | .57** | .52** | .49** | .78** | 0.17 | -0.02 | .46** | 0.12 | -0.30 | -0.17 | .35* | 0.16 | 0.07 |
| SOC (%) | .38* | .37* | -0.31 | -.44* | .60** | .57** | -0.18 | -0.12 | .47** | .74** | .54** | .45* | .52** | .71** | 0.05 | -0.03 | 0.21 | 0.18 | -0.25 | -0.12 | .37* | 0.30 | 0.20 |
| pH | 0.11 | 0.30 | -.53** | -.35* | 0.24 | .48** | -0.04 | -0.15 | .65** | .65** | .60** | .51** | .47** | .65** | 0.06 | 0.03 | .42* | 0.14 | -.42* | -0.09 | .37* | 0.19 | -0.08 |
| K (ppm) | 0.17 | 0.33 | -.48** | -0.30 | 0.23 | .44* | -0.08 | -0.27 | .46** | .57** | .55** | .49** | .54** | .69** | -0.09 | 0.09 | 0.22 | -0.01 | -0.29 | -0.24 | .48** | 0.22 | -0.04 |
| Ca (ppm) | 0.20 | .52** | -.42* | -0.33 | 0.24 | .39* | -0.27 | -0.14 | .60** | .71** | .66** | .57** | .61** | .58** | 0.29 | 0.12 | .50** | 0.18 | -.45** | 0.07 | 0.32 | 0.16 | 0.19 |
| Mg (ppm) | 0.24 | .50** | -.45** | -.37* | .41* | .55** | -0.30 | -0.16 | .42* | .71** | .60** | .53** | .66** | .60** | 0.14 | 0.11 | 0.30 | 0.21 | -.41* | 0.06 | 0.28 | 0.23 | 0.19 |
| Mn (ppm) | .34* | -0.19 | .40* | .62** | 0.18 | -0.10 | -0.22 | .49** | 0.17 | -0.03 | 0.06 | 0.23 | -0.34 | 0.22 | -0.07 | -0.13 | -0.11 | 0.26 | 0.28 | -0.21 | -0.11 | 0.21 | -.36* |
| S (ppm) | -0.04 | 0.30 | -0.12 | 0.07 | -0.12 | -0.08 | -.40* | -0.05 | -0.08 | 0.05 | 0.04 | 0.06 | 0.17 | -0.19 | .40* | 0.03 | 0.05 | 0.08 | -0.26 | .37* | -0.09 | -0.15 | 0.06 |
| Cu (ppm) | 0.09 | .37* | -0.30 | -0.09 | .41* | 0.21 | -0.21 | -0.01 | 0.26 | .45** | .51** | 0.31 | .45** | 0.30 | 0.23 | -0.02 | 0.05 | 0.16 | -0.15 | -0.09 | 0.18 | .46** | 0.21 |
| B (ppm) | 0.15 | 0.28 | -0.26 | -0.31 | 0.11 | 0.14 | -0.2 | -0.10 | .62** | .47** | .47** | .43* | 0.26 | .60** | 0.27 | -0.12 | .38* | 0.14 | -0.31 | -0.12 | 0.14 | 0.11 | -0.03 |
| Zn (ppm) | 0.29 | .48** | -0.32 | -0.20 | .52** | .59** | -.43* | -0.21 | 0.28 | .71** | .48** | .46** | .65** | .59** | 0.11 | 0.21 | 0.30 | 0.13 | -0.34 | 0.11 | 0.21 | 0.14 | 0.23 |
| Fe (ppm) | 0.29 | .37* | -0.05 | -0.16 | .61** | .55** | -0.21 | -0.01 | -0.08 | .52** | 0.33 | 0.25 | .54** | 0.33 | 0.19 | .50** | 0.05 | 0.14 | -0.26 | 0.02 | 0.34 | 0.10 | .36* |
| Na (ppm) | 0.25 | -0.02 | -0.23 | -0.25 | .45** | 0.27 | -0.10 | 0.11 | 0.02 | .37* | 0.33 | 0.01 | 0.10 | 0.04 | 0.05 | 0.09 | -0.03 | 0.05 | -0.30 | 0.16 | 0.11 | 0.15 | 0.22 |
| EC | 0.21 | 0.32 | 0.10 | 0.01 | 0.23 | 0.03 | -0.33 | 0.01 | -0.02 | 0.23 | -0.05 | -0.03 | .36* | 0.02 | .48** | -0.08 | 0.14 | 0.03 | -0.02 | 0.21 | 0.07 | -0.05 | .41* |
| C.E.C | .41* | .51** | -0.18 | -0.19 | .54** | .51** | -.42* | 0.18 | .50** | .82** | .64** | .57** | .63** | .62** | 0.30 | 0.15 | .37* | .37* | -0.34 | 0.01 | 0.25 | 0.23 | 0.32 |
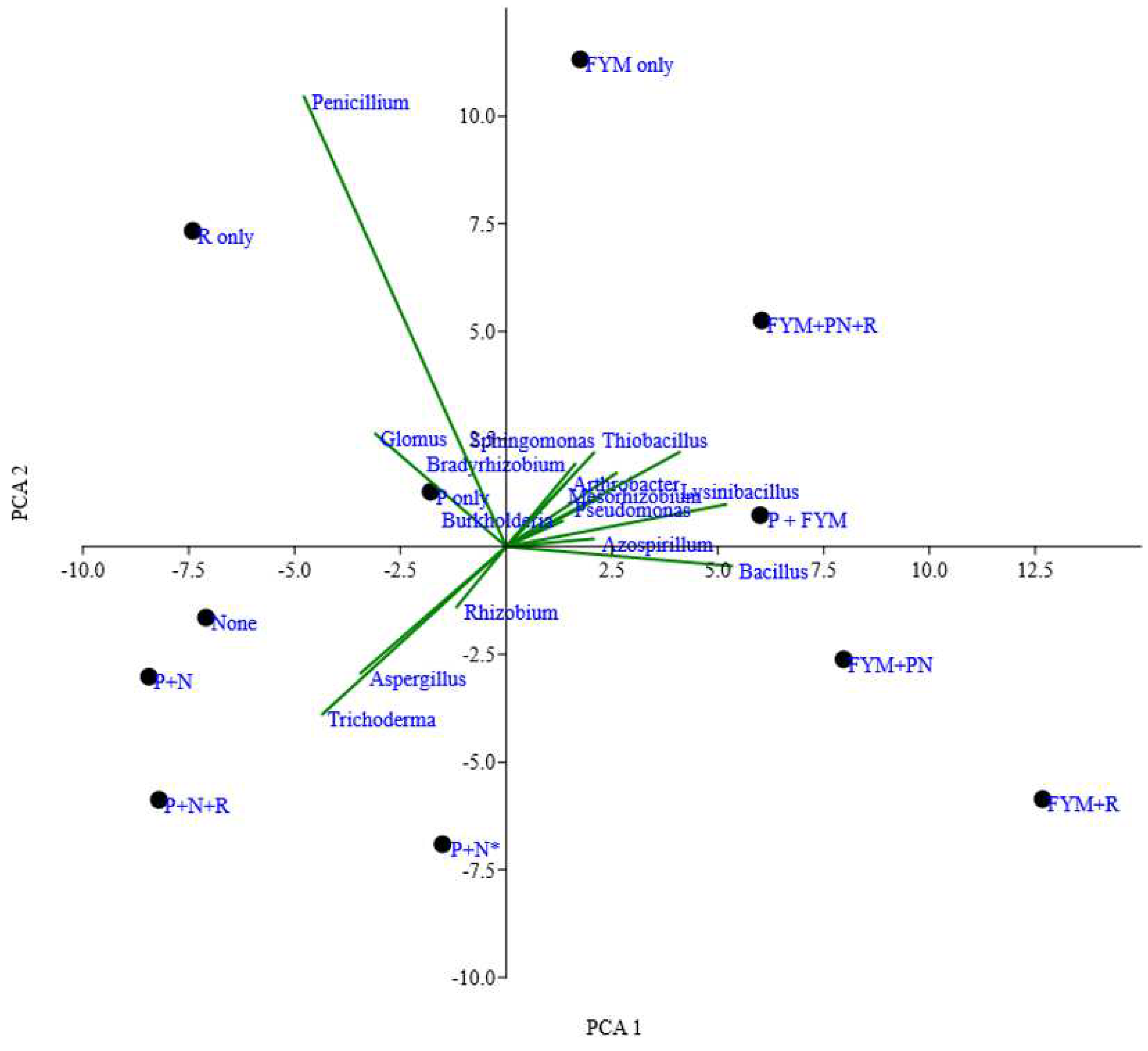
| CCA1 | CCA2 | R2 | P-value | |
|---|---|---|---|---|
|
Zinc solubilizing microbial genera Agrobacterium |
0.91 | 0.42 | 0.27 | 0.02 |
| Arthrobacter | 0.64 | 0.76 | 0.66 | 0.001 |
| Aspergillus | 0.98 | -0.21 | 0.51 | 0.001 |
| Azospirillum | 0.96 | 0.26 | 0.39 | 0.001 |
| Bacillus | 1.00 | 0.03 | 0.57 | 0.001 |
| Bradyrhizobium | 0.90 | -0.43 | 0.42 | 0.001 |
| Burkholderia | 1.00 | -0.01 | 0.03 | 0.67 |
| Enterobacter | 1.00 | 0.10 | 0.02 | 0.73 |
| Glomus | 0.23 | -0.97 | 0.19 | 0.05 |
| Gluconacetobacter | 0.62 | 0.79 | 0.02 | 0.71 |
| Klebsiella | 0.99 | 0.12 | 0.02 | 0.68 |
| Lysinibacillus | 1.00 | -0.05 | 0.76 | 0.001 |
| Mesorhizobium | 0.92 | 0.40 | 0.33 | 0.01 |
| Paenibacillus | 0.81 | 0.58 | 0.07 | 0.38 |
| Pantoea | 0.33 | -0.94 | 0.69 | 0.004 |
| Penicillium | 0.24 | 0.97 | 0.84 | 0.001 |
| Pseudomonas | 0.98 | -0.22 | 0.32 | 0.03 |
| Rhizobium | -1.00 | -0.04 | 0.38 | 0.002 |
| Serratia | -0.09 | -1.00 | 0.02 | 0.64 |
| Sphingomonas | 0.75 | -0.66 | 0.26 | 0.03 |
| Staphylococcus | 0.67 | 0.74 | 0.06 | 0.47 |
| Stenotrophomonas | -0.31 | 0.95 | 0.01 | 0.84 |
| Thiobacillus | 1.00 | 0.04 | 0.59 | 0.001 |
| Trichoderma | 1.00 | -0.01 | 0.67 | 0.001 |
| Xanthomonas | 0.71 | 0.70 | 0.22 | 0.05 |
|
Soil chemical properties Olsen P |
-0.11 | -0.41 | 0.04 | |
| N | 0.71 | 0.45 | 0.001 | |
| Zn | 0.76 | -0.17 | 0.02 | |
| pH | 0.77 | 0.44 | 0.13 | |
| SOC | 0.38 | -0.06 | 0.05 | |
| S | 0.4 | 0.03 | 0.22 | |
| B | 0.55 | 0.62 | 0.69 | |
| Fe | 0.57 | -0.76 | 0.03 | |
| Cu | 0.31 | 0.09 | 0.56 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




