Submitted:
01 September 2023
Posted:
05 September 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. FUEL CELLS
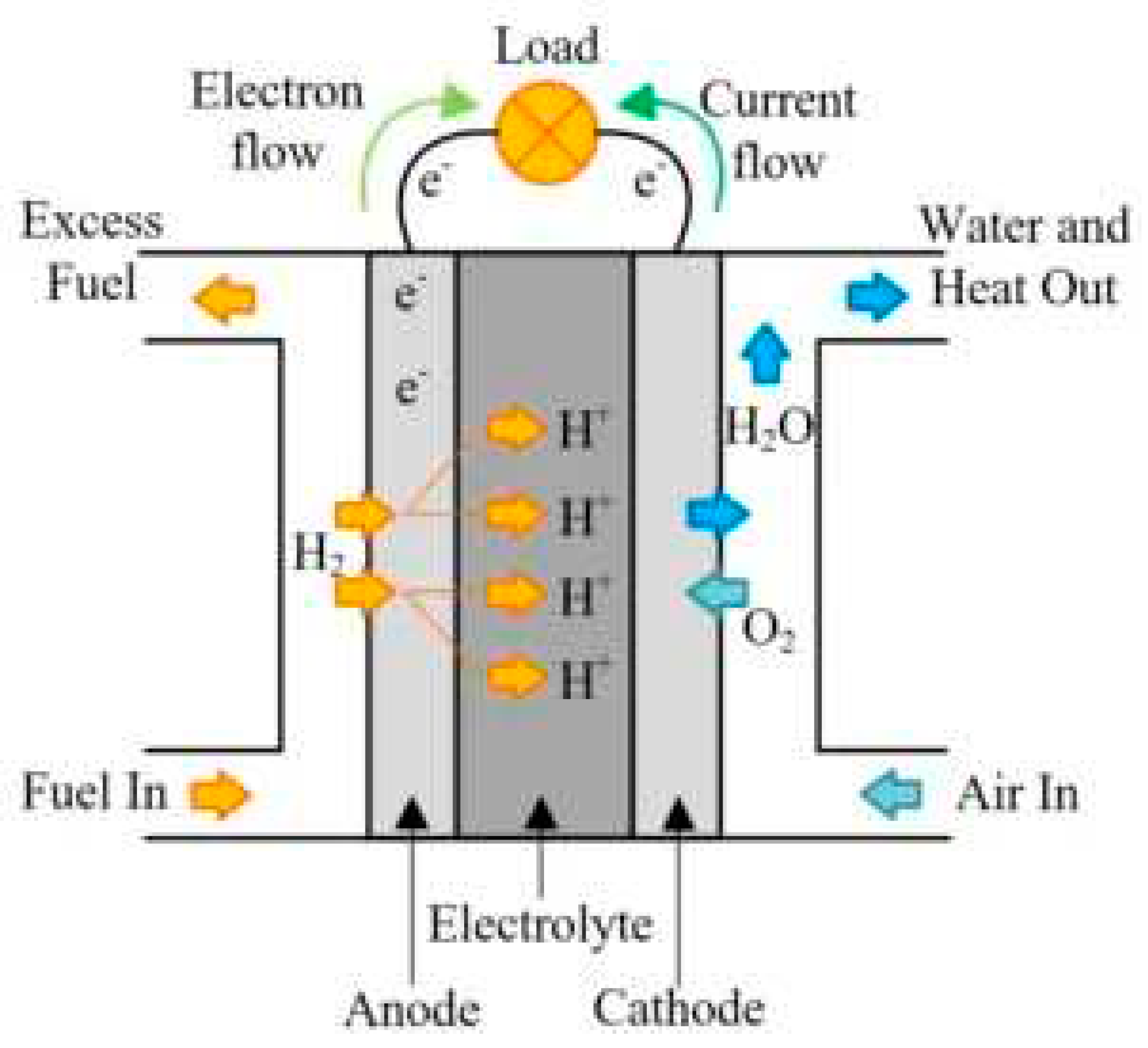
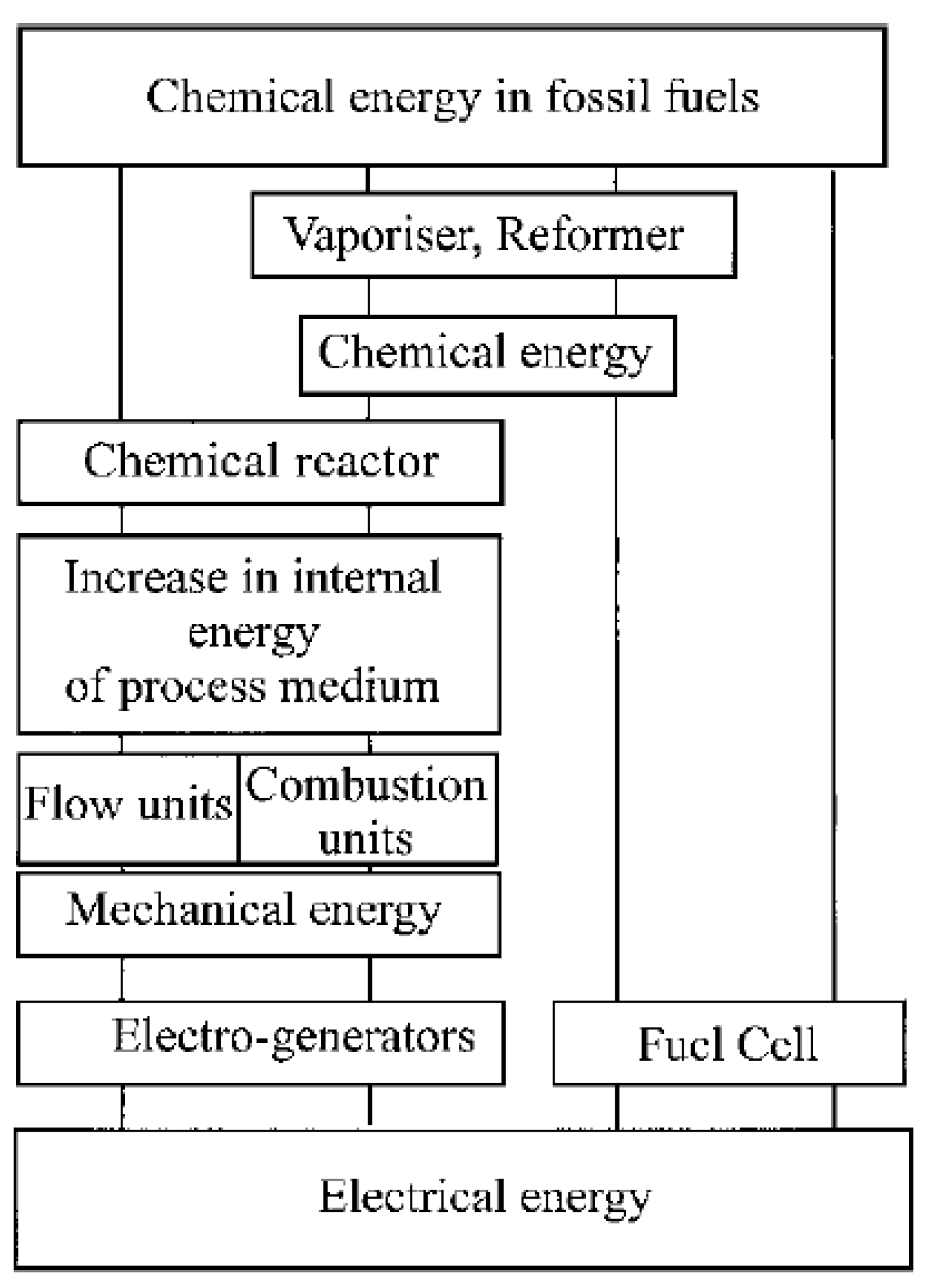
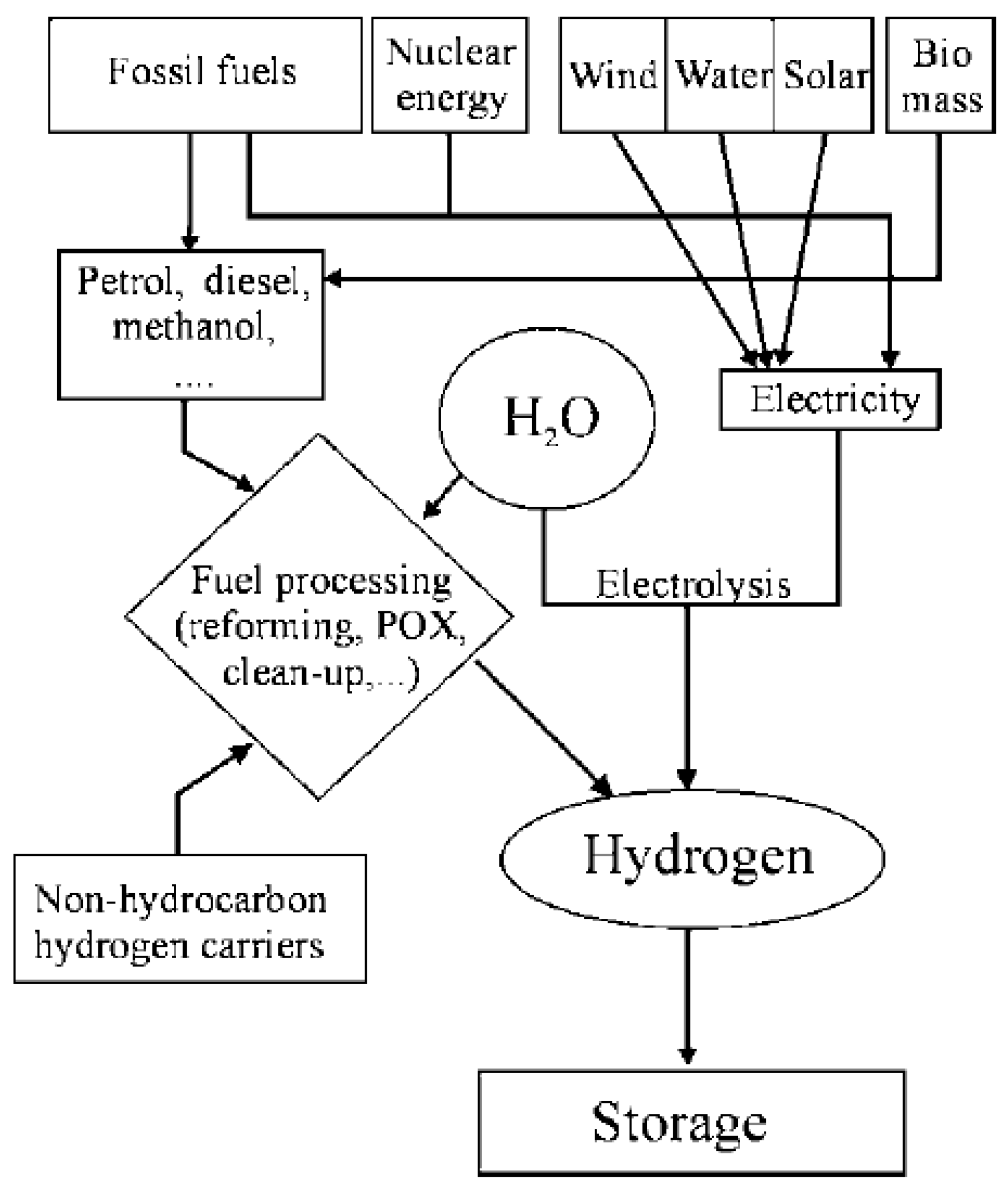
2.1. History of Fuel Cells
2.2. Types of Fuel Cells
| Fuel Cell Type | Common Electrolyte | Operating Temperature | Electrical Efficiency (LHV) | Applications |
|---|---|---|---|---|
| Polymer electrolyte membrane (PEM) | Perfluorosulfonic acid | <120°C | 60% direct H2; 40% reformed fuel | • Backup power Portable power Distributed generation Transportation Specialty vehicles |
| Alkaline (AFC) | Aqueous potassium hydroxide soaked in a porous matrix, or alkaline polymer membrane | <100°C | 60% | • Military Space Backup power Transportation |
| Phosphoric acid (PAFC) | Phosphoric acid soaked in a porous matrix or imbibed in a polymer membrane | 150°–200°C | 40% | Distributed generation |
| Molten carbonate (MCFC) | Molten lithium, sodium, and/or potassium carbonates, soaked in a porous matrix | 600°–700°C | 50% | • Electric utility Distributed generation |
| Solid oxide (SOFC) | Yttria stabilized zirconia | 500°–1,000°C | 60% | • Auxiliary power Electric utility Distributed generation |
| Fuel Cell Type | Advantages | Challenges |
|---|---|---|
| Polymer electrolyte membrane (PEM) | • Solid electrolyte reduces corrosion and electrolyte management problems Low temperature Quick start-up and load following |
• Expensive catalysts Sensitive to fuel impurities |
| Alkaline (AFC) | • Wider range of stable materials allows lower cost components Low temperature Quick start-up |
• Sensitive to CO2 in fuel and air Electrolyte management (aqueous) Electrolyte conductivity (polymer) |
| Phosphoric acid (PAFC) | • Suitable for CHP Increased tolerance to fuel impurities |
• Expensive catalysts Long start-up time Sulfur sensitivity |
| Molten carbonate (MCFC) | • High efficiency Fuel flexibility Suitable for CHP Hybrid/gas turbine cycle |
• High temperature corrosion and breakdown of cell components Long start-up time Low power density |
| Solid oxide (SOFC) | • High efficiency Fuel flexibility Solid electrolyte Suitable for CHP Hybrid/gas turbine cycle |
• High temperature corrosion and breakdown of cell components Long start-up time Limited number of shutdowns |
2.2.1. Alkaline fuel cells
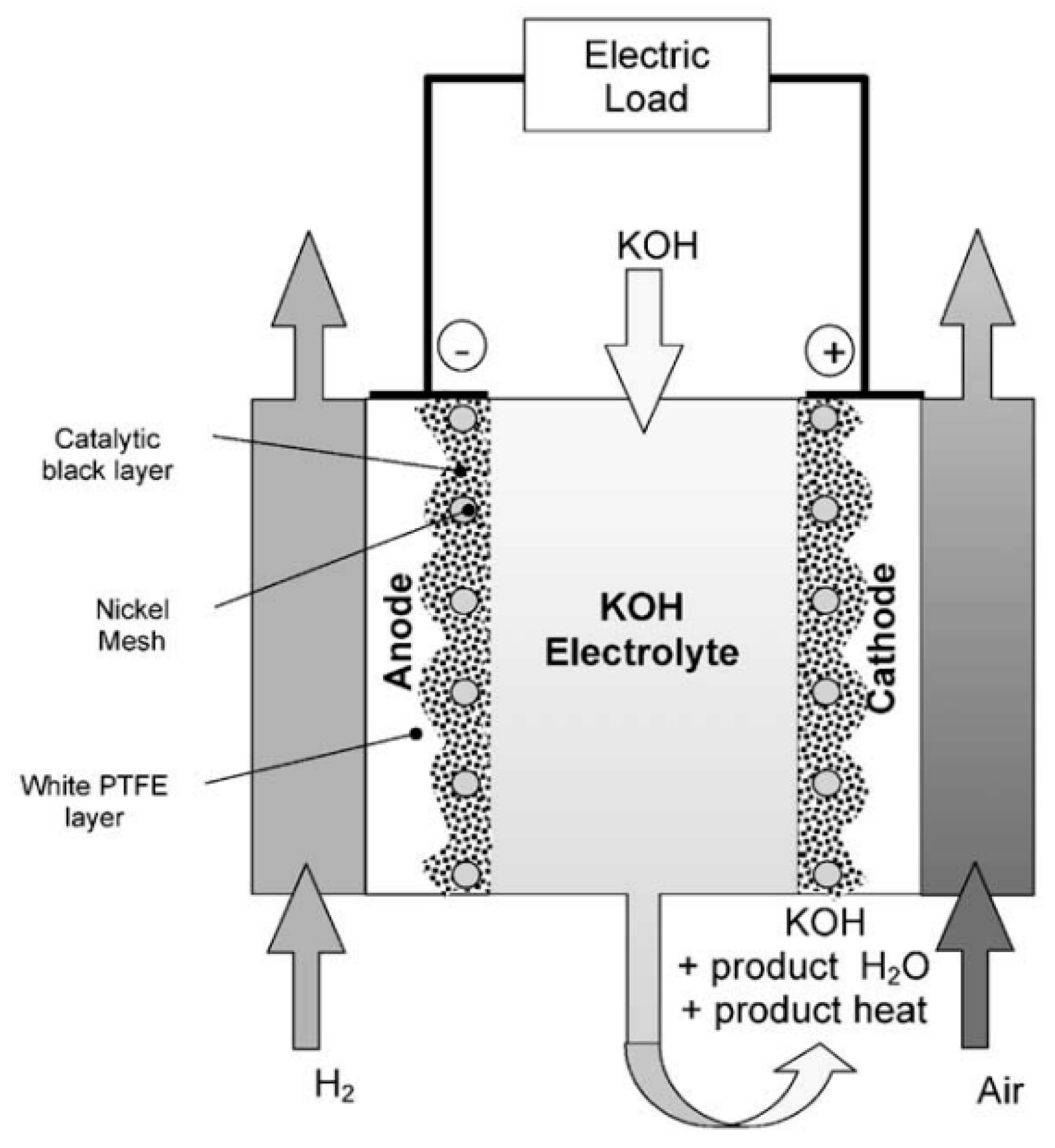
2.2.2. Proton exchange membrane fuel cells
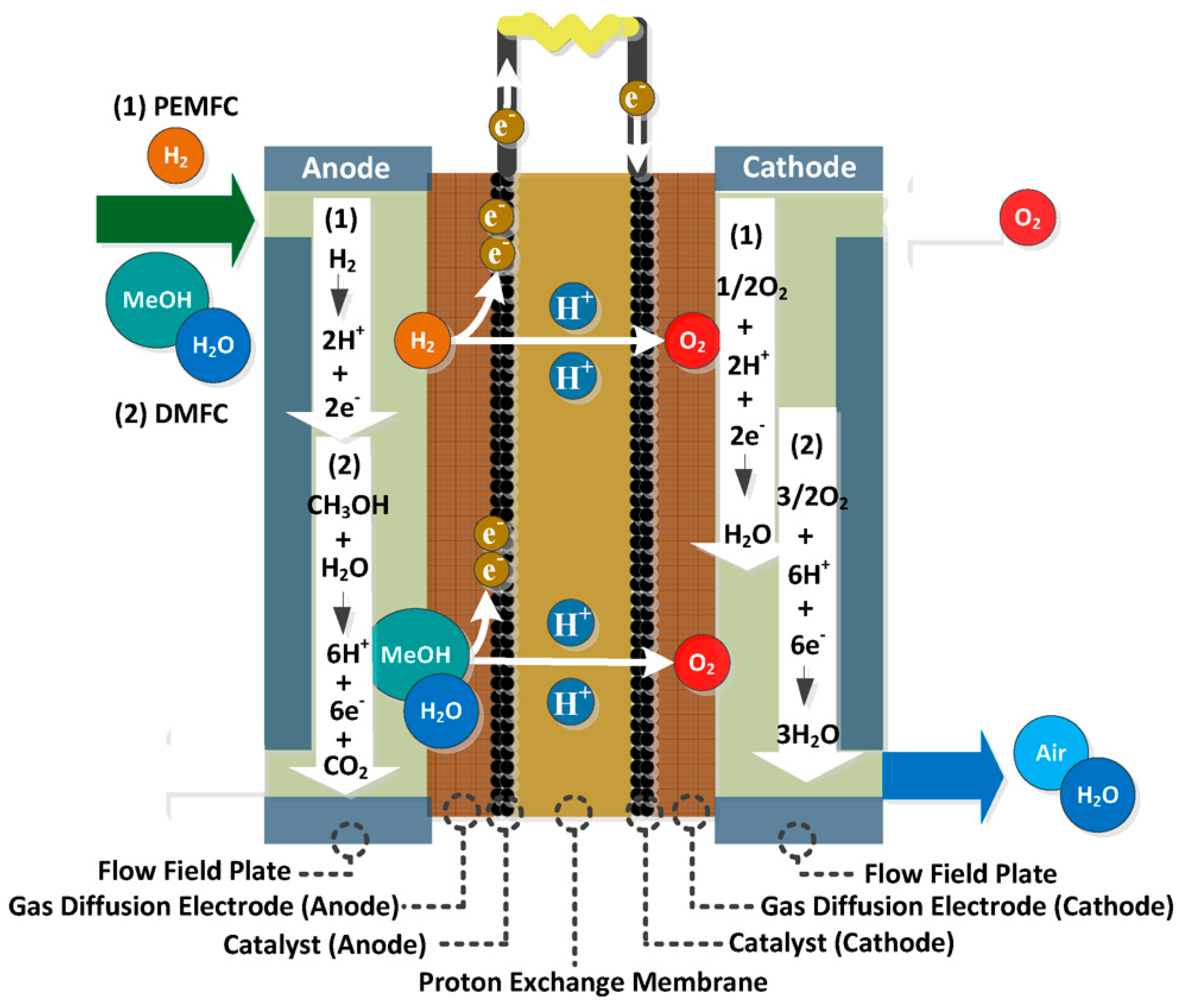
2.2.3. Direct methanol fuel cells
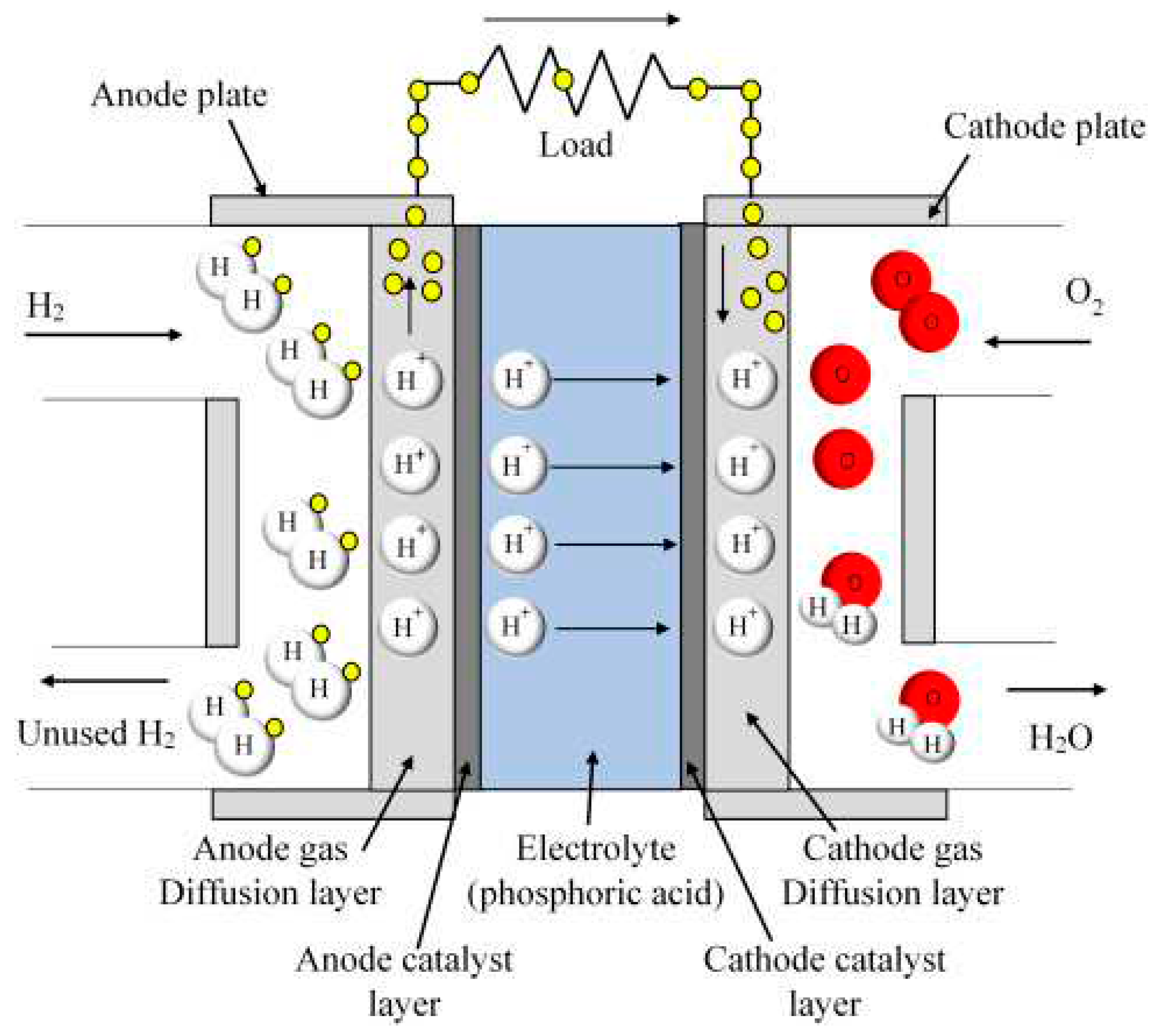
2.2.4. Phosphoric acid fuel cells
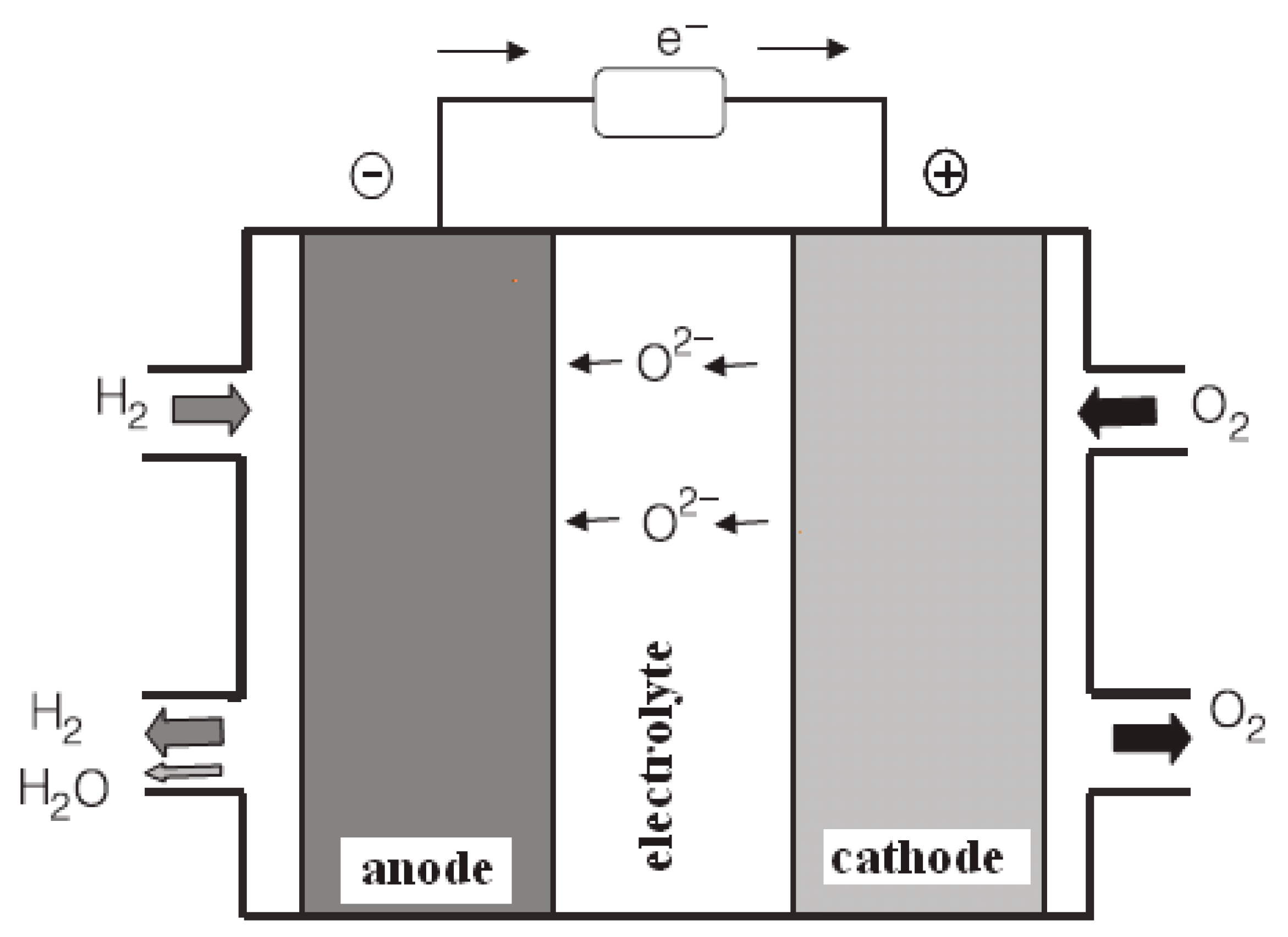
2.2.5. Solid oxide fuel cells
3. PROTON EXCHANGE MEMBRANES
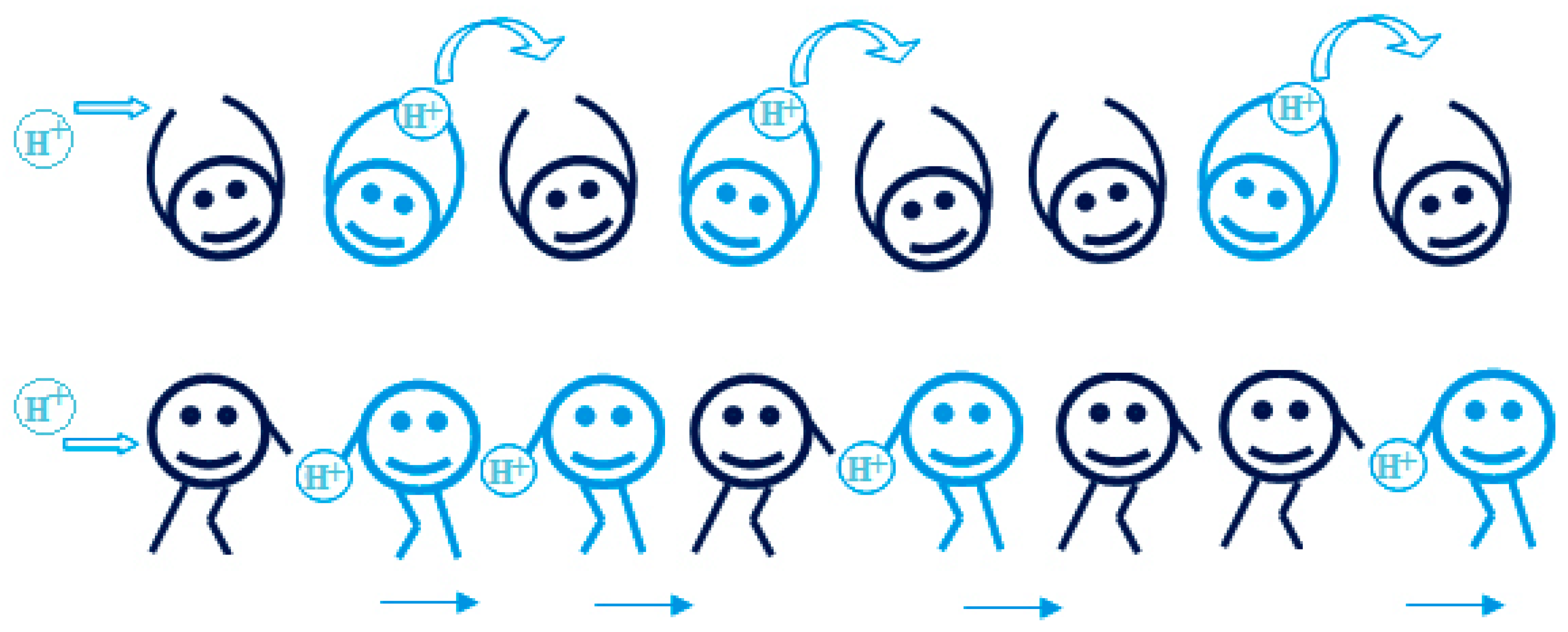
3.1. Proton Conductivity Mechanism
3.2. Proton Conductivity Measurement Methods
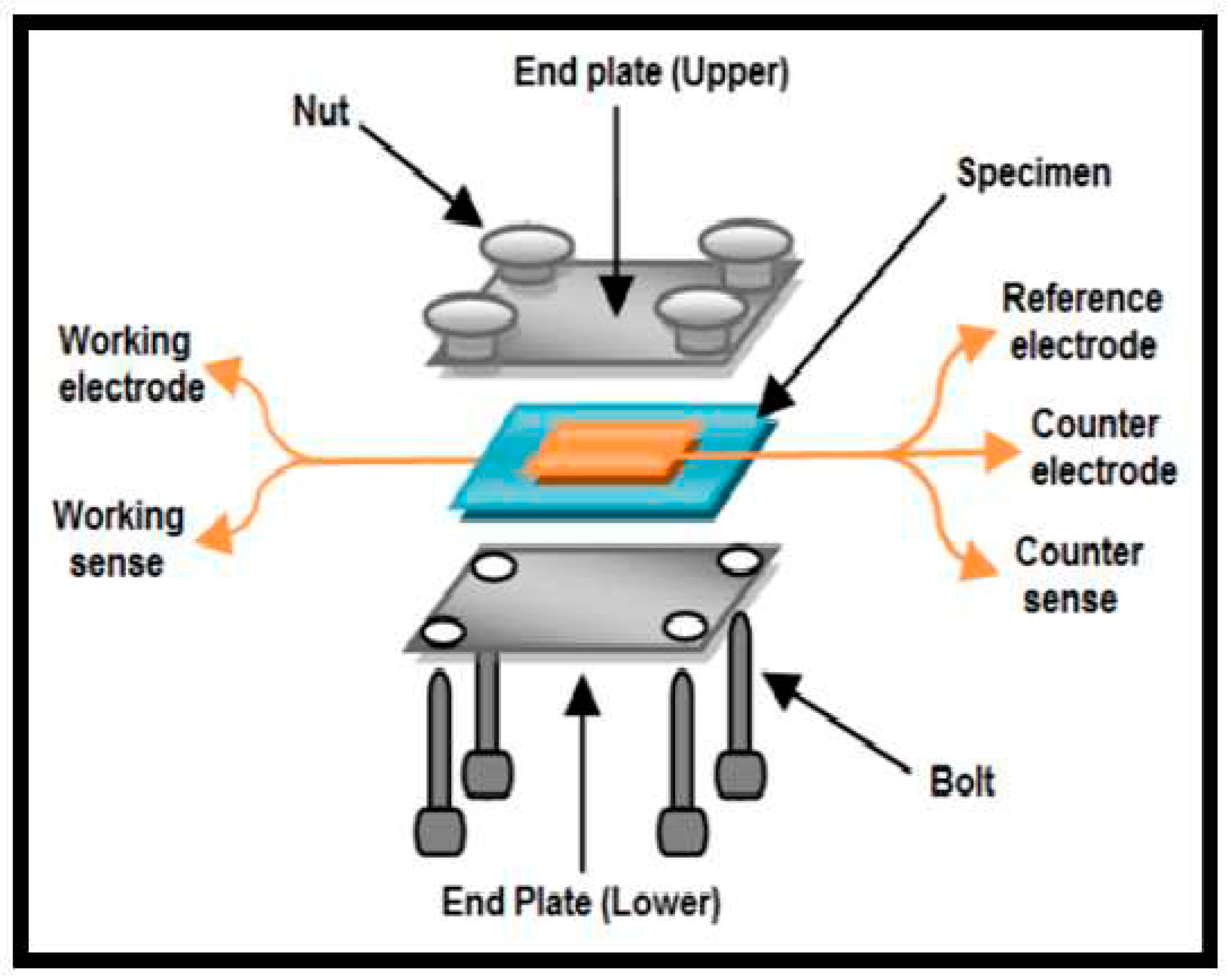
3.3. Some Types of Proton Exchange Membranes
3.3.1. Perfluorosulfonic acid (PFSA) membranes
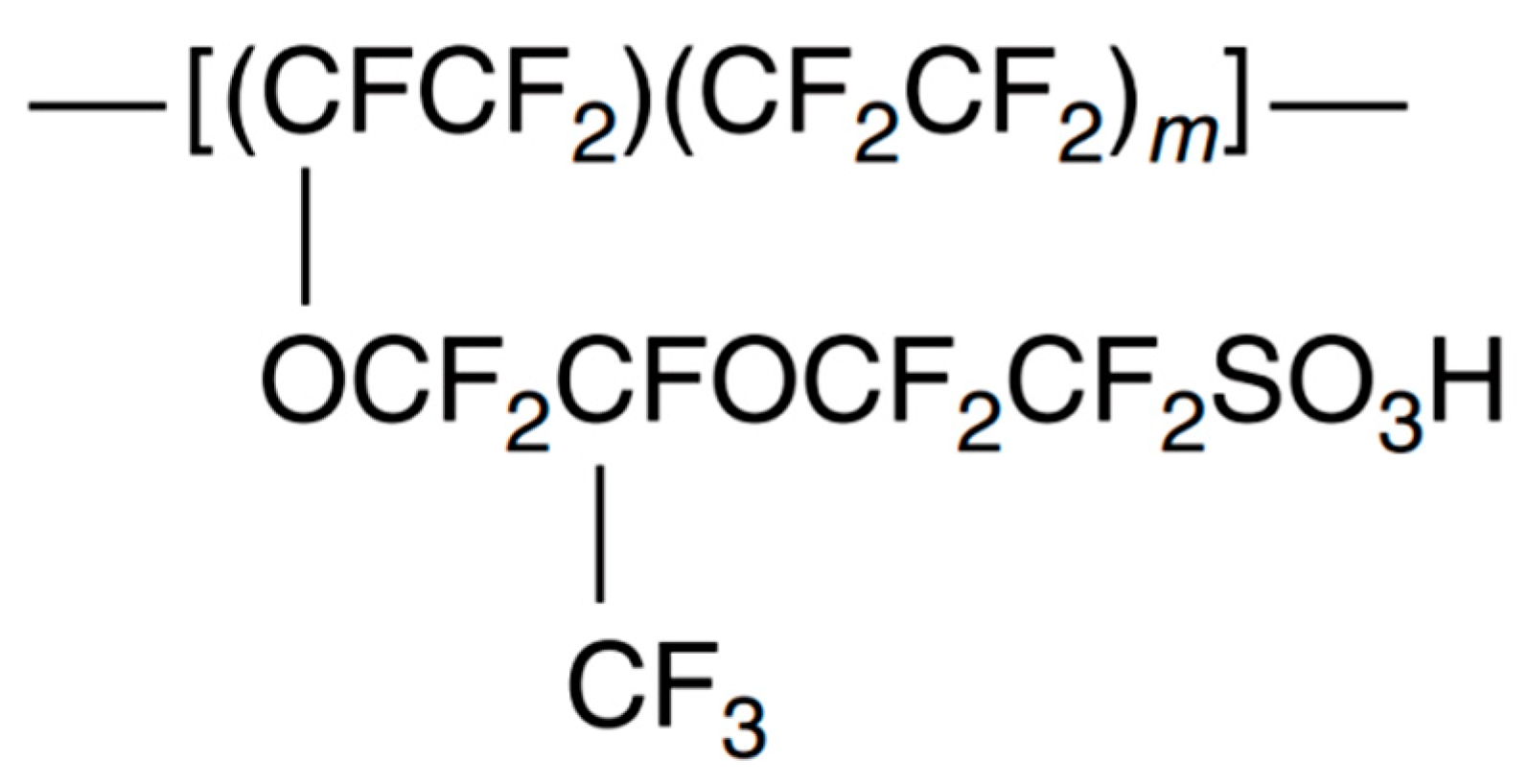
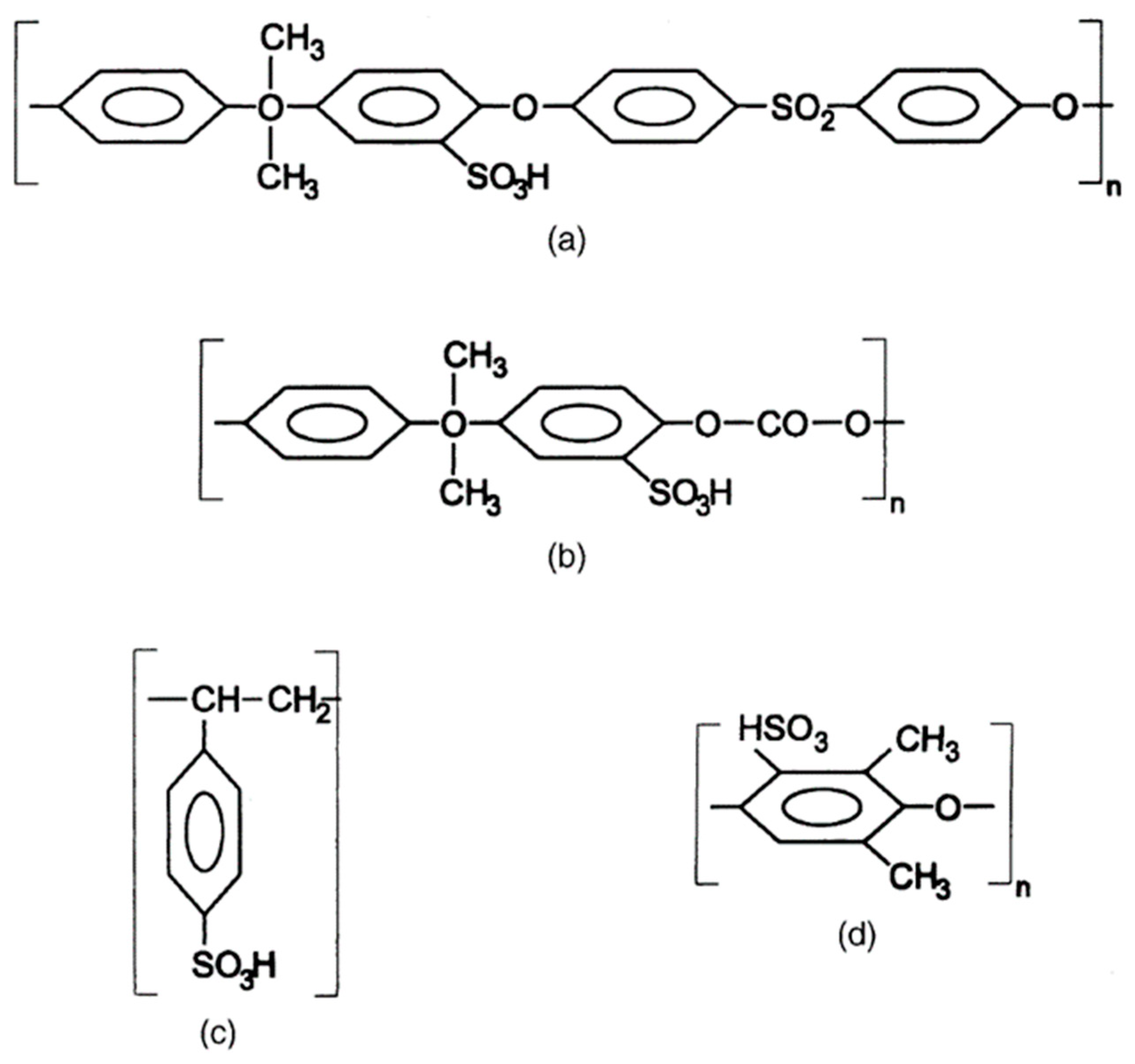
3.3.2. Sulfonated polymer membranes
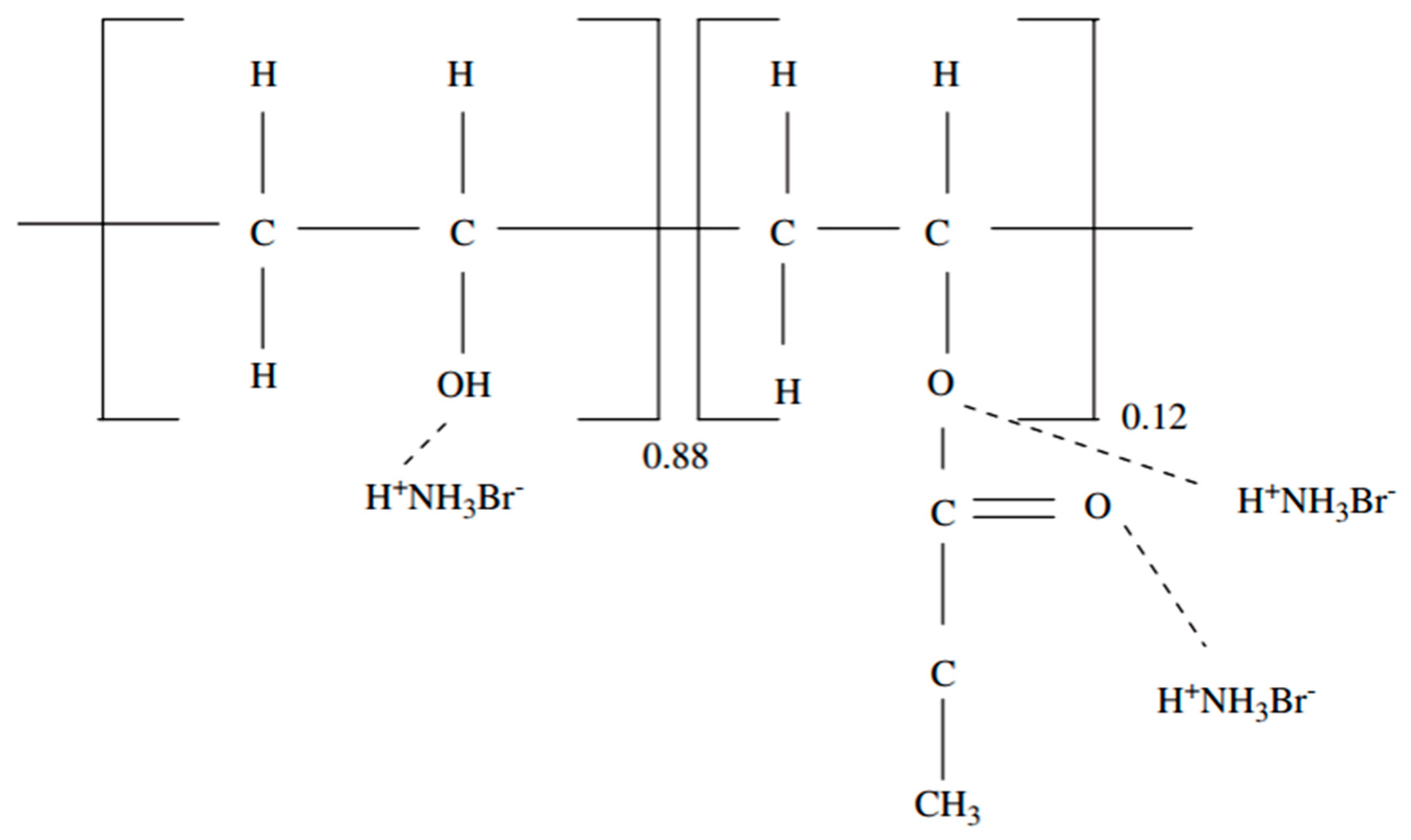
3.3.3. Polymer matrixes
| Conductivity at room temperature (S/cm) | |||
|---|---|---|---|
| H3PO4 concentration (M) | 100 Hz | 1 kHz | 100 kHz |
| 0.00 | 1.74*10-6 | 2.02*10-6 | 6.23*10-6 |
| 0.85 | 3.90*10-5 | 4.13*10-5 | 4.44*10-5 |
| 1.7 | 1.08*10-4 | 1.27*10-4 | 1.45*10-4 |
| 3.4 | 9.84*10-4 | 1.24*10-3 | 1.82*10-3 |
| 5.1 | 3.26*10-3 | 4.30*10-3 | 6.43*10-3 |
4. MATERIALS AND METHODS
4.1. Equipment
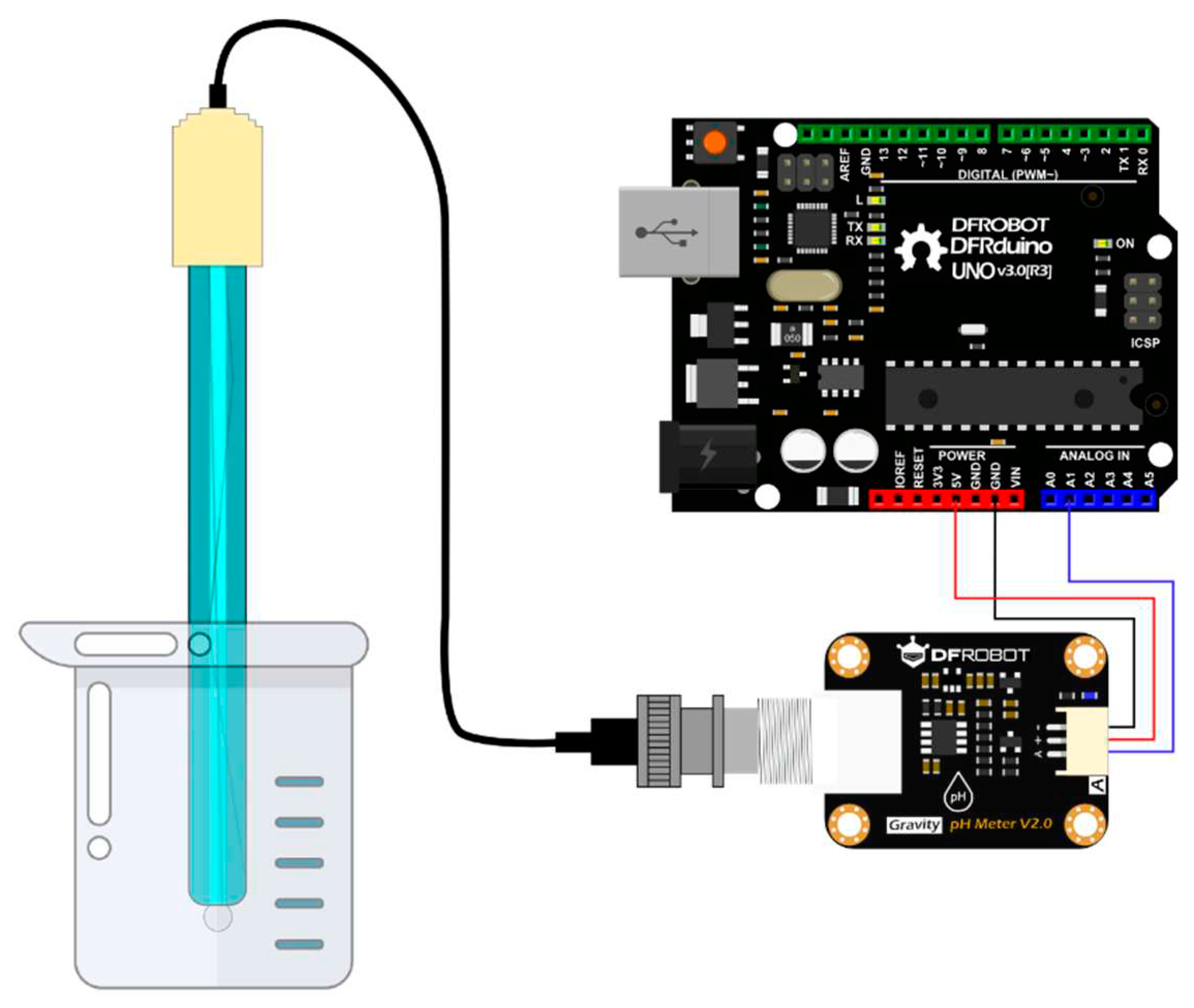
4.1.1. DFRobot’s Gravity: Analog pH Meter V2

- Specifications of pH Probe
- Probe Type: Laboratory Grade
- Detection Range: 0~14
- Temperature Range: 5~60°C
- Zero Point: 7±0.5
- Response Time: <2min
- Internal Resistance: <250MΩ
- Probe Life: >0.5 year (depending on frequency of use)
- Cable Length: 100cm
- Specifications of Signal Conversion Board
- Supply Voltage: 3.3~5.5V
- Output Voltage: 0~3.0V
- Probe Connector: BNC
- Signal Connector: PH2.0-3P
- Measurement Accuracy: ±0.1@25℃
- Dimension: 42mm*32mm/1.66*1.26in
4.1.2. Experiment setup prototype

4.1.3. Arduino Uno

4.1.4. Memmert ULE 500 oven

4.1.5. Ika works RW 20.n S1 overhead stirrer mixer

4.1.6. Polyscience immersion circulator

4.2. Experiment Design
4.3. Materials
4.4. Preparation of the Membranes
4.4.1. (PVA)0.7(NaBr)0.3(H3PO4)xM membranes

4.4.2. PVA/PAMA (poly (acrylic acid-co-maleic acid) membranes
4.4.3. PVA tartaric acid membranes

4.5. Connections of pH Meter
4.6. MATLAB Codes
4.7. Experiment Procedure

4.8. Data Smoothing
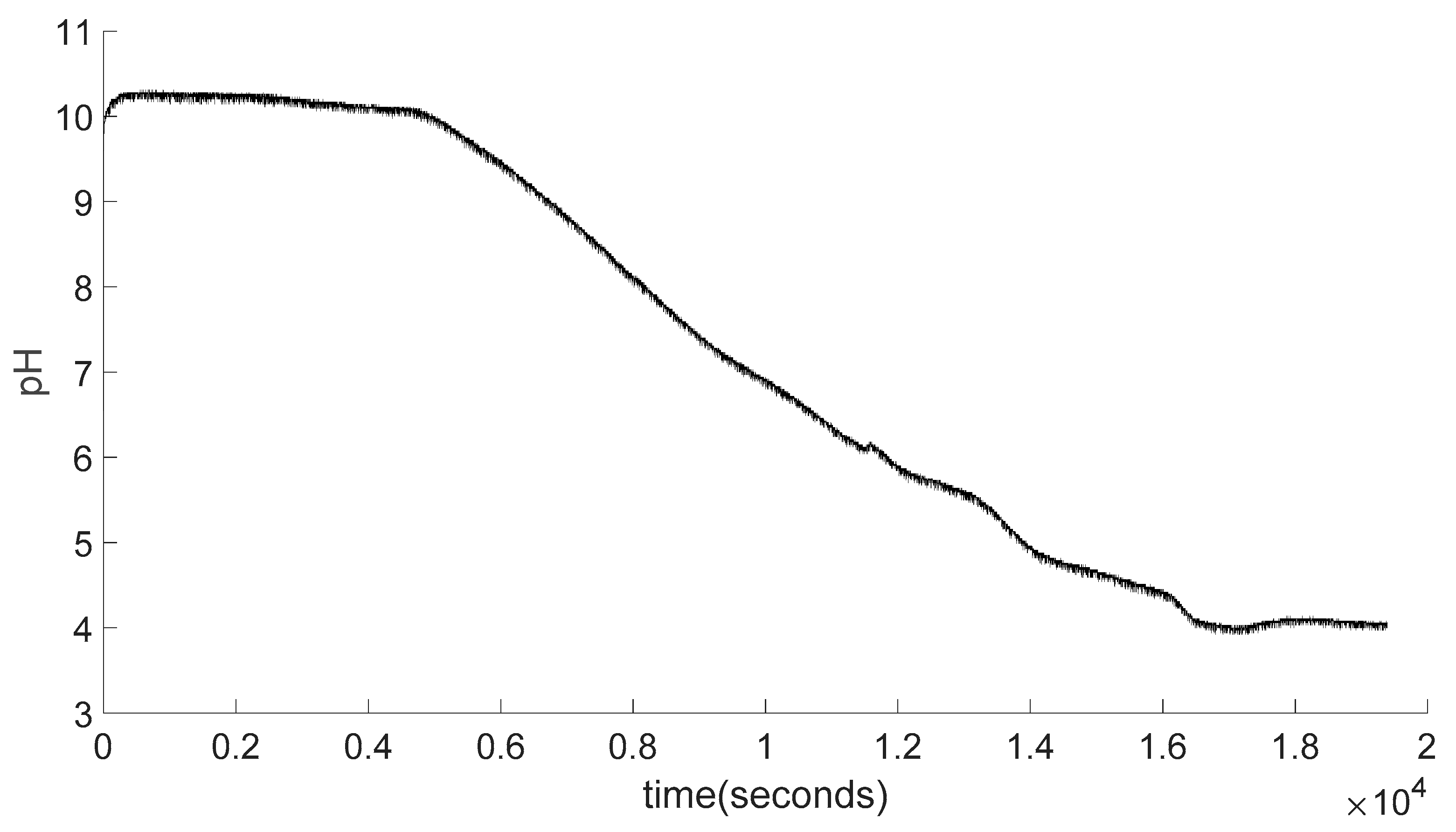

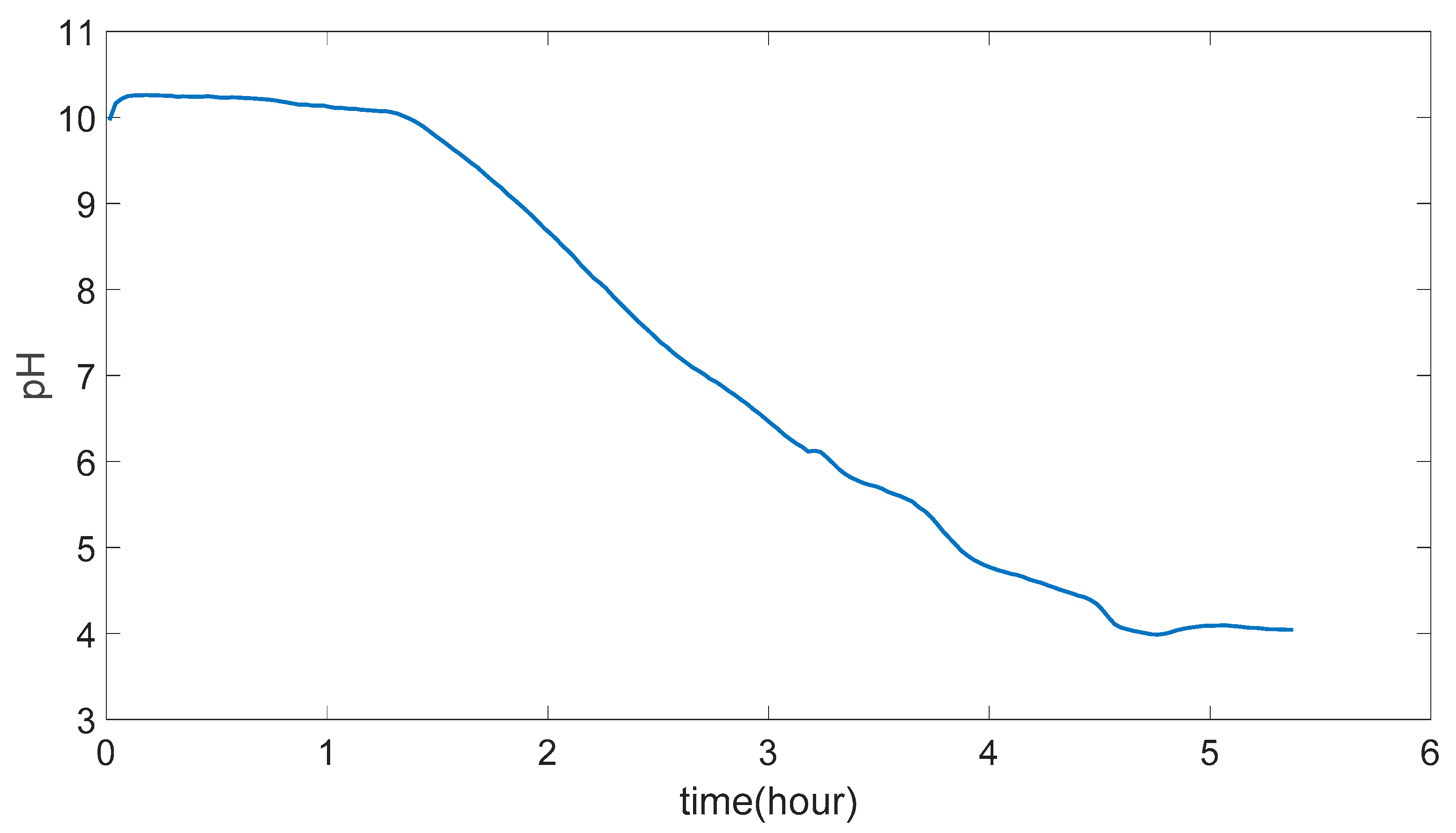
4.9. Swelling Calculations
5. RESULTS AND DISCUSSION
5.1. (PVA)0.7(NaBr)0.3(H3PO4)xM Membranes Results
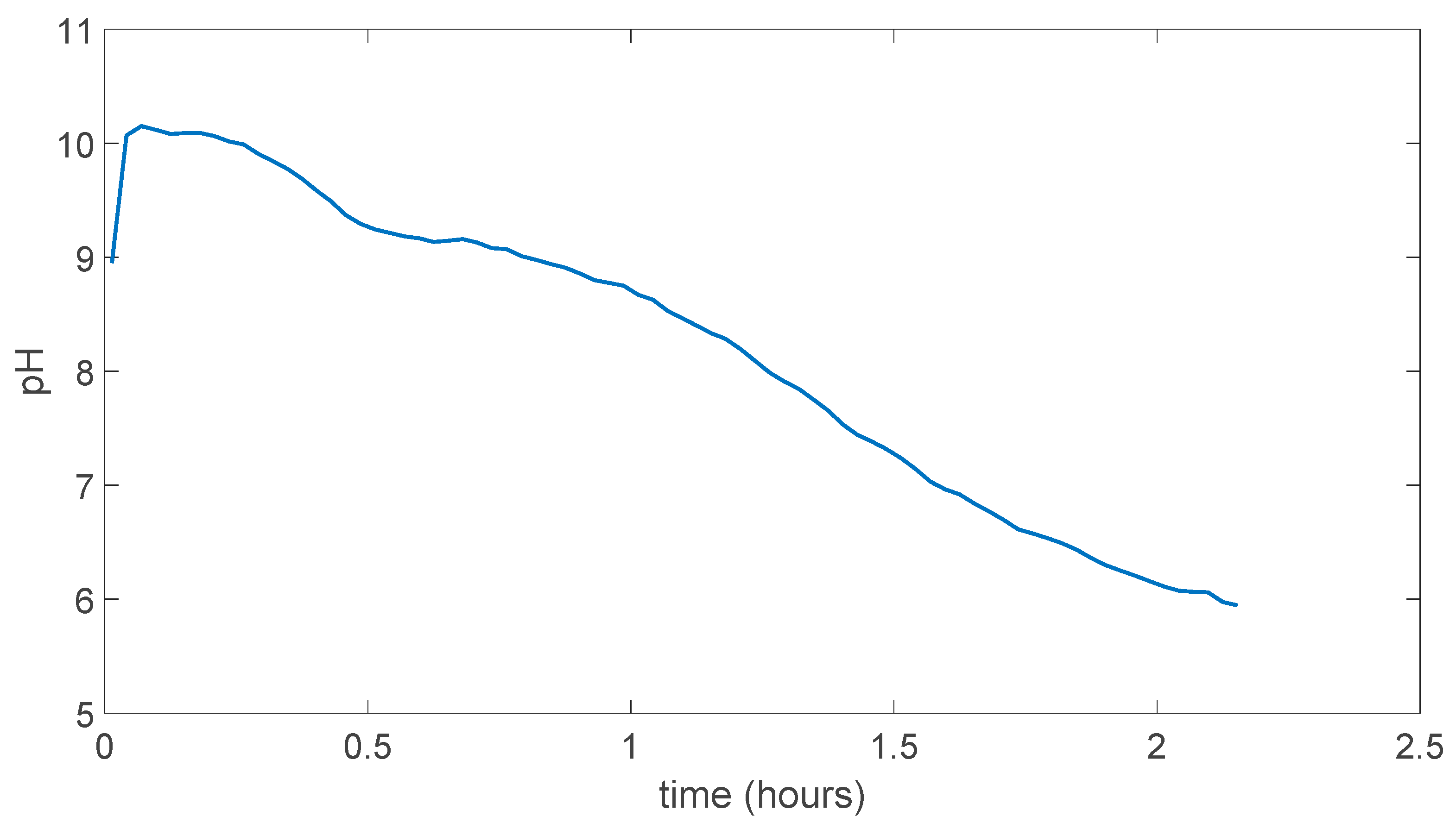
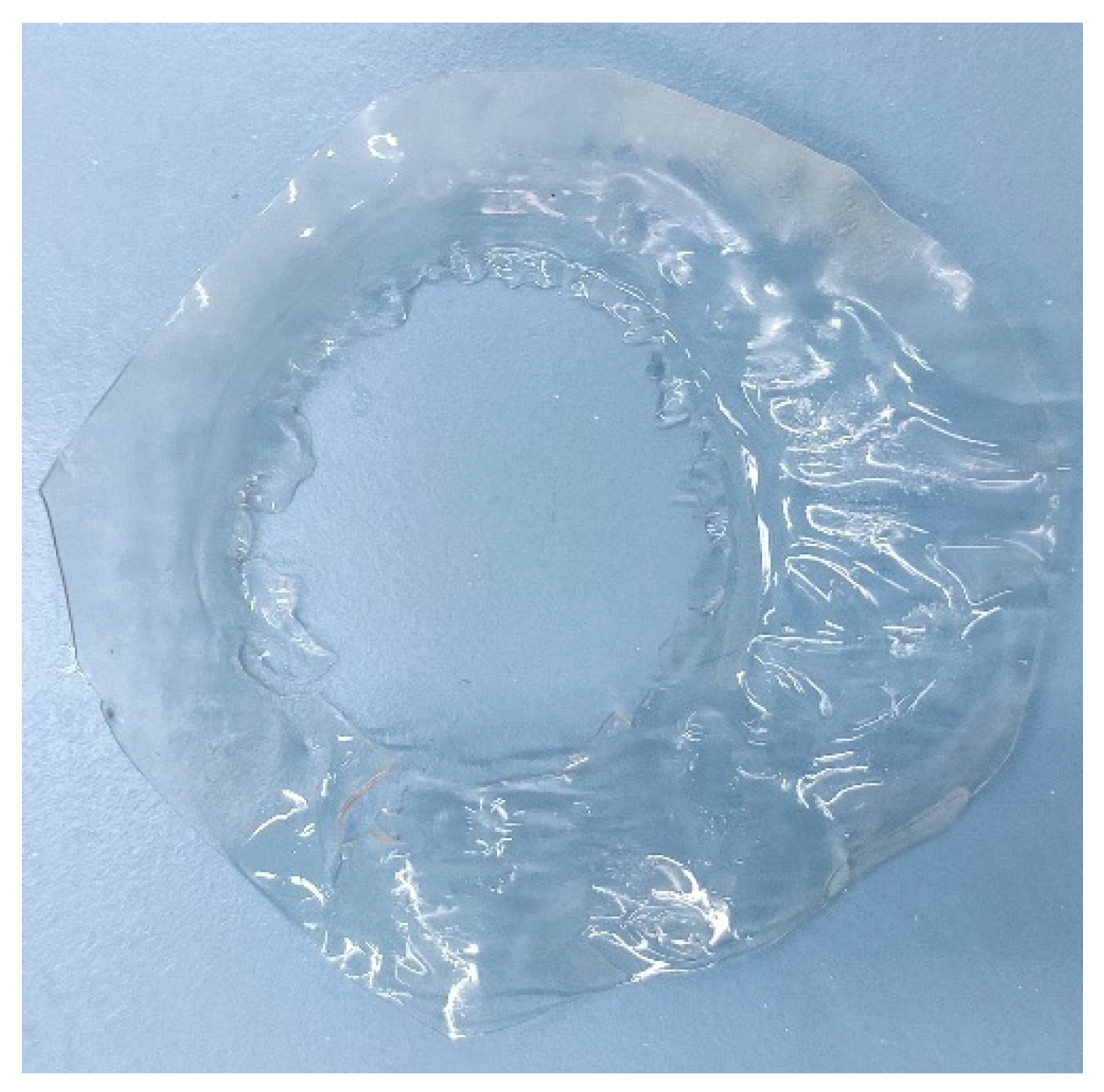
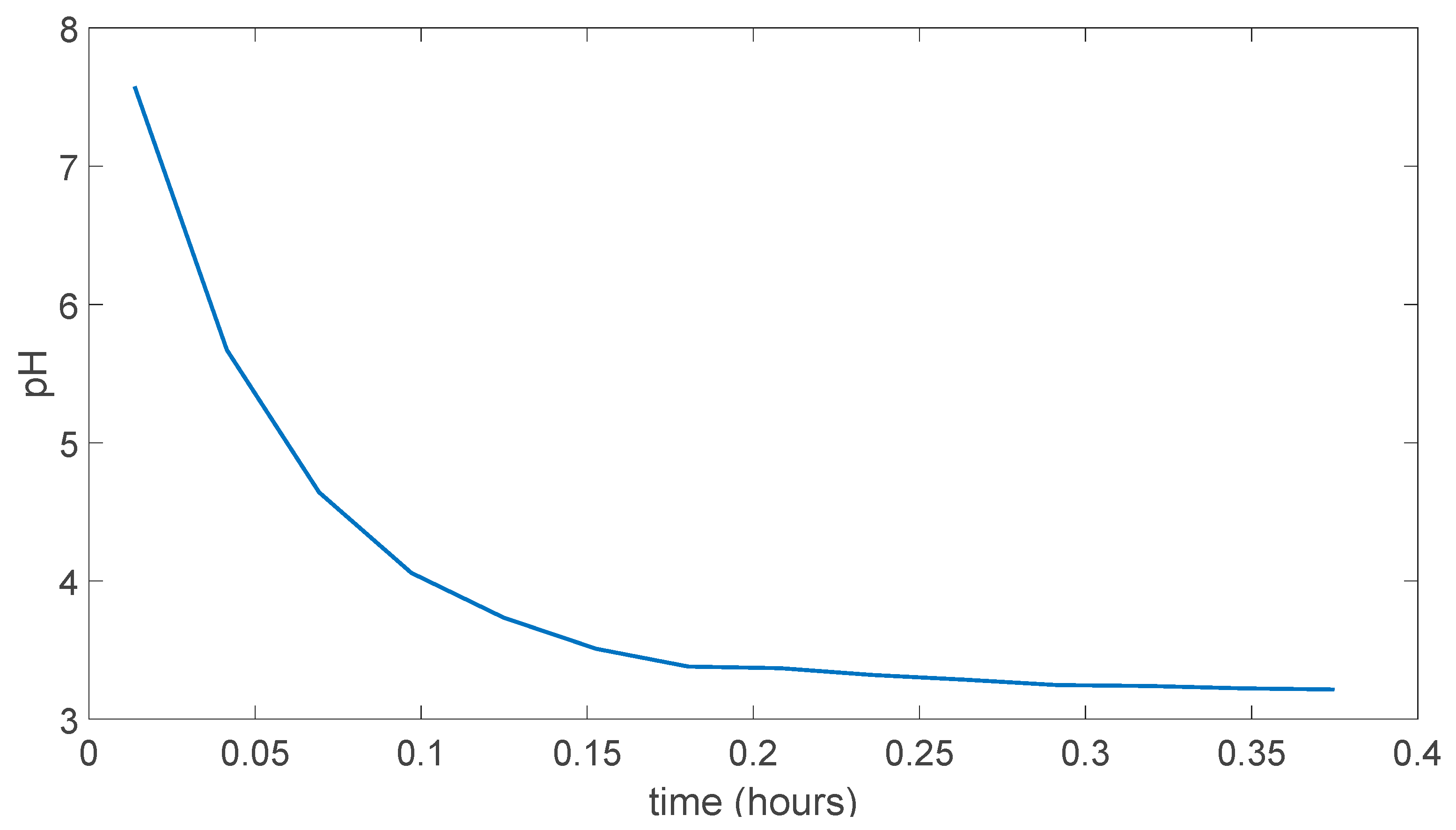
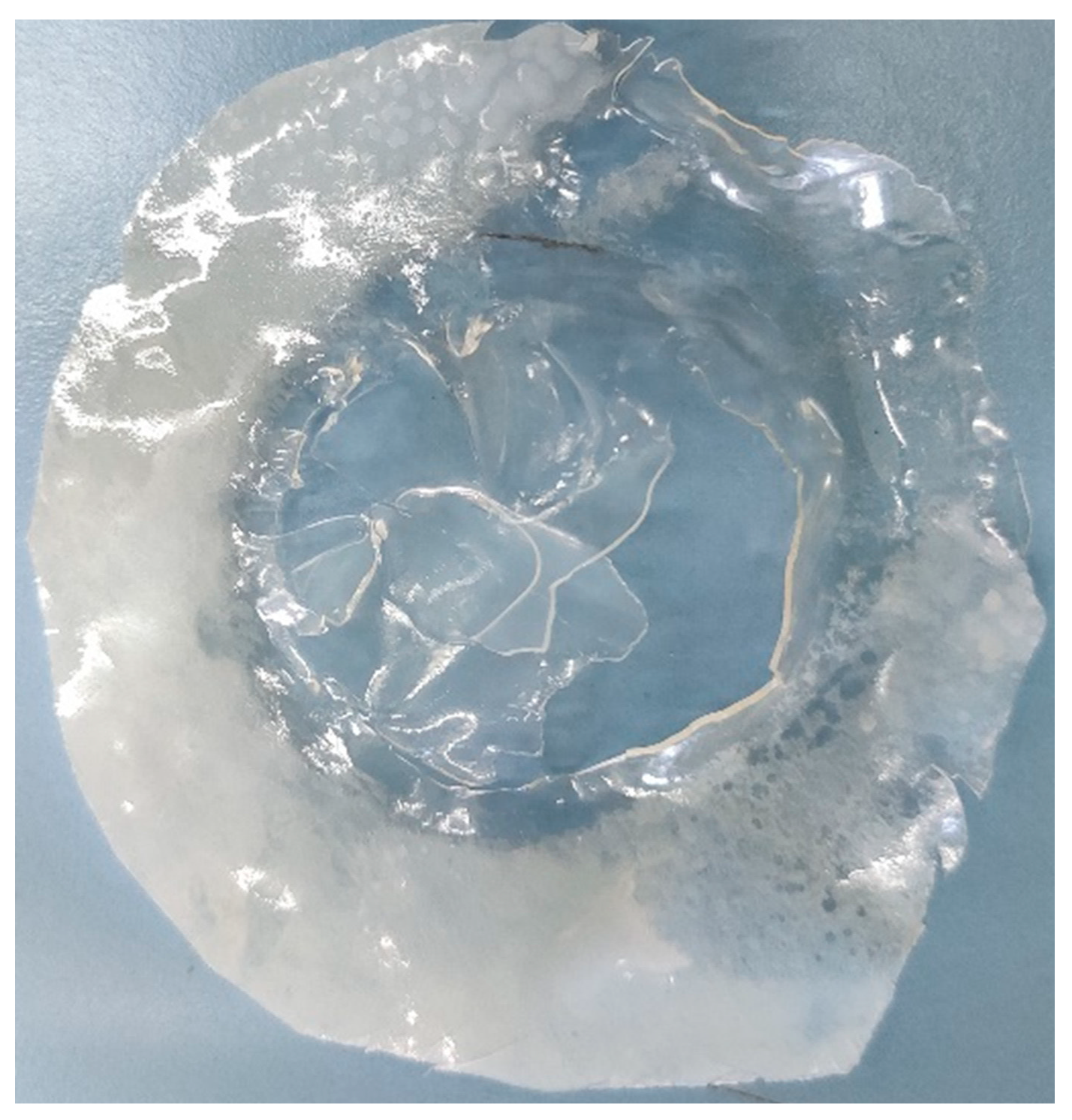
5.2. PVA/PAMA Membranes Results
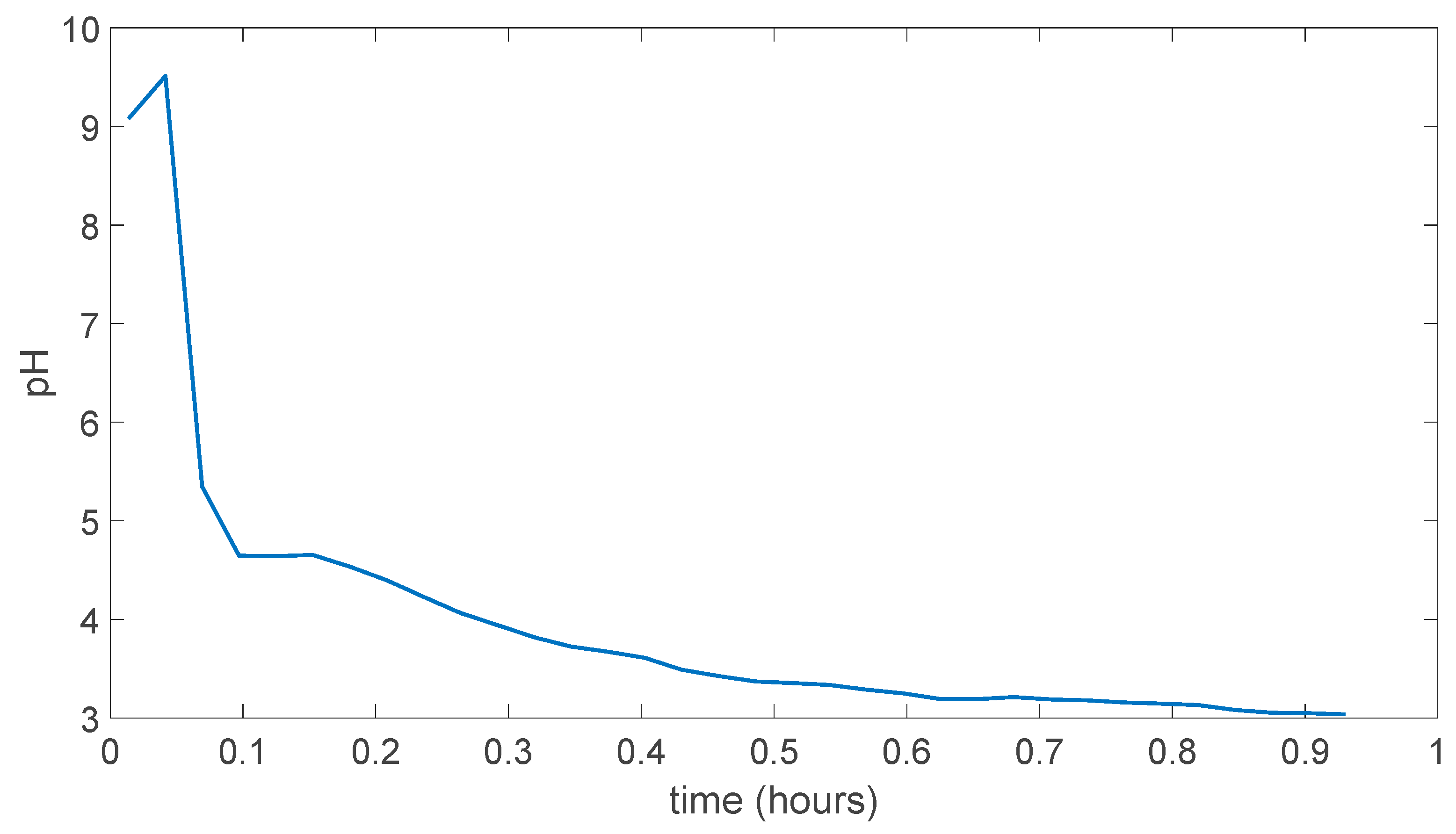

5.3. PVA/Tartaric Acid Membranes Results
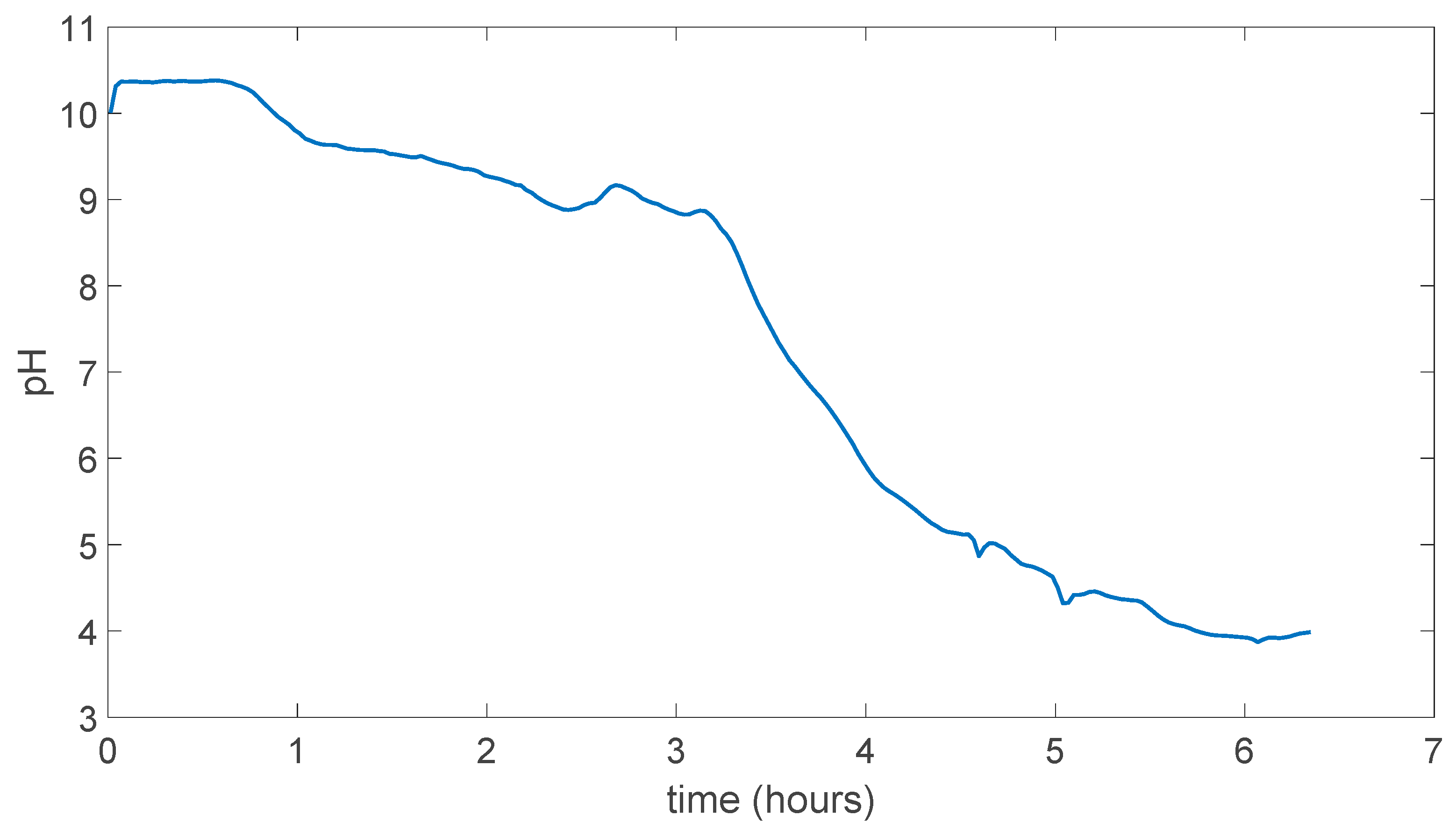
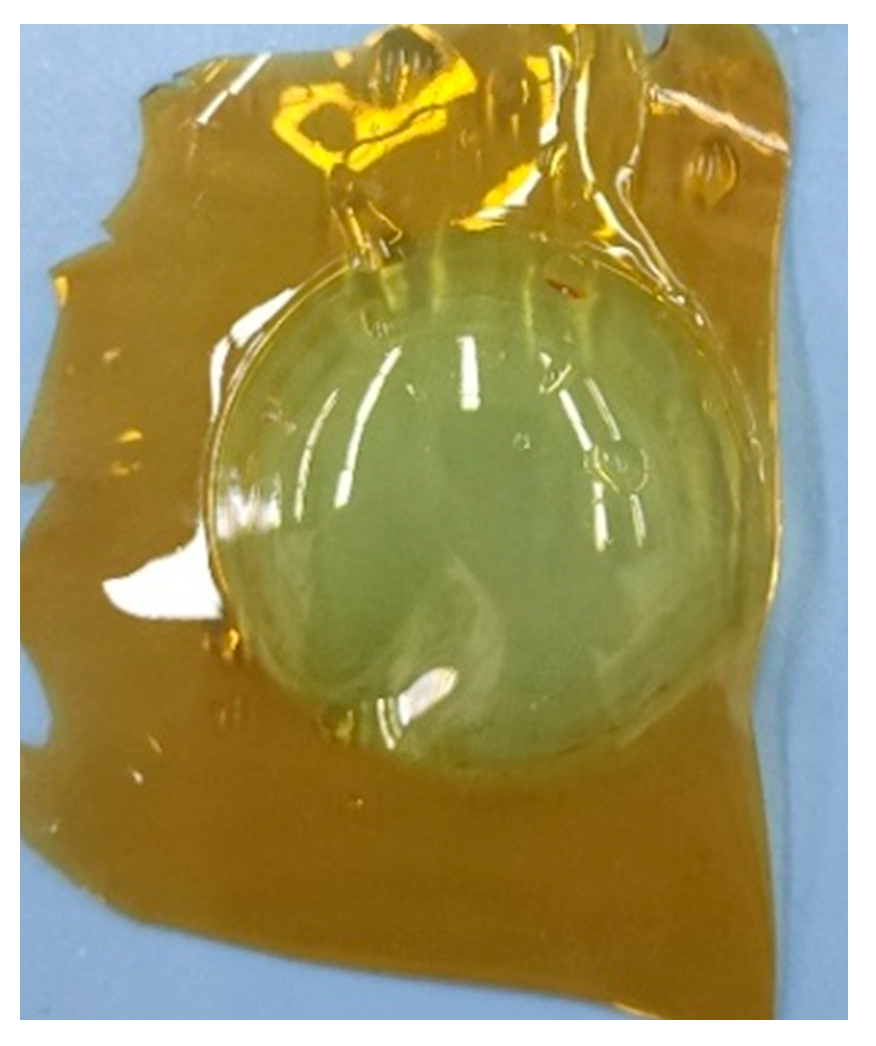
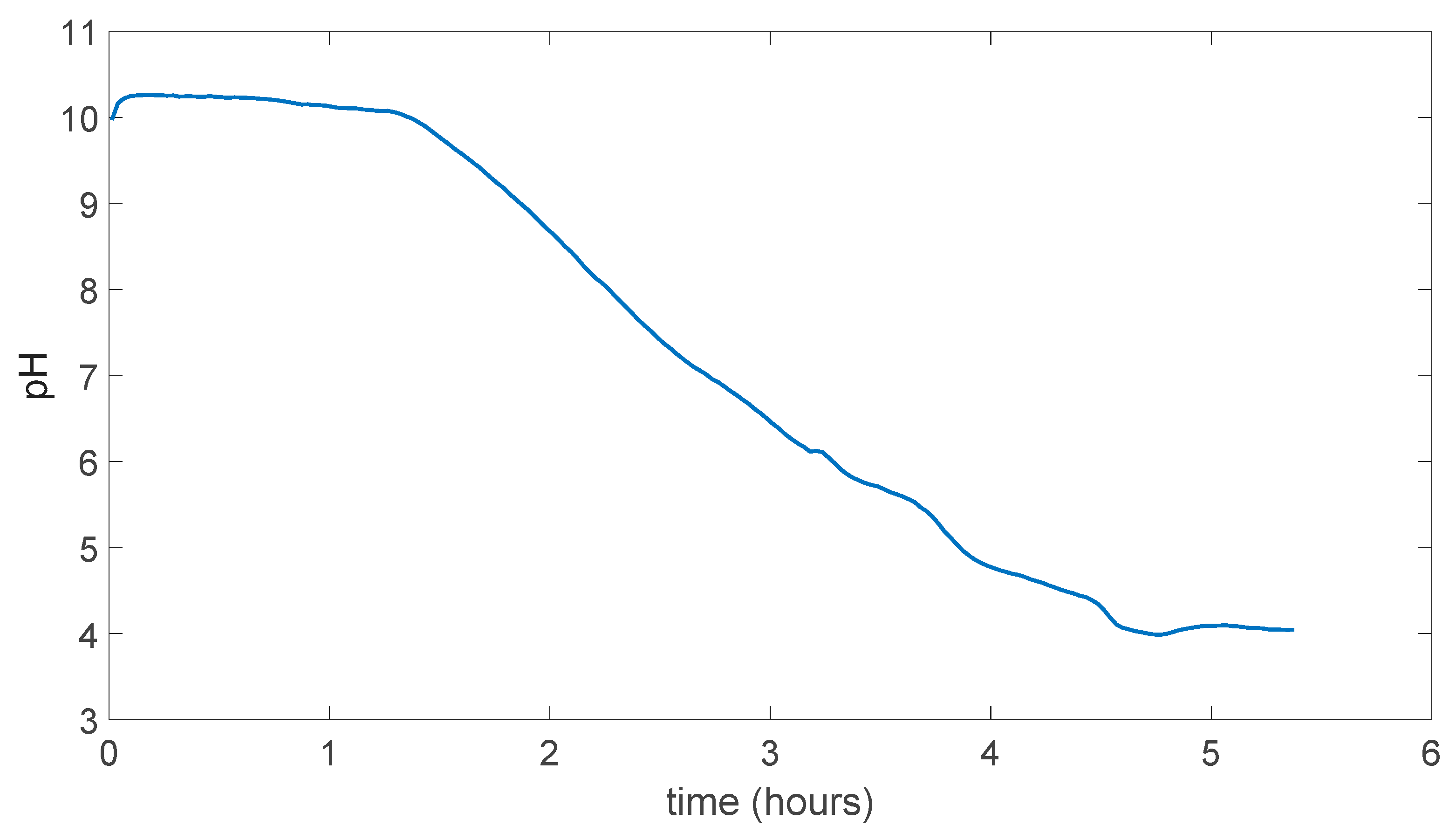

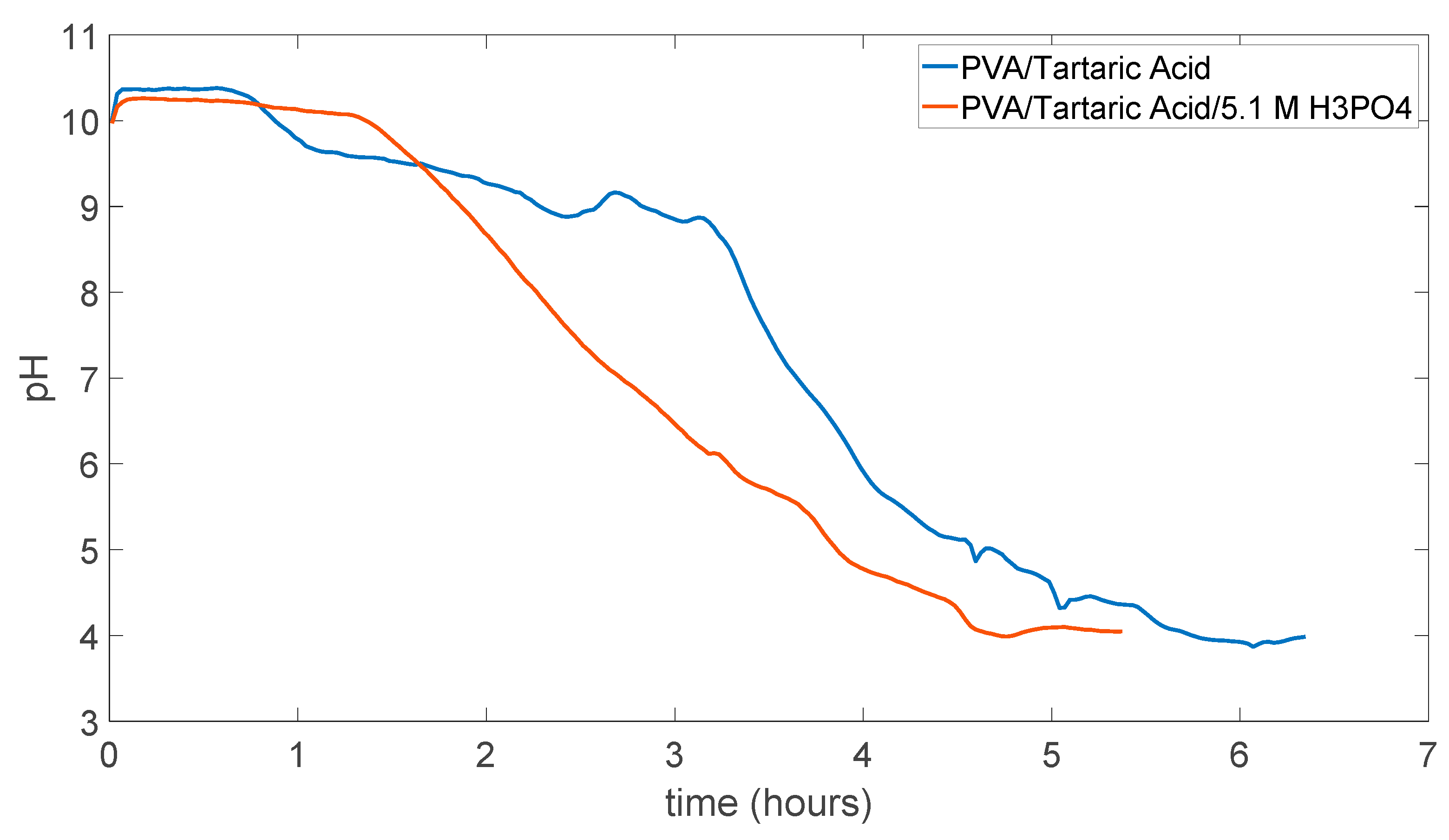
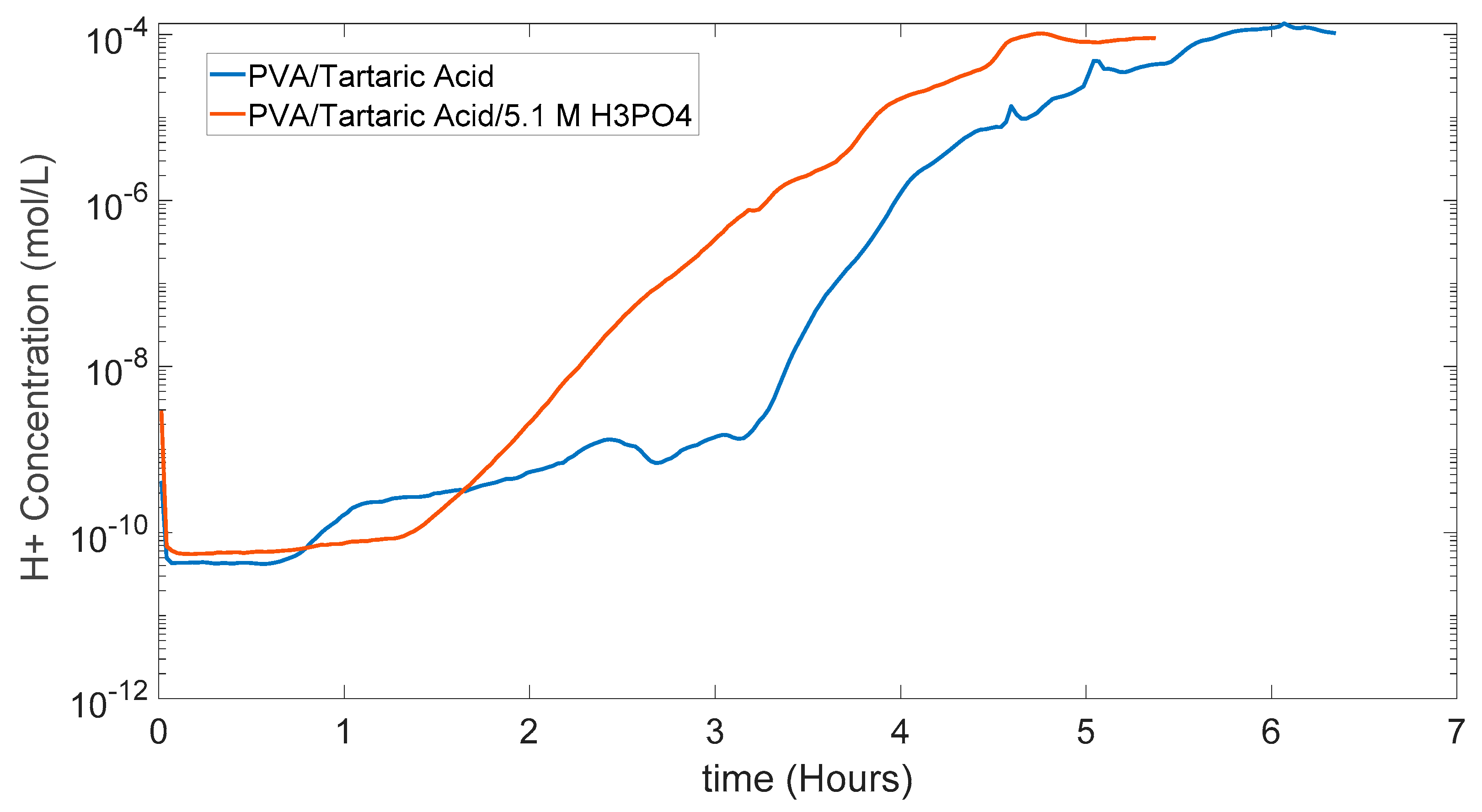
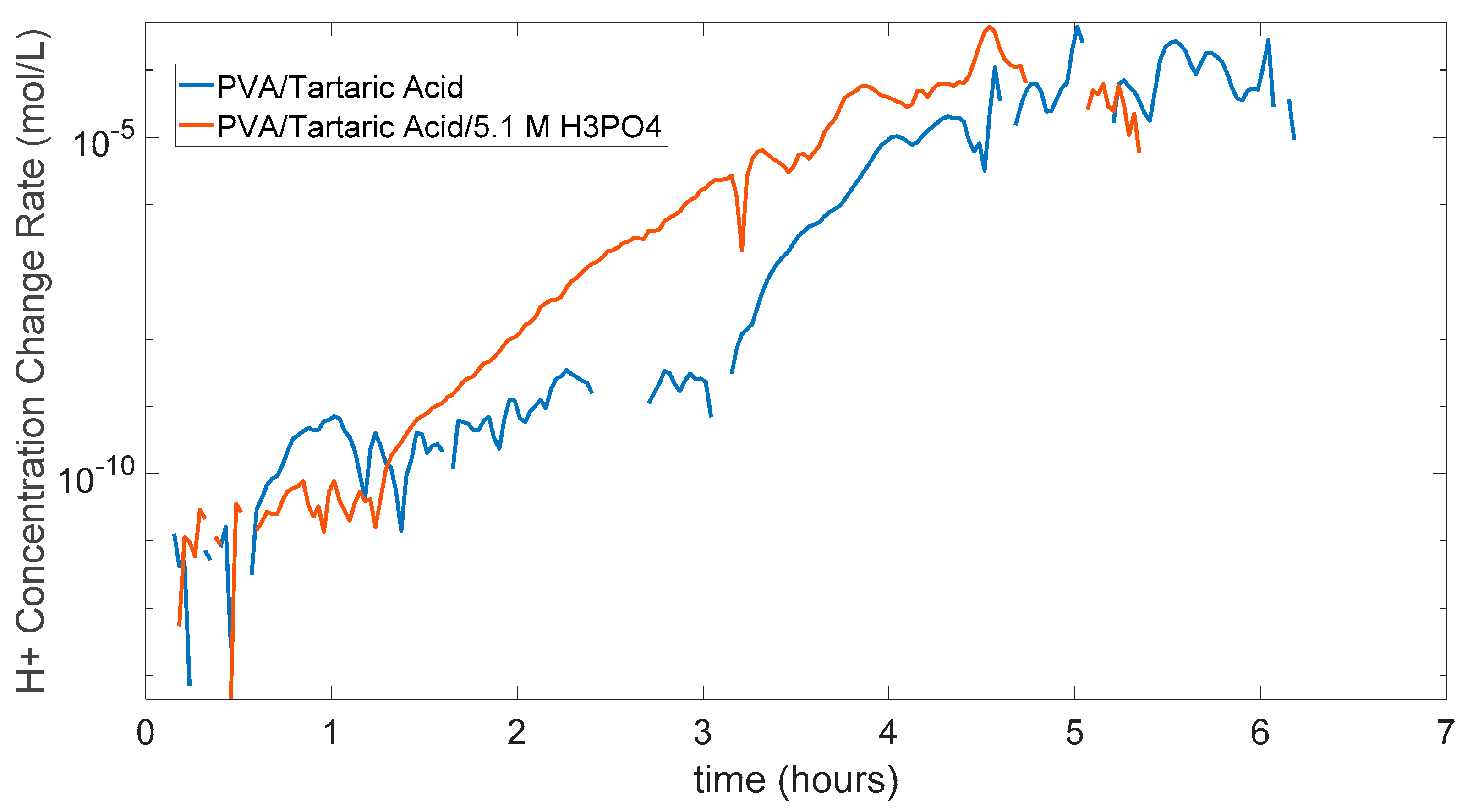
5.4. PDMS (Polydimethylsiloxane) Membrane Results
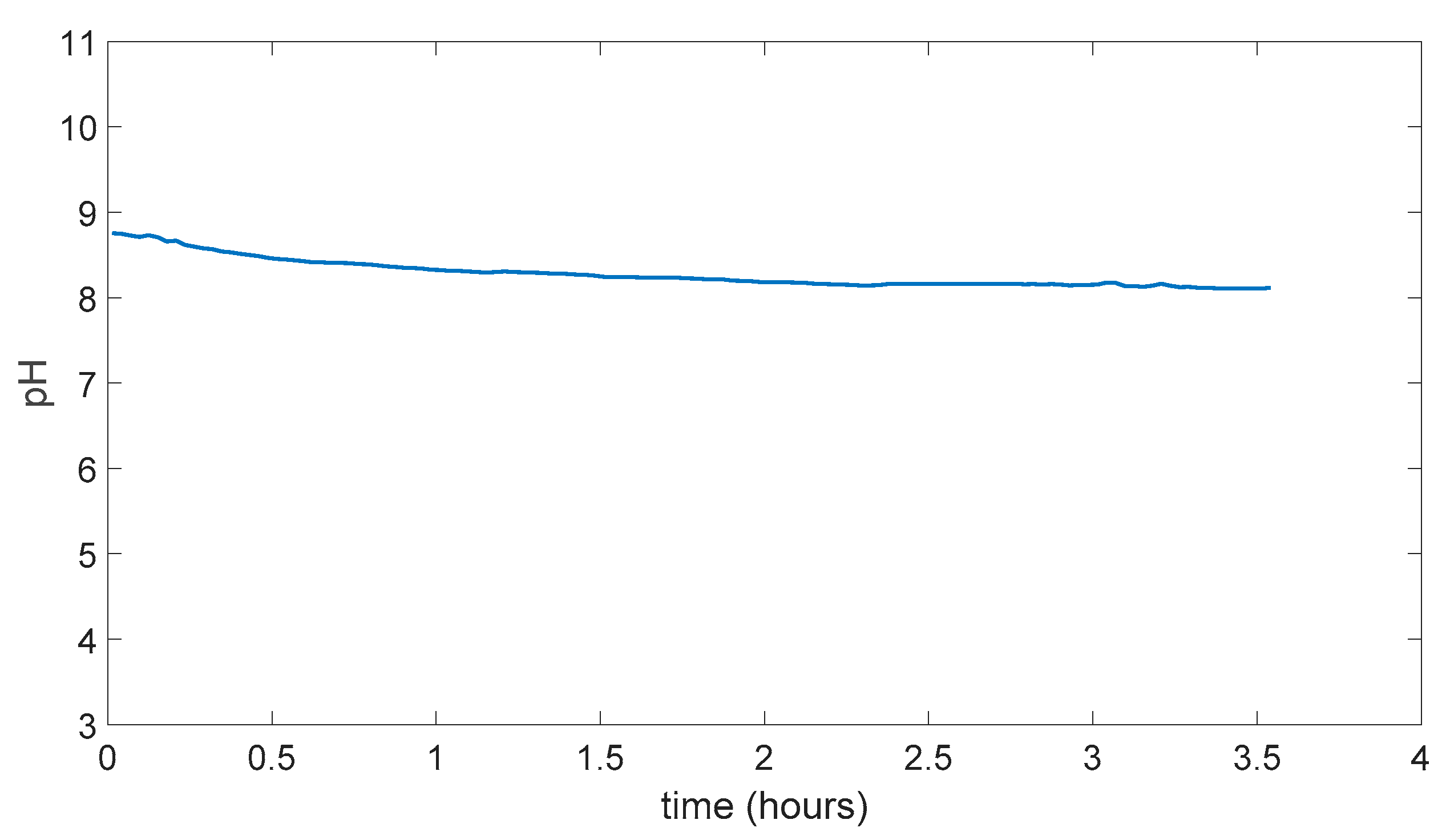
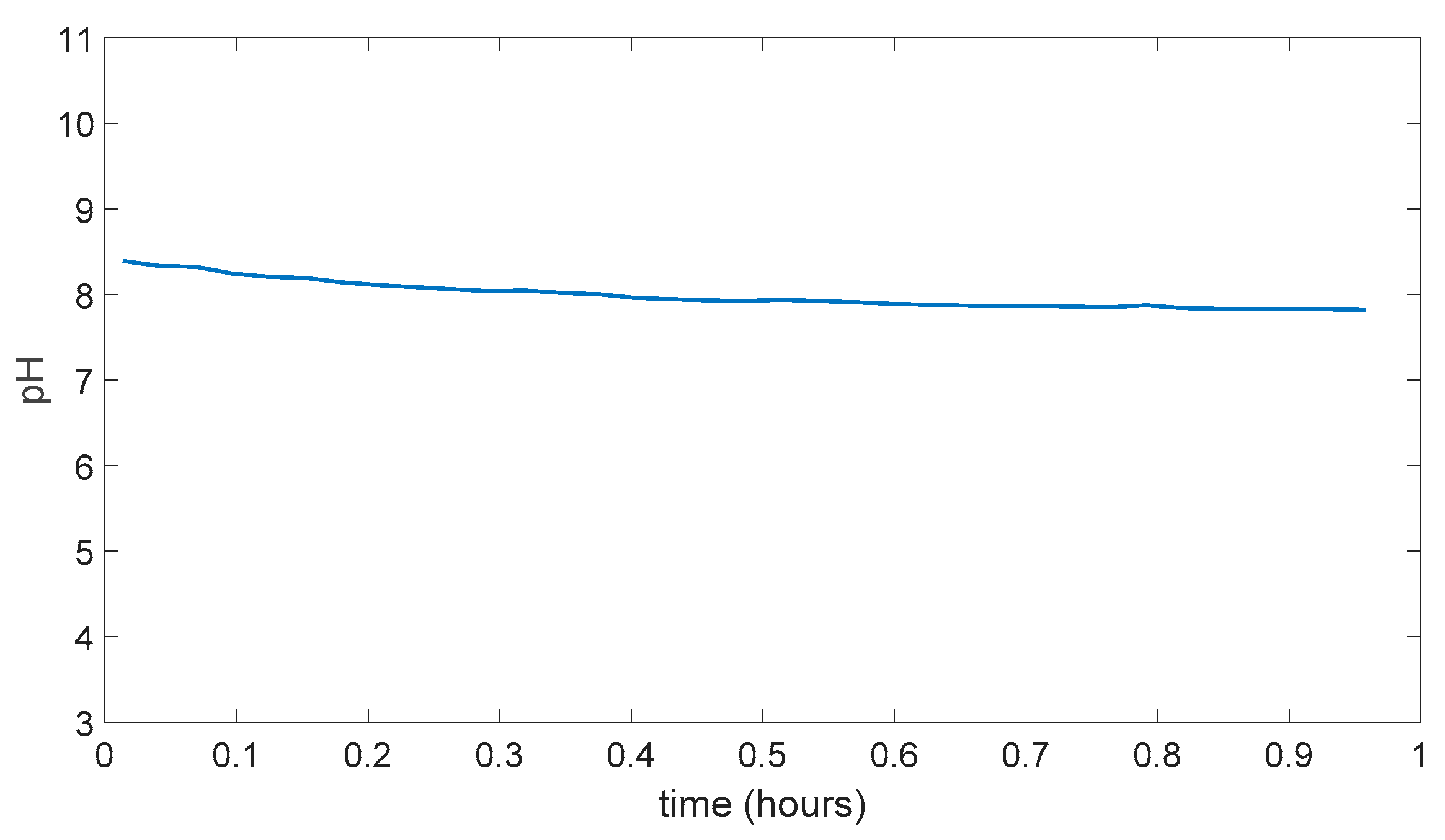
5.5. Swelling Data
| Membrane | Swelling (%) |
|---|---|
| (PVA)0.7(NaBr)0.3(H3PO4)0M | - |
| (PVA)0.7(NaBr)0.3(H3PO4)5.1M | - |
| PVA/PAMA | - |
| PVA/PAMA/5.1 M H3PO4 | - |
| PVA/Tartaric Acid | %16.3 |
| PVA/Tartaric Acid/5.1 M H3PO4 | %8.87 |
| PDMS | %0.116 |
6. CONCLUSION
Acknowledgments
INTERNET REFERENCES
- [1] Figure-1-functional-diagram-of-fuel-cell. Electrical A2Z. (n.d.). Retrieved April 10, 2022, from https://electricala2z.com/renewable-energy/fuel-cell-types-working/attachment/figure-1-functional-diagram-of-fuel-cell/
- [2] Gravity__analog_ph_sensor_meter_kit_v2_sku_sen0161-V2. DFRobot. (n.d.). Retrieved April 10, 2022, from https://wiki.dfrobot.com/Gravity__Analog_pH_Sensor_Meter_Kit_V2_SKU_SEN0161-V2#More_Documents
- [3] Comparison of Fuel Cell Technologies. Energy.gov. (n.d.). Retrieved May 22, 2022, from https://www.energy.gov/eere/fuelcells/comparison-fuel-cell-technologies
- [4] Memmert Ule500 Oven. American Instrument Exchange. (n.d.). Retrieved June 1, 2022, from https://www.americaninstrument.com/products/3735b-oven
- [5] Home. Cole. (n.d.). Retrieved June 1, 2022, from https://www.coleparmer.com/i/polyscience-7306a11b-standard-immersion-circulator-12l-120-vac/1210184
References
- A., H., F. Santos, J. A., Z., R., & Matencio, T. (2011). Ceramic materials for solid oxide fuel cells. Advances in Ceramics - Synthesis and Characterization, Processing and Specific Applications. [CrossRef]
- Ahmad, F., & Sheha, E. (2013). Preparation and physical properties of (PVA)0.7(NaBr)0.3(H3PO4)m solid acid membrane for phosphoric acid – fuel cells. Journal of Advanced Research, 4(2), 155–161. [CrossRef]
- Alberti, G. (2009). Fuel cells – proton-exchange membrane fuel cells membranes. Encyclopedia of Electrochemical Power Sources, 650–666. [CrossRef]
- Andújar, J. M., & Segura, F. (2009). Fuel cells: History and updating. A walk along two centuries. Renewable and Sustainable Energy Reviews, 13(9), 2309–2322. [CrossRef]
- Aung, N. N., Myat, Y. Y., Ngawhirunpat, T., Rojanarata, T., Patrojanasophon, P., Opanasopit, P., & Pamornpathomkul, B. (2019). Evaluation of thermally crosslinked poly(acrylic acid-co-maleic acid) (pama)/poly(vinyl alcohol) (PVA) Microneedle arrays. Key Engineering Materials, 819, 45–50. [CrossRef]
- Baykara, S. Z. (2018). Hydrogen: A brief overview on its sources, production and environmental impact. International Journal of Hydrogen Energy, 43(23), 10605–10614. [CrossRef]
- Boudghene Stambouli, A., & Traversa, E. (2002). Fuel cells, an alternative to standard sources of energy. Renewable and Sustainable Energy Reviews, 6(3), 295–304. [CrossRef]
- Carrette, L., Friedrich, K.A. and Stimming, U. (2000), Fuel Cells: Principles, Types, Fuels, and Applications. ChemPhysChem, 1: 162-193. [CrossRef]
- Colpan, C. O., Nalbant, Y., & Ercelik, M. (2018). 4.28 fundamentals of Fuel Cell Technologies. Comprehensive Energy Systems, 1107–1130. [CrossRef]
- Haile, S. M. (2003). Fuel cell materials and components. Acta Materialia, 51(19), 5981–6000. [CrossRef]
- Hema, M., Selvasekerapandian, S., Sakunthala, A., Arunkumar, D., & Nithya, H. (2008). Structural, vibrational and electrical characterization of PVA–NH4BR polymer electrolyte system. Physica B: Condensed Matter, 403(17), 2740–2747. [CrossRef]
- Kamran, M. (2021). Fuel cell. Renewable Energy Conversion Systems, 221–242. [CrossRef]
- Kumar, V., Kumar, P., Nandy, A., & Kundu, P. P. (2015). Crosslinked inter penetrating network of sulfonated styrene and sulfonated PVDF-co-HFP as electrolytic membrane in a single chamber microbial fuel cell. RSC Advances, 5(39), 30758–30767. [CrossRef]
- Lee, C. H., Park, H. B., Lee, Y. M., & Lee, R. D. (2005). Importance of proton conductivity measurement in polymer electrolyte membrane for fuel cell application. Industrial & Engineering Chemistry Research, 44(20), 7617–7626. [CrossRef]
- Lucia, U. (2014). Overview on fuel cells. Renewable and Sustainable Energy Reviews, 30, 164–169. [CrossRef]
- Mahato, N., Jang, H., Dhyani, A., & Cho, S. (2020). Recent progress in conducting polymers for hydrogen storage and fuel cell applications. In Polymers (Vol. 12, Issue 11, pp. 1–40). MDPI AG. [CrossRef]
- McLean, G. (2002). An assessment of Alkaline Fuel Cell Technology. International Journal of Hydrogen Energy, 27(5), 507–526. [CrossRef]
- Pourzare, K., Mansourpanah, Y., & Farhadi, S. (2016). Advanced nanocompositemembranes for fuel cell applications: A comprehensive review. Biofuel Research Journal, 3(4), 496–513. [CrossRef]
- Rozière, J., & Jones, D. J. (2003). Non-fluorinated polymer materials for proton exchange membrane fuel cells. Annual Review of Materials Research, 33(1), 503–555. [CrossRef]
- Sheha, E. (2010). Preparation and physical properties of (PVA)0.75(NH4BR)0.25(H2SO4)XM solid acid membrane. Journal of Non-Crystalline Solids, 356(43), 2282–2285. [CrossRef]
- Smitha, B. (2003). Synthesis and characterization of proton conducting polymer membranes for fuel cells. Journal of Membrane Science, 225(1-2), 63–76. [CrossRef]
- Sudhakar, Y. N., Selvakumar, M., & Bhat, D. K. (2018). Biopolymer electrolytes for fuel cell applications. Biopolymer Electrolytes, 151–166. [CrossRef]
- Ye, Y.-S., Rick, J., & Hwang, B.-J. (2012). Water soluble polymers as proton exchange membranes for fuel cells. Polymers, 4(2), 913–963. [CrossRef]
- Zuo, Z., Fu, Y., & Manthiram, A. (2012). Novel blend membranes based on acid-base interactions for fuel cells. Polymers, 4(4), 1627–1644. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




