Submitted:
22 August 2023
Posted:
24 August 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
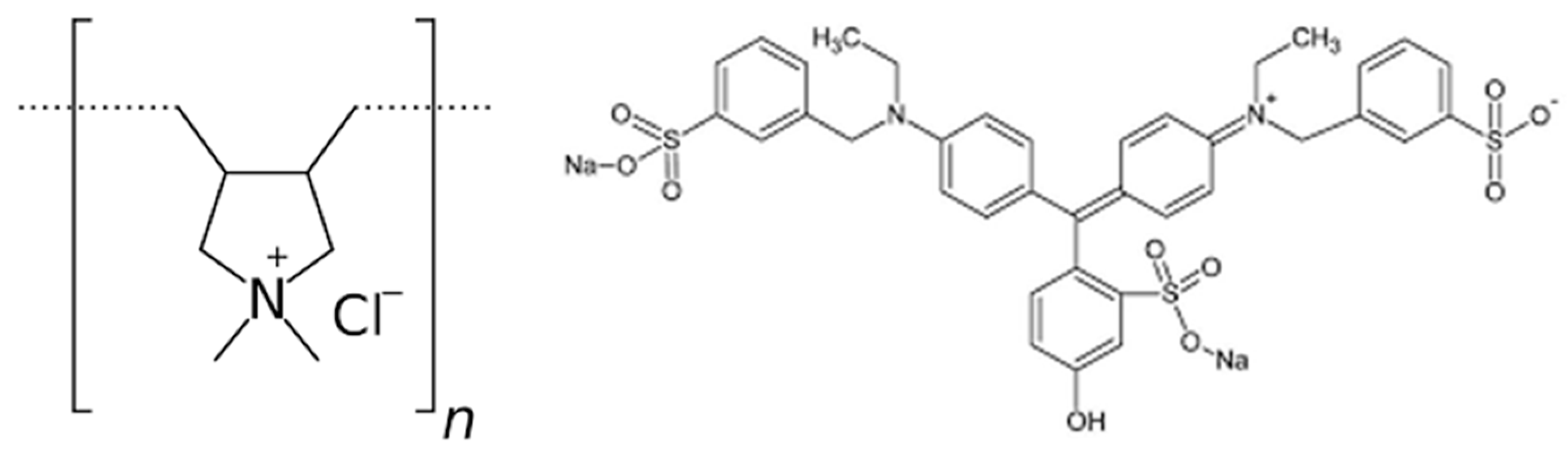
2.1. Materials
2.2. Complexation/calibration experiments description
2.3. Implementation in water with commercial PD
2.4. Influence of pH
2.5. Quantification of poly-DADMAC in coagulation water treatment samples
3. Results
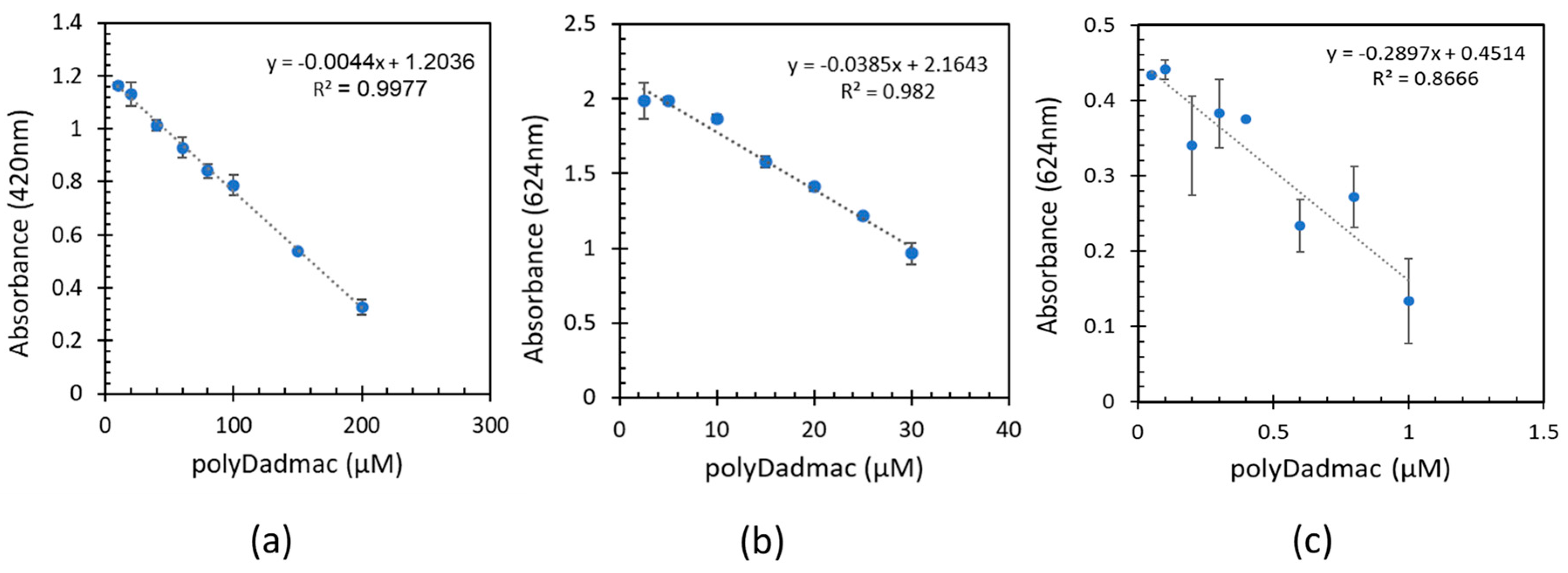
3.1. Quantification of analytical poly-DADMAC concentrations
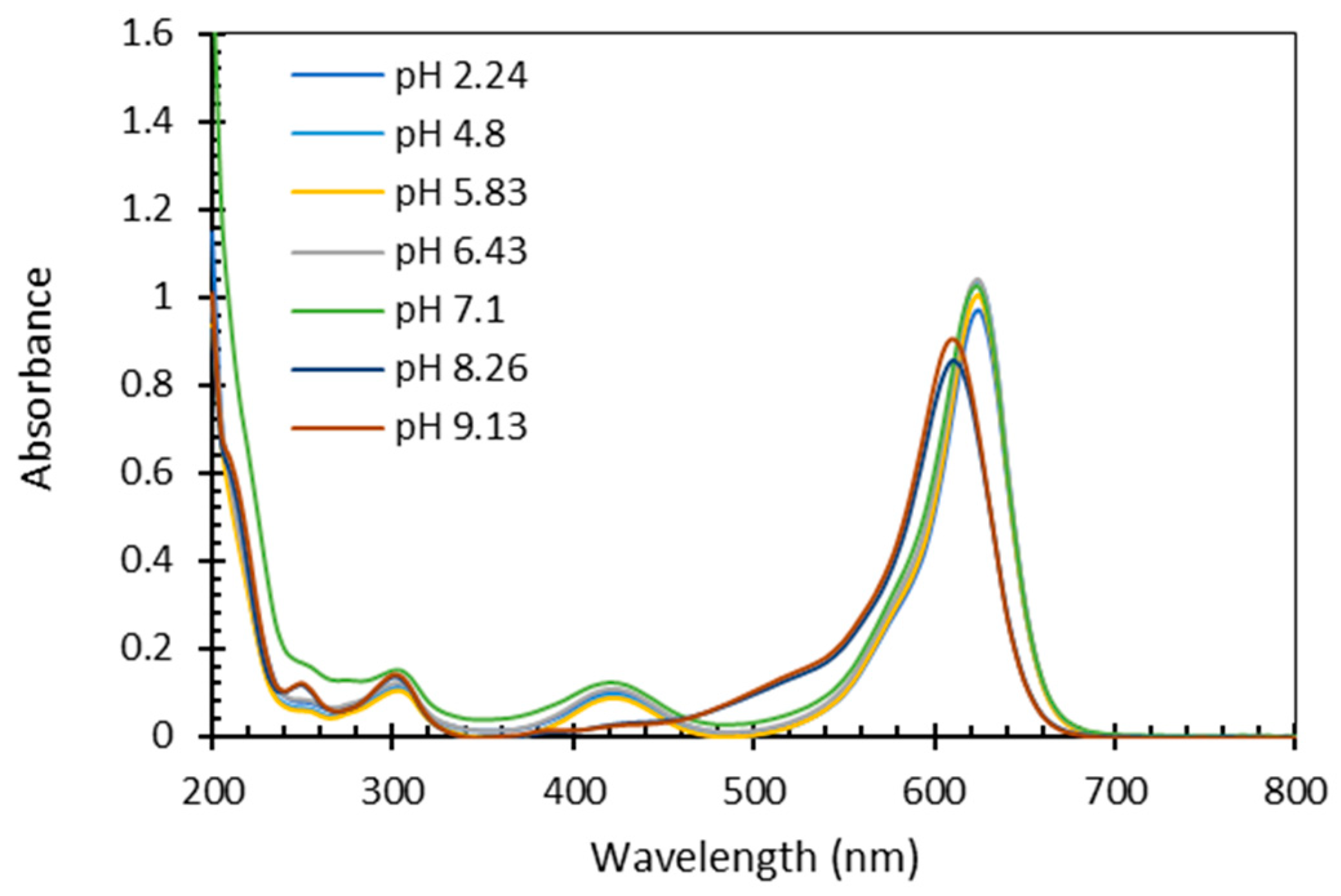
3.2. Influence of pH
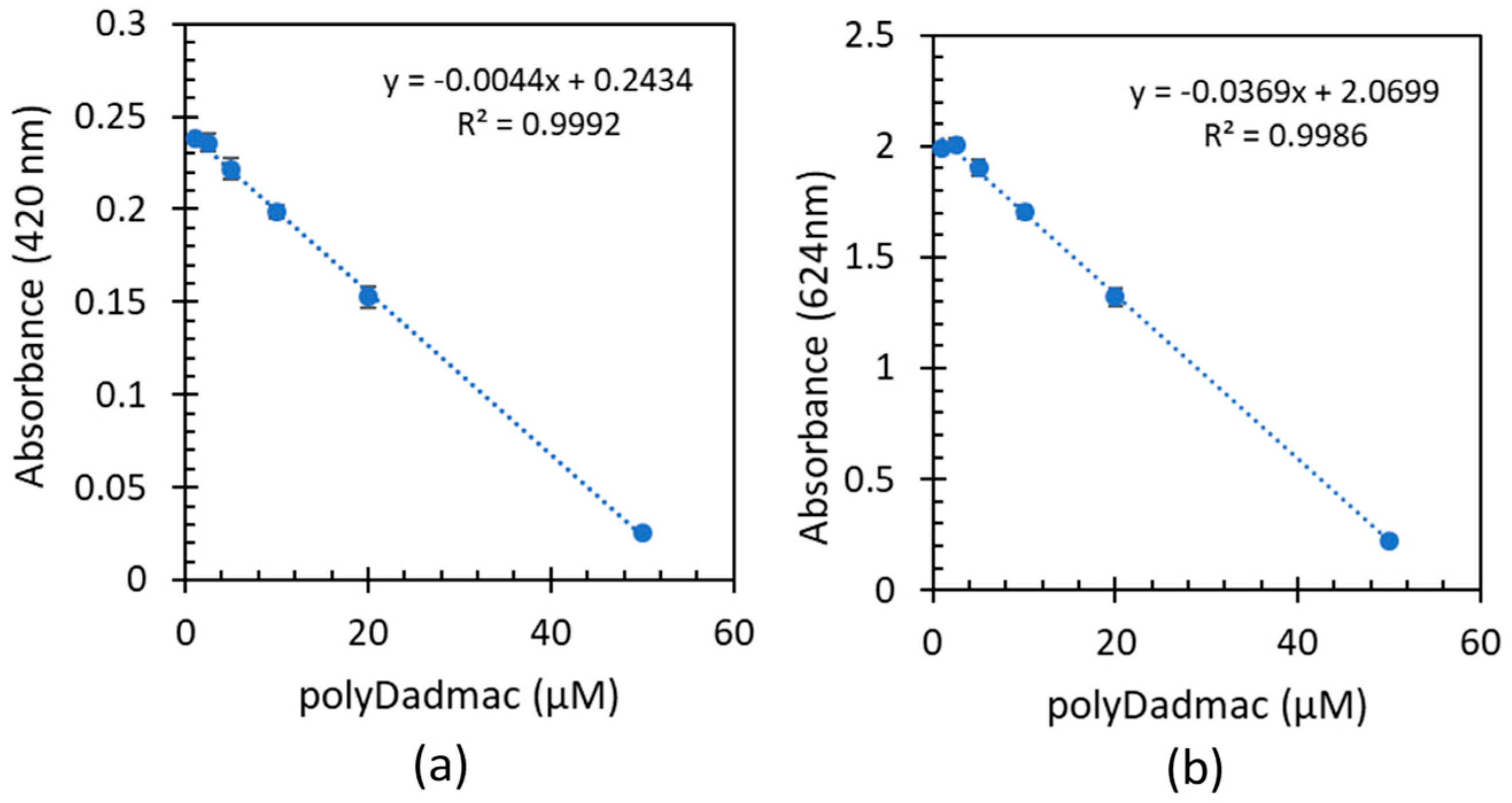
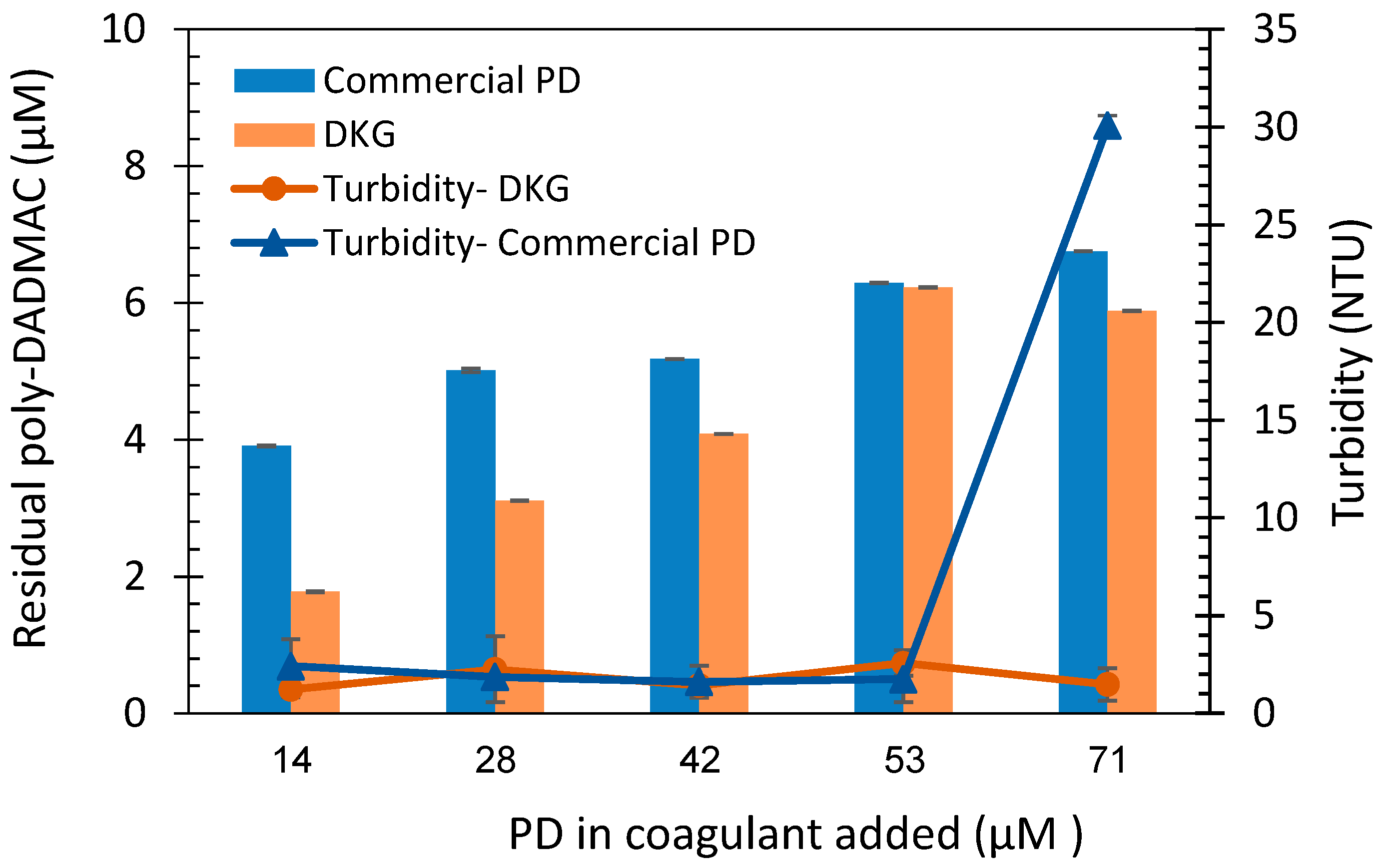
3.3. Quantification of commercial poly-DADMAC
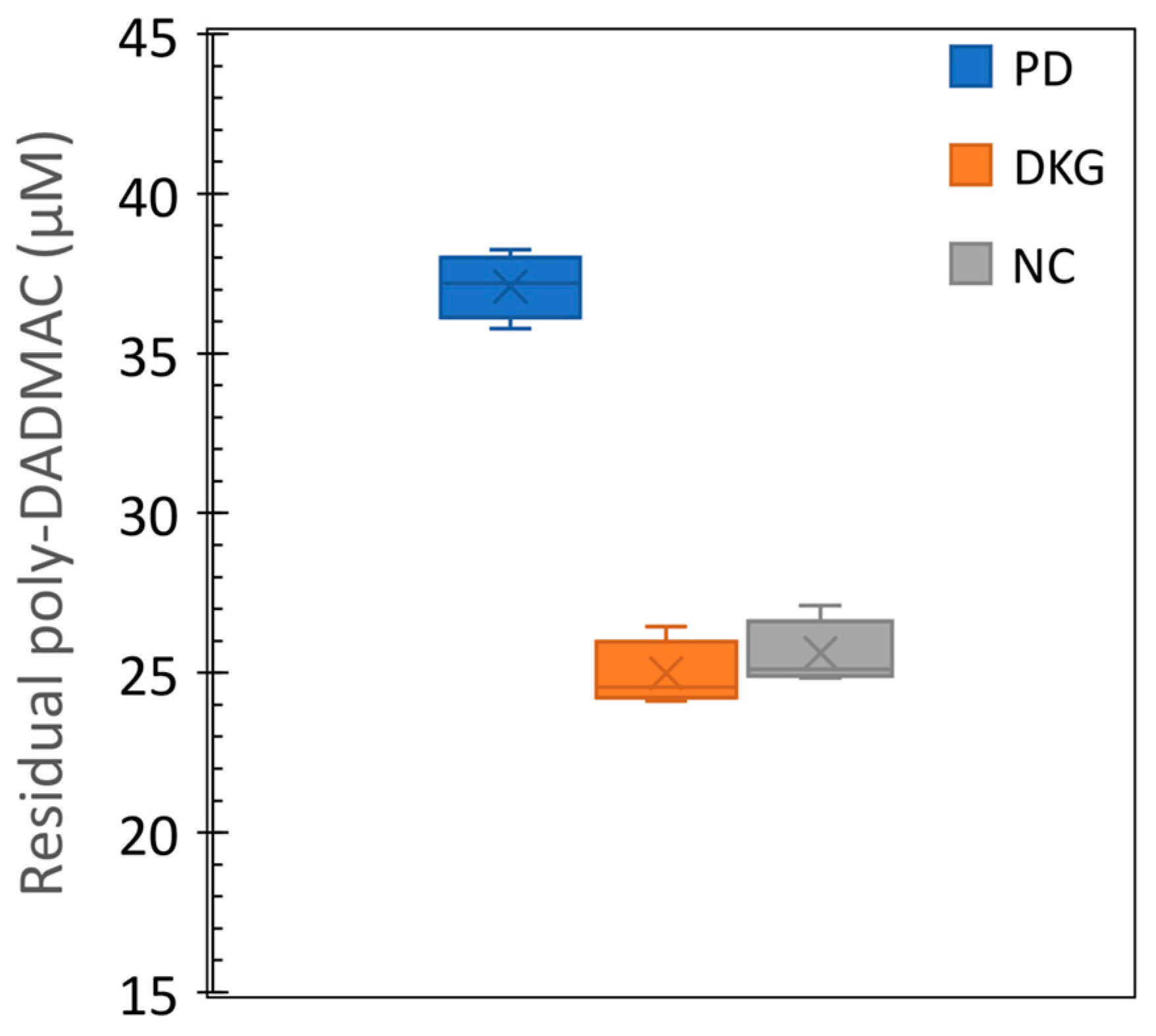
3.4. Quantification of poly-DADMAC in coagulation treatment samples
4. Conclusions
- In this study, a colorimetric quantification method for poly-DADMAC through complexation with fast green dye was developed and evaluated.
- The method exhibited high sensitivity with a detection limit of 0.02 µM (0.0032 mg L-1), meeting regulatory demands according to the world standard limitations.
- Quantification experiments of analytical PD concentrations including low, medium, and high ranges demonstrated a linear correlation between the absorbance and PD concentrations.
- The influence of pH on the quantification process was tested, revealing that the absorbance spectrum of FG remains unchanged within the pH range of 2 to 7, exhibiting an hypsochromic and hyperchromic shift at higher pH. Thus, acidification (or adequate pH measurements) step is necessary before quantification, to ensure FG spectrum is in the required range.
- The quantification method's feasibility was demonstrated in coagulation experiments, estimating remaining PD concentrations in treated water samples.
- The method's feasibility in estimating PD concentrations under "real" coagulation conditions highlights its potential for efficient monitoring and control of coagulation processes, offering a rapid, cost-effective, and sensitive tool for accurate measurement of PD concentrations.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amuda, O.S.; Amoo, I.A. Coagulation/flocculation process and sludge conditioning in beverage industrial wastewater treatment. J. Hazard. Mater. 2007, 141, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Dautzenberg, H.; Görnitz, E.; Jaeger, W. Synthesis and characterization of poly(diallyldimethylammonium chloride) in a broad range of molecular weight. Macromol. Chem. Phys. 1998, 199, 1561–1571. [Google Scholar] [CrossRef]
- Becker, N.S.; Bennett, D.; Bolto, B.; Dixon, D.; Eldridge, R.; Le, N.; Rye, C. Detection of polyelectrolytes at trace levels in water by fluorescent tagging. React. Funct. Polym. 2004, 60, 183–193. [Google Scholar] [CrossRef]
- Razali, M.A.A.; Ahmad, Z.; Ahmad, M.S.B.; Ariffin, A. Treatment of pulp and paper mill wastewater with various molecular weight of polyDADMAC induced flocculation. Chem. Eng. J. 2011, 166, 529–535. [Google Scholar] [CrossRef]
- Ariffin, A.; Razali, M.A.A.; Ahmad, Z. PolyDADMAC and polyacrylamide as a hybrid flocculation system in the treatment of pulp and paper mills waste water. Chem. Eng. J. 2012, 179, 107–111. [Google Scholar] [CrossRef]
- Hamad, M.J.A.; Chirwa, E.M.N. Forward osmosis for water recovery using polyelectrolyte PolyDADMAC and DADMAC draw solutions as a low pressure energy saving process. Desalination 2019, 453, 89–101. [Google Scholar] [CrossRef]
- Sampl, C.; Schaubeder, J.; Hirn, U.; Spirk, S. Interplay of electrolyte concentration and molecular weight of polyDADMAC on cellulose surface adsorption. Int. J. Biol. Macromol. 2023, 239, 124286. [Google Scholar] [CrossRef]
- Ramos, P.; Singh Kalra, S.; Johnson, N.W.; Khor, C.M.; Borthakur, A.; Cranmer, B.; Dooley, G.; Mohanty, S.K.; Jassby, D.; Blotevogel, J.; et al. Enhanced removal of per- and polyfluoroalkyl substances in complex matrices by polyDADMAC-coated regenerable granular activated carbon. Environ. Pollut. 2022, 294, 118603. [Google Scholar] [CrossRef]
- Gabelich, C.J.; Ishida, K.P.; Bold, R.M. Testing of water treatment copolymers for compatibility with polyamide reverse osmosis membranes. Environ. Prog. 2005, 24, 410–416. [Google Scholar] [CrossRef]
- Wang, S.; Liu, C.; Li, Q. Fouling of microfiltration membranes by organic polymer coagulants and flocculants: Controlling factors and mechanisms. Water Res. 2011, 45, 357–365. [Google Scholar] [CrossRef]
- Zhang, B.; Tang, H.; Shen, Y.; Liu, G.; Shi, W. Comparative analysis of membrane fouling mechanisms induced by colloidal polymer: Effects of sodium and calcium ions. J. Colloid Interface Sci. 2022, 608, 780–791. [Google Scholar] [CrossRef]
- Uzun, H.; Kim, D.; Karanfil, T. Removal of wastewater and polymer derived N-nitrosodimethylamine precursors with integrated use of chlorine and chlorine dioxide. Chemosphere 2019, 216, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Padhye, L.P.; Wang, P.; Cho, M.; Kim, J.-H.; Huang, C.-H. N-nitrosodimethylamine (NDMA) formation potential of amine-based water treatment polymers: Effects of in situ chloramination, breakpoint chlorination, and pre-oxidation. J. Hazard. Mater. 2015, 282, 133–140. [Google Scholar] [CrossRef]
- Park, S.H.; Wei, S.; Mizaikoff, B.; Taylor, A.E.; Favero, C.; Huang, C.H. Degradation of amine-based water treatment polymers during chloramination as N-nitrosodimethylamine (NDMA) precursors. Environ. Sci. Technol. 2009, 43, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Padhye, L.P.; Park, S.H. PolyDADMAC and dimethylamine as precursors of N-nitrosodimethylamine during ozonation. Water Qual. Technol. Conf. Expo. 2011 2011, 2304–2315. [Google Scholar]
- Atkinson, A.J.; Fischer, N.; Donovan, S.; Bartlett, J.; Alrehaili, O.; Sinha, S.; Kommineni, S.; Herckes, P.; Westerhoff, P. Purification and removal of the low molecular weight fraction of polyDADMAC reduces N -nitrosodimethylamine formation during water treatment. Environ. Sci. Water Res. Technol. 2020, 6, 2492–2498. [Google Scholar] [CrossRef]
- T Manickum Occurrence, Fate and Preliminary Environmental Risk Assessment of Residual Poly-Diallyldimethyl Ammonium Chloride, and Some Disinfection By-Products, in Treated (Potable), and Environmental, Waters in the Umgeni Water Catchment in Kwazulu-Natal. SM J. Public Heal. Epidemiol. 2017, 3.
- Al Momani, F.A.; Örmeci, B. Measurement of polyacrylamide polymers in water and wastewater using an in-line UV-vis spectrophotometer. J. Environ. Chem. Eng. 2014, 2, 765–772. [Google Scholar] [CrossRef]
- Di Gaudio, F.; Barreca, S.; Orecchio, S. Diallyldimethylammonium Chloride (DADMAC) in Water Treated with Poly-Diallyldimethylammonium Chloride (PDADMAC) by Reversed-Phase Ion-Pair Chromatography—Electrospray Ionization Mass Spectrometry. Separations 2023, 10, 311. [Google Scholar] [CrossRef]
- John, W. Synthesis, properties and analysis of polydadmac for water purification. 2008. [Google Scholar]
- Mwangi, I.W.; Ngila, J.C.; Ndungu, P.; Msagati, T.A.M. Method Development for the Determination of Diallyldimethylammonium Chloride at Trace Levels by Epoxidation Process. Water. Air. Soil Pollut. 2013, 224, 1638. [Google Scholar] [CrossRef]
- Mwangi, I.W.; Catherine Ngila, J.; Ndungu, P.; Msagati, T. Preconcentration and spectrophotometric determination of polyDADMAC in treated water by in situ co-precipitation with naphthalene. Phys. Chem. Earth, Parts A/B/C 2014, 72–75, 54–60. [Google Scholar] [CrossRef]
- Mwangi, I.; Ngila, J.; Ndungu, P. A new spectrophotometric method for determination of residual polydiallyldimethylammonium chloride flocculant in treated water based on a diazotization-coupled ion pair. Water SA 2012, 38, 707–714. [Google Scholar] [CrossRef]
- Gumbi, B.; Ngila, J.C.; Ndungu, P.G. Gold nanoparticles for the quantification of very low levels of poly-diallyldimethylammonium chloride in river water. Anal. Methods 2014, 6, 6963. [Google Scholar] [CrossRef]
- Manickum, T.; John, W.; Toolsee, N.; Rajagopaul, R. Preliminary Performance Evaluation of the Gold Nanoparticle Method for Quantification of Residual Poly-(Diallyldimethyl Ammonium Chloride) in Treated Waters in the Umgeni Water Catchment, Kwazulu-Natal (South Africa). Hydrol. Curr. Res. 2015, 06. [Google Scholar]
- Magubane, S.E.; Ntlhoro, S.; Sabela, M.; Kanchi, S.; Mlambo, M.; Onwubu, S.C.; Mdluli, P.S.; Inamuddin; Asiri, A.M. Novel on-site residual screening of poly-diallyldimethylammonium chloride in treated potable water using gold nanoparticle based lovibond color filters. J. Taiwan Inst. Chem. Eng. 2019, 101, 159–166. [Google Scholar] [CrossRef]
- Majam, S.; Thompson, P.A. Polyelectrolyte determination in drinking water. Water SA 2006, 32, 705–707. [Google Scholar] [CrossRef]
- Thilak Kumar, R.; Umamaheswari, S. FTIR, FTR and UV-Vis analysis of carbamazepine. Res. J. Pharm. Biol. Chem. Sci. 2011, 2, 685–693. [Google Scholar]
- Mendelovits, A.; Prat, T.; Gonen, Y.; Rytwo, G.; Rytwoa, G. Improved colorimetric determination of chitosan concentrations by dye binding. Appl. Spectrosc. 2012, 66, 979–982. [Google Scholar] [CrossRef]
- Venkatesh, N.; Murugadoss, G.; Mohamed, A.A.A.; Kumar, M.R.; Peera, S.G.; Sakthivel, P. A Novel Nanocomposite Based on Triazine Based Covalent Organic Polymer Blended with Porous g-C3N4 for Photo Catalytic Dye Degradation of Rose Bengal and Fast Green. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Rytwo, G.; Lavi, R.; Rytwo, Y.; Monchase, H.; Dultz, S.; König, T.N. Clarification of olive mill and winery wastewater by means of clay-polymer nanocomposites. Sci. Total Environ. 2013, 442, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Rytwo, G. The Use of Clay-Polymer Nanocomposites in Wastewater Pretreatment. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Rytwo, G. Hybrid Clay-Polymer Nanocomposites for the Clarification of Water and Effluents. Recent Pat. Nanotechnol. 2017, 11, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Rytwo, G. US20190152813 Method for production of potable water 2019.
- Sukenik, A.; Beardall, J.; Hadas, O. Photosynthetic characterization of developing and mature akinetes of Aphanizomenon ovalisporum (Cyanoprokaryota). J. Phycol. 2007, 43, 780–788. [Google Scholar] [CrossRef]
- Rytwo, G.; Lavi, R.; König, T.N.; Avidan, L. Direct Relationship Between Electrokinetic Surface-charge Measurement of Effluents and Coagulant Type and Dose. Colloids Interface Sci. Commun. 2014, 1, 27–30. [Google Scholar] [CrossRef]
- Rytwo, G.; Malka, H. A pilot plant for the treatment of cowshed efflFuents. Water Irrig. 2013, 530, 6–9. [Google Scholar]
- Inbar, Y. New Standards for Treated Wastewater Reuse in Israel. In Proceedings of the Wastewater Reuse--Risk Assessment, Decision-Making and Environmental Security; Zaidi, M.K., Ed.; Springer Netherlands: Dordrecht, 2007; pp. 291–296. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



