Submitted:
16 August 2023
Posted:
18 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
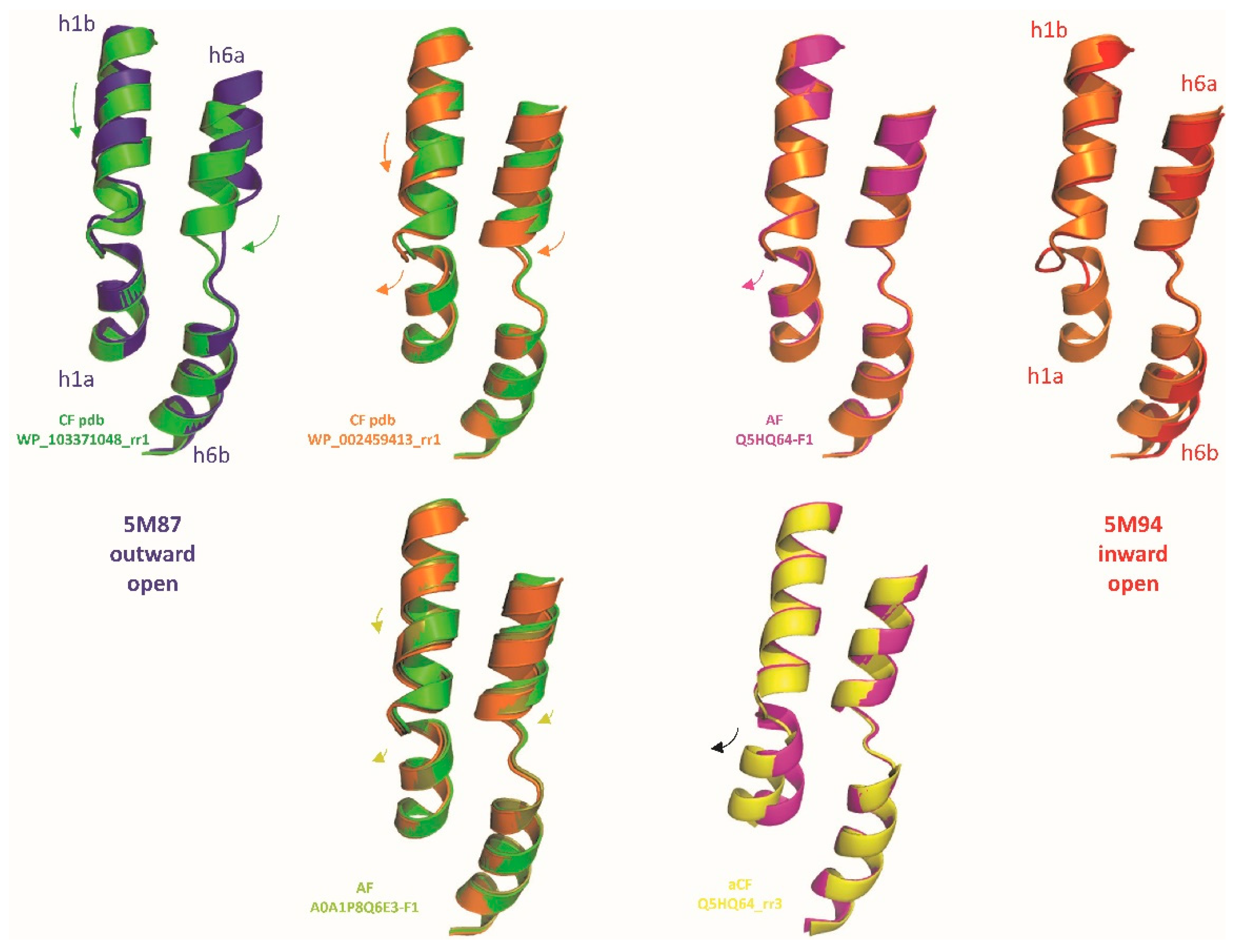
2.1. AF2/CF predict native MCb carrier cycle intermediates
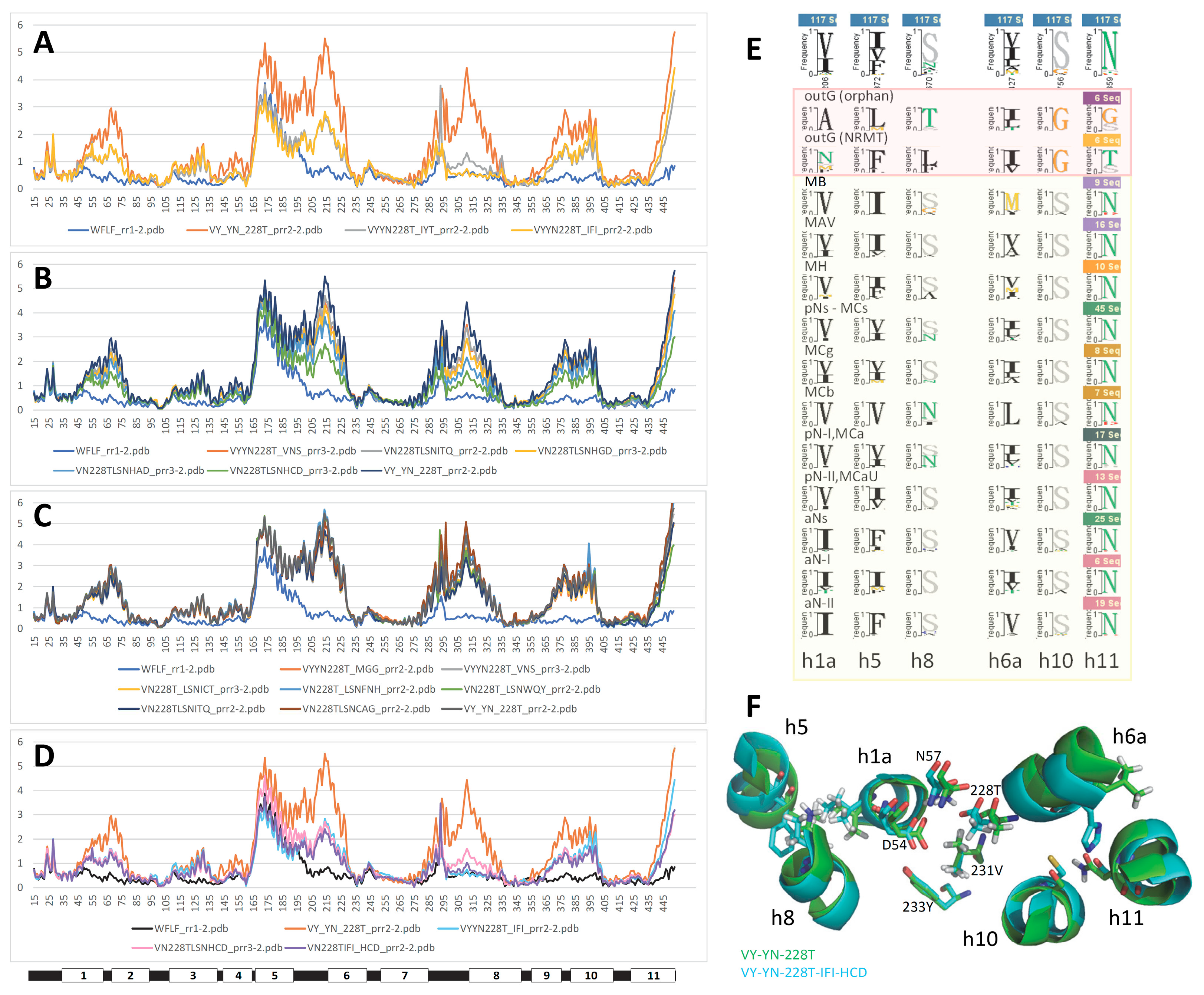
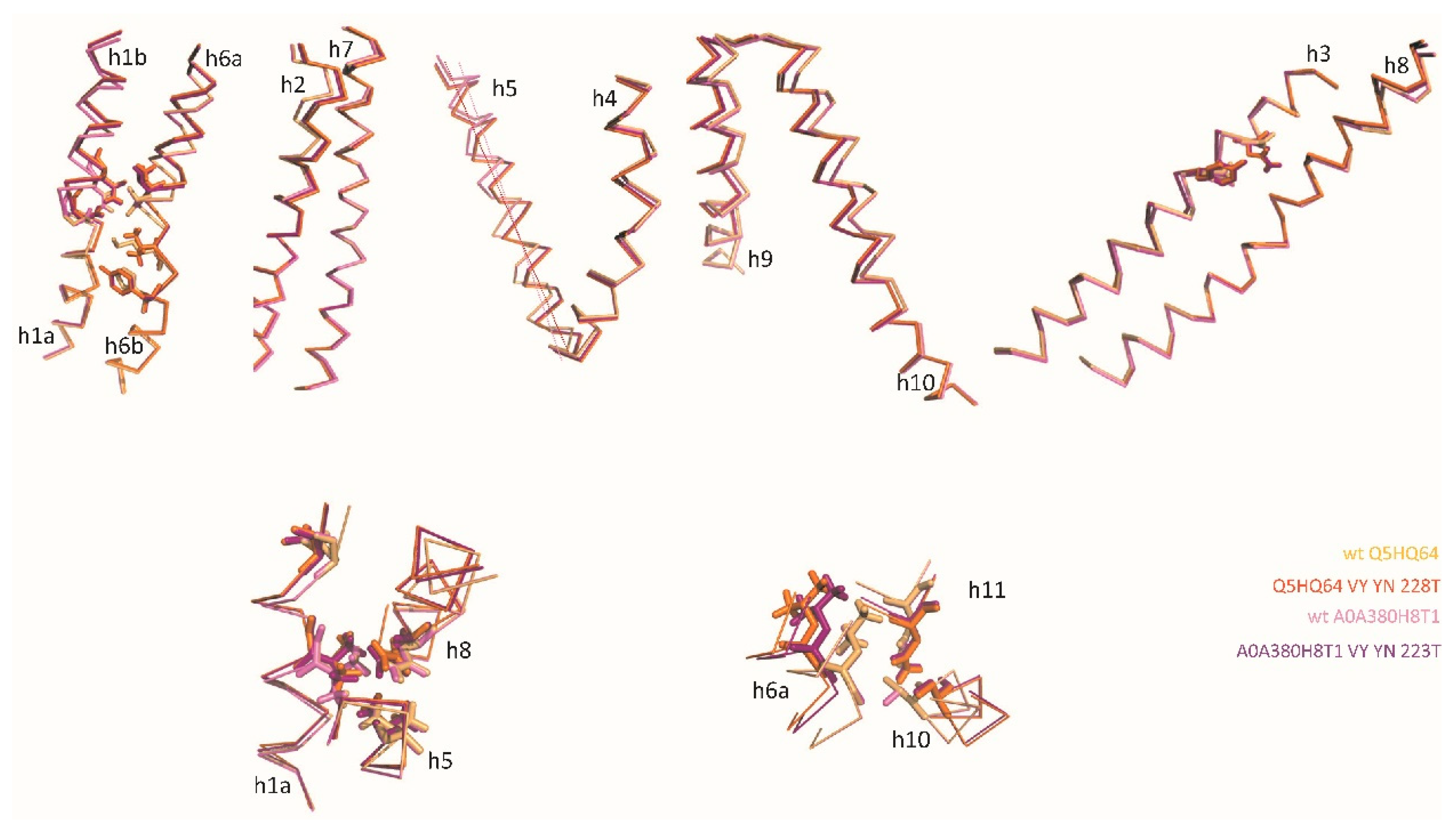
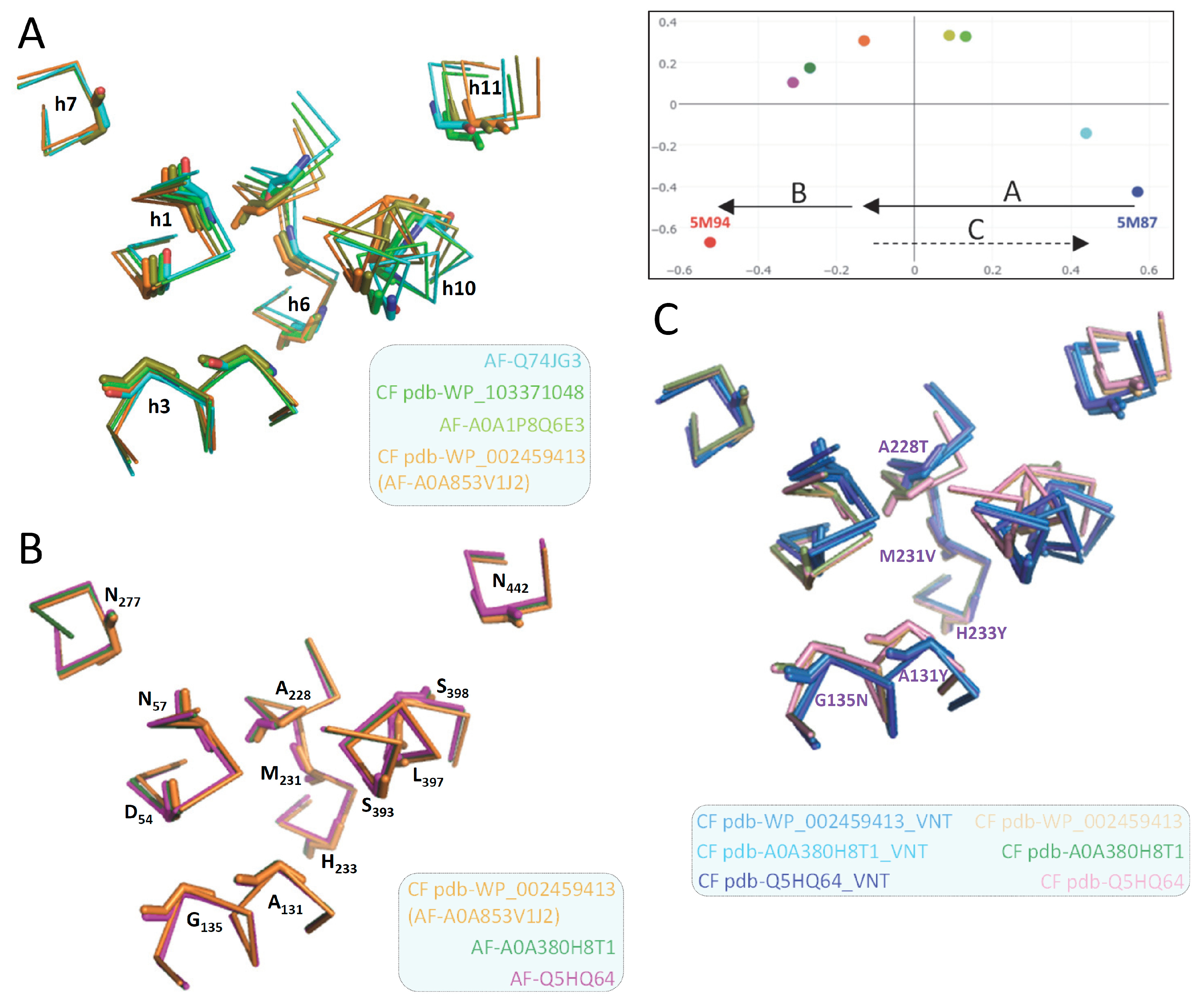
2.2. Site-directed mutagenesis demonstrates the functional significance of MCb Q5HQ64 CF modeling
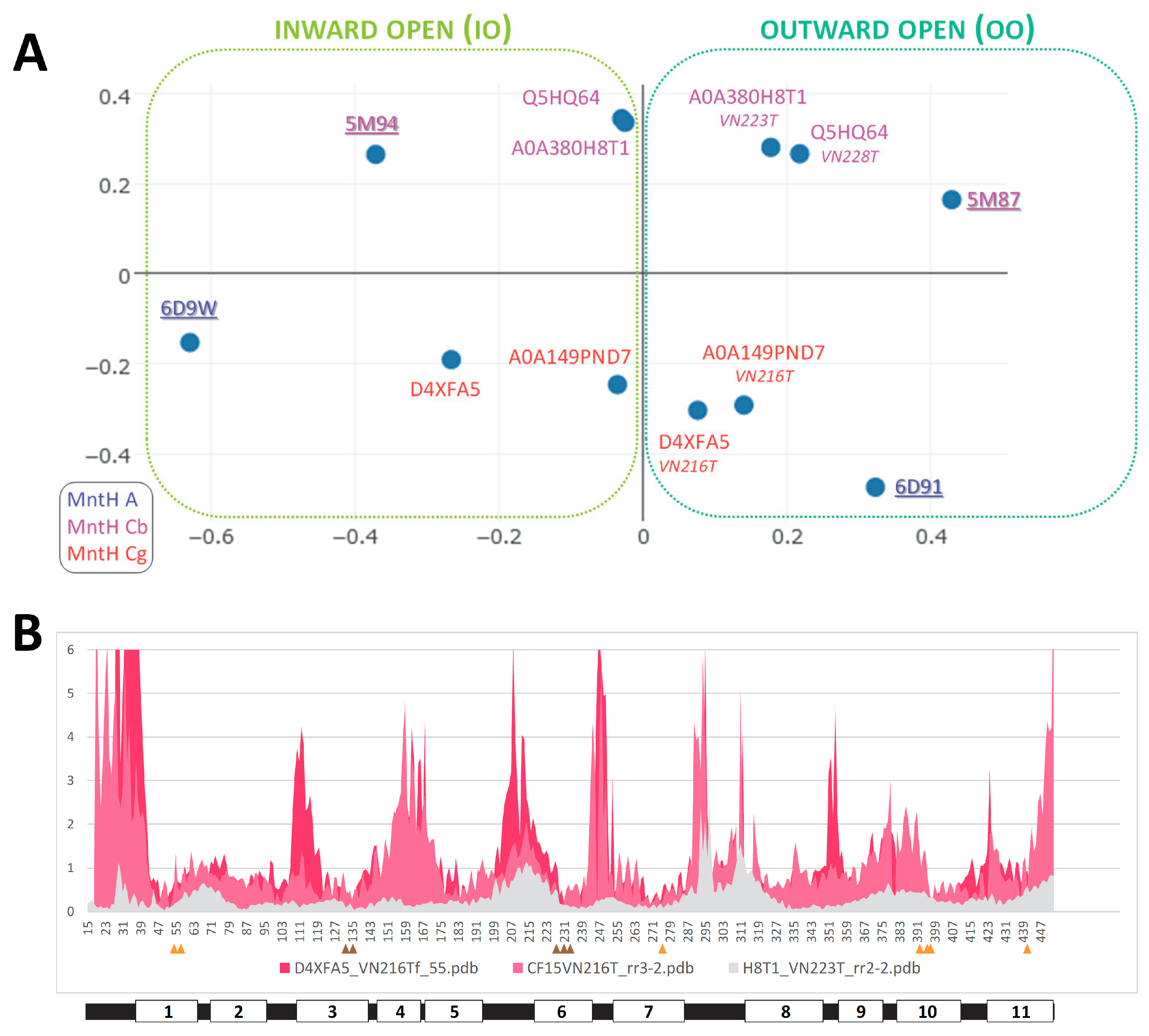
2.3. Central role of h3 in MCb Q5HQ64 in silico IO to OO conformation exchange
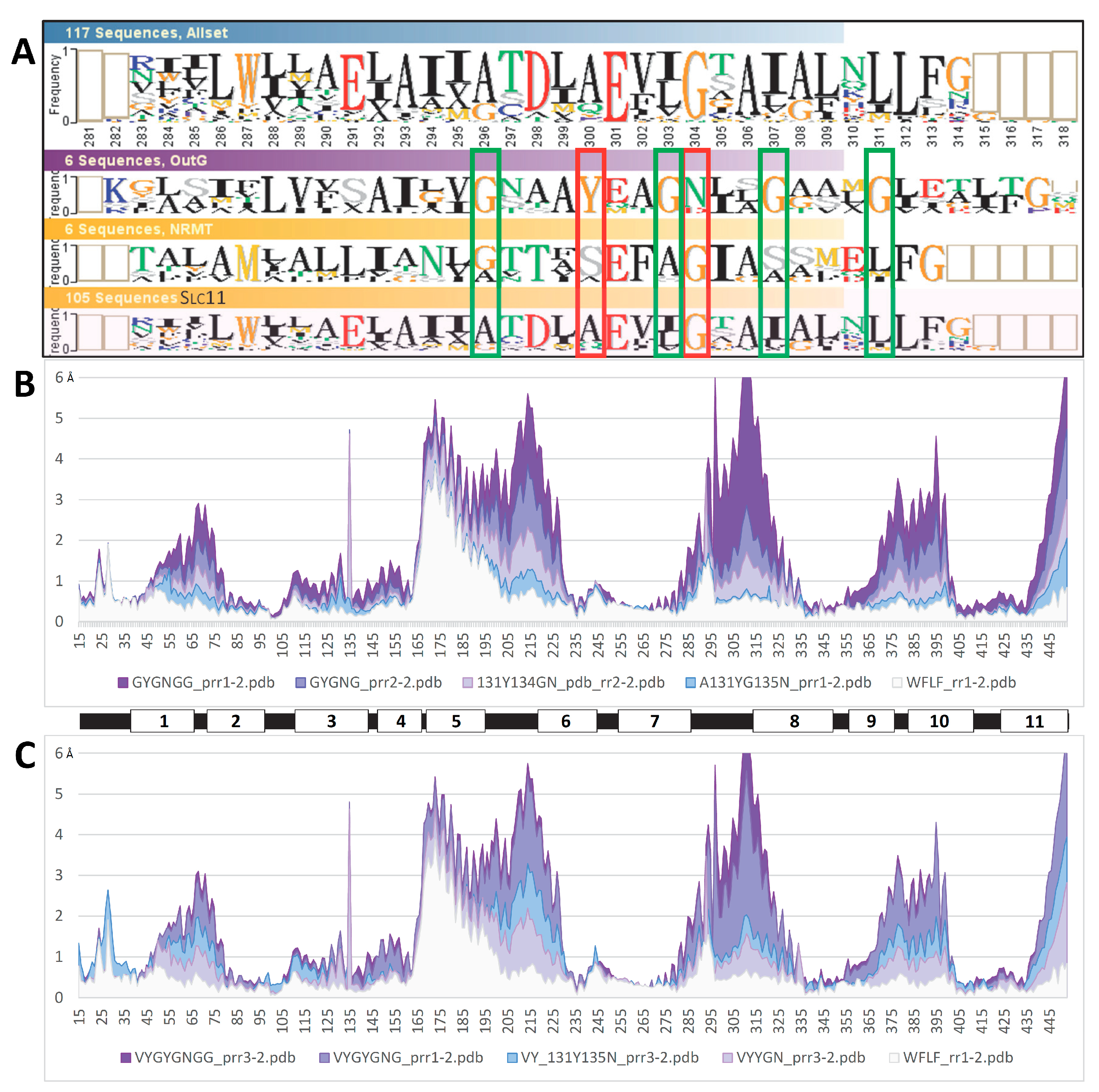
2.4. MCb 3D models are flexible and share alternate communities of networked residues
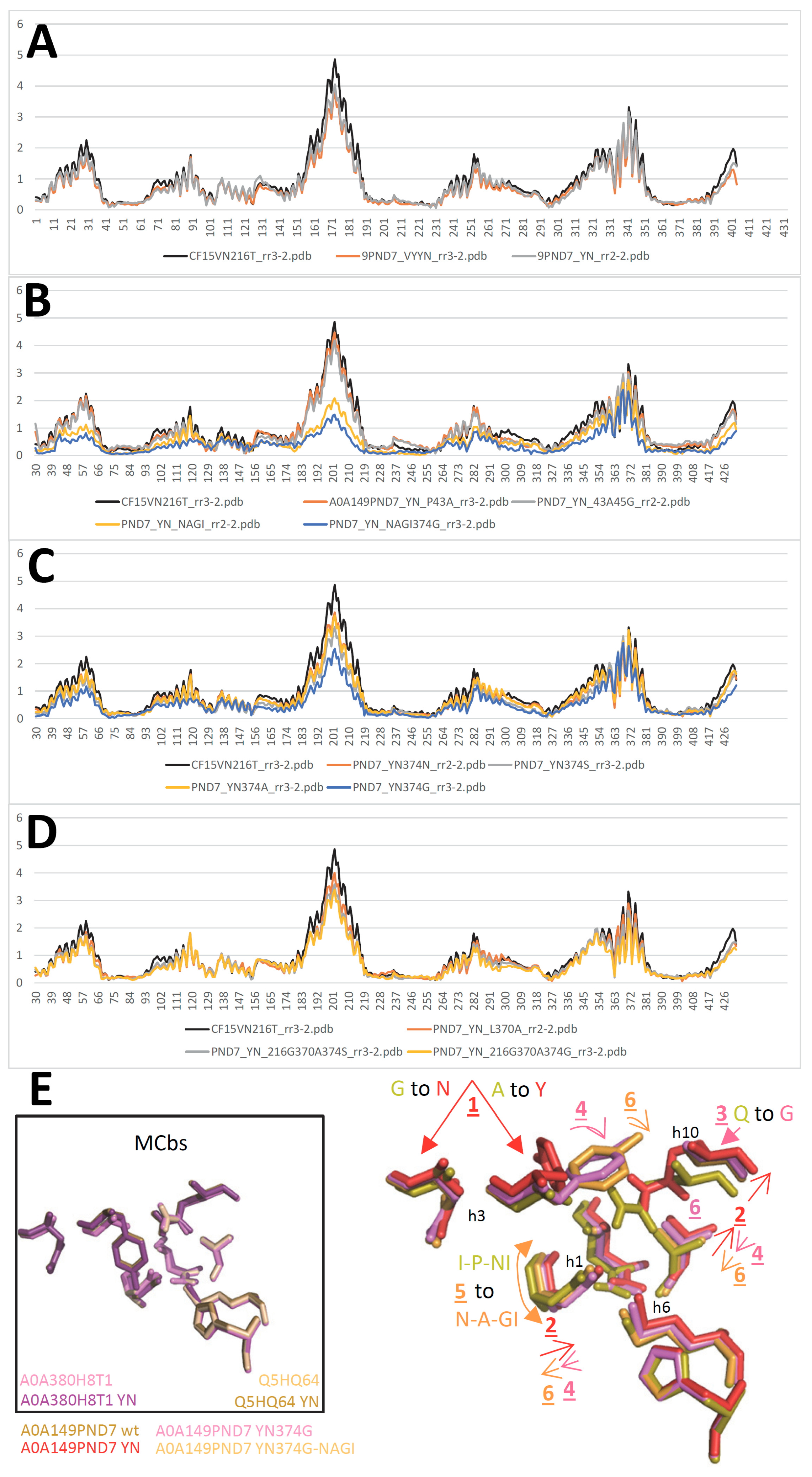
2.5. MCg1 carrier A0A149PND7 undergoes similar in silico conformation switch
2.6. Conserved switch mechanism in extensively divergent MCg1 from Bordetella-Achromobacter
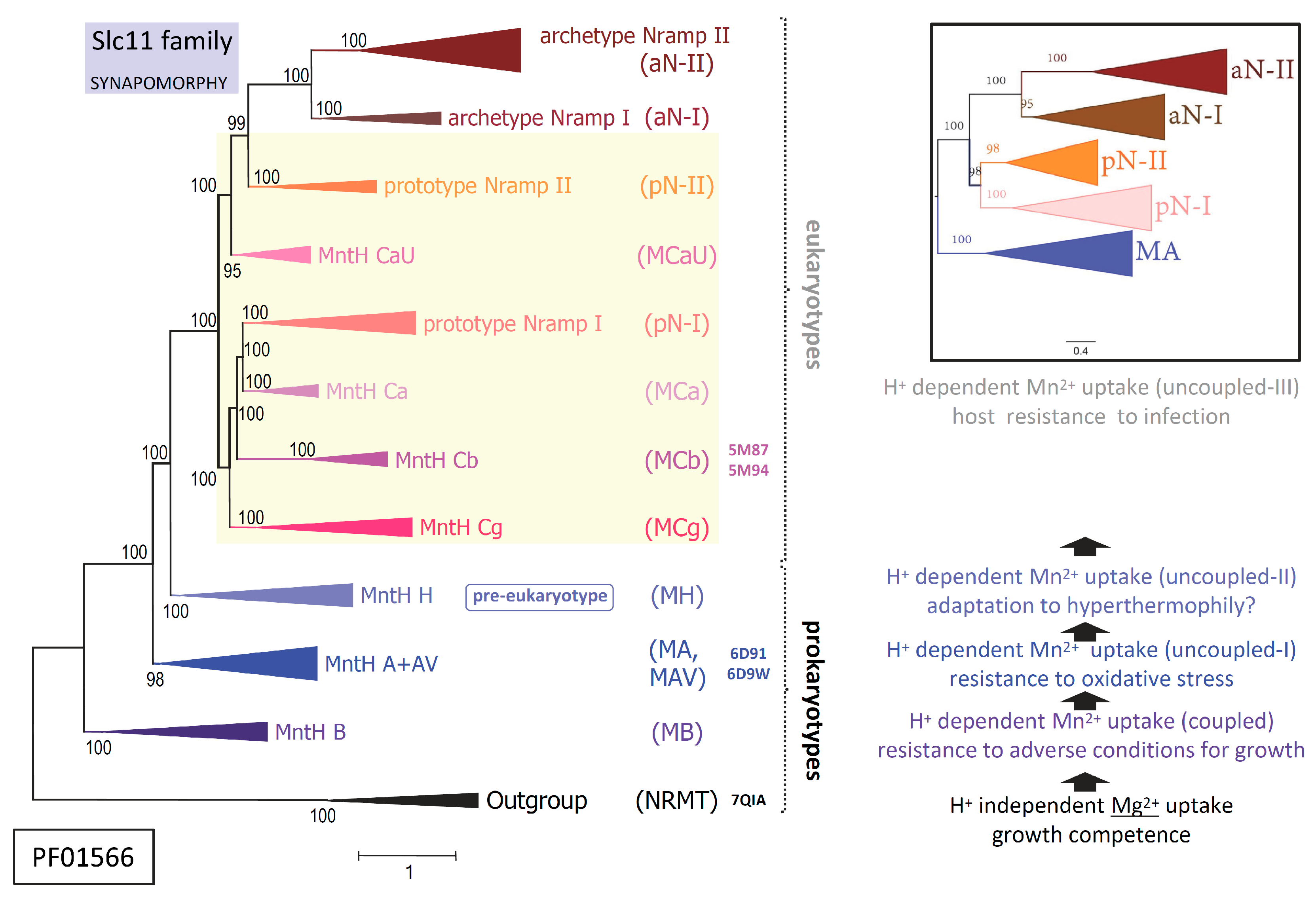
2.7. Slc11 synapomorphy: a conserved framework articulating carrier cycling in divergent 3D contexts
3. Discussion
4. Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Bozzi, A. T.; Gaudet, R. , Molecular Mechanism of Nramp-Family Transition Metal Transport. J Mol Biol 2021, 433, 166991. [Google Scholar] [CrossRef] [PubMed]
- Shawki, A.; Knight, P. B.; Maliken, B. D.; Niespodzany, E. J.; Mackenzie, B. , H(+)-coupled divalent metal-ion transporter-1: functional properties, physiological roles and therapeutics. Curr Top Membr 2012, 70, 169–214. [Google Scholar] [PubMed]
- Forrest, L. R.; Rudnick, G. , The rocking bundle: a mechanism for ion-coupled solute flux by symmetrical transporters. Physiology (Bethesda) 2009, 24, 377–86. [Google Scholar] [CrossRef]
- Yamashita, A.; Singh, S. K.; Kawate, T.; Jin, Y.; Gouaux, E. , Crystal structure of a bacterial homologue of Na+/Cl--dependent neurotransmitter transporters. Nature 2005, 437, 215–23. [Google Scholar] [CrossRef]
- Del Alamo, D.; Meiler, J.; McHaourab, H. S. , Principles of Alternating Access in LeuT-fold Transporters: Commonalities and Divergences. J Mol Biol 2022, 434, 167746. [Google Scholar] [CrossRef] [PubMed]
- Ehrnstorfer, I. A.; Geertsma, E. R.; Pardon, E.; Steyaert, J.; Dutzler, R. , Crystal structure of a SLC11 (NRAMP) transporter reveals the basis for transition-metal ion transport. Nat Struct Mol Biol 2014, 21, 990–6. [Google Scholar] [CrossRef]
- Ehrnstorfer, I. A.; Manatschal, C.; Arnold, F. M.; Laederach, J.; Dutzler, R. , Structural and mechanistic basis of proton-coupled metal ion transport in the SLC11/NRAMP family. Nat Commun 2017, 8, 14033. [Google Scholar] [CrossRef]
- Ramanadane, K.; Straub, M. S.; Dutzler, R.; Manatschal, C. , Structural and functional properties of a magnesium transporter of the SLC11/NRAMP family. Elife 2022, 11. [Google Scholar]
- Cellier, M. F. , Nramp: from sequence to structure and mechanism of divalent metal import. Curr Top Membr 2012, 69, 249–93. [Google Scholar]
- Romei, M.; Sapriel, G.; Imbert, P.; Jamay, T.; Chomilier, J.; Lecointre, G.; Carpentier, M. , Protein folds as synapomorphies of the tree of life. Evolution 2022, 76, 1706–1719. [Google Scholar] [CrossRef]
- Gupta, R. S. , Protein signatures (molecular synapomorphies) that are distinctive characteristics of the major cyanobacterial clades. Int J Syst Evol Microbiol 2009, 59 Pt 10, 2510–26. [Google Scholar] [CrossRef]
- Cellier, M. F. M. , Nramp: Deprive and conquer? Front Cell Dev Biol 2022, 10, 988866. [Google Scholar] [CrossRef] [PubMed]
- Raval, P. K.; Garg, S. G.; Gould, S. B. , Endosymbiotic selective pressure at the origin of eukaryotic cell biology. Elife 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Cellier, M. F.; Courville, P.; Campion, C. , Nramp1 phagocyte intracellular metal withdrawal defense. Microbes Infect 2007, 9, 1662-70. [Google Scholar] [CrossRef]
- Antelo, G. T.; Vila, A. J.; Giedroc, D. P.; Capdevila, D. A. , Molecular Evolution of Transition Metal Bioavailability at the Host-Pathogen Interface. Trends Microbiol 2021, 29, 441–457. [Google Scholar] [CrossRef]
- Morey, J. R.; McDevitt, C. A.; Kehl-Fie, T. E. , Host-imposed manganese starvation of invading pathogens: two routes to the same destination. Biometals 2015, 28, 509–19. [Google Scholar] [CrossRef] [PubMed]
- Čapek, J.; Večerek, B. , Why is manganese so valuable to bacterial pathogens? Front Cell Infect Microbiol 2023, 13, 943390. [Google Scholar] [CrossRef]
- Nelson, N.; Sacher, A.; Nelson, H. , The significance of molecular slips in transport systems. Nat Rev Mol Cell Biol 2002, 3, 876–81. [Google Scholar] [CrossRef]
- Beckstein, O.; Naughton, F. , General principles of secondary active transporter function. Biophys Rev (Melville) 2022, 3, 011307. [Google Scholar] [CrossRef]
- Banerjee, A.; Saha, S.; Tvedt, N. C.; Yang, L. W.; Bahar, I. , Mutually beneficial confluence of structure-based modeling of protein dynamics and machine learning methods. Curr Opin Struct Biol 2023, 78, 102517. [Google Scholar] [CrossRef]
- Bridel, S.; Bouchez, V.; Brancotte, B.; Hauck, S.; Armatys, N.; Landier, A.; Mühle, E.; Guillot, S.; Toubiana, J.; Maiden, M. C. J.; Jolley, K. A.; Brisse, S. , A comprehensive resource for Bordetella genomic epidemiology and biodiversity studies. Nat Commun 2022, 13, 3807. [Google Scholar] [CrossRef] [PubMed]
- Dumolin, C.; Peeters, C.; Ehsani, E.; Tahon, G.; De Canck, E.; Cnockaert, M.; Boon, N.; Vandamme, P. , Achromobacter veterisilvae sp. nov., from a mixed hydrogen-oxidizing bacteria enrichment reactor for microbial protein production. Int J Syst Evol Microbiol 2020, 70, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Kim, S. C.; Chung, S. O.; Lee, H. J. , Achromobacter aestuarii sp. nov., Isolated from an Estuary. Curr Microbiol 2021, 78, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Kumaraswami, M. , Managing Manganese: The Role of Manganese Homeostasis in Streptococcal Pathogenesis. Front Cell Dev Biol 2022, 10, 921920. [Google Scholar] [CrossRef]
- Gabrielaite, M.; Nielsen, F. C.; Johansen, H. K.; Marvig, R. L. , Achromobacter spp. genetic adaptation in cystic fibrosis. Microb Genom 2021, 7. [Google Scholar] [CrossRef]
- Jolley, K. A.; Bray, J. E.; Maiden, M. C. J. , Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res 2018, 3, 124. [Google Scholar] [CrossRef]
- Miton, C. M.; Buda, K.; Tokuriki, N. , Epistasis and intramolecular networks in protein evolution. Curr Opin Struct Biol 2021, 69, 160–168. [Google Scholar] [CrossRef]
- Siedler, S.; Rau, M. H.; Bidstrup, S.; Vento, J. M.; Aunsbjerg, S. D.; Bosma, E. F.; McNair, L. M.; Beisel, C. L.; Neves, A. R. , Competitive Exclusion Is a Major Bioprotective Mechanism of Lactobacilli against Fungal Spoilage in Fermented Milk Products. Appl Environ Microbiol 2020, 86. [Google Scholar] [CrossRef]
- van Mastrigt, O.; Di Stefano, E.; Hartono, S.; Abee, T.; Smid, E. J. , Large plasmidome of dairy Lactococcus lactis subsp. lactis biovar diacetylactis FM03P encodes technological functions and appears highly unstable. BMC Genomics 2018, 19, 620. [Google Scholar] [CrossRef]
- Juritz, E.; Palopoli, N.; Fornasari, M. S.; Fernandez-Alberti, S.; Parisi, G. , Protein conformational diversity modulates sequence divergence. Mol Biol Evol 2013, 30, 79–87. [Google Scholar] [CrossRef]
- Ilgü, H.; Jeckelmann, J. M.; Kalbermatter, D.; Ucurum, Z.; Lemmin, T.; Fotiadis, D. , High-resolution structure of the amino acid transporter AdiC reveals insights into the role of water molecules and networks in oligomerization and substrate binding. BMC Biol 2021, 19, 179. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Berry, S. P.; Wilson, E. A.; Zhang, C. H.; Shekhar, M.; Singharoy, A.; Gaudet, R. , High-resolution structures with bound Mn(2+) and Cd(2+) map the metal import pathway in an Nramp transporter. Elife 2023, 12. [Google Scholar]
- Serrano, F. A.; Yukl, E. T. , Contributions of Conformational Flexibility to High-Affinity Zinc Binding in the Solute Binding Protein AztC. ACS Omega 2022, 7, 3768–3774. [Google Scholar] [CrossRef] [PubMed]
- Martin, J. E.; Waters, L. S. , Regulation of Bacterial Manganese Homeostasis and Usage During Stress Responses and Pathogenesis. Front Mol Biosci 2022, 9, 945724. [Google Scholar] [CrossRef] [PubMed]
- Kajfasz, J. K.; Katrak, C.; Ganguly, T.; Vargas, J.; Wright, L.; Peters, Z. T.; Spatafora, G. A.; Abranches, J.; Lemos, J. A. , Manganese Uptake, Mediated by SloABC and MntH, Is Essential for the Fitness of Streptococcus mutans. mSphere 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Shabayek, S.; Bauer, R.; Mauerer, S.; Mizaikoff, B.; Spellerberg, B. , A streptococcal NRAMP homologue is crucial for the survival of Streptococcus agalactiae under low pH conditions. Mol Microbiol 2016, 100, 589–606. [Google Scholar] [CrossRef]
- Colomer-Winter, C.; Flores-Mireles, A. L.; Baker, S. P.; Frank, K. L.; Lynch, A. J. L.; Hultgren, S. J.; Kitten, T.; Lemos, J. A. , Manganese acquisition is essential for virulence of Enterococcus faecalis. PLoS Pathog 2018, 14, e1007102. [Google Scholar] [CrossRef]
- Kehl-Fie, T. E.; Zhang, Y.; Moore, J. L.; Farrand, A. J.; Hood, M. I.; Rathi, S.; Chazin, W. J.; Caprioli, R. M.; Skaar, E. P. , MntABC and MntH contribute to systemic Staphylococcus aureus infection by competing with calprotectin for nutrient manganese. Infect Immun 2013, 81, 3395–405. [Google Scholar] [CrossRef]
- Currie, B. J. , Melioidosis and Burkholderia pseudomallei : progress in epidemiology, diagnosis, treatment and vaccination. Curr Opin Infect Dis 2022, 35, 517–523. [Google Scholar] [CrossRef]
- Tavares, M.; Kozak, M.; Balola, A.; Sá-Correia, I. , Burkholderia cepacia Complex Bacteria: a Feared Contamination Risk in Water-Based Pharmaceutical Products. Clin Microbiol Rev 2020, 33. [Google Scholar] [CrossRef]
- Li, X.; Hu, Y.; Gong, J.; Zhang, L.; Wang, G. , Comparative genome characterization of Achromobacter members reveals potential genetic determinants facilitating the adaptation to a pathogenic lifestyle. Appl Microbiol Biotechnol 2013, 97, 6413–25. [Google Scholar] [CrossRef]
- Bonis, B. M.; Hunter, R. C. , JMM Profile: Achromobacter xylosoxidans: the cloak-and-dagger opportunist. J Med Microbiol 2022, 71. [Google Scholar] [CrossRef]
- Menetrey, Q.; Aujoulat, F.; Chiron, R.; Jumas-Bilak, E.; Marchandin, H.; Dupont, C. , A new perspective on opportunistic pathogens of the genus Bordetella in cystic fibrosis. J Cyst Fibros 2022, 21, 344–347. [Google Scholar] [CrossRef]
- Storz, J. F. , Compensatory mutations and epistasis for protein function. Curr Opin Struct Biol 2018, 50, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Metzger, B. P. H.; Thornton, J. W. , Epistatic drift causes gradual decay of predictability in protein evolution. Science 2022, 376, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Ghenu, A. H.; Amado, A.; Gordo, I.; Bank, C. , Epistasis decreases with increasing antibiotic pressure but not temperature. Philos Trans R Soc Lond B Biol Sci 2023, 378, 20220058. [Google Scholar] [CrossRef] [PubMed]
- Buda, K.; Miton, C. M.; Fan, X. C.; Tokuriki, N. , Molecular determinants of protein evolvability. Trends Biochem Sci 2023. [Google Scholar] [CrossRef] [PubMed]
- Echave, J. , Evolutionary coupling range varies widely among enzymes depending on selection pressure. Biophys J 2021, 120, 4320–4324. [Google Scholar] [CrossRef]
- Starr, T. N.; Picton, L. K.; Thornton, J. W. , Alternative evolutionary histories in the sequence space of an ancient protein. Nature 2017, 549, 409–413. [Google Scholar] [CrossRef]
- Holm, L.; Laiho, A.; Törönen, P.; Salgado, M. , DALI shines a light on remote homologs: One hundred discoveries. Protein Sci 2023, 32, e4519. [Google Scholar] [CrossRef]
- Bozzi, A. T.; Zimanyi, C. M.; Nicoludis, J. M.; Lee, B. K.; Zhang, C. H.; Gaudet, R. , Structures in multiple conformations reveal distinct transition metal and proton pathways in an Nramp transporter. Elife 2019, 8. [Google Scholar]
- Mirdita, M.; Schütze, K.; Moriwaki, Y.; Heo, L.; Ovchinnikov, S.; Steinegger, M. , ColabFold: making protein folding accessible to all. Nat Methods 2022, 19, 679–682. [Google Scholar] [CrossRef] [PubMed]
- Pasquadibisceglie, A.; Leccese, A.; Polticelli, F. , A computational study of the structure and function of human Zrt and Irt-like proteins metal transporters: An elevator-type transport mechanism predicted by AlphaFold2. Front Chem 2022, 10, 1004815. [Google Scholar] [CrossRef] [PubMed]
- Drew, E. D.; Janes, R. W. , 2StrucCompare: a webserver for visualizing small but noteworthy differences between protein tertiary structures through interrogation of the secondary structure content. Nucleic Acids Res 2019, (W1), W477–w481. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Natarajan, P.; Ye, Y.; Hrabe, T.; Godzik, A. , POSA: a user-driven, interactive multiple protein structure alignment server. Nucleic Acids Res 2014, (Web Server issue), W240–5. [Google Scholar] [CrossRef]
- Felline, A.; Seeber, M.; Fanelli, F. , PSNtools for standalone and web-based structure network analyses of conformational ensembles. Comput Struct Biotechnol J 2022, 20, 640–649. [Google Scholar] [CrossRef]
- Gu, X.; Zou, Y.; Su, Z.; Huang, W.; Zhou, Z.; Arendsee, Z.; Zeng, Y. , An update of DIVERGE software for functional divergence analysis of protein family. Mol Biol Evol 2013, 30, 1713–9. [Google Scholar] [CrossRef]
- Brandt, B. W.; Feenstra, K. A.; Heringa, J. , Multi-Harmony: detecting functional specificity from sequence alignment. Nucleic Acids Res 2010, (Web Server issue), W35–40. [Google Scholar] [CrossRef]
- Shih, A. C.; Lee, D. T.; Peng, C. L.; Wu, Y. W. , Phylo-mLogo: an interactive and hierarchical multiple-logo visualization tool for alignment of many sequences. BMC Bioinformatics 2007, 8, 63. [Google Scholar] [CrossRef]
- Zhang, Z.; Schäffer, A. A.; Miller, W.; Madden, T. L.; Lipman, D. J.; Koonin, E. V.; Altschul, S. F. , Protein sequence similarity searches using patterns as seeds. Nucleic Acids Res 1998, 26, 3986–90. [Google Scholar] [CrossRef]
- Larkin, M. A.; Blackshields, G.; Brown, N. P.; Chenna, R.; McGettigan, P. A.; McWilliam, H.; Valentin, F.; Wallace, I. M.; Wilm, A.; Lopez, R.; Thompson, J. D.; Gibson, T. J.; Higgins, D. G. , Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–8. [Google Scholar] [CrossRef]
- Zimmermann, L.; Stephens, A.; Nam, S. Z.; Rau, D.; Kübler, J.; Lozajic, M.; Gabler, F.; Söding, J.; Lupas, A. N.; Alva, V. , A Completely Reimplemented MPI Bioinformatics Toolkit with a New HHpred Server at its Core. J Mol Biol 2018, 430, 2237–2243. [Google Scholar] [CrossRef] [PubMed]
- Trifinopoulos, J.; Nguyen, L. T.; von Haeseler, A.; Minh, B. Q. , W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 2016, (W1), W232–5. [Google Scholar] [CrossRef] [PubMed]
- Le, S. Q.; Lartillot, N.; Gascuel, O. , Phylogenetic mixture models for proteins. Philos Trans R Soc Lond B Biol Sci 2008, 363, 3965–76. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



