Submitted:
17 August 2023
Posted:
18 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Enzymatic Electrochemical Biosensors for Food Bioprocess Monitoring
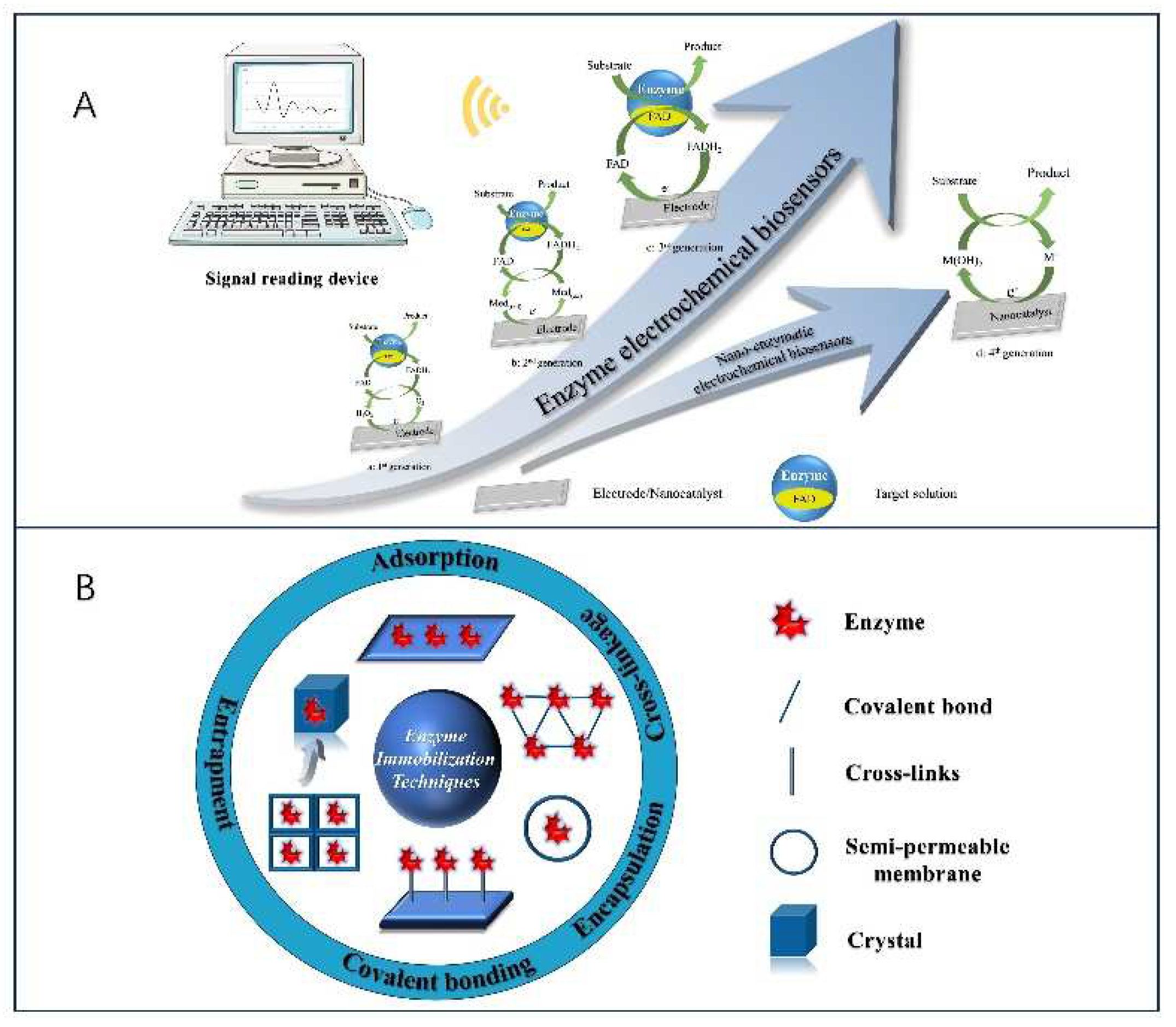
2.1 Principle of Enzyme Electrochemical Biosensor Construction
2.2. Electrochemical Biosensors for Single-Enzyme Systems
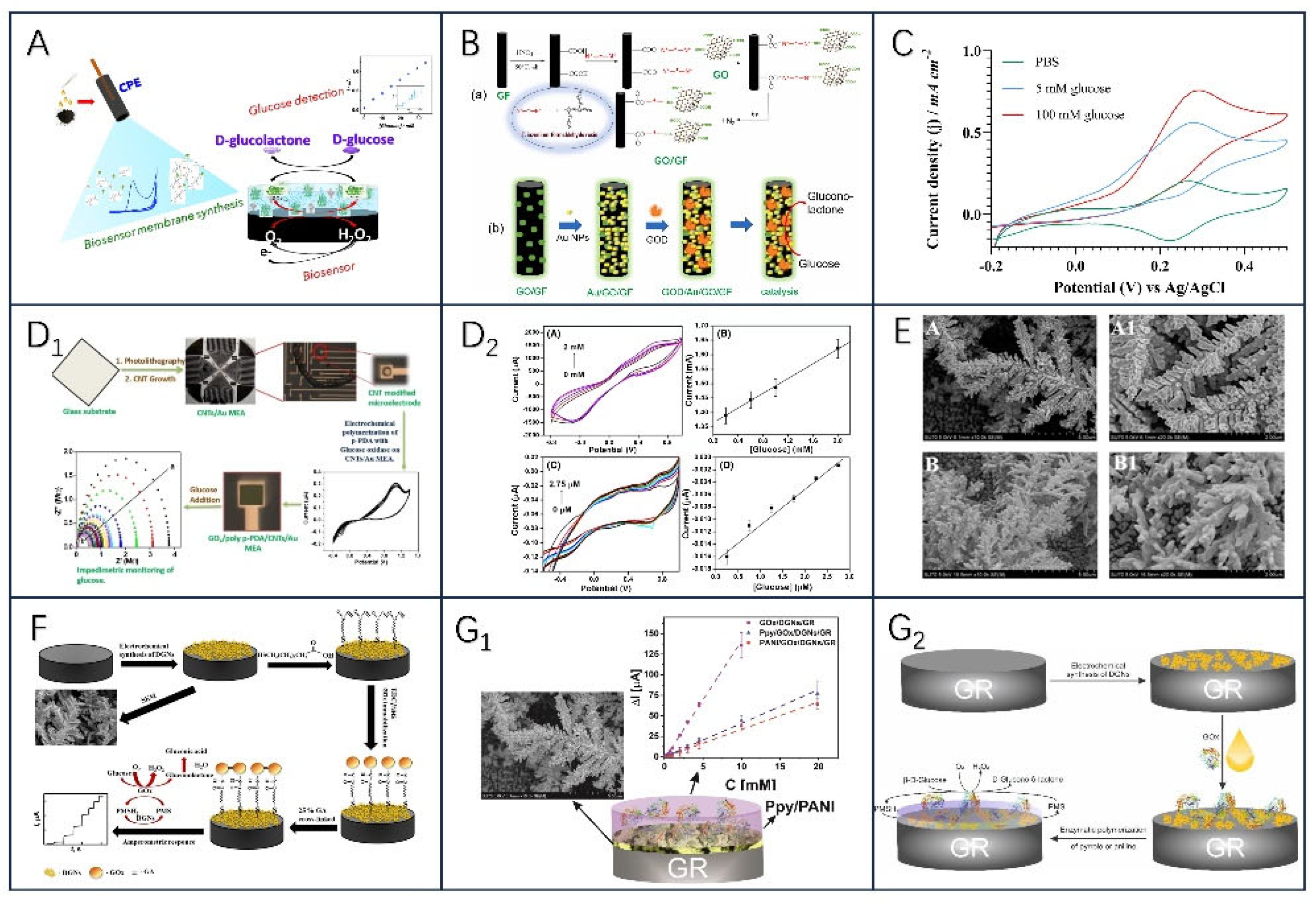
2.2.1. Glucose Oxidase
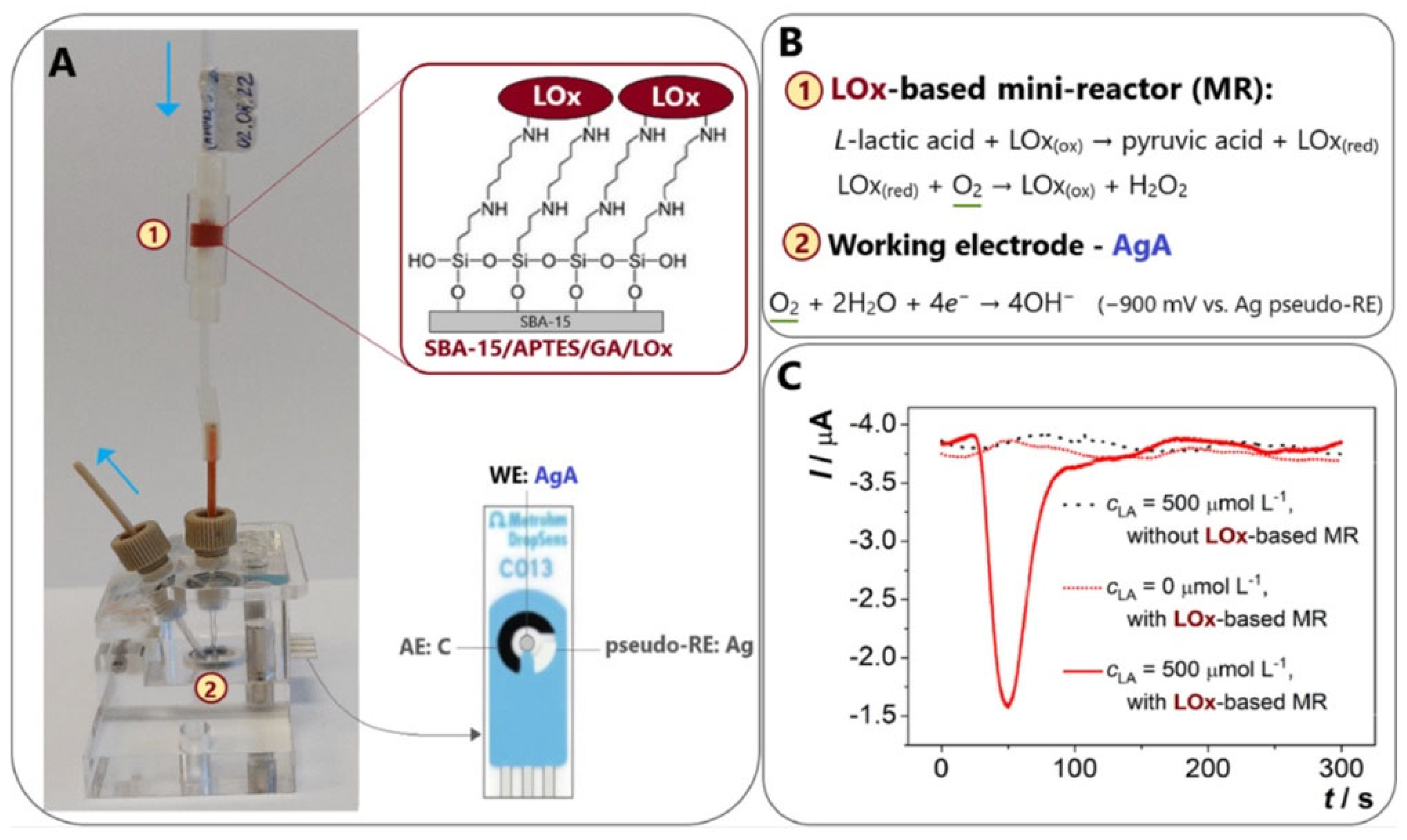
2.2.2. Lactate Oxidase and Lactate Dehydrogenase
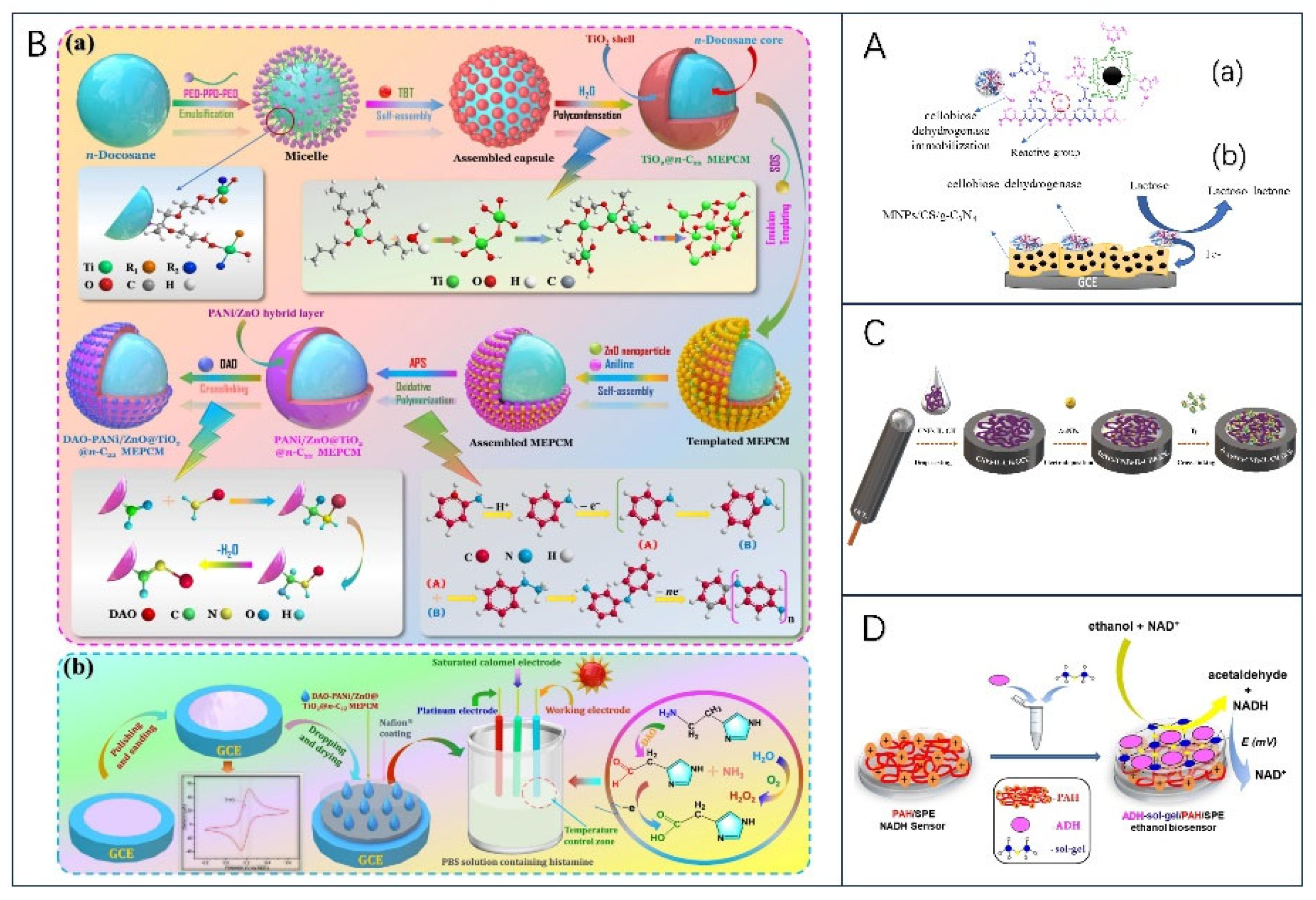
2.2.3. Other enzymes for the Development of Electrochemical Biosensors
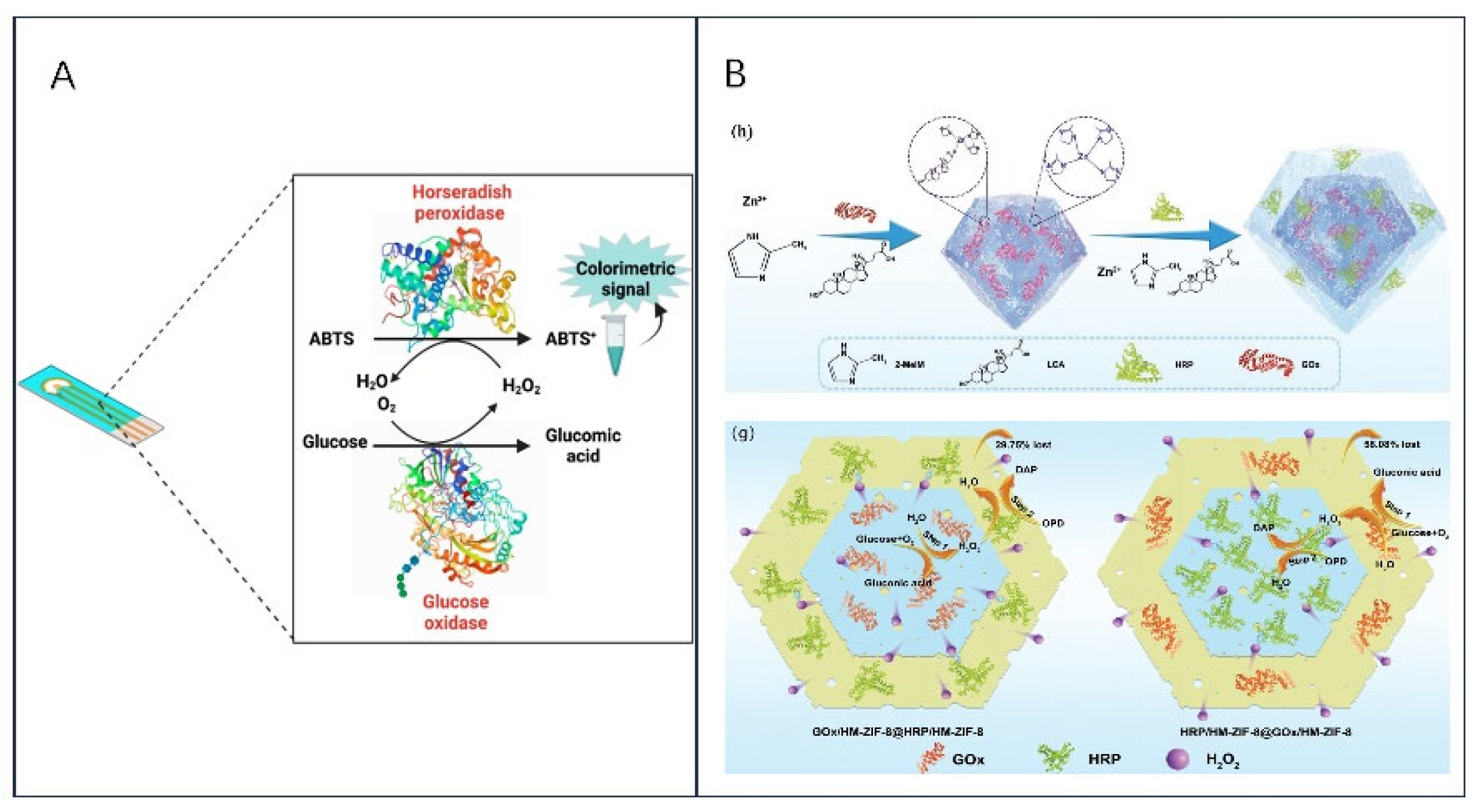
2.3. Electrochemical Biosensors for Multi-Enzyme Systems
2.4. Electrochemical Biosensors for Nano-Enzymatic Systems
3. Nanomaterials for Enzyme Immobilization
3.1. Metal-Based Nanomaterials Modified Electrodes
3.2. Graphene Nanomaterials Modified Electrodes
3.3. Metal-Organic Framework Modified Electrodes
3.4. Carbon Nanotube-Modified Electrodes
3.5. Polymer Modified Electrodes
4. Challenges and Future Trends of Enzyme Electrochemical Biosensors
4.1. Challenges
- (1)
- The major hindrances to the widespread usage of enzyme electrochemical biosensors are still the reusability and stability of these biosensors. Moreover, the complexity of food matrices, harsh environments, and their interference with biorecognition elements can significantly impact the reproducibility and selectivity of biosensors. Henceforth, scientists must prioritize the enhancement of sensor efficacy in forthcoming research endeavors. Specifically, rigorous investigation is necessary to address and resolve the issue of interferences encountered in authentic specimens, ensure the endurance of enzyme-chemical biosensors in adverse surroundings, and assess the impact of varying storage conditions on the biosensors' lifespan [86].
- (2)
- The addition of multiple enzymes to a biosensor in multi-enzyme systems can create complications during biosensor fabrication. Furthermore, it can impose substantial limitations on the characterization and application possibilities of the biosensor. This arises due to variations in the sensitivity to substrates, effectiveness in storage, and conditions required for enzyme immobilization among different enzymes. Hence, a critical consideration in designing a multi-enzyme biosensor is the meticulous selection of enzyme systems. This selection aims to prevent their sensitization to substances other than the target substance and ensure the requisite stability of the biosensor.
- (3)
- Compared with natural enzymes, the catalytic activity of nano enzymes is still relatively low, and most nano enzymes are difficult to catalyze a specific substrate like biological enzymes. Therefore, despite all the advantages of nano enzymes, nano enzymes with high catalytic activity, excellent selectivity, and specificity for constructing nano enzymes-based biosensors still need to be further developed. In the future, integrating biological enzymes or nano enzymes into mesoporous nanomaterials to prepare integrated nano enzymes (INAzymes), or constructing a binding or synergistic mechanism between an enzyme and a nano-enzyme may be a promising strategy to obtain this type of problem.
- (4)
- Achieving high homogeneity, reproducibility and chemical stability in electrode materials is a challenging task that cannot be accomplished by simple synthesis alone. Obtaining these desirable properties requires continuous efforts to advance advanced synthetic methods and their application to the analysis of real samples. Therefore, future prospective studies could prioritize the assessment of the stability of biosensor electrode materials in complex environments. In addition, it would be beneficial to explore more reliable modification strategies to enhance compatibility between biorecognition molecules and electrodes, as well as other potential avenues of exploration.
4.2. Future Development Trends
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clark, L.C.; Lyons, C. ELECTRODE SYSTEMS FOR CONTINUOUS MONITORING IN CARDIOVASCULAR SURGERY. Ann Ny Acad Sci 1962, 102, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Mishra, G.; Barfidokht, A.; Tehrani, F.; Mishra, R. Food Safety Analysis Using Electrochemical Biosensors. Foods 2018, 7, 141. [Google Scholar] [CrossRef] [PubMed]
- Majdinasab, M.; Mishra, R.K.; Tang, X.; Marty, J.L. Detection of Antibiotics in Food: New Achievements in the Development of Biosensors. TrAC Trends in Analytical Chemistry 2020, 127, 115883. [Google Scholar] [CrossRef]
- Ding, J.; Qin, W. Recent Advances in Potentiometric Biosensors. TrAC Trends in Analytical Chemistry 2020, 124, 115803. [Google Scholar] [CrossRef]
- Bankole, O.E.; Verma, D.K.; Chávez González, M.L.; Ceferino, J.G.; Sandoval-Cortés, J.; Aguilar, C.N. Recent Trends and Technical Advancements in Biosensors and Their Emerging Applications in Food and Bioscience. Food Biosci 2022, 47, 101695. [Google Scholar] [CrossRef]
- Purohit, B.; Vernekar, P.R.; Shetti, N.P.; Chandra, P. Biosensor Nanoengineering: Design, Operation, and Implementation for Biomolecular Analysis. Sensors International 2020, 1, 100040. [Google Scholar] [CrossRef]
- Yao, B.; Yao, J.; Fan, Z.; Zhao, J.; Zhang, K.; Huang, W. Rapid Advances of Versatile MXenes for Electrochemical Enzyme-Based Biosensors, Immunosensors, and Nucleic Acid-Based Biosensors. ChemElectroChem 2022, 9. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, L.; Cui, Y. Transdermal Amperometric Biosensors for Continuous Glucose Monitoring in Diabetes. Talanta 2023, 253, 124033. [Google Scholar] [CrossRef]
- Pérez-González, C.; Salvo-Comino, C.; Martín-Pedrosa, F.; García-Cabezón, C.; Rodríguez-Méndez, M.L. Bioelectronic Tongue Dedicated to the Analysis of Milk Using Enzymes Linked to Carboxylated-PVC Membranes Modified with Gold Nanoparticles. Food Control 2023, 145, 109425. [Google Scholar] [CrossRef]
- Hooda, V.; Kumar, V.; Gahlaut, A.; Hooda, V. Alcohol Quantification: Recent Insights into Amperometric Enzyme Biosensors. Artif Cell Nanomed B 2018, 46, 398–410. [Google Scholar] [CrossRef]
- Nguyen, H.H.; Lee, S.H.; Lee, U.J.; Fermin, C.D.; Kim, M. Immobilized Enzymes in Biosensor Applications. Materials 2019, 12, 121. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Soldatkin, O.O.; Dzyadevych, S.V.; Soldatkin, A.P. Electrochemical Biosensors Based on Multienzyme Systems: Main Groups, Advantages and Limitations – A Review. Anal Chim Acta 2020, 1111, 114–131. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, X.; Wang, Q.; Lou, Z.; Li, S.; Zhu, Y.; Qin, L.; Wei, H. Nanomaterials with Enzyme-like Characteristics (Nanozymes): Next-Generation Artificial Enzymes (II). Chem Soc Rev 2019, 48, 1004–1076. [Google Scholar] [CrossRef] [PubMed]
- Kurbanoglu, S.; Erkmen, C.; Uslu, B. Frontiers in Electrochemical Enzyme Based Biosensors for Food and Drug Analysis. TrAC Trends in Analytical Chemistry 2020, 124, 115809. [Google Scholar] [CrossRef]
- Witkowska Nery, E.; Kundys, M.; Jeleń, P.S.; Jönsson-Niedziółka, M. Electrochemical Glucose Sensing: Is There Still Room for Improvement? Anal Chem 2016, 88, 11271–11282. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Biswas, R.; Banerjee, R.; Biswas, P. A Gold Nanoparticle-Intercalated Mesoporous Silica-Based Nanozyme for the Selective Colorimetric Detection of Dopamine. Nanoscale Adv. 2020, 2, 734–745. [Google Scholar] [CrossRef]
- Mohammadi, Z.B.; Zhang, F.; Kharazmi, M.S.; Jafari, S.M. Nano-Biocatalysts for Food Applications; Immobilized Enzymes within Different Nanostructures. Crit Rev Food Sci 2022, 1–19. [Google Scholar] [CrossRef]
- Gupta, R.; Raza, N.; Bhardwaj, S.K.; Vikrant, K.; Kim, K.-H.; Bhardwaj, N. Advances in Nanomaterial-Based Electrochemical Biosensors for the Detection of Microbial Toxins, Pathogenic Bacteria in Food Matrices. Journal of Hazardous Materials 2021, 401, 123379. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Soldatkin, O.O.; Kucherenko, D.Yu.; Soldatkina, O.V.; Dzyadevych, S.V. Advances in Nanomaterial Application in Enzyme-Based Electrochemical Biosensors: A Review. Nanoscale Adv 2019, 1, 4560–4577. [Google Scholar] [CrossRef]
- Mohd Razib, M.S.; Latip, W.; Abdul Rashid, J.I.; Knight, V.F.; Wan Yunus, W.M.Z.; Ong, K.K.; Mohd Kasim, N.A.; Mohd Noor, S.A. An Enzyme-Based Biosensor for the Detection of Organophosphate Compounds Using Mutant Phosphotriesterase Immobilized onto Reduced Graphene Oxide. Journal of Chemistry 2021, 2021, 1–11. [Google Scholar] [CrossRef]
- Yoo, E.-H.; Lee, S.-Y. Glucose Biosensors: An Overview of Use in Clinical Practice. Sensors 2010, 10, 4558–4576. [Google Scholar] [CrossRef] [PubMed]
- Bruen, D.; Delaney, C.; Florea, L.; Diamond, D. Glucose Sensing for Diabetes Monitoring: Recent Developments. Sensors 2017, 17, 1866. [Google Scholar] [CrossRef]
- Vaddiraju, S.; Burgess, D.J.; Tomazos, I.; Jain, F.C.; Papadimitrakopoulos, F. Technologies for Continuous Glucose Monitoring: Current Problems and Future Promises. J Diabetes Sci Technol 2010, 4, 1540–1562. [Google Scholar] [CrossRef]
- Wang, J.; Lu, F. Oxygen-Rich Oxidase Enzyme Electrodes for Operation in Oxygen-Free Solutions. J. Am. Chem. Soc. 1998, 120, 1048–1050. [Google Scholar] [CrossRef]
- Teymourian, H.; Barfidokht, A.; Wang, J. Electrochemical Glucose Sensors in Diabetes Management: An Updated Review (2010–2020). Chem. Soc. Rev. 2020, 49, 7671–7709. [Google Scholar] [CrossRef] [PubMed]
- Çevik, E.; Şenel, M.; Fatih Abasıyanık, M. Construction of Biosensor for Determination of Galactose with Galactose Oxidase Immobilized on Polymeric Mediator Contains Ferrocene. Current Applied Physics 2010, 10, 1313–1316. [Google Scholar] [CrossRef]
- Vostiar, I.; Tkac, J.; Sturdik, E.; Gemeiner, P. Amperometric Urea Biosensor Based on Urease and Electropolymerized Toluidine Blue Dye as a PH-Sensitive Redox Probe. Bioelectrochemistry 2002, 56, 113–115. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Q.; Hu, Z.; Zhang, Y.; Wu, C.; Yang, M.; Wang, P. A Novel Electrochemical Biosensor Based on Dynamic Polymerase-Extending Hybridization for E. Coli O157:H7 DNA Detection. Talanta 2009, 78, 647–652. [Google Scholar] [CrossRef]
- Mayorga Martinez, C.C.; Treo, E.F.; Madrid, R.E.; Felice, C.C. Real-Time Measurement of Glucose Using Chrono-Impedance Technique on a Second Generation Biosensor. Biosensors and Bioelectronics 2011, 29, 200–203. [Google Scholar] [CrossRef]
- Pundir, C.S.; Lata, S.; Narwal, V. Biosensors for Determination of D and L- Amino Acids: A Review. Biosensors and Bioelectronics 2018, 117, 373–384. [Google Scholar] [CrossRef]
- Monteiro, T.; Almeida, M.G. Electrochemical Enzyme Biosensors Revisited: Old Solutions for New Problems. Critical Reviews in Analytical Chemistry 2019, 49, 44–66. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.H.; Lee, S.H.; Lee, U.J.; Fermin, C.D.; Kim, M. Immobilized Enzymes in Biosensor Applications. Materials 2019, 12, 121. [Google Scholar] [CrossRef] [PubMed]
- Tian, K.; Prestgard, M.; Tiwari, A. A Review of Recent Advances in Nonenzymatic Glucose Sensors. Materials Science and Engineering: C 2014, 41, 100–118. [Google Scholar] [CrossRef] [PubMed]
- Si, P.; Huang, Y.; Wang, T.; Ma, J. Nanomaterials for Electrochemical Non-Enzymatic Glucose Biosensors. RSC Adv. 2013, 3, 3487. [Google Scholar] [CrossRef]
- Muguruma, H. Biosensors: Enzyme Immobilization Chemistry. In Encyclopedia of Interfacial Chemistry; Elsevier, 2018; pp. 64–71. [CrossRef]
- Li, J.; Chang, H.; Zhang, N.; He, Y.; Zhang, D.; Liu, B.; Fang, Y. Recent Advances in Enzyme Inhibition Based-Electrochemical Biosensors for Pharmaceutical and Environmental Analysis. Talanta 2023, 253, 124092. [Google Scholar] [CrossRef]
- Jesionowski, T.; Zdarta, J.; Krajewska, B. Enzyme Immobilization by Adsorption: A Review. Adsorption 2014, 20, 801–821. [Google Scholar] [CrossRef]
- Mohamad, N.R.; Marzuki, N.H.C.; Buang, N.A.; Huyop, F.; Wahab, R.A. An Overview of Technologies for Immobilization of Enzymes and Surface Analysis Techniques for Immobilized Enzymes. Biotechnology & Biotechnological Equipment 2015, 29, 205–220. [Google Scholar] [CrossRef]
- Sassolas, A.; Blum, L.J.; Leca-Bouvier, B.D. Immobilization Strategies to Develop Enzymatic Biosensors. Biotechnology Advances 2012, 30, 489–511. [Google Scholar] [CrossRef]
- Bounegru, A.V.; Apetrei, C. Tyrosinase Immobilization Strategies for the Development of Electrochemical Biosensors—A Review. Nanomaterials 2023, 13, 760. [Google Scholar] [CrossRef]
- Liang, S.; Wu, X.-L.; Xiong, J.; Zong, M.-H.; Lou, W.-Y. Metal-Organic Frameworks as Novel Matrices for Efficient Enzyme Immobilization: An Update Review. Coordination Chemistry Reviews 2020, 406, 213149. [Google Scholar] [CrossRef]
- Lee, I.; Probst, D.; Klonoff, D.; Sode, K. Continuous Glucose Monitoring Systems - Current Status and Future Perspectives of the Flagship Technologies in Biosensor Research -. Biosensors and Bioelectronics 2021, 181, 113054. [Google Scholar] [CrossRef] [PubMed]
- Alp, B.; Mutlu, S.; Mutlu, M. Glow-Discharge-Treated Cellulose Acetate (CA) Membrane for a High Linearity Single-Layer Glucose Electrode in the Food Industry. Food Research International 2000, 33, 107–112. [Google Scholar] [CrossRef]
- Valdés-Ramírez, G.; Galicia, L. Biosensing Membrane Base on Ferulic Acid and Glucose Oxidase for an Amperometric Glucose Biosensor. Molecules 2021, 26, 3757. [Google Scholar] [CrossRef] [PubMed]
- Vieira, N.S.; De Souza, F.A.; Da Rocha, R.C.F.; Cestarolli, D.T.; Guerra, E.M. Development of Amperometric Biosensors Using VO2/GOx Films for Detection of Glucose. Materials Science in Semiconductor Processing 2021, 121, 105337. [Google Scholar] [CrossRef]
- Valdés-Ramírez, G.; Galicia, L. Glucose Oxidase Captured into Electropolymerized P-Coumaric Acid towards the Development of a Glucose Biosensor. Chemosensors 2023, 11, 345. [Google Scholar] [CrossRef]
- Jayakumar, K.; Bennett, R.; Leech, D. Electrochemical Glucose Biosensor Based on an Osmium Redox Polymer and Glucose Oxidase Grafted to Carbon Nanotubes: A Design-of-Experiments Optimisation of Current Density and Stability. Electrochimica Acta 2021, 371, 137845. [Google Scholar] [CrossRef]
- Estrada-Osorio, D.V.; Escalona-Villalpando, R.A.; Gutiérrez, A.; Arriaga, L.G.; Ledesma-García, J. Poly-L-Lysine-Modified with Ferrocene to Obtain a Redox Polymer for Mediated Glucose Biosensor Application. Bioelectrochemistry 2022, 146, 108147. [Google Scholar] [CrossRef]
- Wang, F.; Gong, W.; Wang, L.; Chen, Z. Enhanced Amperometric Response of a Glucose Oxidase and Horseradish Peroxidase Based Bienzyme Glucose Biosensor Modified with a Film of Polymerized Toluidine Blue Containing Reduced Graphene Oxide. Microchim Acta 2015, 182, 1949–1956. [Google Scholar] [CrossRef]
- Han, Z.; Zhang, X.; Yuan, H.; Li, Z.; Li, G.; Zhang, H.; Tan, Y. Graphene Oxide/Gold Nanoparticle/Graphite Fiber Microelectrodes for Directing Electron Transfer of Glucose Oxidase and Glucose Detection. Journal of Power Sources 2022, 521, 230956. [Google Scholar] [CrossRef]
- Singh, A.K.; Jaiswal, N.; Tiwari, I.; Ahmad, M.; Silva, S.R.P. Electrochemical Biosensors Based on in Situ Grown Carbon Nanotubes on Gold Microelectrode Array Fabricated on Glass Substrate for Glucose Determination. Microchim Acta 2023, 190, 55. [Google Scholar] [CrossRef]
- Ramanaviciene, A.; German, N.; Kausaite-Minkstimiene, A.; Ramanavicius, A. Glucose Biosensor Based on Dendritic Gold Nanostructures Electrodeposited on Graphite Electrode by Different Electrochemical Methods. Chemosensors 2021, 9, 188. [Google Scholar] [CrossRef]
- Sakalauskiene, L.; Popov, A.; Kausaite-Minkstimiene, A.; Ramanavicius, A.; Ramanaviciene, A. The Impact of Glucose Oxidase Immobilization on Dendritic Gold Nanostructures on the Performance of Glucose Biosensors. Biosensors 2022, 12, 320. [Google Scholar] [CrossRef]
- German, N.; Popov, A.; Ramanavicius, A.; Ramanaviciene, A. Development and Practical Application of Glucose Biosensor Based on Dendritic Gold Nanostructures Modified by Conducting Polymers. Biosensors 2022, 12, 641. [Google Scholar] [CrossRef] [PubMed]
- Kamel, K.S.; Oh, M.S.; Halperin, M.L. L-Lactic Acidosis: Pathophysiology, Classification, and Causes; Emphasis on Biochemical and Metabolic Basis. Kidney International 2020, 97, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Teusink, B.; Molenaar, D. Systems Biology of Lactic Acid Bacteria: For Food and Thought. Current Opinion in Systems Biology 2017, 6, 7–13. [Google Scholar] [CrossRef]
- Elshaghabee, F.M.F.; Rokana, N.; Panwar, H.; Heller, K.J.; Schrezenmeir, J. Probiotics for Dietary Management of Non-Alcoholic Fatty Liver Disease. Environ Chem Lett 2019, 17, 1553–1563. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Topolnikova, Ya.V.; Soldatkin, O.O. Advances in the Biosensors for Lactate and Pyruvate Detection for Medical Applications: A Review. TrAC Trends in Analytical Chemistry 2019, 110, 160–172. [Google Scholar] [CrossRef]
- Yata, V.K. Engineered Nanostructured Materials: Benefits and Risks. Environ Chem Lett 2019, 17, 1523–1527. [Google Scholar] [CrossRef]
- Narayanan, J.S.; Slaughter, G. Lactic Acid Biosensor Based on Lactate Dehydrogenase Immobilized on Au Nanoparticle Modified Microwire Electrode. IEEE Sensors J. 2020, 20, 4034–4040. [Google Scholar] [CrossRef]
- Istrate, O.-M.; Rotariu, L.; Bala, C. Amperometric L-Lactate Biosensor Based upon a Gold Nanoparticles/Reduced Graphene Oxide/Polyallylamine Hydrochloride Modified Screen-Printed Graphite Electrode. Chemosensors 2021, 9, 74. [Google Scholar] [CrossRef]
- Rahman, M.M.; Shiddiky, M.J.A.; Rahman, Md.A.; Shim, Y.-B. A Lactate Biosensor Based on Lactate Dehydrogenase/Nictotinamide Adenine Dinucleotide (Oxidized Form) Immobilized on a Conducting Polymer/Multiwall Carbon Nanotube Composite Film. Analytical Biochemistry 2009, 384, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Nikolaus, N.; Strehlitz, B. Amperometric Lactate Biosensors and Their Application in (Sports) Medicine, for Life Quality and Wellbeing. Microchim Acta 2008, 160, 15–55. [Google Scholar] [CrossRef]
- Tvorynska, S.; Barek, J.; Josypcuk, B. High-Performance Amperometric Biosensor for Flow Injection Analysis Consisting of a Replaceable Lactate Oxidase-Based Mini-Reactor and a Silver Amalgam Screen-Printed Electrode. Electrochimica Acta 2023, 445, 142033. [Google Scholar] [CrossRef]
- Ozoglu, O.; Uzunoglu, A.; Unal, M.A.; Gumustas, M.; Ozkan, S.A.; Korukluoglu, M.; Gunes Altuntas, E. Electrochemical Detection of Lactate Produced by Foodborne Presumptive Lactic Acid Bacteria. Journal of Bioscience and Bioengineering 2023, 135, 313–320. [Google Scholar] [CrossRef]
- Zaccheria, F.; Mariani, M.; Scotti, N.; Psaro, R.; Ravasio, N. Catalytic Upgrading of Lactose: A Rest Raw Material from the Dairy Industry. Green Chem. 2017, 19, 1904–1910. [Google Scholar] [CrossRef]
- Nasiri, H.; Baghban, H.; Teimuri-Mofrad, R.; Mokhtarzadeh, A. Graphitic Carbon Nitride/Magnetic Chitosan Composite for Rapid Electrochemical Detection of Lactose. International Dairy Journal 2023, 136, 105489. [Google Scholar] [CrossRef]
- de Brito, A.R.; dos Santos Reis, N.; Oliveira, P.C.; Rezende, D.V.B.; Monteiro, G.P.; Soares, G.A.; de Jesus, R.S.; Santos, A.S.; Salay, L.C.; de Oliveira, J.R.; et al. Development of Amperometric Biosensor in Modified Carbon Paste with Enzymatic Preparation Based on Lactase Immobilized on Carbon Nanotubes. J Food Sci Technol 2020, 57, 1342–1350. [Google Scholar] [CrossRef]
- De Brito, A.R.; De Jesus, R.S.; Tavares, I.M.D.C.; Silva, F.N.; Santana, N.B.; Barbosa Ferrão, S.P.; Bilal, M.; De Santana Santos, A.; Salay, L.C.; De Oliveira, J.R.; et al. Application of the Electrochemical Biosensor in the Detection of Lactose in Skimmed Milk. Surfaces and Interfaces 2021, 22, 100839. [Google Scholar] [CrossRef]
- Villalonga, A.; Sánchez, A.; Mayol, B.; Reviejo, J.; Villalonga, R. Electrochemical Biosensors for Food Bioprocess Monitoring. Current Opinion in Food Science 2022, 43, 18–26. [Google Scholar] [CrossRef]
- Bollella, P.; Gorton, L. Enzyme Based Amperometric Biosensors. Current Opinion in Electrochemistry 2018, 10, 157–173. [Google Scholar] [CrossRef]
- Tian, X.; Liu, H.; Liu, H.; Wang, X. Immobilizing Diamine Oxidase on Electroactive Phase-Change Microcapsules to Construct Thermoregulatory Smart Biosensor for Enhancing Detection of Histamine in Foods. Food Chemistry 2022, 397, 133759. [Google Scholar] [CrossRef] [PubMed]
- Erden, P.E.; Kaçar Selvi, C.; Kılıç, E. A Novel Tyramine Biosensor Based on Carbon Nanofibers, 1-Butyl-3-Methylimidazolium Tetrafluoroborate and Gold Nanoparticles. Microchemical Journal 2021, 170, 106729. [Google Scholar] [CrossRef]
- Istrate, O.-M.; Rotariu, L.; Bala, C. Amperometric L-Lactate Biosensor Based upon a Gold Nanoparticles/Reduced Graphene Oxide/Polyallylamine Hydrochloride Modified Screen-Printed Graphite Electrode. Chemosensors 2021, 9, 74. [Google Scholar] [CrossRef]
- Wu, L.; Pu, H.; Sun, D.-W. Novel Techniques for Evaluating Freshness Quality Attributes of Fish: A Review of Recent Developments. Trends in Food Science & Technology 2019, 83, 259–273. [Google Scholar] [CrossRef]
- Maddaloni, L.; Grasso, S.; De Gara, L.; Pennazza, G.; Zompanti, A.; Rapa, M.; Ruggieri, R.; Vinci, G.; Santonico, M. An Electrochemical Sensor for Monitoring Biogenic Amines in Anchovies as Quality and Safety Index. Sensors and Actuators B: Chemical 2021, 347, 130648. [Google Scholar] [CrossRef]
- Ahangari, H.; Kurbanoglu, S.; Ehsani, A.; Uslu, B. Latest Trends for Biogenic Amines Detection in Foods: Enzymatic Biosensors and Nanozymes Applications. Trends Food Sci Tech 2021, 112, 75–87. [Google Scholar] [CrossRef]
- Kaçar, C.; Erden, P.E.; Dalkiran, B.; İnal, E.K.; Kiliç, E. Amperometric Biogenic Amine Biosensors Based on Prussian Blue, Indium Tin Oxide Nanoparticles and Diamine Oxidase– or Monoamine Oxidase–Modified Electrodes. Anal Bioanal Chem 2020, 412, 1933–1946. [Google Scholar] [CrossRef]
- Liu, Y.; He, Y.; Li, H.; Jia, D.; Fu, L.; Chen, J.; Zhang, D.; Wang, Y. Biogenic Amines Detection in Meat and Meat Products: The Mechanisms, Applications, and Future Trends. Journal of Future Foods 2024, 4, 21–36. [Google Scholar] [CrossRef]
- Li, B.; Lu, S. The Importance of Amine-Degrading Enzymes on the Biogenic Amine Degradation in Fermented Foods: A Review. Process Biochemistry 2020, 99, 331–339. [Google Scholar] [CrossRef]
- Gao, X.; Li, C.; He, R.; Zhang, Y.; Wang, B.; Zhang, Z.-H.; Ho, C.-T. Research Advances on Biogenic Amines in Traditional Fermented Foods: Emphasis on Formation Mechanism, Detection and Control Methods. Food Chemistry 2023, 405, 134911. [Google Scholar] [CrossRef]
- Prasanna Kumar, S.; Parashuram, L.; Suhas, D.P.; Krishnaiah, P. Carboxylated Graphene-Alcohol Oxidase Thin Films Modified Graphite Electrode as an Electrochemical Sensor for Electro-Catalytic Detection of Ethanol. Materials Science for Energy Technologies 2020, 3, 159–166. [Google Scholar] [CrossRef]
- Pham, T.-T.D.; Phan, L.M.T.; Park, J.; Cho, S. Review—Electrochemical Aptasensor for Pathogenic Bacteria Detection. J. Electrochem. Soc. 2022, 169, 087501. [Google Scholar] [CrossRef]
- Shan, X.; Kuang, D.; Feng, Q.; Wu, M.; Yang, J. A Dual-Mode Ratiometric Aptasensor for Accurate Detection of Pathogenic Bacteria Based on Recycling of DNAzyme Activation. Food Chemistry 2023, 423, 136287. [Google Scholar] [CrossRef] [PubMed]
- Kurbanoglu, S.; Erkmen, C.; Uslu, B. Frontiers in Electrochemical Enzyme Based Biosensors for Food and Drug Analysis. TrAC Trends in Analytical Chemistry 2020, 124, 115809. [Google Scholar] [CrossRef]
- Xiong, X.; Tan, Y.; Mubango, E.; Shi, C.; Regenstein, J.M.; Yang, Q.; Hong, H.; Luo, Y. Rapid Freshness and Survival Monitoring Biosensors of Fish: Progress, Challenge, and Future Perspective. Trends in Food Science & Technology 2022, 129, 61–73. [Google Scholar] [CrossRef]
- Yang, L.; Bai, R.; Xie, B.; Zhuang, N.; Lv, Z.; Chen, M.; Dong, W.; Zhou, J.; Jiang, M. A Biosensor Based on Oriented Immobilization of an Engineered L-Glutamate Oxidase on a Screen-Printed Microchip for Detection of l-Glutamate in Fermentation Processes. Food Chemistry 2023, 405, 134792. [Google Scholar] [CrossRef]
- Backes, E.; Kato, C.G.; Corrêa, R.C.G.; Peralta Muniz Moreira, R.D.F.; Peralta, R.A.; Barros, L.; Ferreira, I.C.F.R.; Zanin, G.M.; Bracht, A.; Peralta, R.M. Laccases in Food Processing: Current Status, Bottlenecks and Perspectives. Trends in Food Science & Technology 2021, 115, 445–460. [Google Scholar] [CrossRef]
- Yang, L.; Guo, X.-Y.; Zheng, Q.-H.; Zhang, Y.; Yao, L.; Xu, Q.-X.; Chen, J.-C.; He, S.-B.; Chen, W. Construction of Platinum Nanozyme by Using Carboxymethylcellulose with Improved Laccase-like Activity for Phenolic Compounds Detection. Sensors and Actuators B: Chemical 2023, 393, 134165. [Google Scholar] [CrossRef]
- Nguyen, H.H.; Lee, S.H.; Lee, U.J.; Fermin, C.D.; Kim, M. Immobilized Enzymes in Biosensor Applications. Materials 2019, 12, 121. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Soldatkin, O.O.; Dzyadevych, S.V.; Soldatkin, A.P. Electrochemical Biosensors Based on Multienzyme Systems: Main Groups, Advantages and Limitations – A Review. Analytica Chimica Acta 2020, 1111, 114–131. [Google Scholar] [CrossRef]
- Li, Z.; Li, F.; Cai, L.; Chen, Z.; Qin, L.; Gao, X.-D. One-Pot Multienzyme Synthesis of Rare Ketoses from Glycerol. J. Agric. Food Chem. 2020, 68, 1347–1353. [Google Scholar] [CrossRef]
- Yang, K.; Li, F.; Qiao, Y.; Zhou, Q.; Hu, Z.; He, Y.; Yan, Y.; Xu, L.; Madzak, C.; Yan, J. Design of a New Multienzyme Complex Synthesis System Based on Yarrowia Lipolytica Simultaneously Secreted and Surface Displayed Fusion Proteins for Sustainable Production of Fatty Acid-Derived Hydrocarbons. ACS Sustainable Chem. Eng. 2018, 6, 17035–17043. [Google Scholar] [CrossRef]
- Monošík, R.; Ukropcová, D.; Streďanský, M.; Šturdík, E. Multienzymatic Amperometric Biosensor Based on Gold and Nanocomposite Planar Electrodes for Glycerol Determination in Wine. Analytical Biochemistry 2012, 421, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-Ramírez, L.; Rostro-Alanis, M.; Rodríguez-Rodríguez, J.; Sosa-Hernández, J.E.; Melchor-Martínez, E.M.; Iqbal, H.M.N.; Parra-Saldívar, R. Enzyme (Single and Multiple) and Nanozyme Biosensors: Recent Developments and Their Novel Applications in the Water-Food-Health Nexus. Biosensors 2021, 11, 410. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Li, M.; Luo, Z.; Qi, L.; Yu, L.; Zhang, S.; Liu, H. Designed Synthesis of Compartmented Bienzyme Biocatalysts Based on Core–Shell Zeolitic Imidazole Framework Nanostructures. Small 2023, 19, 2206606. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Shen, H.; Zhou, Z.; Huang, Z.; Chao, H.; Song, J.; Su, P.; Yang, Y. Janus DNA Bridges Metal-Organic Frameworks and Graphene Oxide for Convenient and Efficient Multienzyme Co-Immobilization with Boosted Activity. Applied Surface Science 2021, 570, 151242. [Google Scholar] [CrossRef]
- Arana-Peña, S.; Carballares, D.; Morellon-Sterlling, R.; Berenguer-Murcia, Á.; Alcántara, A.R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Enzyme Co-Immobilization: Always the Biocatalyst Designers’ Choice…or Not? Biotechnology Advances 2021, 51, 107584. [Google Scholar] [CrossRef]
- Xu, K.; Chen, X.; Zheng, R.; Zheng, Y. Immobilization of Multi-Enzymes on Support Materials for Efficient Biocatalysis. Front. Bioeng. Biotechnol. 2020, 8, 660. [Google Scholar] [CrossRef]
- Zou, L.; Wang, S.; Qiu, J. Preparation and Properties of a Glucose Biosensor Based on an Ionic Liquid-Functionalized Graphene/Carbon Nanotube Composite. New Carbon Materials 2020, 35, 12–19. [Google Scholar] [CrossRef]
- Bagal-Kestwal, D.R.; Chiang, B.-H. Electrochemical Invertase Probes with Nanocomposite of Microfibrillated Cellulose-Tragacanth Gum-Metal Nanoparticles for Direct Sucrose Analysis in Sweetened Beverages. J. Electrochem. Soc. 2019, 166, B720–B727. [Google Scholar] [CrossRef]
- Gursoy, O.; Sen Gursoy, S.; Cogal, S.; Celik Cogal, G. Development of a New Two-Enzyme Biosensor Based on Poly(Pyrrole-Co-3,4-Ethylenedioxythiophene) for Lactose Determination in Milk. Polym Eng Sci 2018, 58, 839–848. [Google Scholar] [CrossRef]
- Halpin, G.; McEntee, S.; Dwyer, C.; Lawless, F.; Dempsey, E. Lactose Biosensor Development and Deployment in Dairy Product Analysis. J. Electrochem. Soc. 2022, 169, 037528. [Google Scholar] [CrossRef]
- Pyeshkova, V.M.; Dudchenko, O.Y.; Soldatkin, O.O.; Alekseev, S.A.; Seker, T.; Kurc, B.A.; Dzyadevych, S.V. Development of Three-Enzyme Lactose Amperometric Biosensor Modified by Nanosized Poly (Meta-Phenylenediamine) Film. Appl Nanosci 2022, 12, 1267–1274. [Google Scholar] [CrossRef]
- Giménez-Gómez, P.; Gutiérrez-Capitán, M.; Capdevila, F.; Puig-Pujol, A.; Fernández-Sánchez, C.; Jiménez-Jorquera, C. Monitoring of Malolactic Fermentation in Wine Using an Electrochemical Bienzymatic Biosensor for L-Lactate with Long Term Stability. Analytica Chimica Acta 2016, 905, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Jin, K.; Luo, X.; Piao, J.; Wang, F. Electrochemical Biosensor Based on Chitosan- and Thioctic-Acid-Modified Nanoporous Gold Co-Immobilization Enzyme for Glycerol Determination. Chemosensors 2022, 10, 258. [Google Scholar] [CrossRef]
- Yokus, M.A.; Songkakul, T.; Pozdin, V.A.; Bozkurt, A.; Daniele, M.A. Wearable Multiplexed Biosensor System toward Continuous Monitoring of Metabolites. Biosensors and Bioelectronics 2020, 153, 112038. [Google Scholar] [CrossRef]
- Jalalvand, A.R. An Intelligent and Novel Electrochemical Biosensor for Simultaneous Enzymatic Biosensing of Cholesterol and Glucose in the Presence of Uric Acid Based on First-and Second-Order Calibration Methods. Microchemical Journal 2023, 191, 108824. [Google Scholar] [CrossRef]
- Liu, A.; Lang, Q.; Liang, B.; Shi, J. Sensitive Detection of Maltose and Glucose Based on Dual Enzyme-Displayed Bacteria Electrochemical Biosensor. Biosensors and Bioelectronics 2017, 87, 25–30. [Google Scholar] [CrossRef]
- Liu, L.; Chen, C.; Chen, C.; Kang, X.; Zhang, H.; Tao, Y.; Xie, Q.; Yao, S. Poly(Noradrenalin) Based Bi-Enzyme Biosensor for Ultrasensitive Multi-Analyte Determination. Talanta 2019, 194, 343–349. [Google Scholar] [CrossRef]
- Mahmudunnabi, R.G.; Farhana, F.Z.; Kashaninejad, N.; Firoz, S.H.; Shim, Y.-B.; Shiddiky, M.J.A. Nanozyme-Based Electrochemical Biosensors for Disease Biomarker Detection. Analyst 2020, 145, 4398–4420. [Google Scholar] [CrossRef]
- Wong, E.L.S.; Vuong, K.Q.; Chow, E. Nanozymes for Environmental Pollutant Monitoring and Remediation. Sensors 2021, 21, 408. [Google Scholar] [CrossRef] [PubMed]
- Arshad, F.; Mohd-Naim, N.F.; Chandrawati, R.; Cozzolino, D.; Ahmed, M.U. Nanozyme-Based Sensors for Detection of Food Biomarkers: A Review. RSC Adv. 2022, 12, 26160–26175. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, X.; Wang, Q.; Lou, Z.; Li, S.; Zhu, Y.; Qin, L.; Wei, H. Nanomaterials with Enzyme-like Characteristics (Nanozymes): Next-Generation Artificial Enzymes (II). Chem. Soc. Rev. 2019, 48, 1004–1076. [Google Scholar] [CrossRef] [PubMed]
- Torres Castillo, N.E.; Melchor-Martínez, E.M.; Ochoa Sierra, J.S.; Ramírez-Torres, N.M.; Sosa-Hernández, J.E.; Iqbal, H.M.N.; Parra-Saldívar, R. Enzyme Mimics In-Focus: Redefining the Catalytic Attributes of Artificial Enzymes for Renewable Energy Production. International Journal of Biological Macromolecules 2021, 179, 80–89. [Google Scholar] [CrossRef]
- Huang, Y.; Mu, X.; Wang, J.; Wang, Y.; Xie, J.; Ying, R.; Su, E. The Recent Development of Nanozymes for Food Quality and Safety Detection. J. Mater. Chem. B 2022, 10, 1359–1368. [Google Scholar] [CrossRef]
- Stasyuk, N.; Smutok, O.; Demkiv, O.; Prokopiv, T.; Gayda, G.; Nisnevitch, M.; Gonchar, M. Synthesis, Catalytic Properties and Application in Biosensorics of Nanozymes and Electronanocatalysts: A Review. Sensors 2020, 20, 4509. [Google Scholar] [CrossRef]
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; et al. Intrinsic Peroxidase-like Activity of Ferromagnetic Nanoparticles. Nature Nanotech 2007, 2, 577–583. [Google Scholar] [CrossRef]
- Ahangari, H.; Kurbanoglu, S.; Ehsani, A.; Uslu, B. Latest Trends for Biogenic Amines Detection in Foods: Enzymatic Biosensors and Nanozymes Applications. Trends in Food Science & Technology 2021, 112, 75–87. [Google Scholar] [CrossRef]
- Kurup, C.P.; Ahmed, M.U. Nanozymes towards Personalized Diagnostics: A Recent Progress in Biosensing. Biosensors 2023, 13, 461. [Google Scholar] [CrossRef]
- Dong, W.; Zhuang, Y.; Li, S.; Zhang, X.; Chai, H.; Huang, Y. High Peroxidase-like Activity of Metallic Cobalt Nanoparticles Encapsulated in Metal–Organic Frameworks Derived Carbon for Biosensing. Sensors and Actuators B: Chemical 2018, 255, 2050–2057. [Google Scholar] [CrossRef]
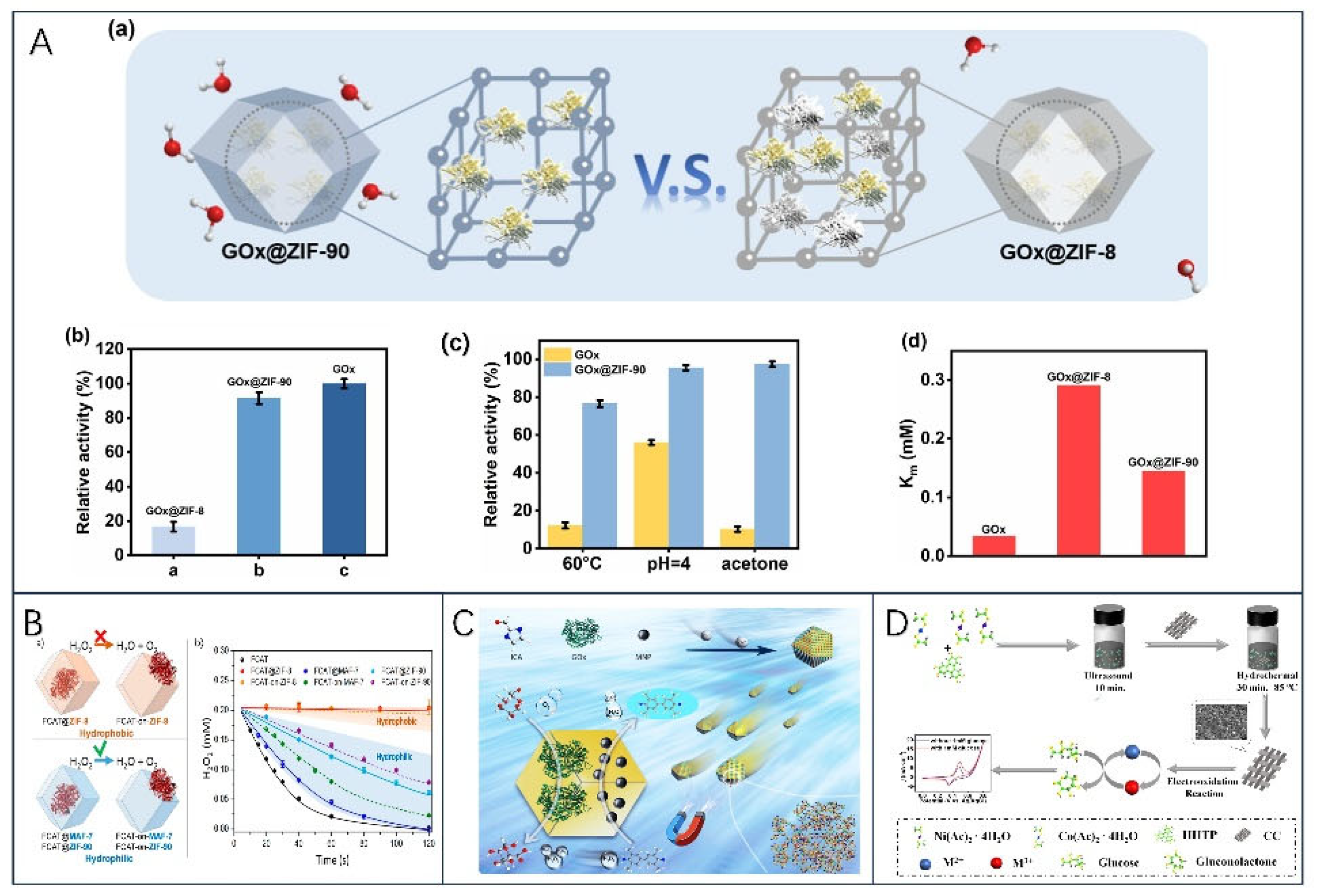
- Wang, X.; Lan, P.C.; Ma, S. Metal–Organic Frameworks for Enzyme Immobilization: Beyond Host Matrix Materials. ACS Cent. Sci. 2020, 6, 1497–1506. [Google Scholar] [CrossRef]
- Huang, Y.; Ren, J.; Qu, X. Nanozymes: Classification, Catalytic Mechanisms, Activity Regulation, and Applications. Chem. Rev. 2019, 119, 4357–4412. [Google Scholar] [CrossRef]
- Wang, W.; Gunasekaran, S. Nanozymes-Based Biosensors for Food Quality and Safety. TrAC Trends in Analytical Chemistry 2020, 126, 115841. [Google Scholar] [CrossRef]
- Chong, Y.; Liu, Q.; Ge, C. Advances in Oxidase-Mimicking Nanozymes: Classification, Activity Regulation and Biomedical Applications. Nano Today 2021, 37, 101076. [Google Scholar] [CrossRef]
- Su, Z.; Du, T.; Liang, X.; Wang, X.; Zhao, L.; Sun, J.; Wang, J.; Zhang, W. Nanozymes for Foodborne Microbial Contaminants Detection: Mechanisms, Recent Advances, and Challenges. Food Control 2022, 141, 109165. [Google Scholar] [CrossRef]
- Smutok, O.; Kavetskyy, T.; Prokopiv, T.; Serkiz, R.; Šauša, O.; Novák, I.; Švajdlenková, H.; Maťko, I.; Gonchar, M.; Katz, E. Biosensor Based on Peroxidase-Mimetic Nanozyme and Lactate Oxidase for Accurate L-Lactate Analysis in Beverages. Biosensors 2022, 12, 1042. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.-Y.; Wan, C.-Q.; Wang, Y.-X.; Shen, X.-F.; Pang, Y.-H. Bismuth-Based Metal-Organic Framework Peroxidase-Mimic Nanozyme: Preparation and Mechanism for Colorimetric-Converted Ultra-Trace Electrochemical Sensing of Chromium Ion. Journal of Hazardous Materials 2023, 451, 131148. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, Z.; Guan, J. Single-Atom Nanozyme-Based Electrochemical Sensors for Health and Food Safety Monitoring. Food Chemistry 2023, 425, 136518. [Google Scholar] [CrossRef]
- Stasyuk, N.; Gayda, G.; Zakalskiy, A.; Zakalska, O.; Serkiz, R.; Gonchar, M. Amperometric Biosensors Based on Oxidases and PtRu Nanoparticles as Artificial Peroxidase. Food Chemistry 2019, 285, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Stasyuk, N.; Gayda, G.; Demkiv, O.; Darmohray, L.; Gonchar, M.; Nisnevitch, M. Amperometric Biosensors for L-Arginine Determination Based on L-Arginine Oxidase and Peroxidase-Like Nanozymes. Applied Sciences 2021, 11, 7024. [Google Scholar] [CrossRef]
- Song, G.; Zhang, J.; Huang, H.; Wang, X.; He, X.; Luo, Y.; Li, J.; Huang, K.; Cheng, N. Single-Atom Ce-N-C Nanozyme Bioactive Paper with a 3D-Printed Platform for Rapid Detection of Organophosphorus and Carbamate Pesticide Residues. Food Chemistry 2022, 387, 132896. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Q.; Pang, X.; Luo, Y.; Huang, K.; He, X.; Yao, Z.; Li, J.-C.; Cheng, N. Fe-N-C Nanozyme Mediated Bioactive Paper-3D Printing Integration Technology Enables Portable Detection of Lactose in Milk. Sensors and Actuators B: Chemical 2022, 368, 132111. [Google Scholar] [CrossRef]
- Liu, L.; Du, J.; Liu, W.; Guo, Y.; Wu, G.; Qi, W.; Lu, X. Enhanced His@AuNCs Oxidase-like Activity by Reduced Graphene Oxide and Its Application for Colorimetric and Electrochemical Detection of Nitrite. Anal Bioanal Chem 2019, 411, 2189–2200. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Luo, L.; Zhang, L.; Kang, Y.; Sun, H.; Dan, J.; Sun, J.; Zhang, W.; Yue, T.; Wang, J. Oxidase-like Fe–Mn Bimetallic Nanozymes for Colorimetric Detection of Ascorbic Acid in Kiwi Fruit. LWT 2022, 154, 112821. [Google Scholar] [CrossRef]
- Han, X.; Liu, L.; Gong, H.; Luo, L.; Han, Y.; Fan, J.; Xu, C.; Yue, T.; Wang, J.; Zhang, W. Dextran-Stabilized Fe-Mn Bimetallic Oxidase-like Nanozyme for Total Antioxidant Capacity Assay of Fruit and Vegetable Food. Food Chemistry 2022, 371, 131115. [Google Scholar] [CrossRef]
- Xu, S.; Wang, S.; Guo, L.; Tong, Y.; Wu, L.; Huang, X. Nanozyme-Catalysed CRISPR-Cas12a System for the Preamplification-Free Colorimetric Detection of Lead Ion. Analytica Chimica Acta 2023, 1243, 340827. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Wang, F.; Chi, H.; Zhao, G.; Zhang, Y.; Li, T.; Wei, Q. Electrochemical Aptasensor Based on Gold Modified Thiol Graphene as Sensing Platform and Gold-Palladium Modified Zirconium Metal-Organic Frameworks Nanozyme as Signal Enhancer for Ultrasensitive Detection of Mercury Ions. Journal of Colloid and Interface Science 2022, 606, 510–517. [Google Scholar] [CrossRef]
- Wu, F.; Wang, H.; Lv, J.; Shi, X.; Wu, L.; Niu, X. Colorimetric Sensor Array Based on Au2Pt Nanozymes for Antioxidant Nutrition Quality Evaluation in Food. Biosensors and Bioelectronics 2023, 236, 115417. [Google Scholar] [CrossRef]
- Wu, K.; Feng, Y.; Li, Y.; Li, L.; Liu, R.; Zhu, L. S-Doped Reduced Graphene Oxide: A Novel Peroxidase Mimetic and Its Application in Sensitive Detection of Hydrogen Peroxide and Glucose. Anal Bioanal Chem 2020, 412, 5477–5487. [Google Scholar] [CrossRef]
- Dinh Van, T.; Dang Thi Thuy, N.; Dao Vu Phuong, T.; Nguyen Thi, N.; Nguyen Thi, T.; Nguyen Phuong, T.; Vu Van, T.; Vuong-Pham, H.; Phuong Dinh, T. High-Performance Nonenzymatic Electrochemical Glucose Biosensor Based on AgNP-Decorated MoS2 Microflowers. Current Applied Physics 2022, 43, 116–123. [Google Scholar] [CrossRef]
- Cao, B.; Gao, G.; Zhang, J.; Zhang, Z.; Sun, T. A Smartphone-Assisted Colorimetric Sensor Based on Fe1-XS Nanozyme for Detection of Glucose and Ascorbic-Acid in Soft Drinks. Microchemical Journal 2023, 193, 109018. [Google Scholar] [CrossRef]
- Xu, S.; Wu, F.; Mu, F.; Dai, B. The Preparation of Fe-Based Peroxidase Mimetic Nanozymes and for the Electrochemical Detection of Histamine. Journal of Electroanalytical Chemistry 2022, 908, 116088. [Google Scholar] [CrossRef]
- Chen, L.; Huang, W.; Hao, M.; Yang, F.; Shen, H.; Yu, S.; Wang, L. Rapid and Ultrasensitive Activity Detection of α-Amylase Based on γ-Cyclodextrin Crosslinked Metal-Organic Framework Nanozyme. International Journal of Biological Macromolecules 2023, 242, 124881. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xiao, S.; Zeng, M.; Xie, H.; Gan, N. Dual-Mode Colorimetric-Electrochemical Biosensor for Vibrio Parahaemolyticus Detection Based on CuO2 Nanodot-Encapsulated Metal-Organic Framework Nanozymes. Sensors and Actuators B: Chemical 2023, 387, 133835. [Google Scholar] [CrossRef]
- Reyes-De-Corcuera, J.I.; Olstad, H.E.; García-Torres, R. Stability and Stabilization of Enzyme Biosensors: The Key to Successful Application and Commercialization. Annu. Rev. Food Sci. Technol. 2018, 9, 293–322. [Google Scholar] [CrossRef]
- Bollella, P.; Gorton, L. Enzyme Based Amperometric Biosensors. Current Opinion in Electrochemistry 2018, 10, 157–173. [Google Scholar] [CrossRef]
- Hitaishi, V.; Clement, R.; Bourassin, N.; Baaden, M.; De Poulpiquet, A.; Sacquin-Mora, S.; Ciaccafava, A.; Lojou, E. Controlling Redox Enzyme Orientation at Planar Electrodes. Catalysts 2018, 8, 192. [Google Scholar] [CrossRef]
- Nemiwal, M.; Zhang, T.C.; Kumar, D. Enzyme Immobilized Nanomaterials as Electrochemical Biosensors for Detection of Biomolecules. Enzyme and Microbial Technology 2022, 156, 110006. [Google Scholar] [CrossRef]
- Mohamad Nor, N.; Ridhuan, N.S.; Abdul Razak, K. Progress of Enzymatic and Non-Enzymatic Electrochemical Glucose Biosensor Based on Nanomaterial-Modified Electrode. Biosensors 2022, 12, 1136. [Google Scholar] [CrossRef]
- Kilic, N.M.; Singh, S.; Keles, G.; Cinti, S.; Kurbanoglu, S.; Odaci, D. Novel Approaches to Enzyme-Based Electrochemical Nanobiosensors. Biosensors 2023, 13, 622. [Google Scholar] [CrossRef]
- Martinkova, P. Main Streams in the Construction of Biosensors and Their Applications. Int. J. Electrochem. Sci. 2017, 7386–7403. [Google Scholar] [CrossRef]
- Naresh, Varnakavi. ; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.; Jiang, H.; Fang, Y.; Wang, L.; Jiang, D. Emerging Electrochemical Biosensing Approaches for Detection of Allergen in Food Samples: A Review. Trends in Food Science & Technology 2022, 121, 93–104. [Google Scholar] [CrossRef]
- Cerrato-Alvarez, M.; Bernalte, E.; Bernalte-García, M.J.; Pinilla-Gil, E. Fast and Direct Amperometric Analysis of Polyphenols in Beers Using Tyrosinase-Modified Screen-Printed Gold Nanoparticles Biosensors. Talanta 2019, 193, 93–99. [Google Scholar] [CrossRef]
- Narayanan, J.S.; Slaughter, G. Lactic Acid Biosensor Based on Lactate Dehydrogenase Immobilized on Au Nanoparticle Modified Microwire Electrode. IEEE Sensors J. 2020, 20, 4034–4040. [Google Scholar] [CrossRef]
- Salvo-Comino, C.; Martín-Bartolomé, P.; Pura, J.L.; Perez-Gonzalez, C.; Martin-Pedrosa, F.; García-Cabezón, C.; Rodríguez-Méndez, M.L. Improving the Performance of a Bioelectronic Tongue Using Silver Nanowires: Application to Milk Analysis. Sensors and Actuators B: Chemical 2022, 364, 131877. [Google Scholar] [CrossRef]
- Sadak, O. One-Pot Scalable Synthesis of RGO/AuNPs Nanocomposite and Its Application in Enzymatic Glucose Biosensor. Nanocomposites 2021, 7, 44–52. [Google Scholar] [CrossRef]
- Beitollahi, H.; Tajik, S.; Garkani Nejad, F.; Safaei, M. Recent Advances in ZnO Nanostructure-Based Electrochemical Sensors and Biosensors. J. Mater. Chem. B 2020, 8, 5826–5844. [Google Scholar] [CrossRef]
- Baruah, S.; Maibam, B.; Borah, C.K.; Agarkar, T.; Kumar, A.; Kumar, S. A Highly Receptive ZnO-Based Enzymatic Electrochemical Sensor for Glucose Sensing. IEEE Sensors J. 2021, 21, 14601–14608. [Google Scholar] [CrossRef]
- Wang, Y.; Song, W.; Zhao, H.; Ma, X.; Yang, S.; Qiao, X.; Sheng, Q.; Yue, T. DNA Walker-Assisted Aptasensor for Highly Sensitive Determination of Ochratoxin A. Biosensors and Bioelectronics 2021, 182, 113171. [Google Scholar] [CrossRef]
- Hui, Y.; Peng, H.; Zhang, F.; Zhang, L.; Liu, Y.; Jia, R.; Song, Y.; Wang, B. An Ultrasensitive Sandwich-Type Electrochemical Aptasensor Using Silver Nanoparticle/Titanium Carbide Nanocomposites for the Determination of Staphylococcus Aureus in Milk. Microchim Acta 2022, 189, 276. [Google Scholar] [CrossRef]
- Zhang, Y.; Wan, Q.; Yang, N. Recent Advances of Porous Graphene: Synthesis, Functionalization, and Electrochemical Applications. Small 2019, 15, 1903780. [Google Scholar] [CrossRef] [PubMed]
- Taniselass, S.; Arshad, M.K.M.; Gopinath, S.C.B. Graphene-Based Electrochemical Biosensors for Monitoring Noncommunicable Disease Biomarkers. Biosensors and Bioelectronics 2019, 130, 276–292. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Lai, G.; Yu, A. Electroanalysis of Dopamine Using Polydopamine Functionalized Reduced Graphene Oxide-Gold Nanocomposite. 2014. [CrossRef]
- Zhang, X.; Liao, Q.; Chu, M.; Liu, S.; Zhang, Y. Structure Effect on Graphene-Modified Enzyme Electrode Glucose Sensors. Biosensors and Bioelectronics 2014, 52, 281–287. [Google Scholar] [CrossRef]
- Gong, H.; Hu, X.; Zeng, R.; Li, Y.; Xu, J.; Li, M.; Tang, D. CRISPR/Cas12a-Based Photoelectrochemical Sensing of MicroRNA on Reduced Graphene Oxide-Anchored Bi2WO6 Coupling with Catalytic Hairpin Assembly. Sensors and Actuators B: Chemical 2022, 369, 132307. [Google Scholar] [CrossRef]
- Zewdu, M.; Sandhu, T.; Kaur, N.; Abebe, M.; Olu, F.; Sabherwal, P. Synergetic Effect of Three-in-One Nanocomposite Based on AuNPs and RGO-MWCNTs for Ultrasensitive Electrochemical Bio-Diagnostic Applications. J. Electrochem. Soc. 2023, 170, 047513. [Google Scholar] [CrossRef]
- Bisht, N.; Patel, M.; Dwivedi, N.; Kumar, P.; Mondal, D.P.; Srivastava, A.K.; Dhand, C. Bio-Inspired Polynorepinephrine Based Nanocoatings for Reduced Graphene Oxide/Gold Nanoparticles Composite for High-Performance Biosensing of Mycobacterium Tuberculosis. Environmental Research 2023, 227, 115684. [Google Scholar] [CrossRef] [PubMed]
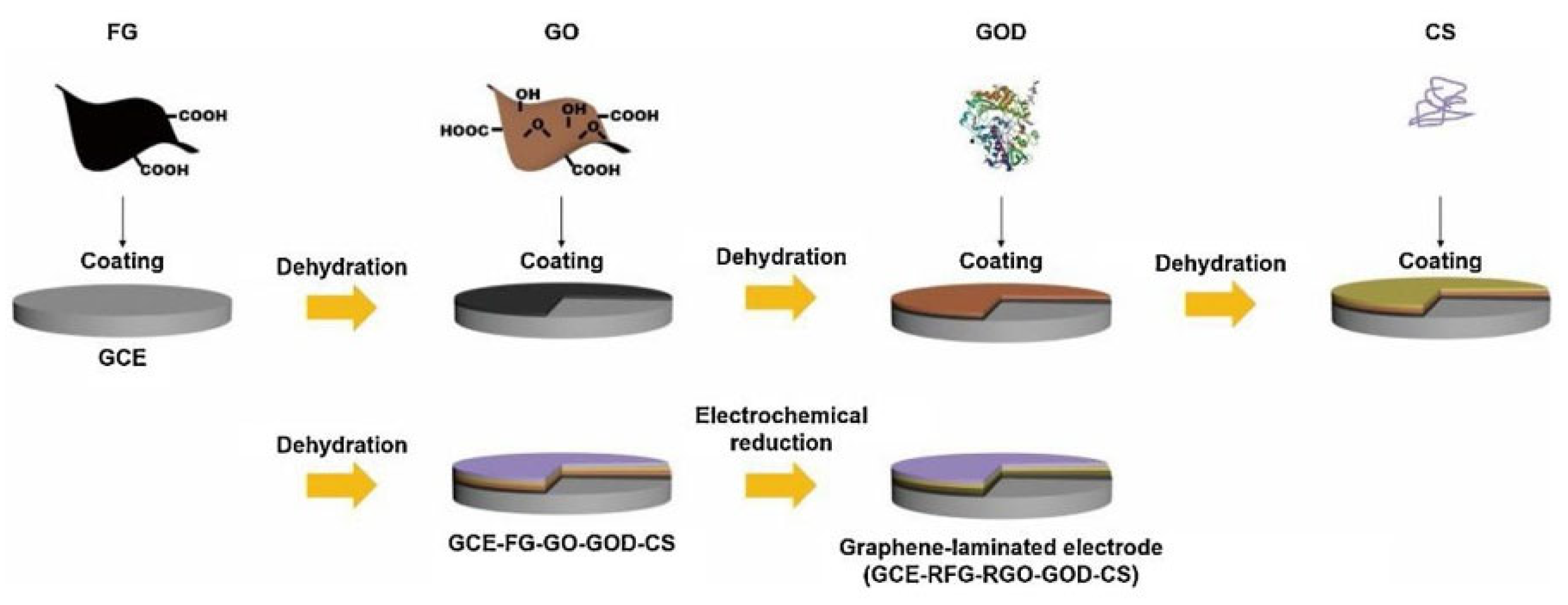
- Hao, Y.; Fang, M.; Xu, C.; Ying, Z.; Wang, H.; Zhang, R.; Cheng, H.-M.; Zeng, Y. A Graphene-Laminated Electrode with High Glucose Oxidase Loading for Highly-Sensitive Glucose Detection. Journal of Materials Science & Technology 2021, 66, 57–63. [Google Scholar] [CrossRef]
- Fang, M.; Hao, Y.; Ying, Z.; Wang, H.; Cheng, H.-M.; Zeng, Y. Controllable Edge Modification of Multi-Layer Graphene for Improved Dispersion Stability and High Electrical Conductivity. Appl Nanosci 2019, 9, 469–477. [Google Scholar] [CrossRef]
- Reddy, Y.V.M.; Shin, J.H.; Palakollu, V.N.; Sravani, B.; Choi, C.-H.; Park, K.; Kim, S.-K.; Madhavi, G.; Park, J.P.; Shetti, N.P. Strategies, Advances, and Challenges Associated with the Use of Graphene-Based Nanocomposites for Electrochemical Biosensors. Advances in Colloid and Interface Science 2022, 304, 102664. [Google Scholar] [CrossRef]
- Ramanaviciene, A.; German, N.; Kausaite-Minkstimiene, A.; Ramanavicius, A. Glucose Biosensor Based on Dendritic Gold Nanostructures Electrodeposited on Graphite Electrode by Different Electrochemical Methods. Chemosensors 2021, 9, 188. [Google Scholar] [CrossRef]
- Sakalauskiene, L.; Popov, A.; Kausaite-Minkstimiene, A.; Ramanavicius, A.; Ramanaviciene, A. The Impact of Glucose Oxidase Immobilization on Dendritic Gold Nanostructures on the Performance of Glucose Biosensors. Biosensors 2022, 12, 320. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.; Aukstakojyte, R.; Gaidukevic, J.; Lisyte, V.; Kausaite-Minkstimiene, A.; Barkauskas, J.; Ramanaviciene, A. Reduced Graphene Oxide and Polyaniline Nanofibers Nanocomposite for the Development of an Amperometric Glucose Biosensor. Sensors 2021, 21, 948. [Google Scholar] [CrossRef]
- Zheng, Y.; Zheng, S.; Xue, H.; Pang, H. Metal-Organic Frameworks/Graphene-Based Materials: Preparations and Applications. Adv. Funct. Mater. 2018, 28, 1804950. [Google Scholar] [CrossRef]
- Fan, M.; Liao, D.; Aboud, M.F.A.; Shakir, I.; Xu, Y. A Universal Strategy toward Ultrasmall Hollow Nanostructures with Remarkable Electrochemical Performance. Angew. Chem. Int. Ed. 2020, 59, 8247–8254. [Google Scholar] [CrossRef]
- Bu, F.; Chen, W.; Aboud, M.F.A.; Shakir, I.; Gu, J.; Xu, Y. Microwave-Assisted Ultrafast Synthesis of Adjustable Bimetal Phosphide/Graphene Heterostructures from MOFs for Efficient Electrochemical Water Splitting. J. Mater. Chem. A 2019, 7, 14526–14535. [Google Scholar] [CrossRef]
- Palakollu, V.N.; Chen, D.; Tang, J.-N.; Wang, L.; Liu, C. Recent Advancements in Metal-Organic Frameworks Composites Based Electrochemical (Bio)Sensors. Microchim Acta 2022, 189, 161. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Han, X.; Liu, Y.; Li, H.; Cui, Y. Metal–Covalent Organic Frameworks (MCOFs): A Bridge Between Metal–Organic Frameworks and Covalent Organic Frameworks. Angew. Chem. Int. Ed. 2020, 59, 13722–13733. [Google Scholar] [CrossRef]
- Zhang, Z.; Lou, Y.; Guo, C.; Jia, Q.; Song, Y.; Tian, J.-Y.; Zhang, S.; Wang, M.; He, L.; Du, M. Metal–Organic Frameworks (MOFs) Based Chemosensors/Biosensors for Analysis of Food Contaminants. Trends in Food Science & Technology 2021, 118, 569–588. [Google Scholar] [CrossRef]
- Sharanyakanth, P.S.; Radhakrishnan, M. Synthesis of Metal-Organic Frameworks (MOFs) and Its Application in Food Packaging: A Critical Review. Trends in Food Science & Technology 2020, 104, 102–116. [Google Scholar] [CrossRef]
- Cheng, W.; Tang, X.; Zhang, Y.; Wu, D.; Yang, W. Applications of Metal-Organic Framework (MOF)-Based Sensors for Food Safety: Enhancing Mechanisms and Recent Advances. Trends in Food Science & Technology 2021, 112, 268–282. [Google Scholar] [CrossRef]
- Du, T.; Huang, L.; Wang, J.; Sun, J.; Zhang, W.; Wang, J. Luminescent Metal-Organic Frameworks (LMOFs): An Emerging Sensing Platform for Food Quality and Safety Control. Trends in Food Science & Technology 2021, 111, 716–730. [Google Scholar] [CrossRef]
- Liu, J.; Wu, D.; Zhu, N.; Wu, Y.; Li, G. Antibacterial Mechanisms and Applications of Metal-Organic Frameworks and Their Derived Nanomaterials. Trends in Food Science & Technology 2021, 109, 413–434. [Google Scholar] [CrossRef]
- Xiao, J.; Fan, C.; Xu, T.; Su, L.; Zhang, X. An Electrochemical Wearable Sensor for Levodopa Quantification in Sweat Based on a Metal–Organic Framework/Graphene Oxide Composite with Integrated Enzymes. Sensors and Actuators B: Chemical 2022, 359, 131586. [Google Scholar] [CrossRef]
- Chen, C.; Xu, H.; Zhan, Q.; Zhang, Y.; Wang, B.; Chen, C.; Tang, H.; Xie, Q. Preparation of Novel HKUST-1-Glucose Oxidase Composites and Their Application in Biosensing. Microchim Acta 2023, 190, 10. [Google Scholar] [CrossRef]
- Liu, X.; Chen, W.; Lian, M.; Chen, X.; Lu, Y.; Yang, W. Enzyme Immobilization on ZIF-67/MWCNT Composite Engenders High Sensitivity Electrochemical Sensing. Journal of Electroanalytical Chemistry 2019, 833, 505–511. [Google Scholar] [CrossRef]
- Ge, D.; Li, M.; Wei, D.; Zhu, N.; Wang, Y.; Li, M.; Zhang, Z.; Zhao, H. Enhanced Activity of Enzyme Encapsulated in Hydrophilic Metal-Organic Framework for Biosensing. Chemical Engineering Journal 2023, 469, 144067. [Google Scholar] [CrossRef]
- Liang, W.; Xu, H.; Carraro, F.; Maddigan, N.K.; Li, Q.; Bell, S.G.; Huang, D.M.; Tarzia, A.; Solomon, M.B.; Amenitsch, H.; et al. Enhanced Activity of Enzymes Encapsulated in Hydrophilic Metal–Organic Frameworks. J. Am. Chem. Soc. 2019, 141, 2348–2355. [Google Scholar] [CrossRef]
- Ji, Y.; Gao, W.; Zhang, S.; Li, B.; Huang, H.; Zhang, X. Confining Natural/Mimetic Enzyme Cascade in an Amorphous Metal-Organic Framework for the Construction of Recyclable Biomaterials with Catalytic Activity. Langmuir 2022, 38, 927–936. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, Q.; Zhangsun, H.; Zhao, S.; Zhao, Y.; Wang, L. Carbon Cloth-Supported Nanorod-like Conductive Ni/Co Bimetal MOF: A Stable and High-Performance Enzyme-Free Electrochemical Sensor for Determination of Glucose in Serum and Beverage. Food Chemistry 2021, 349, 129202. [Google Scholar] [CrossRef]
- Pundir, M.; Prasher, P.; Vasić, K.; Leitgeb, M.; Kumar, A.; Prakash, R.; Knez, Ž.; Pandey, J.K.; Kumar, S. Enzyme Modified CNTs for Biosensing Application: Opportunities and Challenges. Colloid and Interface Science Communications 2021, 44, 100506. [Google Scholar] [CrossRef]
- Sheng, K.; Jiang, H.; Fang, Y.; Wang, L.; Jiang, D. Emerging Electrochemical Biosensing Approaches for Detection of Allergen in Food Samples: A Review. Trends in Food Science & Technology 2022, 121, 93–104. [Google Scholar] [CrossRef]
- Singh, A.K.; Jaiswal, N.; Tiwari, I.; Ahmad, M.; Silva, S.R.P. Electrochemical Biosensors Based on in Situ Grown Carbon Nanotubes on Gold Microelectrode Array Fabricated on Glass Substrate for Glucose Determination. Microchim Acta 2023, 190, 55. [Google Scholar] [CrossRef]
- Kumari, A.; Rajeev, R.; Benny, L.; Sudhakar, Y.N.; Varghese, A.; Hegde, G. Recent Advances in Carbon Nanotubes-Based Biocatalysts and Their Applications. Advances in Colloid and Interface Science 2021, 297, 102542. [Google Scholar] [CrossRef]
- Song, H.; Gao, G.; Ma, C.; Li, Y.; Shi, J.; Zhou, X.; Zhu, Z. A Hybrid System Integrating Xylose Dehydrogenase and NAD+ Coupled with PtNPs@MWCNTs Composite for the Real-Time Biosensing of Xylose. Analyst 2020, 145, 5563–5570. [Google Scholar] [CrossRef] [PubMed]
- Maity, D.; C. R., M.; R.T., R.K. Glucose Oxidase Immobilized Amine Terminated Multiwall Carbon Nanotubes/Reduced Graphene Oxide/Polyaniline/Gold Nanoparticles Modified Screen-Printed Carbon Electrode for Highly Sensitive Amperometric Glucose Detection. Materials Science and Engineering: C 2019, 105, 110075. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, B.K.; Ahmad, R.; Mousa, H.M.; Kim, I.-G.; Kim, J.I.; Neupane, M.P.; Park, C.H.; Kim, C.S. High-Performance Glucose Biosensor Based on Chitosan-Glucose Oxidase Immobilized Polypyrrole/Nafion/Functionalized Multi-Walled Carbon Nanotubes Bio-Nanohybrid Film. Journal of Colloid and Interface Science 2016, 482, 39–47. [Google Scholar] [CrossRef]
- Karim, M.R.; Alam, M.M.; Aijaz, M.O.; Asiri, A.M.; Dar, M.A.; Rahman, M.M. Fabrication of 1,4-Dioxane Sensor Based on Microwave Assisted PAni-SiO2 Nanocomposites. Talanta 2019, 193, 64–69. [Google Scholar] [CrossRef]
- Gerard, M. Application of Conducting Polymers to Biosensors. Biosensors and Bioelectronics 2002, 17, 345–359. [Google Scholar] [CrossRef]
- Kucherenko, I.S.; Soldatkin, O.O.; Kucherenko, D.Yu.; Soldatkina, O.V.; Dzyadevych, S.V. Advances in Nanomaterial Application in Enzyme-Based Electrochemical Biosensors: A Review. Nanoscale Adv. 2019, 1, 4560–4577. [Google Scholar] [CrossRef]
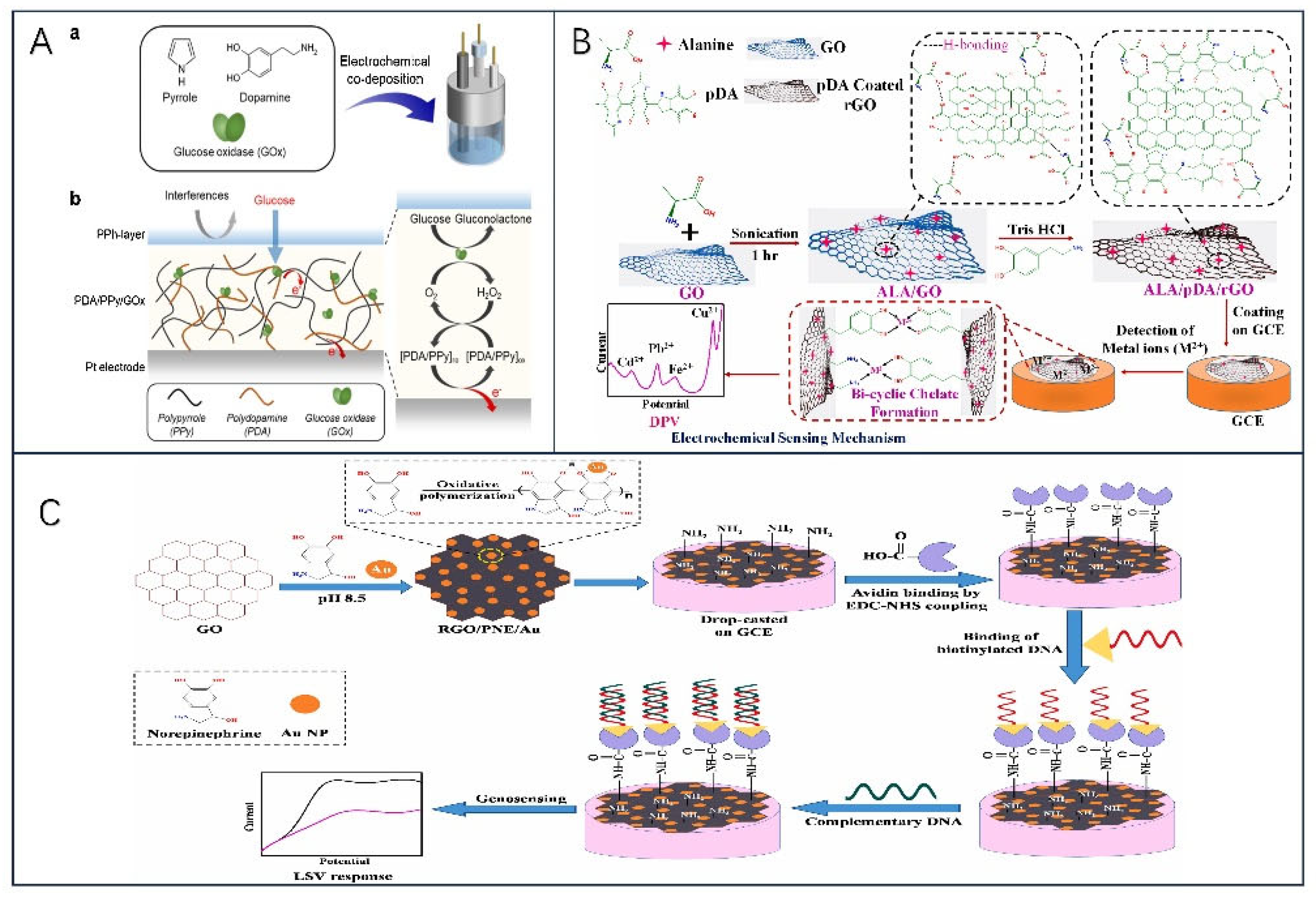
- Ramanavicius, A.; Rekertaitė, A.I.; Valiūnas, R.; Valiūnienė, A. Single-Step Procedure for the Modification of Graphite Electrode by Composite Layer Based on Polypyrrole, Prussian Blue and Glucose Oxidase. Sensors and Actuators B: Chemical 2017, 240, 220–223. [Google Scholar] [CrossRef]
- Shen, F.; Arshi, S.; Magner, E.; Ulstrup, J.; Xiao, X. One-Step Electrochemical Approach of Enzyme Immobilization for Bioelectrochemical Applications. Synthetic Metals 2022, 291, 117205. [Google Scholar] [CrossRef]
- Li, J.; Bi, X.; Tamulevičius, S.; Erts, D.; Chang, C.-F.; Gu, Y. Fabrication of a Biocompatible and Continuous Glucose Biosensor with the Poly(3,4-Ethylenedioxythiophene) Modified Electrode. Journal of the Taiwan Institute of Chemical Engineers 2019, 104, 1–7. [Google Scholar] [CrossRef]
- Senel, M.; Dervisevic, M.; Esser, L.; Dervisevic, E.; Dyson, J.; Easton, C.D.; Cadarso, V.J.; Voelcker, N.H. Enhanced Electrochemical Sensing Performance by in Situ Electrocopolymerization of Pyrrole and Thiophene-Grafted Chitosan. International Journal of Biological Macromolecules 2020, 143, 582–593. [Google Scholar] [CrossRef]
- Ou, J.; Ma, J.; Wang, F.; Li, W.; Fang, X.; Lei, S.; Amirfazli, A. Unexpected Superhydrophobic Polydopamine on Cotton Fabric. Progress in Organic Coatings 2020, 147, 105777. [Google Scholar] [CrossRef]
- Prabhakar, P.; Sen, R.K.; Patel, M.; Shruti; Dwivedi, N. ; Singh, S.; Kumar, P.; Chouhan, M.; Yadav, A.K.; Mondal, D.P.; et al. Development of Copper Impregnated Bio-Inspired Hydrophobic Antibacterial Nanocoatings for Textiles. Colloids and Surfaces B: Biointerfaces 2022, 220, 112913. [Google Scholar] [CrossRef]
- Lee, M.; Kim, S.; Jang, M.; Park, H.S.; Lee, J.Y. One-Pot Electrochemical Fabrication of High Performance Amperometric Enzymatic Biosensors Using Polypyrrole and Polydopamine. Journal of Industrial and Engineering Chemistry 2021, 97, 316–325. [Google Scholar] [CrossRef]
- Patel, M.; Bisht, N.; Prabhakar, P.; Sen, R.K.; Kumar, P.; Dwivedi, N.; Ashiq, M.; Mondal, D.P.; Srivastava, A.K.; Dhand, C. Ternary Nanocomposite-Based Smart Sensor: Reduced Graphene Oxide/Polydopamine/Alanine Nanocomposite for Simultaneous Electrochemical Detection of Cd2+, Pb2+, Fe2+, and Cu2+ Ions. Environmental Research 2023, 221, 115317. [Google Scholar] [CrossRef]
- Jeon, W.-Y.; Kim, H.-H.; Choi, Y.-B. Development of a Glucose Sensor Based on Glucose Dehydrogenase Using Polydopamine-Functionalized Nanotubes. Membranes 2021, 11, 384. [Google Scholar] [CrossRef]
- Liu, Y.; Nan, X.; Shi, W.; Liu, X.; He, Z.; Sun, Y.; Ge, D. A Glucose Biosensor Based on the Immobilization of Glucose Oxidase and Au Nanocomposites with Polynorepinephrine. RSC Adv. 2019, 9, 16439–16446. [Google Scholar] [CrossRef]
- Baldoneschi, V.; Palladino, P.; Banchini, M.; Minunni, M.; Scarano, S. Norepinephrine as New Functional Monomer for Molecular Imprinting: An Applicative Study for the Optical Sensing of Cardiac Biomarkers. Biosensors and Bioelectronics 2020, 157, 112161. [Google Scholar] [CrossRef]
- Lu, Z.; Teo, B.M.; Tabor, R.F. Recent Developments in Polynorepinephrine: An Innovative Material for Bioinspired Coatings and Colloids. J. Mat. Chem. B 2022, 10, 7895–7904. [Google Scholar] [CrossRef] [PubMed]
- Forouzanfar, S.; Alam, F.; Khakpour, I.; Baboukani, A.R.; Pala, N.; Wang, C. Highly Sensitive Lactic Acid Biosensors Based on Photoresist Derived Carbon. IEEE Sensors J. 2020, 20, 8965–8972. [Google Scholar] [CrossRef]
- Bollella, P.; Gorton, L. Enzyme Based Amperometric Biosensors. Current Opinion in Electrochemistry 2018, 10, 157–173. [Google Scholar] [CrossRef]
- Katz, E.; Bückmann, A.F.; Willner, I. Self-Powered Enzyme-Based Biosensors. J. Am. Chem. Soc. 2001, 123, 10752–10753. [Google Scholar] [CrossRef]
- Pinyou, P.; Blay, V.; Muresan, L.M.; Noguer, T. Enzyme-Modified Electrodes for Biosensors and Biofuel Cells. Mater. Horiz. 2019, 6, 1336–1358. [Google Scholar] [CrossRef]
- Kim, T.H.; Hahn, Y.K.; Kim, M.S. Recent Advances of Fluid Manipulation Technologies in Microfluidic Paper-Based Analytical Devices (ΜPADs) toward Multi-Step Assays. Micromachines 2020, 11, 269. [Google Scholar] [CrossRef]
- Yan, Y.; Zhao, D.; Li, W.; Li, X.; Chang, Y.; Zhang, Q.; Liu, M. An Origami Paper-Based Analytical Device for Rapid and Sensitive Analysis of Acrylamide in Foods. Micromachines 2021, 13, 13. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Z.; Wang, X. A Microfluidic PET-Based Electrochemical Glucose Sensor. Micromachines 2022, 13, 552. [Google Scholar] [CrossRef]
- Pagkali, V.; Soulis, D.; Kokkinos, C.; Economou, A. Fully Drawn Electrochemical Paper-Based Glucose Biosensors Fabricated by a High-Throughput Dual-Step Pen-on-Paper Approach with Commercial Writing Stationery. Sensors and Actuators B: Chemical 2022, 358, 131546. [Google Scholar] [CrossRef]
| Model enzymes | Support Materials | Target substance | Linear reaction range (μM) | Detection Limit (μM) | Application | References |
|---|---|---|---|---|---|---|
| HRP and GOx | G-IL/CNTs | Glucose | 0.004-5 mm | 3.99×10-7 M | Determination in real samples | [100] |
| INV and GOx | INVWM-GOx-Au/CuNPs-MFC-IGT/AuSPE | sucrose | 0.1 nM-10 μM | 0.1 nM | Direct Sucrose Analysis in Sweetened Beverages | [101] |
| GaOx and β-gal | P(Py-co-EDOT)-NaDBS | Lactose | 0.2–2.3 mM | 1.4 x 10-5 M | Lactose Determination in Milk Samples | [102] |
| GOx and β-gal | Chitosan/Enzyme(s)/Chitosan/GA | lactose | 5.83 x 10-3-1.65 x 10-2 M | 1.38 mM | Determination of lactose in dairy products | [103] |
| GOx,β-gal and mutarotase | PmPD | lactose | 0.01-1.25 mM | 0.005 mM | Determination of lactose in dairy products | [104] |
| HRP and LOx | Electrosynthesis PPy film | lactose | 1 x 10-6-1 x 10-4 M | 5.2 x 10-7 M | Monitor malolactic fermentation for winemaking | [105] |
| GK and GPO | GK/GPO/CHIT/TA/NPG/AuE | Glycerol | 0.1-5 mM | 77.08 μM | Control of wine quality | [106] |
| GOx and LOx | Flexible electrode array with gold nanoparticles and Prussian blue | GlucoseLactose | 60-1000 μM 5–20 mM | / | medical diagnosis | [107] |
| GOx, CO and HRP | MIPs/MWCNTs-IL/GCE | GlucoseCholesterol | 1-18 pM0.5-15 pM | 0.81 pM 0.23 pM | medical diagnosis | [108] |
| GA-bacteria and GDH-bacteria | MWNTs/GCE | MaltoseGlucose | 0.2–10 mM0.1–2.0 mM | 0.1 mM0.04 mM | Monitoring of food production and fermentation processes | [109] |
| HRP and GOx | Polynoradrenalin/Polyaniline electrode | GlucoseH2O2Cr(III)Cr(VI) | 0.50 μM–0.42 mM50–3.02 × 1040.01 ~ 3.85.0 × 10−4~6.0 × 10−3 | 0.08100.012.0 × 10−4 | Determination in real samples | [110] |
| Enzyme mimicked | Nanomaterials | Target substance | Linear range | Detection limit | Application | References |
|---|---|---|---|---|---|---|
| Oxidase | His@AuNCs/RGO | Nitrites | 2.5-5700 μM | 0.5 μM | Detection of nitrite in sausage samples | [134] |
| Oxidase | FeMnzyme | AA | 8 μM-56 μM | 0.88 μM | Determination of AA in actual kiwifruit fruit | [135] |
| Oxidase | Dex-FeMnzyme | TAC | 1 μM-30 μM | 1.17 μM | Practical applications in fruit and vegetable foods | [136] |
| Oxidase | MnO2 NRs | Pb2+ | 0.8–2500 nM | 0.54 nM | Detection in actual sample oils, wines, and spirits | [137] |
| Peroxidase | AuPd@UiO-67 | Hg2+ | 1.0 nM-1.0 mM | 0.16 nM | Actual measurements of tap water and lake water | [138] |
| Peroxidase | Au2Pt NPs | TAC | / | <0.2 μM | Determination of TAC in real samples (milk, green tea, and orange juice) | [139] |
| Peroxidase | S-rGO | H2O2 glucose | 0.1-1 μM1-100 μM | 0.042 μM0.38 μM | Determination of glucose in real samples | [140] |
| Peroxidase | AgNPs/MoS2-MF | glucose | 1-15 mM | 1.0 mM | Detection of glucose concentration in real samples | [141] |
| Peroxidase | Fe1-xS | Glucose AA | 200-700 μM 10-500 μM | 37 μM 53 μM | Detection of glucose and AA in actual beverages | [142] |
| Peroxidase | FeCo NCs | Histamine | 1-5000 nM | 0.79 nM | Detection of histamine in actual crab samples | [143] |
| Peroxidase | MOF-919-NH2@γ-CD | α-amylase activity | 0-200 U L-1 | 0.12 U L-1 | Determination of alpha-amylase activity in real distillers yeast samples | [144] |
| Peroxidase | PBA-CP@MOF | VP | 102-108 CFU mL-110-108 CFU mL-1 | 30 CFU mL-15 CFU mL-1 | Detection of VP in actual shrimp samples | [145] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




