Submitted:
10 August 2023
Posted:
11 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical approval
2.2. Groups and dosage
2.3. Preparation of the dried alginate beads
2.4. Parasitological analysis
2.5. Performance parameters
2.6. Statistical analysis
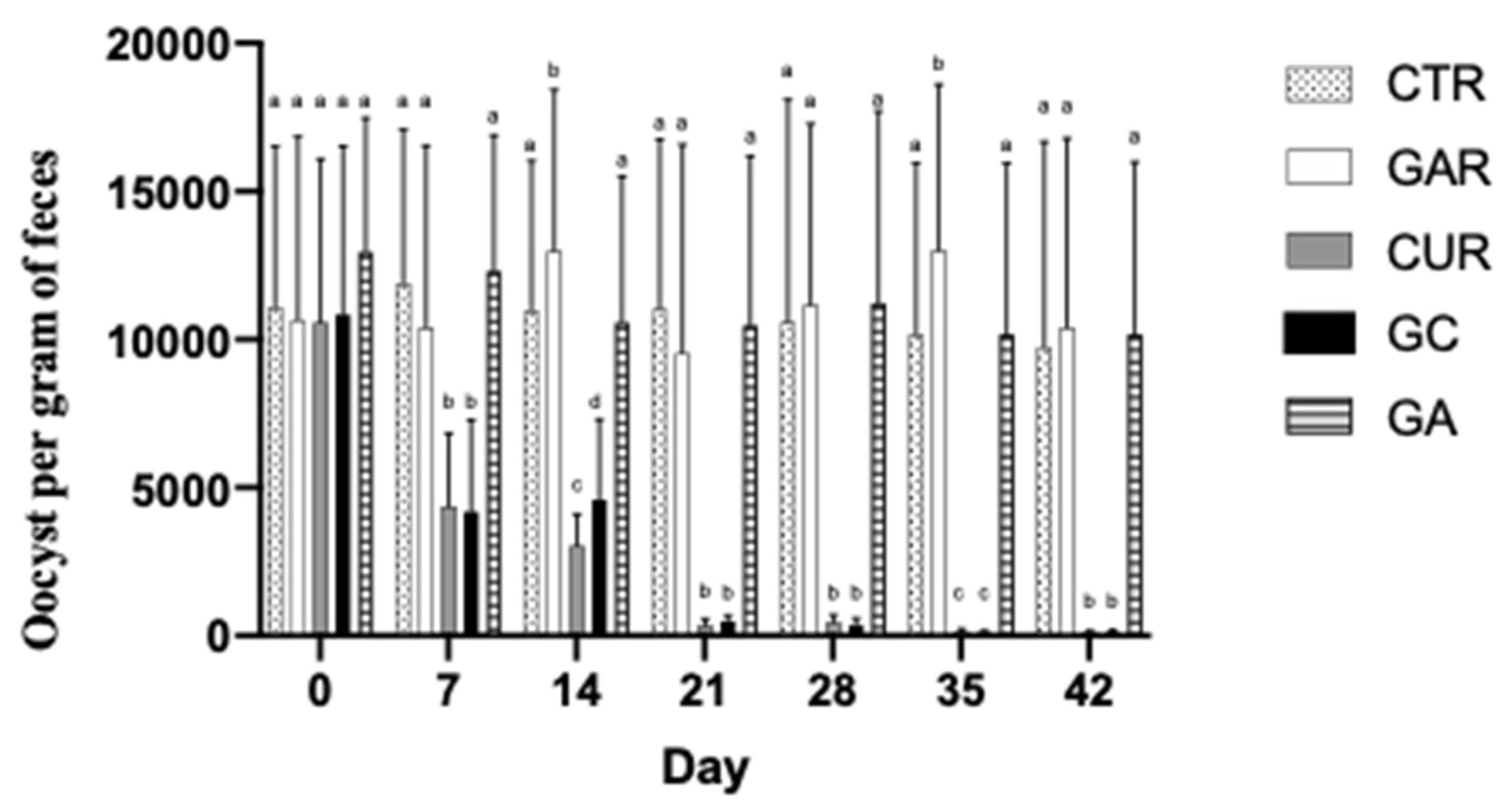
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. Food and Agriculture Organization of the United Nations. Gateway to poultry production and products. 2022. Available online: https://www.fao.org/poultry-production-products/en/ (accessed on July 2023).
- UNA. Unión Nacional de Avicultores. Compendio de indicadores económicos del sector avícola. 2021. Available online: https://una.org.mx/indicadores-economicos/ (accessed on July 2023).
- Taylor, M.A.; Coop, R.L.; Wall, R. Veterinary parasitology; Willey-Blackwell: Oxford, UK, 2016. [Google Scholar]
- Fatoba, A.J.; Adeleke, M.A. Diagnosis and control of chicken coccidiosis: a recent update. J Parasit Dis. 2018, 42, 483–493. [Google Scholar] [CrossRef]
- Chapman, H.D. Coccidiosis in the turkey. Avian Pathol. 2008, 37, 205–223. [Google Scholar] [CrossRef]
- Vrba, V.; Pakandl, M. Coccidia of turkey: from isolation, characterization and comparison to molecular phylogeny and molecular diagnostics. International journal for parasitology. 2014, 44, 985–1000. [Google Scholar] [CrossRef]
- Fatoba, A.J.; Adeleke, M.A. Diagnosis and control of chicken coccidiosis: a recent update. Journal of Parasitic Diseases. 2018, 42, 483–493. [Google Scholar] [CrossRef]
- Chapman, H.D.; Rathinam, T. Focused review: the role of drug combinations for the control of coccidiosis in commercially reared chickens. International Journal for Parasitology: Drugs and Drug Resistance. 2022, 18, 32–42. [Google Scholar] [CrossRef]
- Chapman, H.D. Milestones in avian coccidiosis research: a review. Poult Sci. 2014, 93, 501–511. [Google Scholar] [CrossRef]
- Madlala, T.; Okpeku, M.; Adeleke, M.A. Understanding the interactions between Eimeria infection and gut microbiota, towards the control of chicken coccidiosis: a review. Parasite. 2021, 28, 48. [Google Scholar] [CrossRef]
- El-Shall, N.A.; Abd El-Hack, M.E.; Albaqami, N.M.; Khafaga, A.F.; Taha, A.E.; Swelum, A.A.; et al. Phytochemical control of poultry coccidiosis: a review. Poult Sci. 2022, 101, 101542. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Kruthiventi, A.K.; Doble, M. Synergism between natural products and antibiotics against infectious diseases. Phytomedicine. 2008, 15, 639–652. [Google Scholar] [CrossRef]
- Kim, J.E.; Lillehoj, H.S.; Hong, Y.H.; Kim, G.B.; Lee, S.H.; Lillehoj, E.P.; et al. Dietary Capsicum and Curcuma longa oleoresins increase intestinal microbiome and necrotic enteritis in three commercial broiler breeds. Res Vet Sci. 2015, 102, 150–158. [Google Scholar] [CrossRef]
- Yadav, S.; Teng, P.Y.; Souza Dos Santos, T.; Gould, R.L.; Craig, S.W.; Lorraine Fuller, A.; et al. The effects of different doses of curcumin compound on growth performance, antioxidant status, and gut health of broiler chickens challenged with Eimeria species. Poult Sci. 2020, 99, 5936–5945. [Google Scholar] [CrossRef]
- Chattopadhyay, I.; Biswas, K.; Bandyopadhyay, U.; Banerjee, R.K. Turmeric and curcumin: Biological actions and medicinal applications. Current science. 2004, 44–53. [Google Scholar]
- Adjei-Mensah, B.; Atuahene, C.C. Avian coccidiosis and anticoccidial potential of garlic (Allium sativum, L.) in broiler production: a review. Journal of Applied Poultry Research. Journal of Applied Poultry Research. 2022, 100314. [Google Scholar]
- Shabkhiz, M.A.; Pirouzifard, M.K.; Pirsa, S.; Mahdavinia, G.R. Alginate hydrogel beads containing Thymus daenensis essential oils/Glycyrrhizic acid loaded in β-cyclodextrin. Investigation of structural, antioxidant/antimicrobial properties and release assessment. Journal of Molecular Liquids. 2021, 344, 117738. [Google Scholar] [CrossRef]
- Noppakundilograt, S.; Piboon, P.; Graisuwan, W.; Nuisin, R.; Kiatkamjornwong, S. Encapsulated eucalyptus oil in ionically cross-linked alginate microcapsules and its controlled release. Carbohydr Polym. 2015, 131, 23–33. [Google Scholar] [CrossRef]
- Gholamian, S.; Nourani, M.; Bakhshi, N. Formation and characterization of calcium alginate hydrogel beads filled with cumin seeds essential oil. Food Chemistry. 2021, 338, 128143. [Google Scholar] [CrossRef]
- Zhang, Y.; Gong, J.; Yu, H.; Guo, Q.; Defelice, C.; Hernandez, M.; et al. Alginate-whey protein dry powder optimized for target delivery of essential oils to the intestine of chickens. Poult Sci. 2014, 93, 2514–2525. [Google Scholar] [CrossRef]
- Voo W-P, Ravindra, P. ; Tey B-T, Chan E-S. Comparison of alginate and pectin-based beads for production of poultry probiotic cells. Journal of bioscience and bioengineering. 2011, 111, 294–299. [Google Scholar] [CrossRef]
- Gomez-Garcia, J.; Chavez-Carbajal, A.; Segundo-Arizmendi, N.; Baron-Pichardo, M.G.; Mendoza-Elvira, S.E.; Hernandez-Baltazar, E.; et al. Efficacy of Salmonella bacteriophage S1 delivered and released by alginate beads in a chicken model of infection. Viruses. 2021, 13, 1932. [Google Scholar] [CrossRef]
- Khoshtinat, K.; Barzegar, M.; Sahari, M.A.; Hamidi, Z. Encapsulation of Iranian Garlic Oil with β-cyclodextrin: Optimization and its Characterization. 2017.
- Hazra, K.; Kumar, R.; Sarkar, B.K.; Chowdary, Y.A.; Devgan, M.; Ramaiah, M. UV-visible spectrophotometric estimation of curcumin in nanoformulation. International journal of pharmacognosy. 2015, 2, 127–130. [Google Scholar]
- Figueroa CJA, Jasso, V.C.; Liébano, H.E.; Martínez, L.P.; Rodríguez VRI, Zárate RJJ. Examen Coproparasitoscópico. In: Técnicas para el diagnóstico de parásitos con importancia en salud pública y veterinaria. AMPAVE-CONASA, editor. Mexico, D.F.: AMPAVE-CONASA; 2015. 517 p.
- Clarkson, M.J. The life history and pathogenicity of Eimeria meleagrimitis Tyzzer 1929, in the turkey poult. Parasitology. 1959, 49, 70–82. [Google Scholar] [CrossRef]
- Gutierrez, O.L.; Sumano, L.H.; Zamora, Q.M. Administration of enrofloxacin and capsaicin to chickens to achieve higher maximal serum concentrations. Veterinary record. 2002, 150, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, B.; De Backer, P.; Remon, J.P. Drug administration to poultry. Advanced Drug Delivery Reviews. 2002, 54, 795–803. [Google Scholar] [CrossRef]
- Roura, E.; Baldwin, M.W.; Klasing, K.C. The avian taste system: Potential implications in poultry nutrition. Animal Feed Science and Technology. 2013, 180, 1–9. [Google Scholar] [CrossRef]
- Liu H-X, Rajapaksha, P. ; Wang, Z.; Kramer, N.E.; Marshall, B.J. An update on the sense of taste in chickens: A better-developed system than previously appreciated. Journal of nutrition & food sciences. 2018, 8. [Google Scholar]
- Severino, P.; da Silva, C.F.; Andrade, L.N.; de Lima Oliveira, D.; Campos, J.; Souto, E.B. Alginate Nanoparticles for Drug Delivery and Targeting. Curr Pharm Des. 2019, 25, 1312–1334. [Google Scholar] [CrossRef] [PubMed]
- Gheorghita Puscaselu, R.; Lobiuc, A.; Dimian, M.; Covasa, M. Alginate: From Food Industry to Biomedical Applications and Management of Metabolic Disorders. Polymers (Basel). 2020, 12. [Google Scholar] [CrossRef]
- Gutierrez, L.; Tapia, G.; Gutierrez, E.; Sumano, H. Evaluation of a Tasteless Enrofloxacin Pharmaceutical Preparation for Cats. Naive Pooled-Sample Approach to Study Its Pharmacokinetics. Animals (Basel). 2021, 11. [Google Scholar]
- Gutierrez, L.; Lechuga, T.; Marcos, X.; García-Guzmán, P.; Gutierrez, C.; Sumano, H. Comparative bioavailability of enrofloxacin in dogs when concealed in noncommercial morsels, either as a tablet or as enrofloxacin–alginate dried beads. Journal of Veterinary Pharmacology and Therapeutics. 2021, 44, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, F.; Al-Quraishy, S.; Steinbrenner, H.; Sies, H.; Dkhil, M.A. Towards identifying novel anti-Eimeria agents: trace elements, vitamins, and plant-based natural products. Parasitol Res. 2014, 113, 3547–3556. [Google Scholar] [CrossRef] [PubMed]
- Sidiropoulou, E.; Skoufos, I.; Marugan-Hernandez, V.; Giannenas, I.; Bonos, E.; Aguiar-Martins, K.; et al. Anticoccidial Study of Oregano and Garlic Essential Oils and Effects on Growth Performance, Fecal Oocyst Output, and Intestinal Microbiota. Front Vet Sci. 2020, 7, 420. [Google Scholar] [CrossRef]
- Gamboa Izurieta, M.F. Evaluación de diferentes niveles de Cúrcuma longa (Cúrcuma), como pigmetante natural en dietas a base de sorgo, para la implementación de pollos broiler. 2016.
- Llerena Lima, G.S. Efecto de tres niveles de harina de palillo (Curcuma longa, L.) en la pigmentación y comportamiento productivo de pollos Broiler en Pucallpa. 2016.
- Al-Rubaei, Z.M.; Mohammad, T.U.; Ali, L.K. Effects of local curcumin on oxidative stress and total antioxidant capacity in vivo study. Pak J Biol Sci. 2014, 17, 1237–1241. [Google Scholar] [CrossRef] [PubMed]
- Balaji, S.; Chempakam, B. Toxicity prediction of compounds from turmeric (Curcuma longa L). Food Chem Toxicol. 2010, 48, 2951–2959. [Google Scholar] [CrossRef] [PubMed]
- Ahad, S.; Tanveer, S.; Nawchoo, I.A.; Malik, T.A. Anticoccidial activity of Artemisia vestita (Anthemideae, Asteraceae) - a traditional herb growing in the Western Himalayas, Kashmir, India. Microb Pathog. 2017, 104, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Burt, S.A.; Tersteeg-Zijderveld, M.H.; Jongerius-Gortemaker, B.G.; Vervelde, L.; Vernooij, J.C. In vitro inhibition of Eimeria tenella invasion of epithelial cells by phytochemicals. Vet Parasitol. 2013, 191, 374–378. [Google Scholar] [CrossRef]
- Galli, G.M.; Da Silva, A.S.; Biazus, A.H.; Reis, J.H.; Boiago, M.M.; Topazio, J.P.; et al. Feed addition of curcumin to laying hens showed anticoccidial effect and improved egg quality and animal health. Res Vet Sci. 2018, 118, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lillehoj, H.S.; Lee, S.H.; Lillehoj, E.P.; Bravo, D. Improved resistance to Eimeria acervulina infection in chickens due to dietary supplementation with garlic metabolites. Br J Nutr. 2013, 109, 76–88. [Google Scholar] [CrossRef]


| Species | CTR | GAR | CUR | GC |
|---|---|---|---|---|
| E. gallopavonis | 20.64% | 22.29% | 8.94% | 14.25% |
| E. subrotunda | 6.09% | 16.96% | 23.59% | 11.79% |
| E. innocua | 17.32% | 6.13% | 10.99% | 2.51% |
| E. meleagrimitis | 27.00% | 25.93% | 5.68% | 30.38% |
| E. dispersa | 3.05% | 4.33% | 24.30% | 4.82% |
| E. meleagridis | 8.73% | 9.50% | 21.68% | 6.98% |
| E. adenoieides | 17.18% | 14.86% | 4.82% | 29.27% |
| Group | Weight gain (kg) | Feed consumption (kg) | Feed conversion index |
|---|---|---|---|
| CTR | 3.55 ± 0.2a | 12.34 ± 0.8a | 3.47 ± 0.2a |
| CUR | 3.38 ± 0.3a | 11.80 ± 1.0 a | 3.49 ± 0.2a |
| GAR | 3.20 ± 0.2a | 10.88 ± 0.9a | 3.40 ± 0.1a |
| GA | 4.69 ± 0.3b | 11.09 ± 0.9b | 2.28 ± 0.1b |
| GC | 4.89 ± 0.2b | 11.04 ± 1.0b | 2.25 ± 0.2b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




