3. Discussion
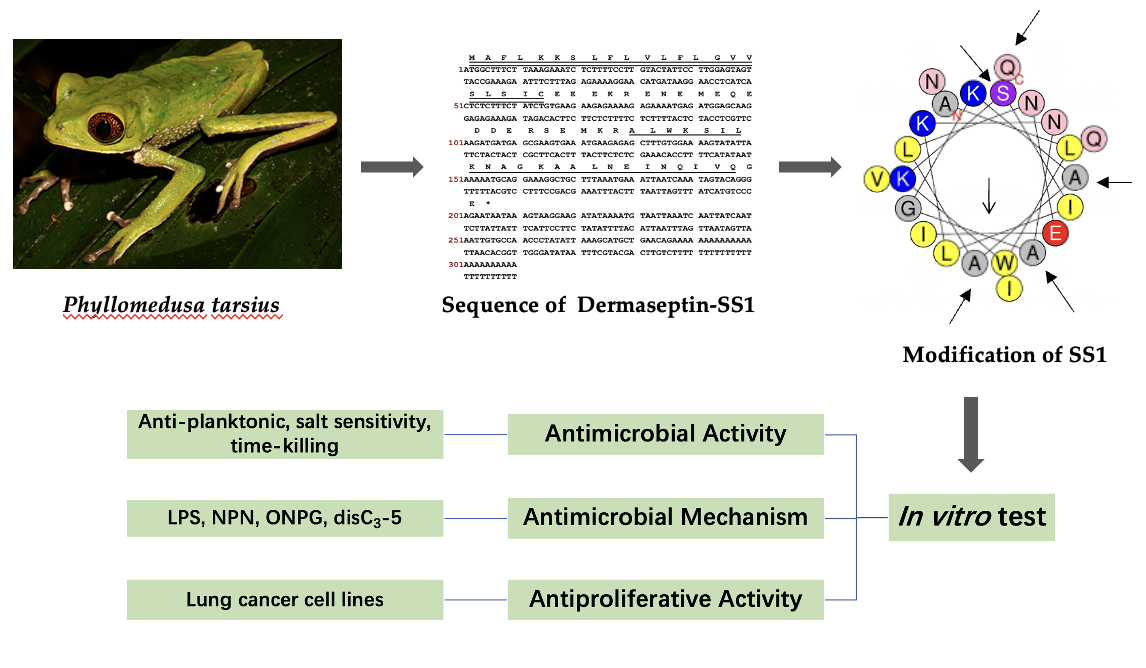
The study reported the identification of a newly discovered antimicrobial peptide, Dermaseptin-SS1, which was separated from the skin secretion of Phyllomedusa tarsius. The anticipated secondary structure of SS1 was determined to be an amphipathic α-helix configuration, which was subsequently validated by the CD spectrum analysis. To assess the potential efficacy of SS1 against drug-resistant bacteria, an in vitro investigation was conducted to examine its anti-planktonic microorganism activity.
Generally, SS1 and its analogues displayed a greater range of antimicrobial activity against Gram-negative bacteria. In a comparison of the TI values of SS1 and its analogues in Table 4, 14V5K showed an obvious antimicrobial therapeutic effect. Therefore, SS1 and 14V5K were selected to measure future antimicrobial activities and mechanisms.
As peptides showed significant antimicrobial activity against
E. coli 8739, additional strains of drug-resistant
E. coli were used in the study. These were; (i)
E. coli 2340: KPC strain panel, bl
KPC+/bla
NDM-; (ii)
E. coli 2469: NDM-1 strain panel, bla
NDM+/bla
KPC-; (iii)
E. coli 2471: NDM-1 strain panel, bla
NDM+/bla
KPC-; (iv)
E. coli 13846: colistin-resistant with MCR-1 positive.
E. coli 2340, 2469 and 2471 belonged to Carbapenem-Resistant
Enterobacteriaceae (CRE) [
12]. Carbapenem antibiotics are mostly β-lactam antibiotics whose principle was to inhibit peptidoglycan synthesis [
13]. The mechanisms of carbapenem resistance were β-lactamase production combined with porin mutations to produce carbapenemase [
14]. CRE had three classifications, including KPC, NDM and OXA-48.
Klebsiella pneumoniae Carbapenemase (KPC) hydrolysed all β-lactam agents encoded by the plasmid-associated gene bla
KPC, which may be difficult to detect using higher breakpoints [
15]. New Delhi Metallo-β-lactamase (NDM) was encoded by the plasmid-associated gene bla
NDM [
16]. However,
E. coli 13846 was a colistin-resistant bacterium with MCR-1 positive and harboured multiple other resistance genes. The MCR-1 protein was a transmembrane protein primarily found in the cellular membrane. Its presence has potentially led to the deterioration of cell membrane integrity, decreasing the MICs of gentamicin, kanamycin, and rifampicin [
17]. The presence of MCR-1 conferred protection to the host against colistin, while simultaneously altering the permeability of the cellular membrane and diminishing resistance to hydrophobic antibiotics. The results in
Table 5 could be explained in that the antimicrobial activities of peptides against
E. coli 2340, 2469 and 2471, were better than those against
E. coli 13846 because
E. coli 13846 had the ability to change the cell membrane structure but the others did not.
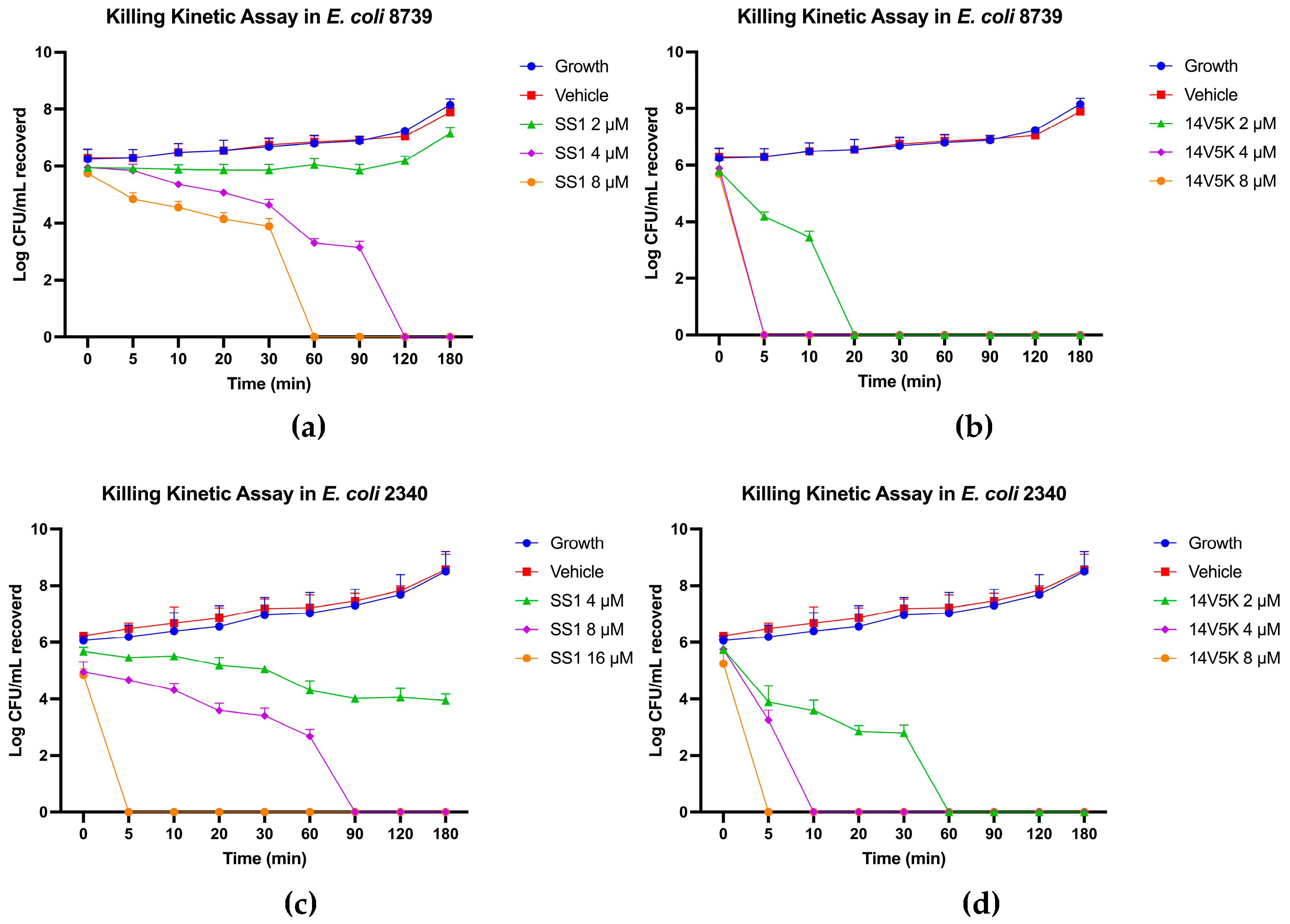
14V5K showed significant superiority in salt sensitivity against both bacteria, as shown in Table 7. The results could be analysed from the perspective of the properties of the peptides. On the one hand, 14V5K had more net charges than SS1, so it was more competitive than most ions. The combination between the negatively charged LPS and peptide 14V5K produced a stronger electrostatic interaction, which was one of the factors of the membrane-targeting mechanism. On the other hand, 14V5K had a higher hydrophobic moment than SS1, and with this elevated hydrophobicity, the peptide could readily accumulate on the phospholipid bilayers of the cell membrane. Therefore, 14V5K had low salt sensitivity. Meanwhile, SS1 displayed long-duration killing for about 2 hours, while 14V5K slightly decreased killing time against bacteria because of the extra net charge and hydrophobic properties.
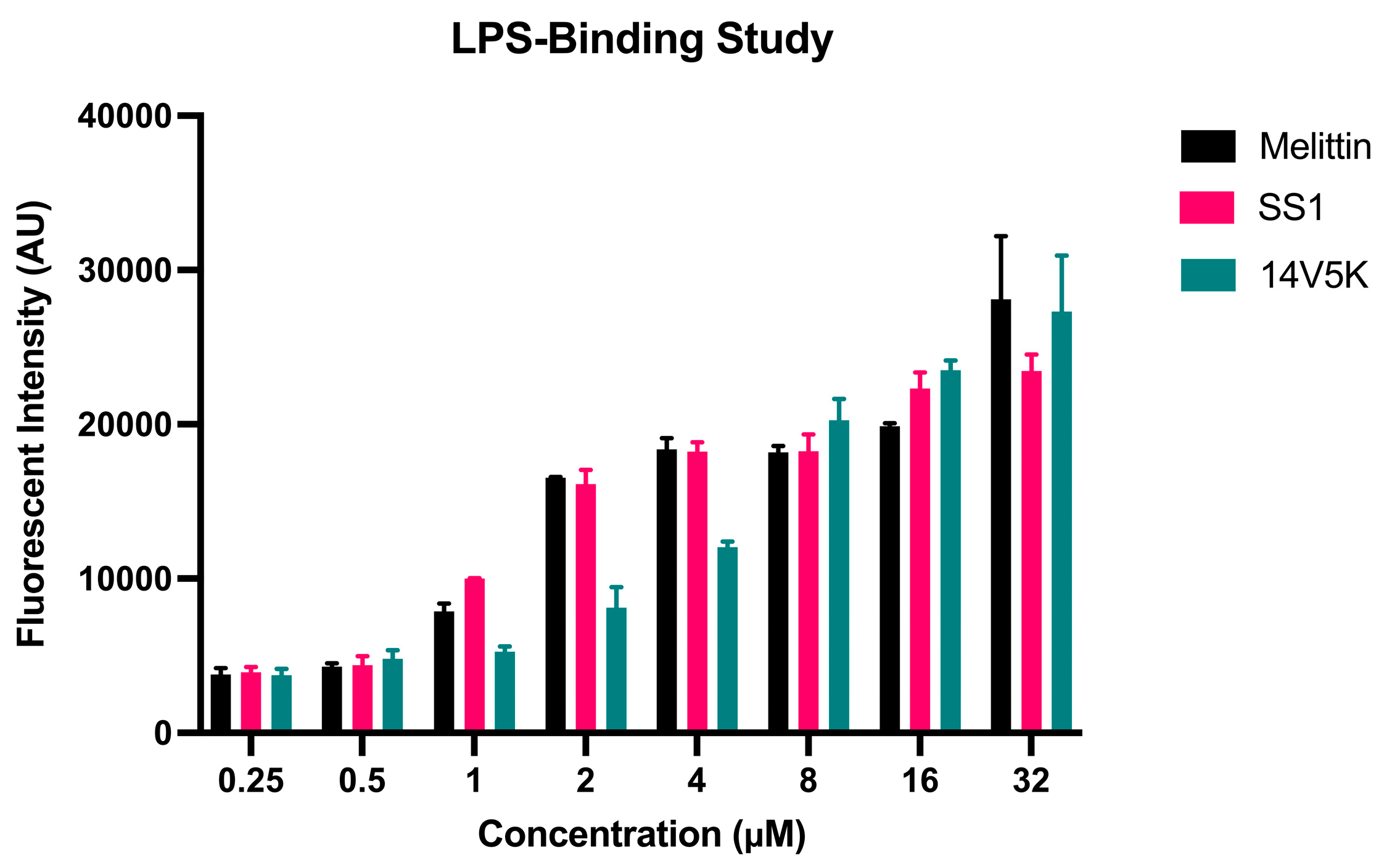
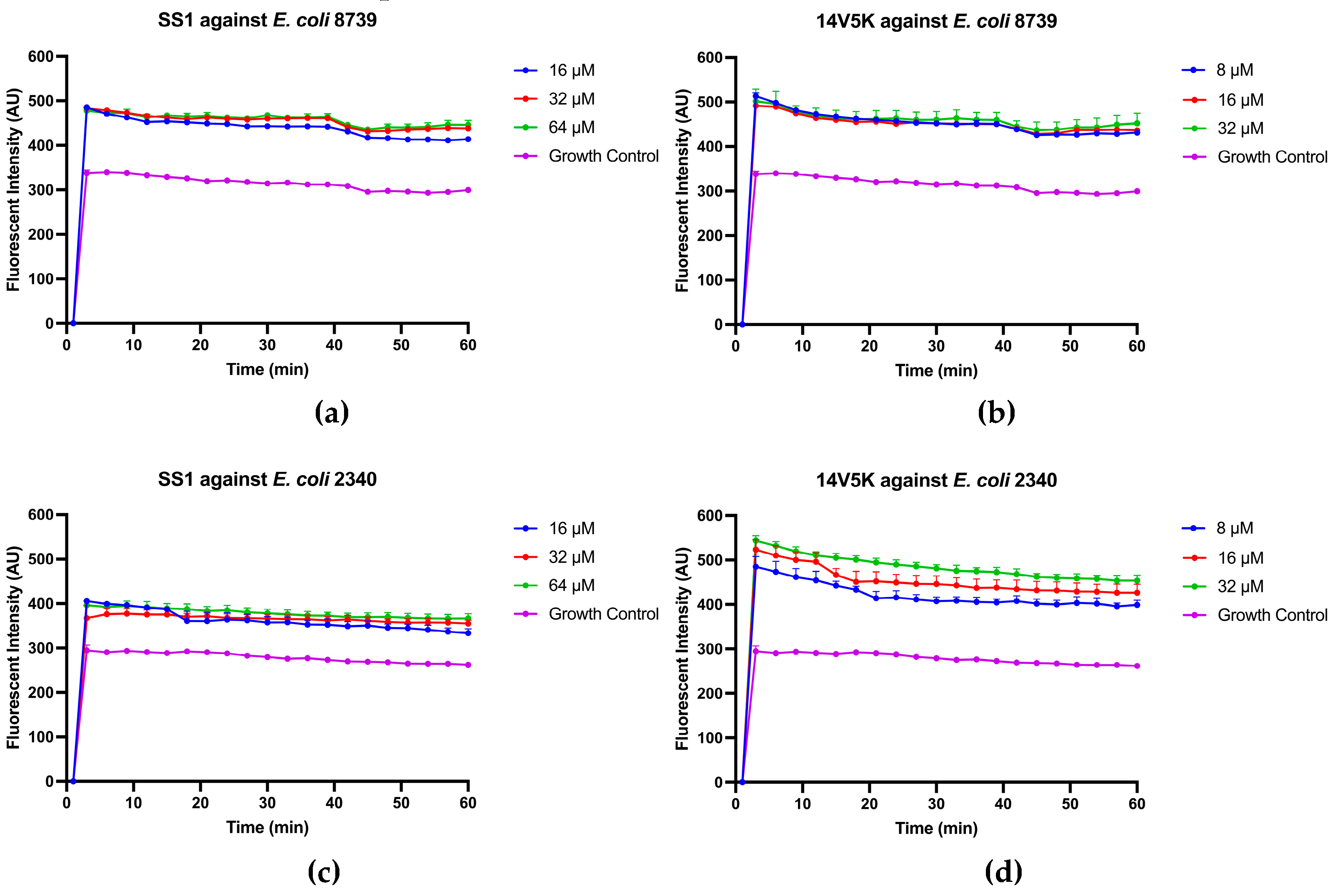
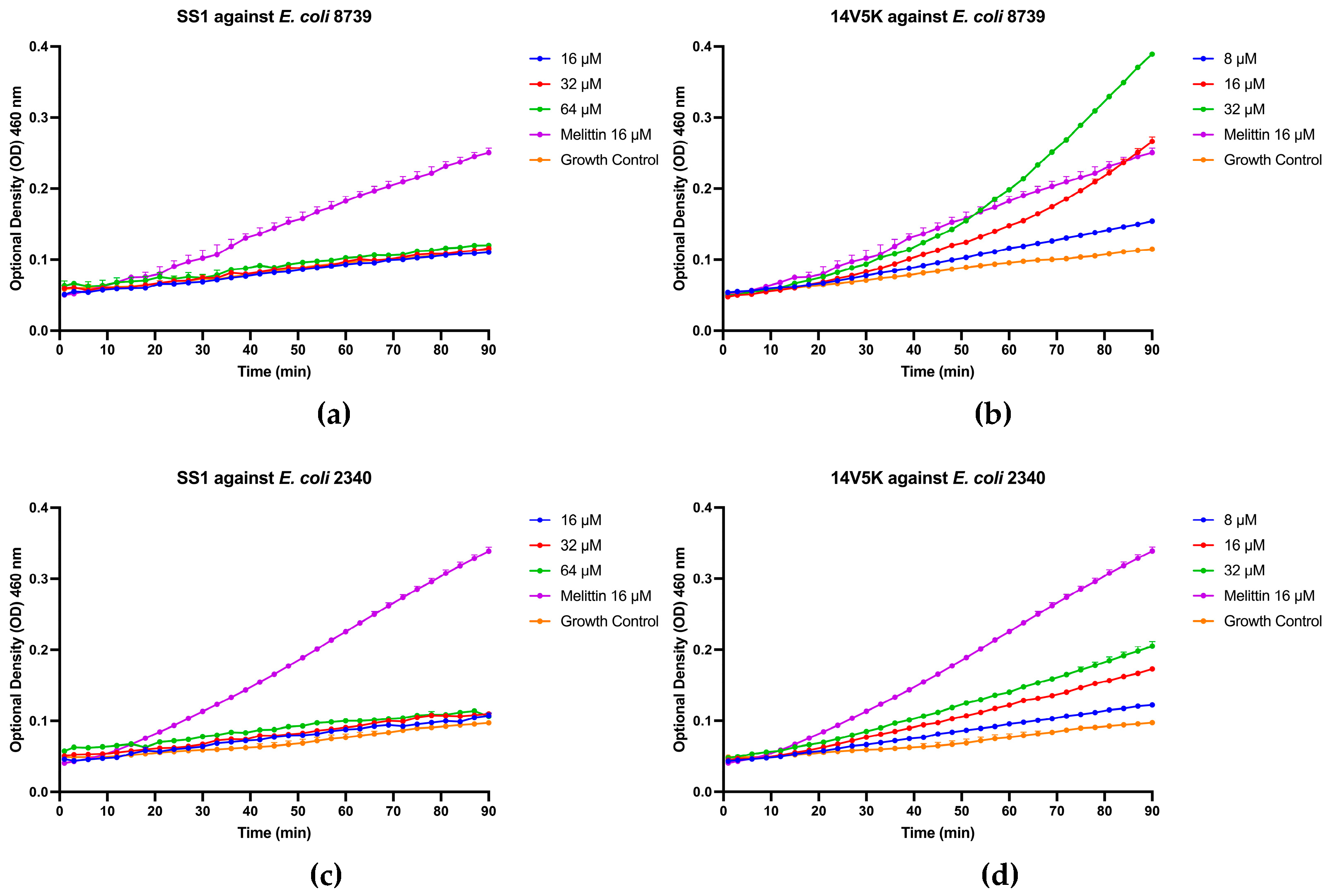
The initial reaction between peptides with positive net charges and the cell membrane occurred with the negatively charged components of the membrane. A fluorescence-based displacement assay was conducted to gauge the specificity of the binding of the peptides to LPS. As anticipated, the antimicrobial peptide SS1 exhibited comparable levels of efficiency in compulsory LPS as melittin. This well-known membrane-targeting peptide could swiftly induce pore formation in bacterial cell membranes and result in cytoplasm leakage [
18]. Once peptides form aggregates on the surface of bacteria and reach a certain concentration threshold, they could traverse the cell wall, thereby destabilising the cell membrane [
19]. However, 14V5K showed weaker LPS-binding ability at MIC concentrations. Therefore, the present study investigated the immediate impacts of peptides on both the external and internal membranes of
E. coli. The alterations in the fluorescence of NPN and the absorbance of ONPG, as observed in the presence of SS1 and 14V5K, provided initial evidence supporting the notion that these peptides illustrate an analogous process of bacterial membrane penetration to melittin, a widely explored antimicrobial peptide known for its membrane-damaging properties [
20].
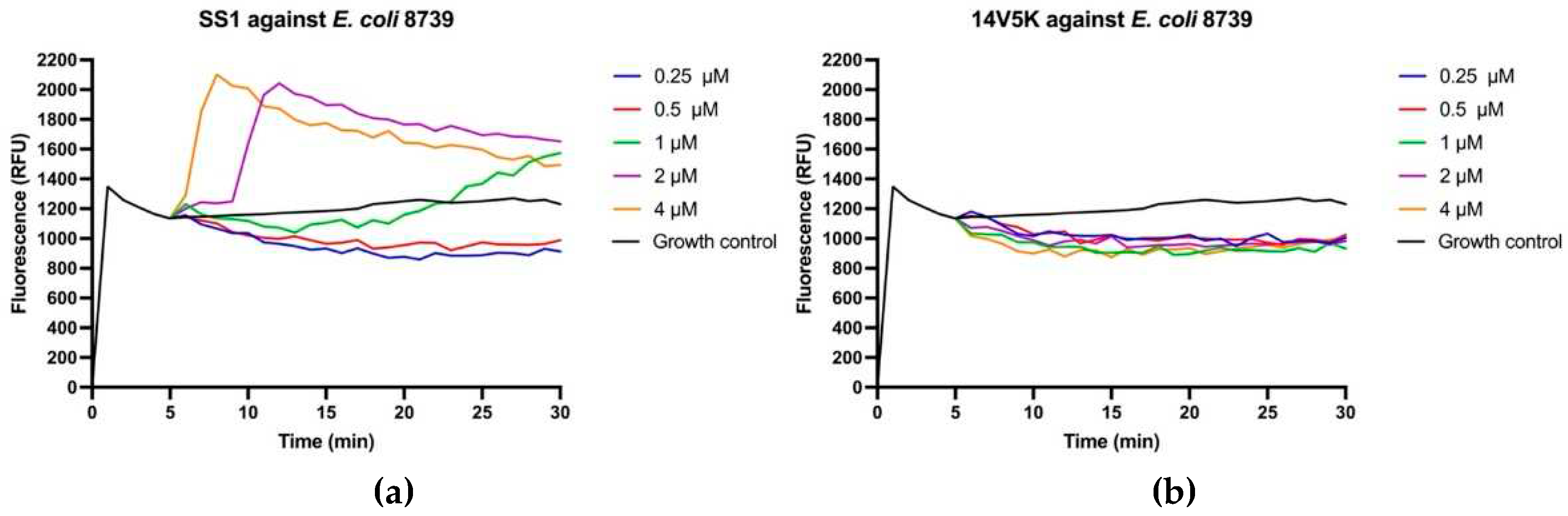
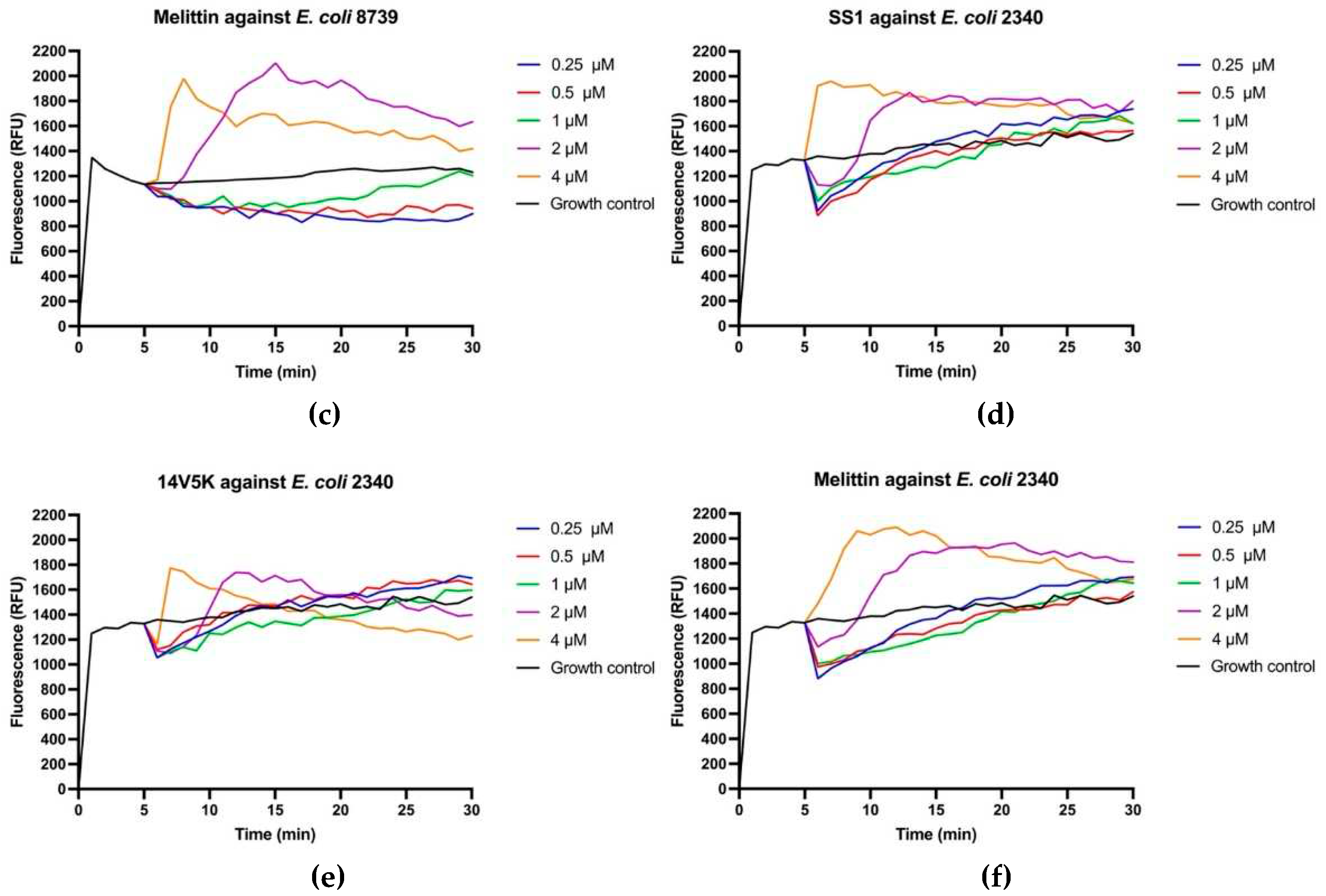
The perturbation of the inner membrane through peptides was investigated utilising diSC
3-5, a probe dependent on membrane potential. According to the results in
Figure 8, peptides showed fluctuant membrane potentials. The disruption of the cytoplasmic membrane potential led to the dissipation of the proton-motivated force, which hindered the transfer of electrons along the breathing pathway and consequently reduced the synthesis of ATP. This reduction in ATP synthesis was crucial for the movement of bacteria that relied on flagella [
11].
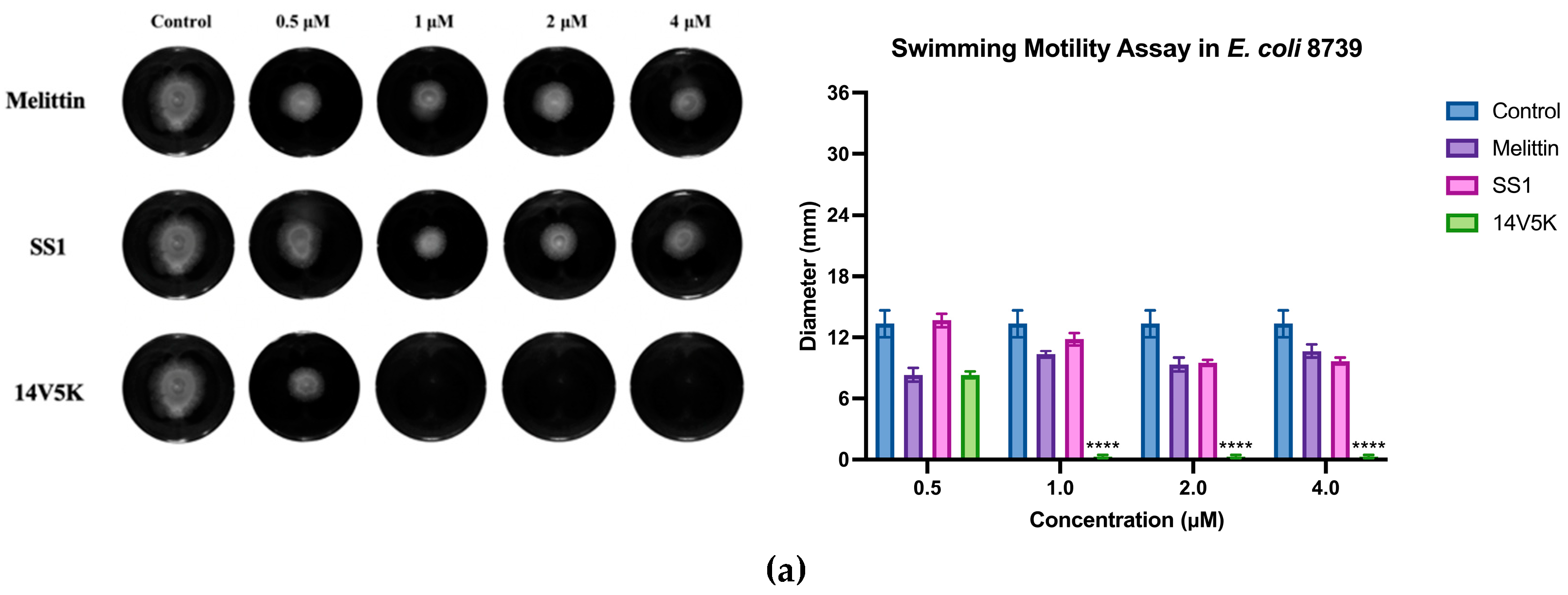
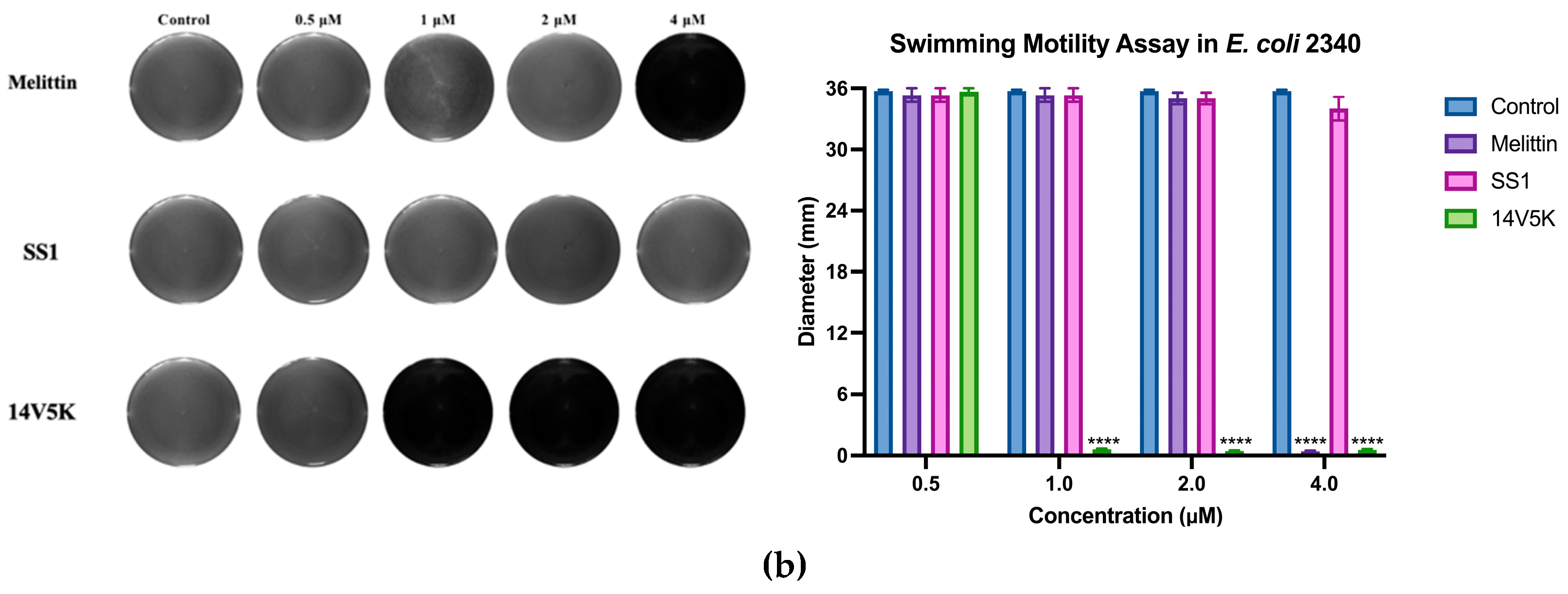
E. coli 8739 and 2340 were used to measure because of the existing flagellum. The study examined the consequences of peptides on bacterial swimming motion on swim plates with low viscosity. The peptide 14V5K demonstrated a more pronounced effect on the reduction of bacterial swimming diameter than that of melittin, whereas SS1 showed a greater capacity for membrane depolarization. Compared to 14V5K, the lower outer membrane binding ability and penetration effect of SS1 could be responsible for the less effective suppression of bacterial swimming motility. This hypothesis was supported by previous research [
21]. The belief that the electrostatic attraction between AMPs and bacterial surfaces plays a crucial role in the effectiveness of AMPs is based on the notion that it allows AMPs to reach the necessary concentrations required for disrupting the bacterial membrane. In summary,
E. coli 8739 and 2340 were killed by SS1 and 14V5K using the membrane-targeting mechanism.
Generally, SS1 and its analogues showed notable antiproliferative effects against both H838 and H460 lung cancer cell lines as displayed in Table 8. However, the antiproliferative activity of L2V was better than its antimicrobial activity. This was based on the membrane-targeting mechanism. A component of the membrane surface, phosphatidylserine (PS), has a higher affinity for L2V than other peptides. In cancer cells, PS is often exposed on the outer membrane leading to the development of a negative surface charge. This charge is associated with a lower pH in the surrounding environment. Membranolysis by AMPs and their selective mode of action on tumour cells can be attributed to the increased anionic nature of the cytoplasmic membrane of these cells. Another suspicion was that L2V had similar high hydrophobicity and hydrophobic moments, some of the forces required to disrupt cell membranes.
SS1 and its analogues showed cytotoxicity mostly at high concentrations in the haemolysis assays, but the IC
50 values of several peptides against HaCat cells were at low concentration levels. Meanwhile, the results of the salt stability assay supplied in
Table 7 revealed that SS1 lost antimicrobial activity partly in the environment with salts, while the salt stability of 14V5K was better than that of SS1. When a peptide bonded to red blood cells (RBCs), the activity of Na
+-K
+-ATPase on the surface of RBCs was inhibited [
22]. The imbalance of intracellular potassium ion concentration led to abnormal ion exchange between the outside and inside of the membrane. In addition, the positively charged region of the peptide interfered with the transport function of anion channels in the erythrocyte and changed the osmotic pressure of the erythrocyte membrane, causing the erythrocyte to swell and lyse. However, peptides may lose activity when potassium ions are low and may then show low cytotoxicity to RBCs. Therefore, the evidence for peptides with low cytotoxicity may be explained by their poor displacement capacity for salts.