Submitted:
02 August 2023
Posted:
03 August 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
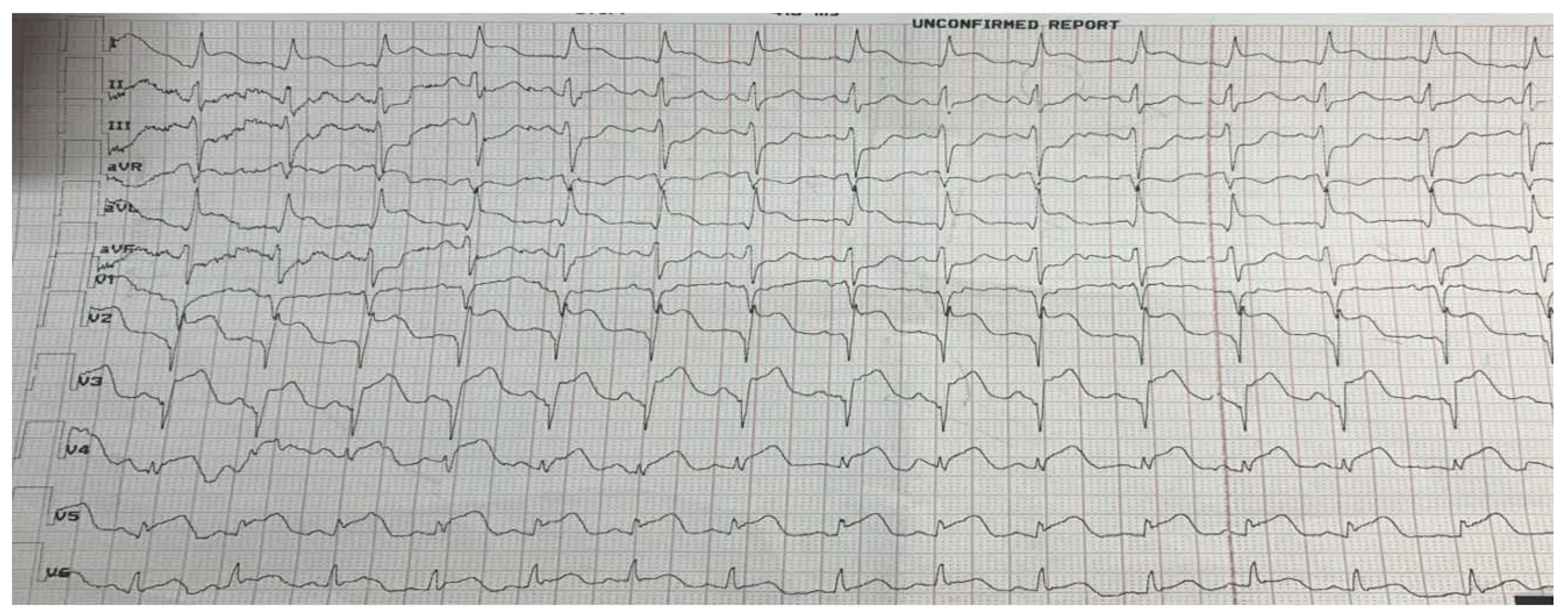
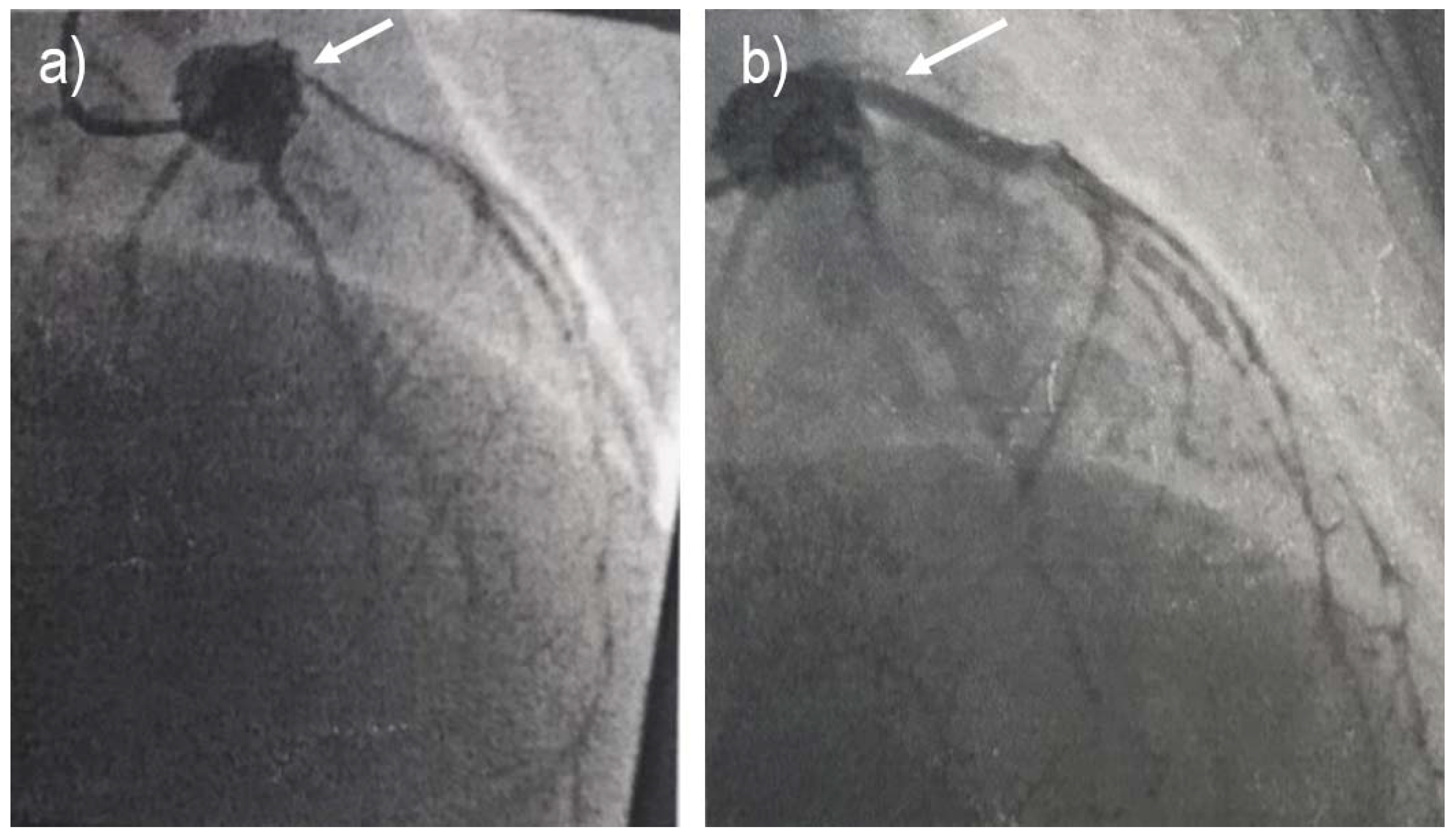
2. Case report
3. Discussion
4. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Regitz-Zagrosek, V.; Roos-Hesselink, J.W.; Bauersachs, J.; Blomström-Lundqvist, C.; Cífková, R.; De Bonis, M.; Iung, B.; Johnson, M.R.; Kintscher, U.; Kranke, P.; et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. Eur Heart J. 2018, 39(34), 3165–241. [Google Scholar] [CrossRef] [PubMed]
- Rossini, R.; Musumeci, G.; Visconti, L.O.; Bramucci, E.; Castiglioni, B.; De Servi, S.; Lettieri, C.; Lettino, M.; Piccaluga, E.; Savonitto, S.; et al. Perioperative management of antiplatelet therapy in patients with coronary stents undergoing cardiac and non-cardiac surgery: a consensus document from Italian cardiological, surgical and anaesthesiological societies. EuroIntervention. 2014, 10(1), 38–46. [Google Scholar] [CrossRef] [PubMed]
- Battinelli, EM.; Marshall, A.; Connors, J.M. The role of thrombophilia in pregnancy. Thrombosis. 2013, 2013, 516420. [Google Scholar] [CrossRef] [PubMed]
- Ormesher, L.; Simcox, L.; Tower, C.; Greer, I.A. Management of inherited thrombophilia in pregnancy. Women's Health (Lond Engl). 2016; 12(4): 433–41. [CrossRef]
- Juan, P.; Stefano, G.; Antonella, S.; Albana, C. Platelets in pregnancy. J Prenat Med. 2011, 5(4), 90–2. [Google Scholar] [PubMed]
- Burke, N.; Flood, K.; Murray, A.; Cotter, B.; Dempsey, M.; Fay, L.; Dicker, P.; Geary, M.P.; Kenny, D.; Malone, F.D. Platelet reactivity changes significantly throughout all trimesters of pregnancy compared with the nonpregnant state: a prospective study. BJOG. 2013, 120(13), 1599–604. [Google Scholar] [CrossRef] [PubMed]
- Verbruggen, M.; Mannaerts, D.; Muys, J.; Jacquemyn, Y. Use of ticagrelor in human pregnancy, the first experience. BMJ Case Rep. 2015; 2015, bcr2015212217. [CrossRef]
- Argentiero, D.; Savonitto, S.; D'Andrea, P.; Iacovelli, F. Ticagrelor and tirofiban in pregnancy and delivery: beyond labels. J Thromb Thrombolysis. 2020; 49(1), 145–8. [CrossRef]
- Tello-Montoliu, A.; Seecheran, N.A.; Angiolillo, D.J. Successful pregnancy and delivery on prasugrel treatment: considerations for the use of dual antiplatelet therapy during pregnancy in clinical practice. J Thromb Thrombolysis. 2013; 36(3), 348–51. [CrossRef]
- Pop, R.; Cebula, H.; Lambert, A.; Mihoc, D.; Manisor, M.; Cindea, C.; Hasiu, A.; Deruelle, P.; Chibbaro, S.; Mertz, L.; et al. Treatment with flow diverter stent during pregnancy. Neuroradiology. 2020, 62(11), 1507–10. [Google Scholar] [CrossRef] [PubMed]
- Reilly, C.R.; Cuesta-Fernandez, A.; Kayaleh, O.R. Successful gestation and delivery using clopidogrel for secondary stroke prophylaxis: a case report and literature review. Arch Gynecol Obstet. 2014; 290(3), 591–4. [CrossRef]
- De Santis, M.; De Luca, C.; Mappa, I.; Cesari, E.; Mazza, A.; Quattrocchi, T.; Caruso, A. Clopidogrel treatment during pregnancy: a case report and a review of literature. Intern Med. 2011, 50(16), 1769–73. [Google Scholar] [CrossRef] [PubMed]
- Klinzing, P.; Markert, U.R.; Liesaus, K.; Peiker, G. Case report: successful pregnancy and delivery after myocardial infarction and essential thrombocythemia treated with clopidogrel. Clin Exp Obstet Gynecol. 2001, 28(4), 215–6. [Google Scholar] [PubMed]
- Santiago-Díaz, P.; Arrebola-Moreno, A.L.; Ramírez-Hernández, J.A.; Melgares-Moreno, R. Platelet antiaggregants in pregnancy. Rev Esp Cardiol (Engl Ed). 2009, 62(10), 1197–8. [CrossRef]
- Al-Aqeedi, R.F.; Al-Nabti. A.D. Drug-eluting stent implantation for acute myocardial infarction during pregnancy with use of glycoprotein IIb/IIIa inhibitor, aspirin and clopidogrel. J Invasive Cardiol. 2008, 20(5), E146-9.
- Valgimigli, M.; Bueno, H.; Byrne, R.A.; Collet, J.P.; Costa, F.; Jeppsson, A.; Jüni, P.; Kastrati, A.; Kolh, P.; Mauri, L,; et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS. Eur Heart J. 2018, 39(3), 213–60. 3. [CrossRef]
- Hansson, E.C.; Malm, C.J.; Hesse, C.; Hornestam, B.; Dellborg, M.; Rexius, H.; Jeppsson, A. Platelet function recovery after ticagrelor withdrawal in patients awaiting urgent coronary surgery. Eur J Cardiothorac Surg. 2017, 51(4), 633–7. [Google Scholar] [CrossRef] [PubMed]
- Malm, C.J.; Hansson, E.C.; Åkesson, J.; Andersson, M.; Hesse, C.; Shams Hakimi, C.; Jeppsson, A. Preoperative platelet function predicts perioperative bleeding complications in ticagrelor-treated cardiac surgery patients: a prospective observational study. Br J Anaesth. 2016, 117(3), 309–15. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, M.; Baryshnikova, E.; Soro, G.; Ballotta, A.; De Benedetti, D.; Conti, D. Multiple electrode whole-blood aggregometry and bleeding in cardiac surgery patients receiving thienopyridines. Ann Thorac Surg. 2011, 91(1), 123–9. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, S.; Woźniak, K.; Hryniewiecki, T.; Kruk, M.; Różański, J.; Kuśmierczyk, M. The predictive value of multiple electrode platelet aggregometry for postoperative bleeding complications in patients undergoing coronary artery bypass graft surgery. Kardiochir Torakochirurgia Pol. 2016; 13(1), 3–9. [CrossRef]
- Aradi, D.; Komócsi, A.; Price, M.J.; Cuisset, T.; Ari, H.; Hazarbasanov, D.; Trenk, D.; Sibbing, D.; Valgimigli, M.; Bonello, L. Efficacy and safety of intensified antiplatelet therapy on the basis of platelet reactivity testing in patients after percutaneous coronary intervention: systematic review and meta-analysis. Int J Cardiol. 2013, 167(5), 2140–8. [Google Scholar] [CrossRef] [PubMed]
- Lenk, E.; Spannagl, M. Platelet function testing-guided antiplatelet therapy. EJIFCC. 2013, 24(3), 90–6. [Google Scholar] [PubMed]
- Mazzeffi, M.A.; Lee, K.; Taylor, B.; Tanaka, K.A. Perioperative management and monitoring of antiplatelet agents: a focused review on aspirin and P2Y12 inhibitors. Korean J Anesthesiol. 2017, 70(4), 379–89. [Google Scholar] [CrossRef] [PubMed]
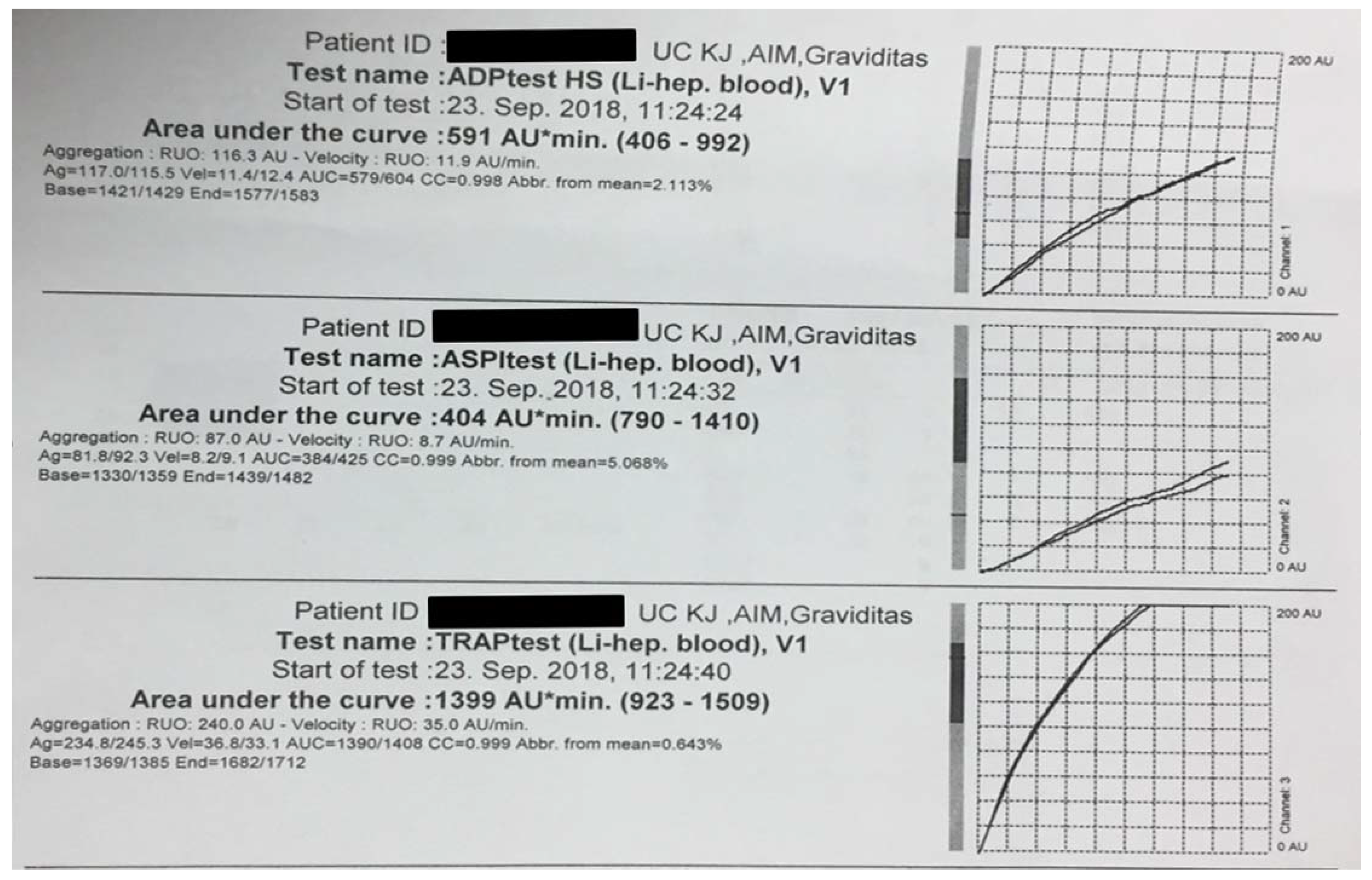
| Hospitalization day | ADP inhibitor drug | ADP test HS (AU*min) |
ASPI test (AU*min) |
TRAP test (AU*min) |
| Day 1 | ticagrelor | 591 | 404 | 1399 |
| Day 2 | clopidogrel | 1081 | 889 | 1984 |
| Day 3 | clopidogrel | 1263 | 1109 | 1718 |
| Day 4 | ticagrelor | 567 | 1320 | 1807 |
| Day 5 | ticagrelor | 535 | 646 | 1679 |
| Day 6 | ticagrelor | 373 | 373 | 1761 |
| Day 9 | ticagrelor | 315 | 512 | 1352 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




