Submitted:
01 August 2023
Posted:
02 August 2023
You are already at the latest version
Abstract
Keywords:
Introduction
On the origin of Mycolactone-Producing Mycobacteria (MPM) species assignment
History of BU diagnosis and M. ulcerans DNA PCR testing
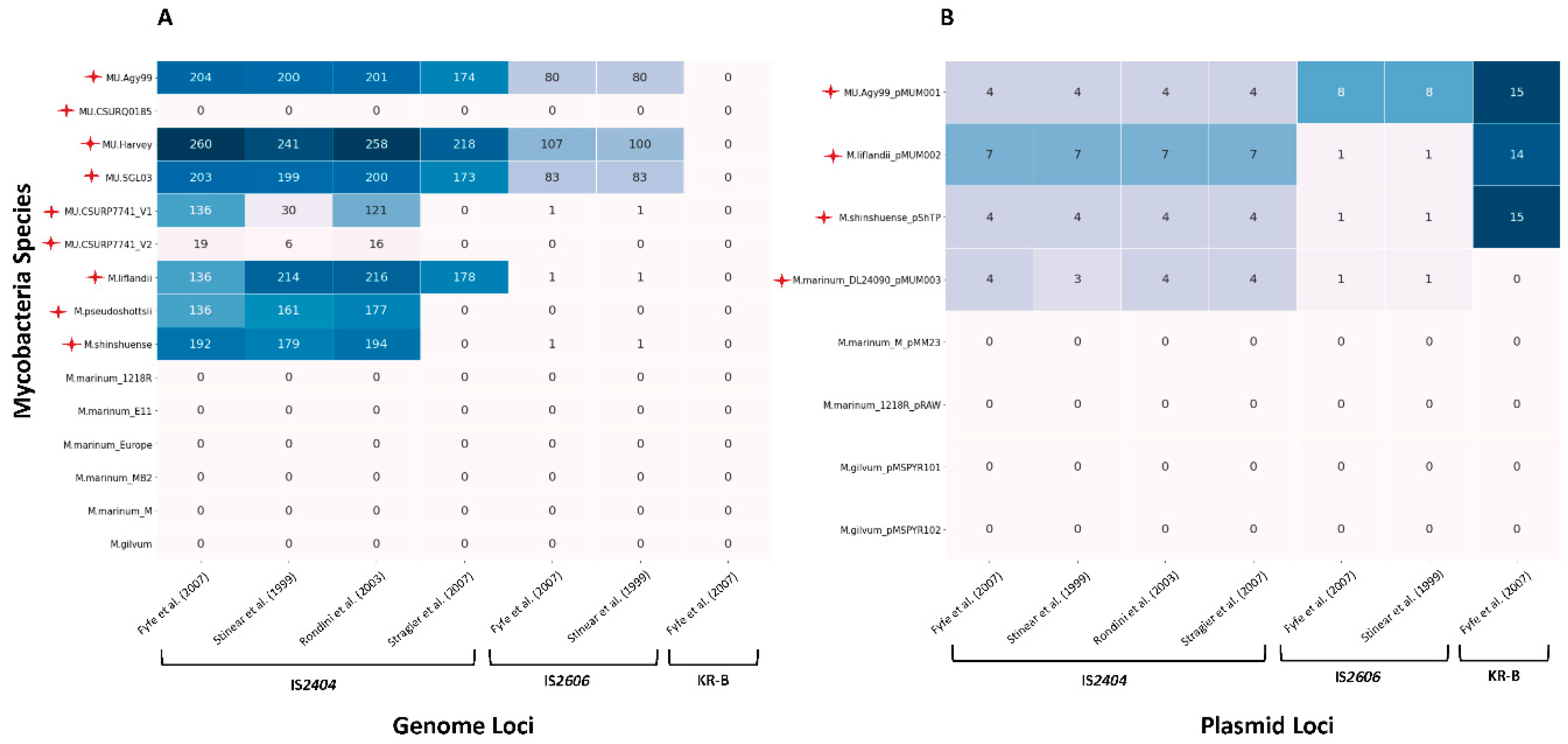
Lack of specificity of (q)PCR-based tests for M. ulcerans variant detection
Implementation of MIRU-VNTR to assess genotypic diversity
Implication for the diagnosis and treatment of BU
Conclusions
Material & Methods
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ablordey, A., Amissah, D.A., Aboagye, I.F., Hatano, B., Yamazaki, T., Sata, T., et al. (2012). Detection of Mycobacterium ulcerans by the loop mediated isothermal amplification method. PLoS Neglect. Trop. Dis 6, e1590. [CrossRef]
- Ablordey, A., Fonteyne, P., Stragier, P., Vandamme, P., and Portaels, F. (2007). Identification of a new variable number tandem repeat locus in Mycobacterium ulcerans for potential strain discrimination among African isolates. Clin. Microbiol. Infect. 13, 734–736. [CrossRef]
- Ablordey, A., Swings, J., Hubans, C., Chemlal, K., Locht, C., Portaels, F., et al. (2005). Multilocus Variable-Number Tandem Repeat Typing of Mycobacterium ulcerans. J. Clin. Microbiol. 43, 1546–1551. [CrossRef]
- Ahlstrom, C., Barkeman, H.W., Stevenson, K., Zadoks, R.N., Biek, R., Kao, R., et al. (2015). Limitations of variable number of tandem repeat typing identified through whole genome sequencing of Mycobacterium avium subsp. paratuberculosis on a national and herd level. BMC Genomics 16, 161. [CrossRef]
- Aubry, A., Jarlier, V., Escolano, S., Truffot-Pernot, C., and Cambau, E. (2000). Antibiotic susceptibility pattern of Mycobacterium marinum. Antimicrob. Agents Chemo. 44, 3133-3136. [CrossRef]
- Benbow, E., Kimbirauskas, M., McIntosh, R., Williamson, M.D., Quaye, H., Boakye, C., et al. (2014). Aquatic Macroinvertebrate Assemblages of Ghana, West Africa: Understanding the Ecology of a Neglected Tropical Disease. EcoHealth 11, 168–183. [CrossRef]
- Bonamonte, D., De Vito, D., Vestita, M., Delvecchio, S., Ranieri, L. D., Santantonio, et al. (2013). Aquarium-borne Mycobacterium marinum skin infection. Report of 15 cases and review of the literature. Europ. J. Dermatol. 23, 510–516.
- Buchanan, T.M.H, Nomaguchi, H., Anderson, D.C., Young, R.A., Gillis, T.P., Britton, W.J., et al. (1987). Characterization of antibofy-reactive epitopes on the 65-kilodalton protein of Mycobacterium leprae. Infect. Immun. 55, 1000–1003. [CrossRef]
- Bull, T.J, Sidi-Boumedine, K., McMinn, E.J., Stevenson, K., Pickup, R., and Hermon-Taylor, J. (2003). Mycobacterial interspersed repetitive units (MIRU) differentiate Mycobacterium avium subspecies paratuberculosis from other species of the Mycobacterium avium complex. Cell Probes 17, 157–164. [CrossRef]
- Camacho, C., Coulouris, G., Avagyan, V., Ma, N., Papadopoulos, J., Bealer, K. et al. (2009). BLAST+: architecture and applications. BMC bioinformatics 10, 1–9. [CrossRef]
- Chemlal, K., Huys, G., Fonteyne, P-A., Vincent, V., Lopez, G., Rigouts, J., et al. (2001a). Evaluation of PCR-restriction profile analysis and IS2404 restriction fragment length polymorphisms and amplified fragment length polymorphisms fingerprinting for identification and typing of Mycobacterium ulcerans and M. marinum. J. Clin. Microbiol. 39, 3272–3278. [CrossRef]
- Chemlal, K., De Ridder, K., Fonteyne, P-A., Meyers, W.M., Swings, J., and Portaels, F. (2001b). The use of IS2404 restriction fragment length polymorphisms suggests the diversity of Mycobacterium ulcerans from different geographical areas. Am. J. Trop. Med. Hyg. 64, 270–273.
- Combe, M., Couppié, P., Blaizot, R., Valentini, A., and Gozlan, R. E. (2020). Are all Buruli ulcers caused by Mycobacterium ulcerans? Br. J. Dermatol. 183, 968–970. [CrossRef]
- Combe, M., Velvin, C. J., Morris, A., Garchitorena, A., Carolan, K., Sanhueza, D., et al. (2017). Global and local environmental changes as drivers of Buruli ulcer emergence. Emerg. Microbes Infect. 6. [CrossRef]
- Demangel, C., Stinear, T. P., and Cole, S. T. (2009). Buruli ulcer: reductive evolution enhances pathogenicity of Mycobacterium ulcerans. Nat. Rev. Microbiol. 7, 50–60. [CrossRef]
- Diaz, D., Döbeli, H., Yeboah-Manu, D., Mensah-Quaino, E., Friedlein, A., Soder, N., et al. (2006). Use of the immunodominant 18-kiloDalton small heat shock protein as a serological marker for exposure to Mycobacterium ulcerans. Clin. Vaccine Immunol. 13, 1314–11321. [CrossRef]
- Dixit, P. and Kotra, L.P. (2007). Diseases caused by acid-fast bacteria. Eds S.J. Enna, D.B. Bylund,xPharm: the comprehensive pharmacology reference. Elsevier, pp. 1-5. [CrossRef]
- Doig, K. D., Holt, K. E., Fyfe, J. A. M., Lavender, C. J., Eddyani, M., Portaels, F., et al. (2012). On the origin of Mycobacterium ulcerans, the causative agent of Buruli ulcer. BMC Genomics 13. [CrossRef]
- Dong, J., Olano, J. P., McBride, J. W., and Walker, D. H. (2008). Emerging pathogens: Challenges and successes of molecular diagnostics. J. Mol. Diagnostics 10, 185–197. [CrossRef]
- Fyfe, J. A. M., Lavender, C. J., Johnson, P. D. R., Globan, M., Sievers, A., Azuolas, J., et al. (2007). Development and application of two multiplex real-time PCR assays for the detection of Mycobacterium ulcerans in clinical and environmental samples. Appl. Environ. Microbiol. 73, 4733–4740. [CrossRef]
- George, K.M. (1999). Mycolactone: a polyketide toxin from Mycobacterium ulcerans required for virulence. Science 283, 854-857. [CrossRef]
- Giles-Vernick, T., Owona-Ntsama, J., Landier, J., and Eyangoh, S. (2015). The puzzle of Buruli ulcer transmission, ethno-ecological history and the end of “love” in the Akonolinga district, Cameroon. Soc. Sci. Med. 129, 20–27. [CrossRef]
- Griffith, D.E. (2007). on behalf of the ATS Mycobacterial Diseases Subcommittee: An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 175, 367–415.
- Guerrero, C., Bernasconi, C., Burki, D., Bodmer, T., and Talenti, A. (1995). A novel insertion element from Mycobacteriul avium, IS1245, is a specific target for analysis of strain relatedness. J. Clin. Microbiol. 33, 304–307. [CrossRef]
- Guillemi, E. C., Tomassone, L., and Farber, M. D. (2015). Tick-borne Rickettsiales: Molecular tools for the study of an emergent group of pathogens. J. Microbiol. Methods 119, 87–97. [CrossRef]
- Hance, A.J., Grandchamp, B., Lévy-Frébault, V., Lecossier, D., Rauzier, J., Bocart, D., and Gicquel, B. (1989). Detection and identification of mycobacteria by amplification pf mycobacterial DNA. Molec. Microbiol. 3, 843–849. [CrossRef]
- Hayman, J. (1991). Postulated epidemiology of Mycobacterium ulcerans infection. Int. J. Epidemiol. 20, 1093–1098. [CrossRef]
- Hilty, M., Käser, M., Zinsstag, J., Stinear, T., and Pluschke, G. (2007). Analysis of the Mycobacterium ulcerans genome sequence reveals new loci for variable number tandem repeats (VNTR) typing. Microbiology 153, 1483–1487. [CrossRef]
- Hilty, M., Yeboah-Manu, D., Boakye, D., Mensah-Quainoo, E., Rondini, S., Schelling, E., et al. (2006). Genetic Diversity in Mycobacterium ulcerans Isolates from Ghana Revealed by a Newly Identified Locus Containing a Variable Number of Tandem Repeats. J. Bacteriol. 188, 1462–1465. [CrossRef]
- Hofer, M., Hirschel, B., Kirschner, P., Beghetti, M., Kaelin, A., Siegrist, C-.A., et al. (1993). Brief report: dissaminated osteomyelitis from Mycobacterium ulcerans after snakebite. N. Engl. J. Med. 328, 1007–1009. [CrossRef]
- Jackson, K., Edwards, R., Leslie, D.E., and Hayman, J. (1995). Molecular method for typing Mycobacterium ulcerans. J. Clin. Microbiol. 33, 2250–2253. [CrossRef]
- Johnson, M.G., and Stout, J.E. (2015). Twenty-eight cases of Mycobacterium marinum infection: retrospective case series and literature review. Infection 43, 655–662 (2015). [CrossRef]
- Jones, K., Patel, N., Levy, M., Storeygard, A., Balk, D., Gittleman, J., et al. (2008). Global trends in emerging infectious diseases. Nature 451, 990–993. [CrossRef]
- Kim, H., Kim, S. H., Shim, T. S., Kim, M. N., Bai, G. H., Park, Y. G., et al. (2005). Differentiation of Mycobacterium species by analysis of the heat-shock protein 65 gene (hsp65). Int. J. Syst. Evol. Microbiol. 55, 1649–1656. [CrossRef]
- Lavender, C. J., Stinear, T. P., Johnson, P. D. R., Azuolas, J., Benbow, M. E., Wallace, J. R., et al. (2008). Evaluation of VNTR typing for the identification of Mycobacterium ulcerans in environmental samples from Victoria, Australia. FEMS Microbiol. Lett. 287, 250–255. [CrossRef]
- Le Turnier, P., and Epelboin, L. (2019). Mise au point sur la leptospirose. La Revue de Médecine Interne 40, 306–312. [CrossRef]
- MacCULLUM, P. (1948). A new mycobacterial infection in man; clinical aspects. J. Pathol. Bacteriol. 60, 93–102.
- Marston, B.J., Diallo, M.O., Horsburgh, C.R., Diomande, I., Saki, M.Z., Kanga, J.M., et al. (1995). Emergence of Buruli ulcer disease in the Daola region of Côte d’Ivoire. Am. J. Tropl. Med. Hyg. 52, 219–224. [CrossRef]
- Matsumoto, M., Machida, Y., Kanemaru, M., Yamamoto, M., Sano, M. and Kato, G. (2022). Infection with Mycobacterium pseudoshottsii in cultured yellowtail Seriola quinqueradiata in Owase Bay, Japan. Fish Pathol. 57, 35-40. [CrossRef]
- McIntosh, M., Williamson, H., Benbow, E., Kimbirauskas, R., Quaye, C., Boakye, D., et al. (2014).Associations between Mycobacterium ulcerans and aquatic plant communities of West Africa: implications for Buruli ulcer disease. EcoHealth 11, 184-196. [CrossRef]
- Merritt, R. W., Walker, E. D., Small, P. L. C., Wallace, J. R., Johnson, P. D. R., Benbow, M. E., et al. (2010). Ecology and Transmission of Buruli Ulcer Disease: A Systematic Review. PLoS Negl. Trop. Dis. 4, e911. [CrossRef]
- Meyers, W.M. (1994). Mycobacterial infections of the skin. In G. Seifert (ed.) Trop. Dermatol. Springer-Verlag, Heidelberg, Germany.
- Mve-Obiang, A., Lee, R. E., Umstot, E. S., Trott, K. A., Grammer, T. C., Parker, J. M., et al. (2005). A newly discovered mycobacterial pathogen isolated from laboratory colonies of Xenopus species with lethal infections produces a novel form of mycolactone, the Mycobacterium ulcerans macrolide toxin. Infect. Immun. 73, 3307–3312. [CrossRef]
- Narh, C. A., Mosi, L., Quaye, C., Dassi, C., Konan, D. O., Tay, S. C. K., et al. (2015). Source Tracking Mycobacterium ulcerans Infections in the Ashanti Region, Ghana. PLoS Negl. Trop. Dis. 9, 1–18. [CrossRef]
- Narh, C.A., Mosi, L., Quaye, C., Tay, S.C.K., Bonfoh, B., and de Souza, D.K. (2014). Genotyping Tools for Mycobacterium ulcerans Drawbacks and Future Prospects. Mycobact. Dis. 04. [CrossRef]
- Nakanaga, K., Hoshino, Y., Yotsu, R. R., Makino, M., and Ishii, N. (2011). Nineteen cases of Buruli ulcer diagnosed in Japan from 1980 to 2010. J. clin. microbiol. 49, 3829–3836. [CrossRef]
- Nakanaga, K., Ishii, N., Suzuki, K., Tanigawa, K., Goto, M., Okabe, T., et al. (2007). “Mycobacterium ulcerans subsp. shinshuense” isolated from a skin ulcer lesion: Identification based on 16S rRNA gene sequencing. J. Clin. Microbiol. 45, 3840–3843. [CrossRef]
- Nguetta, A., Coulibaly, N.D., Kouamé-Elogne, N.C., Acquah, K.J.R., Amon, A.C., Kouamé, K., et al. (2018). Phenotypic and Genotypic Characterization of Mycobacteria Isolates from Buruli Ulcer Suspected Patients Reveals the Involvement of Several Mycobacteria in Chronic Skin Lesions. Am. J. Microbiol. Res. 6, 79–87. [CrossRef]
- Nigou, J., Gilleron, M., and Puzo, G. (2003). Lipoarabinomannans: from structure to biosynthesis. Biochimie 85, 153-166. [CrossRef]
- Njiru, Z.K., Yeboah-Manu, D., Stinear, T.P., and Fyfe, J.A.M. (2012). Rapid and sensitive detection of Mycobacterium ulcerans by use of a loop-mediated isothermal amplification test. J. Clin. Microbiol. 50, 1737-1741. [CrossRef]
- Ohtsuka, M., Kikuchi, N., Yamamoto, T., Suzutani, T., Nakanaga, K., Suzuki, K., et al. (2014). Buruli ulcer caused by Mycobacterium ulcerans subsp shinshuense. JAMA Dermatol. 150, 64–67. [CrossRef]
- Picardeau, M., Bull, T.J., Vincent, V. (1997). Identification and characterization of IS-like elements in Mycobacterium gordonae. FEMS Microbiol Lett. 154, 95–102. [CrossRef]
- Pidot, S. J., Asiedu, K., Käser, M., Fyfe, J. a M., and Stinear, T. P. (2010). Mycobacterium ulcerans and other Mycolactone-producing mycobacteria should be considered a single species. PLoS Negl. Trop. Dis. 4, 6–8. [CrossRef]
- Portaels, F., Aguiar, J., Fissette, K., Fonteyne, P.A., de Beenhouwer, H., de Rijk, P., et al. (1997). Direct detection and identification of Mycobacterium ulcerans in clinical speciemns by PCR and oligonucleotide-specific capture plate hybridization. J. Clin. Microbiol. 35, 1097-1100.
- Portaels, F., Fonteyne, P. A., De Beenhouwer, H., De Rijk, P., Guédénon, A., Hayman, J., et al. (1996). Variability in 3′ end of 16S rRNA sequence of Mycobacterium ulcerans is related to geographic origin of isolates. J. Clin. Microbiol. 34, 962–965. [CrossRef]
- Radomski, N., Kreitmann, L., Mcintosh, F., and Behr, M. A. (2013). The Critical Role of DNA Extraction for Detection of Mycobacteria in Tissues. PLoS ONE 8, 1–8. [CrossRef]
- Ranger, B. S., Mahrous, E. A., Mosi, L., Adusumilli, S., Lee, R. E., Colorni, A., et al. (2006). Globally distributed mycobacterial fish pathogens produce a novel plasmid-encoded toxic macrolide, mycolactone F. Infect. Immun. 74, 6037–6045. [CrossRef]
- Reynaud, Y., Millet, J., Couvin, D., Rastogi, N., and Brown, C. (2015). Heterogeneity among Mycobacterium ulcerans from French Guiana Revealed by Multilocus Variable Number Tandem Repeat Analysis ( MLVA ). 1–9. [CrossRef]
- Rezazadeh, E., Moazeni, maryam, and Sabokbar, A. (2016). Use of cost effective and rapid molecular tools for identification of Candida species, opportunistic pathogens. Curr. Med. Mycol. 2, 1–4. [CrossRef]
- Rhodes, M. W., Kator, H., McNabb, A., Deshayes, C., Reyrat, J. M., Brown-Elliott, B. A., et al. (2005). Mycobacterium pseudoshottsii sp. nov., a slowly growing chromogenic species isolated from Chesapeake Bay striped bass (Morone saxatilis). Int. J. Syst. Evol. Microbiol. 55, 1139–1147. [CrossRef]
- Rondini, S., Mensah-Quainoo, E., Troll, H., Bodmer, T., and Pluschke, G. (2003). Development and application of real-time PCR assay for quantification of Mycobacterium ulcerans DNA. J. Clin. Microbiol. 41, 4231–4237. [CrossRef]
- Ross, B. C., Johnson, P. D. R., Oppedisano, F., Marino, L., Sievers, a., Stinear, T., et al. (1997a). Detection of Mycobacterium ulcerans in environmental samples during an outbreak of ulcerative disease. Appl. Environ. Microbiol. 63, 4135–4138.
- Ross, B. C., Marino, L., Oppedisano, F., Edwards, R., Robins-Browne, R. M., and Johnson, P. D. R. (1997b). Development of a PCR assay for rapid diagnosis of Mycobacterium ulcerans infection. J. Clin. Microbiol. 35, 1696–1700. [CrossRef]
- Saad, J., Combe, M., Hammoudi, N., Couppié, P., Blaizot, R., Jedir, F. et al. (2019). Whole-genome sequence of Mycobacterium ulcerans CSURP7741, a French Guianan clinical isolate. Microbiol. Resour. Announc. 8, e00215-19. [CrossRef]
- Shinoda, N., Nakamura, H., and Watanabe, M. (2016). Detection of Mycobacterium ulcerans by real-time PCR with improved primers. Trop. Med. Health 44. [CrossRef]
- Stanford, J.L. and Gunthorpe, W.J. (1971). A study of some fast-growing scotochromogenic mycobacteria including species descritpions of Mycobacterium gilvum (new species) and Mycobacterium duvalii (new species). Br. J. Exp. Pathol. 52, 627-673.
- Stinear, T. P., Davies, J. K., Jenkin, G. A., Hayman, J. A., Oppedisano, F., and Johnson, P. D. R. (2000b). Identification of Mycobacterium ulcerans in the environment from regions in southeast Australia in which it is endemic with sequence capture-PCR. Appl. Environ. Microbiol. 66, 3206–3213. [CrossRef]
- Stinear, T., Davies, J. K., Jenkin, G. A., Portaels, F., Ross, B. C., Oppedisano, F., et al. (2000a). A simple PCR method for rapid genotype analysis of Mycobacterium ulcerans. J. Clin. Microbiol. 38, 1482–1487. [CrossRef]
- Stinear, T.P., Pryor, M.J., Porter, J.L., and Cole, S.T. (2005). Functional analysis and annotation of the virulence plasmid pMUM001 from Mycobacterium ulcerans. Microbiol. 151, 683-692. [CrossRef]
- Stinear, T., Ross, B. C., Davies, J. K., Marino, L., Robins-Browne, R. M., Oppedisano, F., et al. (1999). Identification and characterization of IS2404 and IS2606: Two distinct repeated sequences for detection of Mycobacterium ulcerans by PCR. J. Clin. Microbiol. 37, 1018–1023. [CrossRef]
- Stinear, T.P., Seemann, T., Pidot, S., Frigui, W., Reysset, G., Garnier, T., et al. (2007). Reductive evolution and niche adaptation inferred from the genome of Mycobacterium ulcerans, the causative agent of Buruli ulcer. Genome Research 17, 192-200. [CrossRef]
- Stragier, P., Ablordey, A., Durnez, L., and Portaels, F. (2007). VNTR analysis differentiates Mycobacterium ulcerans and IS2404 positive mycobacteria. Syst. Appl. Microbiol. 30, 525–530. [CrossRef]
- Stragier, P., Ablordey, A., and Meyers, W. M. (2005). Using Mycobacterial Interspersed Repetitive Units. 187, 1639–1647. [CrossRef]
- Supply, P., Magdalena, J., Himpens, S., and Locht, C. (1997). Identification of novel intergenic repetitive units in a mycobacterial two-component system operon. Mol. Microbiol. 26, 991–1003. [CrossRef]
- Suykerbuyk, P., Vleminckx, K., Pasmans, F., Stragier, P., Ablordey, A., Tran, H.T., et al. (2007). Mycobacterium liflandii infection in European colony of Silurana tropicalis. Emerg. Infec. Dis. 13, 743-746.
- Tobias, N. J., Doig, K. D., Medema, M. H., Chen, H., Haring, V., Moore, R., et al. (2013). Complete genome sequence of the frog pathogen Mycobacterium ulcerans ecovar Liflandii. J. Bacteriol. 195, 556–564. [CrossRef]
- Trott, K. A., Stacy, B. A., Lifland, B. D., Diggs, H. E., Harland, R. M., Khokha, M. K., et al. (2004). Characterization of a Mycobacterium ulcerans -like infection in a colony of African tropical clawed frogs (Xenopus tropicalis). Comp. Med. 54, 309–317.
- Tsukamura, M., Kaneda, K., Imaeda, T., and Mikoshiba, H. (1989). A taxonomic study on mycobacterium which caused a skin ulcer in a Japanese girl and resembled Mycobacterium ulcerans. Kekkaku 64, 691–697.
- Tsukamura, M., and Mikoshiba, H. (1982). A New Mycobacterium which Caused Skin Infection. Microbiol. Immunol. 26, 951–955. [CrossRef]
- Ucko, M., Colorni, A., Kvitt, H., Diamant, A., Zlotkin, A., and Knibb, W. R. (2002). Strain variation in Mycobacterium marinum fish isolates. Appl. Environ. Microbiol. 68, 5281–5287. [CrossRef]
- Untergasser, A., Nijveen, H., Rao, X., Bisseling, T., Geurts, R., & Leunissen, J.A. (2007). Primer3Plus, an enhanced web interface to Primer3. Nucleic acids research, 35, W71-W74. [CrossRef]
- Van Trang, N., ThiBich Vu, H., ThiHong Le, N., Huang, P. Jiang, X., and Duc Anh, D. (2014). Association between norovirus and rotavirus infection and histo-blood group antigen types in Vietnamese children. J. Clin. Microbiol. 52, 1366–1374. [CrossRef]
- Williamson, H. R., Benbow, M. E., Nguyen, K. D., Beachboard, D. C., Kimbirauskas, R. K., McIntosh, M. D., et al. (2008). Distribution of Mycobacterium ulcerans in Buruli ulcer endemic and non-endemic aquatic sites in Ghana. PLoS Negl. Trop. Dis. 2. [CrossRef]
- Williamson, H., Phillips, R., Sarfo, S., Wansbrough-Jones, M., and Small, P. (2014). Genetic diversity of PCR-positive, culture-negative and culture-positive Mycobacterium ulcerans isolated from buruli ulcer patients in Ghana. PLoS ONE 9: e88007. [CrossRef]
- Yip, M. J., Porter, J. L., Fyfe, J. A. M., Lavender, C. J., Portaels, F., Rhodes, M., et al. (2007). Evolution of Mycobacterium ulcerans and other mycolactone-producing mycobacteria from a common Mycobacterium marinum progenitor. J. Bacteriol. 189, 2021–2029. [CrossRef]
- Ye, J., Coulouris, G., Zaretskaya, I., Cutcutache, I., Rozen, S., and Madden, T. (2012). Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 13:134. [CrossRef]

| MPM | First isolation | Origin | Host | Colony coloration | Growth rate | Growth temperature | Mycolactone | Source |
|---|---|---|---|---|---|---|---|---|
| M. ulcerans | 1948 | Australia | Human | Greenish or brownish yellow | Slow growth (>4 weeks incubation) | 30°C-33°C | A/B | MacCallum, Buckle, Tolhurst and Sissons (1948) |
|
M. shinshuense (strain 753) |
1980 | Japan | Human | Yellowish, pigmentation in dark | Slow growth (>3 weeks incubation) | 28°C | A/B | Tsukamura and Mikoshiba (1982) |
|
M. marinum (strain CC240299) |
2002 | Israel | Koi (Cyprinus carpio) | NA | NA | NA | F | Ucko et al. (2002) |
|
M. marinum (strain DL240490) |
2002 | Red Sea | European sea bass (Dicentrarchus labrax) | NA | NA | NA | F | Ucko et al. (2002) |
|
M. marinum (strain DL045) |
2002 | Mediterranean Sea | European sea bass (Dicentrarchus labrax) | NA | NA | NA | F | Ucko et al. (2002) |
|
M. liflandii (strain 128FXT) |
2004 | US | African tropical clawed frogs (Xenopus tropicalis) | Buff-colored, non-pigmented | Slow growth (>4 weeks incubation) | 28°C | E | Trott et al. (2004) |
| M. pseudoshottsii (strain L15) | 2005 | US | Striped bass (Morone saxatilis) | Pale-yellow to gold, non-pigmented | Slow growth (>4 weeks incubation) |
23°C | F | Rhodes et al. (2005) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




