Submitted:
26 July 2023
Posted:
28 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Design of RT-LAMP primers for SARS CoV-2genome detection
2.2. Sample Inactivation and treatment for direct assay
2.3. RT-LAMP assay
2.4. Primers cross-reactivity (in silico sequences analysis)
2.5. Analytical Specificity of the RT-LAMP Assay
2.6. Repeatability, Reproducibility and Detection limit of the RT-LAMP assay
2.7. Clinical evaluation of the RT-LAMP assay for SARS-CoV-2 detection
3. Results and discussion
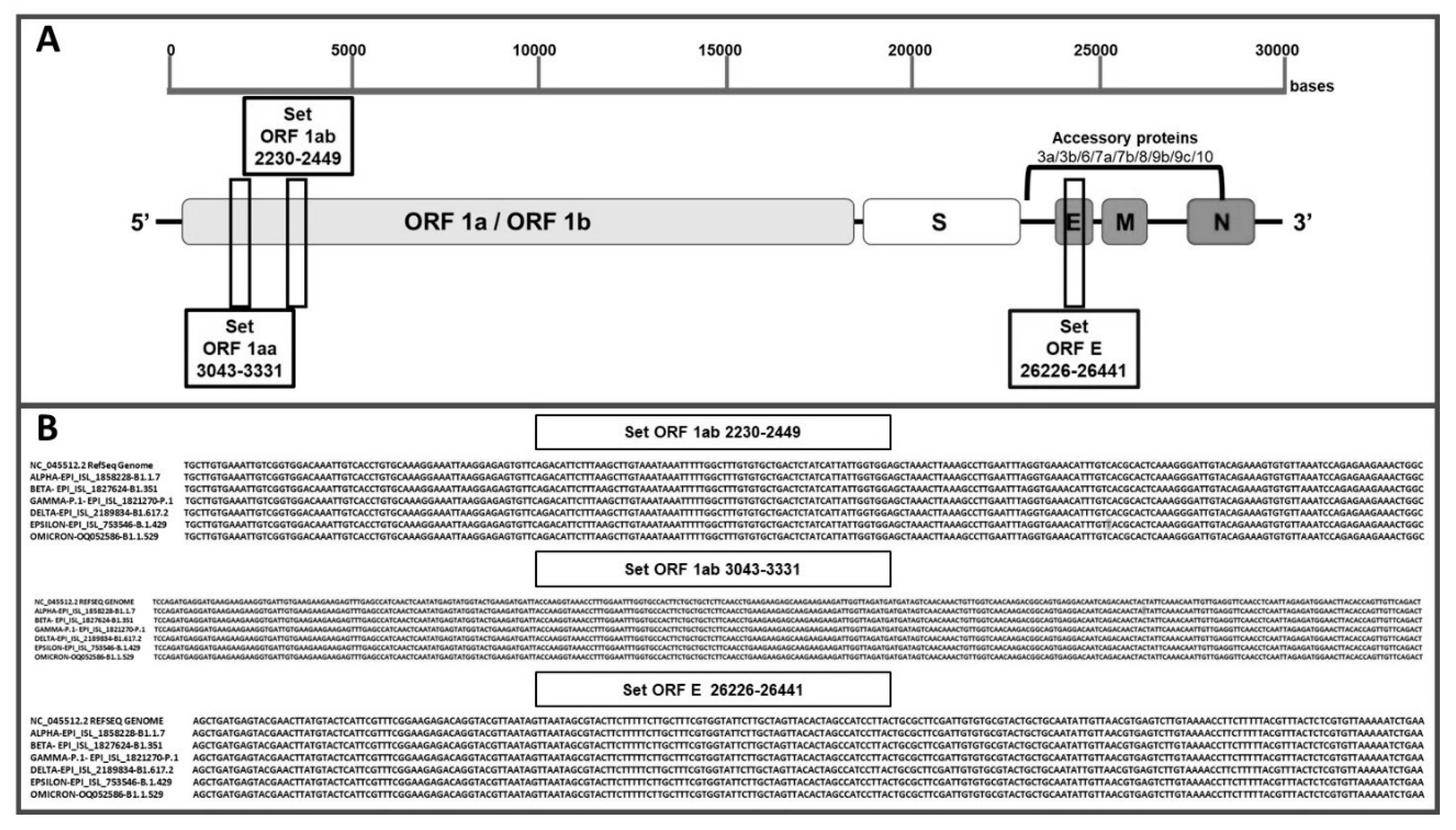
3.1. Design and specificity of ORF1aa, ORF1ab and gene E primers for SARS-CoV-2 target sequence identification
3.2. Evaluation of primer annealing with SARS-CoV-2 variants and mutations
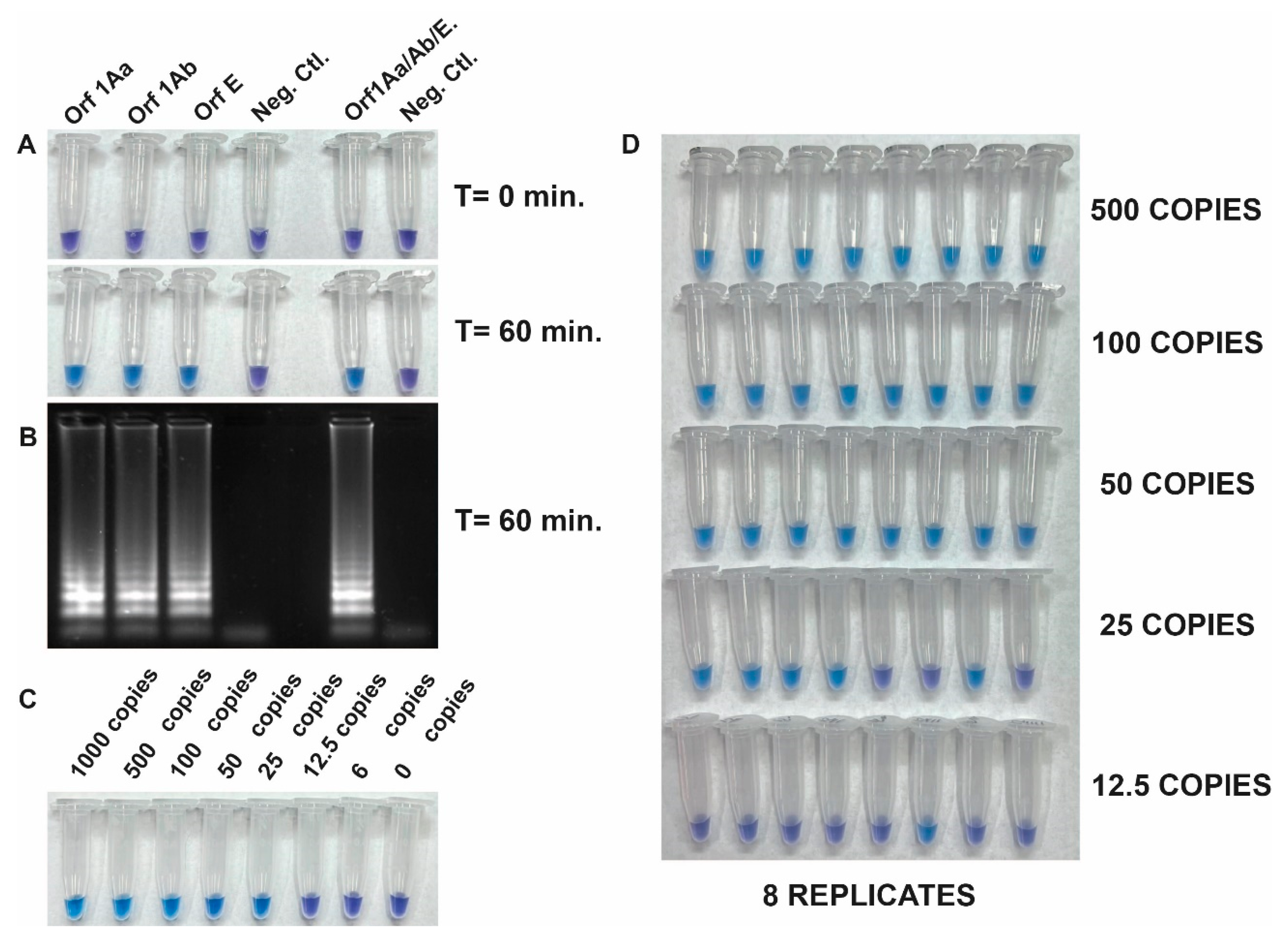
3.3. Development of an RT-LAMP assay and its Analytical Sensitivity Testing
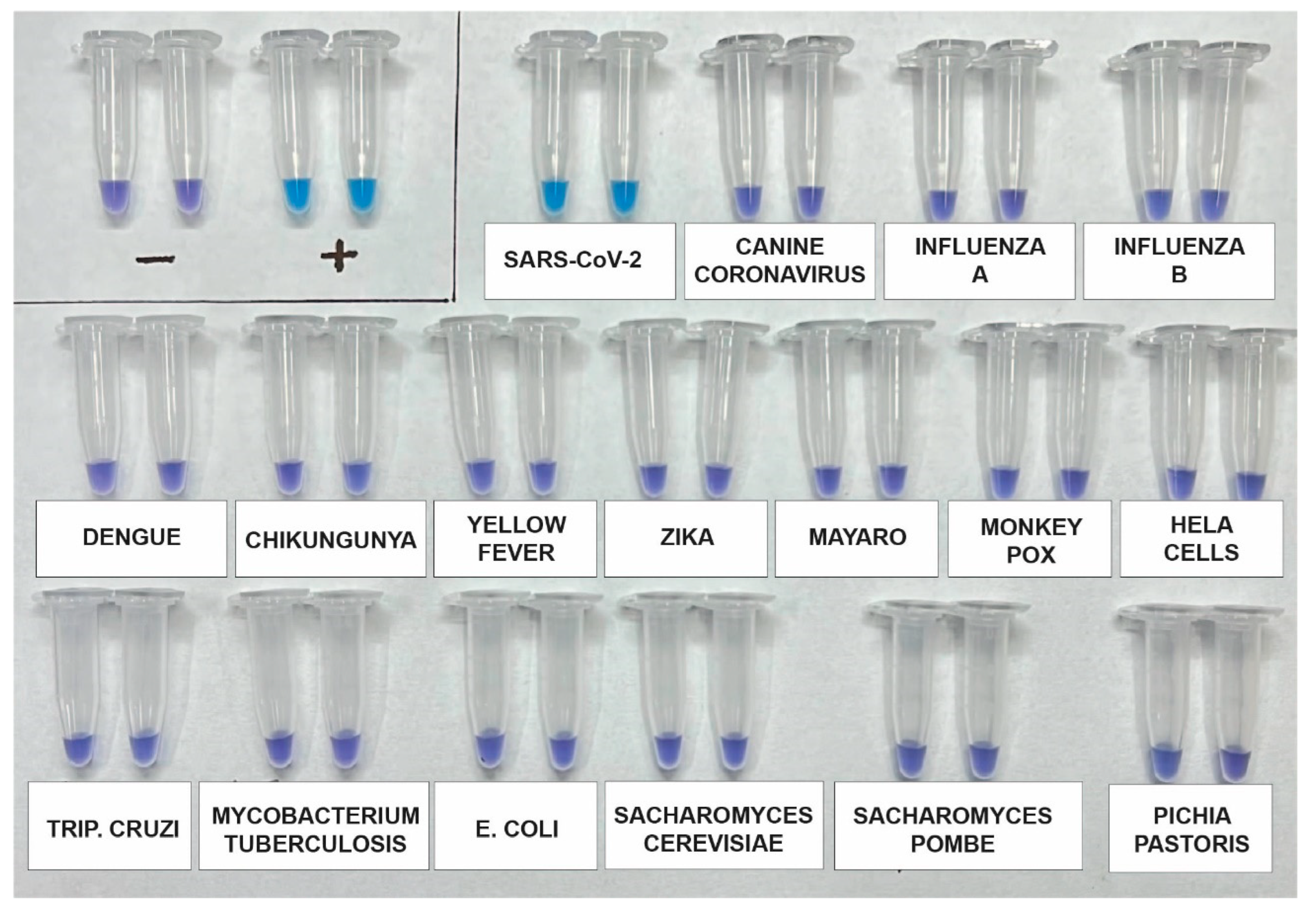
3.4. Analytical Specificity Testing
3.5. Clinical Validation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethical approval
Acknowledgments
Conflicts of Interest
References
- Drosten, C.; Göttig, S.; Schilling, S.; Asper, M.; Panning, M.; Schmitz, H.; Günther, S. Rapid Detection and Quantification of RNA of Ebola and Marburg Viruses, Lassa Virus, Crimean-Congo Hemorrhagic Fever Virus, Rift Valley Fever Virus, Dengue Virus, and Yellow Fever Virus by Real-Time Reverse Transcription-PCR. J. Clin. Microbiol. 2002, 40, 2323–2330. [Google Scholar] [CrossRef]
- Espy, M.J.; Uhl, J.R.; Sloan, L.M.; Buckwalter, S.P.; Jones, M.F.; Vetter, E.A.; Yao, J.D.C.; Wengenack, N.L.; Rosenblatt, J.E.; Cockerill, F.R.; et al. Real-Time PCR in Clinical Microbiology: Applications for Routine Laboratory Testing. Clin. Microbiol. Rev. 2006, 19, 165–256. [Google Scholar] [CrossRef]
- Mackay, I.M.; Arden, K.E.; Nitsche, A. SURVEY AND SUMMARY SURVEY AND SUMMARY Real-Time PCR in Virology. Nucleic Acids Res. 2002, 30, 1292–1305. [Google Scholar] [CrossRef]
- Boehme, C.C.; Nabeta, P.; Henostroza, G.; Raqib, R.; Rahim, Z.; Gerhardt, M.; Sanga, E.; Hoelscher, M.; Notomi, T.; Hase, T.; et al. Operational Feasibility of Using Loop-Mediated Isothermal Amplification for Diagnosis of Pulmonary Tuberculosis in Microscopy Centers of Developing Countries. J. Clin. Microbiol. 2007, 45, 1936–1940. [Google Scholar] [CrossRef]
- Tomita, N.; Mori, Y.; Kanda, H.; Notomi, T. Loop-Mediated Isothermal Amplification (LAMP) of Gene Sequences and Simple Visual Detection of Products. Nat. Protoc. 2008, 3, 877–882. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-Mediated Isothermal Amplification of DNA. Nucleic Acids Res. 2000, 28. [Google Scholar] [CrossRef]
- Mori, Y.; Notomi, T. Loop-Mediated Isothermal Amplification (LAMP): A Rapid, Accurate, and Cost-Effective Diagnostic Method for Infectious Diseases. J. Infect. Chemother. 2009, 15, 62–69. [Google Scholar] [CrossRef]
- Law, J.W.F.; Mutalib, N.S.A.; Chan, K.G.; Lee, L.H. Rapid Metho Ds for the Detection of Foodborne Bacterial Pathogens: Principles, Applications, Advantages and Limitations. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef]
- Thai, H.T.C.; Le, M.Q.; Vuong, C.D.; Parida, M.; Minekawa, H.; Notomi, T.; Hasebe, F.; Morita, K. Development and Evaluation of a Novel Loop-Mediated Isothermal Amplification Method for Rapid Detection of Severe Acute Respiratory Syndrome Coronavirus. J. Clin. Microbiol. 2004, 42, 1956–1961. [Google Scholar] [CrossRef]
- Reboud, J.; Xu, G.; Garrett, A.; Adriko, M.; Yang, Z.; Tukahebwa, E.M.; Rowell, C.; Cooper, J.M. Vector Control Division, Ministry of Health. 2019. [CrossRef]
- Nagamine, K.; Hase, T.; Notomi, T. Accelerated Reaction by Loop-Mediated Isothermal Amplification Using Loop Primers. Mol. Cell. Probes 2002, 16, 223–229. [Google Scholar] [CrossRef]
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A Novel Coronavirus Outbreak of Global Health Concern Published. Lancet 2020, 395, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Lu, Y.; Meng, Y.; Ye, Z.; Wang, L.; Wang, R.; Zheng, Q.; Wu, H.; Wu, J. Field Detection of Citrus Huanglongbing Associated with ’ Candidatus Liberibacter Asiaticus’ by Recombinese Polymerase Amplification within 15 Min. J. Agric. Food Chem. 2018, 66, 5473–5480. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lu, H.; Thai, S.; Li, X.; Hui, J.; Tang, H.; Zhai, S.; Sun, L.; Wang, T. Development and Validation of an Algorithm to Identify Drug-Induced Anaphylaxis in the Beijing Pharmacovigilance Database. Int. J. Clin. Pharm. 2018, 40, 862–869. [Google Scholar] [CrossRef]
- Yao, M.; Jiang, F.; Zhang, Y.; Li, Y.; Pang, B.; Liang, H.; Kou, Z.; Jiang, X.; Wen, H.; Xu, Y. SARS-CoV-2 Variant of Concern 202 012/01 (B.1.1.7) in a Traveller from the UK to China. J Travel Med 2021, 28. [Google Scholar] [CrossRef] [PubMed]
- Iacobucci, G. Covid-19: New UK Variant May Be Linked to Increased Death Rate, Early Data Indicate. BMJ 2021, 372, 1–2. [Google Scholar] [CrossRef]
- Tegally, H.; Wilkinson, E.; Giovanetti, M.; Iranzadeh, A.; Fonseca, V.; Giandhari, J.; Doolabh, D.; Pillay, S.; San, E.J.; Msomi, N.; et al. Emergence and Rapid Spread of a New Severe Acute Respiratory Syndrome-Related Coronavirus 2 (SARS-CoV-2) Lineage with Multiple Spike Mutations in South Africa. medRxiv 2020, 2. [Google Scholar] [CrossRef]
- Voloch, C.M.; Ronaldo da Silva Francisco, J.; Almeida, L.G.P. de; Cardoso, C.C.; Brustolini, O.J.; Gerber, A.L.; Guimarães, A.P. de C.; Mariani, D.; Costa, R.M. da; Orlando C. Ferreira, J.; et al. Genomic Characterization of a Novel SARS-CoV-2 Lineage from Rio de Janeiro, Brazil. J. Virol. 2021, 95, 19–21. [Google Scholar] [CrossRef]
- Long, S.W.; Olsen, R.J.; Christensen, P.A.; Subedi, S.; Olson, R.; Davis, J.J. SHORT COMMUNICATION Sequence Analysis of 20 , 453 Severe Acute Respiratory Syndrome Coronavirus 2 Genomes from the Houston Metropolitan Area Identi Fi Es the Emergence and Widespread Distribution of Multiple Isolates of All Major Variants of Concern. 2021, 191, 983–992.
- Tian, D.; Sun, Y.; Xu, H.; Ye, Q. The Emergence and Epidemic Characteristics of the Highly Mutated SARS-CoV-2 Omicron Variant. J. Med. Virol. 2022, 94, 2376–2383. [Google Scholar] [CrossRef]
- Ahmed, W.; Bivins, A.; Smith, W.J.M.; Metcalfe, S.; Stephens, M.; Jennison, A. V.; Moore, F.A.J.; Bourke, J.; Schlebusch, S.; McMahon, J.; et al. Detection of the Omicron (B.1.1.529) Variant of SARS-CoV-2 in Aircraft Wastewater. Sci. Total Environ. 2022, 820. [Google Scholar] [CrossRef]
- Lyke, K.E.; Atmar, R.L.; Dominguez Islas, C.; Posavad, C.M.; Szydlo, D.; PaulChourdhury, R.; Deming, M.E.; Eaton, A.; Jackson, L.A.; Branche, A.R.; et al. Rapid Decline in Vaccine-Boosted Neutralizing Antibodies Against SARS-CoV-2 Omicron Variant. Cell reports Med. 2022, 3, 1–7. [Google Scholar] [CrossRef]
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.M.; Wang, W.; Song, Z.G.; Hu, Y.; Tao, Z.W.; Tian, J.H.; Pei, Y.Y.; et al. A New Coronavirus Associated with Human Respiratory Disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.Y.; Huang, Y.; Lau, S.K.P.; Yuen, K.Y. Coronavirus Genomics and Bioinformatics Analysis. Viruses 2010, 2, 1805–1820. [Google Scholar] [CrossRef] [PubMed]
- Schoeman, D.; Fielding, B.C. Coronavirus Envelope Protein: Current Knowledge. Virol. J. 2019, 16. [Google Scholar] [CrossRef] [PubMed]
| Target | Primer | Sequence |
|---|---|---|
| ORF1AA | R1Aa-2F3 | TGCTTGTGAAATTGTCGGT |
| R1Aa-2B3 | GCCAGTTTCTTCTCTGGAT | |
| R1Aa-2FIP | TCAGCACACAAAGCCAAAAATTTAT-AAATTGTCACCTGTGCAAAG | |
| R1Aa-2BIP | TATTGGTGGAGCTAAACTTAAAGCC-ACACTTTCTGTACAATCCCTT | |
| R1Aa-2LP | CTTAAAGAATGTCTGAACACTCTCC | |
| R1Aa-2LB | GAATTTAGGTGAAACATTTGTCACG | |
| ORF1AB | R1Ab-1F3(R1AB) | TCCAGATGAGGATGAAGAAGA |
| R1Ab-1B3 | AGTCTGAACAACTGGTGTAAG | |
| R1Ab-1FIP | AGAGCAGCAGAAGTGGCACAGGTGATTGTGAAGAAGAAGAG | |
| R1Ab-1BIP | TCAACCTGAAGAAGAGCAAGAACTGATTGTCCTCACTGCC | |
| R1Ab-1LF | CTCATATTGAGTTGATGGCTCA | |
| R1Ab-1LB | ACAAACTGTTGGTCAACAAGAC | |
| ORF E | pE-3F3(PE3) | AGCTGATGAGTACGAACTT |
| pE-3B3 | TTCAGATTTTTAACACGAGAGT | |
| pE-3FIP | ACCACGAAAGCAAGAAAAAGAAGTATTCGTTTCGGAAGAGACAG | |
| pE-3BIP | TTGCTAGTTACACTAGCCATCCTTAGGTTTTACAAGACTCACGT | |
| pE-3LF | ACGCTATTAACTATTAACGTAC | |
| pE-3LB | CTGCGCTTCGATTGTGTGCGT |
| Reference technique RT-PCR |
TOTAL | ||
| Positive samples | Negative Samples | ||
| NEOKIT positives | 126 | 0 | 126 |
| NEOKIT negatives | 13 | 53 | 66 |
| TOTAL | 139 | 53 | 192 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




