Submitted:
27 July 2023
Posted:
28 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
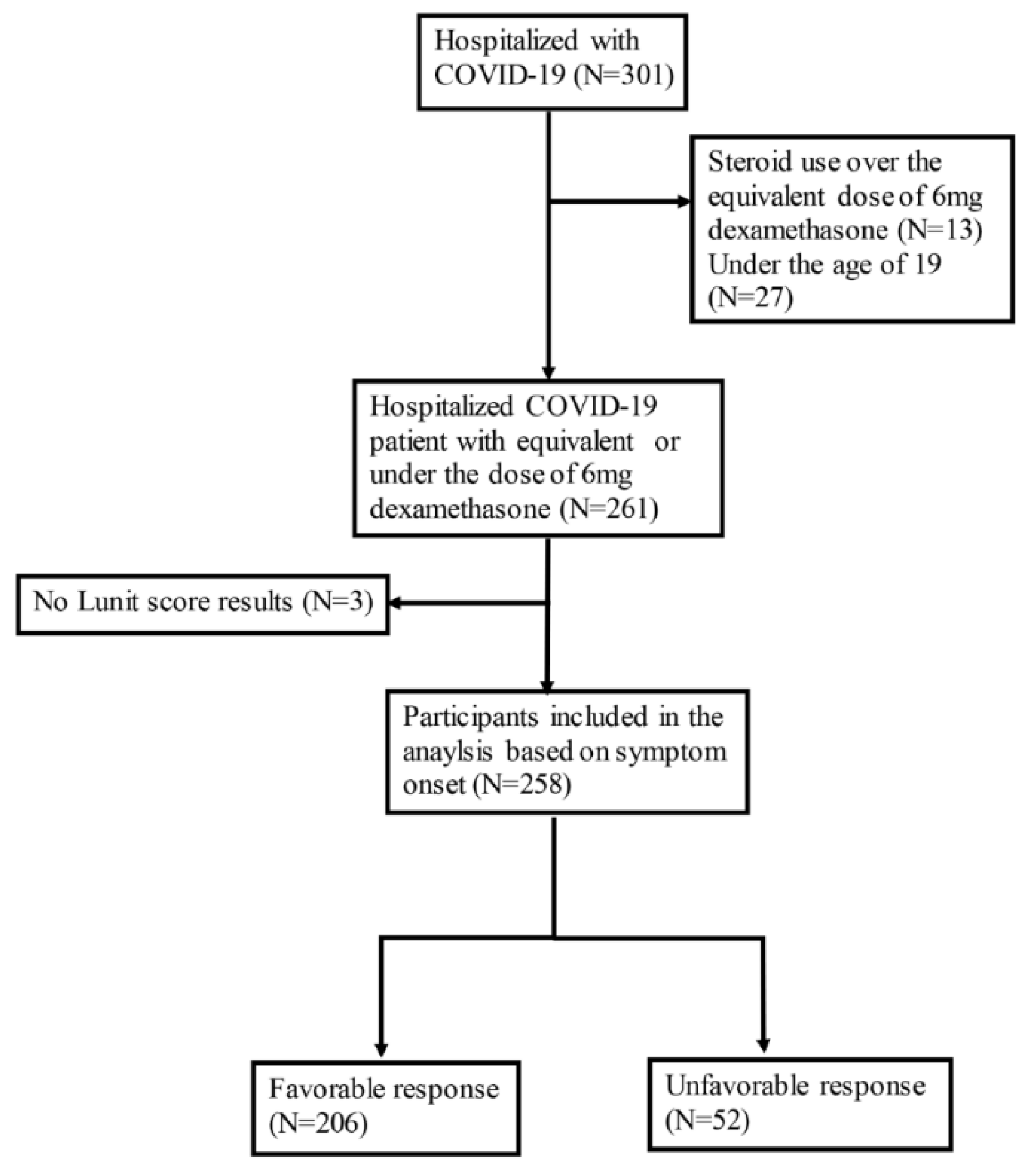
2.1. Study design and population
2.2. Data collection
2.4. Statistical analysis
3. Results
3.1. Baseline characteristics
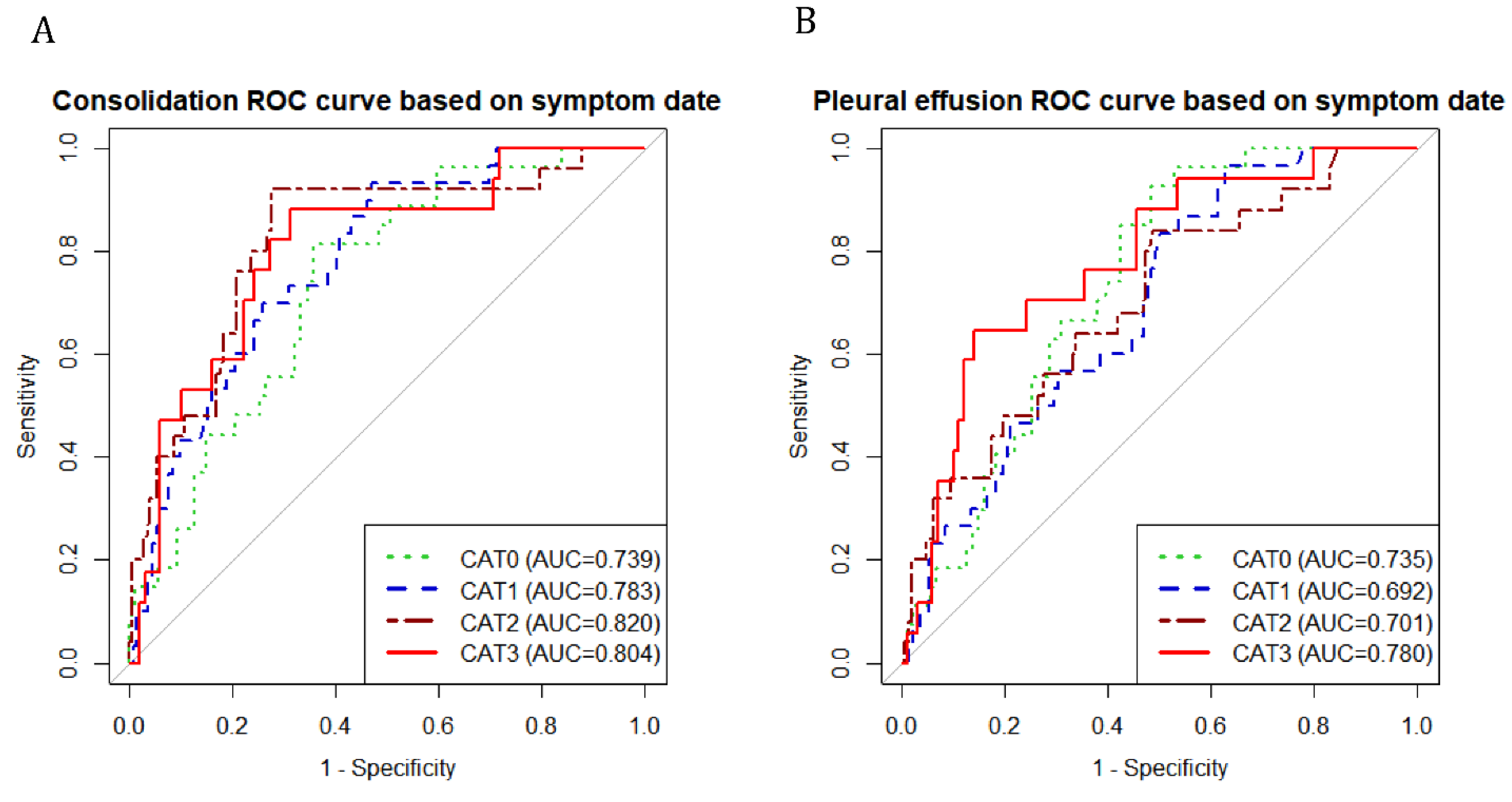
3.2. AI-CXR score as a factor associated with unfavorable corticosteroid response
3.3. Association between AI-CXR scores and other laboratory tests correlated with unfavorable corticosteroid response
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus-2 |
| CXR | chest radiographs |
| AI | artificial intelligence |
| AI-CXR score | artificial intelligence-generated chest radiograph abnormality score |
| DM | Diabetes mellitus |
| COPD | chronic obstructive pulmonary disease chronic kidney disease |
| CKD | chronic kidney disease |
| CCI | Charlson comorbidity index |
| WBC | white blood cell count |
| CRP | C-reactive protein |
| IL | interleukin |
| PCT | procalcitonin |
| ROC | receiver operating characteristic |
| aOR | adjusted odds ratio |
| CI | confidence interval. |
References
- Weekly epidemiological update on COVID-19 - 18 May 2023. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---18-may-2023 (accessed on June 15th,2023).
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med 2021, 384, 693-704. [CrossRef]
- Tomazini, B.M.; Maia, I.S.; Cavalcanti, A.B.; Berwanger, O.; Rosa, R.G.; Veiga, V.C.; Avezum, A.; Lopes, R.D.; Bueno, F.R.; Silva, M.; et al. Effect of Dexamethasone on Days Alive and Ventilator-Free in Patients With Moderate or Severe Acute Respiratory Distress Syndrome and COVID-19: The CoDEX Randomized Clinical Trial. Jama 2020, 324, 1307-1316. [CrossRef]
- Munch, M.W.; Myatra, S.N.; Vijayaraghavan, B.K.T.; Saseedharan, S.; Benfield, T.; Wahlin, R.R.; Rasmussen, B.S.; Andreasen, A.S.; Poulsen, L.M.; Cioccari, L.; et al. Effect of 12 mg vs 6 mg of Dexamethasone on the Number of Days Alive Without Life Support in Adults With COVID-19 and Severe Hypoxemia: The COVID STEROID 2 Randomized Trial. Jama 2021, 326, 1807-1817. [CrossRef]
- Maskin, L.P.; Bonelli, I.; Olarte, G.L.; Palizas, F., Jr.; Velo, A.E.; Lurbet, M.F.; Lovazzano, P.; Kotsias, S.; Attie, S.; Lopez Saubidet, I.; et al. High- Versus Low-Dose Dexamethasone for the Treatment of COVID-19-Related Acute Respiratory Distress Syndrome: A Multicenter, Randomized Open-Label Clinical Trial. J Intensive Care Med 2022, 37, 491-499. [CrossRef]
- Moore, J.B.; June, C.H. Cytokine release syndrome in severe COVID-19. Science 2020, 368, 473-474.
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. Journal of Medical Virology 2020, 92, 424-432. [CrossRef]
- Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available online: https://www.covid19treatmentguidelines.nih.gov/about-the-guidelines/whats-new/ (accessed on June 15th,2023).
- Prescott, H.C.; Rice, T.W. Corticosteroids in COVID-19 ARDS: Evidence and Hope During the Pandemic. JAMA 2020, 324, 1292-1295. [CrossRef]
- Wang, J.; Yang, W.; Chen, P.; Guo, J.; Liu, R.; Wen, P.; Li, K.; Lu, Y.; Ma, T.; Li, X. The proportion and effect of corticosteroid therapy in patients with COVID-19 infection: A systematic review and meta-analysis. PloS one 2021, 16, e0249481. [CrossRef]
- Carsana, L.; Sonzogni, A.; Nasr, A.; Rossi, R.S.; Pellegrinelli, A.; Zerbi, P.; Rech, R.; Colombo, R.; Antinori, S.; Corbellino, M. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study. The Lancet infectious diseases 2020, 20, 1135-1140. [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. New England Journal of Medicine 2020, 383, 120-128.
- Liang, W.; Liang, H.; Ou, L.; Chen, B.; Chen, A.; Li, C.; Li, Y.; Guan, W.; Sang, L.; Lu, J. Development and validation of a clinical risk score to predict the occurrence of critical illness in hospitalized patients with COVID-19. JAMA internal medicine 2020, 180, 1081-1089. [CrossRef]
- D'Cruz, R.F.; Waller, M.D.; Perrin, F.; Periselneris, J.; Norton, S.; Smith, L.-J.; Patrick, T.; Walder, D.; Heitmann, A.; Lee, K. Chest radiography is a poor predictor of respiratory symptoms and functional impairment in survivors of severe COVID-19 pneumonia. ERJ open research 2021, 7. [CrossRef]
- Chassagnon, G.; Vakalopoulou, M.; Battistella, E.; Christodoulidis, S.; Hoang-Thi, T.N.; Dangeard, S.; Deutsch, E.; Andre, F.; Guillo, E.; Halm, N.; et al. AI-driven quantification, staging and outcome prediction of COVID-19 pneumonia. Med Image Anal 2021, 67, 101860. [CrossRef]
- Baltazar, L.R.; Manzanillo, M.G.; Gaudillo, J.; Viray, E.D.; Domingo, M.; Tiangco, B.; Albia, J. Artificial intelligence on COVID-19 pneumonia detection using chest xray images. Plos one 2021, 16, e0257884. [CrossRef]
- Jiao, Z.; Choi, J.W.; Halsey, K.; Tran, T.M.L.; Hsieh, B.; Wang, D.; Eweje, F.; Wang, R.; Chang, K.; Wu, J. Prognostication of patients with COVID-19 using artificial intelligence based on chest x-rays and clinical data: a retrospective study. The Lancet Digital Health 2021, 3, e286-e294. [CrossRef]
- Haghanifar, A.; Majdabadi, M.M.; Choi, Y.; Deivalakshmi, S.; Ko, S. Covid-cxnet: Detecting covid-19 in frontal chest x-ray images using deep learning. Multimedia Tools and Applications 2022, 1-31. [CrossRef]
- Shin, H.J.; Son, N.-H.; Kim, M.J.; Kim, E.-K. Diagnostic performance of artificial intelligence approved for adults for the interpretation of pediatric chest radiographs. Scientific Reports 2022, 12, 10215. [CrossRef]
- Nam, J.G.; Park, S.; Hwang, E.J.; Lee, J.H.; Jin, K.-N.; Lim, K.Y.; Vu, T.H.; Sohn, J.H.; Hwang, S.; Goo, J.M. Development and validation of deep learning–based automatic detection algorithm for malignant pulmonary nodules on chest radiographs. Radiology 2019, 290, 218-228. [CrossRef]
- Ahn, J.S.; Ebrahimian, S.; McDermott, S.; Lee, S.; Naccarato, L.; Di Capua, J.F.; Wu, M.Y.; Zhang, E.W.; Muse, V.; Miller, B. Association of Artificial Intelligence–Aided Chest Radiograph Interpretation With Reader Performance and Efficiency. JAMA Network Open 2022, 5, e2229289-e2229289. [CrossRef]
- People with Certain Medical Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html#immunocompromised (accessed on December 23rd, 2022).
- Hwang, E.J.; Goo, J.M.; Yoon, S.H.; Beck, K.S.; Seo, J.B.; Choi, B.W.; Chung, M.J.; Park, C.M.; Jin, K.N.; Lee, S.M. Use of artificial intelligence-based software as medical devices for chest radiography: a position paper from the Korean Society of Thoracic Radiology. Korean Journal of Radiology 2021, 22, 1743. [CrossRef]
- Lee, S.; Shin, H.J.; Kim, S.; Kim, E.-K. Successful implementation of an artificial intelligence-based computer-aided detection system for chest radiography in daily clinical practice. Korean Journal of Radiology 2022, 23, 847-852. [CrossRef]
- Kim, E.Y.; Kim, Y.J.; Choi, W.-J.; Jeon, J.S.; Kim, M.Y.; Oh, D.H.; Jin, K.N.; Cho, Y.J. Concordance rate of radiologists and a commercialized deep-learning solution for chest X-ray: Real-world experience with a multicenter health screening cohort. PloS one 2022, 17, e0264383. [CrossRef]
- Murakami, K.; Sano, H.; Tode, N.; Tsukita, Y.; Sato, K.; Narita, D.; Kimura, N.; Matsumoto, S.; Ono, Y.; Iwasaki, C. Clinical features of COVID-19 patients with rebound phenomenon after corticosteroid therapy. BMJ Open Respiratory Research 2022, 9, e001332. [CrossRef]
- Gao, Y.; Xiong, X.; Jiao, X.; Yu, Y.; Chi, J.; Zhang, W.; Chen, L.; Li, S.; Gao, Q. PRCTC: a machine learning model for prediction of response to corticosteroid therapy in COVID-19 patients. Aging (Albany NY) 2022, 14, 54. [CrossRef]
- Yang, R.; Li, X.; Liu, H.; Zhen, Y.; Zhang, X.; Xiong, Q.; Luo, Y.; Gao, C.; Zeng, W. Chest CT severity score: an imaging tool for assessing severe COVID-19. Radiology: Cardiothoracic Imaging 2020, 2. [CrossRef]
- Abdollahi, I.; Nabahati, M.; Javanian, M.; Shirafkan, H.; Mehraeen, R. Can initial chest CT scan predict status and clinical outcomes of COVID-19 infection? A retrospective cohort study. Egyptian Journal of Radiology and Nuclear Medicine 2021, 52, 1-10. [CrossRef]
- Zhou, S.; Chen, C.; Hu, Y.; Lv, W.; Ai, T.; Xia, L. Chest CT imaging features and severity scores as biomarkers for prognostic prediction in patients with COVID-19. Annals of translational medicine 2020, 8. [CrossRef]
- Solinas, C.; Perra, L.; Aiello, M.; Migliori, E.; Petrosillo, N. A critical evaluation of glucocorticoids in the management of severe COVID-19. Cytokine & growth factor reviews 2020, 54, 8-23. [CrossRef]
- Drozdov, I.; Szubert, B.; Reda, E.; Makary, P.; Forbes, D.; Chang, S.L.; Ezhil, A.; Puttagunta, S.; Hall, M.; Carlin, C. Development and prospective validation of COVID-19 chest X-ray screening model for patients attending emergency departments. Scientific reports 2021, 11, 1-14. [CrossRef]
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nature Reviews Microbiology 2021, 19, 141-154.
- Chong, W.H.; Saha, B.K.; Conuel, E.; Chopra, A. The incidence of pleural effusion in COVID-19 pneumonia: state-of-the-art review. Heart & Lung 2021, 50, 481-490. [CrossRef]
- Jacobi, A.; Chung, M.; Bernheim, A.; Eber, C. Portable chest X-ray in coronavirus disease-19 (COVID-19): A pictorial review. Clin Imaging 2020, 64, 35-42. [CrossRef]
- Lombardi, Y.; Azoyan, L.; Szychowiak, P.; Bellamine, A.; Lemaitre, G.; Bernaux, M.; Daniel, C.; Leblanc, J.; Riller, Q.; Steichen, O. External validation of prognostic scores for COVID-19: a multicenter cohort study of patients hospitalized in Greater Paris University Hospitals. Intensive care medicine 2021, 47, 1426-1439. [CrossRef]
- Ding, L.; Zhang, W.; Zhang, F.; Huang, C.; Yang, M.; Tang, Z.; Li, Y.; Mi, J.; Zhong, W. Prognostic role and diagnostic power of seven indicators in COVID-19 patients. Frontiers in Medicine 2021, 8. [CrossRef]
- Yoon, S.H.; Lee, J.H.; Kim, B.-N. Chest CT findings in hospitalized patients with SARS-CoV-2: delta versus Omicron variants. Radiology 2023, 306, 252-260.
- Jin, K.N.; Kim, E.Y.; Kim, Y.J.; Lee, G.P.; Kim, H.; Oh, S.; Kim, Y.S.; Han, J.H.; Cho, Y.J. Diagnostic effect of artificial intelligence solution for referable thoracic abnormalities on chest radiography: a multicenter respiratory outpatient diagnostic cohort study. European radiology 2022, 32, 3469-3479. [CrossRef]
- Hwang, E.J.; Kim, H.; Yoon, S.H.; Goo, J.M.; Park, C.M. Implementation of a deep learning-based computer-aided detection system for the interpretation of chest radiographs in patients suspected for COVID-19. Korean journal of radiology 2020, 21, 1150. [CrossRef]
| Total | Unfavorable* (N=52) | Favorable (N=206) |
p-value | |
|---|---|---|---|---|
| Age (years) | 64.21±18.88 | 69.67±16.52 | 62.83±19.19 | <0.001 |
| Sex (male), n (%) | 147 (57.0) | 42 (80.8) | 105 (51.0) | <0.001 |
| Re-infection, n (%) | 2 (0.8) | 0 (0.0) | 2 (1.0) | 0.371b |
| Comorbidities, n (%) | ||||
| DM | 76 (29.5) | 17 (32.7) | 59 (28.6) | 0.220 |
| COPD | 19 (7.4) | 4 (7.7) | 15 (7.3) | 0.882† |
| CHF | 16 (6.2) | 3 (5.8) | 13 (6.3) | 0.873† |
| CKD | 7 (2.7) | 2 (3.8) | 5 (2.4) | 0.243† |
| Chronic liver Dz. | 5 (1.9) | 1 (1.9) | 4 (1.9) | 1.00b |
| Malignancy | 42 (16.3) | 14 (26.9) | 28 (13.6) | <0.001 |
| CCI | 1 [0-3] | 1.5 [0-4] | 1 [0-2] | <0.001 |
| Immunocompromised, n (%) | 44 (17.1) | 17 (32.7) | 27 (13.1) | <0.001 |
| Outcomes | ||||
| Condition at discharge, n (%) | <0.001 | |||
| Normal discharge | 205 (79.4) | 0 (0.0) | 205 (99.5) | |
| Transfer | 51 (19.8) | 51 (98.1) | 0 (0.0) | |
| Death | 1 (0.4) | 1 (1.9) | 0 (0.0) | |
| Others | 1 (0.4) | 0 (0.0) | 1 (0.5) | |
| Hospital days | 8 [6-12] | 4 [1-11.75] | 8 [6-12] | <0.001 |
| Treatments | ||||
| Oxygen requirements, n (%) | <0.001 | |||
| None | 99 (38.4) | 0 (0.0) | 99 (48.1) | |
| Low flow oxygen | 101 (39.1) | 3 (5.8) | 98 (47.6) | |
| High flow oxygen | 55 (21.3) | 46 (88.5) | 9 (4.4) | |
| Mechanical ventilation | 3 (1.2) | 3 (5.8) | 0 (0.0) | |
| Monoclonal antibody,n (%) | 10 (3.9) | 0 (0.0) | 10 (4.9) | <0.001 |
| Tocilizumab,n (%) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Antiviral agents,n (%) | 152 (58.9) | 40 (76.9) | 112 (54.4) | <0.001 |
| Remdesivir | 146 (96.1) | 39 (97.5) | 107 (95.5) | |
| Nirmatrevir/lopinavir | 3 (2.0) | 1 (2.5) | 2 (1.8) | |
| Molnuprevir | 3 (2.0) | 0 (0.0) | 3 (2.7) | |
| Antibacterial agents,n (%) | 201 (77.9) | 50 (96.2) | 151 (73.3) | <0.001 |
| Vaccination,n (%) | 118 (49.0) | 21(43.8) | 97 (50.3) | 0.115 |
| Primary vaccination | 97 (82.2) | 20 (95.2) | 77 (79.4) | |
| Booster | 21 (17.8) | 1(4.8) | 20 (20.6) | |
| Corticosteroid Treatment | ||||
| Types, n (%) | 1.000 | |||
| Dexamethasone | 243 (94.2) | 51 (98.1) | 192 (93.2) | |
| Methylprednisolone | 8 (3.1) | 1 (1.9) | 7 (3.4) | |
| Prednisolone | 4 (1.5) | 0 (0.0) | 4 (1.9) | |
| Hydrocortisone | 3 (1.1) | 0 (0.0) | 3 (1.4) | |
| Doses, n (%) | 0.300 | |||
| 6mg equivalent | 255 (98.5) | 52 (100.0) | 203 (98.5) | |
| less | 3 (1.2) | 0 (0.0) | 3 (1.5) | |
| Days of steroid initiation‡ | 4 [2-7] | 3 [2-6] | 4 [2-7] | 0.271 |
| Treatment duration | 5 [4-8] | 3 [1.25-8] | 6 [4-8] | 0.350 |
| Variables | Univariate | Multivariable | |||
|---|---|---|---|---|---|
| OR* (95% CI) | p-value | aOR† (95% CI) | p-value | ||
| Total | Consolidation score (%) | 1.030 (1.017-1.042) | <0.001 | 1.022 (1.010-1.035) | <0.001 |
| Pleural effusion score (%) | 1.020 (1.009-1.032) | 0.001 | 1.013 (1.001-1.026) | 0.040 | |
| Category 0‡ | Consolidation score (%) | 1.025 (1.011-1.039) | <0.001 | 1.025 (1.006-1.045) | 0.010 |
| Pleural effusion score (%) | 1.016 (0.999-1.033) | 0.068 | 1.003 (0.984-1.021) | 0.780 | |
| Category 1§ | Consolidation score (%) | 1.035 (1.018-1.053) | <0.001 | 1.03 (1.011-1.051) | 0.002 |
| Pleural effusion score (%) | 1.020 (1.004-1.035) | 0.013 | 1.017 (0.999-1.035) | 0.070 | |
| Category 2‖ | Consolidation score (%) | 1.057 (1.022-1.093) | 0.001 | 1.052 (1.015-1.089) | 0.005 |
| Pleural effusion score (%) | 1.025 (1.010-1.040) | 0.001 | 1.022 (1.003-1.042) | 0.020 | |
| Category 3¶ | Consolidation score (%) | 1.058 (1.006-1.113) | 0.028 | 1.033 (0.988-1.080) | 0.158 |
| Pleural effusion score (%) | 1.022 (1.006-1.039) | 0.006 | 1.003 (0.979-1.027) | 0.809 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




