Submitted:
17 July 2023
Posted:
18 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture and microgravity parameters
2.2. Cell viability assay
2.3. Measurements of glucose and lactate levels
2.4. Western Blot Analysis
2.5. Transmission Electron Microscopy analysis
2.6. Confocal live cell imaging analysis for mitochondria function
2.6.1. JC1 assay
2.6.2. MitoSOX Assay
2.6.3. Reactive Oxygen Species Detection
2.7. RNA isolation, RT-PCR and qRT-PCR analyses
2.8. Statistical Analyses
3. Results
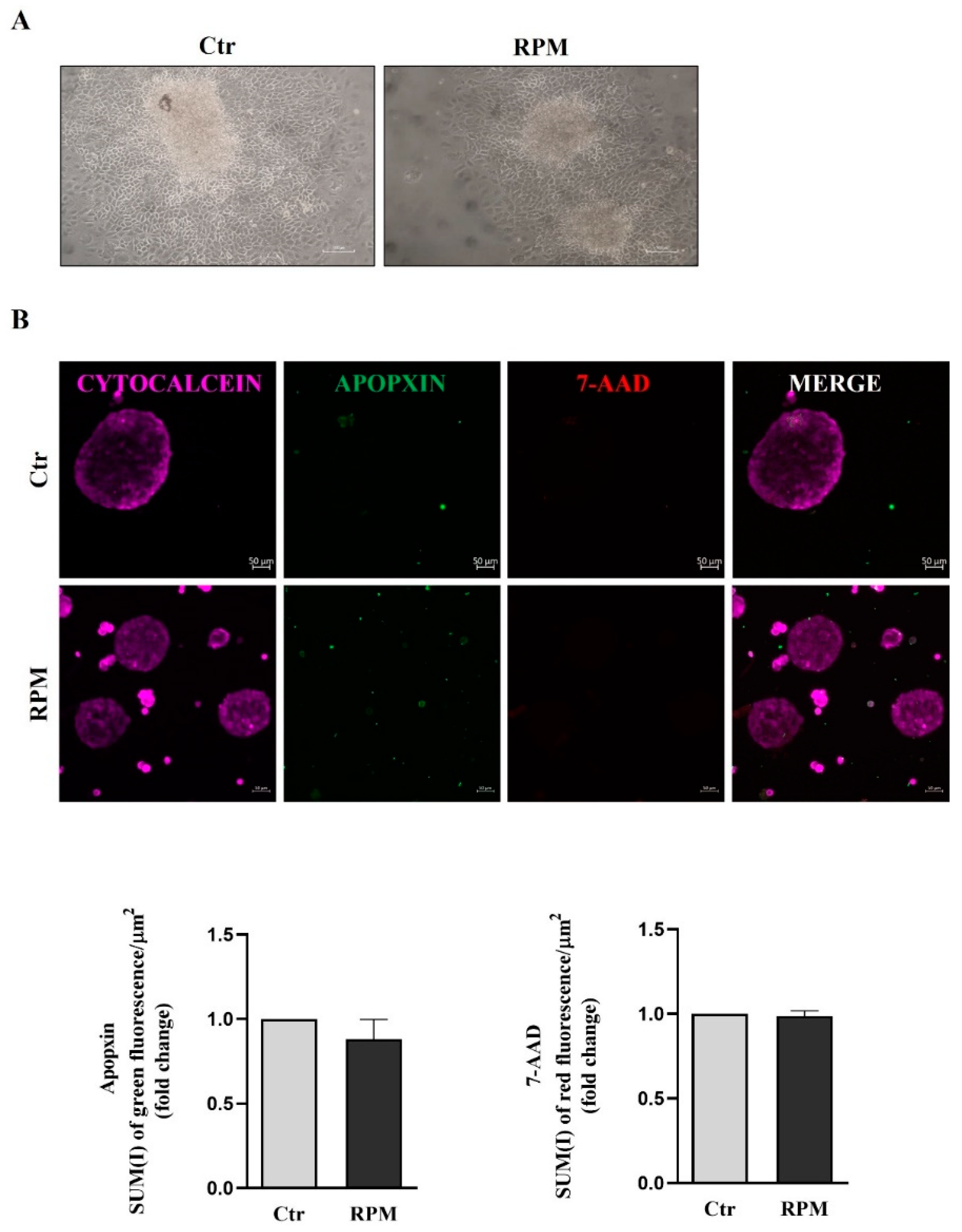
3.1. TCam-2 spheroids viability
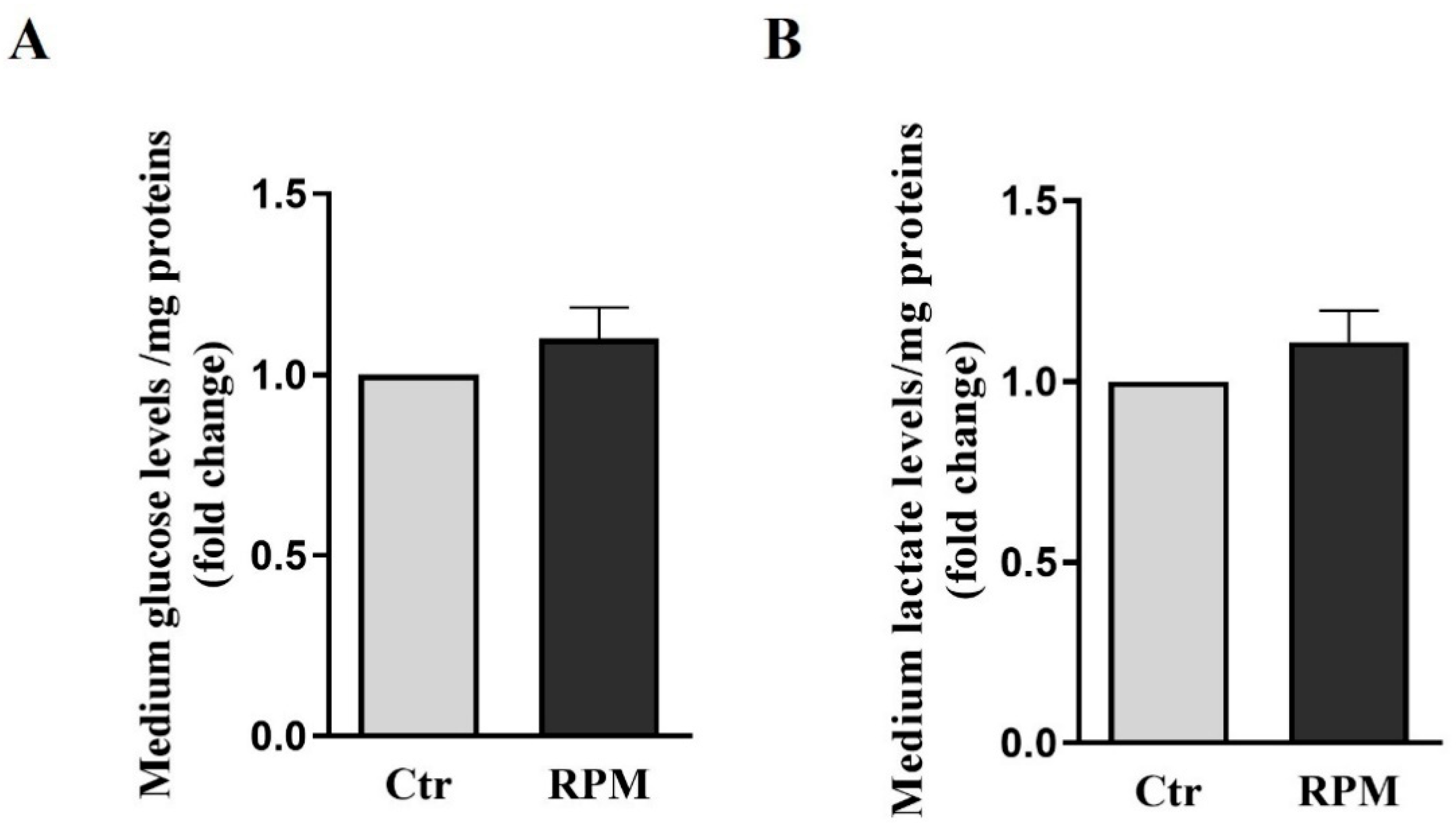
3.2. Metabolic features: glucose consumption and lactate production
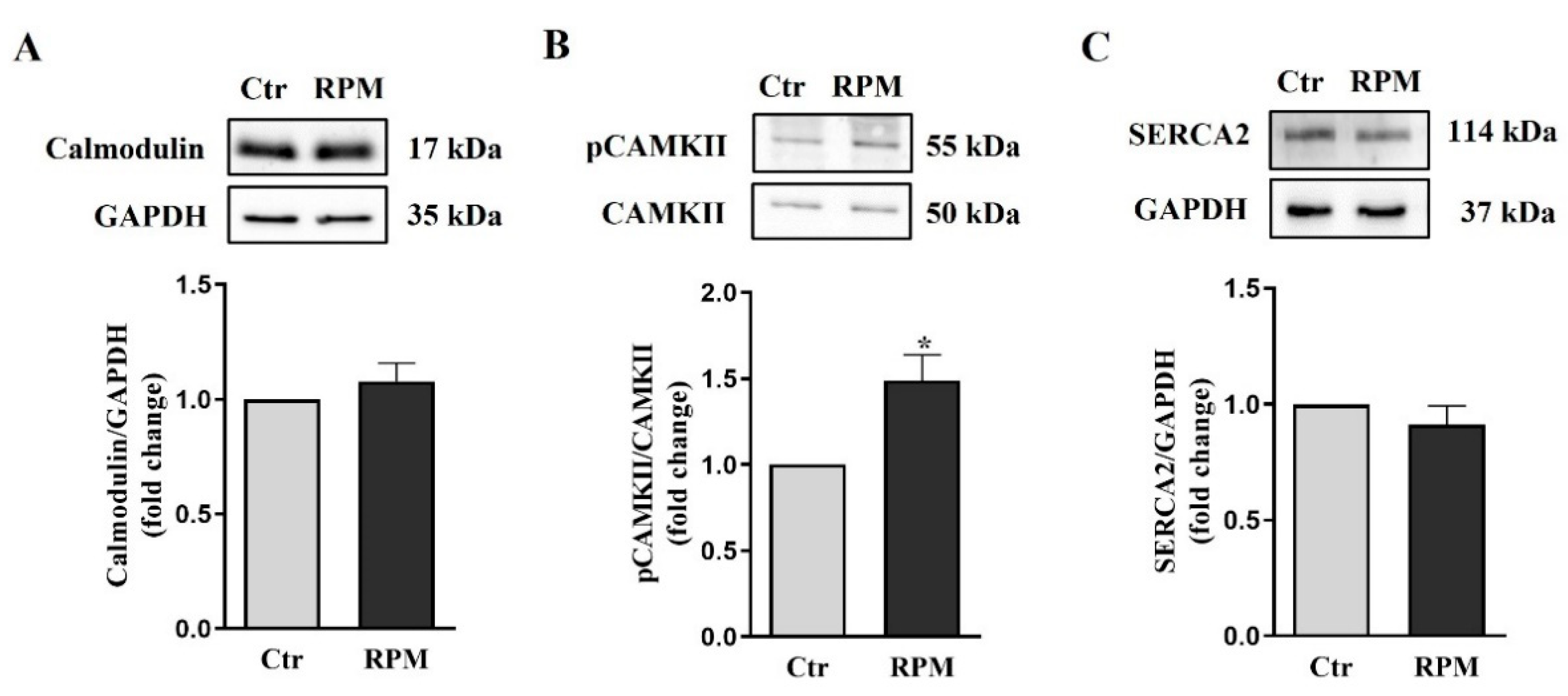
3.3. Intracellular calcium handling
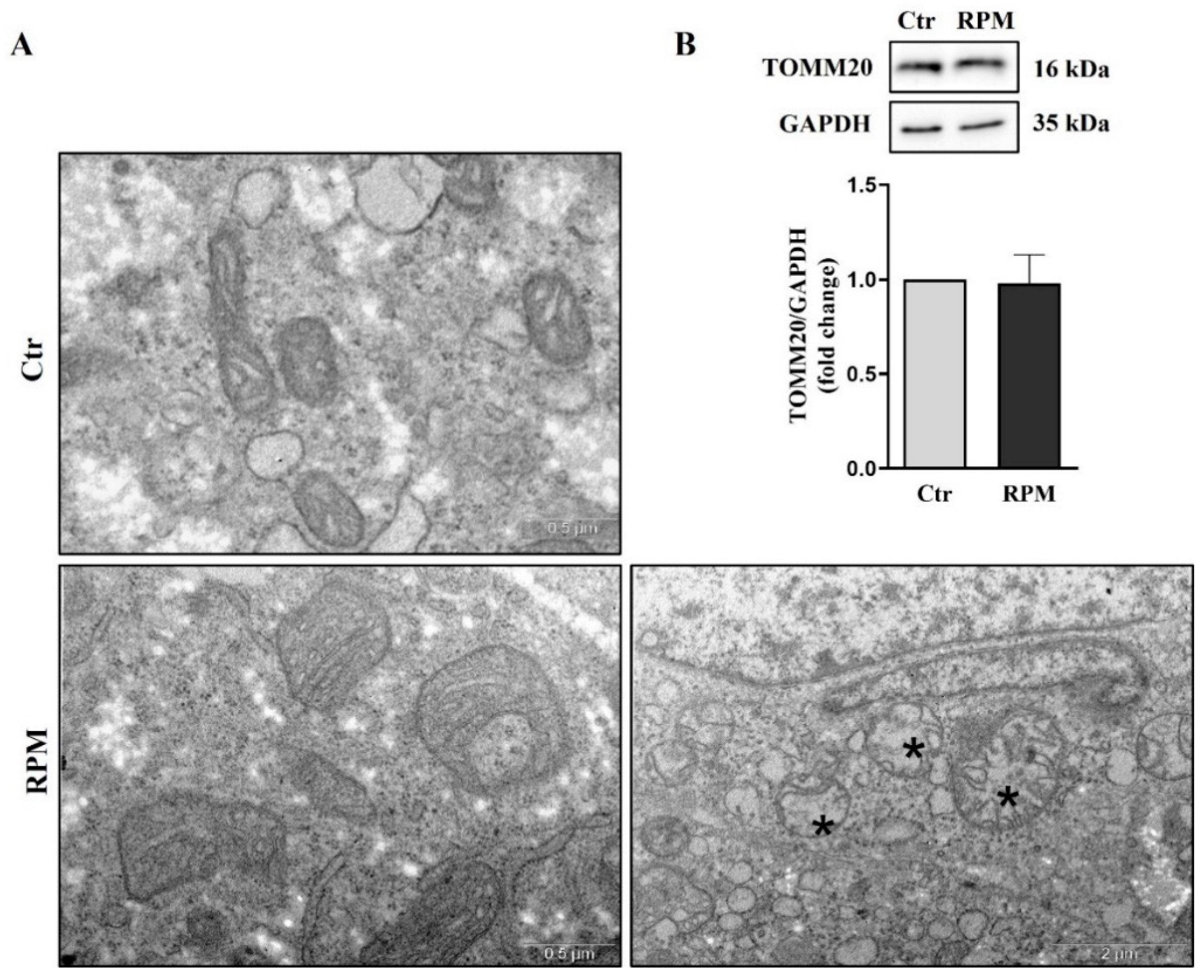
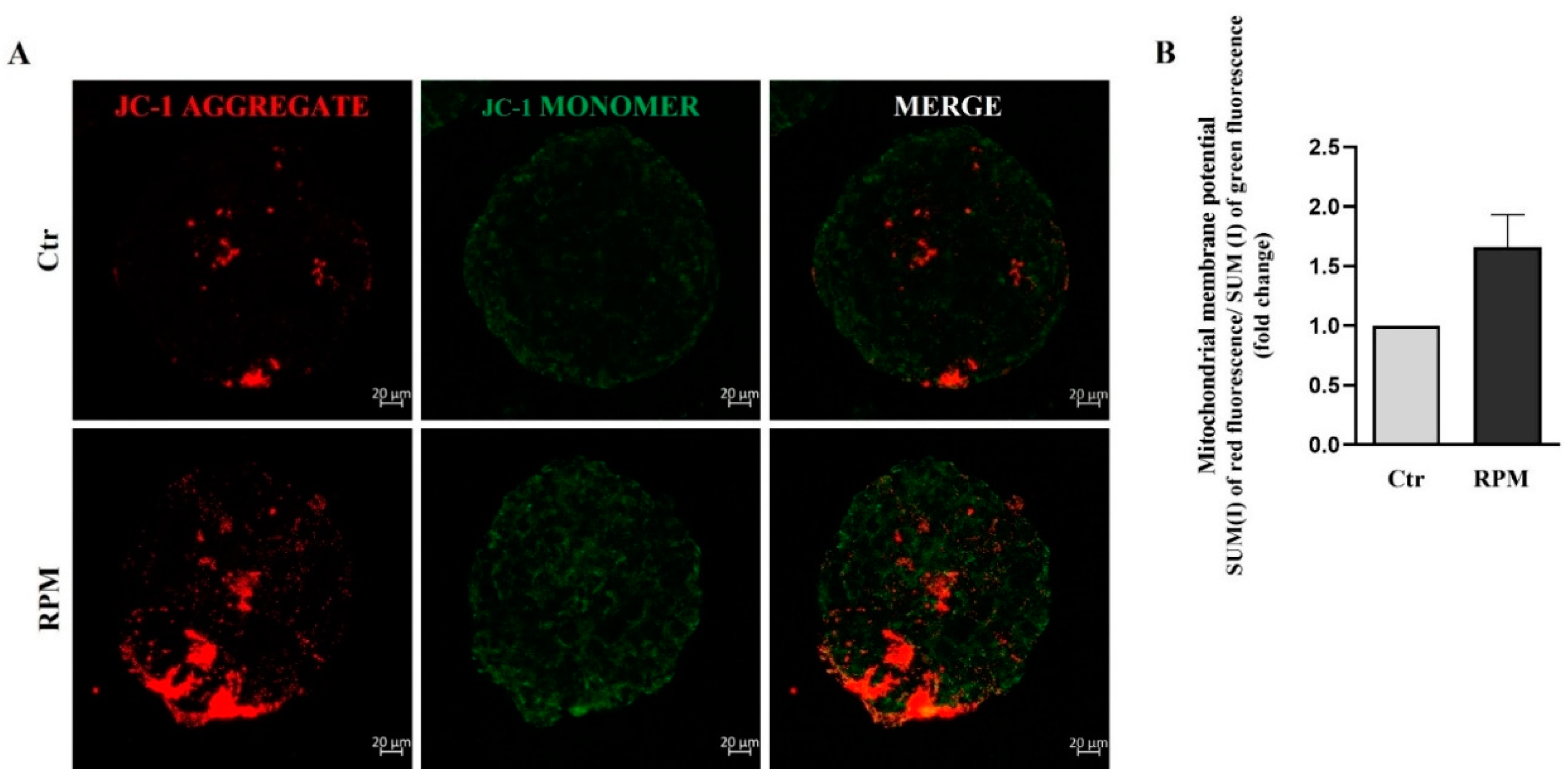
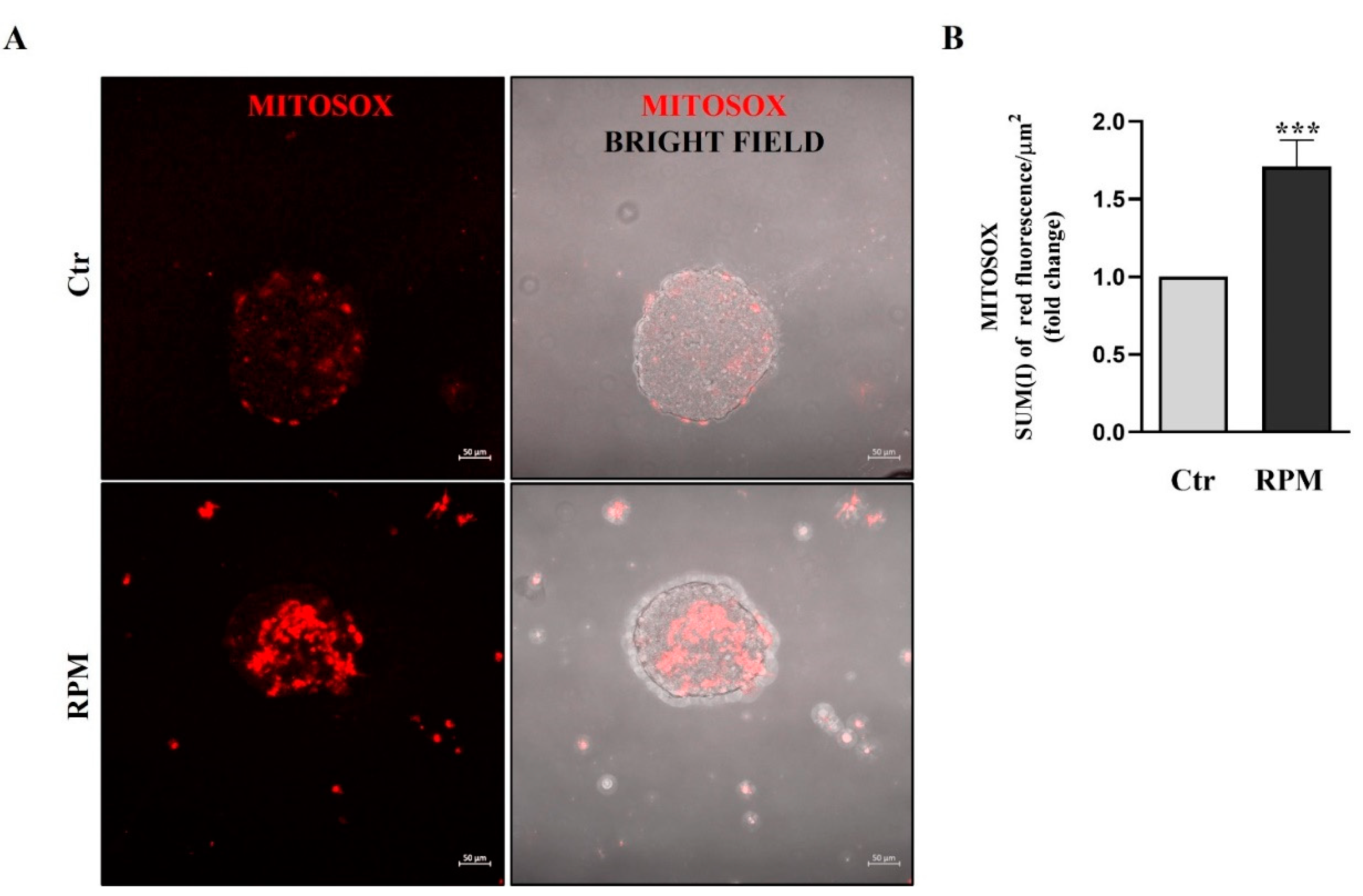
3.4. Mitochondria ultrastructure and functional features
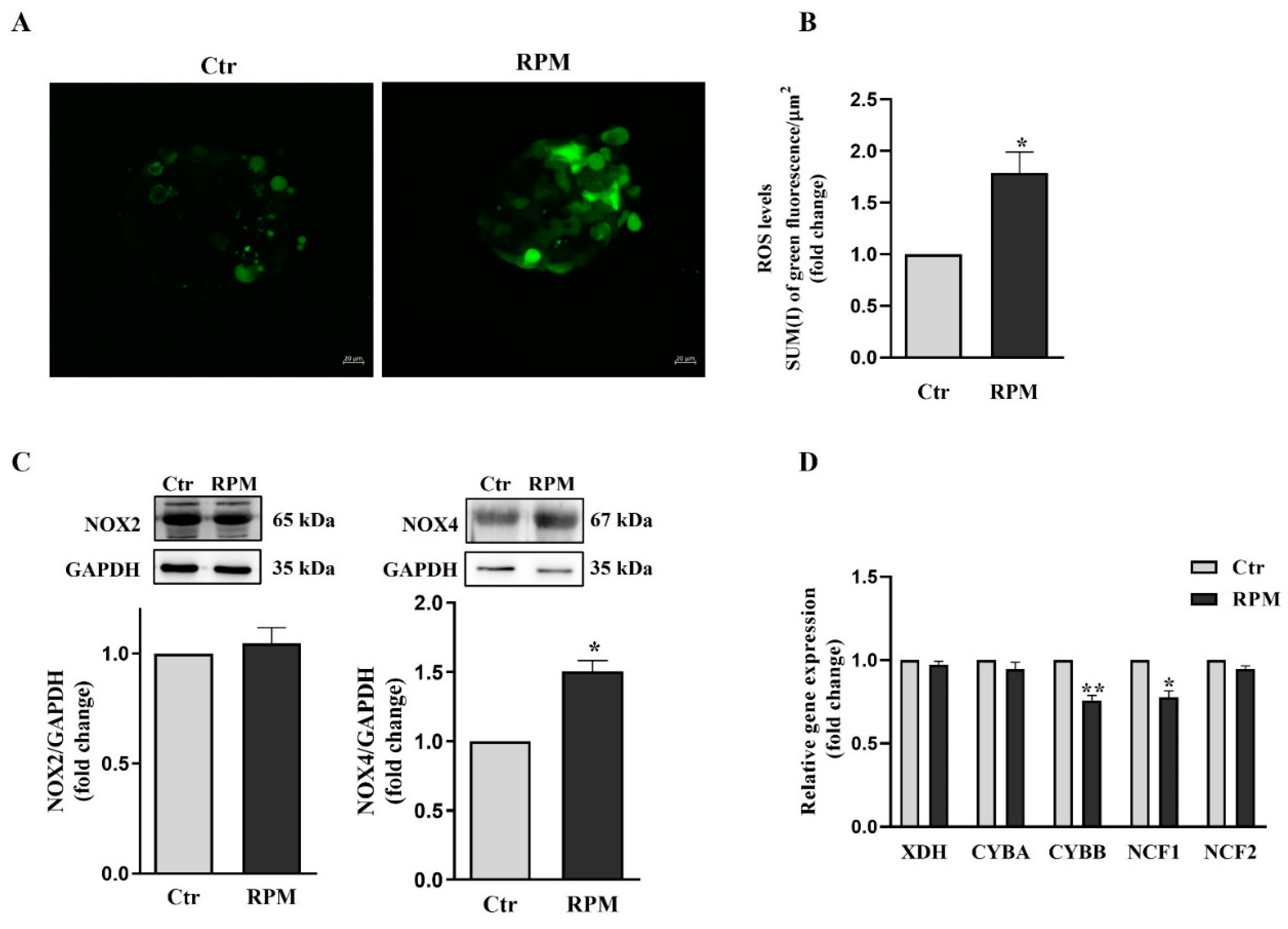
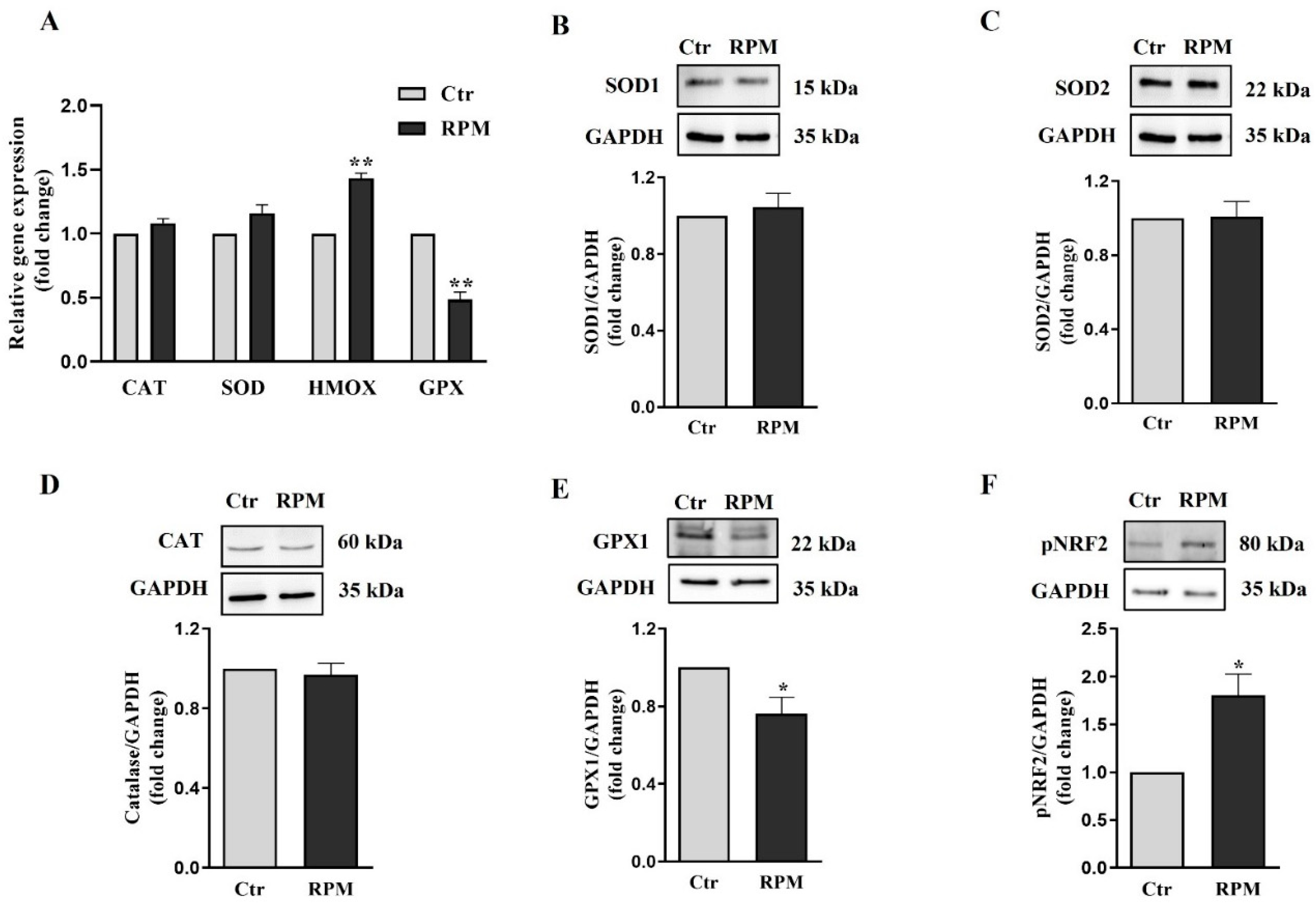
3.5. Oxidative balance in TCam-2 spheroids
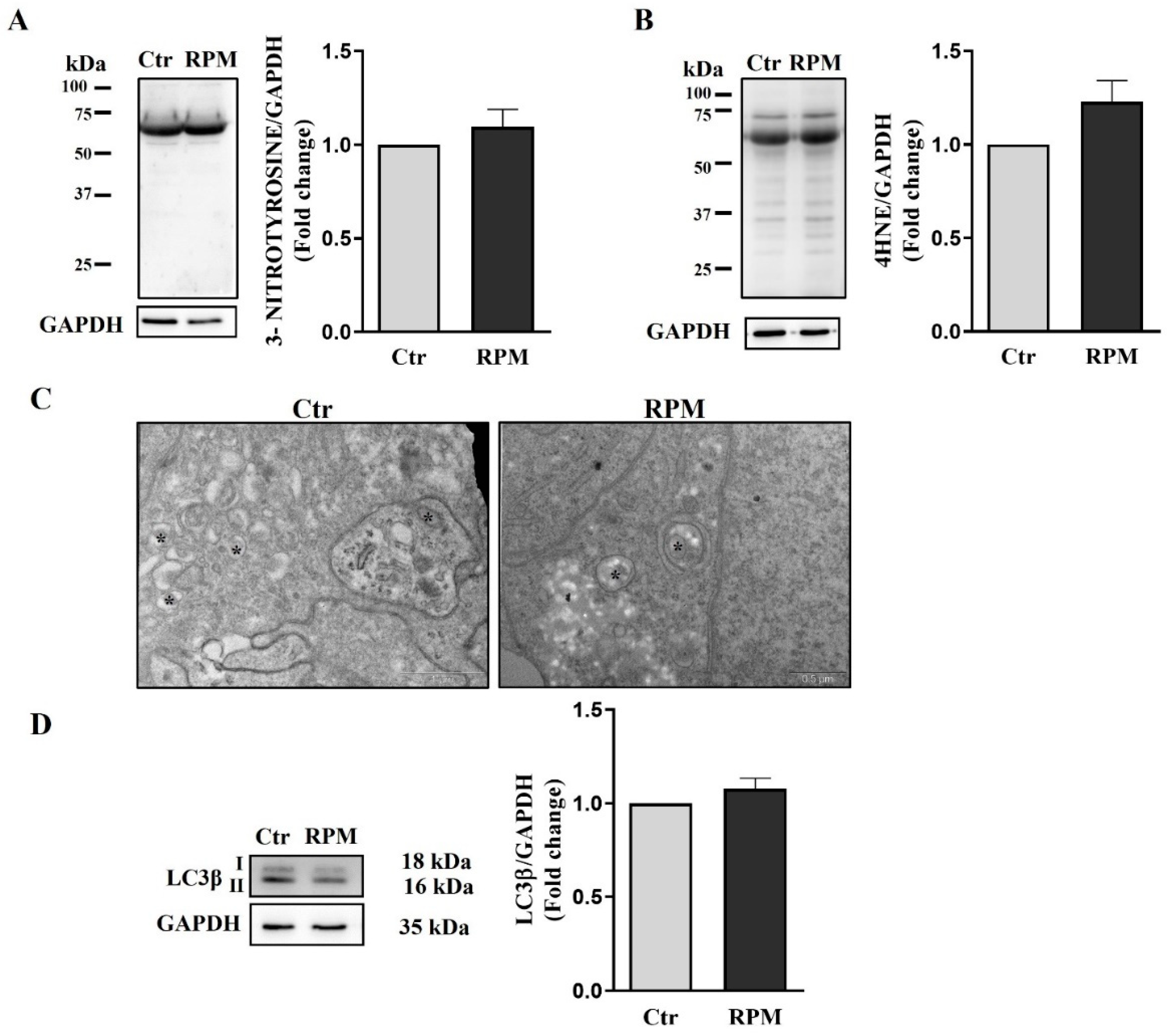
3.6. Oxidative damage and autophagy are not influenced by simulated microgravity exposure
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- B. Prasad et al., “Exploration of space to achieve scientific breakthroughs,” Biotechnology Advances, vol. 43, no. January, 2020. [CrossRef]
- Z. S. Patel et al., “Red risks for a journey to the red planet: The highest priority human health risks for a mission to Mars,” npj Microgravity, vol. 6, no. 1, pp. 1–13, 2020. [CrossRef]
- M. Löbrich and P. A. Jeggo, “Hazards of human spaceflight,” Science, vol. 364, no. 6436, pp. 127–128, 2019. [CrossRef]
- E. Afshinnekoo et al., “Fundamental Biological Features of Spaceflight: Advancing the Field to Enable Deep-Space Exploration,” Cell, vol. 183, no. 5, pp. 1162–1184, 2020. [CrossRef]
- R. L. Cromwell, J. L. Huff, L. C. Simonsen, and Z. S. Patel, “Earth-Based Research Analogs to Investigate Space-Based Health Risks,” New Space, vol. 9, no. 4, pp. 204–216, 2021. [CrossRef]
- F. Ferranti, M. Del Bianco, and C. Pacelli, “Advantages and limitations of current microgravity platforms for space biology research,” Applied Sciences (Switzerland), vol. 11, no. 1, pp. 1–18, 2021. [CrossRef]
- D. Ingber, “How cells (might) sense microgravity,” The FASEB Journal, vol. 13, no. 9001, 1999. [CrossRef]
- H. P. Nguyen, P. H. Tran, K. S. Kim, and S. G. Yang, “The effects of real and simulated microgravity on cellular mitochondrial function,” npj Microgravity, vol. 7, no. 1, 2021. [CrossRef]
- F. Ran, L. An, Y. Fan, H. Hang, and S. Wang, “Simulated microgravity potentiates generation of reactive oxygen species in cells,” Biophysics Reports, vol. 2, no. 5–6, pp. 100–105, 2016. [CrossRef]
- S. Dikalov, “Cross talk between mitochondria and NADPH oxidases,” Free Radical Biology and Medicine, vol. 51, no. 7, pp. 1289–1301, 2011. [CrossRef]
- H. Sies and D. P. Jones, “Reactive oxygen species (ROS) as pleiotropic physiological signalling agents,” Nature Reviews Molecular Cell Biology, vol. 21, no. 7, pp. 363–383, 2020. [CrossRef]
- M. Ighodaro and O. A. Akinloye, “First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid,” Alexandria Journal of Medicine, vol. 54, no. 4, pp. 287–293, 2018. [CrossRef]
- G. Napolitano, G. Fasciolo, and P. Venditti, “Mitochondrial management of reactive oxygen species,” Antioxidants, vol. 10, no. 11, pp. 1–29, 2021. [CrossRef]
- K. Yoshida et al., “Intergenerational effect of short-term spaceflight in mice,” iScience, vol. 24, no. 7, p. 102773, 2021. [CrossRef]
- T. Matsumura et al., “Male mice, caged in the International Space Station for 35 days, sire healthy offspring,” Scientific Reports, vol. 9, no. 1, pp. 1–8, 2019. [CrossRef]
- R. T. Jennings and P. A. Santy, “Reproduction in the space environment: Part II. Concerns for human reproduction.,” Obstetrical & gynecological survey, vol. 45, no. 1, pp. 7–17, Jan. 1990. [CrossRef]
- K. Ahrari, T. S. Omolaoye, N. Goswami, H. Alsuwaidi, and S. S. du Plessis, “Effects of space flight on sperm function and integrity: A systematic review,” Frontiers in Physiology, vol. 13, no. August, pp. 1–14, 2022. [CrossRef]
- M. Boada et al., “Microgravity effects on frozen human sperm samples,” Journal of Assisted Reproduction and Genetics, vol. 37, no. 9, pp. 2249–2257, 2020. [CrossRef]
- T. Ikeuchi et al., “Human sperm motility in a microgravity environment,” Reproductive Medicine and Biology, vol. 4, no. 2, pp. 161–168, 2005. [CrossRef]
- M. Pellegrini et al., “Microgravity promotes differentiation and meiotic entry of postnatal mouse male germ cells,” PLoS One, vol. 5, no. 2, 2010. [CrossRef]
- S. Di Agostino, F. Botti, A. Di Carlo, C. Sette, and R. Geremia, “Meiotic progression of isolated mouse spermatocytes under simulated microgravity,” Reproduction, vol. 128, no. 1, pp. 25–32, 2004. [CrossRef]
- L. H. Yan et al., “Simulated microgravity conditions and carbon ion irradiation induce spermatogenic cell apoptosis and sperm DNA damage,” Biomedical and Environmental Sciences, vol. 26, no. 9, pp. 726–734, 2013. [CrossRef]
- J. A. Hadley, J. C. Hall, A. O’Brien, and R. Ball, “Effects of a simulated microgravity model on cell structure and function in rat testis and epididymis,” Journal of Applied Physiology, vol. 72, no. 2, pp. 748–759, Feb. 1992. [CrossRef]
- G. Ricci, R. Esposito, A. Catizone, and M. Galdieri, “Direct effects of microgravity on testicular function: Analysis of hystological, molecular and physiologic parameters,” Journal of Endocrinological Investigation, vol. 31, no. 3, pp. 229–237, 2008. [CrossRef]
- R. J. Aitken, “Impact of oxidative stress on male and female germ cells: implications for fertility.,” Reproduction (Cambridge, England), vol. 159, no. 4, pp. R189–R201, Apr. 2020. [CrossRef]
- M. Giebler, T. Greither, and H. M. Behre, “Differential regulation of PIWI-LIKE 2 expression in primordial germ cell tumor cell lines by promoter methylation,” Frontiers in Genetics, vol. 9, no. SEP, pp. 1–8, 2018. [CrossRef]
- C. Morabito, S. Guarnieri, A. Catizone, C. Schiraldi, G. Ricci, and M. A. Mariggiò, “Transient increases in intracellular calcium and reactive oxygen species levels in TCam-2 cells exposed to microgravity,” Scientific Reports, vol. 7, no. 1, pp. 1–11, 2017. [CrossRef]
- F. Ferranti et al., “Cytoskeleton modifications and autophagy induction in TCam-2 Seminoma cells exposed to simulated microgravity,” BioMed Research International, vol. 2014, 2014. [CrossRef]
- Q. Sun et al., “PER1 interaction with GPX1 regulates metabolic homeostasis under oxidative stress,” Redox Biology, vol. 37, no. August, 2020. [CrossRef]
- J. F. Reichard, G. T. Motz, and A. Puga, “Heme oxygenase-1 induction by NRF2 requires inactivation of the transcriptional repressor BACH1,” Nucleic Acids Research, vol. 35, no. 21, pp. 7074–7086, 2007. [CrossRef]
- Z. Meng et al., “HMOX1 upregulation promotes ferroptosis in diabetic atherosclerosis,” Life Sciences, vol. 284, no. September, p. 119935, 2021. [CrossRef]
- L. Vitek, J. Hinds Terry, D. Stec, and C. Tiribelli, “The physiology of bilirubin: health and disease equilibrium,” Trends in Molecular Medicine, vol. 29, Feb. 2023. [CrossRef]
- N. Meyer et al., “AT 101 induces early mitochondrial dysfunction and HMOX1 (heme oxygenase 1) to trigger mitophagic cell death in glioma cells,” Autophagy, vol. 14, no. 10, pp. 1693–1709, 2018. [CrossRef]
- Q. Ma, “Role of Nrf2 in oxidative stress and toxicity,” Annual Review of Pharmacology and Toxicology, vol. 53, no. 1, pp. 401–426, 2013. [CrossRef]
- F. He, X. Ru, and T. Wen, “NRF2, a transcription factor for stress response and beyond,” International Journal of Molecular Sciences, vol. 21, no. 13, pp. 1–23, 2020. [CrossRef]
- Q. Wang et al., “CaMKII oxidation is a critical performance/disease trade-off acquired at the dawn of vertebrate evolution,” Nature Communications, vol. 12, no. 1, pp. 1–17, 2021. [CrossRef]
- C. V. C. Junho, W. Caio-Silva, M. Trentin-Sonoda, and M. S. Carneiro-Ramos, “An Overview of the Role of Calcium/Calmodulin-Dependent Protein Kinase in Cardiorenal Syndrome,” Frontiers in Physiology, vol. 11, no. July, pp. 1–11, 2020. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



