Submitted:
12 July 2023
Posted:
13 July 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
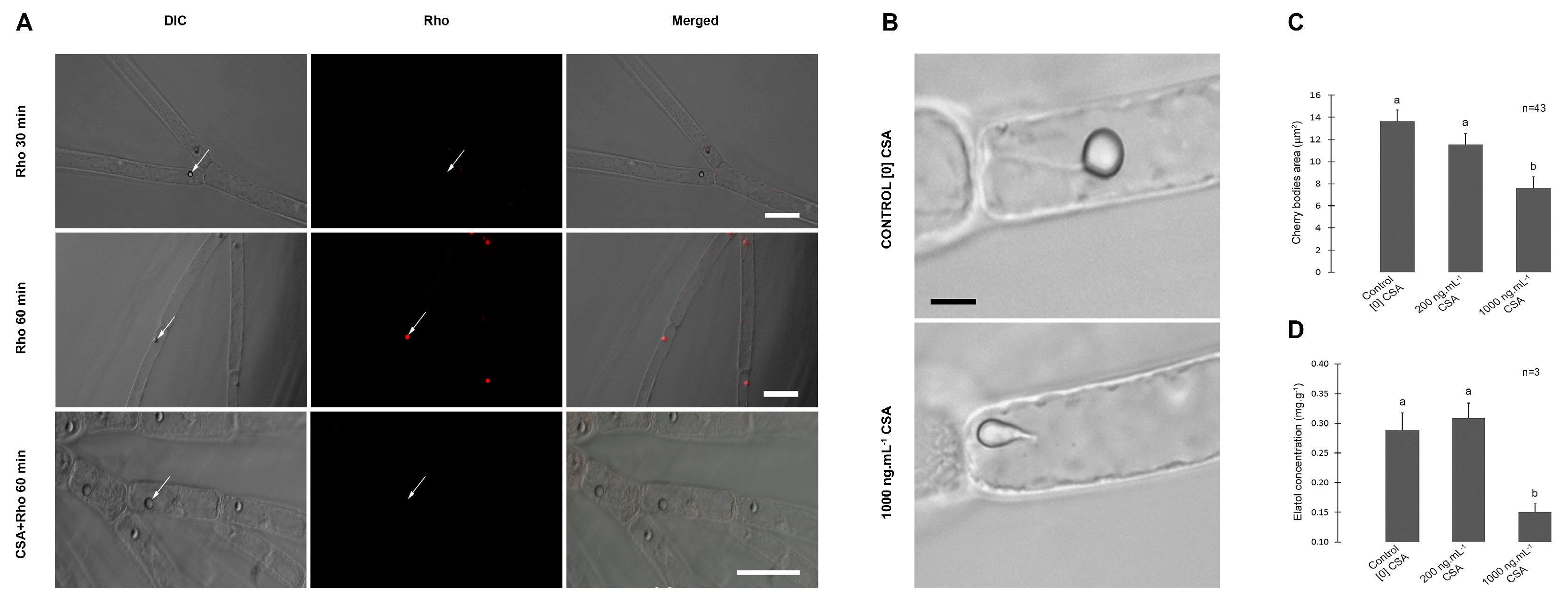
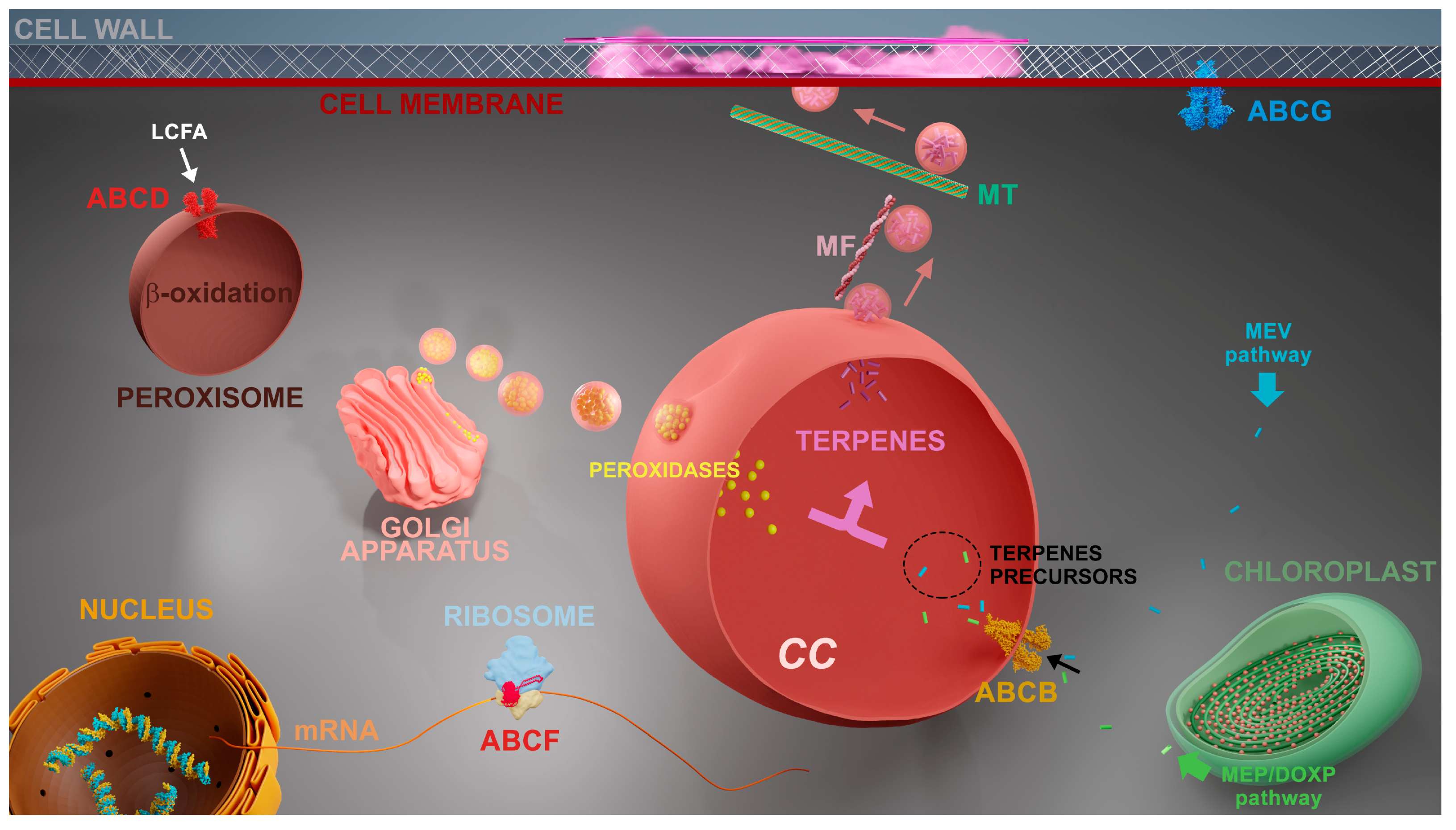
2.1. Intracellular localization of ABC proteins
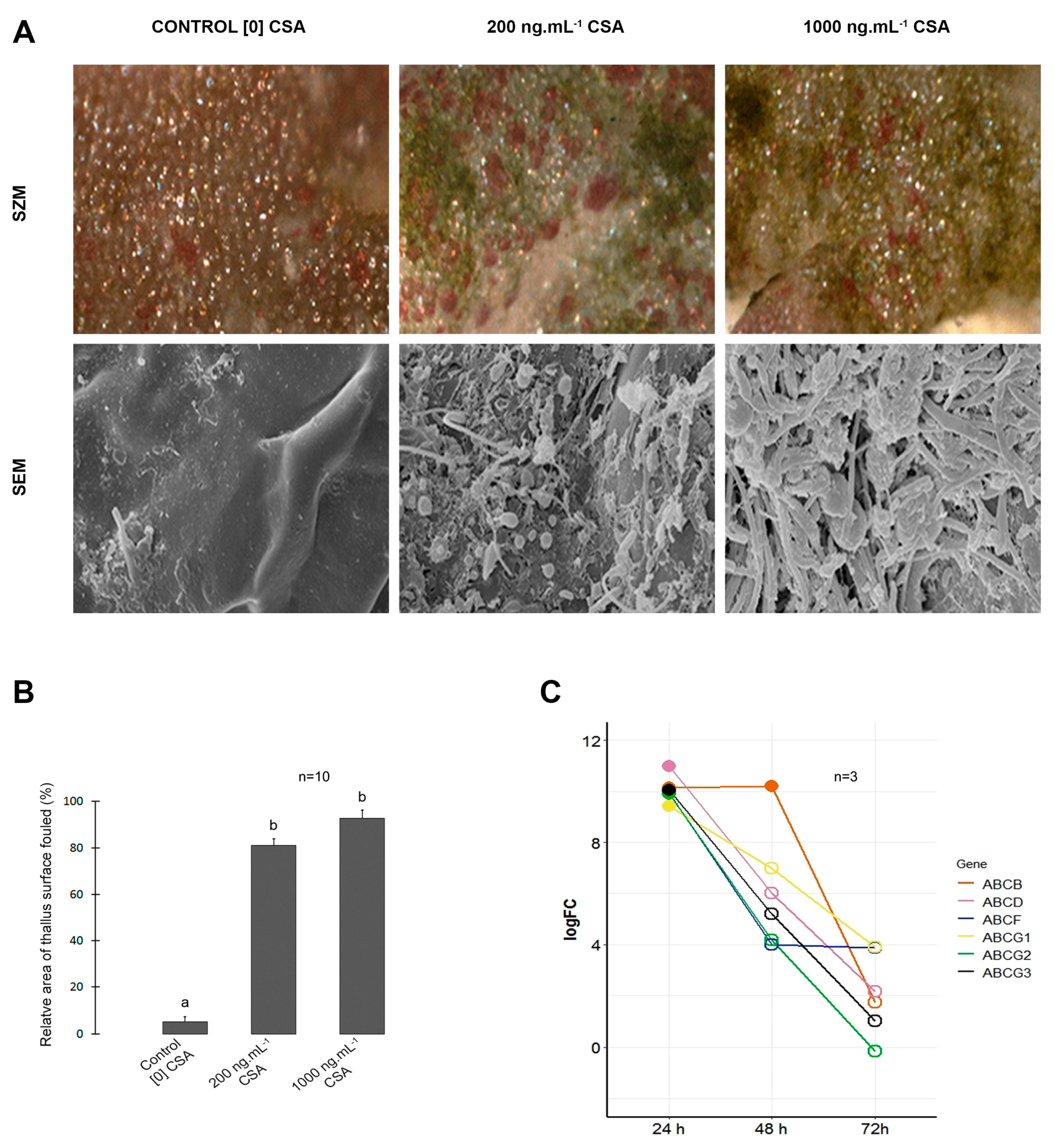
2.2. Morphology of corps en cerise (CC), elatol content and chemical defense
2.3. Statistical analysis
2.4. Expression level of ABC protein candidate genes
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Harizani, M.; Ioannou, E.; Roussis, V. The Laurencia paradox: an endless source of chemodiversity. Prog. Chem. Org. Nat. Prod. 2016, 102, 91–252. [Google Scholar] [CrossRef]
- Sudatti, D.B; Rodrigues, S.V.; Pereira, R.C. Quantitative GC-ECD analysis of halogenated metabolites: determination of surface and within-thallus elatol of Laurencia obtusa. J. Chem. Ecol. 2006, 32, 835–843. [Google Scholar] [CrossRef]
- Paul, N.A.; Cole, L.; De Nys, R.; Steinberg, P.D. Ultrastructure of the gland cells of the red alga Asparagopsis armata (Bonnemaisoniaceae). J. Phycol. 2006, 42, 637–645. [Google Scholar] [CrossRef]
- Paradas, W.C.; Crespo, T.M.; Salgado, L.T.; Andrade, L.R.; Soares, A.R.; Hellio, C.; Paranhos, R.R.; Hill, L.J.; Souza, G.M.; Kelecom, A.G.A.C.; et al. Mevalonosomes: specific vacuoles containing the mevalonate pathway in Plocamium brasiliense cortical cells (Rhodophyta). J. Phycol. 2015, 51, 225–235. [Google Scholar] [CrossRef]
- Young, D.N.; Howard, B.M.; Fenical, W. Subcellular localization of brominated secondary metabolites in the red alga Laurencia snyderae. J. Phycol. 1980, 16, 182–185. [Google Scholar] [CrossRef]
- Salgado, L.T.; Viana, N.B.; Andrade, L.R.; Leal, R.N.; Da Gama, B.A.P.; Pereira, R.C.; Amado Filho, G.M. Intra-cellular storage, transport and exocytosis of halogenated compounds in marine red alga Laurencia obtusa. J. Struct. Biol. 2008, 162, 345–355. [Google Scholar] [CrossRef]
- Reis, V.M.; Oliveira, L.S.; Passos, R.M.F.; Viana, N.B.; Mermelstein, C.; Sant’Anna, C.; Pereira, R.C.; Paradas, W.C.; Thompson, F.L.; Amado-Filho, G.M.; et al. Traffic of secondary metabolites to cell surface in the red alga Laurencia dendroidea depends on a two-step transport by the cytoskeleton. PLoS One 2018, 8, e63929. [Google Scholar] [CrossRef]
- Sudatti, D.B.; Rodrigues, S.V.; Coutinho, R.; Da Gama, B.A.P.; Salgado, L.T.; Amado-Filho, G.M.; Pereira, R.C. Transport and defensive role of elatol at the surface of the red seaweed Laurencia obtusa (Ceramiales, Rhodophyta). J. Phycol. 2008, 44, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Paradas, W.C.; Salgado, L.T.; Sudatti, D.B.; Crapez, M.A.; Fujii, M.T.; Coutinho, R.; Pereira, R.C.; Amado Filho, G.M. Induction of halogenated vesicle transport in cells of the red seaweed Laurencia obtusa. Biofouling 2010, 26, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.S.; Tschoeje, D.A.; Oliveira, A.S.; Hill, L.J.; Paradas, W.C.; Salgado, L.T.; Thompson, C.C.; Pereira, R.C.; Thompson, F.L. New Insights on the terpenome of the red seaweed Laurencia dendroidea (Florideophyceae, Rhodophyta). Mar. Drugs 2015, 13, 879–902. [Google Scholar] [CrossRef]
- Oliveira, L.S.; Tschoeke, D.A.; Lopes, A.C.R.M.; Sudatii, D.B.; Meirelles, P.M.; Thompson, C.C.; Pereira, R.C.; Thompson, F.L. Molecular mechanisms for microbe recognition and defense by the red seaweed Laurencia dendroidea. MSphere 2017, 2, e00094–17. [Google Scholar] [CrossRef]
- Brito, F.R.; Martinoia, E. The vacuolar transportome of plant specialized metabolites. Plant Cell Physiol. 2018, 59, 1326–1336. [Google Scholar] [CrossRef]
- Verrier, P.J.; Bird, D.; Burla, B.; Dassa, E.; Forestier, C.; Geisler, M.; Klein, M.; Kolukisaoglu, U.; Lee, Y.; Martinoia, E.; et al. Plant ABC proteins - a unified nomenclature and updated inventory. Trends Plant Sci. 2008, 13, 151–159. [Google Scholar] [CrossRef]
- Hwang, J.U.; Song, W.-Y.; Hong, D.; Ko, D.; Yamaoka, Y.; Jang, S.; Yim, S.; Lee, E.; Khare, D.; Kim, K.; et al. Plant ABC transporters enable many unique aspects of a terrestrial plant’s lifestyle. Mol. Plant. 2016, 9, 338–355. [Google Scholar] [CrossRef]
- Shitan, N. Secondary metabolites in plants: Transport and self-tolerance mechanisms. Biosci. Biotechnol. Biochem. 2016, 80, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Lane, T.S.; Rempe, C.S.; Davitt, J.; Station, M.E.; Peng, Y.; Soltis, D.E.; Melkonian, M.; Deyholos, M.; Leebens-Mack, C.M.; Rothfels, C.J.; et al. Diversity of ABC transporter genes across the plant kingdom and their potential utility in biotechnology. BMC Biotechnol. 2016, 16, 47. [Google Scholar] [CrossRef] [PubMed]
- Ritter, A.; Dittami, S.M.; Goultquer, S.; Correa, J.A.; Boyen, C.; Potin, P.; Tonon, T. Transcriptomic and metabolomic analysis of copper stress acclimation in Ectocarpus siliculosus highlights signaling and tolerance mechanisms in brown algae. BMC Plant Biol. 2014, 14, 116. [Google Scholar] [CrossRef]
- Fierro, C.; Lópes-Cristoffanini, C.; Meynard, A.; Lovazzano, C.; Castañeda, F.; Guajardo, E.; Conttreras-Porcia, L. Expression profile of desiccation tolerance factors in intertidal seaweed species during the tidal cycle. Planta 2017, 245, 1149–1164. [Google Scholar] [CrossRef] [PubMed]
- Sudatti, D.B.; Duarte, H.M.; Soares, A.R.; Salgado, L.T.; Pereira, R.C. New ecological role of seaweed secondary metabolites as autotoxic and allelopathic. Front. Plant Sci. 2020, 11, 347. [Google Scholar] [CrossRef]
- Qadir, M.; O’Loughlin, K.L.; Fricke, S.M.; Williamson, N.A.; Greco, W.R.; Minderman, H.; Baer, M.R. Cyclosporin A is a broad-spectrum multidrug resistance modulator. Clin. Cancer Res. 2005, 11, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Demissie, Z.A.; Tarnowycz, M.; Adai, A.M.; Sarjer, L. A lavender ABC transporter confers resistance to monoterpene toxicity in yeast. Planta 2019, 249, 139–144. [Google Scholar] [CrossRef]
- Otani, M.; Shitan, N.; Sakai, K.; Martinoia, E.; Sato, F.; Yazaki, K. Characterization of vacuolar transport of the endogenous alkaloid berberine in Coptis japonica. Plant Physiol. 2005, 138, 1939–1946. [Google Scholar] [CrossRef] [PubMed]
- Charton, L.; Plett, A.; Linka, N. Plant peroxisomal solute transporter proteins. J. Integr. Plant Biol. 2019, 61, jipb–12790. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre, F.; Boutry, M. Towards identification of the substrates of ATP-binding cassette transporters. Plant Physiol. 2018, 178, 18–39. [Google Scholar] [CrossRef] [PubMed]
- Forster, S.; Thumser, A.E.; Hood, N.; Plant, N. Characterization of Rhodamine-123 as a tracer dye for use in in vitro drug transport assays. PLoS One 2012, 7, e33253. [Google Scholar] [CrossRef]
- Schindelin, J.; Rueden, C.T.; Hiner, M.C.; Eliceiri, K.W. The ImageJ ecosystem: An open platform for biomedical image analysis. Mol. Rep. Dev. 2015, 82, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.P.B.; Duytschaever, G.; Tonon, L.A.C.; Dias, G.M.; Mesquita, M.; Cnockaert, M.; Francini-Filho, R.B.; De Vos, P.; Thompson, C.C.; Thompson, F.L. Vibrio madracius sp. nov. isolated from Madracis decactis (Scleractinia) in St Peter & St Paul Archipelago, Mid-Atlantic Ridge, Brazil. Curr. Microbiol. 2014, 69, 405–411. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z; Thomspn, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef]
- Finn, R.D.; Clements, J.; Eddy, S.R. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 2011, 39, W29–W37. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. EdgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 139–140. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



