Submitted:
03 July 2023
Posted:
03 July 2023
You are already at the latest version
Abstract
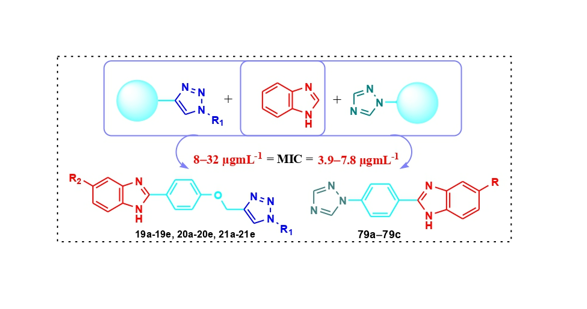
Keywords:
1. Introduction
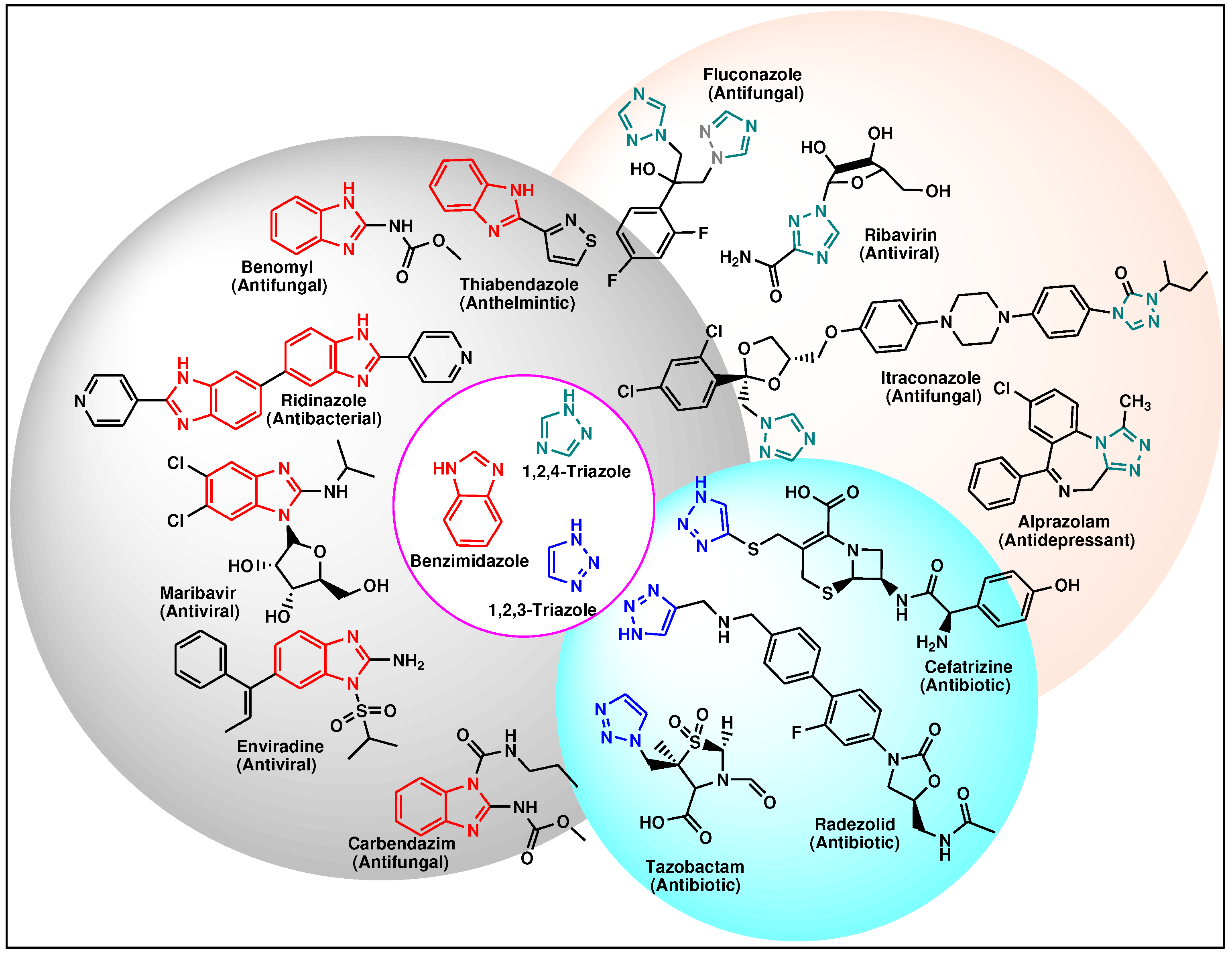
2. Synthesis and antimicrobial activities of benzimidazole-1,2,3-triazoles
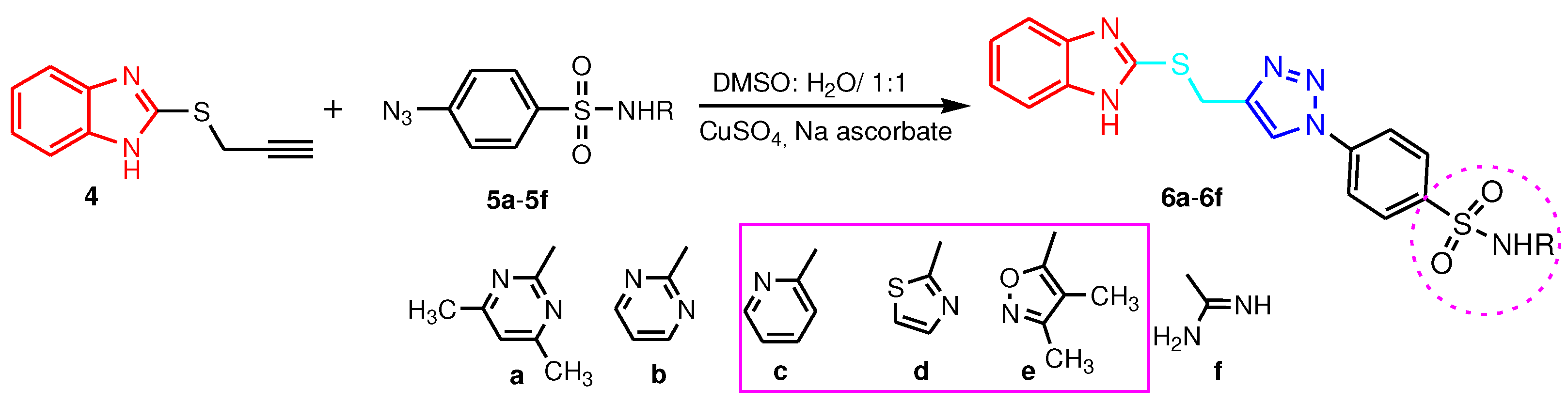
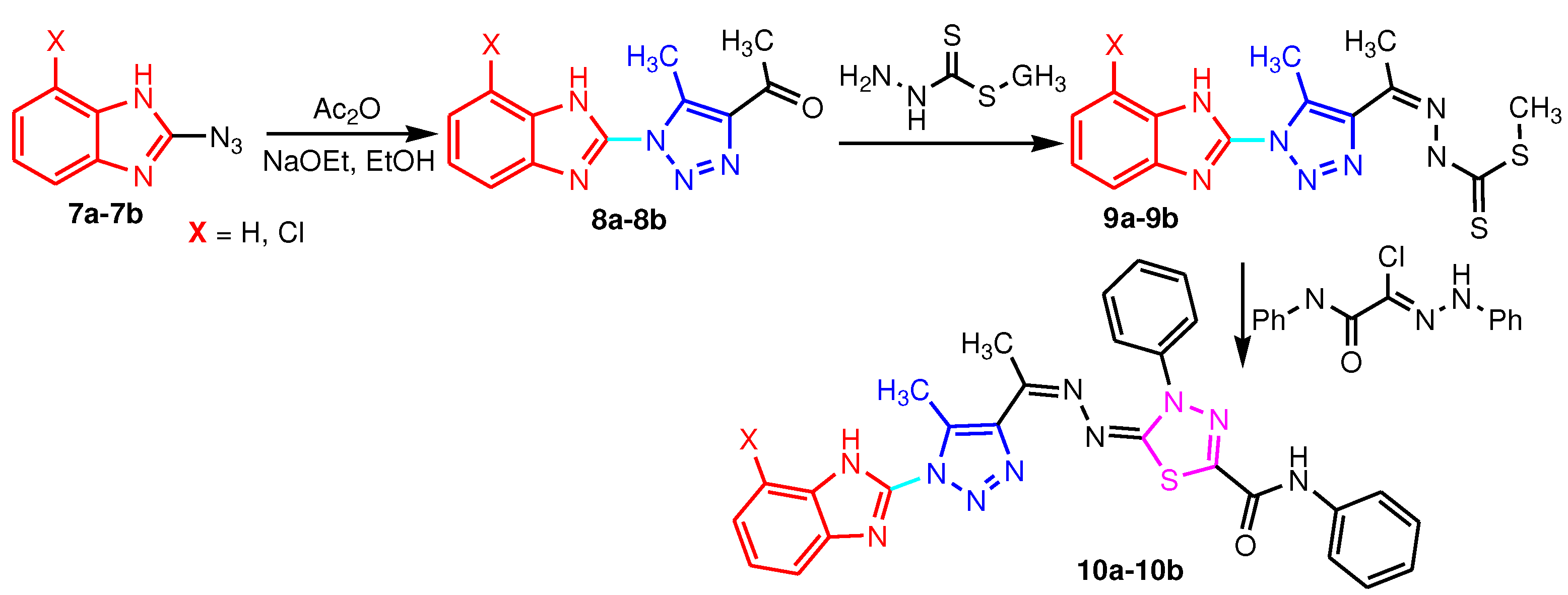
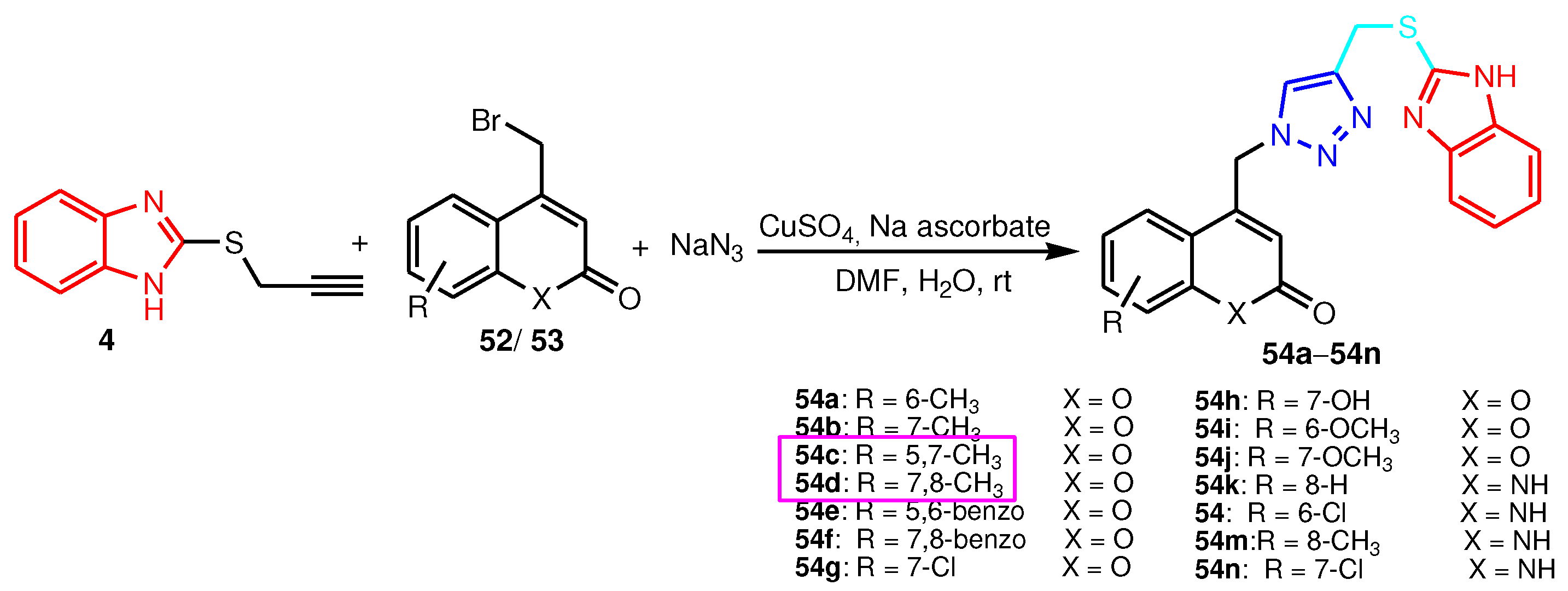
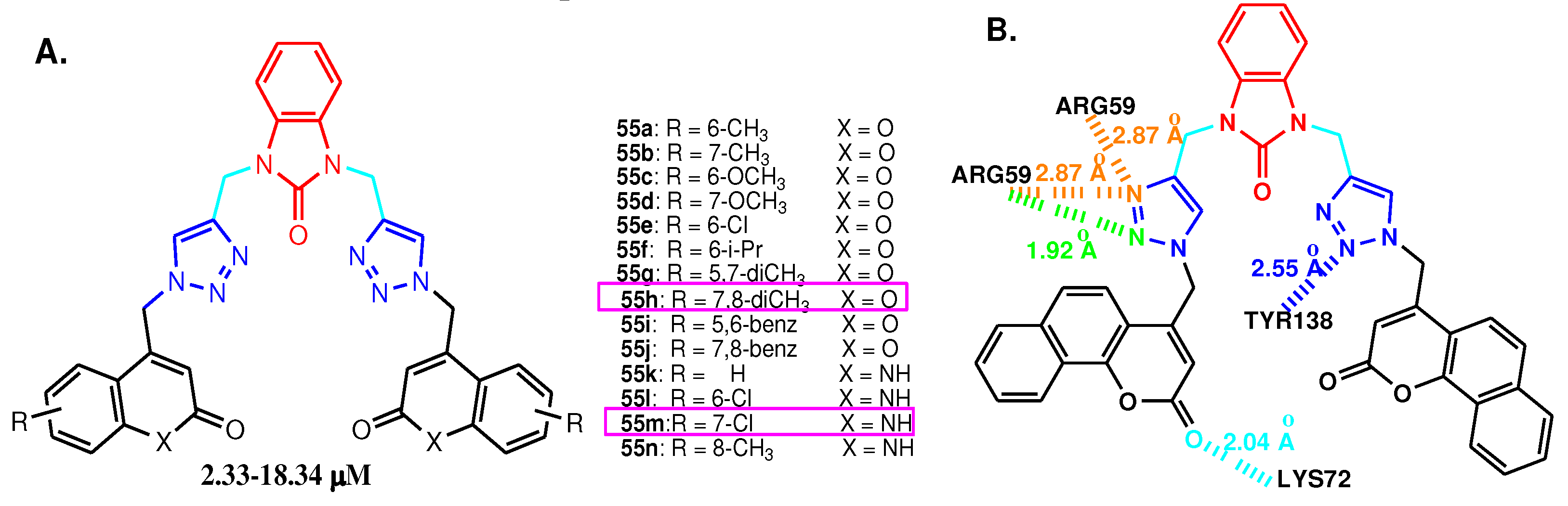
2.1. 2-Benzimidazole-R(Ar)-1,4-disubstituted-1,2,3-triazole hybrids
| Hybrids | Inhibition zone diameters using the agar diffusion method (mm) | ||||
|---|---|---|---|---|---|
| S. aureus | E. coli | P. aeruginosa | A. niger | C. albicans | |
| 8a | 15 ± 0.14 | 12 ± 1.08 | 22 ± 1.01 | - | - |
| 8b | - | 5 ± 0.2 | - | 30 ± 1.16 | 27 ± 1.1 |
| 9a | 23 ± 0.8 | - | 13 ± 0.65 | - | - |
| 9b | - | - | 12 ± 0.8 | 14 ± 0.15 | 19 ± 1.04 |
| 10a | 24 ± 0.6 | 25 ± 0.9 | 17 ± 0.75 | 20 ± 0.9 | 16 ± 0.89 |
| 10b | 29 ± 1.2 | 21 ± 1.14 | 19 ± 0.79 | 18 ± 0.12 | 14 ± 0.58 |
| Ciprofloxacin | 20 ± 0.9 | 23 ± 1.02 | 21 ± 0.9 | - | - |
| Nystatin | - | - | - | 22 ± 0.18 | 23 ± 1.15 |
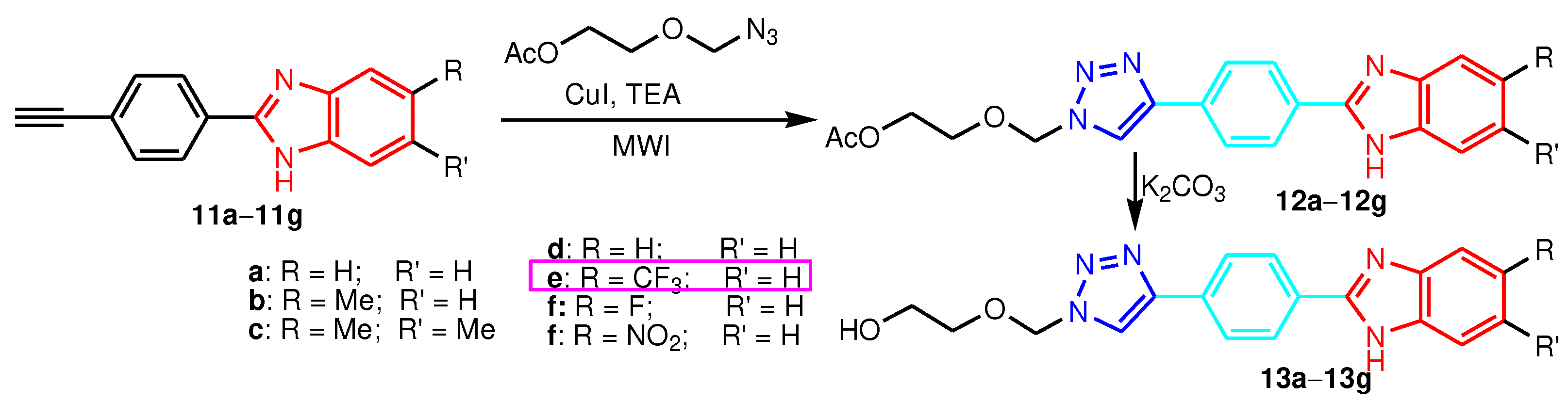
2.2. 1-Benzimidazole-R(Ar)-1,4-disubstituted-1,2,3-triazole hybrids
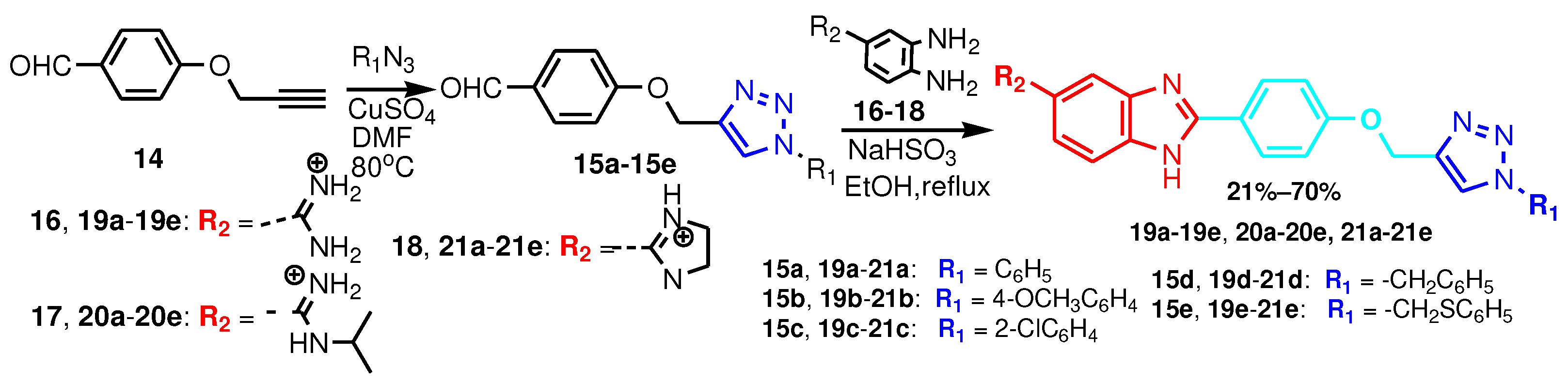
2.3. 1,2-. bis-substitutedBenzimidazoles-R(Ar)-1,4-disubstituted-1,2,3-triazole
2.4. Benzimidazole-R(Ar)-1,2,3-triazole hybrids as antitubecular agents
3. Synthesis and antimicrobial activities of benzimidazole-1,2,4-triazoles
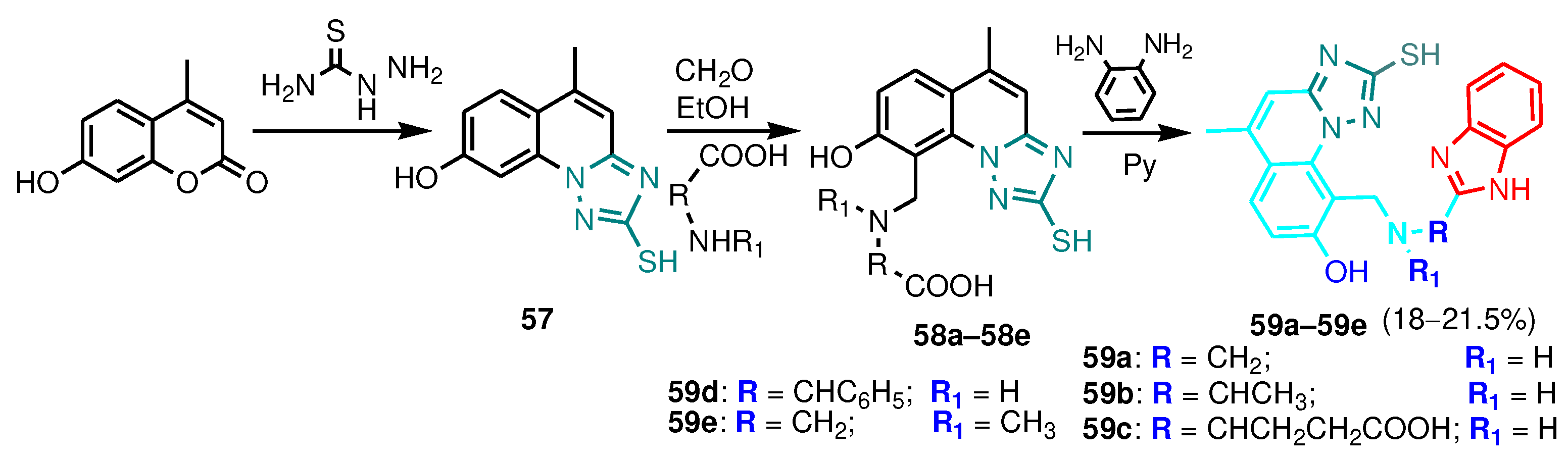
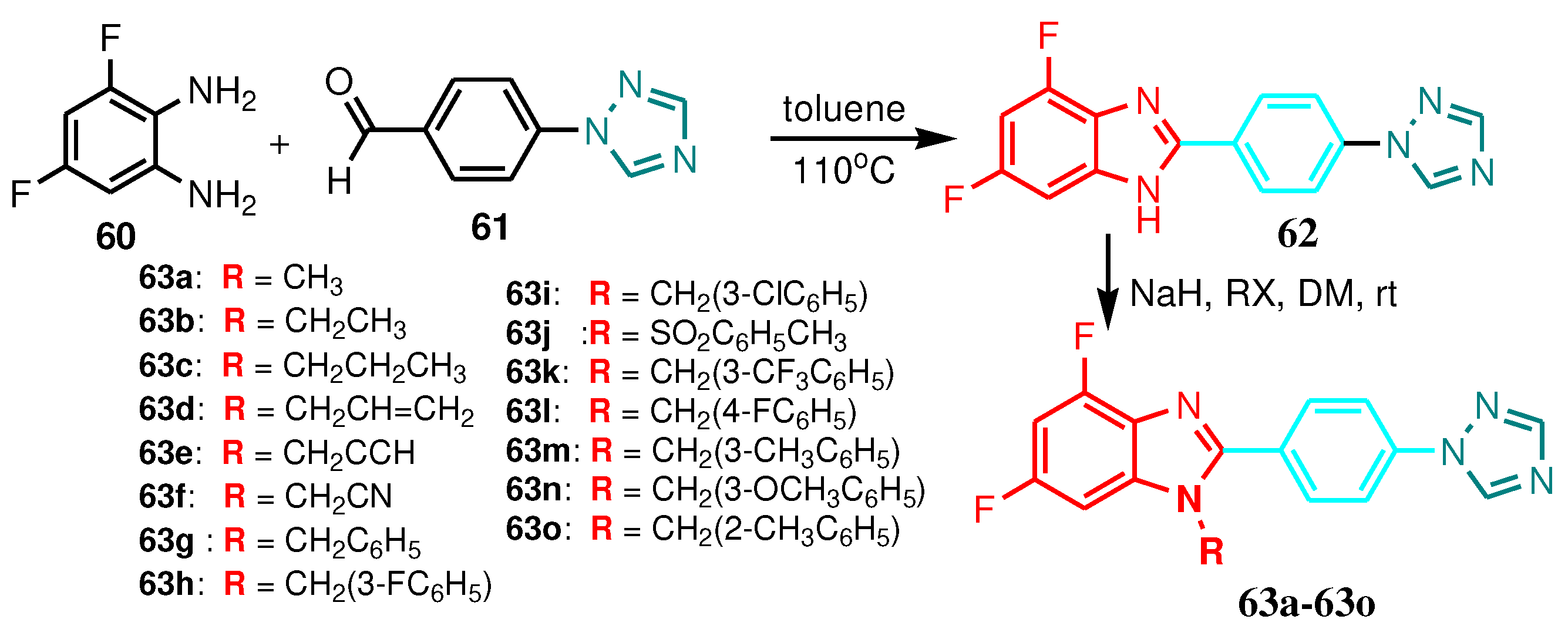
3.1. 2-Benzimidazole-R(Ar)-1-(1,2,4-triazole)
3.2. 1-Benzimidazole-R(Ar)-2-1,2,3-triazole
3.3. 2-Benzimidazole-R(Ar)-2-1,2,4-triazole
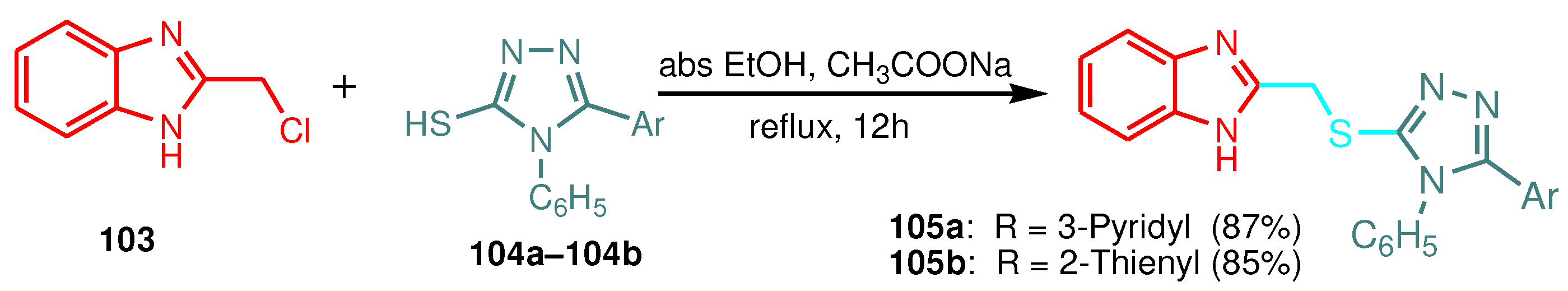
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kabir, E.; Uzzaman,M. A review on biological and medicinal impact of heterocyclic compounds. Results in Chemistry 2022, 100606. [Google Scholar] [CrossRef]
- Nishanth Rao, R.; Jena, S.; Mukherjee, M.; Maiti, B; Chanda, K. Green synthesis of biologically active heterocycles of medicinal importance: a review. Environ. Chem. Lett. 2021, 19, 3315–3358. [Google Scholar] [CrossRef]
- Ebenezer, O.; Oyetunde-Joshua, F.; Omotoso, O.D.; Shapi, M. Benzimidazole and its derivatives: Recent Advances (2020–2022). Results in Chemistry 2023, 5, 100925. [Google Scholar] [CrossRef]
- Bansal, Y.; Kaur, M.; Bansal, G. Antimicrobial Potential of Benzimidazole Derived Molecules. Mini. Rev. Med. Chem. 2019, 19(8), 624–646. [Google Scholar] [CrossRef] [PubMed]
- Marinescu, M. Synthesis of Antimicrobial Benzimidazole–Pyrazole Compounds and Their Biological Activities. Antibiotics 2021, 10, 1002. [Google Scholar] [CrossRef]
- Deswal, L.; Verma, V.; Kumar, D.; Deswal, Y.; Kumar, A. ; Kumar, R; Parshad, M., Ed.; Bhatia, M. Synthesis, antimicrobial and α-glucosidase inhibition of new benzimidazole-1,2,3-triazole-indoline derivatives: a combined experimental and computational venture. Chem. Pap. 2022, 76, 7607–7622. [Google Scholar] [CrossRef]
- Raducka, A.; Świątkowski, M.; Korona-Głowniak, I.; Kaproń, B.; Plech, T.; Szczesio, M.; Gobis, K.; Szynkowska-Jóźwik, M.I.; Czylkowska, A. Zinc Coordination Compounds with Benzimidazole Derivatives: Synthesis, Structure, Antimicrobial Activity and Potential Anticancer Application. Int. J. Mol. Sci. 2022, 23, 6595. [Google Scholar] [CrossRef]
- Zalaru, C.; Dumitrascu, F.; Draghici, C.; Tarcomnicu, I.; Marinescu, M.; Nitulescu, G.M.; Tatia, R.; Moldovan, L.; Popa, M.; Chifiriuc, M.C. New Pyrazolo-Benzimidazole Mannich Bases with Antimicrobial and Antibiofilm Activities. Antibiotics 2022, 11, 1094. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Su, S.; Zhou, Q.; Tang, X.; Liu, T. , Peng, F.; He, M.; Luo, H.; Xue, W. Antibacterial and antiviral activities and action mechanism of flavonoid derivatives with a benzimidazole moiety. J. Saudi Chem. Soc. 2021, 25, 101194. [Google Scholar] [CrossRef]
- Kanwal, A.; Ahmad, M.; Aslam, S.; Naqvi, S.A.R.; Jawwad Saif, M.J. Molecular-biological prolems of drug design and mehanism of drug action. Recent advances n antiviral benyimidayole derivatives: a mini review. Pharm. Chem. J. 2019, 53 (3), 179-187. [CrossRef]
- Brishty, S.R.; Hossain, Md. J.; Khandaker, M.U.; Faruque, M.R.I.; Osman, H.; Rahman, S.M.A. A Comprehensive Account on Recent Progress in Pharmacological Activities of Benzimidazole Derivatives. Front. Pharmacol. 2021, 12, 762807. [Google Scholar] [CrossRef]
- Yhou, S.; Huang, G. Synthesis of anti-allergic drugs. RSC Adv. 2020, 10, 5874. [Google Scholar] [CrossRef]
- Vasil’ev, P. M.; Kalitin, K. Yu.; Spasov, A. A.; Grechko, O. Yu.; Poroikov, V. V.; Filimonov, D. A.; Anisimova, V. A. Search for new dugs prediction and study of anticonvulsant properties of benzimidazole derivatives. Pharm. Chem. J. 2017, 50(12), 775–780. [Google Scholar] [CrossRef]
- Lee, Y.T.; Tan, YJ.; OOn, C.E. Benzimidazole and its derivatives as cancer therapeutics: The potential role from traditional to precision medicine. Acta Pharmaceutica Sinica B 2023, 13(2), 478–497. [Google Scholar] [CrossRef] [PubMed]
- Satija, G.; Sharma, B.; Madan,A.; Iqubal, A; Shaquiquzzaman, M.; Akhter, M.; Parvez, S.; Khan, M.A.; Alam, M.M. Benzimidazole based derivatives as anticancer agents: Structure activity relationship analysis for various targets. J. Heterocyclic. Chem. 2021, 59 (1), 22-66. [CrossRef]
- Song, B.; Park, E.Y.; Kim, K.J.; Ki, S.H. Repurposing of Benzimidazole Anthelmintic Drugs as Cancer Therapeutics. Cancers 2022, 14, 4601. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.L.; Li, D.D.; Chen, X.; Wang, Y.Z.; Xu, J.-J.; Jiang, Z.-Y.; You, Q.-D.; Guo, X.-K. Proton pump inhibitors selectively suppress MLL rearranged leukemia cells via disrupting MLL1-WDR5 protein-protein interaction. Eur. J. Med. Chem. 2020, 188, 112027. [Google Scholar] [CrossRef]
- Argirova,M.A.; Georgieva, M.K.; Hristova-Avakumova, N.G.; Vuchev, D.I.; Popova-Daskalova, G.V.; Anichina, K.K.; Yancheva, D.Y. New 1H-benzimidazole-2-yl hydrazones with combined antiparasitic and antioxidant activity. RSC Adv. 2021, 11, 39848–39868. [CrossRef]
- Valderas-García, E.; Häberli, C.; Álvarez-Bardón, M.; Escala, N.; Castilla-Gómez de Agüero, V.; de la Vega, J.; del Olmo, E.; Balaña-Fouce, R.; Keiser, J.; Martínez-Valladares, M. Benzimidazole and aminoalcohol derivatives show in vitro anthelmintic activity against Trichuris muris and Heligmosomoides polygyrus. Parasites & Vectors. [CrossRef]
- Kamat, V.; Yallur, B.C.; Poojary, B.; Patil, V.B.; Nayak, S.P.; Krishna, P.M.; Joshi, S.D. Synthesis, molecular docking, antibacterial, and anti-inflammatory activities of benzimidazole-containing tricyclic systems. J. Chin. Chem. Soc. 2021, 68(6), 1055–1066. [Google Scholar] [CrossRef]
- Moharana, A.K.; Dash, R.N.; Mahanandia, N.C.; Subudhi, B.B. Synthesis and anti-inflammatory activity evaluation of some benzimidazoled erivatives. Pharm. Chem. J. 2022, 56, 1070–1074. [Google Scholar] [CrossRef]
- Veerasamy, R.; Roy, A.; Karunakaran, R.; Rajak, H. Structure–Activity Relationship Analysis of Benzimidazoles as Emerging Anti-Inflammatory Agents: An Overview. Pharmaceuticals 2021, 14, 663. [Google Scholar] [CrossRef]
- Iqbal, H.; Verma, A.K.; Yadav, P.; Alam, S.; Shafiq, M.; Mishra, D.; Khan, F.; Hanif, K.; Negi, A.S.; Chanda, D. Antihypertensive Effect of a Novel Angiotensin II Receptor Blocker Fluorophenyl Benzimidazole: Contribution of cGMP, Voltage-dependent Calcium Channels, and BKCa Channels to Vasorelaxant Mechanisms. Front. Pharmacol. 2021, 30. [Google Scholar] [CrossRef]
- Tajane, P. S. , Sawant, R. L. An updated review on benzimidazole derivatives as potential antihypertensive agents. Int. J. Health Sci. 2022, 6( S1), 7169–7179. [Google Scholar] [CrossRef]
- Aboul-Enein, H.Y.; El Rashedy, A.A. Benzimidazole Derivatives as Antidiabetic Agents. Med. Chem. 2015, 5, 318–325. [Google Scholar] [CrossRef]
- Dik, B.; Coşkun, D.; Bahcivan, E.; Unez, K. Potential antidiabetic activity of benzimidazole derivative albendazole and lansoprazole drugs in different doses in experimental type 2 diabetic rats. Turk. J. Med. Sci. 2021, 51, 1578–1585. [Google Scholar] [CrossRef]
- Farid, S.M.: Noori, M.; Montazer, M.N.; Ghomi, M.K.; Mollazadeh, M.; Dastyafteh, N.; Irajie C.; Zomorodian, K.; Mirfazli, S.S.; Mojtabavi, S.; Faramarzi, M.A.; Larijani, B.; Iraji, A.; Mahdavi, M. Synthesis and structure–activity relationship studies of benzimidazole-thioquinoline derivatives as α-glucosidase inhibitors. Sci. Rep. 2023, 13, 4392. [CrossRef]
- Stanton, J.B.; Schneider, D.A.; Dinkel, K.D.; Balmer, B.F.; Baszler, T.V.; Mathison, B.A.; Boykin, D.W.; Kumar, A. Discovery of a Novel, Monocationic, Small-Molecule Inhibitor of Scrapie Prion Accumulation in Cultured Sheep Microglia and Rov Cells. Plos One 2012, 7 (11), | e51173. [CrossRef]
- Dinparast, L.; Zengin, G.; Bahadori, M.B. Cholinesterases Inhibitory Activity of 1H-benzimidazole Derivatives. Biointerface Res in Appl. Chem. 2021, 11. [Google Scholar] [CrossRef]
- Adalat, B.; Rahim, F.; Taha, M.; Alshamrani, F.J.; Anouar, E.H.; Uddin, N.; Shah, S.A.A.; Ali, Z.; Zakaria, Z.A. Synthesis of Benzimidazole–Based Analogs as Anti Alzheimer’s Disease Compounds and Their Molecular Docking Studies. Molecules 2020, 25, 4828. [Google Scholar] [CrossRef]
- Cheretaev, I. V.; Korenyuk, I. I.; Nozdrachev, A.D. Neurotropic, Psychoactive, and Analgesic Properties of Benzimidazole and Its Derivatives: Physiological Mechanisms. Neurosci. Behav. Physiol. 2018, 48(7), 848–853. [Google Scholar] [CrossRef]
- Maltsev, D.V.; Spasov, A.A.; Vassiliev, P.M.; Skripka, M.O.; Miroshnikov, M.V.; Kochetkov, A.N.; Eliseeva, N.V.; Lifanova, Y.V.; Kuzmenko, T.A.; Divaeva, L.N.; Morkovnik, A.S. Synthesis and Pharmacological Evaluation of Novel 2,3,4,5-tetrahydro[1,3]diazepino[1,2-a]benzimidazole Derivatives as Promising Anxiolytic and Analgesic Agents. Molecules 2021, 26, 6049. [Google Scholar] [CrossRef]
- Yen, T.L.; Wu, .P.; Chng, C.L.; Yang, W.B.; Jayakumar, T.; Geraldine, P.; Chou, C.M.; Chang, C.Y.; Lu, W.J.; Sheu, J.R. Novel synthetic benzimidazole-derived oligosaccharide, M3BIM, prevents ex vivo platelet aggregation and in vivo thromboembolism. J. Biomed. Sci. 2016, 23, 26. [CrossRef]
- Zhang, T.; Liu, Q.; Ren, Y. Design, synthesis and biological activity evaluation of novel methyl substituted benzimidazole derivatives. Tetrahedron 2020, 76. [Google Scholar] [CrossRef]
- Zhang, B. Comprehensive review on the anti-bacterial activity of 1,2,3-triazole hybrids. Eur. J. Med. Chem. 2019, 168, 357–372. [Google Scholar] [CrossRef]
- Strzelecka, M.; Świątek, P. 1,2,4-Triazoles as Important Antibacterial Agents. Pharmaceuticals 2021, 14, 224. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.A.; Nesaragi, A.R.; Rodríguez-Berrios, R.R.; Hampton, S.M.; Bugarin, A.; Patil, S.A. Coumarin Triazoles as Potential Antimicrobial Agents. Antibiotics 2023, 12, 160. [Google Scholar] [CrossRef]
- Kazeminejad, Z.; Marzi, M.; Shiroudi, A.; Kouhpayeh, S.A.; Farjam, M.; Zarenezhad, E. Novel 1, 2, 4-Triazoles as Antifungal Agents. Review. BioMed Res. Int. 2022, 4584846, 1–39. [Google Scholar] [CrossRef]
- Sharma, A.; Agrahari, A.K.; Rajkhowa, S.; Tiwari, V.K. Emerging impact of triazoles as anti-tubercular agent. Eur. J. Med.Chem. 2022, 238, 114454. [Google Scholar] [CrossRef] [PubMed]
- El-Shoukrofy, M.S.; Atta, A.; Fahmy, S.; Sriram, D.; Mahran, M.A.; Labouta, I.M. New tetrahydropyrimidine-1,2,3-triazole clubbed compounds: Antitubercular activity and Thymidine Monophosphate Kinase (TMPKmt) inhibition. Bioorg. Chem. 2023, 131, 106312. [Google Scholar] [CrossRef] [PubMed]
- Ravisankar, N.; Sarathi, N.; Maruthavanan, T.; Ramasundaram, S.; Ramesh, M.; Sankar, C.; Umamatheswari, S.; Kanthimathi, G.; Oh, T.H. Synthesis, antimycobacterial screening, molecular docking, ADMET prediction and pharmacological evaluation on novel pyran-4-one bearing hydrazone, triazole and isoxazole moieties: Potential inhibitors of SARS CoV-2. Synthesis, antimycobacterial screening, molecular docking, ADMET prediction and pharmacological evaluation on novel pyran-4-one bearing hydrazone, triazole and isoxazole moieties: Potential inhibitors of SARS CoV-2. J. Mol. Struct. 2023, 1285, 135461. [Google Scholar] [CrossRef]
- Musa, A.; Abulkhair, H.S.; Aljuhani, A.; Rezki, N.; Abdelgawad, M.A.; Shalaby, K.; El-Ghorab, A.H.; Aouad, M.R. Phenylpyrazolone-1,2,3-triazole Hybrids as Potent Antiviral Agents with Promising SARS-CoV-2 Main Protease Inhibition Potential. Pharmaceuticals 2023, 16, 463. [Google Scholar] [CrossRef]
- Seliem, I.A.; Panda, S.S.; Girgis, A.S.; Moatasim, Y.; Kandeil, A.; Mostafa, A.; Ali, M.A.; Nossier, E.S.; Rasslan, F.; Srour, A.M.; Sakhuja, R.; Ibrahim, T.S.; Abdel-samii, Z.K.M.; Al-Mahmoudy, A.M.M. New quinoline-triazole conjugates: Synthesis, and antiviral properties against SARS-CoV-2. Bioorg. Chem. 2021, 114, 105117. [Google Scholar] [CrossRef] [PubMed]
- Venkatesham, P.; Schols, D.; Persoons, L.; Claes, S.; Sangolkar, A.A.; Chedupaka, R.; Vedula, R.R. Synthesis of novel thioalkylated triazolothiazoles and their promising in-vitro antiviral activity. J. Mol. Struct. 2023, 1286, 135573. [Google Scholar] [CrossRef]
- Pinheiro, N.G.; Gonzaga, D.T.G.; da Silva, A.R.; Fuly, A.C.; von Ranke, N.L.; Rodrigues, C.R.; Magalhães, B.Q.; Pereira, J.S.; Pacheco, P.A.F.; Silva, A.C.; Ferreira, V.F.; de Carvalho da Silva, F.; Faria, R.X. Triazoles with inhibitory action on P2X7R impaired the acute inflammatory response in vivo and modulated the hemostatic balance in vitro and ex vivo. Inflamm. Res. 2023, 72, 237–250. [Google Scholar] [CrossRef]
- Demchenko, S.; Lesyk, R.; Yadlovskyi, O.; Holota, S.; Yarmoluk, S.; Tsyhankov, S.; Demchenko, A. Fused Triazole-Azepine Hybrids as Potential Non-Steroidal Antiinflammatory Agents. Sci. Pharm. 2023, 91, 26. [Google Scholar] [CrossRef]
- Bozorov, K.; Zhao, J.; Aisa, H.A. 1,2,3-Triazole-containing hybrids as leads in medicinal chemistry: A recent overview. Bioorg. Med. Chem. 2019, 27, 3511–3531. [Google Scholar] [CrossRef]
- Hashem, H.E.; Amr, A. E-G.E.; Nossier, E.S.; Anwar,, M.M.; Azmy, E.M. New Benzimidazole-1,2,4-Triazole-, and 1,3,5-Triazine-Based Derivatives as Potential EGFRWT and EGFRT790M Inhibitors: Microwave-Assisted Synthesis, Anticancer Evaluation, and Molecular Docking Study. ACS Omega 2022, 7, 7155–7171. [Google Scholar] [CrossRef]
- Othman, D.I.A.; Hamdi, A.; Tawfik, S.S.; Elgazar, A.A.; Mostafa, A.S. Identification of new benzimidazole-triazole hybrids as anticancer agents: multi-target recognition, in vitro and in silico studies. J. Enzyme Inhib. Med. Chem. 2023, 38. [Google Scholar] [CrossRef]
- Gupta, O.; Pradhan, G, T. Chawla, G. An updated review on diverse range of biological activities of 1,2,4-triazole derivatives: Insight into structure activity relationship. J. Mol. Struct. 2023, 1274, Part 2, 134487. [CrossRef]
- Abu-Melha, S.; Azher, O.A.; Alaysuy, O.; Alnoman, R.B.; Abualnaja, M.M.; Althagafi, I.; El-Metwaly, N.M. Synthesis, molecular modeling and antioxidant activity of new thiadiazolyl-triazole analogues. J. Saudi. Chem. Soc. 2023, 27(2), 101596. [Google Scholar] [CrossRef]
- Dawbaa, S.; Nuha, D.; Evren, A.E.; Cankiliç, M.Y.; Yurttaş, L.; Turan, G. New oxadiazole/triazole derivatives with antimicrobial and antioxidant properties. J. Mol. Struct. 2023, 1282, 135213. [Google Scholar] [CrossRef]
- Zhao, W.; Song, M.; Hua, Y.; Zhu, Y.; Liu, W.; Xia, Q.; Deng, X.; Huang, Y. Design, Synthesis, and Pharmacology of New Triazole-Containing Quinolinones as CNS Active Agents. Molecules 2023, 28, 1987. [Google Scholar] [CrossRef]
- Dixit, D.; Verma, P.K.; Marwaha, R.K. A review on ‘triazoles’: their chemistry, synthesis and pharmacological potentials. J. Iran. Chem. Soc. 2021, 18, 2535–2565. [Google Scholar] [CrossRef]
- Fallah, Z.; Tajbakhsh, M.; Alikhani, M.; Larijani, B.; Faramarzi, M.A.; Hamedifar, H.; Mohammadi-Khanaposhtani, M.; Mahdavi, M. A review on synthesis, mechanism of action, and structure-activity relationships of 1,2,3-triazole-based α-glucosidase inhibitors as promising anti-diabetic agents. J. Mol. Struct. 2022, 1255, 132469. [Google Scholar] [CrossRef]
- Rahim, F.; Ullah, H.; Hussain, R.; Taha, M.; Khan, S.; Nawaz, M.; Nawaz, F.; Gilani, S.J.; Bin Jumah, M.N. Thiadiazole based triazole/hydrazone derivatives: Synthesis, in vitro α-glucosidase inhibitory activity and in silico molecular docking study. J. Mol. Struct. 2023, 1287, 135619. [Google Scholar] [CrossRef]
- Kumar, S.; Khokra, S.L.; Yadav, A. Triazole analogues as potential pharmacological agents: a brief review. Future J. Pharm. Sci. 2021, 7, 106. [Google Scholar] [CrossRef]
- Kaproń, B.; Łuszczki, J.J.; Siwek, A.; Karcz, T.; Nowak, G.; Zagaja, M.; Andres-Mach, M.; Stasiłowicz, A.; Cielecka-Piontek, J.; Kocki, J.; Plech, T. Preclinical evaluation of 1,2,4-triazole-based compounds targeting voltage-gated sodium channels (VGSCs) as promising anticonvulsant drug candidates. Bioorg. Chem. 2020, 94, 103355. [Google Scholar] [CrossRef]
- Chu, X.M.; Wang, C.; Wang, W.-L.; Liang, L.L.; Liu, W.; Gong, K.K.; Sun, K.L. Triazole derivatives and their antiplasmodial and antimalarial activities. Eur. J. Med. Chem. 2019, 166, 206–223. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Peng, Y.; Zhu, L.; Wang, S.; Ji, J.; Rakesh, K.P. Triazole derivatives as inhibitors of Alzheimer's disease: Current developments and structure-activity relationships. Eur. J. Med. Chem. 2019, 180, 656–672. [Google Scholar] [CrossRef]
- Khan, S.A.; Akhtar, M.J.; Gogoi, U.; Meenakshi, D.U.; Das, A. An Overview of 1,2,3-triazole-Containing Hybrids and Their Potential Anticholinesterase Activities. Pharmaceuticals 2023, 16, 179. [Google Scholar] [CrossRef] [PubMed]
- Sooknual, P.; Pingaew, R.; Phopin, K.; Ruankham, W.; Prachayasittikul, S.; Ruchirawat, S.; Prachayasittikul, V. Synthesis and neuroprotective effects of novel chalcone-triazole hybrids. Bioorg. Chem. 2020, 105, 104384. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, S.; Almarghalani, D.A.; James, A.W.; Raza, Md K.; Kausar, T.; Nayeem, S.M.; Hoda, N.; Shah, Z.A. Synthesis and Pharmacological Evaluation of Novel Triazole-Pyrimidine Hybrids as Potential Neuroprotective and Anti-neuroinflammatory Agents. Pharm. Res. 2023, 40,167–185. [CrossRef]
- Li, G.; Hilgenfeld, R.; Whitley, R; De Clercq, E. Therapeutic strategies for COVID-19: progress and lessons learned. Nat. Rev. Drug. Discov. 2023, 1-27. [CrossRef]
- Panahi, Y.; Gorabi, A.M.; Talaei, S.; Beiraghdar, F.; Akbarzadeh, A.; Tarhriz, V.; Mellatyar, H. An overview on the treatments and prevention against COVID-19. Virol. J. 2023, 20, 23. [Google Scholar] [CrossRef] [PubMed]
- Uematsu, T.; Takano, T.; Matsui, H.; Kobayashi, N.; Ōmura, S.; Hanaki, H. Prophylactic administration of ivermectin attenuates SARS-CoV-2 induced disease in a Syrian Hamster Model. The Journal of Antibiotics 2023, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.K.; Misra, S. Antimicrobials in COVID-19: strategies for treating a COVID-19 pandemic. J. Basic Clin. Physiol. Pharmacol. 2022, 1–16. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.W. Current and emerging immunomodulators for treatment of SARS-CoV2 infection (COVID-19). Expert Opin. Pharmacother. 2022, 1–6. [Google Scholar] [CrossRef]
- Fazio, S.; Bellavite, P. Early Multi-Target Treatment of Mild-to-Moderate COVID-19, Particularly in Terms of Non-Steroidal Anti-Inflammatory Drugs and Indomethacin. BioMed 2023, 3, 177–194. [Google Scholar] [CrossRef]
- Mallikanti, V.; Thumma, V.; Matta, R.; Valluru, K.R.; Sharma Konidena, L.N.; Boddu, L.S.; Pochampally, J. Chem. Data Collections . 2023, 45, 101034. [Google Scholar] [CrossRef]
- Güzel, E.; Çevik, U.A.; Evren, A.E.; Bostancı, H.E.; Gül, U.D.; Kayış, U.; Özkay, Y.; Kaplancıklı, Z.A. Synthesis of Benzimidazole-1,2,4-triazole Derivatives as Potential Antifungal Agents Targeting 14α-Demethylase. ACS Omega 2023, 8, 4369–4384. [Google Scholar] [CrossRef] [PubMed]
- Ghobadi, E.; Hashemi, S.M.; Fakhim, H.; Hosseini-khah, Z.; Badali, H.; Emami, S. Design, synthesis and biological activity of hybrid antifungals derived from fluconazole and mebendazole. Eur. J. Med. Chem. 2023, 249, 115146. [Google Scholar] [CrossRef] [PubMed]
- Youssif, B.G.M.; Mohamed, Y.A.M.; Salim, M.T.A.; Inagaki, F.; Mukai, C.; Abdu-Allah, H.H.M. Synthesis of some benzimidazole derivatives endowed with 1,2,3-triazole as potential inhibitors of hepatitis C virus. Acta Pharm. 2016, 66, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Al-Humaidi, J.Y.; Shaaban, M.M.; Rezki, N.; Aouad, M.R.; Zakaria, M.; Jaremko, M.; Hagar, M.; Elwakil, B.H. 1,2,3-Triazole-Benzofused Molecular Conjugates as Potential Antiviral Agents against SARS-CoV-2 Virus Variants. Life 2022, 12, 1341. [Google Scholar] [CrossRef] [PubMed]
- Kondengadan, S.M.; Bansal, S.; Yang, C.; Liu, D.; Fultz, Z.; Wang, B. Click chemistry and drug delivery: A bird's-eye view. Acta Pharm. Sin. B. 2023, 13, 1990–2016. [Google Scholar] [CrossRef] [PubMed]
- de Souza, R.O.M.A.; de Mariz Miranda, L.S. Strategies Towards the Synthesis of N2-Substituted 1,2,3-Triazoles. An. Acad. Bras. Ciênc. 2019, 91(Suppl. 1), e20180751. [CrossRef]
- Dai, J. : Tian, S; Yang, X.; Liu, Z. Synthesis methods of 1,2,3-/1,2,4-triazoles: A review. Front. Chem. 2022, 10, 891484. [Google Scholar] [CrossRef]
- De Nino, A.; Maiuolo, L.; Costanzo, P.; Algieri, V.; Jiritano, A.; Olivito, F.; Tallarida, M.A. Recent Progress in Catalytic Synthesis of 1,2,3-Triazoles. Catalysts 2021, 11, 1120. [Google Scholar] [CrossRef]
- Koranne, A; Kurrey, K. ; Kumar, P.; Gupta, S.; Jha, K.V.; Ravi, R.; Sahu, K.P.; Anamika; Jha, A.K. Metal catalyzed C–H functionalization on triazole rings. RSC Adv. 2022, 12, 27534. [CrossRef]
- Marinescu, M. Chemistry and Applications of Benzimidazole and its Derivatives; Publisher: IntechOpen London, GB, 2019; pp. 1–213. [Google Scholar]
- Marinescu, M. Chiral benzimidazoles in medicinal chemistry: syntheses and applications. In Benzimidazole: Preparation and Applications; Vestergaard, A.A., Ed.; Publisher: New York, NY, USA, 2020; pp. 87–112. [Google Scholar]
- Zalaru, C.-M.; Marinescu, M. Zalaru, C.-M.; Marinescu, M. Benzimidazole compounds with anti-tumor and antibacterial activities. In Benzimidazole: Preparation and Applications. Vestergaard, A.A. Ed. Publisher: New York, USA, 2020; pp. 221-250.
- Marinescu, M.; Tudorache, D.G.; Marton, G.I.; Zalaru, C.M.; Popa, M.; Chifiriuc, M.C.; Stavarache, C.E.; Constantinescu, C. Density functional theory molecular modeling, chemical synthesis, and antimicrobial behaviour of selected benzimidazole derivatives. J. Mol. Struct. 2017, 463–471. [Google Scholar] [CrossRef]
- Marinescu, M.; Cinteza, L.O.; Marton, G.I.; Chifiriuc, M.C.; Popa, M.; Stanculescu, I.; Zalaru, C.M.; Stavarache, C.E. Synthesis, density functional theory study and in vitro antimicrobial evaluation of new benzimidazole Mannich bases. BMC Chemistry 2020, 14(1), 45. [Google Scholar] [CrossRef]
- Marinescu, M. Nitrogen-containing heterocycles as corrosion inhibitors. In Corrosion Inhibitors: An Overview; Wilkerson, R., Ed.; Publisher: New York, NY, USA, 2020; pp. 161–201. [Google Scholar]
- Qiu, J.; Zou, Y.; Li, S.; Yang, L.; Qiu, Z.; Kong, F.; Gu, X. Discovery of benzimidazole substituted 1, 2, 4-oxadiazole compounds as novel anti-HBV agents with TLR8-agonistic activities. Eur. J. Med. Chem. 2022, 244, 114833. [Google Scholar] [CrossRef]
- Youssif, B.G.M.; Abdel-Moty, S.G.; Sayed, Y.B. Synthesis and biologicalevaluation of some novel 1,2,3-triazol-N-arylidene acetohydrazide incorporating benzimidazole ring moiety as potential antimicrobial agents. J. Curr. Chem. Pharm. Sc. 2014, 4(2), 54–64. [Google Scholar]
- Al-blewi, F.F.; Almehmadi, M.A.; Aouad, M.R.; Bardaweel, S.K.; Sahu, P.K.; Messali, M.; Rezki, N.; El Ashry, L.S.H. Design, synthesis, ADME prediction and pharmacological evaluation of novel benzimidazole-1,2,3-triazole-sulfonamide hybrids as antimicrobial and antiproliferative agents. Chem. Cent. J. 2018, 12, 110. [Google Scholar] [CrossRef]
- Rashdan, H.R.M.; Abdelmonsef, A.H.; Abou-Krisha, M.M.; Yousef, T.A. Synthesis, Identification, Computer-Aided Docking Studies, and ADMET Prediction of Novel Benzimidazo-1,2,3-triazole Based Molecules as Potential Antimicrobial Agents. Molecules 2021, 26, 7119. [Google Scholar] [CrossRef]
- Ouahrouch, A.; Ighachane, H.; Taourirte, M.; Engels, J.W.; Sedra, M.H.; Lazrek, H.B. Benzimidazole-1,2,3-triazole Hybrid Molecules: Synthesis and Evaluation for Antibacterial/Antifungal Activity. Arch. Pharm. Chem. Life Sci. 2014, 347, 748–755. [Google Scholar] [CrossRef]
- Bistrović, A.; Krstulović, L.; Stolić, I.; Drenjancević, D.; Talapko, J.; Taylor, M.C.; Kelly, J.M.; Bajić, M.; Raic-Malić, S. Synthesis, anti-bacterial and anti-protozoal activities of amidinobenzimidazole derivatives and their interactions with DNA and RNA. J. Enzyme Inhib. Med. Chem. 2018, 33, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Rao, Y.J.; Sowjanya, T.; Thirupathi, G.; Murthy, N.Y.S.; Kotapalli, S.S. Synthesis and biological evaluation of novel flavone/triazole/ benzimidazole hybrids and flavone/isoxazole-annulated heterocycles as antiproliferative and antimycobacterial agents. Mol. Div. 2018, 22, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Ashok, D; Gundu, S. ; Aamate, V.K.; Devulapally, M.G. Conventional andmicrowave-assisted synthesis of new indole-tethered benzimidazole-based 1,2,3-triazoles and evaluation of their antimycobacterial, antioxidant and antimicrobial activities. Mol. Div. 2018, 22, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Chandrika, N.T.; Shrestha, S.K.; Ranjan, N.; Sharma, A.; Arya, D.P.; Garneau-Tsodikova, S. New Application of Neomycin B−Bisbenzimidazole Hybrids as Antifungal Agents. ACS Infect. Dis. 2018, 4, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Saber, A.; Anouar, E.H.; Sebbar, G.; El Ibrahimi, B.; Srhir, M.; Hökelek, T.; Mague, J.T.; El Ghayati, L.; Sebbar, N.K.; Essassi, E.M. New 1,2,3-triazole containing benzimidazolone derivatives: Syntheses, crystal structures, spectroscopic characterizations, Hirshfeld surface analyses, DFT calculations, anti-corrosion property anticipation, and antibacterial activities. J. Mol. Struct. 2021, 1242, 130719. [Google Scholar] [CrossRef]
- Mohsen, D.H.; Radhi, A.J.; Shaheed, D.Q.; Abbas, H.K. Synthesis New Benzimidazole Derivatives as Antibacterial. J. Pharm. Negative Results 2022, 13(3), 893–898. [Google Scholar] [CrossRef]
- Rezki, N. Green Microwave Synthesis and Antimicrobial Evaluation of Novel Triazoles. Org. Prep. Proc. Int. 2017, 49, 525–541. [Google Scholar] [CrossRef]
- Aparna, Y.; Nirmala, G.; Subhashini, N.J.P.; Sharada, L.N.; Sreekanth, S. Synthesis and Antimicrobial Activity of Novel Bis-1,2,3-triazol-1H-4-yl-substituted Aryl Benzimidazole-2-thiol Derivatives. Russ. J. Gen. Chem. 2020, 90, 1501–1506. [Google Scholar] [CrossRef]
- Gill, C.; Jadhav, G.; Shaikh, M.; Kale, R.; Ghawalkar, A.; Nagargoje, D.; Shiradkar, M. Clubbed [1,2,3] triazoles by fluorine benzimidazole: A novel approach to H37Rv inhibitors as a potential treatment for tuberculosis. Bioorg. Med. Chem. Lett. 2008, 18, 6244–6247. [Google Scholar] [CrossRef]
- Anand, A.; Kulkarni, M.V.; Joshi, S.D.; Dixit, S.R. One pot Click chemistry: A three component reaction for the synthesis of 2-mercaptobenzimidazole linked coumarinyl triazoles as anti-tubercular agents. Bioorg. Med. Chem. Lett. 2016, 4709–4713. [Google Scholar] [CrossRef]
- Khanapurmath, N. Kulkarni, M.V.; Joshi, S.D.; Kumar, G.N.A. A click chemistry approach for the synthesis of cyclic ureido tethered coumarinyl and 1-aza coumarinyl 1,2,3-triazoles as inhibitors of Mycobacterium tuberculosis H37Rv and their in silico studies. Biorg. Med. Chem. 2019, 115054. [CrossRef]
- Pandey, V.K.; Upadhyay, M.; Upadhyay, M.; Gupta, V.D.; Tandon, M. Benzimidazolyl quinolinyl mercaptotriazoles as potential antimicrobial and antiviral agents. Acta Pharm. 2005, 55, 47–56. [Google Scholar]
- Jadhav, G.R.; Shaikh, M.U.; Kale, R.P.; Shiradkar, M.R.; Gill, C.H. SAR study of clubbed [1,2,4]-triazolyl with fluorobenzimidazoles as antimicrobial and antituberculosis agents. Eur. J. Med. Chem. 2009, 44, 2930–2935. [Google Scholar] [CrossRef]
- Barot, K.P.; Manna, K.S.; Ghate, M.D. Design, synthesis and antimicrobial activities of some novel 1,3,4-thiadiazole, 1,2,4-triazole-5-thione and 1,3-thiazolan-4-one derivatives of benzimidazole. J. Saudi Chem. Soc. 2017, 21, S35–S43. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, Y.; Wang, W.; Wang, S.; Xu, B.; Fan, G.; Dong, G.; Liu, Y.; Yao, J.; Miao, Z.; Zhang, W. Discovery of highly potent triazole antifungal derivatives by heterocycle-benzene bioisosteric replacement. Eur. J. Med. Chem. 2013, 64, 16–22. [Google Scholar] [CrossRef]
- Luo, Y.-L; Baathulaa, K.; Kannekanti, V.K.; Zhou, C.-H.; Cai, G.-X. Novel benzimidazole derived naphthalimide triazoles: synthesis, antimicrobial activity and interactions with calf thymus DNA. Science China Chem. 2015, 58, 483–494. [Google Scholar] [CrossRef]
- Ahmadi, A. Synthesis an antibacterial evaluation of ome novel Mannich bases of benzimidazole derivatives. Bull. Chem. Soc. Ethiop. 2016, 30(3), 421–425. [Google Scholar] [CrossRef]
- Kankate, R.S.; Gide, P.S.; Belsare, D.P. Design, synthesis and antifungal evaluation of novel benzimidazole tertiary amine type of fluconazole analogues. Arab. J. Chem. 2019, 12, 2224–2235. [Google Scholar] [CrossRef]
- Ahuja, R.; Sidhu, A.; Bala, A.; Arora, D.; Sharma, P. Structure based approach for twin-enzyme targeted benzimidazolyl-1,2,4-triazole molecular hybrids as antifungal agents. Arab. J. Chem. 2020, 13, 5832–5848. [Google Scholar] [CrossRef]
- Evren, A.E.; Celik, I.; Akar Cevik, U. Synthesis, molecular docking, in silico ADME and antimicrobial activity studies of some new benzimidazole-triazole derivatives. Cumhuriyet Sci. J. 2021, 42(4), 795–805. [Google Scholar] [CrossRef]
- Ansari, K.F.; Lal, C.; Khitoliya, R.K. Synthesis and biological activity of some triazole-bearing benzimidazole derivatives. J. Serb. Chem. Soc. 2011, 76 (3) 341–352. [CrossRef]
- Tien, C.N.; Cam, D.T.T.; Manh, H.B.; Dang, D.N. Synthesis and Antibacterial Activity of Some Derivatives of 2-Methylbenzimidazole Containing 1,3,4-Oxadiazole or 1,2,4-Triazole Heterocycle. J. Chem. 2016, 2016, 1507049. [Google Scholar] [CrossRef]
- Kantar, G.K.; Mentese, E.; Beris, F.S.; Şasmaz, S.; Kahveci, B. Synthesis and antimicrobial activity of some new triazole bridged benzimidazole substituted phthalonitrile and phthalocyanines. Rev. Roum. Chim. 2018, 63, 59–65. [Google Scholar]
- Nandwana, N.K.; Singh, R.P.; Patel, O.P.S.; Dhiman, S.; Saini, H.K.; Jha, P.N.; Kumar, A. Design and Synthesis of Imidazo/Benzimidazo[1,2-c] quinazoline Derivatives and Evaluation of Their Antimicrobial Activity. ACS Omega 2018, 3, 16338–16346. [Google Scholar] [CrossRef] [PubMed]
- Al-Majidi, S.M.H.; Ibrahim, H.A.R.; AL-issa, A.H. Synthesis and Identification of Some New Derivatives of ([Benzyl Thio) Benzimidazole N-(Methylene-5-Yl)]-4,5-Di Substituted 1,2,4-Triazole and Evaluation of Their Activity as Antimicrobial and Anti-Inflammatory Agents. Iraqi J. Sci. 2021, 62(4), 1054–1065. [Google Scholar] [CrossRef]
- El-masry, A.H.; Fahmy, H.H.; Ali Abdelwahed, S.H. Synthesis and Antimicrobial Activity of Some New Benzimidazole Derivatives. Molecules 2000, 5, 1429–1438. [Google Scholar] [CrossRef]
- Menteşe, E.; Ülker, S.; Kahveci, B. Synthesis and study of α-glucosidase inhibitory, antimicrobial and antioxidant activities of some benzimidazole derivatives containing triazole, thiadiazole, oxaiazole and morpholine rings. Chem. Heterocycl. Comp. 2015, 50(12), 1671–1682. [Google Scholar] [CrossRef]
- Karale, B.K.; Nirmal, P.R.; Akolkar, H.N. Synthesis and in vitro biological screening of some benzimidazolyl anchored azoles. Ind. J. Chem. 2015, 54B, 399–405. [Google Scholar]
- Madawali, I.M.; Gaviraj, E.N; Kalyane, N.V.; Shivakumar, B. A Review On Substituted Benzimidazoles: Biologically Active Compounds. Am. J. Pharm. Tech. Res. 2019, 9, 256–274. [Google Scholar] [CrossRef]
- Eisa, H.M.; Barghash, A.-e.M.; Badr, S.M.; Farahat, A.A. Synthesis and antimicrobial activity of certain benzimidazole and fused benzimidazole derivatives. Ind. J. Chem. 2010, 49B, 1515–1525. [Google Scholar]
- Nevade, S.A.; Lokapure, S.G.; Kalyane, N.V. Synthesis and Pharmacological Evaluation of Some Novel 2-Mercapto Benzimidazole Derivatives. Kor. Che. Soc. 2013, 57, 755–760. [Google Scholar] [CrossRef]
- Can N.O.; Çevik, U.A.; Saglik, B.N.; Levent, S.; Korkut, B.; Özkay, Y.; Kaplancikli, Z.A.; Koparal, A.S. J. Chem. 2017, 9387102. [CrossRef]
- Karaca Gençer, H.; Acar Çevik, U.; Levent, S.; Sağlık, B.N.; Korkut, B.; Özkay, Y.; Ilgın, S.; Öztürk, Y. New Benzimidazole-1,2,4-Triazole Hybrid Compounds: Synthesis, Anticandidal Activity and Cytotoxicity Evaluation. Molecules 2017, 22, 507. [Google Scholar] [CrossRef]
- Aryal, P.; Shakya, B. J. Synthesis, Cytotoxicity, Antibacterial and Antioxidant Activity of New 2-Substituted Benzimidazole Containing 1,2,4-Triazoles. Nepal Chem. Soc. 2023, 43, 34–45. [Google Scholar] [CrossRef]
- Kazeminejad, Z.; Marzi, M.; Shiroudi, A.; Kouhpayeh, S.A.; Farjam, M.; Zarenezhad, E. Novel 1,2,4-Triazoles as Antifungal Agents. BioMed Res. Int. 2022, 4584846. [Google Scholar] [CrossRef]
- Nandha, B.; Nargund, L.V.G.; Nargund, S.L. Design and synthesis of some new imidazole and 1,2,4-triazole substituted fluorobenzimidazoles for antitubercular and antifungal activity. Der Pharma Chem. 2013, 5(6), 317–327. [Google Scholar]
- Tonelli, M.; Paglietti, G.; Boido. V.; Sparatore, F.; Marongiu, F.; Marongiu, E.; La Colla, P.; Loddo, R. Antiviral Activity of Benzimidazole Derivatives. I. Antiviral Activity of1-Substituted-2-[(Benzotriazol-1/2-yl)methyl]benzimidazoles). Chem. Biodivers. 2008, 5, 2386-2401. [CrossRef]
- Kanwal, A.; Ahmad, M.; Aslam, S.; Raza Naqvi, S.A.; Saif, M.J. Recent advances in antiviral benzimidazole derivatives: a mini review. Pharm. Chem. J. 2019, 15(3), 179–187. [Google Scholar] [CrossRef]
- Ion, V.; Matei, A.; Constantinescu, C.; Ionita, I.; Marinescu, M.; Dinescu, M.; Emandi, A. Octahydroacridine thin films grown by matrix-assisted pulsed laser evaporation for non linear optical applications. Mater. Science Semicond. Process. 2015, 36, 78–83. [Google Scholar] [CrossRef]
- Zalaru, C.; Dumitrascu,F.; Draghici, C.; Tarcomnicu, I.; Tatia, R.; Moldovan, L.; Chifiriuc, M.C.; Lazar, V.; Marinescu, M.; Nitulescu, M.G.; Ferbinteanu, M. Synthesis, spectroscopic characterization, DFT study and antimicrobial activity of novel alkylaminopyrazole derivatives, J. Mol. Struct. 2018, 1156,12-21. [CrossRef]
- Zalaru, C.; Dumitrascu, F.; Draghici, C.; Iovu, M.; Marinescu, M.; Tarcomnicu, I.; Nitulescu, G.M. Synthesis and biological screening of some novel 2-(1H-pyrazol-1-yl)-acetamides as lidocaine analogue, Ind. J. Chem. B 2014, 53 B (06), 733–739. [Google Scholar]
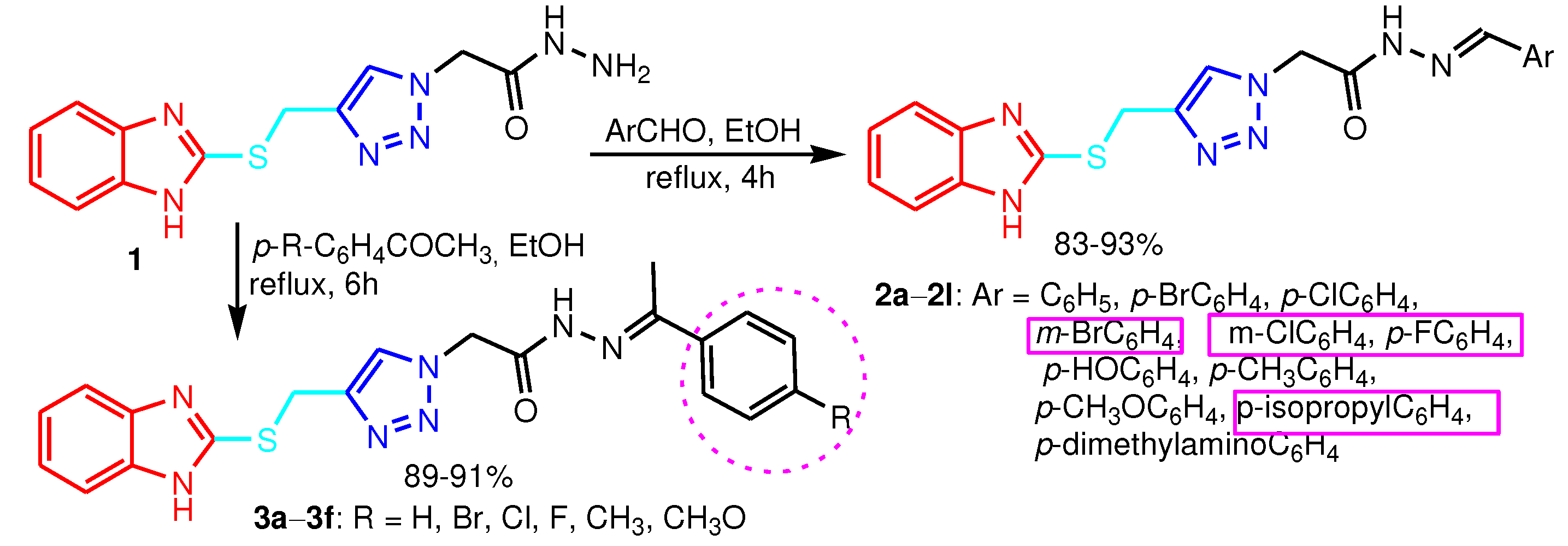
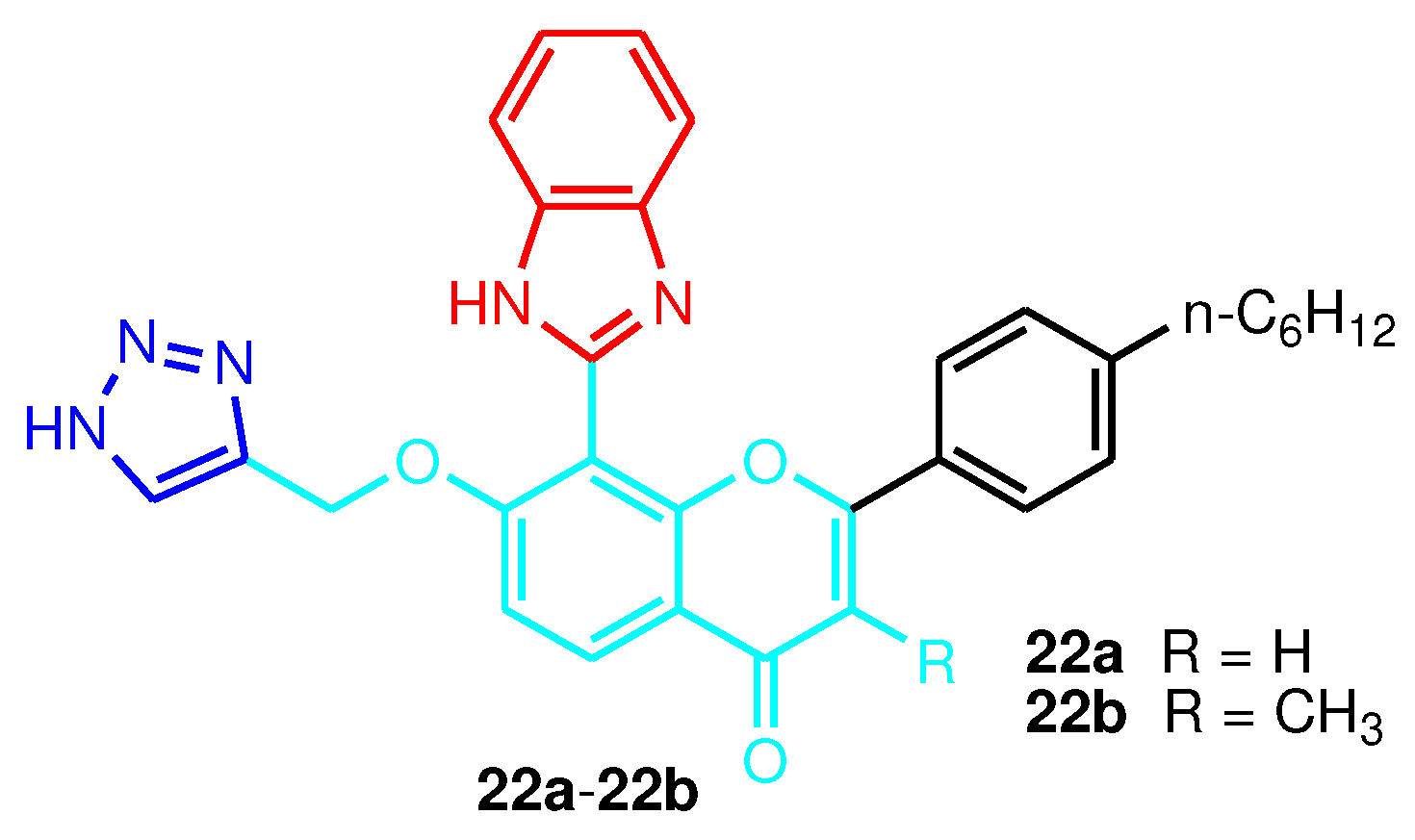
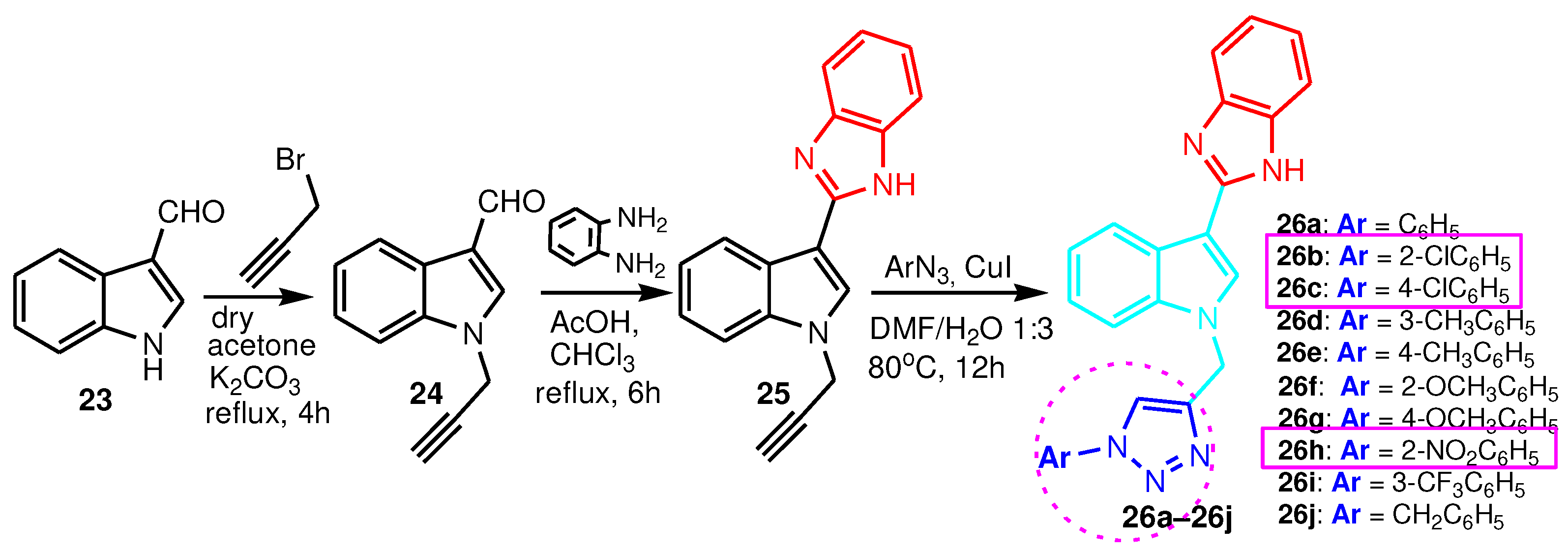
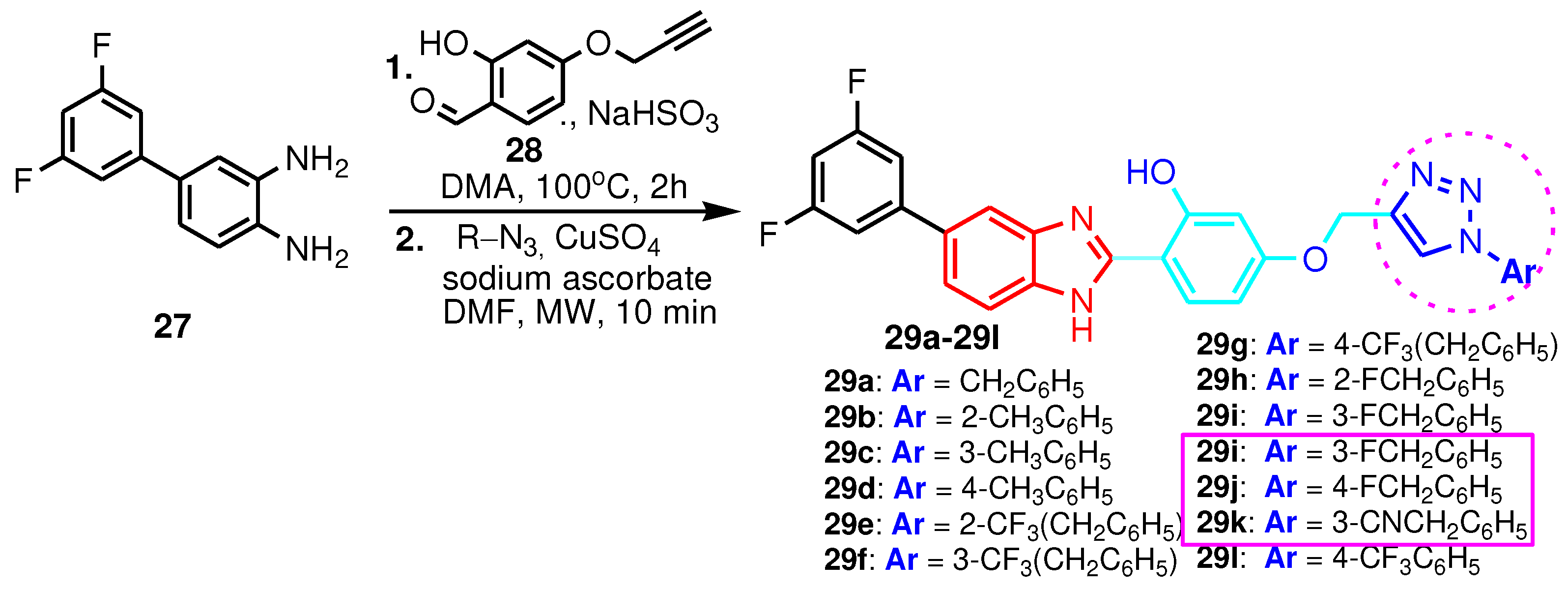
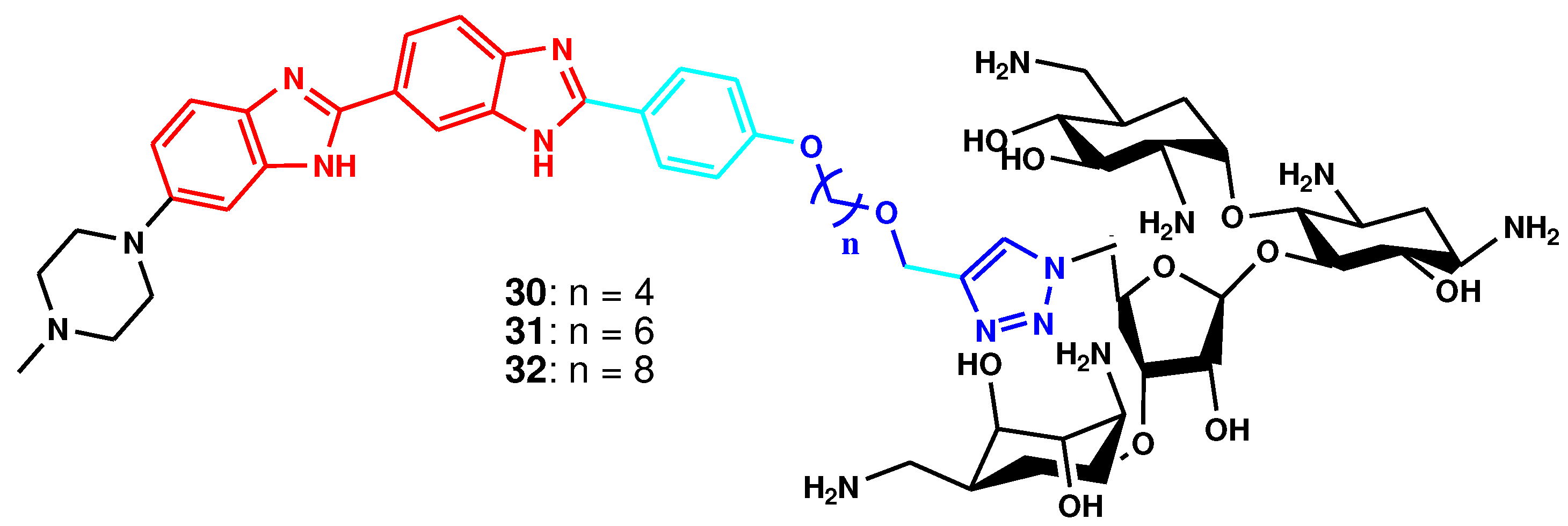
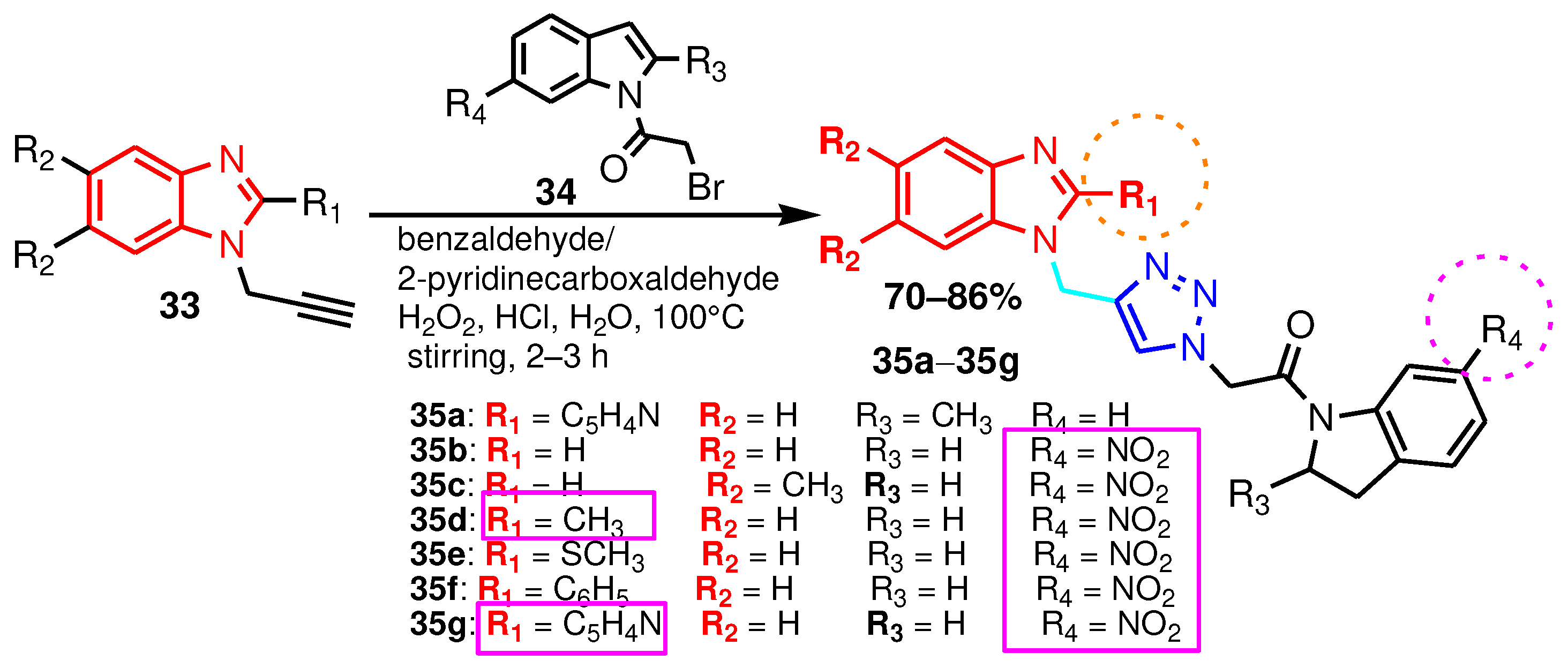
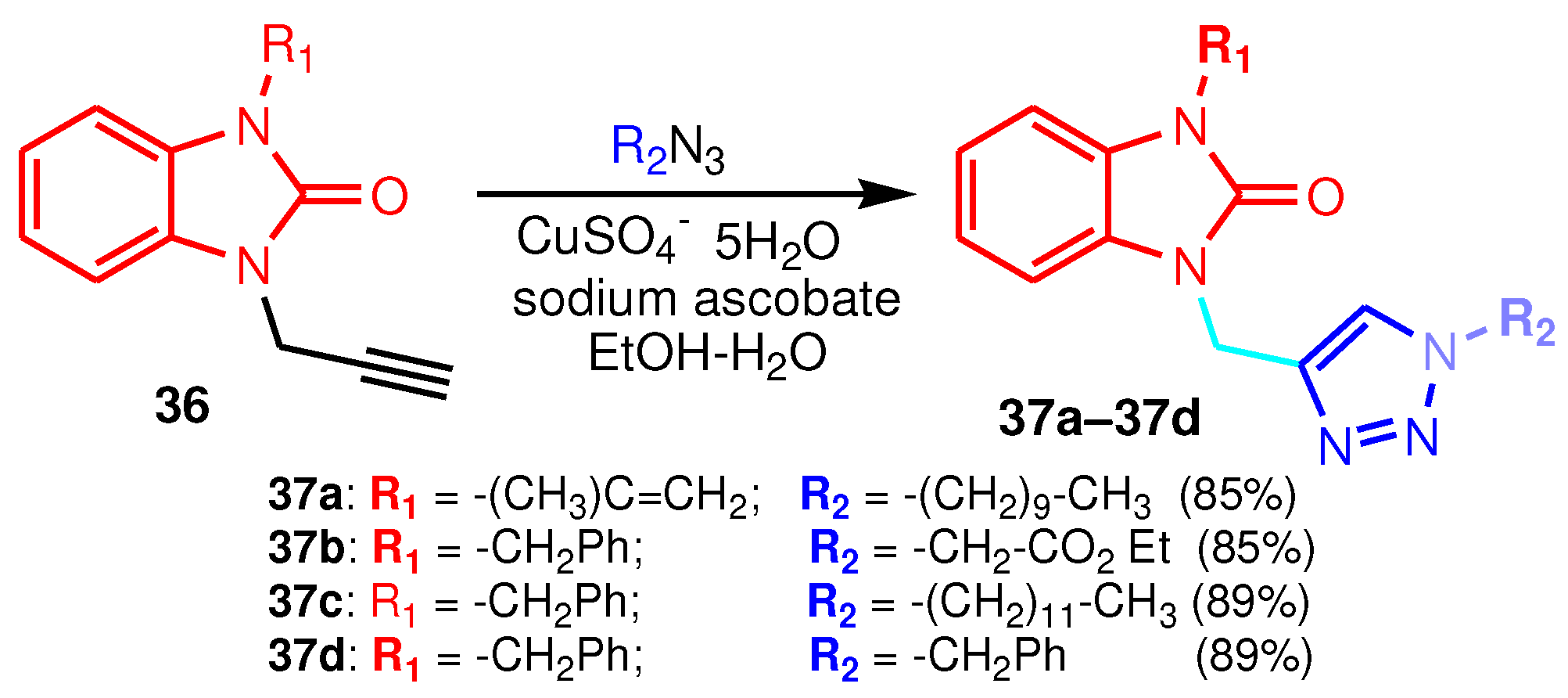
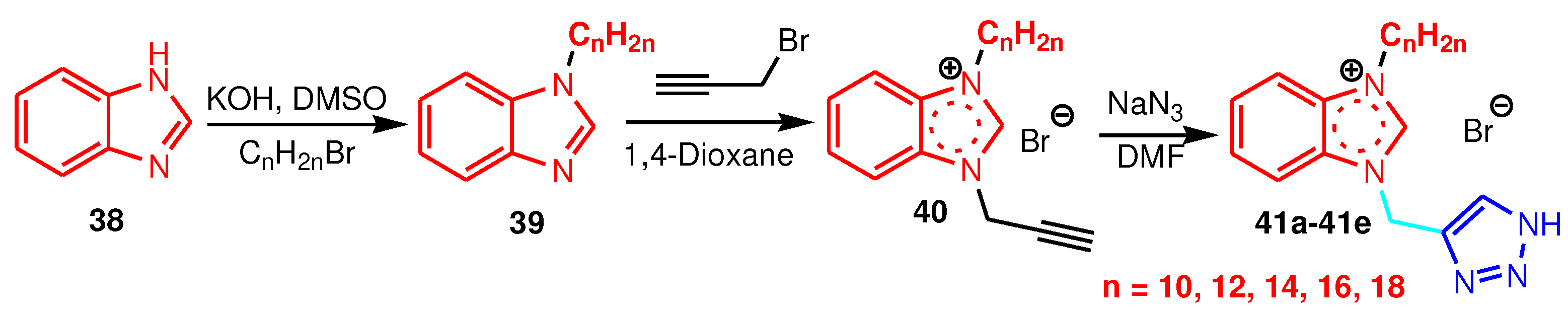
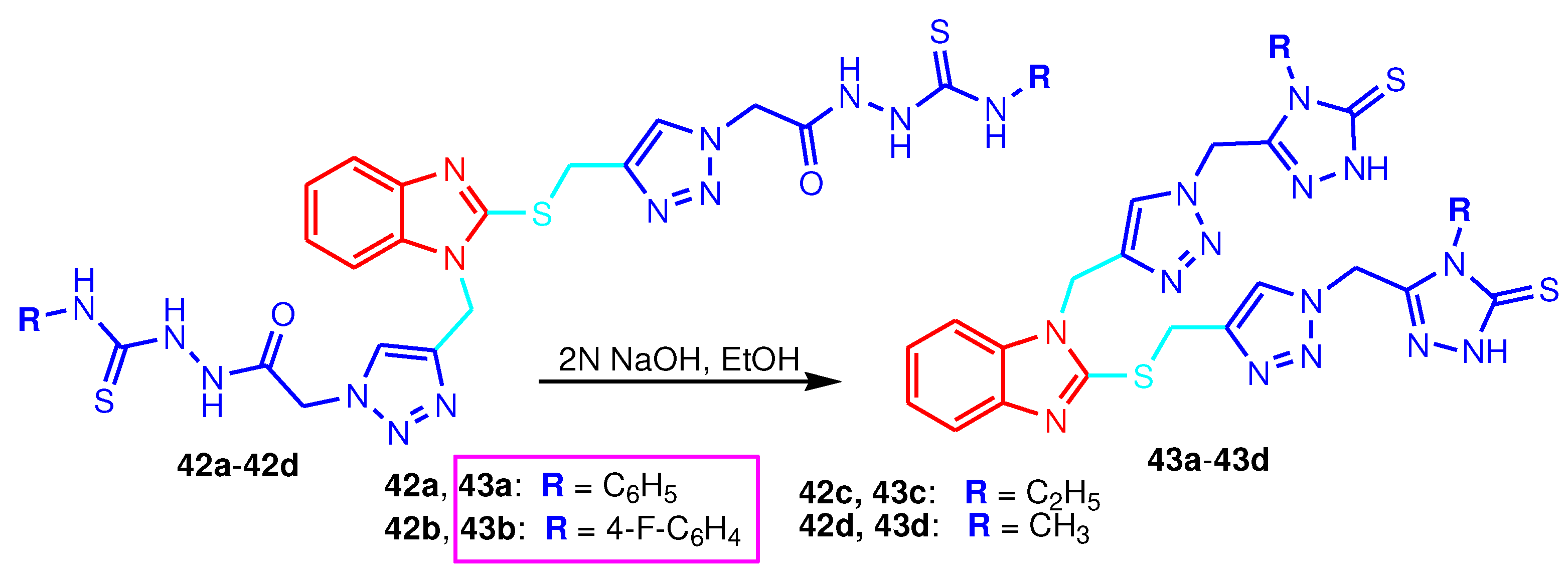
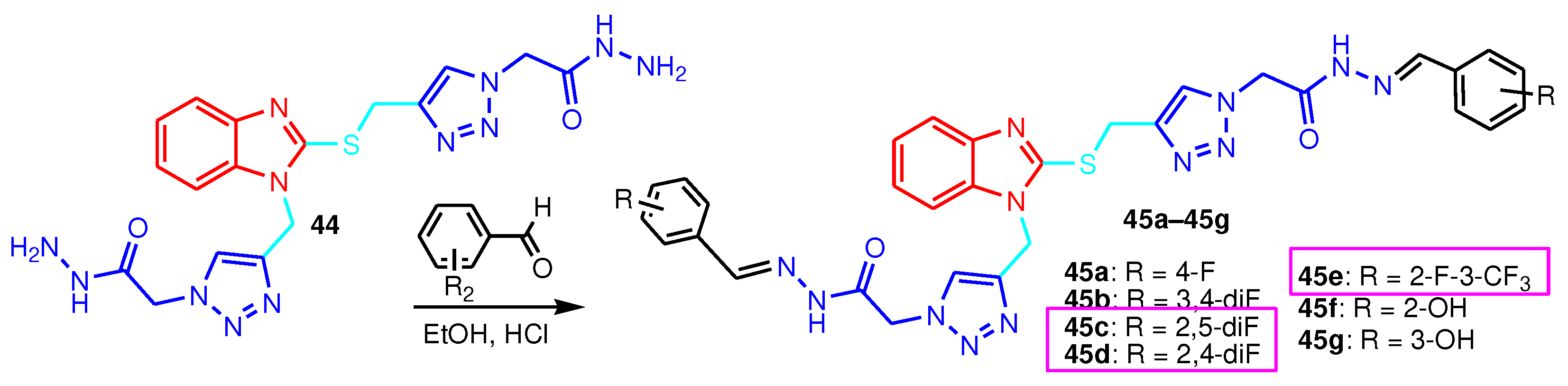
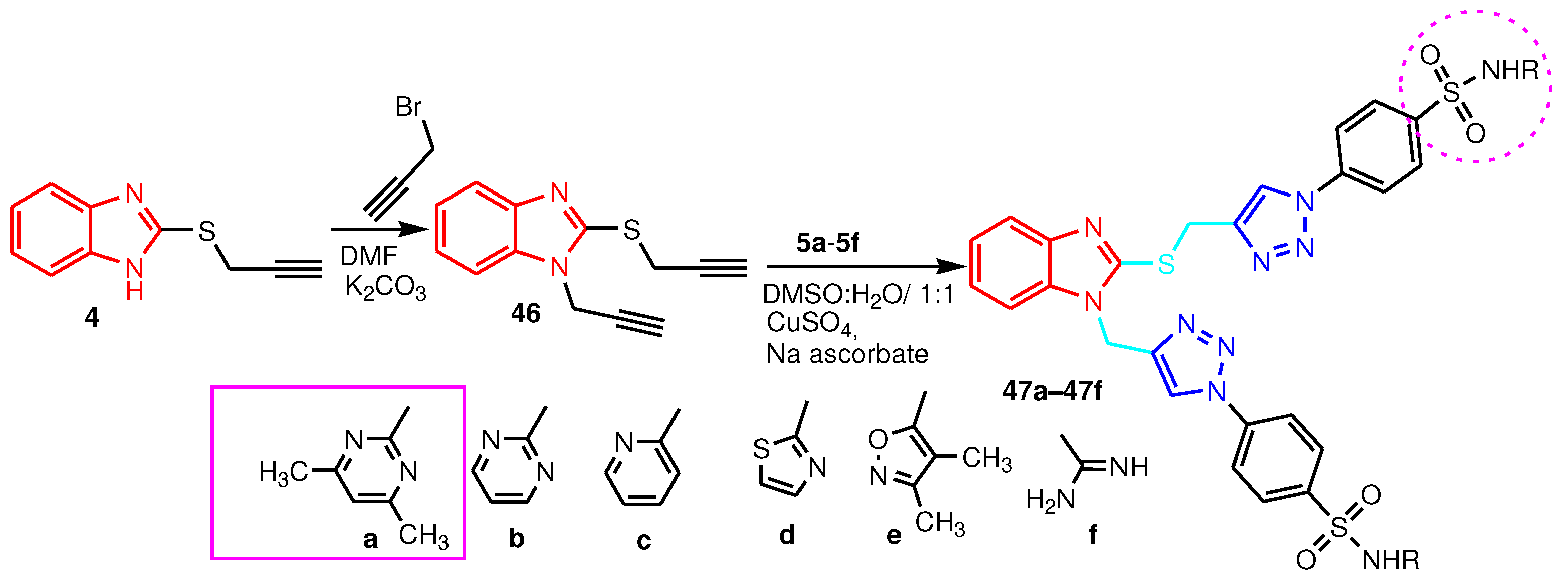
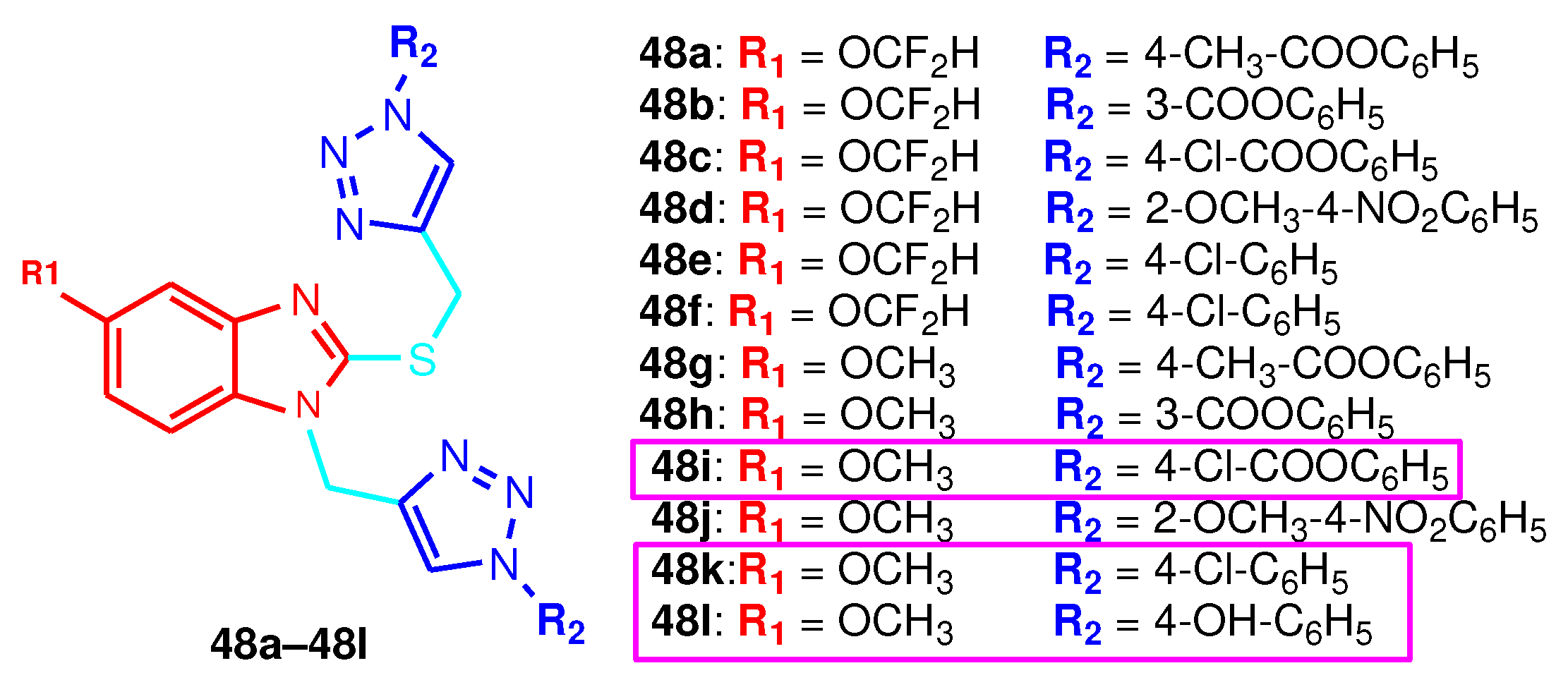
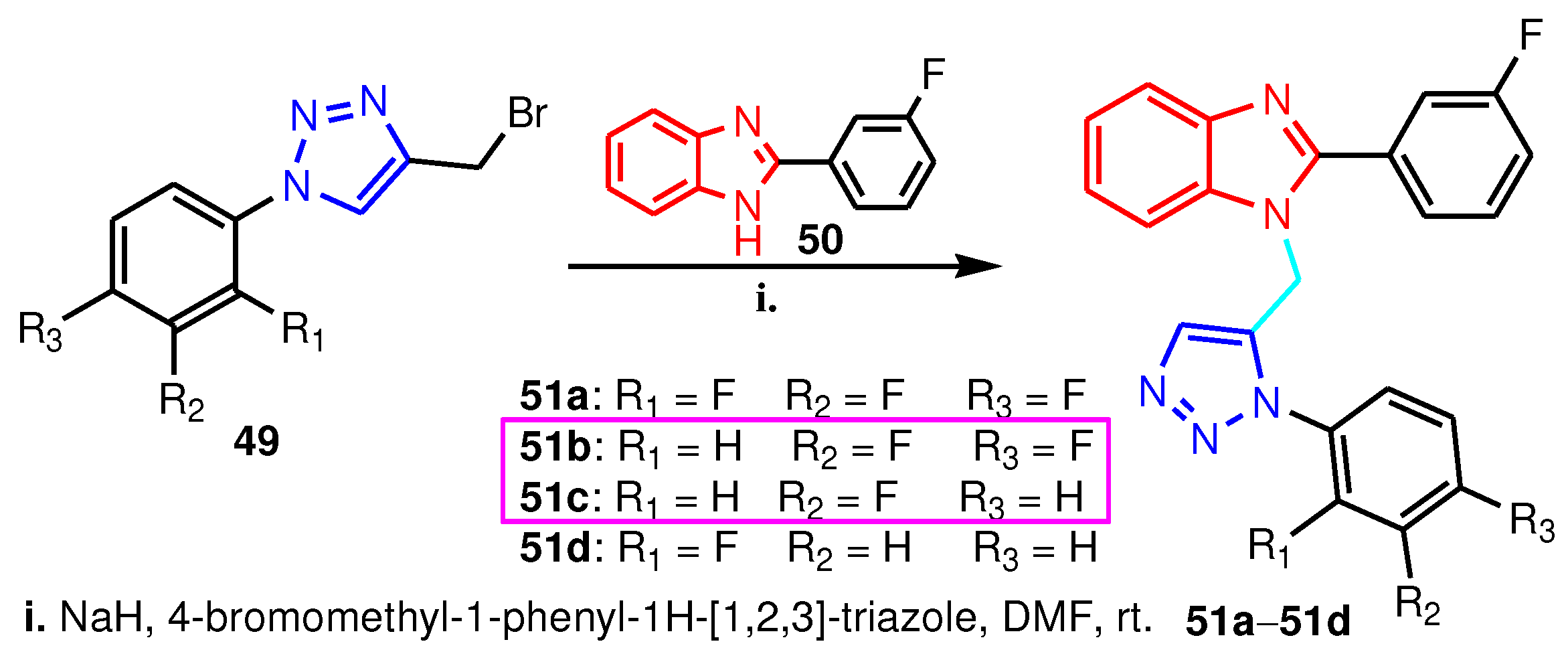
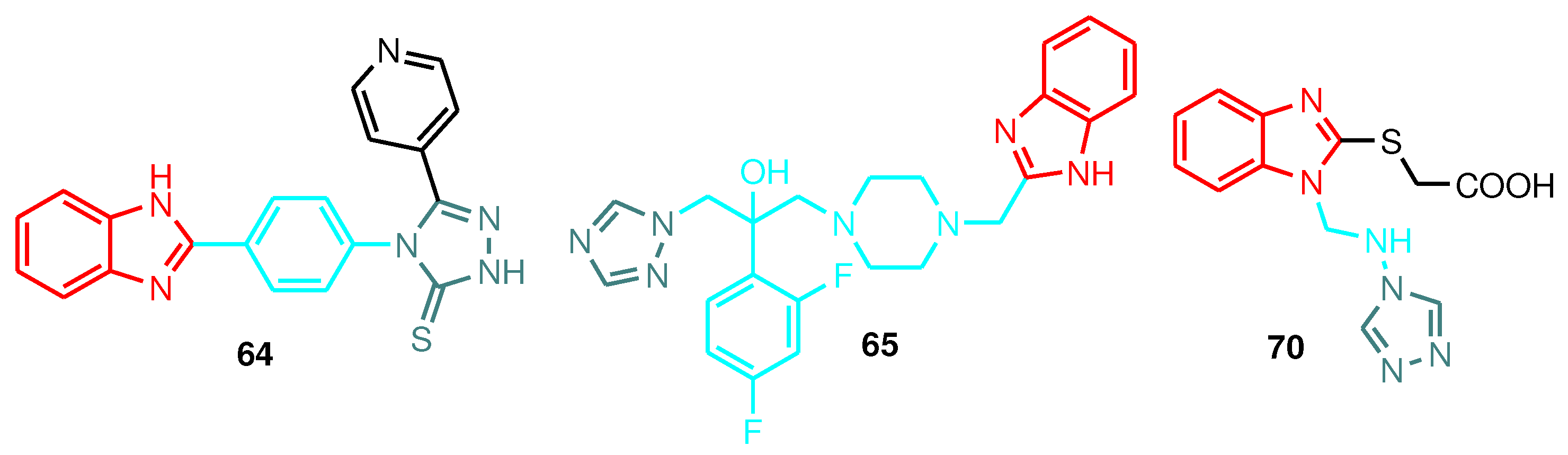
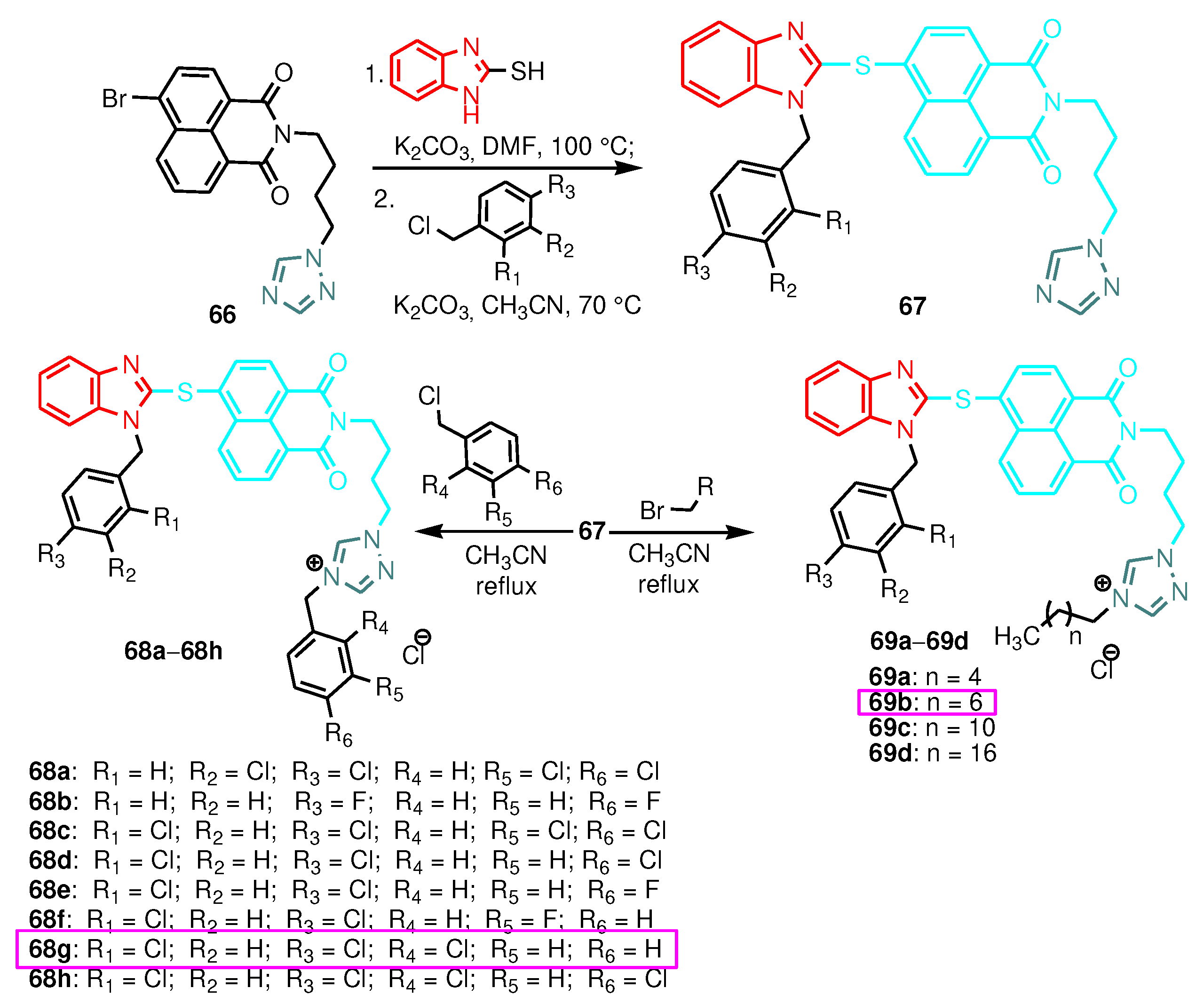
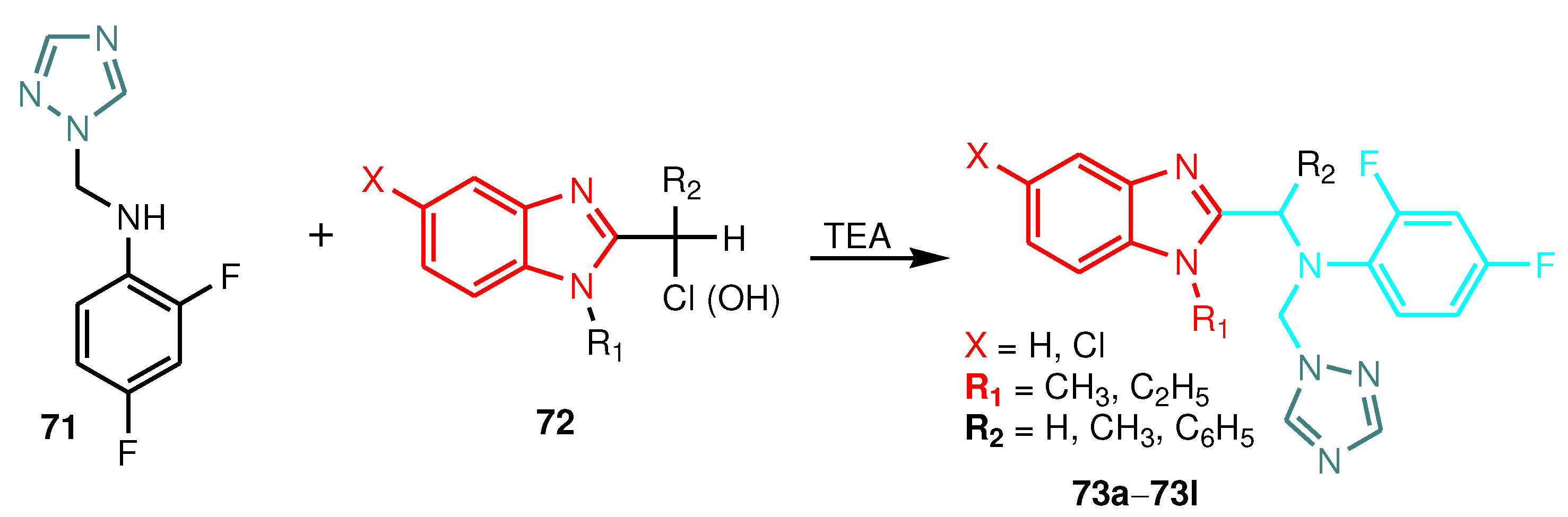
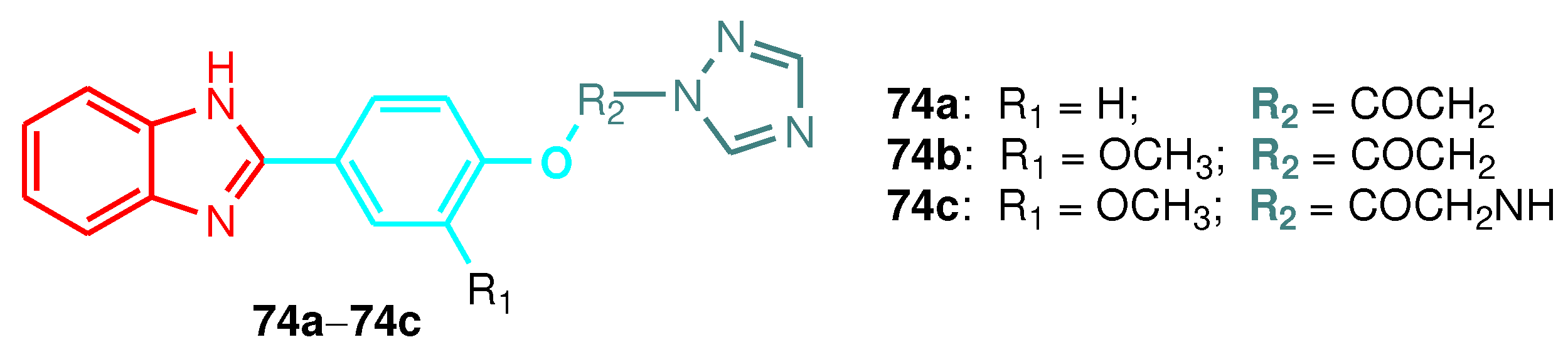
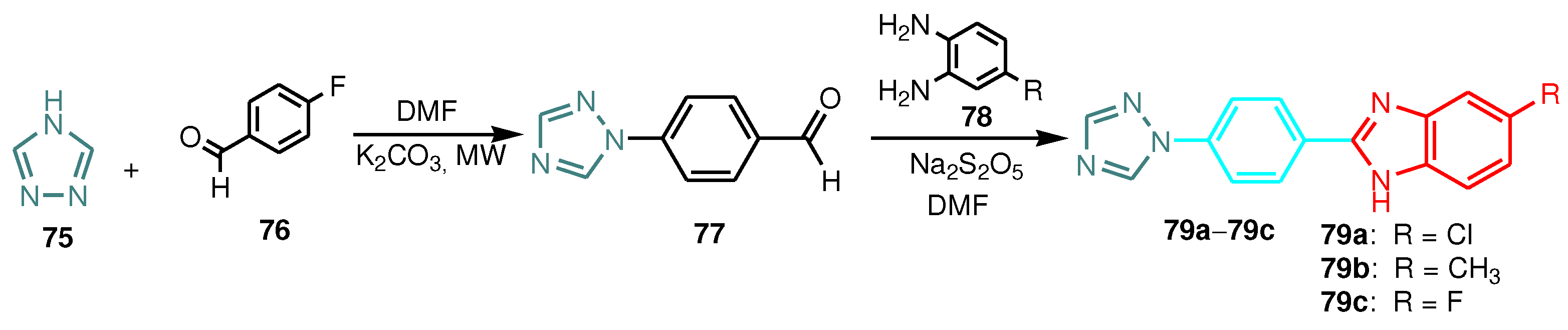
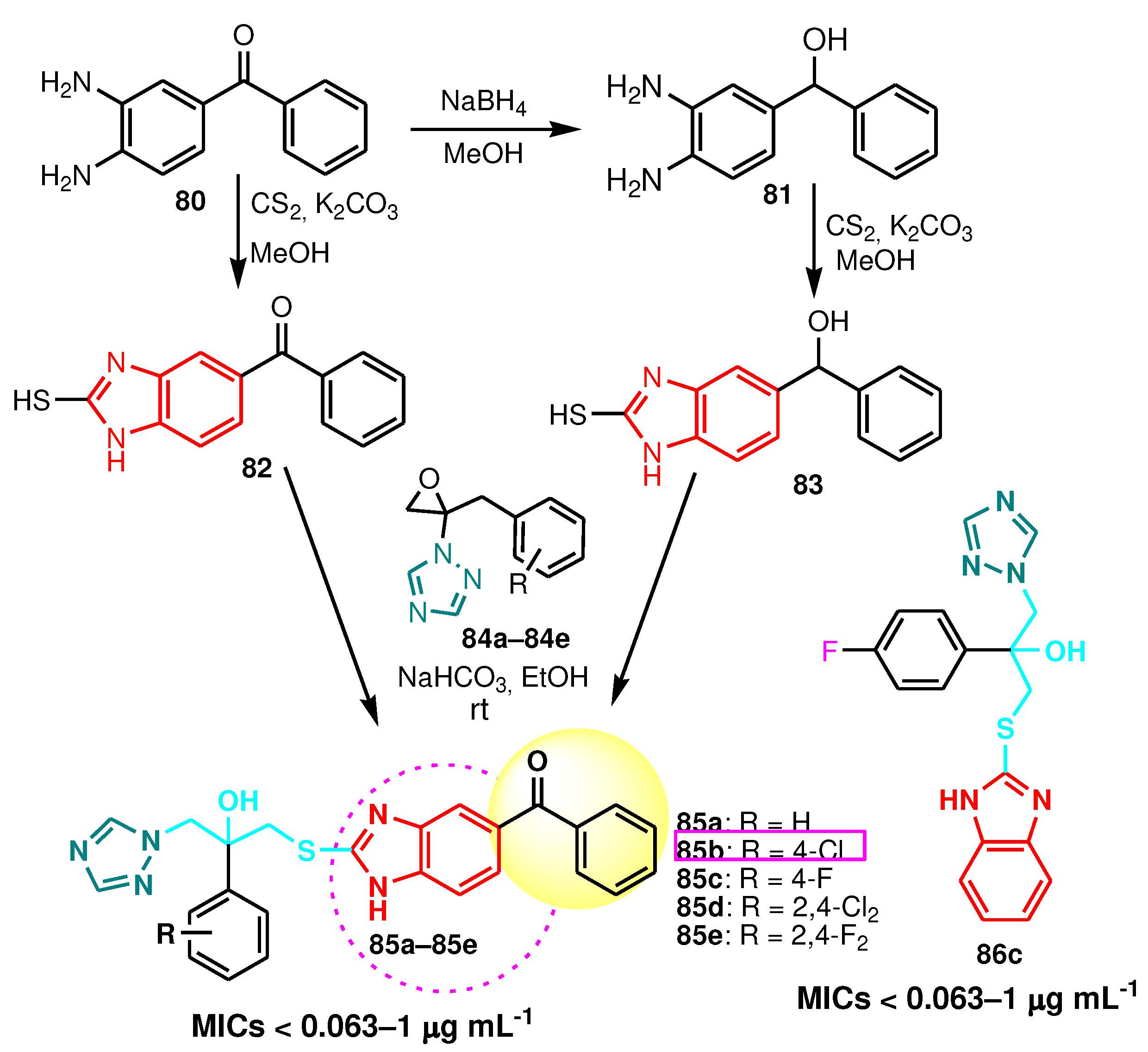
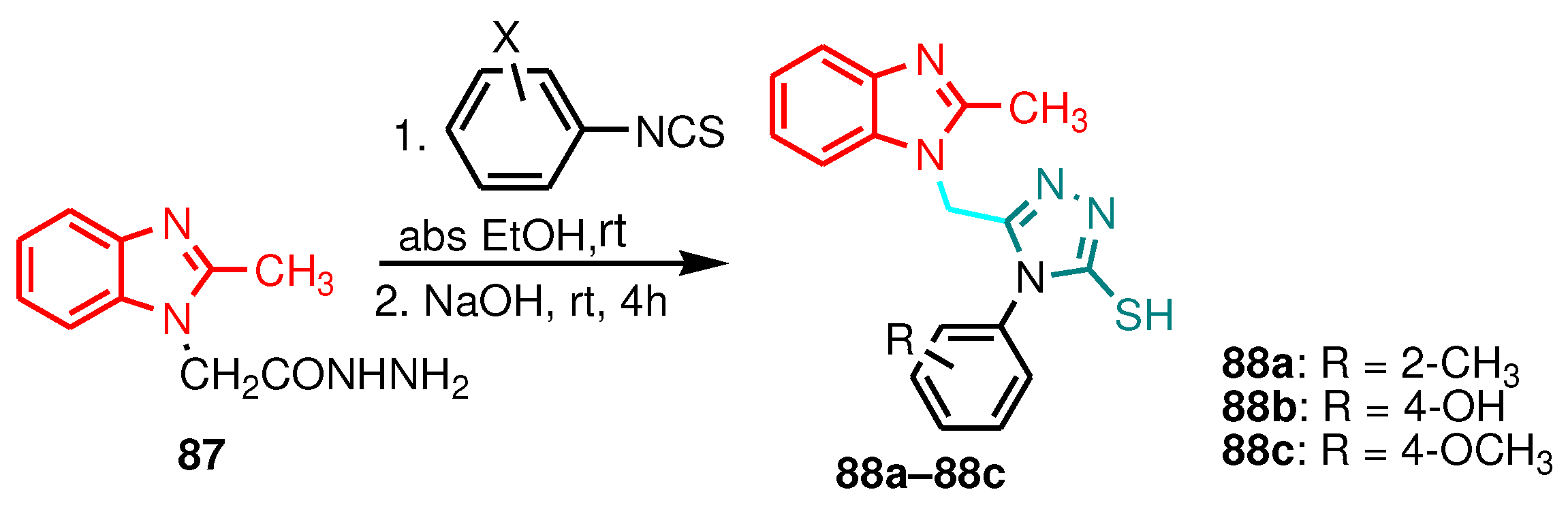
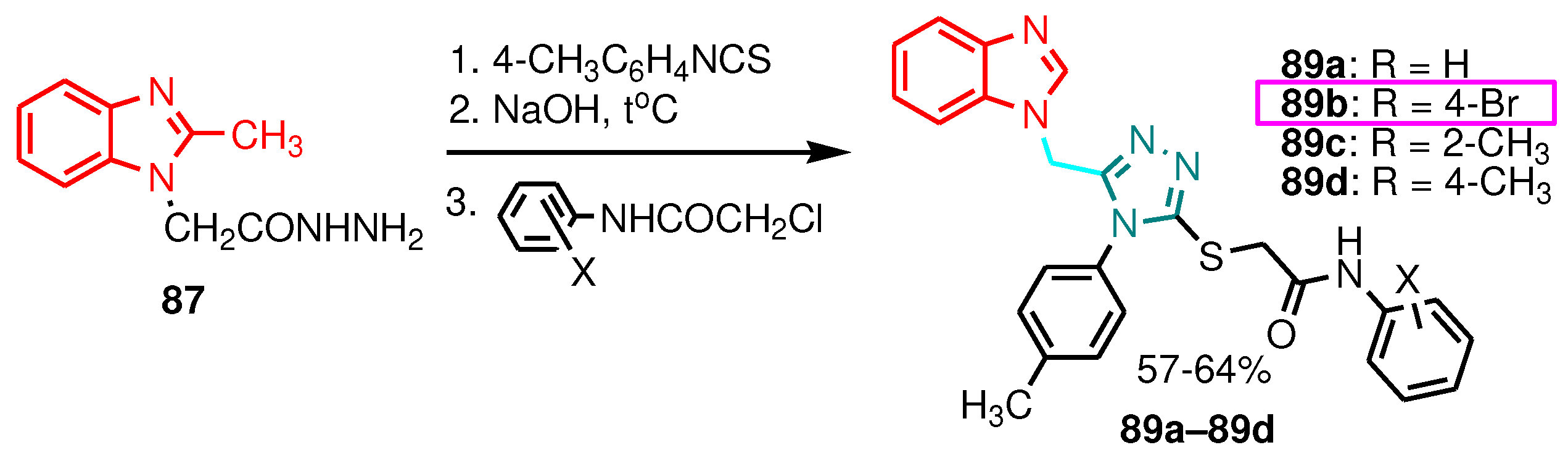
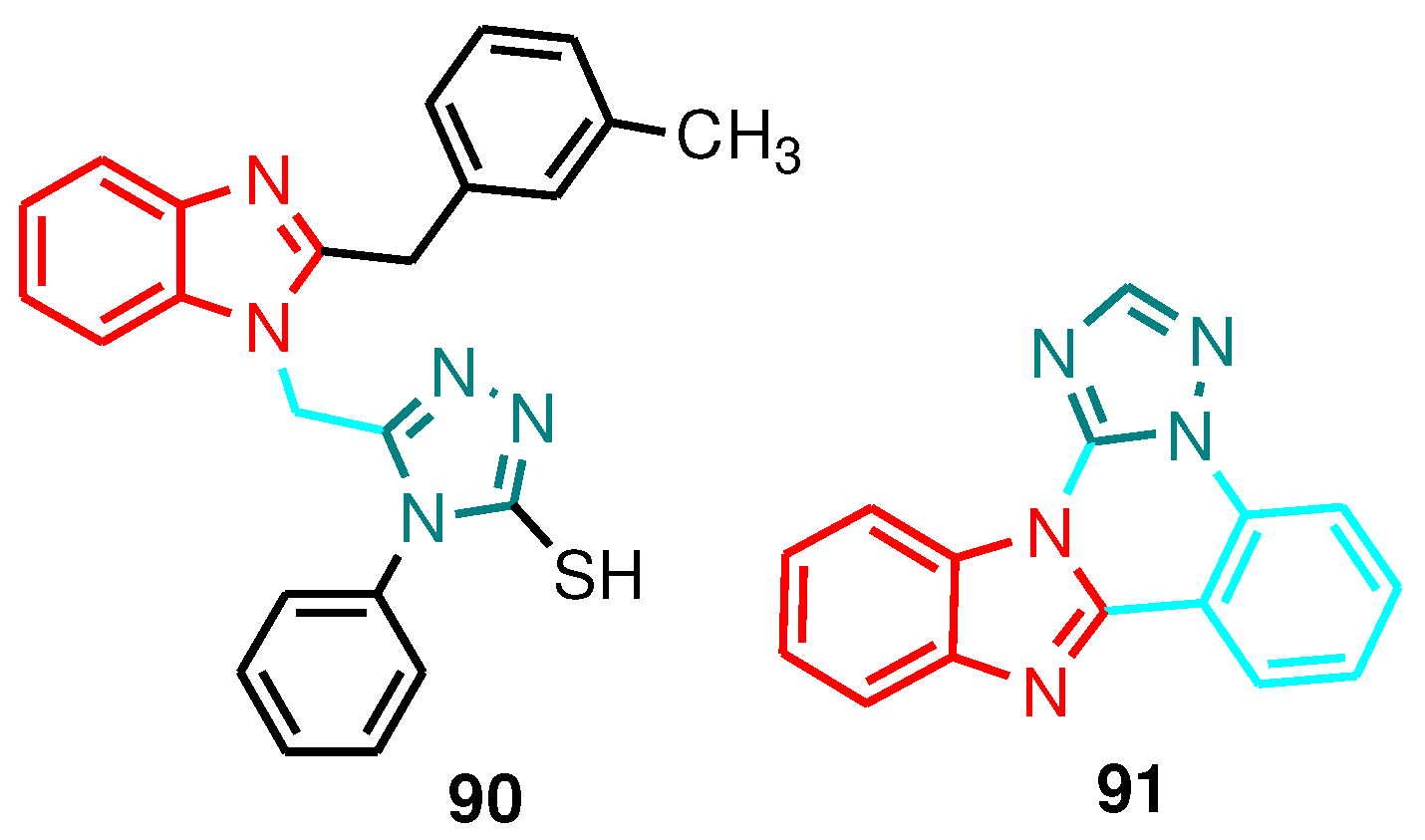
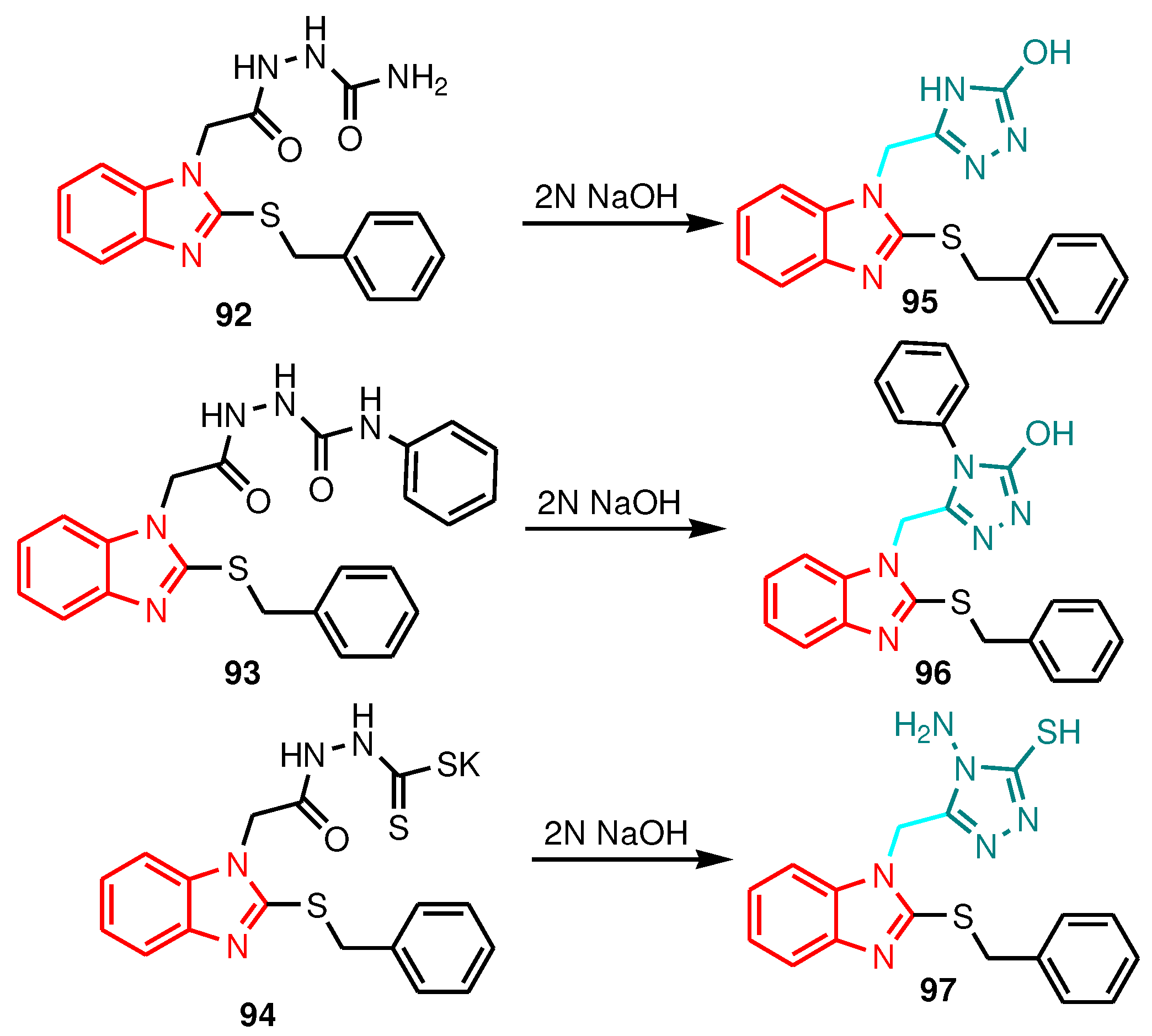
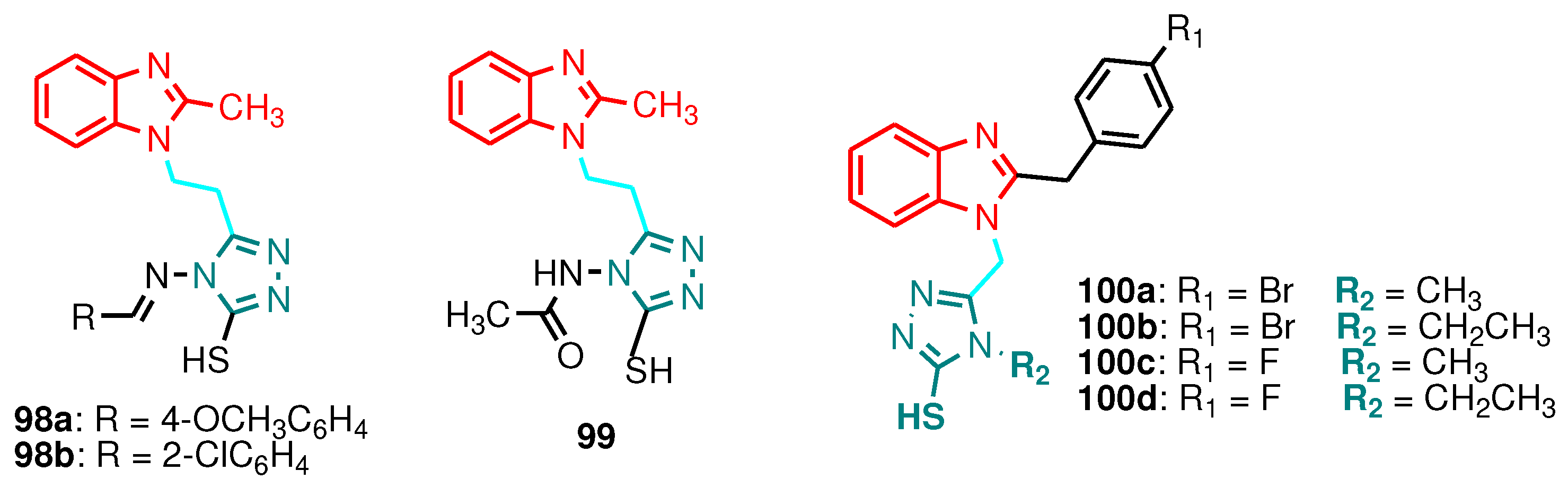
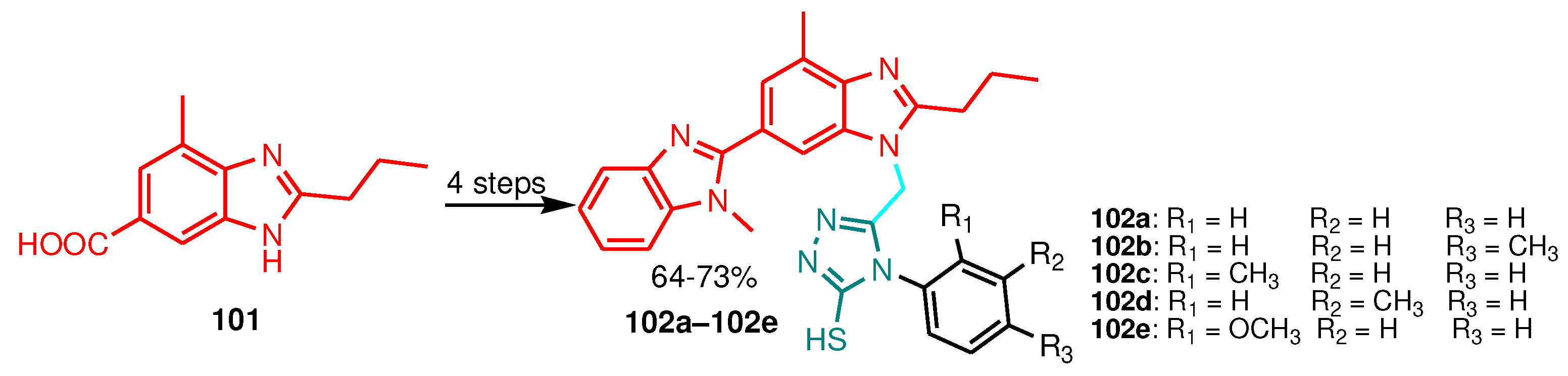
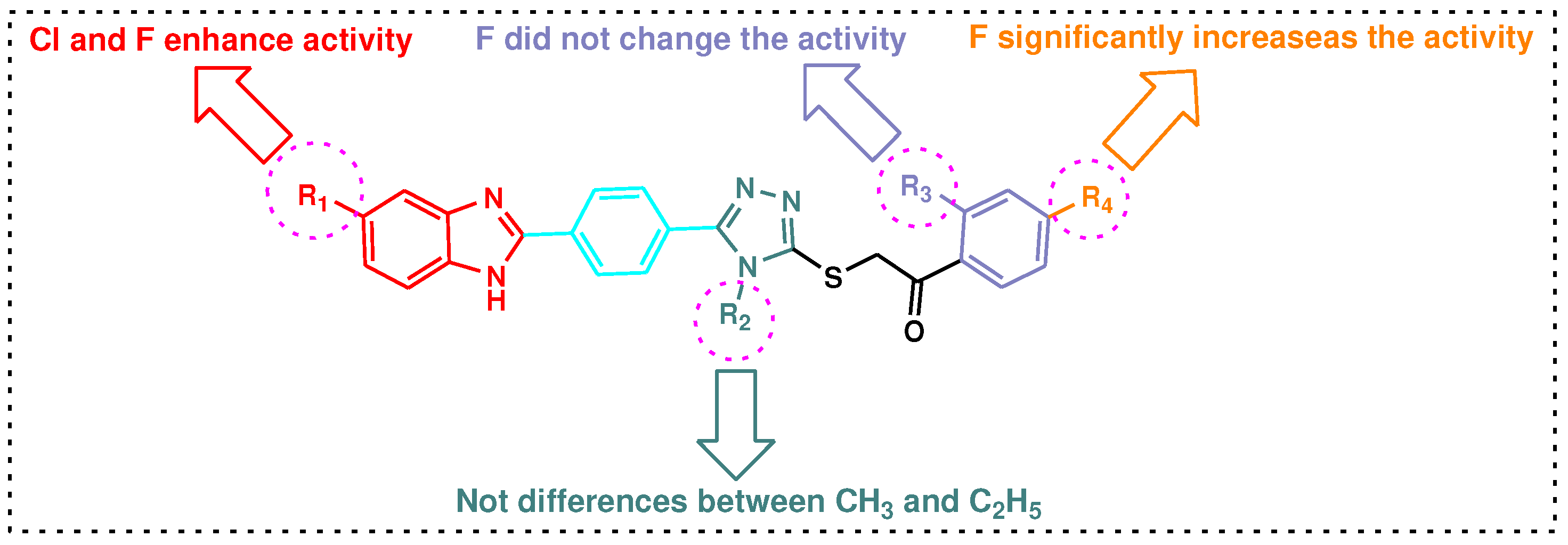
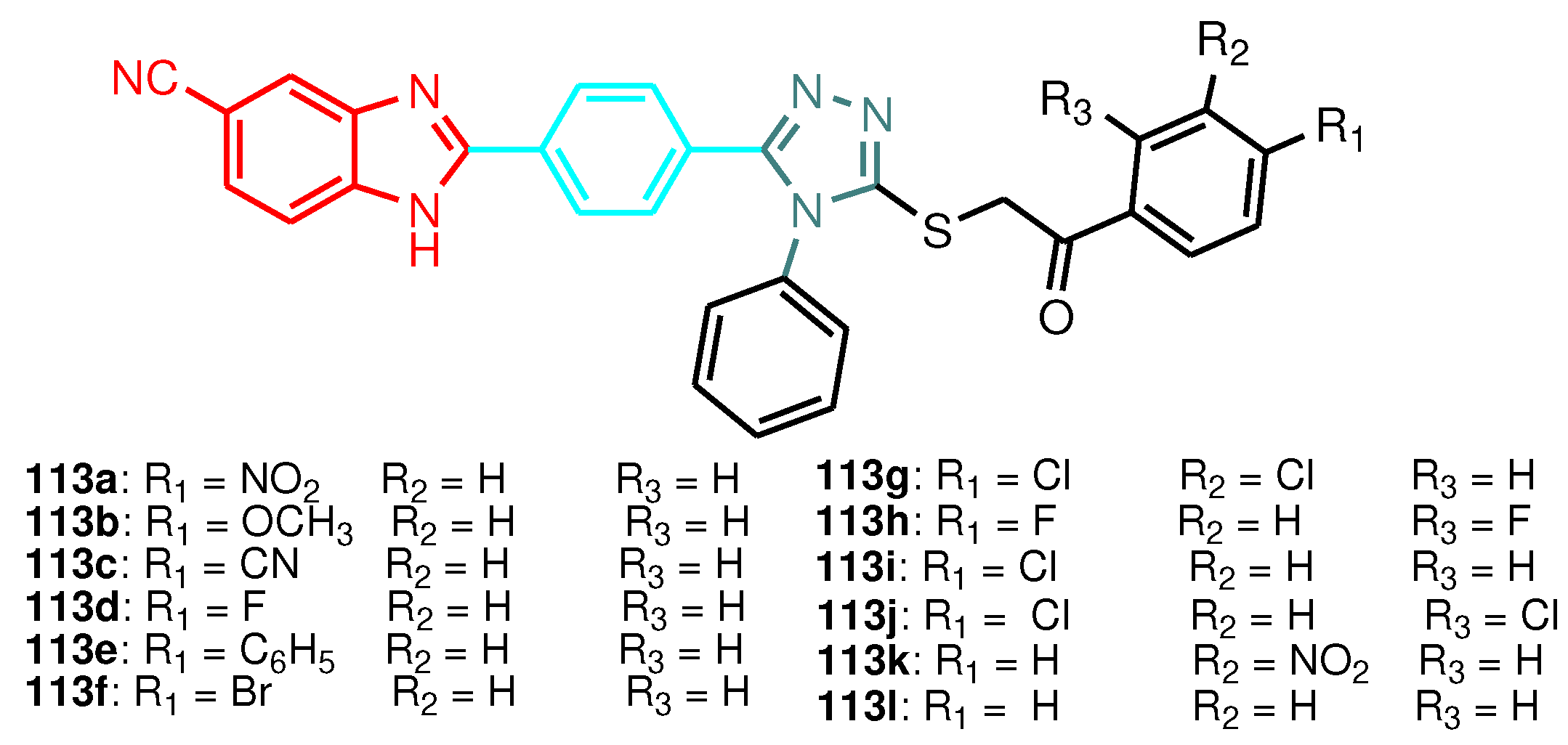
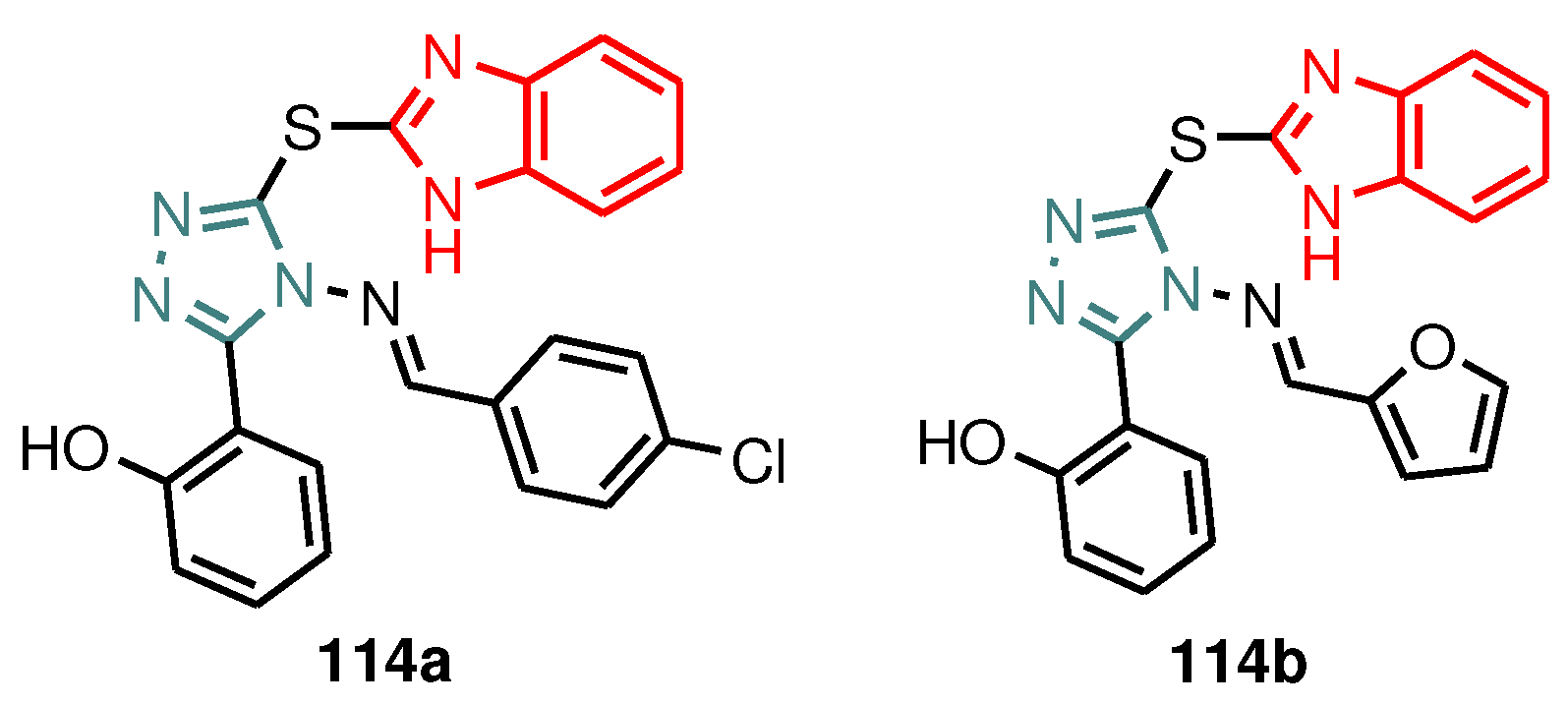
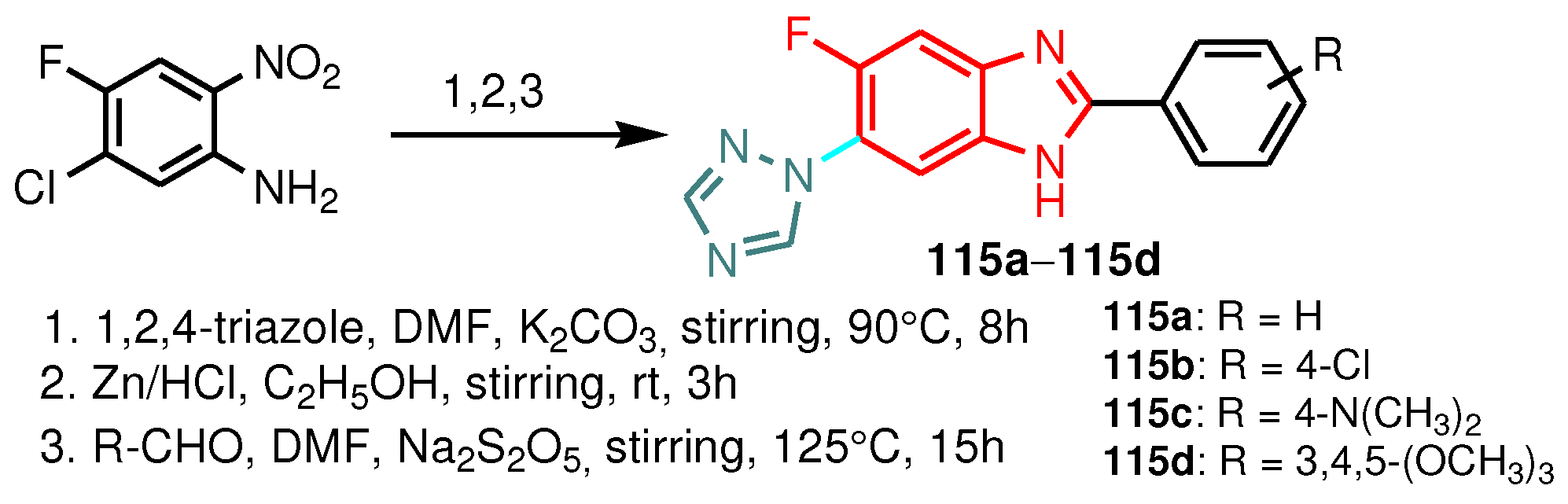
| Compound | Gram-positive organisms | Gram-negative organisms | Fungi organisms | |||
|---|---|---|---|---|---|---|
| B.c. | S.a. | P.a. | E.c. | A.b. | C.a. | |
| 6a | 64 | 64 | 256 | 128 | 128 | 128 |
| 6b | 128 | 128 | 128 | 128 | 256 | 256 |
| 6c | 256 | 128 | 256 | 64 | 256 | 156 |
| 6d | 256 | 128 | 256 | 64 | 256 | 256 |
| 6e | 256 | 128 | 256 | 64 | 256 | 256 |
| 6f | 512 | 512 | 256 | 256 | 512 | 512 |
| Ciprofloxacin | 8 | 4 | 8 | 4 | - | - |
| Compound | S. aureus | E. coli | B. subtilis | S. epidermitis | A. niger | C. albicans |
|---|---|---|---|---|---|---|
| 35a | 0.028 | 0.056 | 0.056 | 0.056 | 0.056 | 0.056 |
| 35b | 0.031 | 0.062 | 0.062 | 0.062 | 0.062 | 0.062 |
| 35c | 0.029 | 0.058 | 0.058 | 0.058 | 0.058 | 0.058 |
| 35d | 0.060 | 0.030 | 0.060 | 0.030 | 0.060 | 0.060 |
| 35e | 0.029 | 0.056 | 0.056 | 0.056 | 0.056 | 0.056 |
| 35f | 0.026 | 0.052 | 0.052 | 0.052 | 0.052 | 0.052 |
| 35g | 0.031 | 0.026 | 0.052 | 0.026 | 0.026 | 0.026 |
| Norfloxacin | 0.020 | 0.039 | 0.039 | 0.039 | - | - |
| Fluconazole | - | - | - | - | 0.04 | 0.020 |
| Compound | Inhibition zone diameters using the agar diffusion method (mm) | |||
|---|---|---|---|---|
| S. aureus | P. aeruginosa | E. coli | S. typhosa | |
| 63a | 28 | 26 | 21 | 19 |
| 63b | 23 | 18 | 16 | 14 |
| 63c | 21 | 23 | 18 | 19 |
| 63d | 20 | 22 | 23 | 23 |
| 63e | 25 | 23 | 21 | 24 |
| 63f | 27 | 26 | 24 | 20 |
| 63g | 19 | 20 | 15 | 13 |
| 63h | 29 | 26 | 22 | 24 |
| 63i | 26 | 22 | 19 | 18 |
| 63j | 14 | 12 | 16 | 16 |
| 63k | 22 | 21 | 20 | 18 |
| 63l | 25 | 23 | 19 | 21 |
| 63m | 21 | 18 | 18 | 16 |
| 63n | 24 | 22 | 22 | 21 |
| 63o | 19 | 21 | 18 | 14 |
| Gentamycin | 34 | 35 | 31 | 30 |
| Compound | F. verticillioides | D. oryzae | C. lunata | F. fujikuroi |
|---|---|---|---|---|
| 74a | 35 | 50 | 28 | 45 |
| 74b | 30 | 25 | 18 | 30 |
| 74c | 16 | 12 | 10 | 15 |
| Carbendazim | 230 | - | - | 150 |
| Propiconazole | 20 | 25 | 22 | 21 |
| Compound | S. aureus, | B. subtilis | S. mutans | P. aeruginosa | C. albicans |
|---|---|---|---|---|---|
| 88a | NT | NT | 16 | 16 | 32 |
| 88b | 8 | 16 | 16 | 16 | NT |
| 88c | 8 | 16 | 32 | 32 | 32 |
| Ampicillin | 2 | 2 | < 1 | 4 | NT |
| Kanamycin | 2 | < 1 | 4 | 2 | NT |
| Compound | Concentration (µg mL-1) | Aspergillus niger | Fusarium oxysporum |
|---|---|---|---|
| 89a | 50 | 50 | - |
| 89b | 50 | 50 | 50 |
| 89c | 50 | 50 | - |
| 89d | 50 | 50 | - |
| Compound (800 µg mL-1) | S. aureus | P. aerugnosa | B. subtilis | A. baumannii | C. albicans |
|---|---|---|---|---|---|
| 95 | 18 | 14 | 15 | - | 10 |
| 96 | 19 | 11 | 12 | - | 11 |
| 97 | 17 | 15 | 14 | 12 | - |
| Amoxicillin | 33 | 32 | 33 | - | - |
| Fluconazole | - | - | - | - | 25 |
| Compound | Minimum inhibitory concentrations (μgmL-1) | |||
|---|---|---|---|---|
| Gram-positive bacteria | Gram-negative bacteria | |||
| B. subtilis | S. aureus | E. coli | P. aeruginosa | |
| 105a | 98 | - | 52 | - |
| 105b | - | - | 65 | - |
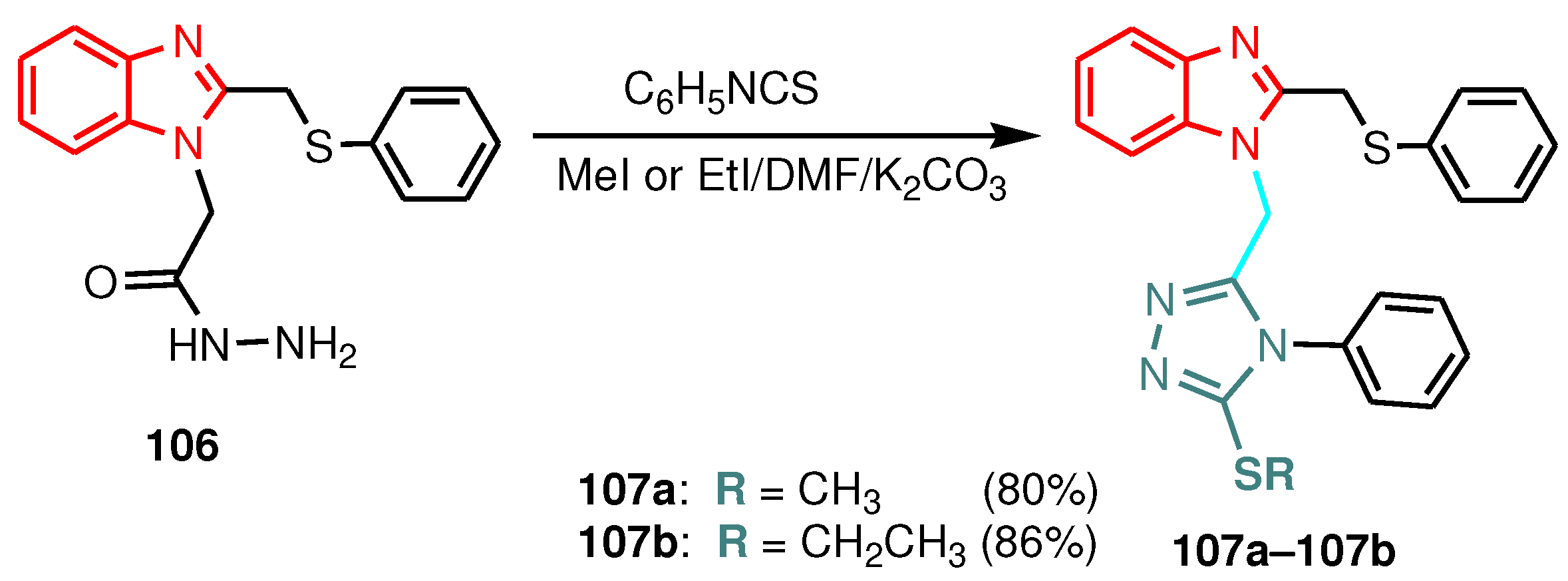
| 107a | 75 | 105 | 62 | - |
| 107b | 79 | - | 72 | - |
| Gentamycin* | 64 | 56 | 72 | 48 |
| No | Compound | Zone of inhibition (mm) | ||
|---|---|---|---|---|
| E. coli | S. aureus | C. albicans | ||
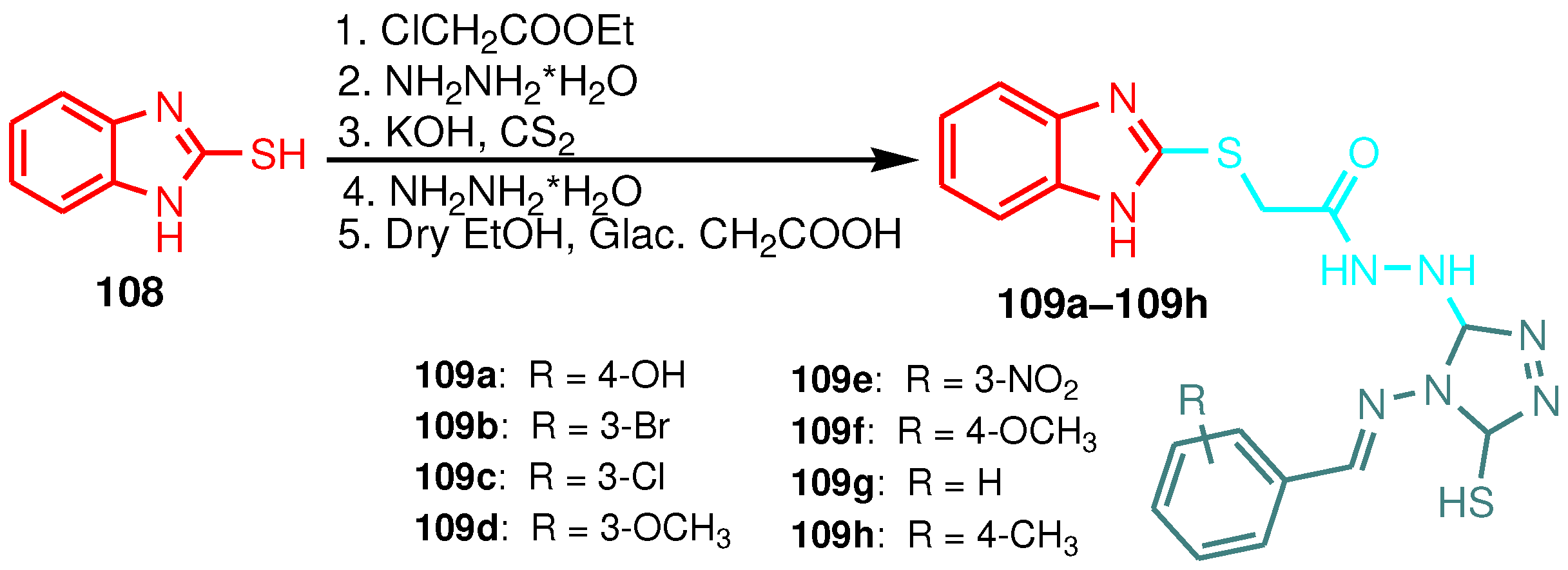
| 1 | 109a | 15 | 13 | 18 |
| 2 | 109b | 13 | 11 | 12 |
| 3 | 109c | 17 | 16 | 14 |
| 4 | 109d | 12 | 13 | 16 |
| 5 | 109e | 13 | 17 | 9 |
| 6 | 109f | 10 | 8 | 11 |
| 7 | 109g | 8 | 11 | 12 |
| 8 | 109h | 12 | 7 | 10 |
| 9 | Ampicilline | 24 | 25 | - |
| 10 | Ketokonazole | - | - | 20 |
| Compound | C. albicans | G. glabrata | C. krusei | C. parapsilosis |
|---|---|---|---|---|
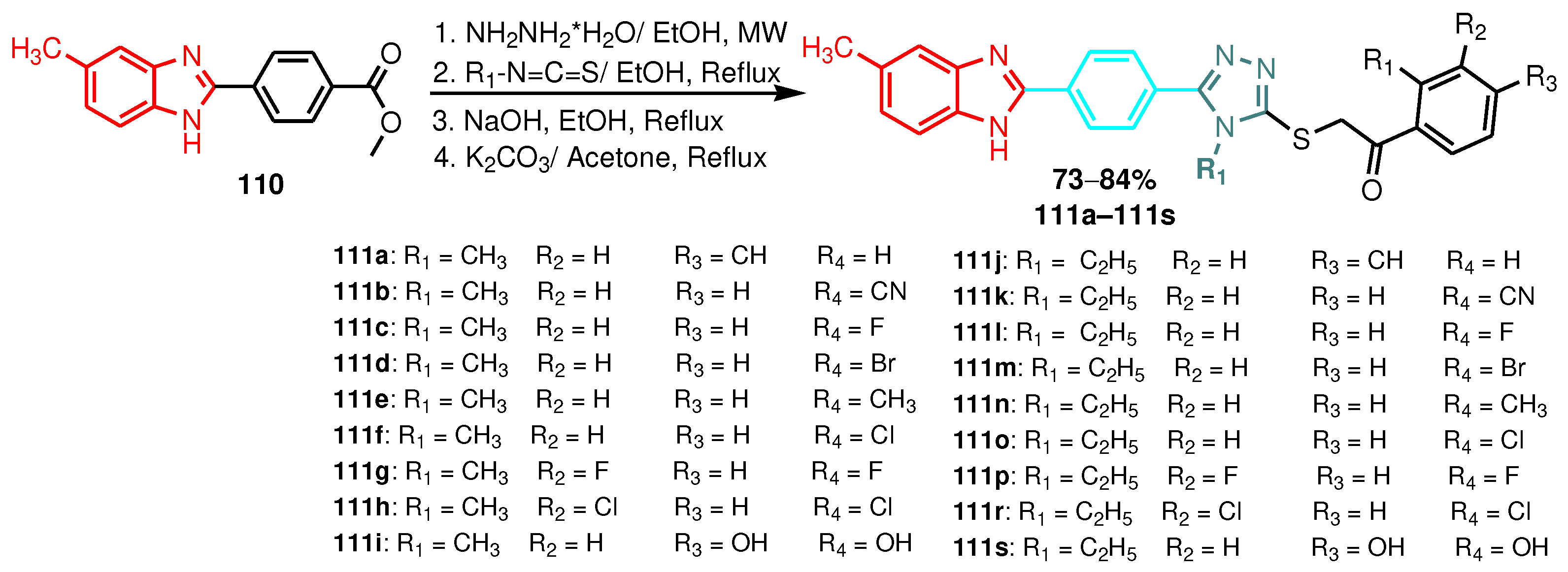
| 111a | 12.5 | 6.25 | 6.25 | 12.5 |
| 111b | 6.25 | 3.12 | 6.25 | 6.25 |
| 111c | 12.5 | 6.25 | 6.25 | 12.5 |
| 111d | 6.25 | 12.5 | 6.25 | 6.25 |
| 111e | 12.5 | 6.25 | 12.5 | 12.5 |
| 111f | 6.25 | 3.12 | 3.12 | 6.25 |
| 111g | 3.12 | 6.25 | 6.25 | 6.25 |
| 111h | 12.5 | 6.25 | 12.5 | 6.25 |
| 111i | 0.78 | 1.56 | 1.56 | 0.78 |
| 111j | 12.5 | 6.25 | 12.5 | 12.5 |
| 111k | 12.5 | 6.25 | 12.5 | 12.5 |
| 111l | 6.25 | 12.5 | 6.25 | 12.5 |
| 111m | 3.12 | 3.12 | 3.12 | 6.25 |
| 111n | 3.12 | 3.12 | 1.56 | 3.12 |
| 111o | 3.12 | 3.12 | 6.25 | 6.25 |
| 111p | 12.5 | 12.52 | 6.25 | 6.25 |
| 111r | 6.25 | 3.12 | 3.12 | 3.12 |
| 111s | 0.78 | 1.56 | 1.56 | 0.78 |
| Ketokonazole | 0.78 | 1.56 | 1.56 | 1.56 |
| Floconazole | 0.78 | 1.56 | 1.56 | 0.78 |
| Compd. | In vitro | In vivo | |||||
|---|---|---|---|---|---|---|---|
| CT50 (µg mL–1) |
EC50 (µg mL–1) |
TI | CPE Inhibition (%) |
Dose (µg per mouse per day) | MST (days) |
Protection (%) |
|
| Anti-JEV | |||||||
| 59a | 125 | 4 | 31 | 30 | 200 | - | - |
| 59b | 125 | 8 | 16 | 90 | 200 | 4 | 16 |
| 59c | - | - | - | - | - | - | - |
| 59d | 125 | 4 | 31 | 30 | 200 | - | - |
| 59e | 250 | 62.5 | 4 | 50 | 200 | 2 | 10 |
| Anti-HSV | |||||||
| 59a | 125 | 62.5 | 2 | 33 | - | - | - |
| 59b | 125 | 62.5 | 2 | 46 | - | - | - |
| 59c | - | - | - | - | - | - | - |
| 59d | 125 | 31.25 | 4 | 53 | 200 | - | - |
| 59e | 250 | 7.8 | 32 | 64 | 200 | - | - |
|
CT50 – 50% cytotoxic concentration, EC50 – 50% effective concentration, TI –therapeutic index (TI= CT50/ EC50) CPE - cytopathic effect, MST – mean survival time | |||||||
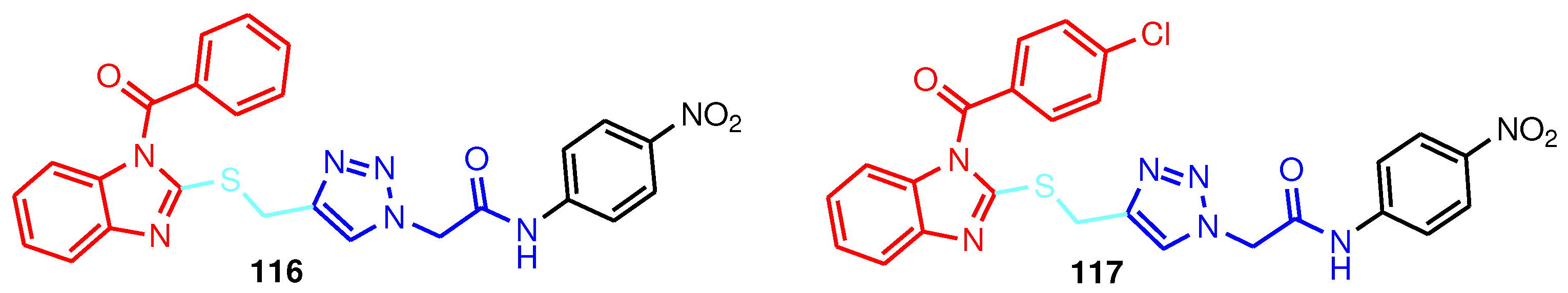
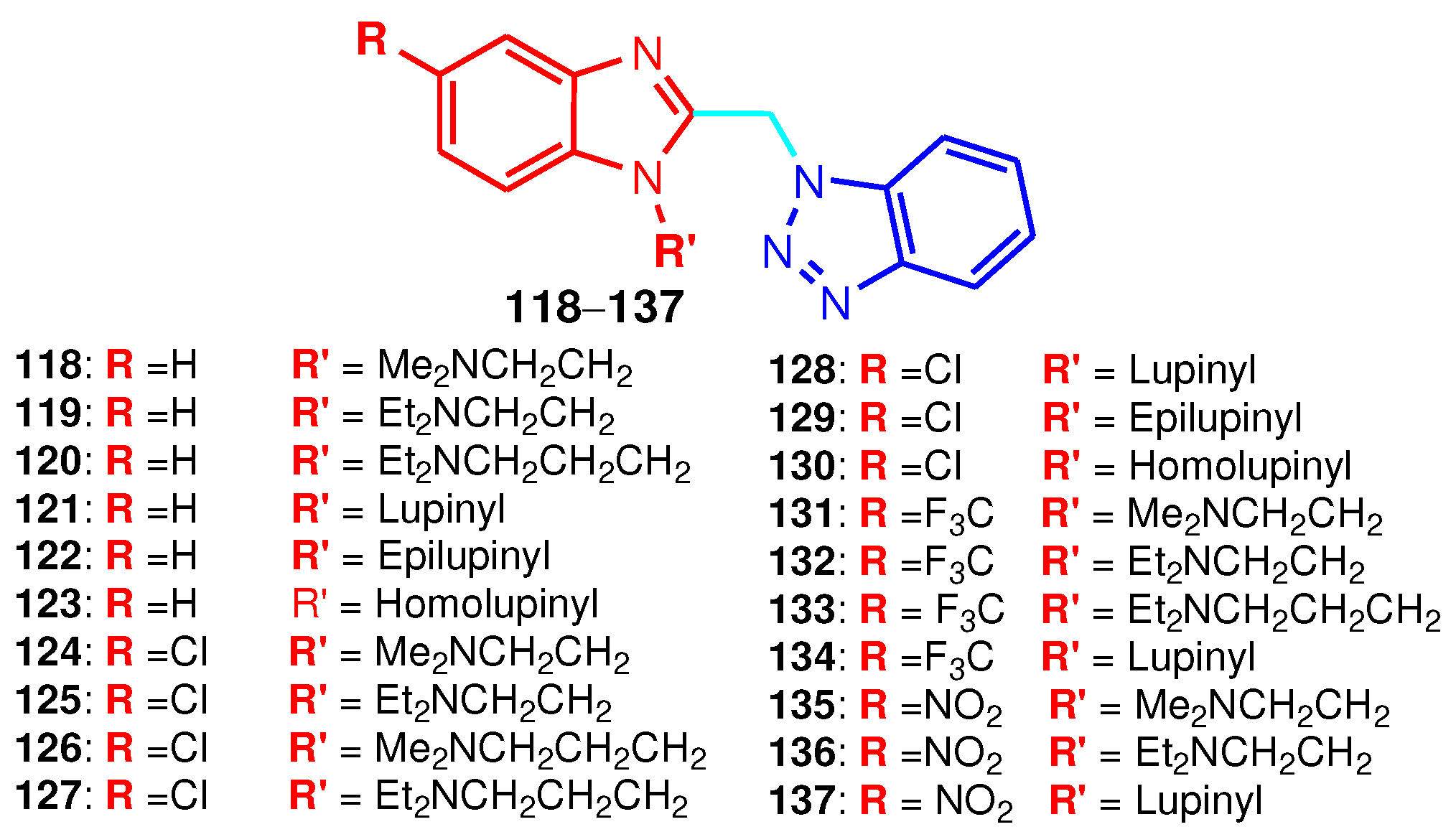
| Compound | Anti-RSV activity | Anti-BVDV activity | Anti-YFV activity | Anti-CVB2 activity |
|---|---|---|---|---|
| 118 | 0.7 | - | - | - |
| 119 | 2.3 | - | - | - |
| 120 | 0.7 | > 100 | 80 | > 100 |
| 121 | 0.7 | 63 | > 90 | > 100 |
| 122 | 0.3 | 53 | > 70 | > 100 |
| 123 | 0.15 | 51 | > 60 | > 100 |
| 124 | 0.03 | - | - | - |
| 125 | 0.7 | - | - | - |
| 126 | 0.06 | 90 | > 100 | > 100 |
| 127 | 0.1 | 72 | > 54 | > 100 |
| 128 | 0.9 | 15 | 6 | 40 |
| 129 | 0.05 | 19 | > 21 | > 88 |
| 130 | 0.02 | 14 | > 20 | 26 |
| 131 | 10.0 | - | - | - |
| 132 | 7.0 | - | - | - |
| 133 | 1.9 | 67 | > 36 | > 100 |
| 134 | > 36 | 15 | > 18 | > 36 |
| 135 | 9 | - | - | - |
| 136 | 11 | 80 | > 45 | > 100 |
| 137 | 23.0 | 80 | 27 | > 83 |
| 6-Azaurine | 1.2 | > 100 | 26 | > 100 |
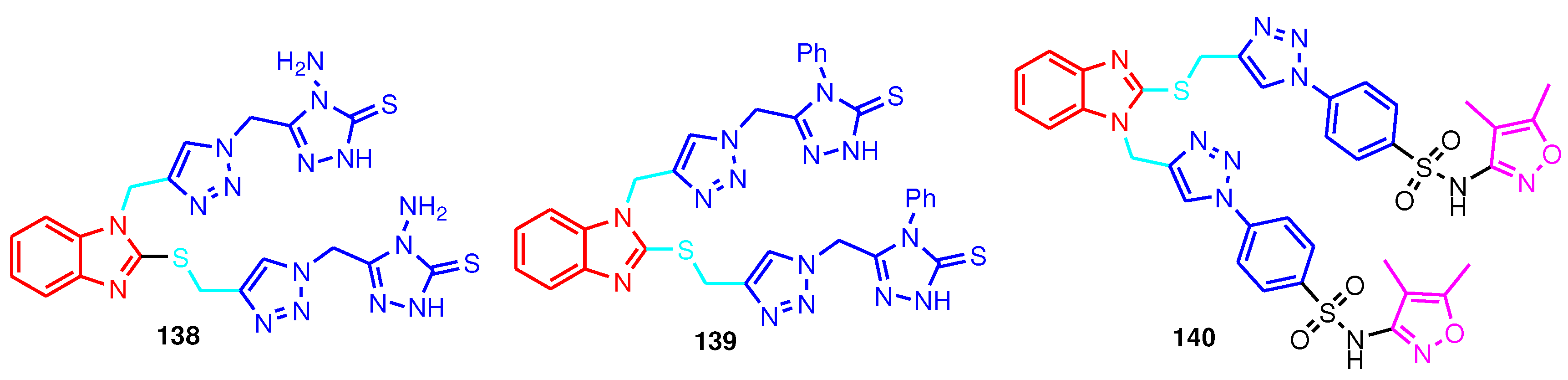
| Compound | CC50 (mg mL-1) | EC50 (mg mL-1) | Selectivity Index (SI) |
|---|---|---|---|
| Ceftazidime | 1045.53 | 85.07 | 12.29 |
| 138 | 1065.51 | 155.05 | 6.87 |
| 139 | 1530.5 | 306.1 | 5.0 |
| 140 | 1028.28 | 80.4 | 12.78 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




