Submitted:
22 June 2023
Posted:
22 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
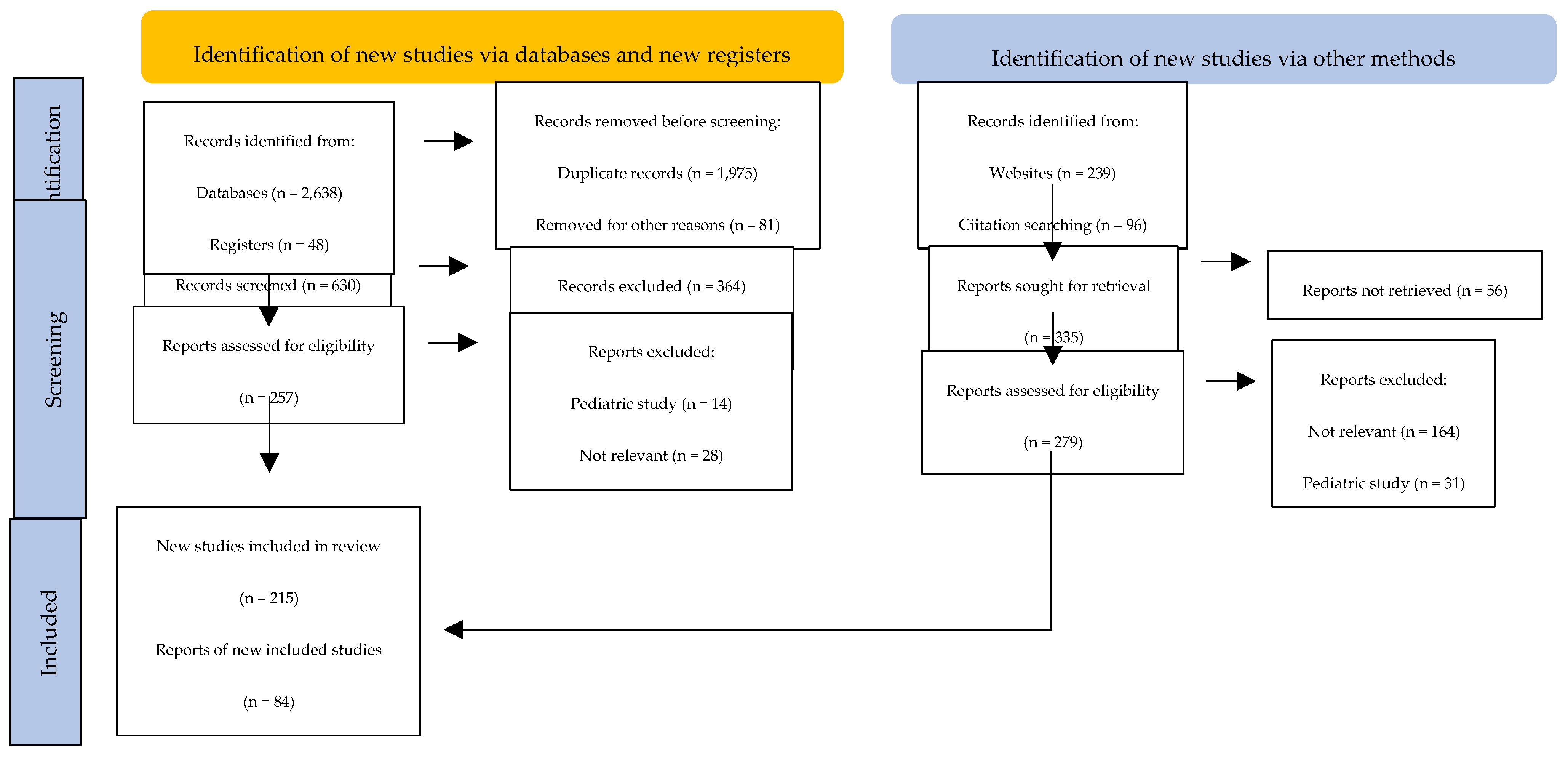
2. Methods (Search Strategy)
3. Major Thyroid Antigens
3.1. Tg
3.2. TPO
3.3. TSHR
4. Minor Thyroid Antigens
4.1. NIS
4.2. PDN
4.3. Meg
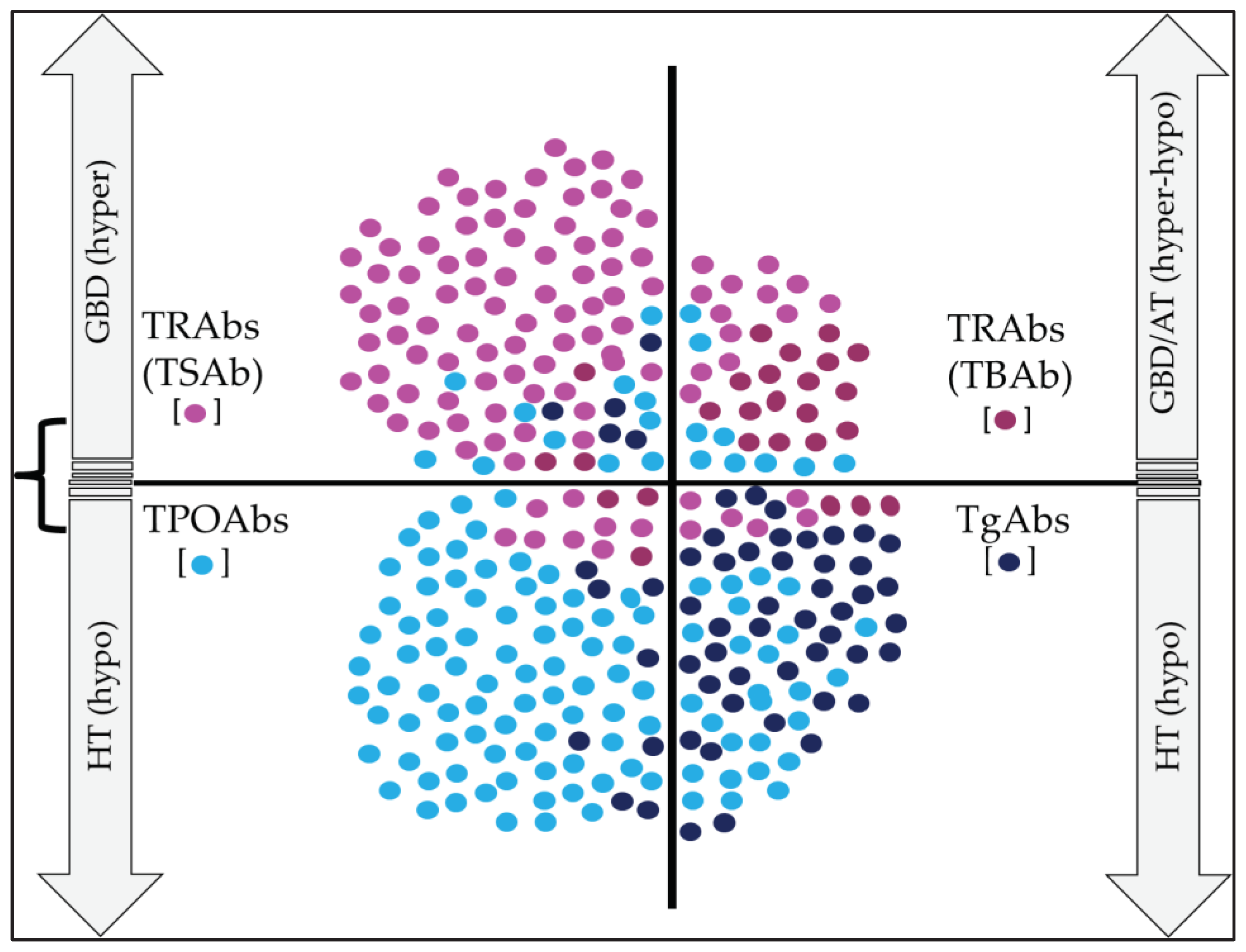
5. Major Thyroid Abs
5.1. TgAbs
5.2. TPOAbs
5.3. TRAbs
5.4. NISAbs and PDNAbs
5.5. MegAbs
6. Clinical Utility of Thyroid Abs in AITD
6.1. Clinical Utility of TgAbs
6.2. Clinical Utility of TPOAbs
6.3. Clinical Utility of TRAbs
- In individuals with HT and hypothyroidism, where there is adequate and stable control with very low doses of levothyroxine, since it is possible in this type of patient that a release of TBAb has occurred, causing a type of transient hypothyroidism, which could have a high recovery rate;
- In a newborn born to a mother with HT, but with the presence of contradictory or bizarre clinical findings;
- In patients with HT and clinical findings suggestive of thyroid ophthalmopathy;
- When in individuals with long-standing hypothyroidism, under treatment with stable doses of levothyroxine, a change to the state of hyperthyroidism is noted;
- When alternating periods of hyperthyroidism and hypothyroidism occur in the same patient.
6.4. Clinical Utility of PDNAbs, NISAbs, MegAbs, and Other Abs
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Skevaki C, Wesemann DR. Antibody repertoire and autoimmunity. J Allergy Clin Immunol. 2023;151(4):898-900. [CrossRef]
- Huffaker MF, Sanda S, Chandran S, Chung SA, St Clair EW, Nepom GT, Smilek DE. Approaches to Establishing Tolerance in Immune Mediated Diseases. Front Immunol. 2021;12:744804. [CrossRef]
- Petersone L, Edner NM, Ovcinnikovs V, Heuts F, Ross EM, Ntavli E, Wang CJ, Walker LSK. T Cell/B Cell Collaboration and Autoimmunity: An Intimate Relationship. Front Immunol. 2018;9:1941. [CrossRef]
- Corneth OBJ, Neys SFH, Hendriks RW. Aberrant B Cell Signaling in Autoimmune Diseases. Cells. 2022;11(21):3391. [CrossRef]
- Samuels H, Malov M, Saha Detroja T, Ben Zaken K, Bloch N, Gal-Tanamy M, Avni O, Polis B, Samson AO. Autoimmune Disease Classification Based on PubMed Text Mining. J Clin Med. 2022;11(15):4345. [CrossRef]
- Hundt JE, Hoffmann MH, Amber KT, Ludwig RJ. Editorial: Autoimmune pre-disease. Front Immunol. 2023;14:1159396. [CrossRef]
- Chi X, Huang M, Tu H, Zhang B, Lin X, Xu H, Dong C, Hu X. Innate and adaptive immune abnormalities underlying autoimmune diseases: the genetic connections. Sci China Life Sci. 2023:1–36. [CrossRef]
- Mazzone R, Zwergel C, Artico M, Taurone S, Ralli M, Greco A, Mai A. The emerging role of epigenetics in human autoimmune disorders. Clin Epigenetics. 2019;11(1):34. [CrossRef]
- Bogusławska J, Godlewska M, Gajda E, Piekiełko-Witkowska A. Cellular and molecular basis of thyroid autoimmunity. Eur Thyroid J. 2022;11(1):e210024. [CrossRef]
- McLachlan SM, Rapoport B. Discoveries in Thyroid Autoimmunity in the Past Century. Thyroid. 2023;33(3):278-286. [CrossRef]
- Rahimova, RR. Autoimmune thyroiditis (review of literature). Klin Lab Diagn. 2022;67(5):286-291. [CrossRef]
- Stasiak M, Lewiński A. New aspects in the pathogenesis and management of subacute thyroiditis. Rev Endocr Metab Disord. 2021;22(4):1027-1039. [CrossRef]
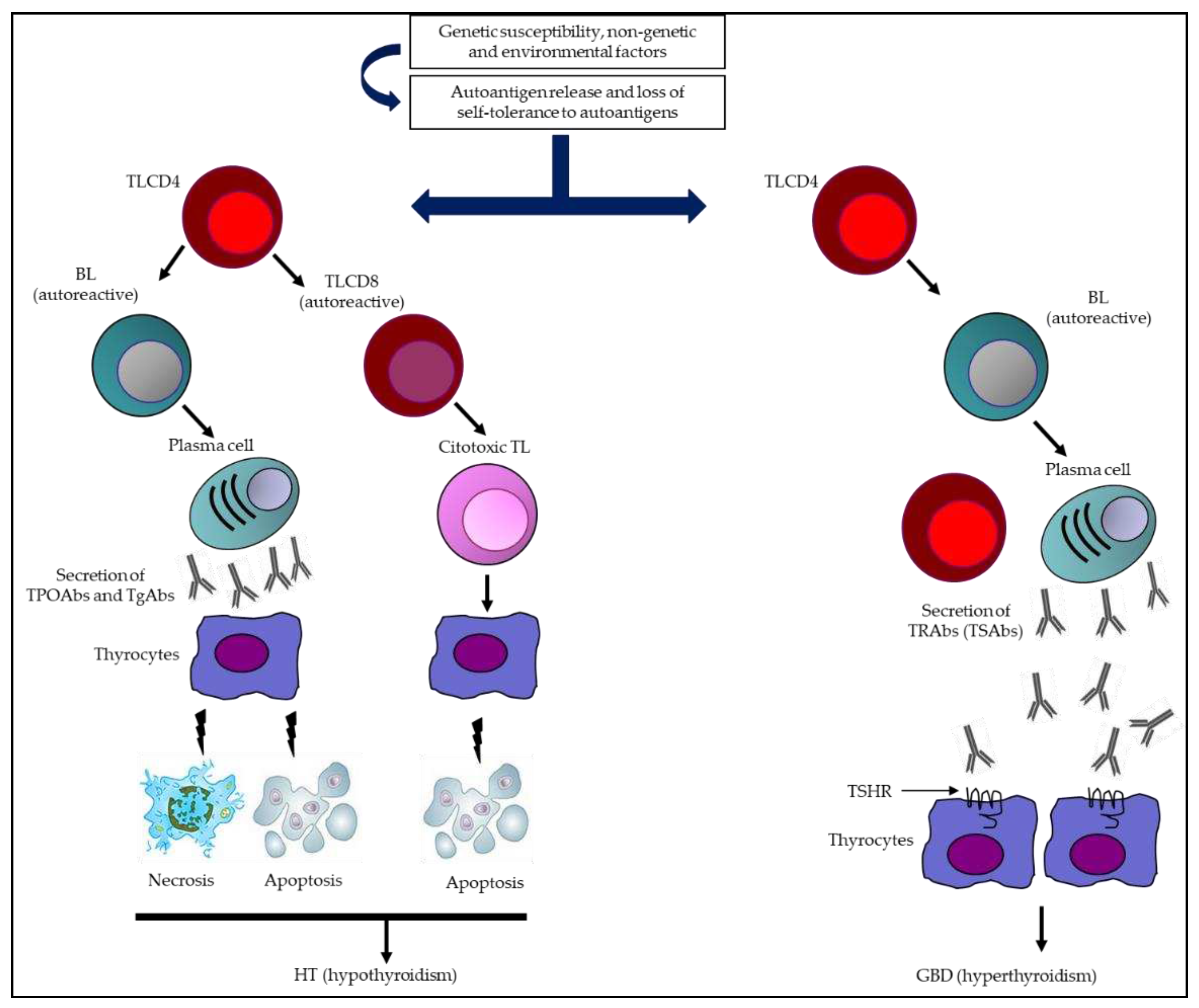
- Vargas-Uricoechea, H. Molecular Mechanisms in Autoimmune Thyroid Disease. Cells. 2023;12(6):918. [CrossRef]
- Daramjav N, Takagi J, Iwayama H, Uchino K, Inukai D, Otake K, Ogawa T, Takami A. Autoimmune Thyroiditis Shifting from Hashimoto's Thyroiditis to Graves' Disease. Medicina (Kaunas). 2023;59(4):757. [CrossRef]
- Wémeau JL, Klein M, Sadoul JL, Briet C, Vélayoudom-Céphise FL. Graves' disease: Introduction, epidemiology, endogenous and environmental pathogenic factors. Ann Endocrinol (Paris). 2018;79(6):599-607. [CrossRef]
- Chang, Y. Graves' Disease is a Thyroid Autoimmune Disorder Identified by Excessive Thyroid Hormone Production. Thyroid Disorders Ther. 2023;12:294.
- Coscia F, Taler-Verčič A, Chang VT, Sinn L, O'Reilly FJ, Izoré T, Renko M, Berger I, Rappsilber J, Turk D, Löwe J. The structure of human thyroglobulin. Nature. 2020;578(7796):627-630. [CrossRef]
- Citterio CE, Rivolta CM, Targovnik HM. Structure and genetic variants of thyroglobulin: Pathophysiological implications. Mol Cell Endocrinol. 2021;528:111227. [CrossRef]
- Tosatto L, Coscia F. A glance at post-translational modifications of human thyroglobulin: potential impact on function and pathogenesis. Eur Thyroid J. 2022;11(3):e220046. [CrossRef]
- Godlewska M, Gawel D, Buckle AM, Banga JP. Thyroid Peroxidase Revisited - What's New? Horm Metab Res. 2019;51(12):765-769. [CrossRef]
- Ruf J, Carayon P. Structural and functional aspects of thyroid peroxidase. Arch Biochem Biophys. 2006;445(2):269-77. [CrossRef]
- Godlewska M, Banga PJ. Thyroid peroxidase as a dual active site enzyme: Focus on biosynthesis, hormonogenesis and thyroid disorders of autoimmunity and cancer. Biochimie. 2019;160:34-45. [CrossRef]
- Williams DE, Le SN, Godlewska M, Hoke DE, Buckle AM. Thyroid Peroxidase as an Autoantigen in Hashimoto's Disease: Structure, Function, and Antigenicity. Horm Metab Res. 2018;50(12):908-921. [CrossRef]
- Mondal S, Raja K, Schweizer U, Mugesh G. Chemistry and Biology in the Biosynthesis and Action of Thyroid Hormones. Angew Chem Int Ed Engl. 2016;55(27):7606-7630. [CrossRef]
- Chu YD, Yeh CT. The Molecular Function and Clinical Role of Thyroid Stimulating Hormone Receptor in Cancer Cells. Cells. 2020;9(7):1730. [CrossRef]
- Vieira IH, Rodrigues D, Paiva I. The Mysterious Universe of the TSH Receptor. Front Endocrinol (Lausanne). 2022;13:944715. [CrossRef]
- Marín-Sánchez A, Álvarez-Sierra D, González O, Lucas-Martin A, Sellés-Sánchez A, Rudilla F, Enrich E, Colobran R, Pujol-Borrell R. Regulation of TSHR Expression in the Thyroid and Thymus May Contribute to TSHR Tolerance Failure in Graves' Disease Patients via Two Distinct Mechanisms. Front Immunol. 2019;10:1695. [CrossRef]
- Kleinau G, Vassart G. TSH Receptor Mutations and Diseases. 2017 Jul 24. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000–. [PubMed]
- Gershengorn MC, Osman R. Molecular and cellular biology of thyrotropin-releasing hormone receptors. Physiol Rev. 1996;76(1):175-191. [CrossRef]
- Rapoport B, McLachlan SM. TSH Receptor Cleavage Into Subunits and Shedding of the A-Subunit; A Molecular and Clinical Perspective. Endocr Rev. 2016;37(2):114-34. [CrossRef]
- Helfinger L, Tate CG. Expression and Purification of the Human Thyroid-Stimulating Hormone Receptor. Methods Mol Biol. 2022;2507:313-325. [CrossRef]
- Führer, D. Constitutive TSH receptor activation as a hallmark of thyroid autonomy. Endocrine. 2020;68(2):274-278. [CrossRef]
- Korta P, Pocheć E. Glycosylation of thyroid-stimulating hormone receptor. Endokrynol Pol. 2019;70(1):86-100. [CrossRef]
- Darrouzet E, Lindenthal S, Marcellin D, Pellequer JL, Pourcher T. The sodium/iodide symporter: state of the art of its molecular characterization. Biochim Biophys Acta. 2014;1838(1 Pt B):244-253. [CrossRef]
- Ravera S, Reyna-Neyra A, Ferrandino G, Amzel LM, Carrasco N. The Sodium/Iodide Symporter (NIS): Molecular Physiology and Preclinical and Clinical Applications. Annu Rev Physiol. 2017;79:261-289.
- Riesco-Eizaguirre G, Santisteban P, De la Vieja A. The complex regulation of NIS expression and activity in thyroid and extrathyroidal tissues. Endocr Relat Cancer. 2021;28(10):T141-T165. [CrossRef]
- Thompson RJ, Fletcher A, Brookes K, Nieto H, Alshahrani MM, Mueller JW, Fine NHF, Hodson DJ, Boelaert K, Read ML, Smith VE, McCabe CJ. Dimerization of the Sodium/Iodide Symporter. Thyroid. 2019;29(10):1485-1498. [CrossRef]
- Portulano C, Paroder-Belenitsky M, Carrasco N. The Na+/I- symporter (NIS): mechanism and medical impact. Endocr Rev. 2014;35(1):106-149. [CrossRef]
- Ravera S, Nicola JP, Salazar-De Simone G, Sigworth FJ, Karakas E, Amzel LM, Bianchet MA, Carrasco N. Structural insights into the mechanism of the sodium/iodide symporter. Nature. 2022;612(7941):795-801.
- Rozenfeld J, Efrati E, Adler L, Tal O, Carrithers SL, Alper SL, Zelikovic I. Transcriptional regulation of the pendrin gene. Cell Physiol Biochem. 2011;28(3):385-396. [CrossRef]
- Dossena S, Nofziger C, Tamma G, Bernardinelli E, Vanoni S, Nowak C, Grabmayer E, Kössler S, Stephan S, Patsch W, Paulmichl M. Molecular and functional characterization of human pendrin and its allelic variants. Cell Physiol Biochem. 2011;28(3):451-466. [CrossRef]
- Dossena S, Bizhanova A, Nofziger C, Bernardinelli E, Ramsauer J, Kopp P, Paulmichl M. Identification of allelic variants of pendrin (SLC26A4) with loss and gain of function. Cell Physiol Biochem. 2011;28(3):467-476. [CrossRef]
- Bizhanova A, Kopp P. Controversies concerning the role of pendrin as an apical iodide transporter in thyroid follicular cells. Cell Physiol Biochem. 2011;28(3):485-490. [CrossRef]
- Twyffels L, Massart C, Golstein PE, Raspe E, Van Sande J, Dumont JE, Beauwens R, Kruys V. Pendrin: the thyrocyte apical membrane iodide transporter? Cell Physiol Biochem. 2011;28(3):491-496. [CrossRef]
- Zheng G, Marino' M, Zhao J, McCluskey RT. Megalin (gp330): a putative endocytic receptor for thyroglobulin (Tg). Endocrinology. 1998;139(3):1462-1465. [CrossRef]
- Marinò M, Pinchera A, McCluskey RT, Chiovato L. Megalin in thyroid physiology and pathology. Thyroid. 2001;11(1):47-56. [CrossRef]
- Marinò M, Zheng G, Chiovato L, Pinchera A, Brown D, Andrews D, McCluskey RT. Role of megalin (gp330) in transcytosis of thyroglobulin by thyroid cells. A novel function in the control of thyroid hormone release. J Biol Chem. 2000;275(10):7125-7137. [CrossRef]
- Goto S, Hosojima M, Kabasawa H, Saito A. The endocytosis receptor megalin: From bench to bedside. Int J Biochem Cell Biol. 2023;157:106393. [CrossRef]
- Lee J, Sul HJ, Kim KH, Chang JY, Shong M. Primary Cilia Mediate TSH-Regulated Thyroglobulin Endocytic Pathways. Front Endocrinol (Lausanne). 2021;12:700083. [CrossRef]
- Soh SB, Aw TC. Laboratory Testing in Thyroid Conditions - Pitfalls and Clinical Utility. Ann Lab Med. 2019;39(1):3-14. [CrossRef]
- Doggui, R. Immunoanalytical profile of thyroglobulin antibodies. Ann Biol Clin (Paris). 2018;76(6):695-704. [CrossRef]
- Dwivedi SN, Kalaria T, Buch H. Thyroid autoantibodies. J Clin Pathol. 2023;76(1):19-28. [CrossRef]
- Li Y, Zhao C, Zhao K, Yu N, Li Y, Yu Y, Zhang Y, Song Z, Huang Y, Lu G, Gao Y, Zhang J, Guo X. Glycosylation of Anti-Thyroglobulin IgG1 and IgG4 Subclasses in Thyroid Diseases. Eur Thyroid J. 2021;10(2):114-124. [CrossRef]
- McIntosh RS, Weetman AP. Molecular analysis of the antibody response to thyroglobulin and thyroid peroxidase. Thyroid. 1997;7(3):471-487. [CrossRef]
- Khan FA, Al-Jameil N, Khan MF, Al-Rashid M, Tabassum H. Thyroid dysfunction: an autoimmune aspect. Int J Clin Exp Med. 2015;8(5):6677-6681.
- Bílek R, Dvořáková M, Grimmichová T, Jiskra J. Iodine, thyroglobulin and thyroid gland. Physiol Res. 2020;69(Suppl 2):S225-S236. [CrossRef]
- McLeod DS, Cooper DS. The incidence and prevalence of thyroid autoimmunity. Endocrine. 2012;42(2):252-265. [CrossRef]
- McLachlan SM, Rapoport B. Thyroid Autoantibodies Display both "Original Antigenic Sin" and Epitope Spreading. Front Immunol. 2017;8:1845. [CrossRef]
- Baker S, Miguel RN, Thomas D, Powell M, Furmaniak J, Smith BR. Cryo-electron microscopy structures of human thyroid peroxidase (TPO) in complex with TPO antibodies. J Mol Endocrinol. 2023;70(3):e220149. [CrossRef]
- Espenbetova M, Kuzmina N, Zubkov A, Akhmetova V, Zamanbekova Z, Krykpaeva A, Zhumanbayeva Z, Amrenova K, Smailova Z, Glushkova N. Epitopes specificity of antibodies to thyroid peroxidase in patients with Graves' disease, Hashimoto's thyroiditis and overlap-syndrome. J Clin Transl Endocrinol. 2022;27:100293. [CrossRef]
- Tian X, Li N, Su R, Dai C, Zhang R. Selenium Supplementation May Decrease Thyroid Peroxidase Antibody Titer via Reducing Oxidative Stress in Euthyroid Patients with Autoimmune Thyroiditis. Int J Endocrinol. 2020;2020:9210572. [CrossRef]
- Williams DE, Le SN, Hoke DE, Chandler PG, Gora M, Godlewska M, Banga JP, Buckle AM. Structural Studies of Thyroid Peroxidase Show the Monomer Interacting With Autoantibodies in Thyroid Autoimmune Disease. Endocrinology. 2020;161(2):bqaa016. [CrossRef]
- Fröhlich E, Wahl R. Thyroid Autoimmunity: Role of Anti-thyroid Antibodies in Thyroid and Extra-Thyroidal Diseases. Front Immunol. 2017;8:521. [CrossRef]
- Nishihara E, Amino N, Kudo T, Ito M, Fukata S, Nishikawa M, Nakamura H, Miyauchi A. Comparison of thyroglobulin and thyroid peroxidase antibodies measured by five different kits in autoimmune thyroid diseases. Endocr J. 2017;64(10):955-961. [CrossRef]
- Wolffenbuttel BHR, Wouters HJCM, Muller Kobold AC, Roozendaal C, Klauw MM. Comparison of four commercially available thyroid peroxidase autoantibody and two thyroglobulin autoantibody assays. Available in: https://assets.researchsquare.com/files/rs-1550125/v1/a9384112-5494-4d6c-9c27-3e8e464faebd.pdf?c=1655112553 (Accessed May 3, 2023).
- La'ulu SL, Slev PR, Roberts WL. Performance characteristics of 5 automated thyroglobulin autoantibody and thyroid peroxidase autoantibody assays. Clin Chim Acta. 2007;376(1-2):88-95. [CrossRef]
- Hu X, Chen Y, Shen Y, Tian R, Sheng Y, Que H. Global prevalence and epidemiological trends of Hashimoto's thyroiditis in adults: A systematic review and meta-analysis. Front Public Health. 2022;10:1020709. [CrossRef]
- Pedersen IB, Knudsen N, Jørgensen T, Perrild H, Ovesen L, Laurberg P. Thyroid peroxidase and thyroglobulin autoantibodies in a large survey of populations with mild and moderate iodine deficiency. Clin Endocrinol (Oxf). 2003;58(1):36-42. [CrossRef]
- Camargo RY, Tomimori EK, Neves SC, Knobel M, Medeiros-Neto G. Prevalence of chronic autoimmune thyroiditis in the urban area neighboring a petrochemical complex and a control area in Sao Paulo, Brazil. Clinics (Sao Paulo). 2006;61(4):307-312. [CrossRef]
- Tammaro A, Pigliacelli F, Fumarola A, Persechino S. Trends of thyroid function and autoimmunity to 5 years after the introduction of mandatory iodization in Italy. Eur Ann Allergy Clin Immunol. 2016;48(3):77-81.
- Ehlers M, Allelein S, Schott M. TSH-receptor autoantibodies: pathophysiology, assay methods, and clinical applications. Minerva Endocrinol. 2018;43(3):323-332. [CrossRef]
- Nicolì F, Lanzolla G, Mantuano M, Ionni I, Mazzi B, Leo M, Sframeli A, Posarelli C, Maglionico MN, Figus M, Nardi M, Marcocci C, Marinò M. Correlation between serum anti-TSH receptor autoantibodies (TRAbs) and the clinical feature of Graves' orbitopathy. J Endocrinol Invest. 2021;44(3):581-585. [CrossRef]
- Kahaly GJ, Diana T, Olivo PD. TSH Receptor Antibodies: Relevance & Utility. Endocr Pract. 2020;26(1):97-106. [CrossRef]
- McLachlan SM, Rapoport B. Thyrotropin-blocking autoantibodies and thyroid-stimulating autoantibodies: potential mechanisms involved in the pendulum swinging from hypothyroidism to hyperthyroidism or vice versa. Thyroid. 2013;23(1):14-24. [CrossRef]
- Da Silva Santos T, Oliveira JC, Freitas C, Couto de Carvalho A. Thyroid-Stimulatory Antibody as a Predictive Factor for Graves' Disease Relapse. Cureus. 2022;14(2):e22190. [CrossRef]
- Arshad I, Zahra T, Vargas-Jerez J. New-Onset Graves' Disease in the Background of Hashimoto's Thyroiditis: Spectrums of the Same Disease With Changing Autoantibodies. Cureus. 2022;14(8):e28296. [CrossRef]
- Kotwal A, Stan M. Thyrotropin Receptor Antibodies-An Overview. Ophthalmic Plast Reconstr Surg. 2018;34(4S Suppl 1):S20-S27. [CrossRef]
- Michalek K, Morshed SA, Latif R, Davies TF. TSH receptor autoantibodies. Autoimmun Rev. 2009;9(2):113-116. [CrossRef]
- Gupta AK, Kumar S. Utility of Antibodies in the Diagnoses of Thyroid Diseases: A Review Article. Cureus. 2022;14(11):e31233. [CrossRef]
- Autilio C, Morelli R, Locantore P, Pontecorvi A, Zuppi C, Carrozza C. Stimulating TSH receptor autoantibodies immunoassay: analytical evaluation and clinical performance in Graves' disease. Ann Clin Biochem. 2018;55(1):172-177. [CrossRef]
- López Ortega JM, Martínez PS, Acevedo-León D, Capell NE. Anti-TSH receptor antibodies (TRAb): Comparison of two third generation automated immunoassays broadly used in clinical laboratories and results interpretation. PLoS ONE.2022;17(7): e0270890.
- Struja T, Jutzi R, Imahorn N, Kaeslin M, Boesiger F, Kutz A, Mundwiler E, Huber A, Kraenzlin M, Mueller B, Meier C, Bernasconi L, Schuetz P. Comparison of Five TSH-Receptor Antibody Assays in Graves' disease: results from an observational pilot study. BMC Endocr Disord. 2019;19(1):38.
- Allelein S, Ehlers M, Goretzki S, Hermsen D, Feldkamp J, Haase M, Dringenberg T, Schmid C, Hautzel H, Schott M. Clinical Evaluation of the First Automated Assay for the Detection of Stimulating TSH Receptor Autoantibodies. Horm Metab Res. 2016;48(12):795-801.
- Liu T, Zhang X, Long L, Zhou L, Chen J, Li M, Gao Y, Zhou X, Han X, Ji L. Clinical evaluation of an automated TSI bridge immunoassay in the diagnosis of Graves' disease and its relationship to the degree of hyperthyroidism. BMC Endocr Disord. 2022;22(1):218.
- Brix TH, Hegedüs L, Weetman AP, Kemp HE. Pendrin and NIS antibodies are absent in healthy individuals and are rare in autoimmune thyroid disease: evidence from a Danish twin study. Clin Endocrinol (Oxf). 2014;81(3):440-444.
- Eleftheriadou AM, Mehl S, Renko K, Kasim RH, Schaefer JA, Minich WB, Schomburg L. Re-visiting autoimmunity to sodium-iodide symporter and pendrin in thyroid disease. Eur J Endocrinol. 2020;183(6):571-580. [CrossRef]
- https://www.biossusa.com/products/bs-0448r (Accesed May 5, 2023).
- https://www.mybiosource.com/polyclonal-nis-human-antibody/sodium-iodide-symporter/2026331 (Accesed May 5, 2023).
- https://www.arp1.com/anti-nai-symporter-nis-polyclonal-antibody-03-16093.html (Accesed may 5, 2023).
- https://www.lsbio.com/antibodies/ihc-plus-slc5a5-antibody-nis-antibody-if-immunofluorescence-ihc-wb-western-ls-b15569/697202?trid=247 (Accesed May 5, 2023).
- https://www.biorbyt.com/nis-antibody-orb11131.html (Accesed May 5, 2023).
- https://www.genetex.com/Product/Detail/NIS-antibody/GTX37599?utm_source=Biocompare&utm_medium=referral&utm_campaign=Biocompare_GeneTex (Accesed May 5, 2023). 5 May.
- https://www.biocompare.com/9776-Antibodies/17754578-SLC26A4-Pendrin-Antibody/?pda=9776|17754578_0_1||1|Pendrin&dfp=true (Accesed May 8, 2023).
- https://www.biocompare.com/9776-Antibodies/8077560-SLC26A4-antibody/?pda=9776|8077560_0_1||2|Pendrin&dfp=true (Accesed May 8, 2023).
- https://www.biocompare.com/9776-Antibodies/4920922-SLC26A4-Pendrin-Mouse-anti-Human-Monoclonal-3D2-Antibody/?pda=9776|4920922_0_0||3|Pendrin (Accesed May 8, 2023).
- https://www.biocompare.com/9776-Antibodies/12145581-Pendrin-Antibody-SLC26A4/?pda=9776|12145581_0_0||4|Pendrin (Accesed May 8, 2023).
- https://www.biocompare.com/9776-Antibodies/14074098-Immunotag-8482-S26A4-Polyclonal-Antibody/?pda=9776|14074098_0_0||5|Pendrin (Accesed May 8, 2023).
- Seissler J, Wagner S, Schott M, Lettmann M, Feldkamp J, Scherbaum WA, Morgenthaler NG. Low frequency of autoantibodies to the human Na(+)/I(-) symporter in patients with autoimmune thyroid disease. J Clin Endocrinol Metab. 2000;85:4630–4634.
- Morris JC, Bergert ER, Bryant WP. Binding of immunoglobulin G from patients with autoimmune thyroid disease to rat sodium-iodide symporter peptides: evidence for the iodide transporter as an autoantigen. Thyroid. 1997;7:527–534.
- Yoshida A, Hisatome I, Taniguchi S, Shirayoshi Y, Yamamoto Y, Miake J, Ohkura T, Akama T, Igawa O, Shigemasa C et al. Pendrin is a novel autoantigen recognized by patients with autoimmune thyroid diseases. J Clin Endocrinol Metab. 2009;94:442–448.
- Ajjan RA, Kemp EH, Waterman EA, Watson PF, Endo T, Onaya T, Weetman AP. Detection of binding and blocking autoantibodies to the human sodium-iodide symporter in patients with autoimmune thyroid disease. J Clin Endocrinol Metab.2000;85:2020–2027.
- Brix TH, Hegedus L, Weetman AP & Kemp HE. Pendrin and NIS antibodies are absent in healthy individuals and are rare in autoimmune thyroid disease: evidence from a Danish twin study. Clinical Endocrinology. 2014;81:440–444.
- Ajjan RA, Findlay C, Metcalfe RA, Watson PF, Crisp M, Ludgate M & Weetman AP. The modulation of the human sodium iodide symporter activity by Graves’ disease sera. J Clin Endocrinol Metab. 1998;83:1217–1221.
- Eleftheriadou AM, Mehl S, Renko K, Kasim RH, Schaefer JA, Minich WB, Schomburg L. Re- visiting autoimmunity to sodium-iodide symporter and pendrin in thyroid disease. Eur J Endocrinol. 2020;183(6):571-580.
- Lisi S, Pinchera A, McCluskey RT, Willnow TE, Refetoff S, Marcocci C, Vitti P, Menconi F, Grasso L, Luchetti F, Collins AB, Marino M. Preferential megalin-mediated transcytosis of low-hormonogenic thyroglobulin: a control mechanism for thyroid hormone release. Proc Natl Acad Sci U S A. 2003;100(25):14858-14863.
- Wang YM, Lee VWS, Wu H, Harris DCH, Alexander SI. Heymann nephritis in Lewis rats. Curr Protoc Immunol. 2015;109:15.29.1-15.29.6.
- Akiyama S, Imai E, Maruyama S. Immunology of membranous nephropathy. F1000Res. 2019;8:F1000 Faculty Rev-734.
- Marinò M, Chiovato L, Friedlander JA, Latrofa F, Pinchera A, McCluskey RT. Serum antibodies against megalin (GP330) in patients with autoimmune thyroiditis. J Clin Endocrinol Metab. 1999;84(7):2468-2474.
- https://www.biocompare.com/Assay-Kit-Product-Search/?search=megalin (Accesed May 10, 2023).
- https://www.mybiosource.com/human-elisa-kits/megalin/3804658 (Accesed May 10, 2023).
- Caturegli P, De Remigis A, Rose NR. Hashimoto thyroiditis: clinical and diagnostic criteria. Autoimmun Rev. 2014;13(4-5):391-7. [CrossRef]
- Esfandiari NH, Papaleontiou M. Biochemical Testing in Thyroid Disorders. Endocrinol Metab Clin North Am. 2017;46(3):631-648. [CrossRef]
- Vargas-Uricoechea H, Agredo-Delgado V, Vargas-Sierra HD, Pinzón-Fernández MV. Prevalence of Functional Alterations and The Effects of Thyroid Autoimmunity on The Levels of TSH in an Urban Population of Colombia: A Population-Based Study. Endocr Metab Immune Disord Drug Targets. 2022. [CrossRef]
- Czarnocka, B. Thyroperoxidase, thyroglobulin, Na(+)/I(-) symporter, pendrin in thyroid autoimmunity. Front Biosci (Landmark Ed). 2011;16(2):783-802. [CrossRef]
- Hadj-Kacem H, Rebuffat S, Mnif-Féki M, Belguith-Maalej S, Ayadi H, Péraldi-Roux S. Autoimmune thyroid diseases: genetic susceptibility of thyroid-specific genes and thyroid autoantigens contributions. Int J Immunogenet. 2009;36(2):85-96. [CrossRef]
- Baloch Z, Carayon P, Conte-Devolx B, Demers LM, Feldt-Rasmussen U, Henry JF, LiVosli VA, Niccoli-Sire P, John R, Ruf J, Smyth PP, Spencer CA, Stockigt JR; Guidelines Committee, National Academy of Clinical Biochemistry. Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid. 2003;13(1):3-126. [CrossRef]
- Demers LM, Spencer CA. Laboratory medicine practice guidelines: laboratory support for the diagnosis and monitoring of thyroid disease. Clin Endocrinol (Oxf). 2003;58(2):138-140. [CrossRef]
- Dhillon-Smith RK, Coomarasamy A. TPO antibody positivity and adverse pregnancy outcomes. Best Pract Res Clin Endocrinol Metab. 2020;34(4):101433. [CrossRef]
- Furmaniak J, Sanders J, Sanders P, Miller-Gallacher J, Ryder MM, Rees Smith B. Practical applications of studies on the TSH receptor and TSH receptor autoantibodies. Endocrine. 2020;68(2):261-264. [CrossRef]
- Kahaly GJ, Bartalena L, Hegedüs L, Leenhardt L, Poppe K, Pearce SH. 2018 European Thyroid Association Guideline for the Management of Graves' Hyperthyroidism. Eur Thyroid J. 2018;7(4):167-186. [CrossRef]
- Parameswaran R, de Jong MC, Kit JLW, Sek K, Nam TQ, Thang TV, Khue NT, Aye TT, Tun PM, Cole T, Miller JA, Villa M, Khiewvan B, Sirinvaravong S, Sin YL, Muhammad R, Jap TS, Agrawal A, Rajput R, Fernando R, Sumanatilleke M, Suastika K, Shong YK, Lang B, Bartalena L, Yang SP; Asian Graves Consortium Study. 2021 Asia-Pacific Graves' Disease Consortium Survey of Clinical Practice Patterns in the Management of Graves' Disease. Endocrine. 2023;79(1):135-142. [CrossRef]
- Hoang TD, Stocker DJ, Chou EL, Burch HB. 2022 Update on Clinical Management of Graves Disease and Thyroid Eye Disease. Endocrinol Metab Clin North Am. 2022;51(2):287-304. [CrossRef]
- Bartalena L, Tanda ML. Current concepts regarding Graves' orbitopathy. J Intern Med. 2022;292(5):692-716. [CrossRef]
- Fatourechi, V. Pretibial myxedema: pathophysiology and treatment options. Am J Clin Dermatol. 2005;6(5):295-309. [CrossRef]
- Trohman RG, Sharma PS, McAninch EA, Bianco AC. Amiodarone and thyroid physiology, pathophysiology, diagnosis and management. Trends Cardiovasc Med. 2019;29(5):285-295. [CrossRef]
- Medić F, Bakula M, Alfirević M, Bakula M, Mucić K, Marić N. Amiodarone and thyroid dysfunction. Acta Clin Croat. 2022;61(2):327-341. [CrossRef]
- Martinez Quintero B, Yazbeck C, Sweeney LB. Thyroiditis: Evaluation and Treatment. Am Fam Physician. 2021;104(6):609-617.
- John M, Jagesh R, Unnikrishnan H, Jalaja MMN, Oommen T, Gopinath D. Utility of TSH Receptor Antibodies in the Differential Diagnosis of Hyperthyroidism in Clinical Practice. Indian J Endocrinol Metab. 2022;26(1):32-37. [CrossRef]
- Mooij CF, Cheetham TD, Verburg FA, Eckstein A, Pearce SH, Léger J, van Trotsenburg ASP. 2022 European Thyroid Association Guideline for the management of pediatric Graves' disease. Eur Thyroid J. 2022;11(1):e210073. [CrossRef]
- Ashkar C, Sztal-Mazer S, Topliss DJ. How to manage Graves' disease in women of childbearing potential. Clin Endocrinol (Oxf). 2023;98(5):643-648. [CrossRef]
- Pyrżak B, Rumińska M, Witkowska-Sędek E, Kucharska A. Follow-Up of Thyroid Function in Children With Neonatal Hyperthyroidism. Front Endocrinol (Lausanne). 2022;13:877119. [CrossRef]
- Orgiazzi, J. Anti-TSH Receptor Antibodies in Clinical Practice. Endocrinol Metab Clin North Am. 2000;29(2):339–355. [CrossRef]
- McLachlan SM, Rapoport B. Thyrotropin-Blocking Autoantibodies and Thyroid-Stimulating Autoantibodies: Potential Mechanisms Involved in the Pendulum Swinging From Hypothyroidism to Hyperthyroidism or Vice Versa. Thyroid. 2013;23(1):14–24. [CrossRef]
- Diana T, Olivo PD, Kahaly GJ. Thyrotropin Receptor Blocking Antibodies. Horm Metab Res. 2018;50(12):853–862. [CrossRef]
- Napolitano G, Bucci I, Di Dalmazi G, Giuliani C. Non-Conventional Clinical Uses of TSH Receptor Antibodies: The Case of Chronic Autoimmune Thyroiditis. Front. Endocrinol.2021; 12:769084. [CrossRef]
- Benvenga S, Bartolone L, Squadrito S, Trimarchi F. Thyroid hormone autoantibodies elicited by diagnostic fine needle biopsy. J Clin Endocrinol Metab. 1997;82(12):4217-4223. [CrossRef]
- Benvenga S, Burek CL, Talor M, Rose NR, Trimarchi F. Heterogeneity of the thyroglobulin epitopes associated with circulating thyroid hormone autoantibodies in Hashimoto's thyroiditis and non-autoimmune thyroid diseases. J Endocrinol Invest. 2002;25(11):977-982. [CrossRef]
- Sakata S, Nakamura S, Miura K. Autoantibodies against thyroid hormones or iodothyronines. Ann Intern Med. 1985;103(4):579-589.
- Ni J, Long Y, Zhang L, Yang Q, Kou C, Li S, Li J, Zhang H. High prevalence of thyroid hormone autoantibody and low rate of thyroid hormone detection interference. J Clin Lab Anal. 2022;36(1):e24124. [CrossRef]
- Ruggeri RM, Galletti M, Mandolfino MG, Aragona P, Bartolone S, Giorgianni G, Alesci D, Trimarchi F, Benvenga S. Thyroid hormone autoantibodies in primary Sjögren syndrome and rheumatoid arthritis are more prevalent than in autoimmune thyroid disease, becoming progressively more frequent in these diseases. J Endocrinol Invest. 2002;25(5):447-454.
- Vita R, Santaguida MG, Virili C, Segni M, Galletti M, Mandolfino M, Di Bari F, Centanni M, Benvenga S. Serum Thyroid Hormone Antibodies Are Frequent in Patients with Polyglandular Autoimmune Syndrome Type 3, Particularly in Those Who Require Thyroxine Treatment. Front Endocrinol (Lausanne). 2017;8:212. [CrossRef]
- Ghazal K, Brabant S, Prie D, Piketty ML. Hormone Immunoassay Interference: A 2021 Update. Ann Lab Med. 2022;42(1):3-23. [CrossRef]
- Doniach D, Roitt IM. In: Gell PGH, Coombs RA, Lachmann PJ, eds. Clinical aspects of immunology. Oxford: Blackwell Science, 1975:1355.
- Ochi, Y. Possible participation of colloid antigen 2 and abhormone (IgG with hormone activity) for the etiology of Grave’s Disease. Med Hypotheses.2019;127:23-25. [CrossRef]
| Molecular characteristics and prevalence | Antigen | |||||
| Tg | TSHR | TPO | PDN | NIS | Meg | |
| Protein | Iodinated glycoprotein | G protein-coupled receptor | Hemoprotein enzyme | Hydrophobic transmembrane glycoprotein | Membrane glycoprotein | Integral membrane protein |
| Amino acids | 2,748 | 743 | 933 | 780 | 643 | 4,655 |
| Molecular weight (kDa) | 660 | 85 | 105-110 | 86 | 70-90 | 600 |
| Thyroid concentration | ++++/++++ | ++/++++ | +++/++++ | +/++++ | +/++++ | +/++++ |
| Epitope localization | Predominantly central region and C-terminus | Predominantly A-subunit | Predominantly myeloperoxidase-like domain and to lesser extent complement control protein-like domain | Apical membrane of thyrocytes | Predominantly extramembranous Regions | Apical surface of thyrocytes |
| Immunogenicity | +++++/+++++ | ++++/+++++ | +++++/+++++ | +++/+++++ | ++/+++++ | +/+++++ |
| Chromosomal location | 8q24 | 14q31 | 2p25 | 7q22-31 | 19p12-13.2 | 2q24-q31 |
| Prevalence of antibody in the general population (%) | TgAbs (5-20) | TRAbs (0-3) | TPOAbs (8-30) | Unknow | Unknow | Unknow |
| Prevalence of antibody in autoimmune thyroiditis ―HT― (%) | TgAbs (80-90) | TRAbs (10-20) | TPOAbs (90-100) | PDNAbs (variable, 9-98%) | NISAbs (7-8) | MegAbs (~50) |
| Prevalence of antibody in GBD (%) | TgAbs (30-60) | TRAbs (90-95) | TPOAbs (80) | PDNAbs (variable, 10-75%) | NISAbs (10-12) | MegAbs (~50) |
| Principle [kit] | CLIA [Architect (Abbott Diagnostics, USA)] | ECLIA [ECLusys (Roche Diagnostics, Germany)] | EIA [AIA-Pack (Tosoh Bioscience, Japan)] | CLEIA [Lumipulse G (Fujirebio Inc, Japan)] | CLEIA [Immulite 2000 (Siemens Healthcare Diagnostics, USA)] |
| Procedure | Two step sandwich | One step competitive | Two step sandwich | Two step sandwich | Two step sandwich |
| TgAbs | |||||
| Assay components | Human Tg coated Microparticles. Acridinium labeled anti-human IgG (mouse monoclonal) | Biotinylated human Tg. Ruthenylated anti-TgAb (mouse monoclonal) | Human Tg coated Microparticles. ALP labeled antihuman IgG (mouse monoclonal | Human Tg coated Microparticles. ALP labeled antihuman IgG (mouse monoclonal). 3-(2’-spiroadamantane)-4-methoxy-4-(3’’-phosphoryloxy)phenyl-1,2-dioxetane disodium salt | Human Tg coated Microparticles. ALP labeled antihuman IgG (mouse monoclonal). 3-(2’-spiroadamantane)-4-methoxy-4-(3’’-phosphoryloxy)phenyl-1,2-dioxetane disodium salt |
| Cut off (IU/mL) | 4.11 | 28 | 13.6 | 12.2 | 40 |
| TPOAbs | |||||
| Assay components | Human TPO coated microparticles. Acridinium labeled anti-human IgG (mouse monoclonal) | Biotinylated human TPO. Ruthenylated anti-TgAb (goat polyclonal) | Human TPO coated Microparticles. ALP labeled antihuman IgG (mouse monoclonal | Human TPO coated Microparticles ALP labeled antihuman IgG (mouse monoclonal). 3-(2’-spiroadamantane)-4-methoxy-4-(3’’-phosphoryloxy)phenyl-1,2-dioxetane disodium salt | Human TPO coated Microparticles ALP labeled antihuman IgG (mouse monoclonal). 3-(2’-spiroadamantane)-4-methoxy-4-(3’’-phosphoryloxy)phenyl-1,2-dioxetane disodium salt |
| Cut off (IU/mL) | 5.6 | 16 | 3.2 | 5.1 | 35 |
| Prevalence of TgAbs and TPOAbs | ||||||||||
| Principle [kit] | CLIA [Architect (Abbott Diagnostics, USA)] | ECLIA [ECLusys (Roche Diagnostics, Germany)] | EIA [AIA-Pack (Tosoh Bioscience, Japan)] | CLEIA [Lumipulse G (Fujirebio Inc, Japan)] | CLEIA [Immulite 2000 (Siemens Healthcare Diagnostics, USA)] | |||||
| Abs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs |
| Hashimoto’s thyroiditis | ++++* | ++ | +++* | ++ | +++* | ++ | ++++* | ++ | ++ | ++ |
| GBD | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ |
| Painless thyroiditis | ++* | + | ++* | + | ++* | + | ++* | + | + | + |
| Healthy controls | +** | +/- | + | +/- | + | +/- | +** | +/- | +/- | +/- |
| Prevalence of TgAbs (alone) or TPOAbs (alone) | ||||||||||
| Abs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs | TgAbs | TPOAbs |
| Hashimoto’s thyroiditis | +* | - | +* | +/- | +* | +/- | +* | - | + | ++ |
| GBD | + | + | +/- | + | +/- | + | + | + | +/- | ++** |
| Painless thyroiditis | ++* | + | ++* | +/- | ++* | +/- | ++* | - | + | +/- |
| Healthy controls | +** | +/- | +/- | +/- | + | +/- | +** | +/- | +/- | +/- |
| TRAbs | ||||
| Immunoassays | ELISA | |||
| EliA anti-TSHR (Thermo Fisher Scientific, Germany) | Elecsys (COBAS, Roche, USA) | BRAHMS TRAK human KRYPTOR (Thermo Fisher Scientific, Germany) | IMMULITE 2000 TSI (Siemens, Healthineers, Germany) | ELISA RSR TRAb Fast (RSR Limited, UK). |
| Sensitivity: 96.6% | Sensitivity: 100% | Sensitivity: >98% | Sensitivity: 98.3% | Sensitivity: 85% |
| Specificity: 99.4% | Specificity: 95.3% | Specificity: almost 100% | Specificity: 97% | Specificity: 100% |
| The lower and upper limit of detection are 1.5 and 80 IU/L, respectively. Intra-assay and inter-assay variance at 3.2 U/L (positive cut-off >3.3 IU/L, negative cut-off <2.9 IU/L) is 10.6% and 11.4%, respectively. | The range is 0.8 – 40 IU/L. The limit of quantification is the lowest analyte concentration that can be reproducibly measured with an intermediate precision CV of ≤ 20 % | The range is 0.27 – 20 IU/L (cut-off 1.8 U/L). Intra-assay variance for the range of 1.2 to 2.0 U/L is < 7.0%. Inter-assay variance for the range of 1.0 to 2.0 U/L is < 18% | The range is 0.10 – 40 IU/L (cut-off 0.55 IU/L). Intra-assay and inter-assay variance at 0.69 IU/L is 4.1% and 5.1%, respectively | The range is 1 – 40 IU/L (positive cut-off ≥1.0 IU/L, lower detection limit at 2 SD 0.16 IU/L). Intra-assay and inter-assay variance at 2.0 and 4.6 IU/L is reported 7.2% and 3.3%, respectively |
| Principle/kit [ref] | Bioss Inc.'s NIS Polyclonal Antibody; USA [87]. | Polyclonal Antibody to NIS, MyBioSource.com; USA [88]. | Anti-NIS Polyclonal Antibody, American Research Products Inc., USA [89]. | SLC5A5 / NIS Monoclonal Antibody, LifeSpan BioSciences; USA [90]. | NIS antibody, Biorbyt; UK [91]. | Anti-NIS antibody, GeneTex; USA [92]. |
| Clonality | Rabbit Polyclonal antibody | Rabbit Polyclonal antibody | Polyclonal antibody | Mouse monoclonal antibody | Human Polyclonal antibody | Rabbit Polyclonal antibody |
| Isotype | IgG | IgG | IgG | IgG1 | IgG | IgG |
| Immunogen | KLH conjugated peptide, mouse NIS | Rabbit polyclonal antibody raised against NIS | Synthetic peptide from the C-terminus of rat NIS | Synthetic peptide corresponding to aa37-54 of human NIS. | KLH conjugated synthetic peptide derived from mouse NIS | KLH conjugated synthetic peptide derived between 535-608 amino acids of human NIS |
| Purity | Protein A purified | Affinity Chromatography | Immunogen affinity purified | Affinity purified | Affinity purified by Protein A | Protein A purified |
| Reactivity | Human, Rat, Pig | Human | Human, Porcine, Rat | Human | Human | Human, rat |
| Applications | WB, ELISA, IHC-P, IHC-F, ICC, IF | WB, ICC, IHC-P, EIA | IHC | IHC, IHC-P, WB | WB | WB, IHC-P, IHC-Fr, IHC |
| Principle/kit [ref] | Polyclonal Rabbit anti-Human SLC26A4 / Pendrin Antibody, LifeSpan BioSciences; USA [93]. | SLC26A4 antibody, MyBioSource.com; USA [94]. | SLC26A4 / Pendrin Monoclonal Antibody, LifeSpan BioSciences; USA [95]. | Pendrin Antibody / SLC26A4, NSJ Bioreagents; USA [96]. | Immunotag™ S26A4 Polyclonal Antibody, G Biosciences; USA [97]. |
| Clonality | IgG Polyclonal | Rabbit Polyclonal | Monoclonal | Polyclonal | Polyclonal |
| Isotype | IgG, epitope: aa287-336 | IgG | IgG2a kappa | IgG | Primary antibody |
| Immunogen | Synthetic peptide located between aa287-336 of human SLC26A4 (O43511, NP_000432). | Amino acids ELNDRFRHKIPVPIPIEVIVTIIATAISYGANLEKNYNAGIVKSIPRGFL | SLC26A4 (NP_000432, aa 674-754). A partial recombinant protein with GST tag. MW of the GST tag alone is 26 KDa. | Amino acids RSLRVIVKEFQRIDVNVYFASLQDYVIEKLEQ | Synthesized peptide derived from part region of human protein |
| Purity | Immunoaffinity purified | Affinity purified | Purified from ascites by Protein A | Antigen affinity purified | Affinity purified |
| Reactivity | Human | Human, mouse | Human | Human | Human |
| Applications | IHC, IHC-P, WB | WB | ELISA | WB | WB, ELISA |
| Antibody | Participants without AITD (controls, %) | GBD (%) | HT (%) | Ratio of prevalence of positivity between subjects with AITD and participants without AITD |
| Studies with NISAbs | 0 | 84 | 15 | 4.6 |
| 0–10 | 0–63 | 0–25.9 | 2.7 | |
| 0.6–3 | 5.6–10.7 | 6.9–20.8 | 1.5 | |
| 0 | 22 | 24 | 1.95 | |
| – | 38 | 27.5 | 1.75 | |
| 0 | 20 | 14 | 0.4 | |
| 1.8 | 12.3 | 7.5 | 0.8 | |
| Studies with PDNAbs | 0.0 | 74 | 97.5 | 1.75 |
| 0.0 | 9.9 | 7.6 | 4.89 | |
| 0 | 13 | 8 | 0.4 | |
| 5.0 | 11.0 | 4.7 | 0.8 |
| Principle/kit (ref) | LRP2 / Megalin Antibody, LifeSpan BioSciences; USA. | Anti-Lrp2 / Megalin Rabbit Monoclonal Antibody, BosterBio; USA. | Rabbit Anti-Megalin/LRP2 Antibody, MyBioSource.com; USA. | HUMAN ANTI HUMAN MEGALIN, Bio-Rad; USA. | Mouse Anti-Human LRP2/Megalin Clone CD7D5 mAb from Cell Sciences; USA. |
| Clonality | Polyclonal | Monoclonal | Polyclonal | Monoclonal | Monoclonal |
| Isotype | IgG, epitope: aa287-336 | Rabbit IgG | IgG | Fab fragment | IgG1 |
| Immunogen | Amino acids 4446-4655 of human LRP2 (NP_004516.2). FHYRRTGSLLPALPKLPSLSSLVKPSENGNGVTFRSGADLNMDIGVSGFGPETAIDRSMAMSEDFVMEMGKQPIIFENPMYSARDSAVKVVQPIQVTVSENVDNKNYGSPINPSEIVPETNPTSPAADGTQVTKWNLFKRKSKQTTNFENPIYAQMENEQKESVAATPPPSPSLPAKPKPPSRRDPTPTYSATEDTFKDTANLVKEDSEV | A synthesized peptide derived from human Lrp2 / Megalin | A synthetic peptide corresponding to the center region of the mouse LRP2/Megalin | Human Megalin (aa sequence 1024-1224) – N1 fusion protein | Purified Human Megalin |
| Purity | Affinity purified | Affinity-chromatography | Protein A and antigen affinity | Affinity purified | Protein G Chromatography |
| Reactivity | Mouse, Rat, Human | Human, Mouse, Rat | Mouse | Human | Human |
| Applications | IHC, IHC-P, WB | WB | IHC-P | WB | IF, IHC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




