Submitted:
12 June 2023
Posted:
19 June 2023
You are already at the latest version
Abstract
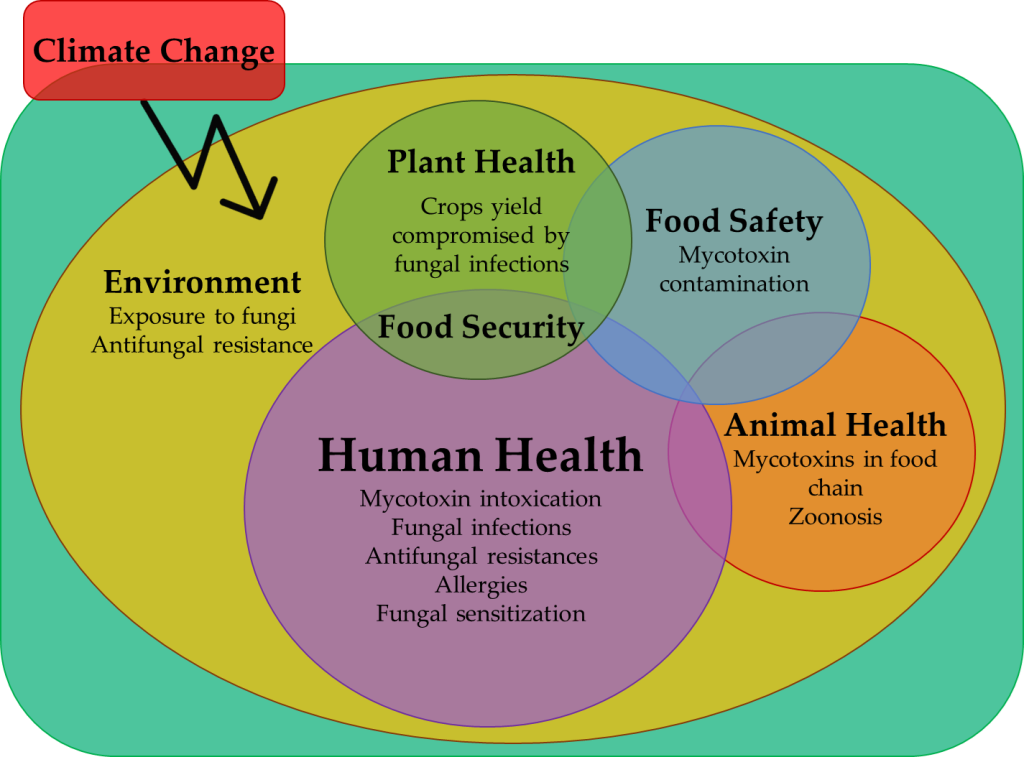
Keywords:
1. Introduction
2. Fungal effects on Plant Health
3. Mycotoxin effects on Animal and Human Health
4. Fungal effects in Human Health
5. Fungal effects in Animal Health
6. Climate Change influence on Fungi
7. Concluding Remarks and Future Perspective
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Blackwell, M. The fungi: 1, 2, 3... 5.1 million species? Am. J. Bot. 2011, 98, 426–438. [Google Scholar] [CrossRef]
- Hawksworth, D.L.; Lücking, R. Fungal diversity revisited: 2.2 to 3.8 million species. The Fungal Kingdom 2017, 79–95. [Google Scholar] [CrossRef]
- Grossart, H.P.; Van den Wyngaert, S.; Kagami, M.; Wurzbacher, C.; Cunliffe, M.; Rojas-Jimenez, K. Fungi in aquatic ecosystems. Nat. Rev. Microbiol. 2019, 17, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Naranjo-Ortiz, M.A.; Gabaldón, T. Fungal evolution: major ecological adaptations and evolutionary transitions. Biol. Rev. 2019, 94, 1443–1476. [Google Scholar] [CrossRef] [PubMed]
- Janowski, D.; Leski, T. Factors in the Distribution of Mycorrhizal and Soil Fungi. Diversity 2022, 14, 1–15. [Google Scholar] [CrossRef]
- Daley, D.K.; Brown, K.J.; Badal, S. Chapter 20 - Fungal Metabolites. In Pharmacognosy: Fundamentals, Applications and Strategy; Badal, S., Delgoda, R., Eds.; Academic Press, 2017; pp. 413–421 ISBN 9780128020999.
- Esheli, M.; Thissera, B.; El-Seedi, H.R.; Rateb, M.E. Fungal Metabolites in Human Health and Diseases—An Overview. Encyclopedia 2022, 2, 1590–1601. [Google Scholar] [CrossRef]
- Elmholt, S. Mycotoxins in the Soil Environment. 2008, 167–203. [CrossRef]
- Afsah-Hejri, L.; Jinap, S.; Hajeb, P.; Radu, S.; Shakibazadeh, S. A review on mycotoxins in food and feed: Malaysia case study. Compr. Rev. Food Sci. Food Saf. 2013, 12, 629–651. [Google Scholar] [CrossRef]
- Seyedmousavi, S.; Bosco, S.D.M.G.; Hoog, S. De; Ebel, F.; Elad, D.; Gomes, R.R.; Jacobsen, I.D.; Jensen, H.E.; Martel, A.; Mignon, B.; et al. Fungal infections in animals: a patchwork of different situations. Med. Mycol. 2018, 56, 165–187. [Google Scholar] [CrossRef]
- Marín, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Reduction of mycotoxins and toxigenic fungi in the mediterranean basin maize chain. Phytopathol. Mediterr. 2012, 51, 93–118. [Google Scholar] [CrossRef]
- Vaali, K.; Tuomela, M.; Mannerström, M.; Heinonen, T.; Tuuminen, T. Toxic Indoor Air Is a Potential Risk of Causing Immuno Suppression and Morbidity—A Pilot Study. J. Fungi 2022, 8. [Google Scholar] [CrossRef]
- Kurup, V.P. Fungal allergens. Curr. Allergy Asthma Rep. 2003, 3, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Bongomin, F.; Gago, S.; Oladele, R.; Denning, D. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Li, S.J.; Yan, J.; Tang, Y.; Cheng, J.P.; Gao, A.J.; Yao, X.; Ruan, J.J.; Xu, B.L. Research Progress on Phytopathogenic Fungi and Their Role as Biocontrol Agents. Front. Microbiol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Simões, D.; Carbas, B.; Soares, A.; Freitas, A.; Silva, A.S.; Brites, C.; Andrade, E. de Assessment of Agricultural Practices for Controlling Fusarium and Mycotoxins Contamination on Maize Grains: Exploratory Study in Maize Farms. Toxins (Basel). 2023, 15, 1–16. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization One Health Available online:. Available online: https://www.who.int/health-topics/one-health#tab=tab_1 (accessed on Mar 8, 2023).
- Carbas, B.; Simões, D.; Soares, A.; Freitas, A.; Ferreira, B.; Carvalho, A.R.F.; Silva, A.S.; Pinto, T.; Diogo, E.; Andrade, E.; et al. Occurrence of fusarium spp. In maize grain harvested in portugal and accumulation of related mycotoxins during storage. Foods 2021, 10, 1–14. [Google Scholar] [CrossRef]
- Fones, H.N.; Bebber, D.P.; Chaloner, T.M.; Kay, W.T.; Steinberg, G.; Gurr, S.J. Threats to global food security from emerging fungal and oomycete crop pathogens. Nat. Food 2020, 1, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Kingsley, K.L.; Bergen, M.S.; Kowalski, K.P.; White, J.F. Fungal disease prevention in seedlings of rice (Oryza sativa) and other grasses by growth-promoting seed-associated endophytic bacteria from invasive phragmites australis. Microorganisms 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Chaiharn, M.; Chunhaleuchanon, S.; Lumyong, S. Screening siderophore producing bacteria as potential biological control agent for fungal rice pathogens in Thailand. World J. Microbiol. Biotechnol. 2009, 25, 1919–1928. [Google Scholar] [CrossRef]
- Mahdavi, F.; Sariah, M.; Maziah, M. Expression of rice thaumatin-like protein gene in transgenic banana plants enhances resistance to Fusarium wilt. Appl. Biochem. Biotechnol. 2012, 166, 1008–1019. [Google Scholar] [CrossRef]
- Ajilogba, C.F.; Babalola, O.O. Integrated management strategies for tomato fusarium wilt. Biocontrol Sci. 2013, 18, 117–127. [Google Scholar] [CrossRef]
- Everts, K.L.; Egel, D.S.; Langston, D.; Zhou, X.G. Chemical management of Fusarium wilt of watermelon. Crop Prot. 2014, 66, 114–119. [Google Scholar] [CrossRef]
- Logrieco, A.; Bottalico, A.; Mulé, G.; Moretti, A.; Perrone, G. Epidemiology of toxigenic fungi and their associated mycotoxins for some Mediterranean crops. Eur. J. Plant Pathol. 2003, 109, 645–667. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, J.; Ma, L.; Li, E.; Ji, B.; Sun, C.; Zhou, J.-J.; Xu, B. First Report of a New Disease Caused by Fusarium tricinctum on Apple Tree in China. Plant Dis. 2022, 106, 1524. [Google Scholar] [CrossRef] [PubMed]
- Aguín, O.; Cao, A.; Pintos, C.; Santiago, R.; Mansilla, P.; Butrón, A. Occurrence of Fusarium species in maize kernels grown in northwestern Spain. Plant Pathol. 2014, 63, 946–951. [Google Scholar] [CrossRef]
- Simões, D.; Diogo, E.; de Andrade, E. First Report of Fusarium andiyazi Presence in Portuguese Maize Kernels. Agriculture 2022, 336. [Google Scholar] [CrossRef]
- FAO Agricultural production statistics 2000–2020; 2022.
- Carvajal-Moreno, M. Mycotoxin challenges in maize production and possible control methods in the 21st century. J. Cereal Sci. 2022, 103, 103293. [Google Scholar] [CrossRef]
- Lino, C.M.; Silva, L.J.G.; Pena, A.; Fernández, M.; Mañes, J. Occurrence of fumonisins B1 and B2 in broa, typical Portuguese maize bread. Int. J. Food Microbiol. 2007, 118, 79–82. [Google Scholar] [CrossRef]
- Zentai, A.; Szeitzné-Szabó, M.; Mihucz, G.; Szeli, N.; Szabó, A.; Kovács, M. Occurrence and risk assessment of fumonisin b1 and b2 mycotoxins in maize-based food products in Hungary. Toxins (Basel). 2019, 11, 1–14. [Google Scholar] [CrossRef]
- Nnadi, N.E.; Carter, D.A. Climate change and the emergence of fungal pathogens. PLoS Pathog. 2021, 17, 1–6. [Google Scholar] [CrossRef]
- Thambugala, K.M.; Daranagama, D.A.; Phillips, A.J.L. Fungi vs. Fungi in Biocontrol : An Overview of Fungal Antagonists Applied Against Fungal Plant Pathogens. Front. Cell. Infect. Microbiol. 2020, 10, 1–19. [Google Scholar] [CrossRef]
- Berger, S.; Chazli, Y.; Babu, A.; Coste, A. Azole resistance in Aspergillus fumigatus: A consequence of antifungal use in agriculture? Front. Microbiol. 2017, 8, 1024. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, M.; Faria-Ramos, I.; Cruz, L.; Pina-Vaz, C.; Gonçalves Rodrigues, A. Genesis of Azole Antifungal Resistance from Agriculture to Clinical Settings. J. Agric. Food Chem. 2015, 63, 7463–7468. [Google Scholar] [CrossRef] [PubMed]
- Dudakova, A.; Spiess, B.; Tangwattanachuleeporn, M.; Sasse, C.; Buchheidt, D.; Weig, M.; Groß, U.; Bader, O. Molecular tools for the detection and deduction of azole antifungal drug resistance phenotypes in Aspergillus species. Clin. Microbiol. Rev. 2017, 30, 1065–1091. [Google Scholar] [CrossRef]
- Snelders, E.; Camps, S.; Karawajczyk, A.; Schaftenaar, G.; Kema, G.; van der Lee, H.; Klaassen, C.; Melchers, W.; Verweij, P. Triazole fungicides can induce cross-resistance to medical triazoles in Aspergillus fumigatus. PLoS One 2012, 7, e31801. [Google Scholar] [CrossRef] [PubMed]
- Melo, A.M.; Stevens, D.A.; Tell, L.A.; Veríssimo, C.; Sabino, R.; Xavier, M.O. Aspergillosis, avian species and the one health perspective: The possible importance of birds in azole resistance. Microorganisms 2020, 8, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.F.; van Zyl, W.H.; Gelderblom, W.C.A. Biologically based methods for control of fumonisin-producing Fusarium species and reduction of the fumonisins. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Khokhar, I.; Haider, M.S.; Mukhtar, I.; Mushtaq, S. Biological control of Aspergillus niger, the cause of Black-rot disease of Allium cepa L. (onion), by Penicillium species. J. Agrobiol. 2013, 29, 23–28. [Google Scholar] [CrossRef]
- Chen, A.; Mao, X.; Sun, Q.; Wei, Z.; Li, J.; You, Y.; Zhao, J.; Jiang, G.; Wu, Y.; Wang, L.; et al. Alternaria Mycotoxins: An Overview of Toxicity, Metabolism, and Analysis in Food. J. Agric. Food Chem 2021, 69, 7817–7830. [Google Scholar] [CrossRef]
- Awuchi, C.G.; Ondari, E.N.; Nwozo, S.; Odongo, G.A.; Eseoghene, I.J.; Twinomuhwezi, H.; Ogbonna, C.U.; Upadhyay, A.K.; Adeleye, A.O.; Okpala, C.O.R. Mycotoxins’ Toxicological Mechanisms Involving Humans, Livestock and Their Associated Health Concerns: A Review. Toxins (Basel). 2022, 14, 1–33. [Google Scholar] [CrossRef]
- Magkos, F. Meat in the human diet: In transition from evolutionary hallmark to scapegoat. Am. J. Clin. Nutr. 2022, 115, 1263–1265. [Google Scholar] [CrossRef]
- BIOMIN DSM World Mycotoxin Survey; 2022.
- Binder, E.M.; Tan, L.M.; Chin, L.J.; Handl, J.; Richard, J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim. Feed Sci. Technol. 2007, 137, 265–282. [Google Scholar] [CrossRef]
- Lye, M.S.; Ghazali, A.A.; Mohan, J.; Alwin, N.; Nair, R.C. An outbreak of acute hepatic encephalopathy due to severe aflatoxicosis in Malaysia. Am. J. Trop. Med. Hyg. 1995, 53, 68–72. [Google Scholar] [PubMed]
- Viegas, S.; Veiga, L.; Malta-Vacas, J.; Sabino, R.; Figueredo, P.; Almeida, A.; Viegas, C.; Carolino, E. Occupational exposure to aflatoxin (AFB1) in poultry production. J. Toxicol. Environ. Heal. - Part A Curr. Issues 2012, 75, 1330–1340. [Google Scholar] [CrossRef] [PubMed]
- Viegas, S.; Veiga, L.; Figueiredo, P.; Almeida, A.; Carolino, E.; Sabino, R.; Verissimo, C.; Viegas, C. Occupational exposure to aflatoxin B1: The case of poultry and swine production. World Mycotoxin J. 2013, 6. [Google Scholar] [CrossRef]
- Viegas, C.; Carolino, E.; Sabino, R.; Viegas, S.; Veríssimo, C. Fungal contamination in swine: a potential occupational health threat. J. Toxicol. Environ. Health. A 2013, 76, 272–280. [Google Scholar] [CrossRef]
- Viegas, C.; Carolino, E.; Malta-Vacas, J.; Sabino, R.; Viegas, S.; Veríssimo, C. Fungal contamination of poultry litter: a public health problem. J. Toxicol. Environ. Health. A 2012, 75, 1341–1350. [Google Scholar] [CrossRef]
- Malta-Vacas, J.; Sabino, R.; Viegas, S.; Viegas, C. Occupational exposure to toxigenic fungi from Aspergillus flavus complex. Toxicol. Lett. 2012, 211, S208. [Google Scholar] [CrossRef]
- Sabino, R.; Faísca, V.; Carolino, E.; Veríssimo, C.; Viegas, C. Occupational exposure to aspergillus by swine and poultry farm workers in portugal. J. Toxicol. Environ. Heal. - Part A Curr. Issues 2012, 75, 1381–1391. [Google Scholar] [CrossRef]
- Perrone, G.; Susca, A. Penicillium Species and Their Associated Mycotoxins. In Mycotoxigenic Fungi. Methods in Molecular Biology; Moretti, A., Susca, A., Eds.; Humana Press: New York, NY, 2017; pp. 107–109. ISBN 978-1-4939-6705-6. [Google Scholar]
- Fumagalli, F.; Ottoboni, M.; Pinotti, L.; Cheli, F. Integrated mycotoxin management system in the feed supply chain: Innovative approaches. Toxins (Basel). 2021, 13. [Google Scholar] [CrossRef]
- Matumba, L.; Namaumbo, S.; Ngoma, T.; Meleke, N.; De Boevre, M.; Logrieco, A.F.; De Saeger, S. Five keys to prevention and control of mycotoxins in grains: A proposal. Glob. Food Sec. 2021, 30, 100562. [Google Scholar] [CrossRef]
- Assefa, T.; Geremew, T. Major mycotoxins occurrence, prevention and control approaches. Biotechnol. Mol. Biol. Rev. 2018, 12, 1–11. [Google Scholar] [CrossRef]
- Gnat, S.; Łagowski, D.; Nowakiewicz, A.; Dyląg, M. A global view on fungal infections in humans and animals: opportunistic infections and microsporidioses. J. Appl. Microbiol. 2021, 131, 2095–2113. [Google Scholar] [CrossRef] [PubMed]
- Scott Weese, J.; Fulford, M.B. Fungal Diseases. In Companion Animal Zoonoses; 2010; pp. 275–298 ISBN 9780813819648.
- Veríssimo, C.; Toscano, C.; Ferreira, T.; Abreu, G.; Simões, H.; Diogo, J.; Carvalho, D.; Santiago, F.; Lima, A.; Queirós, A.M.; et al. Invasive and Subcutaneous Infections Caused by Filamentous Fungi: Report from a Portuguese Multicentric Surveillance Program. Microorganisms 2022, 10. [Google Scholar] [CrossRef]
- Tavares-Bello, E.; Sabino, R.; Simões, H.; Veríssimo, C.; Ussene, E. Tinea Corporis Caused by Trichophyton Benhamiae Transmitted by a Dog: First Case Report in Portugal. Gaz. Médica 2021, 8, 29–33. [Google Scholar] [CrossRef]
- World Health Organization WHO fungal priority pathogens list to guide research, development and public health action; 2022; ISBN 9789240060241.
- Sabino, R.; Veríssimo, C.; Pereira, Á.A.; Antunes, F. Candida auris, an agent of hospital-associated outbreaks: Which challenging issues do we need to have in mind? Microorganisms 2020, 8. [Google Scholar] [CrossRef]
- Kullberg, B.J.; Arendrup, M.C. Invasive Candidiasis. N. Engl. J. Med. 2015, 373, 1445–1456. [Google Scholar] [CrossRef]
- Alcoceba, E.; Gómez, A.; Lara-Esbrí, P.; Oliver, A.; Beltrán, A.F.; Ayestarán, I.; Muñoz, P.; Escribano, P.; Guinea, J. Fluconazole-resistant Candida parapsilosis clonally related genotypes: first report proving the presence of endemic isolates harbouring the Y132F ERG11 gene substitution in Spain. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2022, 28, 1113–1119. [Google Scholar] [CrossRef]
- Sabino, R. Aspergillus and Health. Microorganisms 2022, 10, 4–7. [Google Scholar] [CrossRef]
- Sabino, R.; Veríssimo, C. Novel Clinical and Laboratorial Challenges in Aspergillosis. Microorganisms 2022, 10, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, T.; Yamasaki, A.; Funaki, Y.; Harada, T.; Okazaki, R.; Hasegawa, Y.; Sueda, Y.; Fukushima, T.; Morita, M.; Yamamoto, A.; et al. An onion farmer with a case of hypersensitivity pneumonitis caused by Aspergillus niger. Respir. Med. Case Reports 2018, 23, 60–62. [Google Scholar] [CrossRef]
- Van Der Linden, J.W.M.; Warris, A.; Verweij, P.E. Aspergillus species intrinsically resistant to antifungal agents. Med. Mycol. 2011, 49, S82–S89. [Google Scholar] [CrossRef] [PubMed]
- Thompson III, G.R. HHS Public Access endemic mycoses : an initiative of the European Confederation of for Human and Animal Mycology. Lancet Infect Dis 2021, 21, e364–e374. [Google Scholar] [CrossRef] [PubMed]
- Gnat, S.; Nowakiewicz, A.; Dyla, M. A global view on fungal infections in humans and animals : infections caused by dimorphic fungi and dermatophytoses. J. Appl. Microbiol. 2021, 131, 2688–2704. [Google Scholar] [CrossRef] [PubMed]
- Al-Hatmi, A.M.S.; Meis, J.F.; de Hoog, G.S. Fusarium: Molecular Diversity and Intrinsic Drug Resistance. PLoS Pathog. 2016, 12, 1–8. [Google Scholar] [CrossRef]
- Seed, P.C. The human mycobiome. Cold Spring Harb. Perspect. Med. 2015, 5. [Google Scholar] [CrossRef]
- Belvoncikova, P.; Splichalova, P.; Videnska, P.; Gardlik, R. The Human Mycobiome: Colonization, Composition and the Role in Health and Disease. J. Fungi 2022, 8. [Google Scholar] [CrossRef]
- Tell, L.A. Aspergillosis in mammals and birds: Impact on veterinary medicine. Med. Mycol. 2005, 43, 71–73. [Google Scholar] [CrossRef]
- Guillot, J.; Guérin, C.; Chermette, R. Histoplasmosis in Animals. In Emerging and Epizootic Fungal Infections in Animals; 2018; pp. 115–128 ISBN 9783319720937.
- Brömel, C.; Sykes, J.E. Epidemiology, diagnosis, and treatment of blastomycosis in dogs and cats. Clin. Tech. Small Anim. Pract. 2005, 20, 233–239. [Google Scholar] [CrossRef]
- Velegraki, A.; Cafarchia, C.; Gaitanis, G.; Iatta, R.; Boekhout, T. Malassezia Infections in Humans and Animals: Pathophysiology, Detection, and Treatment. PLoS Pathog. 2015, 11, 1–6. [Google Scholar] [CrossRef]
- Byrnes, E.J.; Bartlett, K.H.; Perfect, J.R.; Heitman, J. Cryptococcus gattii: An emerging fungal pathogen infecting humans and animals. Microbes Infect. 2011, 13, 895–907. [Google Scholar] [CrossRef]
- Chomel, B.B. Emerging and Re-emerging zoonoses of dogs and cats. Animals 2014, 4, 434–445. [Google Scholar] [CrossRef]
- Carnero, L.C.G.; Pérez, N.E.L.; Hernández, S.E.G.; Álvarez, J.A.M. Immunity and treatment of sporotrichosis. J. Fungi 2018, 4. [Google Scholar] [CrossRef]
- Bossche, H. Vanden Mechanisms of antifungal resistance. Rev. Iberoam. Micol. 1997, 14, 44–49. [Google Scholar]
- Snelders, E.; Van Der Lee, H.; Kuijpers, J.; Rijs, A.; Varga, J.; Samson, R.; Mellado, E.; Donders, A.; Melchers, W.; Verweij, P. Emergence of azole resistance in Aspergillus fumigatus and spread of a single resistance mechanism. PLoS Med. 2008, 5, e219. [Google Scholar] [CrossRef] [PubMed]
- Corredor-Moreno, P.; Saunders, D.G.O. Expecting the unexpected: factors influencing the emergence of fungal and oomycete plant pathogens. New Phytol. 2019, 225, 118–125. [Google Scholar] [CrossRef]
- Mcclure, M.L.; Hranac, C.R.; Haase, C.G.; Mcginnis, S.; Dickson, B.G.; Hayman, D.T.S.; Mcguire, L.P.; Lausen, C.L.; Plowright, R.K.; Fuller, N.; et al. Projecting the compound effects of climate change and white-nose syndrome on North American bat species. Clim. Chang. Ecol. 2022, 3. [Google Scholar] [CrossRef]
- Rollins-Smith, L.A. Global Amphibian Declines, Disease, and the Ongoing Battle between Batrachochytrium Fungi and the Immune System. Herpetologica 2020, 76, 178–188. [Google Scholar] [CrossRef]
- Neblett Fanfair, R.; Benedict, K.; Bos, J.; Bennett, S.D.; Lo, Y.-C.; Adebanjo, T.; Etienne, K.; Deak, E.; Derado, G.; Shieh, W.-J.; et al. Necrotizing Cutaneous Mucormycosis after a Tornado in Joplin, Missouri, in 2011. N. Engl. J. Med. 2012, 367, 2214–2225. [Google Scholar] [CrossRef] [PubMed]
- Garcia-solache, M.A.; Casadevall, A. Hypothesis: global warming will bring new fungal diseases for mammals. MBio 2010, 1, e00061–10. [Google Scholar] [CrossRef]
- Akinbobola, A.B.; Kean, R.; Hanifi, S.A.M.; Quilliam, R.S. Environmental reservoirs of the drug- resistant pathogenic yeast Candida auris. PLoS Pathog. 2023, 19, 1–14. [Google Scholar] [CrossRef]
- Alan Pounds, J.; Bustamante, M.R.; Coloma, L.A.; Consuegra, J.A.; Fogden, M.P.L.; Foster, P.N.; La Marca, E.; Masters, K.L.; Merino-Viteri, A.; Puschendorf, R.; et al. Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 2006, 439, 161–167. [Google Scholar] [CrossRef]
- Pegg, K.G.; Coates, L.M.; Neill, W.T.O.; Turner, D.W.; Dita, M.; Carpentier, S.C. The Epidemiology of Fusarium Wilt of Banana. 2019, 10, 1–19. [CrossRef]
- Zingales, V.; Taroncher, M.; Martino, P.A.; Ruiz, M.-J.; Caloni, F. Climate Change and Effects on Molds and Mycotoxins. Toxins (Basel). 2022, 14. [Google Scholar] [CrossRef]
- Leggieri, M.C.; Toscano, P.; Battilani, P. Predicted Aflatoxin B1 Increase in Europe Due to Climate Change: Actions and Reactions at Global Level. Toxins (Basel). 2021, 13. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



