Submitted:
15 June 2023
Posted:
16 June 2023
Read the latest preprint version here
Abstract
Keywords:
Introduction:
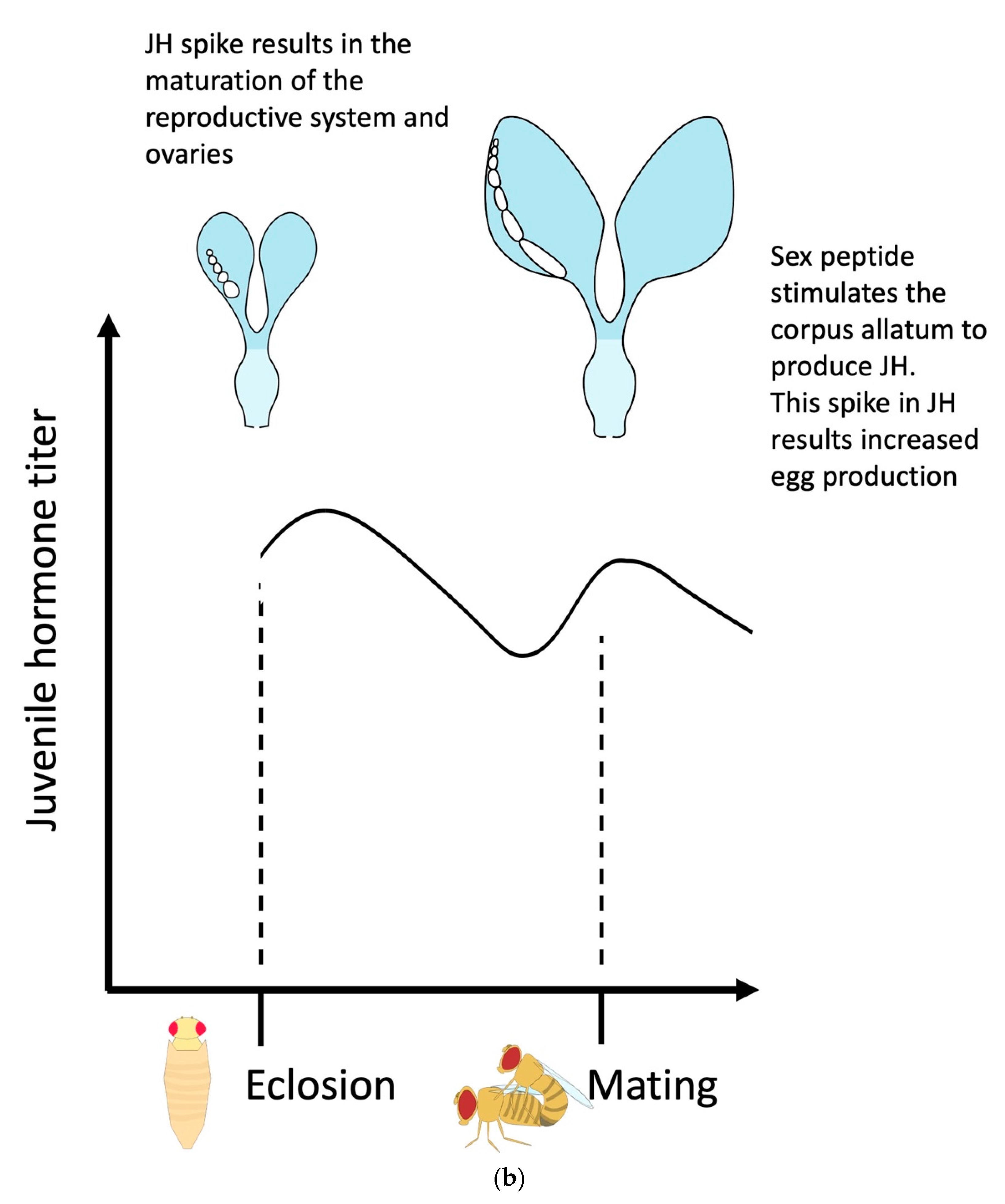
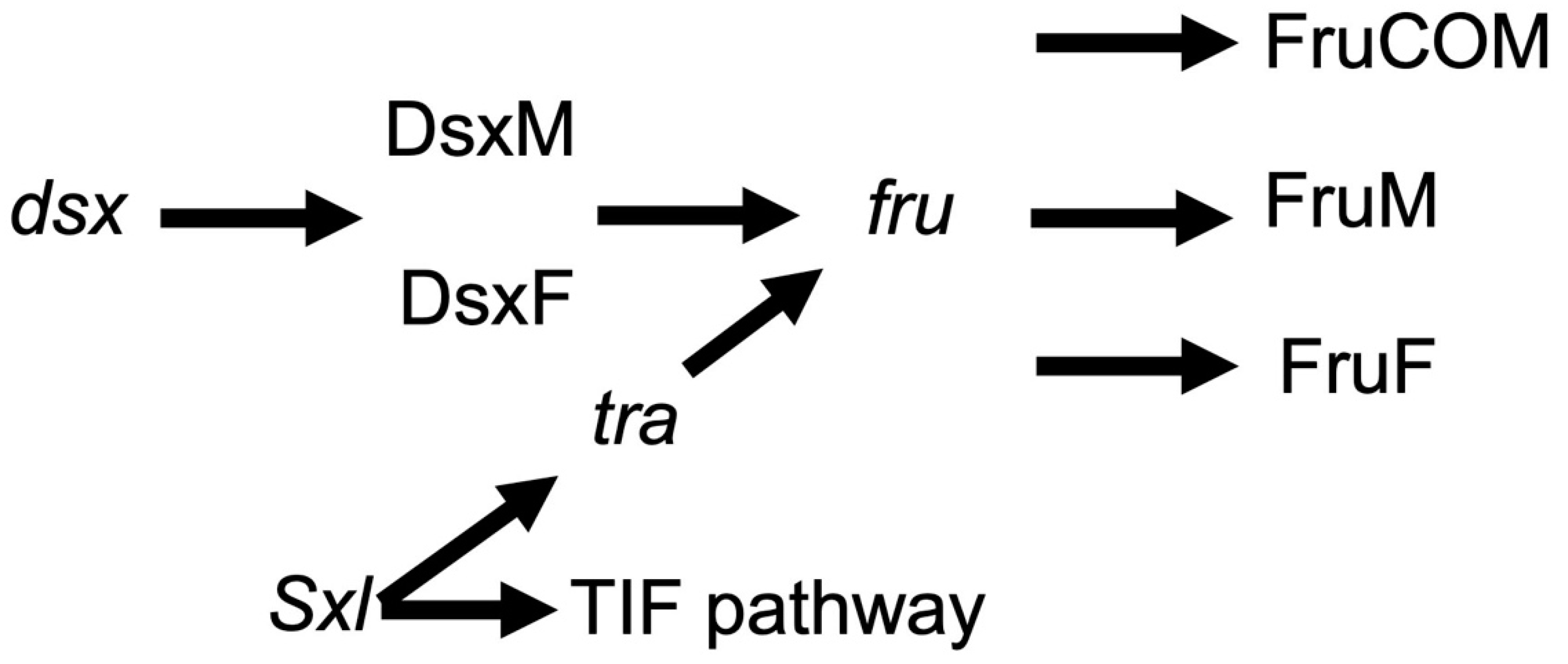
Major sex determination genes as indicators of central neurons involved in female reproduction
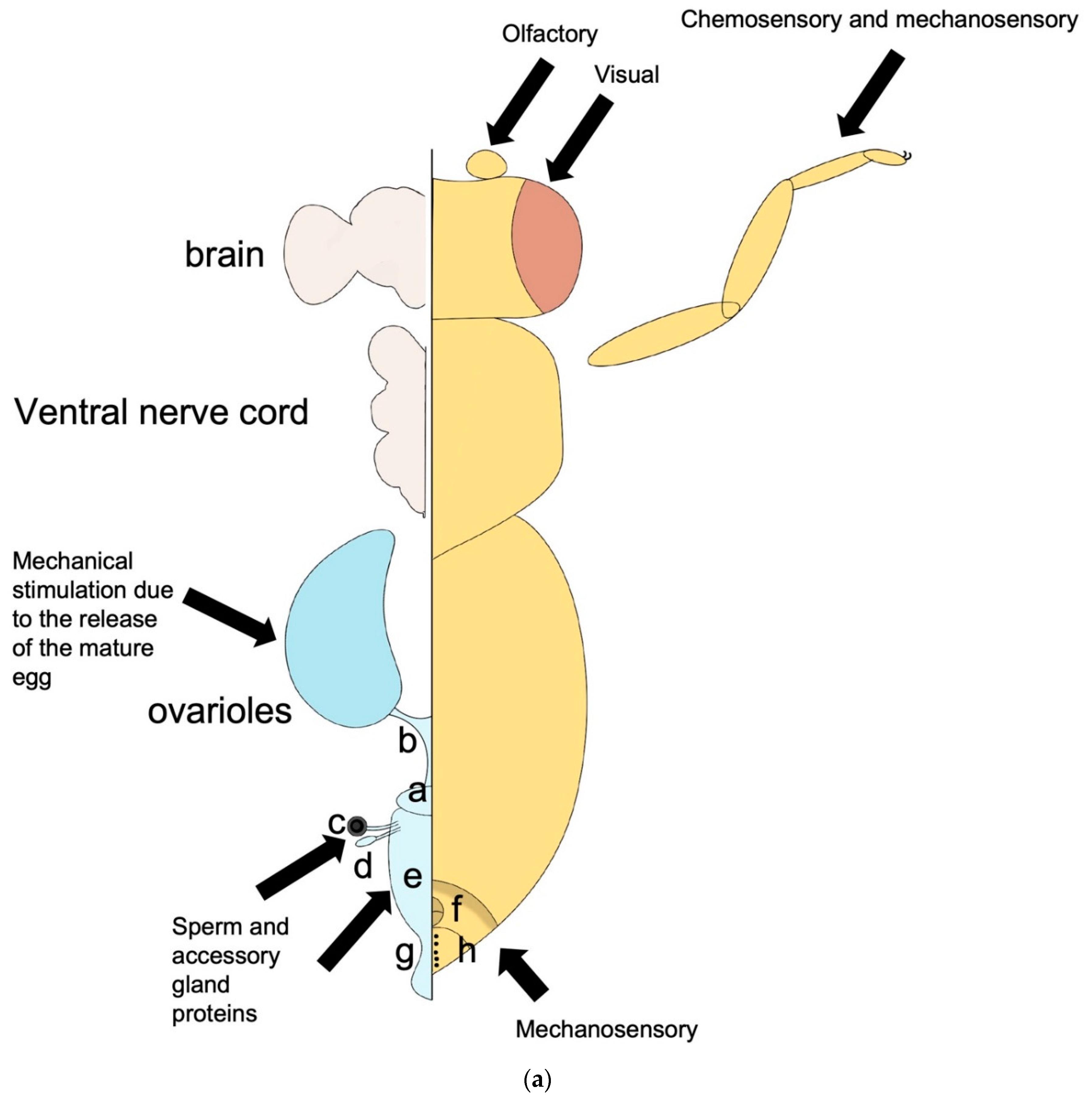
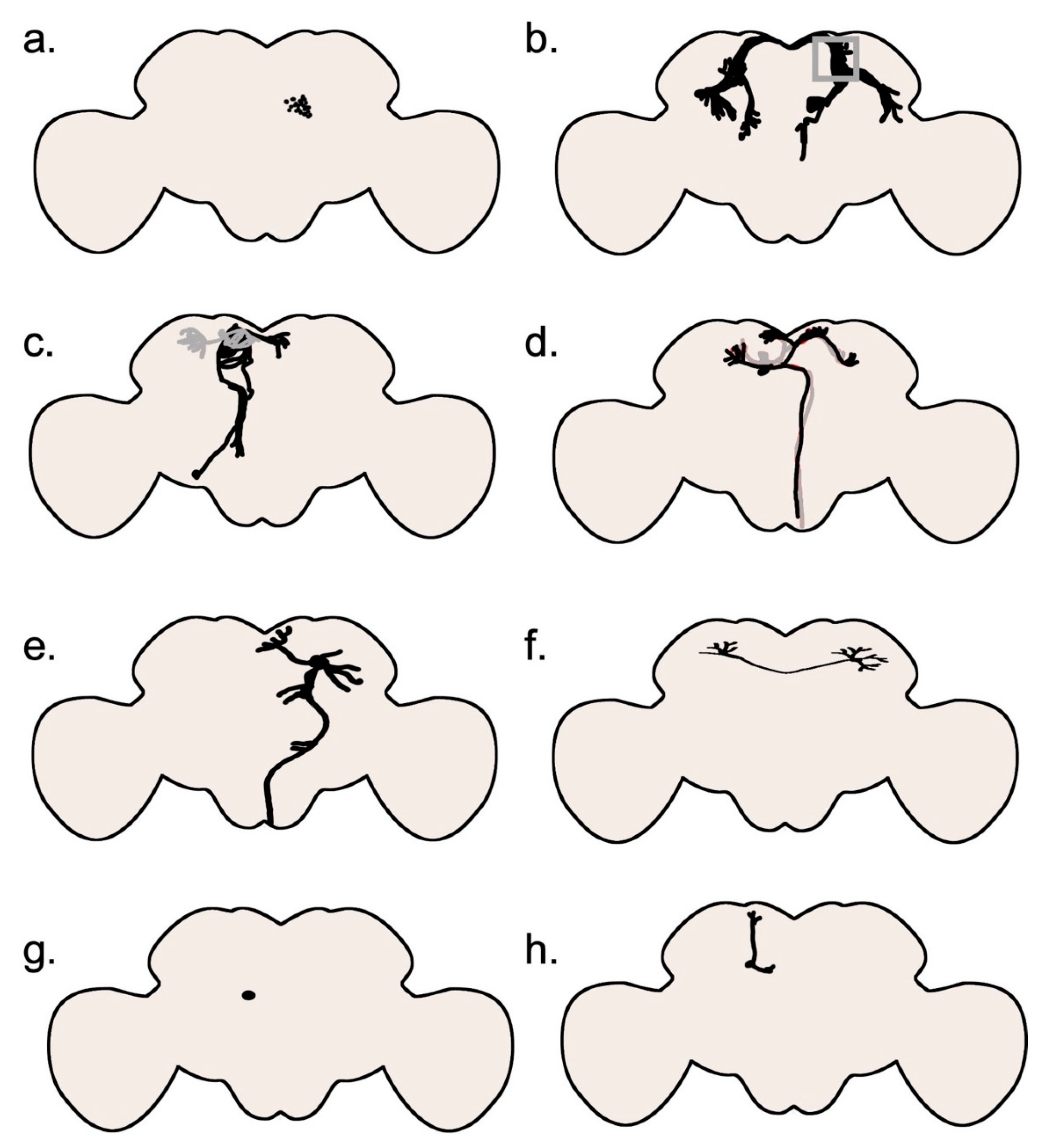
Central nervous system: The Brain
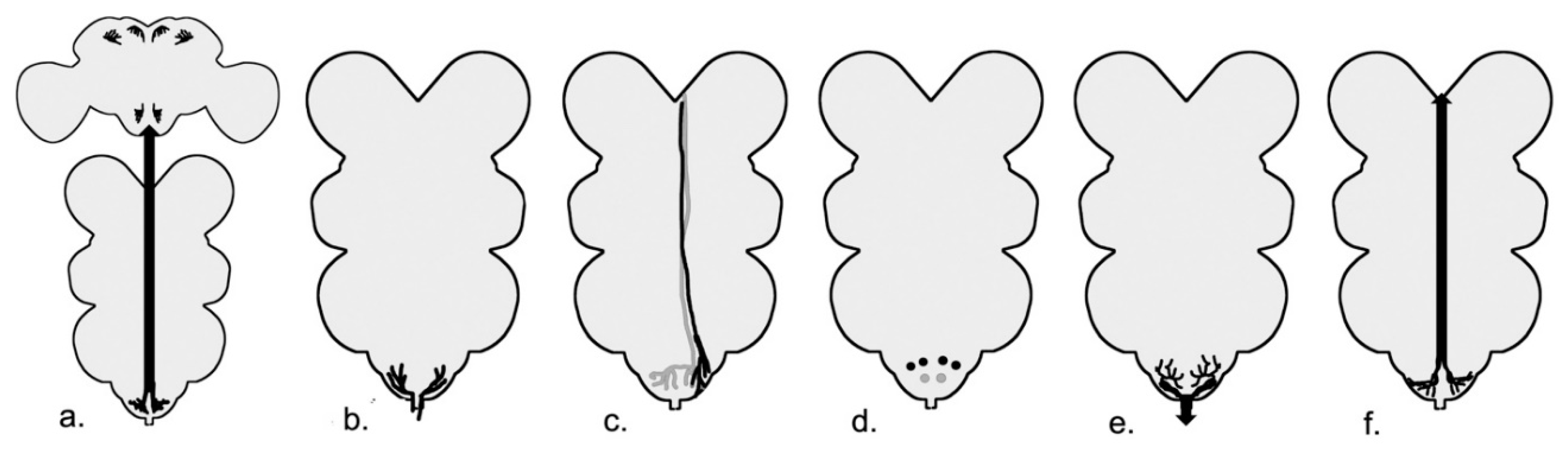
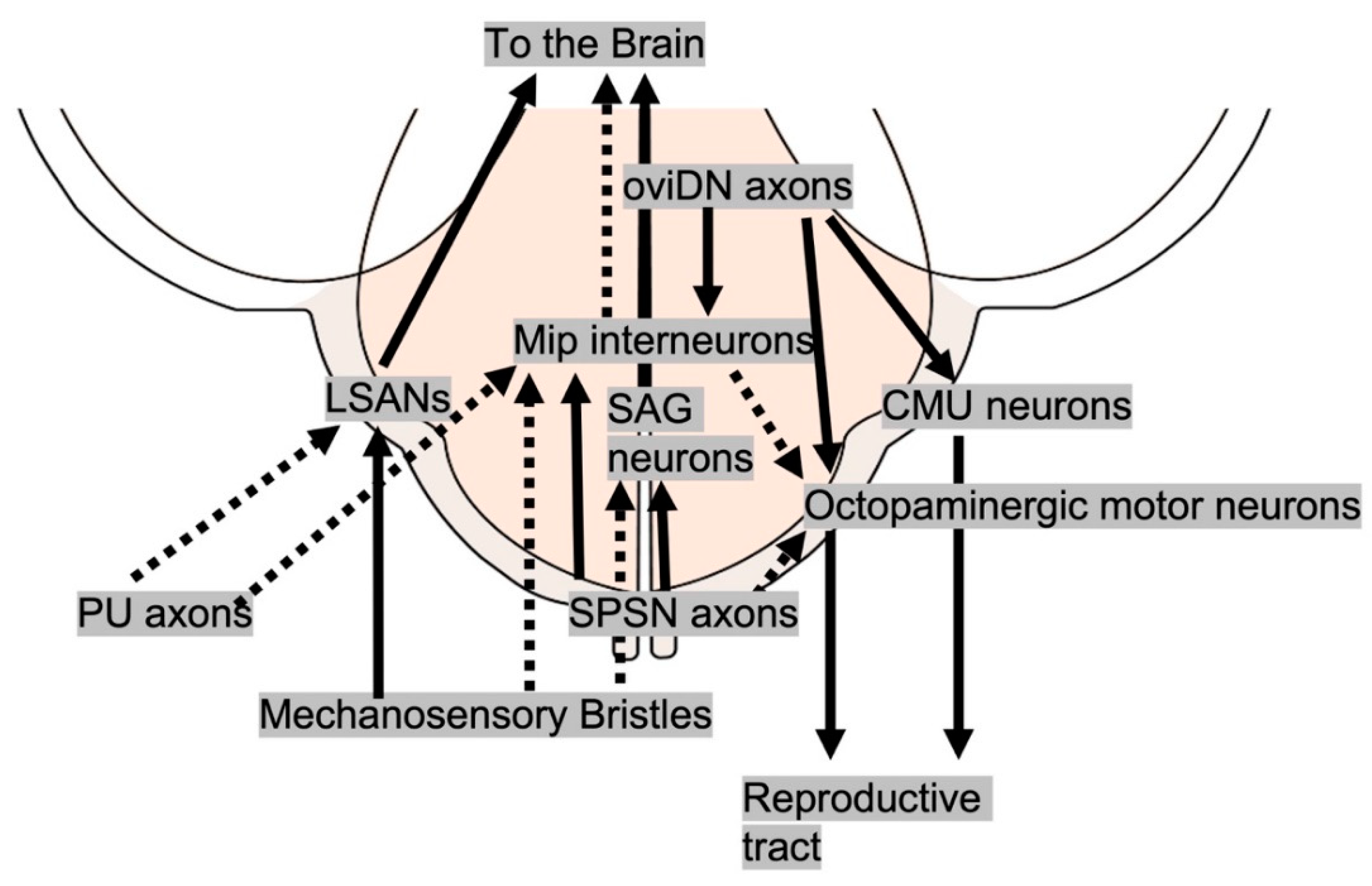
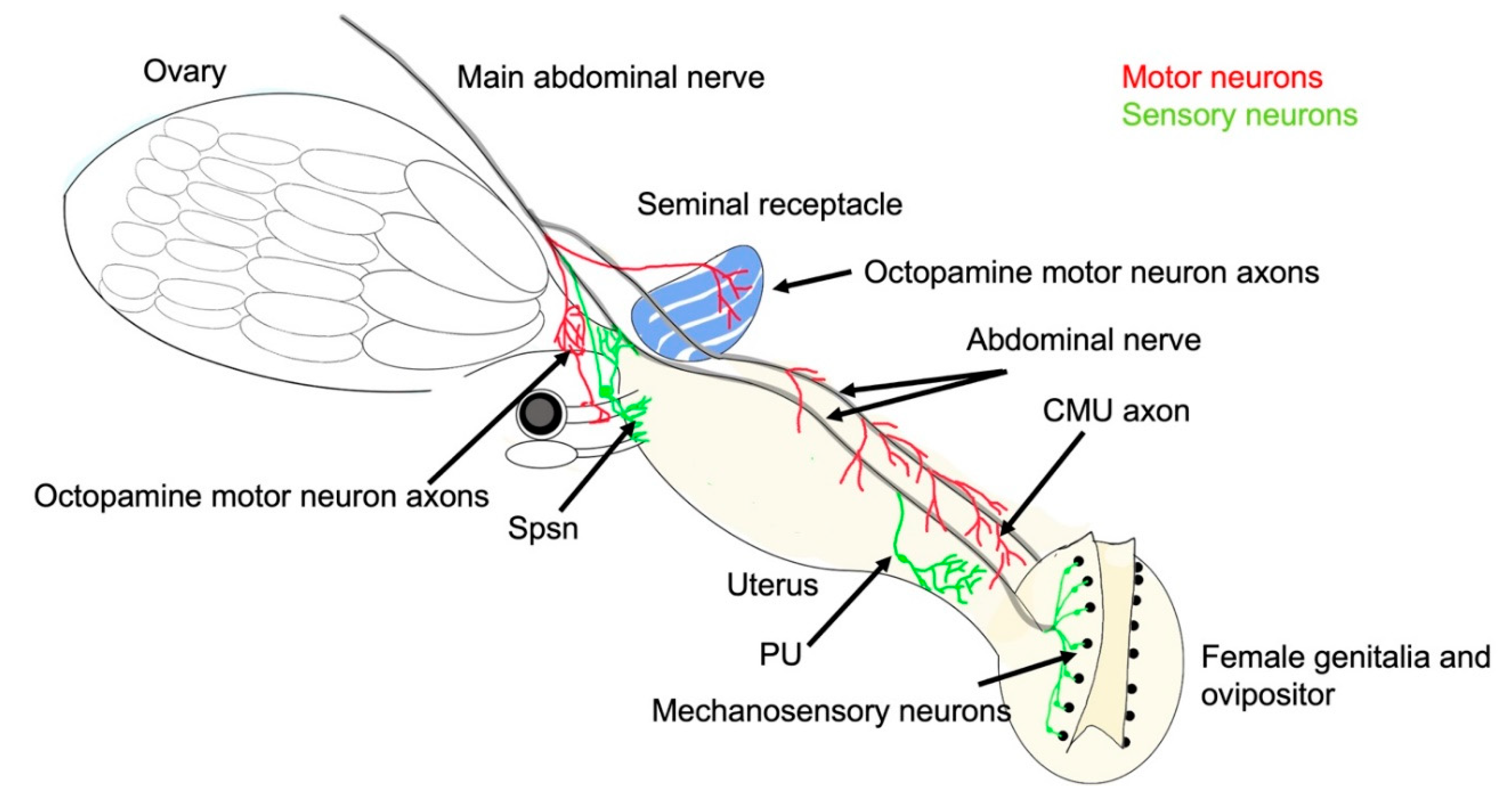
The abdominal neuromere in the ventral nerve cord:
Sensory neurons that influence egg-laying:
Why is neuronal control of egg-laying important in light of evolution?
Funding
Acknowledgements
References
- Adams, E.M.; Wolfner, M.F. Seminal proteins but not sperm induce morphological changes in the Drosophila melanogaster female reproductive tract during sperm storage. Journal of Insect Physiology 2007, 53, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.C.; Fernández, M.P.; Yu, Q.; Leary, G.P.; Leung, A.K.W.; Kavanaugh, M.P.; Kravitz, E.A.; Certel, S.J. Octopamine Neuromodulation Regulates Gr32a-Linked Aggression and Courtship Pathways in Drosophila Males. PLoS Genetics 2014, 10, e1004356. [Google Scholar] [CrossRef] [PubMed]
- Anholt, R.R.H.; O’Grady, P.; Wolfner, M.F.; Harbison, S.T. Evolution of Reproductive Behavior. Genetics 2020, 214, 49–73. [Google Scholar] [CrossRef] [PubMed]
- Atallah, J.; Teixeira, L.; Salazar, R.; Zaragoza, G.; Kopp, A. The making of a pest: The evolution of a fruit-penetrating ovipositor in Drosophila suzukii and related species. Proceedings of the Royal Society B: Biological Sciences 2014, 281, 20132840. [Google Scholar] [CrossRef] [PubMed]
- Auer, T.O.; Khallaf, M.A.; Silbering, A.F.; Zappia, G.; Ellis, K.; Álvarez-Ocaña, R.; Arguello, J.R.; Hansson, B.S.; Jefferis, G.S.X.E.; Caron, S.J.C.; Knaden, M.; Benton, R. Olfactory receptor and circuit evolution promote host specialization. Nature 2020, 579, 402–408. [Google Scholar] [CrossRef]
- Avila, F.W.; Mattei, A.L.; Wolfner, M.F. Sex peptide receptor is required for the release of stored sperm by mated Drosophila melanogaster females. Journal of Insect Physiology 2015, 76, 1–6. [Google Scholar] [CrossRef]
- Avila, F.W.; Sirot, L.K.; LaFlamme, B.A.; Rubinstein, C.D.; Wolfner, M.F. Insect Seminal Fluid Proteins: Identification and Function. Annual Review of Entomology 2011, 56, 21–40. [Google Scholar] [CrossRef]
- Azevedo, R.B.R.; French, V.; Partridge, L. Life-History Consequences of Egg Size in Drosophila Melanogaster. The American Naturalist 1997, 150, 250–282. [Google Scholar] [CrossRef]
- Baker, B.S.; Wolfner, M.F. A molecular analysis of doublesex, a bifunctional gene that controls both male and female sexual differentiation in Drosophila melanogaster. Genes & Development 1988, 2, 477–489. [Google Scholar] [CrossRef]
- Bang, A.G.; Hartenstein, V.; Posakony, J.W. Hairless is required for the development of adult sensory organ precursor cells in Drosophila. Development 1991, 111, 89. [Google Scholar] [CrossRef]
- Bates, A.S.; Janssens, J.; Jefferis, G.S.; Aerts, S. Neuronal cell types in the fly: Single-cell anatomy meets single-cell genomics. Current Opinion in Neurobiology 2019, 56, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Bath, E.; Bowden, S.; Peters, C.; Reddy, A.; Tobias, J.A.; Easton-Calabria, E.; Seddon, N.; Goodwin, S.F.; Wigby, S. Sperm and sex peptide stimulate aggression in female Drosophila. Nature Ecology & Evolution 2017, 1, 0154. [Google Scholar] [CrossRef]
- Bath, E.; Morimoto, J.; Wigby, S. The developmental environment modulates mating-induced aggression and fighting success in adult female Drosophila. Functional Ecology 2018, 32, 2542–2552. [Google Scholar] [CrossRef] [PubMed]
- Belgacem, Y.H.; Martin, J.-R. Neuroendocrine control of a sexually dimorphic behavior by a few neurons of the pars intercerebralis in Drosophila. Proceedings of the National Academy of Sciences 2002, 99, 15154–15158. [Google Scholar] [CrossRef]
- Bilen, J.; Atallah, J.; Azanchi, R.; Levine, J.D.; Riddiford, L.M. Regulation of onset of female mating and sex pheromone production by juvenile hormone in Drosophila melanogaster. Proceedings of the National Academy of Sciences 2013, 110, 18321–18326. [Google Scholar] [CrossRef]
- Billeter, J.-C.; Rideout, E.J.; Dornan, A.J.; Goodwin, S.F. Control of Male Sexual Behavior in Drosophila by the Sex Determination Pathway. Current Biology 2006, 16, R766–R776. [Google Scholar] [CrossRef]
- Billeter, J.-C.; Wolfner, M.F. Chemical Cues that Guide Female Reproduction in Drosophila melanogaster. Journal of Chemical Ecology 2018, 44, 750–769. [Google Scholar] [CrossRef]
- Cachero, S.; Ostrovsky, A.D.; Yu, J.Y.; Dickson, B.J.; Jefferis, G.S.X.E. Sexual Dimorphism in the Fly Brain. Current Biology 2010, 20, 1589–1601. [Google Scholar] [CrossRef]
- Carlsson, M.A.; Diesner, M.; Schachtner, J.; Nässel, D.R. Multiple neuropeptides in the Drosophila antennal lobe suggest complex modulatory circuits. The Journal of Comparative Neurology 2010, 518, 3359–3380. [Google Scholar] [CrossRef]
- Casares, F.; Sánchez, L.; Guerrero, I.; Sánchez-Herrero, E. The genital disc of Drosophila melanogaster. Development Genes and Evolution 1997, 207, 216–228. [Google Scholar] [CrossRef]
- Castellanos, M.C.; Tang, J.C.Y.; Allan, D.W. Female-biased dimorphism underlies a female-specific role for post-embryonic Ilp7 neurons in Drosophila fertility. Development 2013, 140, 3915–3926. [Google Scholar] [CrossRef]
- Chapman, T.; Arnqvist, G.; Bangham, J.; Rowe, L. Sexual conflict. Trends in Ecology & Evolution 2003, 18, 41–47. [Google Scholar] [CrossRef]
- Chiang, A.-S.; Burns, E.L.; Schal, C. Ovarian regulation of cyclic changes in size and activity of corpus allatum cells in Blattella germanica. Journal of Insect Physiology 1991, 37, 907–917. [Google Scholar] [CrossRef]
- Chow, C.Y.; Wolfner, M.F.; Clark, A.G. The Genetic Basis for Male × Female Interactions Underlying Variation in Reproductive Phenotypes of Drosophila. Genetics 2010, 186, Article 4. [Google Scholar] [CrossRef]
- Court, R.; Namiki, S.; Armstrong, J.D.; Börner, J.; Card, G.; Costa, M.; Dickinson, M.; Duch, C.; Korff, W.; Mann, R.; Merritt, D.; Murphey, R.K.; Seeds, A.M.; Shirangi, T.; Simpson, J.H.; Truman, J.W.; Tuthill, J.C.; Williams, D.W.; Shepherd, D. A Systematic Nomenclature for the Drosophila Ventral Nerve Cord. Neuron 2020, 107, 1071–1079e2. [Google Scholar] [CrossRef]
- Crava, C.M.; Zanini, D.; Amati, S.; Sollai, G.; Crnjar, R.; Paoli, M.; Rossi-Stacconi, M.V.; Rota-Stabelli, O.; Tait, G.; Haase, A.; Romani, R.; Anfora, G. Structural and transcriptional evidence of mechanotransduction in the Drosophila suzukii ovipositor. Journal of Insect Physiology 2020, 125, 104088. [Google Scholar] [CrossRef]
- Cury, K.M.; Axel, R. Flexible neural control of transition points within the egg-laying behavioral sequence in Drosophila. Nature Neuroscience 2023, 26, 1054–1067. [Google Scholar] [CrossRef]
- DeFazio, R.A.; Moenter, S.M. Estradiol Feedback Alters Potassium Currents and Firing Properties of Gonadotropin-Releasing Hormone Neurons. Molecular Endocrinology 2002, 16, 2255–2265. [Google Scholar] [CrossRef]
- Dekker, T.; Ibba, I.; Siju, K.P.; Stensmyr, M.C.; Hansson, B.S. Olfactory Shifts Parallel Superspecialism for Toxic Fruit in Drosophila melanogaster Sibling, D. sechellia. Current Biology 2006, 16, 101–109. [Google Scholar] [CrossRef]
- Demir, E.; Dickson, B.J. Fruitless Splicing Specifies Male Courtship Behavior in Drosophila. Cell 2005, 121, 785–794. [Google Scholar] [CrossRef]
- Deutsch, D.; Pacheco, D.; Encarnacion-Rivera, L.; Pereira, T.; Fathy, R.; Clemens, J.; Girardin, C.; Calhoun, A.; Ireland, E.; Burke, A.; Dorkenwald, S.; McKellar, C.; Macrina, T.; Lu, R.; Lee, K.; Kemnitz, N.; Ih, D.; Castro, M.; Halageri, A.; … Murthy, M. The neural basis for a persistent internal state in Drosophila females. ELife 2020, 9, e59502. [Google Scholar] [CrossRef]
- Domínguez, M.; Campuzano, S. Asense, a member of the Drosophila achaete-scute complex, is a proneural and neural differentiation gene. The EMBO Journal 1993, 12, 2049–2060. [Google Scholar] [CrossRef]
- Dorkenwald, S.; McKellar, C.E.; Macrina, T.; Kemnitz, N.; Lee, K.; Lu, R.; Wu, J.; Popovych, S.; Mitchell, E.; Nehoran, B.; Jia, Z.; Bae, J.A.; Mu, S.; Ih, D.; Castro, M.; Ogedengbe, O.; Halageri, A.; Kuehner, K.; Sterling, A.R.; … Seung, H.S. FlyWire: Online community for whole-brain connectomics. Nature Methods 2022, 19, 119–128. [Google Scholar] [CrossRef]
- Duckhorn, J.C.; Cande, J.; Metkus, M.C.; Song, H.; Altamirano, S.; Stern, D.L.; Shirangi, T.R. Regulation of Drosophila courtship behavior by the Tlx/tailless-like nuclear receptor, dissatisfaction. Current Biology 2022, 32, 1703–1714e3. [Google Scholar] [CrossRef]
- Duménil, C.; Woud, D.; Pinto, F.; Alkema, J.T.; Jansen, I.; Van Der Geest, A.M.; Roessingh, S.; Billeter, J.-C. Pheromonal Cues Deposited by Mated Females Convey Social Information about Egg-Laying Sites in Drosophila Melanogaster. Journal of Chemical Ecology 2016, 42, 259–269. [Google Scholar] [CrossRef]
- Durkin, S.M.; Chakraborty, M.; Abrieux, A.; Lewald, K.M.; Gadau, A.; Svetec, N.; Peng, J.; Kopyto, M.; Langer, C.B.; Chiu, J.C.; Emerson, J.J.; Zhao, L. Behavioral and Genomic Sensory Adaptations Underlying the Pest Activity of Drosophila suzukii. Molecular Biology and Evolution 2021, 38, 2532–2546. [Google Scholar] [CrossRef]
- Eberhard, W.G. Female control: Sexual selection by cryptic female choice. Princeton University Press.
- Eisses 1996, K. Th. The influence of 2-propanol and acetone on oviposition rate and oviposition site preference for acetic acid and ethanol of Drosophila melanogaster. Behavior Genetics 1997, 27, 171–180. [Google Scholar] [CrossRef]
- Engels, W.R.; Preston, C.R. HYBRID DYSGENESIS IN DROSOPHILA MELANOGASTER: THE BIOLOGY OF FEMALE AND MALE STERILITY. Genetics 1979, 92, 161–174. [Google Scholar] [CrossRef]
- Evans, D.S.; Cline, T.W. Drosophila switch gene Sex-lethal can bypass its switch-gene target transformer to regulate aspects of female behavior. Proceedings of the National Academy of Sciences 2013, 110. [Google Scholar] [CrossRef]
- Fan, P.; Manoli, D.S.; Ahmed, O.M.; Chen, Y.; Agarwal, N.; Kwong, S.; Cai, A.G.; Neitz, J.; Renslo, A.; Baker, B.S.; Shah, N.M. Genetic and Neural Mechanisms that Inhibit Drosophila from Mating with Other Species. Cell 2013, 154, 89–102. [Google Scholar] [CrossRef]
- Feng, K.; Palfreyman, M.T.; Häsemeyer, M.; Talsma, A.; Dickson, B.J. Ascending SAG Neurons Control Sexual Receptivity of Drosophila Females. Neuron 2014, 83, 135–148. [Google Scholar] [CrossRef]
- Fowler, E.K.; Leigh, S.; Rostant, W.G.; Thomas, A.; Bretman, A.; Chapman, T. Memory of social experience affects female fecundity via perception of fly deposits. BMC Biology 2022, 20, 244. [Google Scholar] [CrossRef] [PubMed]
- Frazee, S.R.; Harper, A.R.; Afkhami, M.; Wood, M.L.; McCrory, J.C.; Masly, J.P. Interspecific introgression reveals a role of male genital morphology during the evolution of reproductive isolation in Drosophila. Evolution 2021, 75, 989–1002. [Google Scholar] [CrossRef]
- Frazee, S.R.; Masly, J.P. Multiple sexual selection pressures drive the rapid evolution of complex morphology in a male secondary genital structure. Ecology and Evolution 2015, 5, 4437–4450. [Google Scholar] [CrossRef]
- Garaulet, D.L.; Moro, A.; Lai, E.C. A double-negative gene regulatory circuit underlies the virgin behavioral state. Cell Reports 2021, 36, 109335. [Google Scholar] [CrossRef]
- Garbe, D.S.; Vigderman, A.S.; Moscato, E.; Dove, A.E.; Vecsey, C.G.; Kayser, M.S.; Sehgal, A. Changes in Female Drosophila Sleep following Mating Are Mediated by SPSN-SAG Neurons. Journal of Biological Rhythms 2016, 31, 551–567. [Google Scholar] [CrossRef]
- García-Bellido, A.; Santamaria, P. DEVELOPMENTAL ANALYSIS OF THE ACHAETE-SCUTE SYSTEM OF DROSOPHILA MELANOGASTER. Genetics 1978, 88, 469–486. [Google Scholar] [CrossRef]
- Goto, J.; Mikawa, Y.; Koganezawa, M.; Ito, H.; Yamamoto, D. Sexually Dimorphic Shaping of Interneuron Dendrites Involves the Hunchback Transcription Factor. Journal of Neuroscience 2011, 31, 5454–5459. [Google Scholar] [CrossRef]
- Grossfield, J.; Sakri, B. Divergence in the neural control of oviposition in Drosophila. Journal of Insect Physiology 1972, 18, 237–241. [Google Scholar] [CrossRef]
- Grunwald Kadow, I.C.; Gompel, N. Sensory Evolution: Making Sense of the Noni Scent. Current Biology 2020, 30, R712–R715. [Google Scholar] [CrossRef]
- Guntur, A.R.; Gou, B.; Gu, P.; He, R.; Stern, U.; Xiang, Y.; Yang, C.-H. H2O2-Sensitive Isoforms of Drosophila melanogaster TRPA1 Act in Bitter-Sensing Gustatory Neurons to Promote Avoidance of UV During Egg-Laying. Genetics 2017, 205, 749–759. [Google Scholar] [CrossRef]
- Gwadz, R.W.; Spielman, A. Corpus allatum control of ovarian development in Aedes aegypti. Journal of Insect Physiology 1973, 19, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.C.; Greenspan, R.J. GENETIC ANALYSIS OF DROSOPHILA NEUROBIOLOGY. Annual Review of Genetics 1979, 13, 127–195. [Google Scholar] [CrossRef]
- Harshman, L.G.; Zera, A.J. The cost of reproduction: The devil in the details. Trends in Ecology & Evolution 2007, 22, 80–86. [Google Scholar] [CrossRef]
- Hasebe, M.; Shiga, S. Oviposition-promoting pars intercerebralis neurons show period -dependent photoperiodic changes in their firing activity in the bean bug. Proceedings of the National Academy of Sciences 2021, 118, e2018823118. [Google Scholar] [CrossRef]
- Häsemeyer, M.; Yapici, N.; Heberlein, U.; Dickson, B.J. Sensory Neurons in the Drosophila Genital Tract Regulate Female Reproductive Behavior. Neuron 2009, 61, 511–518. [Google Scholar] [CrossRef]
- Haussmann, I.U.; Hemani, Y.; Wijesekera, T.; Dauwalder, B.; Soller, M. Multiple pathways mediate the sex-peptide-regulated switch in female Drosophila reproductive behaviours. Proceedings of the Royal Society B: Biological Sciences 2013, 280, 20131938. [Google Scholar] [CrossRef]
- Hehlert, P.; Zhang, W.; Göpfert, M.C. Drosophila Mechanosensory Transduction. Trends in Neurosciences 2021, 44, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Heifetz, Y.; Lung, O.; Frongillo, E.A.; Wolfner, M.F. The Drosophila seminal fluid protein Acp26Aa stimulates release of oocytes by the ovary. Current Biology 2000, 10, 99–102. [Google Scholar] [CrossRef]
- Heifetz, Y.; Wolfner, M.F. Mating, seminal fluid components, and sperm cause changes in vesicle release in the Drosophila female reproductive tract. Proceedings of the National Academy of Sciences 2004, 101, 6261–6266. [Google Scholar] [CrossRef] [PubMed]
- Herndon, L.A.; Wolfner, M.F. A Drosophila seminal fluid protein, Acp26Aa, stimulates egg laying in females for 1 day after mating. Proceedings of the National Academy of Sciences 1995, 92, 10114–10118. [Google Scholar] [CrossRef]
- Hudson, A.M.; Petrella, L.N.; Tanaka, A.J.; Cooley, L. Mononuclear muscle cells in Drosophila ovaries revealed by GFP protein traps. Developmental Biology 2008, 314, 329–340. [Google Scholar] [CrossRef]
- Isaac, R.E.; Kim, Y.-J.; Audsley, N. The degradome and the evolution of Drosophila sex peptide as a ligand for the MIP receptor. Peptides 2014, 53, 258–264. [Google Scholar] [CrossRef]
- Ishimoto, H.; Kamikouchi, A. Molecular and neural mechanisms regulating sexual motivation of virgin female Drosophila. Cellular and Molecular Life Sciences 2021, 78, 4805–4819. [Google Scholar] [CrossRef]
- Jafar-Nejad, H. Senseless and Daughterless confer neuronal identity to epithelial cells in the Drosophila wing margin. Development 2006, 133, 1683–1692. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.-H.; Chae, H.-S.; Kim, Y.-J. Female-specific myoinhibitory peptide neurons regulate mating receptivity in Drosophila melanogaster. Nature Communications 2017, 8, 1630. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.M.; Heberlein, U. Tissue-Specific Activation of a Single Gustatory Receptor Produces Opposing Behavioral Responses in Drosophila. Genetics 2012, 192, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Jowett, T.; Postlethwait, J.H. The regulation of yolk polypeptide synthesis in Drosophila ovaries and fat body by 20-hydroxyecdysone and a juvenile hormone analog. Developmental Biology 1980, 80, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Karageorgi, M.; Bräcker, L.B.; Lebreton, S.; Minervino, C.; Cavey, M.; Siju, K.P.; Grunwald Kadow, I.C.; Gompel, N.; Prud’homme, B. Evolution of Multiple Sensory Systems Drives Novel Egg-Laying Behavior in the Fruit Pest Drosophila suzukii. Current Biology 2017, 27, 847–853. [Google Scholar] [CrossRef]
- Kienzle, R.; Groß, L.B.; Caughman, S.; Rohlfs, M. Resource use by individual Drosophila suzukii reveals a flexible preference for oviposition into healthy fruits. Scientific Reports 2020, 10, 3132. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Bartalska, K.; Audsley, N.; Yamanaka, N.; Yapici, N.; Lee, J.-Y.; Kim, Y.-C.; Markovic, M.; Isaac, E.; Tanaka, Y.; Dickson, B.J. MIPs are ancestral ligands for the sex peptide receptor. Proceedings of the National Academy of Sciences 2010, 107, 6520–6525. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Hachiya, T.; Koganezawa, M.; Tazawa, T.; Yamamoto, D. Fruitless and Doublesex Coordinate to Generate Male-Specific Neurons that Can Initiate Courtship. Neuron 2008, 59, 759–769. [Google Scholar] [CrossRef]
- Kimura, K.; Sato, C.; Koganezawa, M.; Yamamoto, D. Drosophila Ovipositor Extension in Mating Behavior and Egg Deposition Involves Distinct Sets of Brain Interneurons. PLOS ONE 2015, 10, e0126445. [Google Scholar] [CrossRef]
- Kimura, K.-I.; Ote, M.; Tazawa, T.; Yamamoto, D. Fruitless specifies sexually dimorphic neural circuitry in the Drosophila brain. Nature 2005, 438, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Kubli, E. Sex-peptides: Seminal peptides of the Drosophila male. Cellular and Molecular Life Sciences (CMLS) 2003, 60, 1689–1704. [Google Scholar] [CrossRef]
- Kurz, C.L.; Charroux, B.; Chaduli, D.; Viallat-Lieutaud, A.; Royet, J. Peptidoglycan sensing by octopaminergic neurons modulates Drosophila oviposition. ELife 2017, 6, e21937. [Google Scholar] [CrossRef]
- Kvitsiani, D.; Dickson, B.J. Shared neural circuitry for female and male sexual behaviours in Drosophila. Current Biology 2006, 16, R355–R356. [Google Scholar] [CrossRef]
- LaFever, L.; Drummond-Barbosa, D. Direct Control of Germline Stem Cell Division and Cyst Growth by Neural Insulin in Drosophila. Science 2005, 309, 1071–1073. [Google Scholar] [CrossRef]
- Lee, H.; Choi, H.W.; Zhang, C.; Park, Z.-Y.; Kim, Y.-J. A Pair of Oviduct-Born Pickpocket Neurons Important for Egg-Laying in Drosophila melanogaster. Molecules and Cells 2016, 39, 573–579. [Google Scholar] [CrossRef]
- LeVasseur-Viens, H.; Polak, M.; Moehring, A.J. No evidence for external genital morphology affecting cryptic female choice and reproductive isolation in Drosophila: GENITAL SHAPE AND SEXUAL SELECTION. Evolution 2015, 69, 1797–1807. [Google Scholar] [CrossRef]
- Lin, C.-C.; Prokop-Prigge, K.A.; Preti, G.; Potter, C.J. Food odors trigger Drosophila males to deposit a pheromone that guides aggregation and female oviposition decisions. ELife 2015, 4, e08688. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, B.; Zhang, L.; Yang, T.; Zhang, Z.; Gao, Z.; Zhang, W. A neural circuit encoding mating states tunes defensive behavior in Drosophila. Nature Communications 2020, 11, 3962. [Google Scholar] [CrossRef] [PubMed]
- Manning, A. The control of sexual receptivity in female Drosophila. Animal Behaviour, 1967; 15, 239–250. [Google Scholar] [CrossRef]
- Markow, T.A.; Beall, S.; Matzkin, L.M. Egg size, embryonic development time and ovoviviparity in Drosophila species: Ovoviviparity in Drosophila species. Journal of Evolutionary Biology 2009, 22, 430–434. [Google Scholar] [CrossRef]
- Matsumoto, S.; Brown, M.R.; Suzuki, A.; Lea, A.O. Isolation and characterization of ovarian ecdysteroidogenic hormones from the mosquito, Aedes aegypti. Insect Biochemistry 1989, 19, 651–656. [Google Scholar] [CrossRef]
- McMillan, I.; Fitz-Earle, M.; Robson, D.S. QUANTITATIVE GENETICS OF FERTILITY I. LIFETIME EGG PRODUCTION OF DROSOPHILA MELANOGASTER—THEORETICAL. Genetics 1970, 65, 349–353. [Google Scholar] [CrossRef] [PubMed]
- McQueen, E.W.; Afkhami, M.; Atallah, J.; Belote, J.M.; Gompel, N.; Heifetz, Y.; Kamimura, Y.; Kornhauser, S.C.; Masly, J.P.; O’Grady, P.; Peláez, J.; Rebeiz, M.; Rice, G.; Sánchez-Herrero, E.; Santos Nunes, M.D.; Santos Rampasso, A.; Schnakenberg, S.L.; Siegal, M.L.; Takahashi, A.; … Yassin, A. A standardized nomenclature and atlas of the female terminalia of Drosophila melanogaster. Fly 2022, 16, 128–151. [Google Scholar] [CrossRef] [PubMed]
- Mezzera, C.; Brotas, M.; Gaspar, M.; Pavlou, H.J.; Goodwin, S.F.; Vasconcelos, M.L. Ovipositor Extrusion Promotes the Transition from Courtship to Copulation and Signals Female Acceptance in Drosophila melanogaster. Current Biology 2020, 30, 3736–3748e5. [Google Scholar] [CrossRef]
- Milyaev, N.; Osumi-Sutherland, D.; Reeve, S.; Burton, N.; Baldock, R.A.; Armstrong, J.D. The Virtual Fly Brain browser and query interface. Bioinformatics 2012, 28, 411–415. [Google Scholar] [CrossRef]
- Min, S.; Chae, H.-S.; Jang, Y.-H.; Choi, S.; Lee, S.; Jeong, Y.T.; Jones, W.D.; Moon, S.J.; Kim, Y.-J.; Chung, J. Identification of a Peptidergic Pathway Critical to Satiety Responses in Drosophila. Current Biology 2016, 26, 814–820. [Google Scholar] [CrossRef]
- Mirth, C.K.; Nogueira Alves, A.; Piper, M.D. Turning food into eggs: Insights from nutritional biology and developmental physiology of Drosophila. Current Opinion in Insect Science 2019, 31, 49–57. [Google Scholar] [CrossRef]
- Monastirioti, M. Distinct octopamine cell population residing in the CNS abdominal ganglion controls ovulation in Drosophila melanogaster. Developmental Biology 2003, 264, 38–49. [Google Scholar] [CrossRef]
- Monastirioti, M.; Linn, Jr. ; C. E.; White, K. Characterization of Drosophila Tyramine β-Hydroxylase Gene and Isolation of Mutant Flies Lacking Octopamine. The Journal of Neuroscience 1996, 16, 3900–3911. [Google Scholar] [CrossRef]
- Moshitzky, P.; Fleischmann, I.; Chaimov, N.; Saudan, P.; Klauser, S.; Kubli, E.; Applebaum, S.W. Sex-peptide activates juvenile hormone biosynthesis in the Drosophila melanogaster corpus allatum. Archives of Insect Biochemistry and Physiology, 1996; 32, 363–374. [Google Scholar]
- Nakamura, M.; Okano, H.; Blendy, J.A.; Montell, C. Musashi, a neural RNA-binding protein required for Drosophila adult external sensory organ development. Neuron 1994, 13, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Newell, N.R.; Ray, S.; Dalton, J.E.; Fortier, J.C.; Kao, J.Y.; Chang, P.L.; Nuzhdin, S.V.; Arbeitman, M.N. The Drosophila Post-mating Response: Gene Expression and Behavioral Changes Reveal Perdurance and Variation in Cross-Tissue Interactions. G3 Genes|Genomes|Genetics 2020, 10, 967–983. [Google Scholar] [CrossRef]
- Nijhout, H.F. Insect Hormones. Princeton University Press. [CrossRef]
- Nojima 2021, T.; Rings, A.; Allen, A.M.; Otto, N.; Verschut, T.A.; Billeter, J.-C.; Neville, M.C.; Goodwin, S.F. A sex-specific switch between visual and olfactory inputs underlies adaptive sex differences in behavior. Current Biology 2021, 31, 1175–1191e6. [Google Scholar] [CrossRef]
- Ogawa, H.; Kagaya, K.; Saito, M.; Yamaguchi, T. Neural mechanism for generating and switching motor patterns of rhythmic movements of ovipositor valves in the cricket. Journal of Insect Physiology 2011, 57, 326–338. [Google Scholar] [CrossRef]
- Park, J.-H.; Kwon, J.Y. A systematic analysis of Drosophila gustatory receptor gene expression in abdominal neurons which project to the central nervous system. Molecules and Cells 2011, 32, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Parker, G.A. SEXUAL SELECTION AND SEXUAL CONFLICT. In Sexual Selection and Reproductive Competition in Insects (pp. 123–166). Elsevier. [CrossRef]
- Partridge 1979, L.; Fowler, K.; Trevitt, S.; Sharp, W. An examination of the effects of males on the survival and egg-production rates of female Drosophila melanogaster. Journal of Insect Physiology 1986, 32, 925–929. [Google Scholar] [CrossRef]
- Partridge, L.; Green, A.; Fowler, K. Effects of egg-production and of exposure to males on female survival in Drosophila melanogaster. Journal of Insect Physiology 1987, 33, 745–749. [Google Scholar] [CrossRef]
- Price, C.S.C.; Kim, C.H.; Gronlund, C.J.; Coyne, J.A. CRYPTIC REPRODUCTIVE ISOLATION IN THE DROSOPHILA SIMULANS SPECIES COMPLEX. Evolution 2001, 55, 81–92. [Google Scholar] [CrossRef]
- Reguera, P. Flexible oviposition behavior in the golden egg bug (Phyllomorpha laciniata) and its implications for offspring survival. Behavioral Ecology 2002, 13, 70–74. [Google Scholar] [CrossRef]
- Rezával, C.; Nojima, T.; Neville, M.C.; Lin, A.C.; Goodwin, S.F. Sexually Dimorphic Octopaminergic Neurons Modulate Female Postmating Behaviors in Drosophila. Current Biology 2014, 24, 725–730. [Google Scholar] [CrossRef]
- Rezával, C.; Pavlou, H.J.; Dornan, A.J.; Chan, Y.-B.; Kravitz, E.A.; Goodwin, S.F. Neural Circuitry Underlying Drosophila Female Postmating Behavioral Responses. Current Biology 2012, 22, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Dickson, B.J. Sex Peptide Receptor and Neuronal TOR/S6K Signaling Modulate Nutrient Balancing in Drosophila. Current Biology 2010, 20, 1000–1005. [Google Scholar] [CrossRef] [PubMed]
- Rideout, E.J.; Dornan, A.J.; Neville, M.C.; Eadie, S.; Goodwin, S.F. Control of sexual differentiation and behavior by the doublesex gene in Drosophila melanogaster. Nature Neuroscience 2010, 13, 458–466. [Google Scholar] [CrossRef]
- R’Kha, S.; Moreteau, B.; Coyne, J.A.; David, J.R. Evolution of a lesser fitness trait: Egg production in the specialist Drosophila sechellia. Genetical Research 1997, 69, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Valentín, R.; López-González, I.; Jorquera, R.; Labarca, P.; Zurita, M.; Reynaud, E. Oviduct contraction in Drosophila is modulated by a neural network that is both, octopaminergic and glutamatergic. Journal of Cellular Physiology 2006, 209, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Rosenheim, J.A.; Heimpel, G.E.; Mangel, M. Egg maturation, egg resorption and the costliness of transient egg limitation in insects. Proceedings of the Royal Society of London. Series B: Biological Sciences 2000, 267, 1565–1573. [Google Scholar] [CrossRef]
- Roy, S.; Saha, T.T.; Zou, Z.; Raikhel, A.S. Regulatory Pathways Controlling Female Insect Reproduction. Annual Review of Entomology 2018, 63, 489–511. [Google Scholar] [CrossRef]
- Rubinstein, C.D.; Wolfner, M.F. Drosophila seminal protein ovulin mediates ovulation through female octopamine neuronal signaling. Proceedings of the National Academy of Sciences 2013, 110, 17420–17425. [Google Scholar] [CrossRef]
- Sanders, L.E.; Arbeitman, M.N. Doublesex establishes sexual dimorphism in the Drosophila central nervous system in an isoform-dependent manner by directing cell number. Developmental Biology 2008, 320, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Yamamoto, D. Mutually exclusive expression of sex-specific and non-sex-specific fruitless gene products in the Drosophila central nervous system. Gene Expression Patterns 2022, 43, 119232. [Google Scholar] [CrossRef] [PubMed]
- Sawala, A.; Gould, A.P. The sex of specific neurons controls female body growth in Drosophila. PLOS Biology 2017, 15, e2002252. [Google Scholar] [CrossRef]
- Schlegel, P.; Costa, M.; Jefferis, G.S. Learning from connectomics on the fly. Current Opinion in Insect Science 2017, 24, 96–105. [Google Scholar] [CrossRef]
- Schwenke, R.A.; Lazzaro, B.P. Juvenile Hormone Suppresses Resistance to Infection in Mated Female Drosophila melanogaster. Current Biology 2017, 27, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Chung, P.; Wong, A.; Siwanowicz, I.; Kent, C.F.; Long, X.; Heberlein, U. A Neural Circuit Encoding the Experience of Copulation in Female Drosophila. Neuron 2019, 102, 1025–1036e6. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, A.B.; Wheelock, G.D.; Hagedorn, H.H.; Baker, F.C.; Tsai, L.W.; Schooley, D.A. Juvenile hormone and juvenile hormone esterase in adult females of the mosquito Aedes aegypti. Journal of Insect Physiology 1986, 32, 867–877. [Google Scholar] [CrossRef]
- Shapiro, H. THE RATE OF OVIPOSITION IN THE FRUIT FLY, DROSOPHILA. The Biological Bulletin 1932, 63, 456–471. [Google Scholar] [CrossRef]
- Shapiro, J.P.; Hagedorn, H.H. Juvenile hormone and the development of ovarian responsiveness to a brain hormone in the mosquito, Aedes aegypti. General and Comparative Endocrinology 1982, 46, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Shelly, T.E. Defense of Oviposition Sites by Female Oriental Fruit Flies (Diptera: Tephritidae). The Florida Entomologist 1999, 82, 339. [Google Scholar] [CrossRef]
- Shirangi, T.R.; Taylor, B.J.; McKeown, M. A double-switch system regulates male courtship behavior in male and female Drosophila melanogaster. Nature Genetics 2006, 38, 1435–1439. [Google Scholar] [CrossRef] [PubMed]
- Simpson, P. Lateral inhibition and the development of the sensory bristles of the adult peripheral nervous system of Drosophila. Development 1990, 109, 509. [Google Scholar] [CrossRef] [PubMed]
- Sirot, L.K.; Wong, A.; Chapman, T.; Wolfner, M.F. Sexual Conflict and Seminal Fluid Proteins: A Dynamic Landscape of Sexual Interactions. Cold Spring Harbor Perspectives in Biology 2015, 7, a017533. [Google Scholar] [CrossRef] [PubMed]
- Soller, M.; Bownes, M.; Kubli, E. Mating and Sex Peptide Stimulate the Accumulation of Yolk in Oocytes of Drosophila Melanogaster. European Journal of Biochemistry 1997, 243, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Stensmyr, M.C.; Dweck, H.K.M.; Farhan, A.; Ibba, I.; Strutz, A.; Mukunda, L.; Linz, J.; Grabe, V.; Steck, K.; Lavista-Llanos, S.; Wicher, D.; Sachse, S.; Knaden, M.; Becher, P.G.; Seki, Y.; Hansson, B.S. A Conserved Dedicated Olfactory Circuit for Detecting Harmful Microbes in Drosophila. Cell 2012, 151, 1345–1357. [Google Scholar] [CrossRef]
- Sturtevant, A.H. GENETIC STUDIES ON DROSOPHILA SIMULANS. I. INTRODUCTION. HYBRIDS WITH DROSOPHILA MELANOGASTER. Genetics 1920, 5, 488–500. [Google Scholar] [CrossRef]
- Tantawy, A.O.; Vetukhiv, M.O. Effects of Size on Fecundity, Longevity and Viability in Populations of Drosophila pseudoobscura. The American Naturalist 1960, 94, 395–403. [Google Scholar] [CrossRef]
- Tracey Chapman & Linda Partridge. Female fitness in Drosophila melanogaster: An interaction between the effect of nutrition and of encounter rate with males. Proceedings of the Royal Society of London. Series B: Biological Sciences 1996, 263, 755–759. [CrossRef]
- Van Wielendaele, P.; Badisco, L.; Vanden Broeck, J. Neuropeptidergic regulation of reproduction in insects. General and Comparative Endocrinology 2013, 188, 23–34. [Google Scholar] [CrossRef]
- Veenstra, J.A. What the loss of the hormone neuroparsin in the melanogaster subgroup of Drosophila can tell us about its function. Insect Biochemistry and Molecular Biology 2010, 40, 354–361. [Google Scholar] [CrossRef]
- Walker, S.J.; Corrales-Carvajal, V.M.; Ribeiro, C. Postmating Circuitry Modulates Salt Taste Processing to Increase Reproductive Output in Drosophila. Current Biology 2015, 25, 2621–2630. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, G.; Miao, C.; Zhao, M.; Wang, B.; Guo, X. Nonanal modulates oviposition preference in female Helicoverpa assulta (Lepidoptera: Noctuidae) via the activation of peripheral neurons. Pest Management Science 2020, 76, 3159–3167. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, K.; Forknall, N.; Parekh, R.; Dickson, B.J. Circuit and Behavioral Mechanisms of Sexual Rejection by Drosophila Females. Current Biology 2020, 30, 3749–3760e3. [Google Scholar] [CrossRef]
- Wang, F.; Wang, K.; Forknall, N.; Patrick, C.; Yang, T.; Parekh, R.; Bock, D.; Dickson, B.J. Neural circuitry linking mating and egg laying in Drosophila females. Nature 2020, 579, 101–105. [Google Scholar] [CrossRef]
- Wang, K.; Wang, F.; Forknall, N.; Yang, T.; Patrick, C.; Parekh, R.; Dickson, B.J. Neural circuit mechanisms of sexual receptivity in Drosophila females. Nature 2021, 589, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Waterbury, J.A.; Jackson, L.L.; Schedl, P. Analysis of the Doublesex Female Protein in Drosophila melanogaster: Role in Sexual Differentiation and Behavior and Dependence on Intersex. Genetics 1999, 152, 1653–1667. [Google Scholar] [CrossRef] [PubMed]
- Wheelock, G.D.; Petzel, D.H.; Gillett, J.D.; Beyenbach, K.W.; Hagedorn, H.H. Evidence for hormonal control of diuresis after a blood meal in the mosquito Aedes aegypti. Archives of Insect Biochemistry and Physiology 1988, 7, 75–89. [Google Scholar] [CrossRef]
- White, M.A.; Chen, D.S.; Wolfner, M.F. She’s got nerve: Roles of octopamine in insect female reproduction. Journal of Neurogenetics 2021, 35, 132–153. [Google Scholar] [CrossRef]
- Wieschaus, E. Wieschaus, E. A Combined Genetic and Mosaic Approach to the Study of Oogenesis in Drosophila. In O. Siddiqi 1980, P. Babu, L.M. Hall, & J. C. Hall (Eds.), Development and Neurobiology of Drosophila (pp. 85–94). Springer US. [CrossRef]
- Wigby, S.; Chapman, T. Sex Peptide Causes Mating Costs in Female Drosophila melanogaster. Current Biology 2005, 15, 316–321. [Google Scholar] [CrossRef]
- Williams, G.C. Natural Selection, the Costs of Reproduction, and a Refinement of Lack’s Principle. The American Naturalist 1966, 100, 687–690. [Google Scholar] [CrossRef]
- Williams, T.D. Mechanisms Underlying the Costs of Egg Production. BioScience 2005, 55, 39. [Google Scholar] [CrossRef]
- Wong, A.; Albright, S.N.; Wolfner, M.F. Evidence for structural constraint on ovulin, a rapidly evolving Drosophila melanogaster seminal protein. Proceedings of the National Academy of Sciences 2006, 103, 18644–18649. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.; Lange, A.B. Octopamine modulates a central pattern generator associated with egg-laying in the locust, Locusta migratoria. Journal of Insect Physiology 2014, 63, 1–8. [Google Scholar] [CrossRef]
- Wu, C.-L.; Fu, T.-F.; Chou, Y.-Y.; Yeh, S.-R. A Single Pair of Neurons Modulates Egg-Laying Decisions in Drosophila. PLOS ONE 2015, 10, e0121335. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.S.; Januszewski, M.; Lu, Z.; Takemura, S.; Hayworth, K.J.; Huang, G.; Shinomiya, K.; Maitin-Shepard, J.; Ackerman, D.; Berg, S.; Blakely, T.; Bogovic, J.; Clements, J.; Dolafi, T.; Hubbard, P.; Kainmueller, D.; Katz, W.; Kawase, T.; Khairy, K.A.; … Plaza, S.M. A Connectome of the Adult Drosophila Central Brain [Preprint]. Neuroscience. [CrossRef]
- Xu 2020, J.; Tan, A.; Palli, S.R. The function of nuclear receptors in regulation of female reproduction and embryogenesis in the red flour beetle, Tribolium castaneum. Journal of Insect Physiology 2010, 56, 1471–1480. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, D.; Koganezawa, M. Genes and circuits of courtship behaviour in Drosophila males. Nature Reviews Neuroscience 2013, 14, 681–692. [Google Scholar] [CrossRef]
- Yang, C. -h. ; Belawat, P.; Hafen, E.; Jan, L.Y.; Jan, Y.-N. Drosophila Egg-Laying Site Selection as a System to Study Simple Decision-Making Processes. Science 2008, 319, 1679–1683. [Google Scholar] [CrossRef]
- Yapici, N.; Kim, Y.-J.; Ribeiro, C.; Dickson, B.J. A receptor that mediates the post-mating switch in Drosophila reproductive behaviour. Nature 2008, 451, 33–37. [Google Scholar] [CrossRef]
- Zhang, C.; Kim, A.J.; Rivera-Perez, C.; Noriega, F.G.; Kim, Y.-J. The insect somatostatin pathway gates vitellogenesis progression during reproductive maturation and the post-mating response. Nature Communications 2022, 13, 969. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, B.; Roy, S.; Saha, T.T.; Kokoza, V.A.; Li, M.; Raikhel, A.S. MicroRNA-309 targets the Homeobox gene SIX4 and controls ovarian development in the mosquito Aedes aegypti. Proceedings of the National Academy of Sciences 2016, 113. [Google Scholar] [CrossRef]
- Zhou, C.; Pan, Y.; Robinett, C.C.; Meissner, G.W.; Baker, B.S. Central Brain Neurons Expressing doublesex Regulate Female Receptivity in Drosophila. Neuron 2014, 83, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Zhu, E.Y.; Guntur, A.R.; He, R.; Stern, U.; Yang, C.-H. Egg-Laying Demand Induces Aversion of UV Light in Drosophila Females. Current Biology 2014, 24, 2797–2804. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



