Submitted:
03 June 2023
Posted:
05 June 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
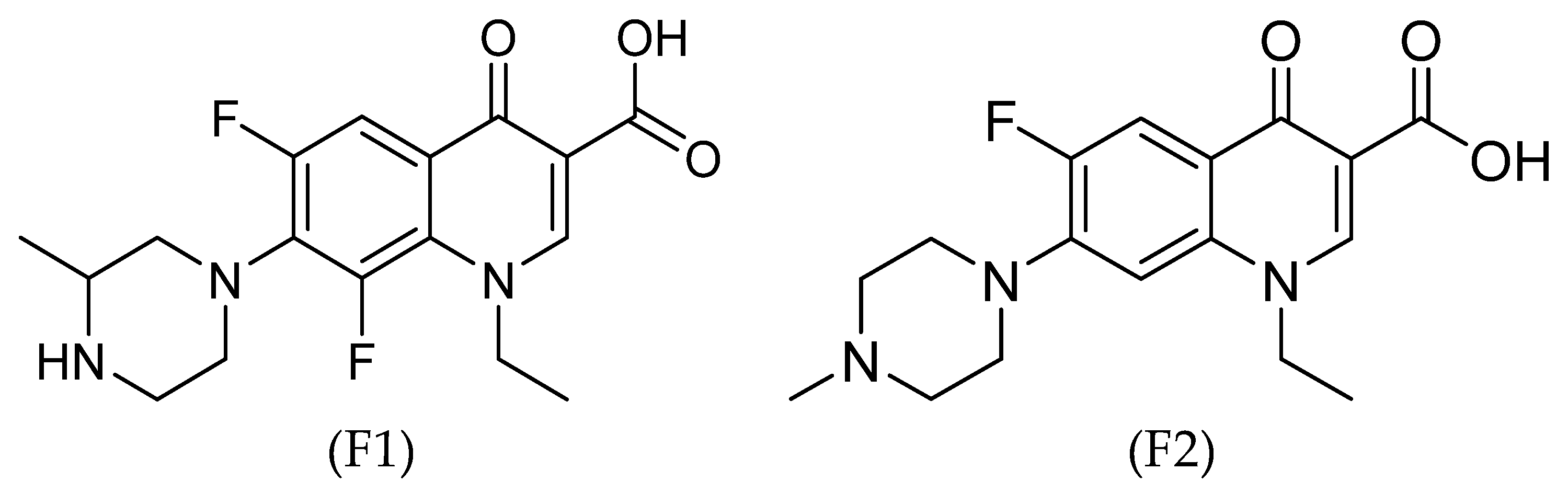
2.1. Materials
2.2. Synthetic Methods
2.3. Characterization Methods
2.4. Antimicrobial Assays
| Microbe | Abbreviation |
| (A) Gram-positive bacterial strains | |
| Bacillus subtilis | B. subtilis |
| Streptococcus pneumoniae | S. pneumoniae |
| Staphylococcus aureus | S. aureus |
| (B) Gram-negative bacterial strains | |
| Escherichia coli | E. coli |
| Pseudomonas aeruginosa | P. aeruginosa |
| (C) Fungal strains | |
| Aspergillus niger | A. niger |
| Penicillium sp. | - |
| Candida albicans | C. albicans |
3. Results and Discussion
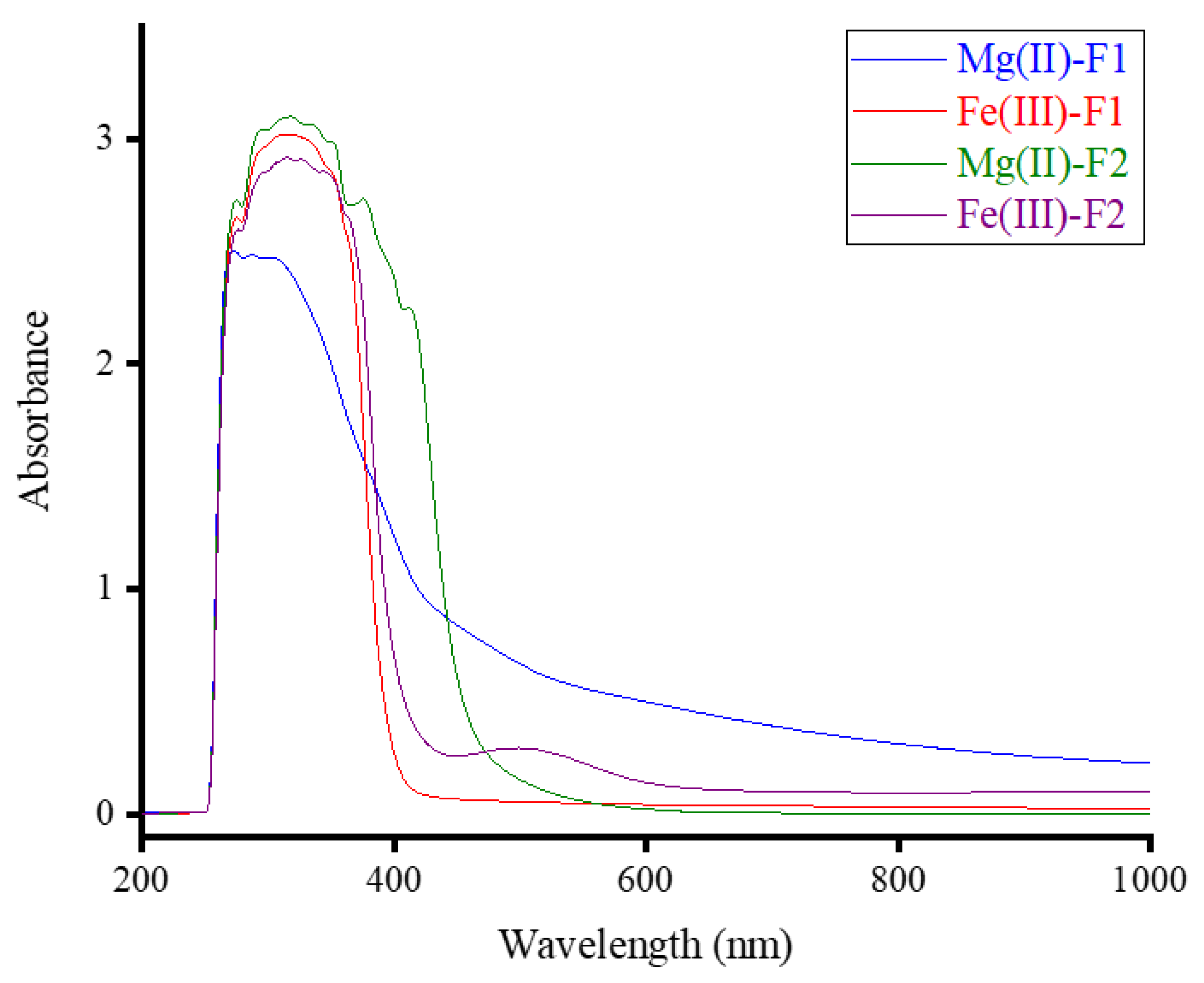
3.1. Compositions and UV-Visible Spectra
| Complex | Elemental results (%) Found (Calculated) |
|||||
|---|---|---|---|---|---|---|
| C | H | N | Cl | H2O | Metal | |
| Mg(II)-F1 | 44.06 (43.95) |
5.35 (5.17) |
8.93 (9.05) |
7.50 (7.64) |
11.45 (11.63) |
5.46 (5.24) |
| Ca(II)-F1 | 40.73 (40.97) |
5.10 (5.22) |
8.59 (8.44) |
7.30 (7.12) |
14.62 (14.46) |
7.94 (8.05) |
| Zn(II)-F1 | 43.60 (43.48) |
4.18 (4.26) |
9.10 (8.95) |
7.43 (7.56) |
4.00 (3.84) |
13.70 (13.93) |
| Fe(III)-F1 | 34.96 (34.84) |
4.31 (4.61) |
7.30 (7.17) |
17.94 (18.16) |
12.09 (12.30) |
9.43 (9.54) |
| Mg(II)-F2 | 45.60 (45.73) |
5.76 (5.60) |
9.57 (9.41) |
7.80 (7.95) |
12.32 (12.10) |
5.70 (5.45) |
| Ca(II)-F2 | 42.39 (42.51) |
5.99 (5.83) |
8.86 (8.75) |
7.56 (7.39) |
14.77 (15.00) |
8.26 (8.35) |
| Zn(II)-F2 | 45.35 (45.21) |
4.79 (4.88) |
9.45 (9.31) |
7.70 (7.86) |
3.92 (3.99) |
14.62 (14.49) |
| Fe(III)-F2 | 35.70 (35.94) |
5.00 (5.11) |
7.22 (7.40) |
18.95 (18.74) |
12.85 (12.69) |
9.97 (9.84) |
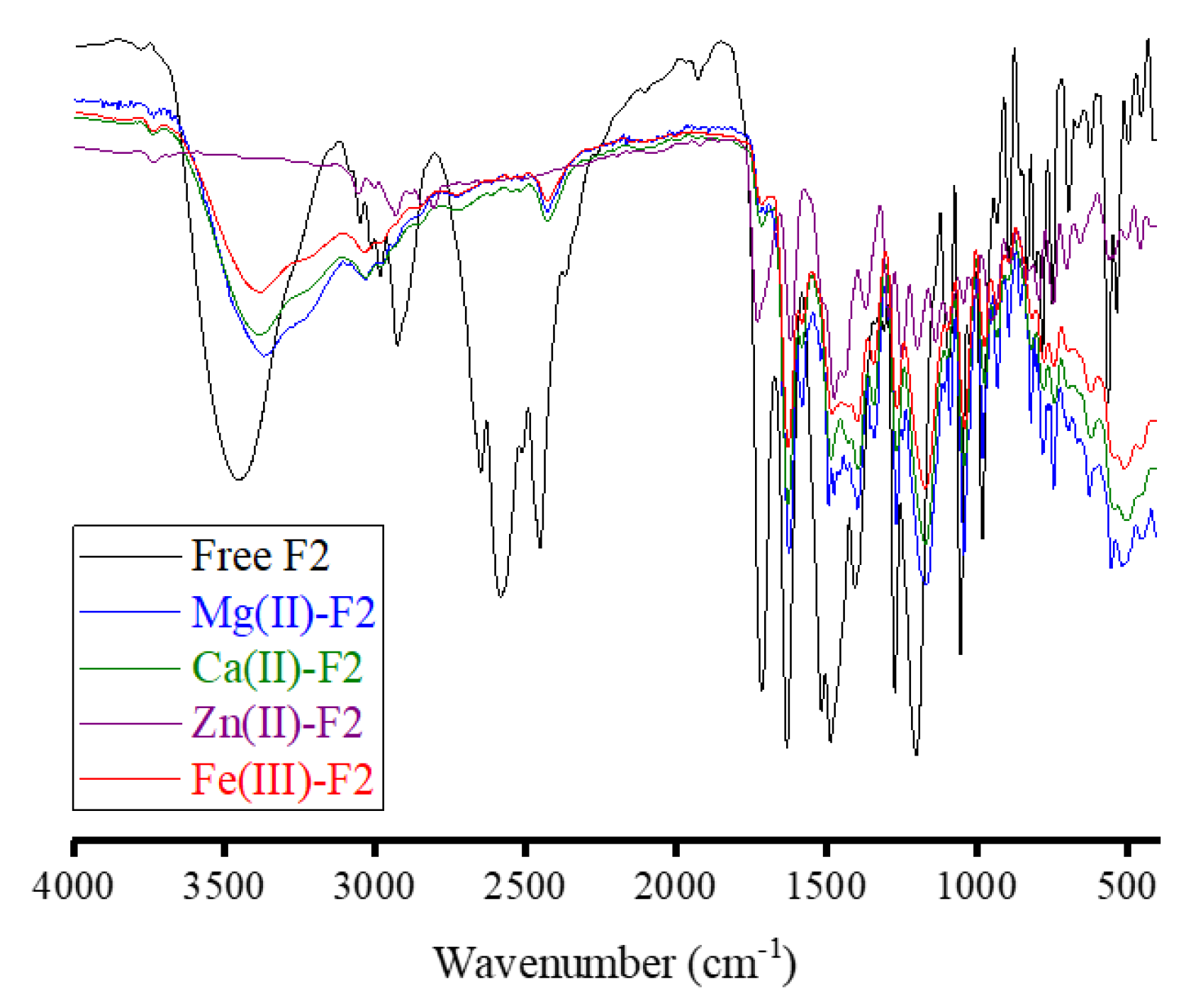
3.2. FT-IR Spectra
3.2.1. Complexes of F1
3.2.2. Complexes of F2
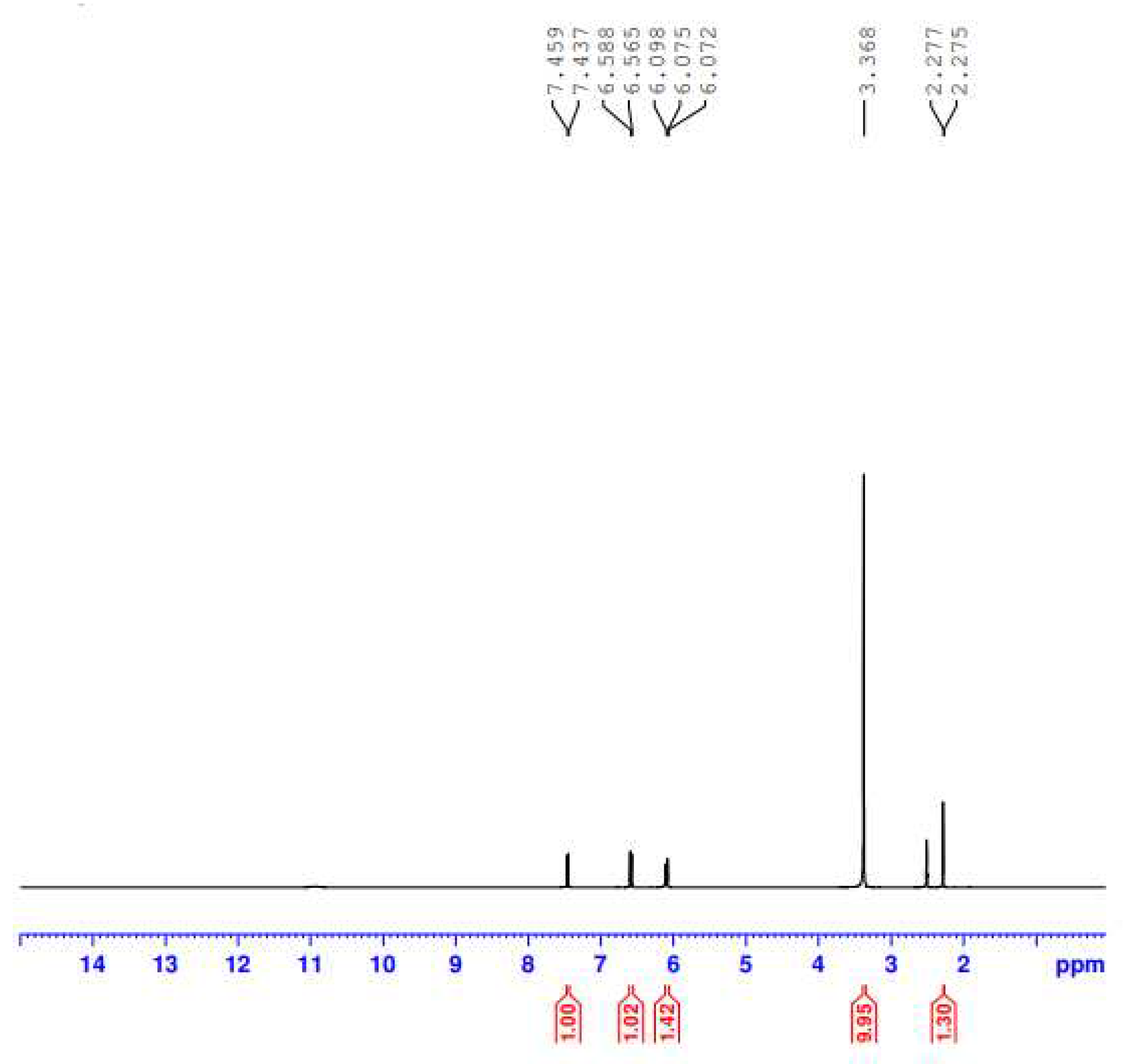
3.3. H NMR Spectral Analysis
| Free F1 | F1-Fe(III) | Assignments |
| 1.323-1.762 | 2.275, 2.277 | δ 3H, -CH3; -CH2CH3 δ 3H, -CH3; attached to piperazine ring |
| 3.546-4.206 | 6.072, 6.075, 6.98 | δ 7H, piperazine ring protons |
| 4.632-4.685 | 6.565, 6.588 | δ 2H, -CH2; -CH2CH3 |
| 7.796, 8.902 | 7.437, 7.459 | δ H, -CH; benzene ring and pyridine ring |
| 10.198-10.315 | - | δ H, -NH2+; piperazine ring |
| 11.783 | - | δ H, -COOH |
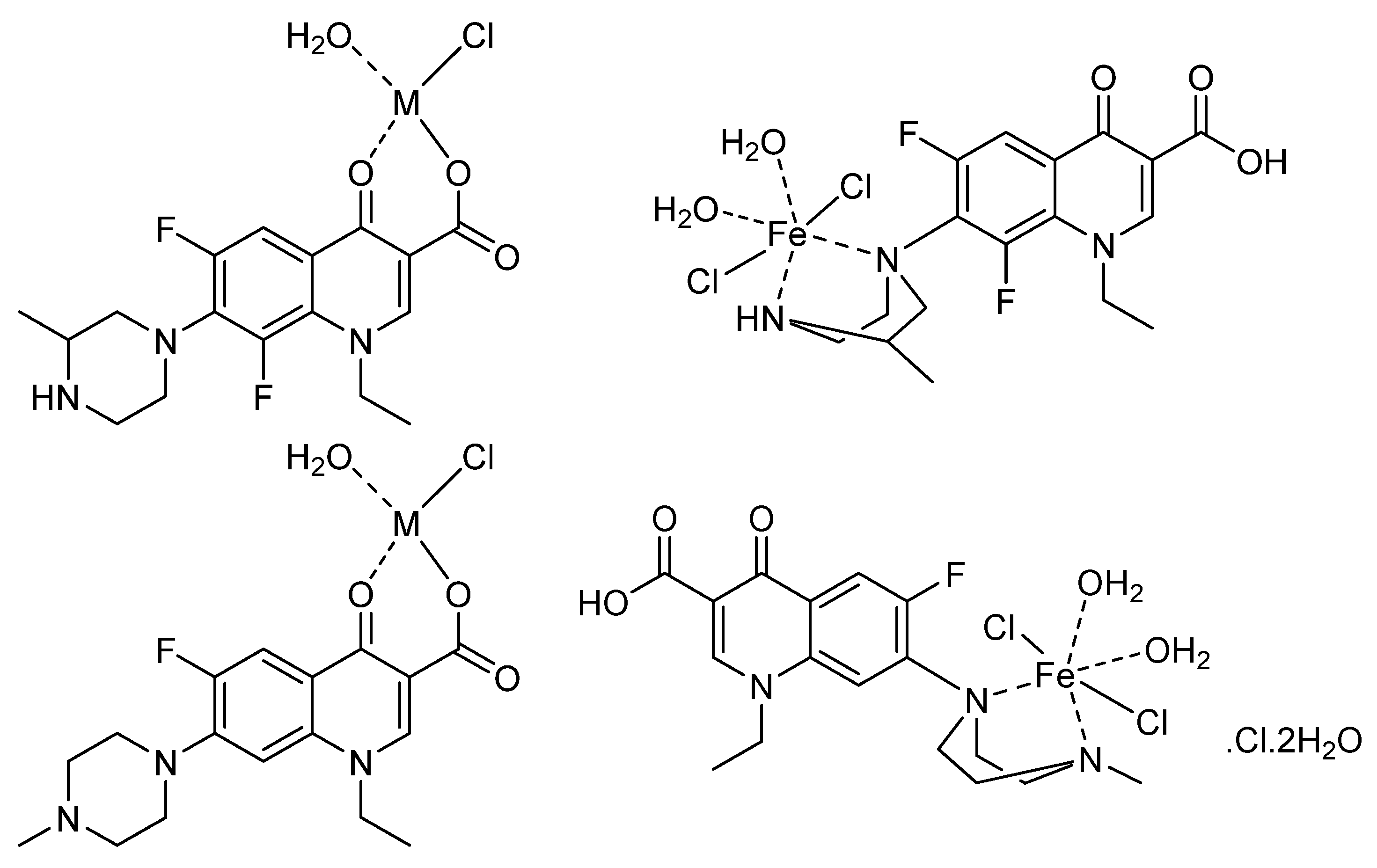
3.4. Proposed Structures
3.5. XRD, SEM, and TEM Results
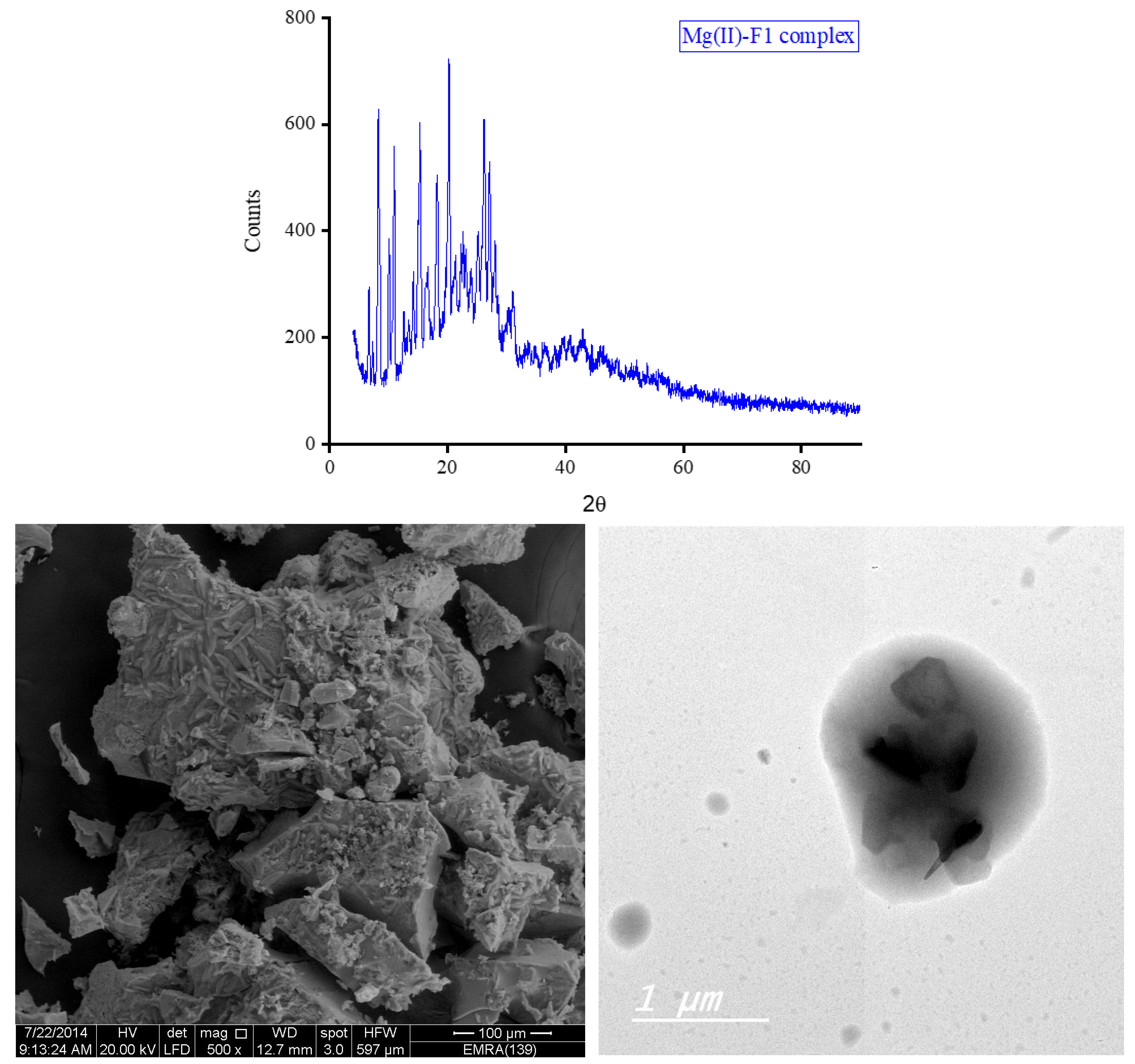
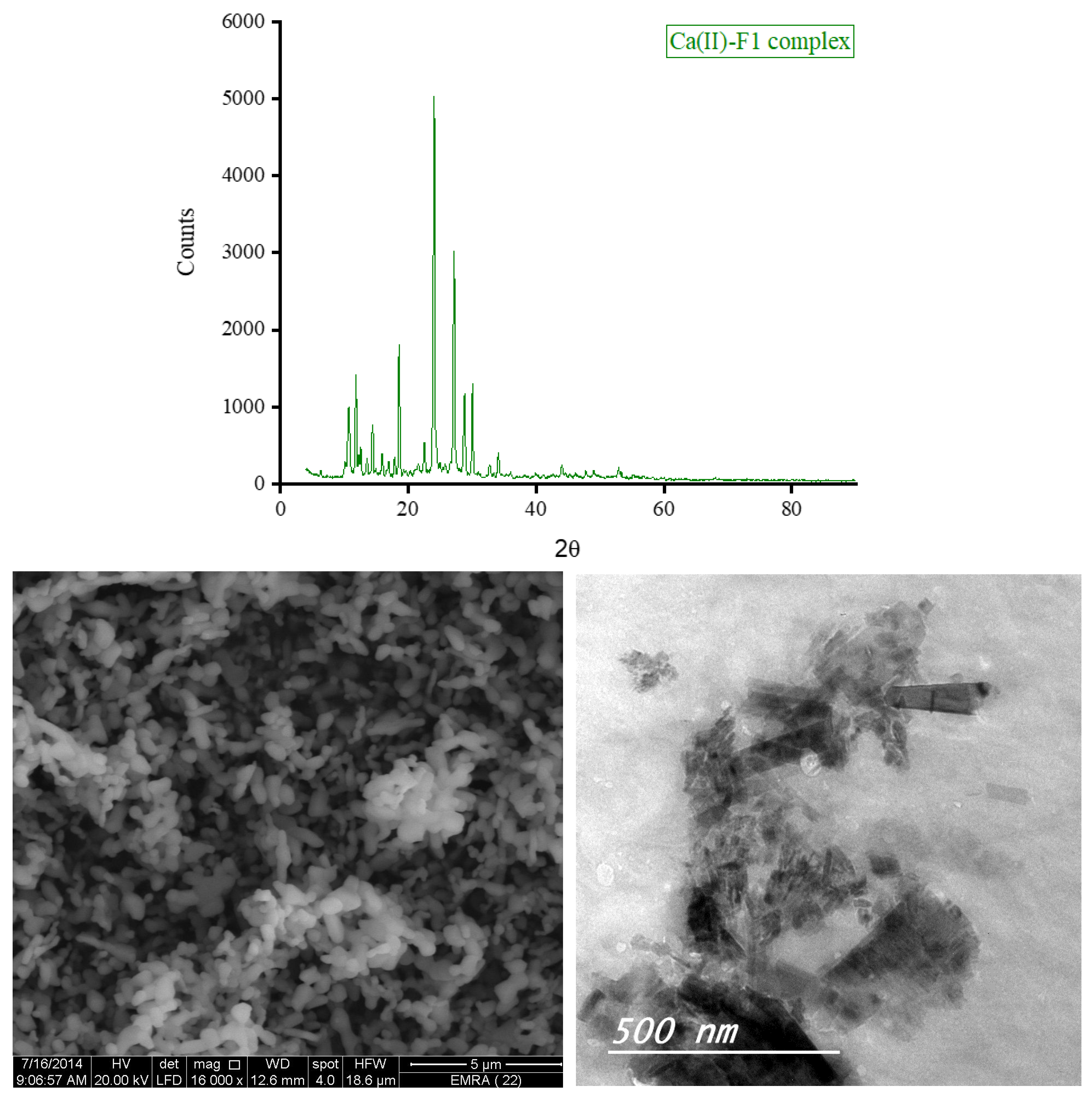
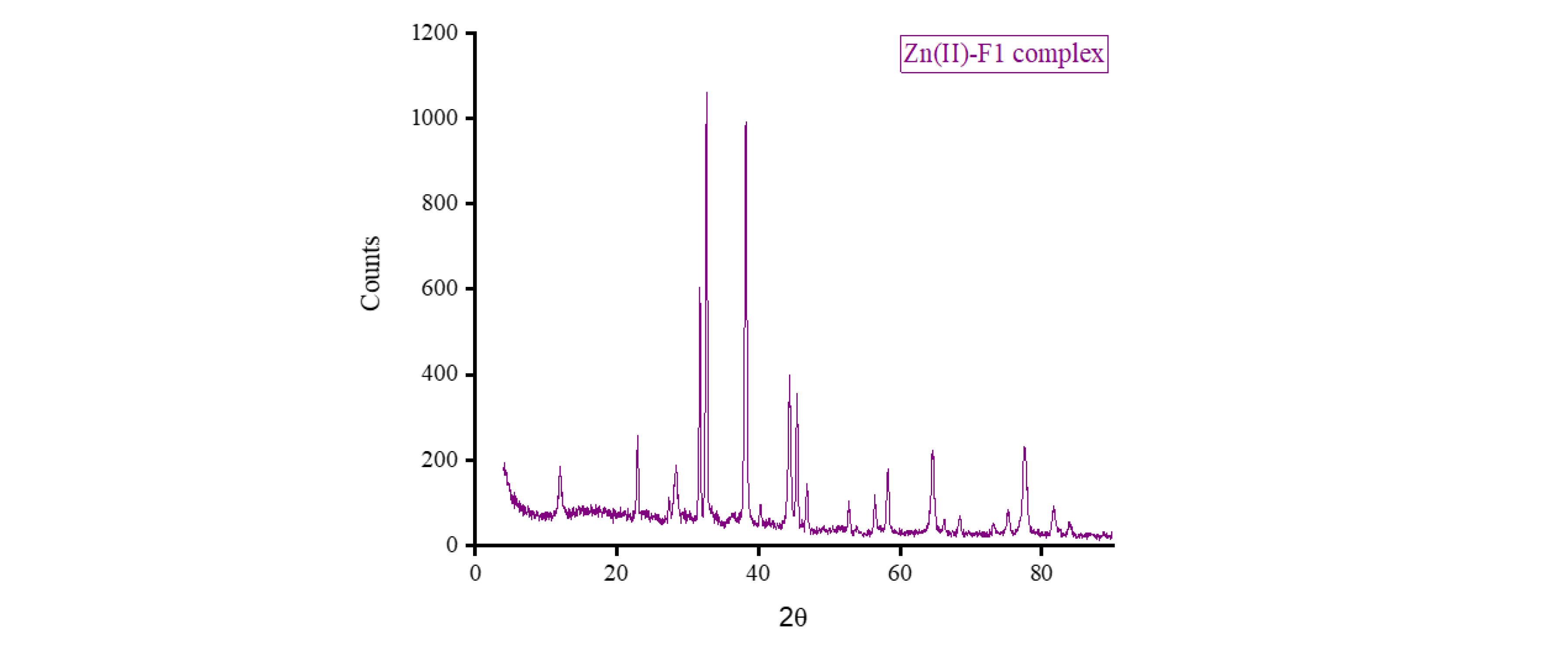

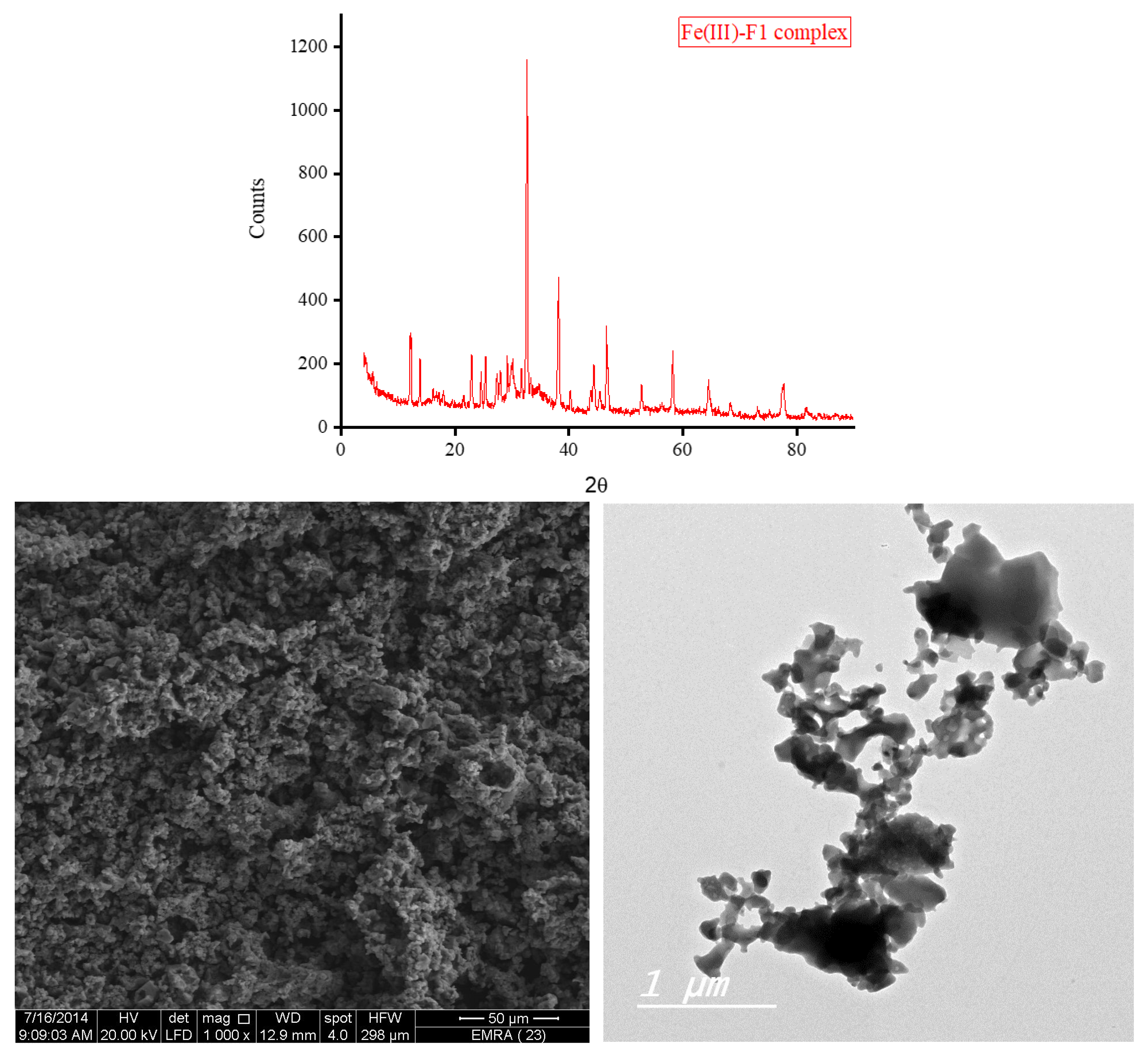
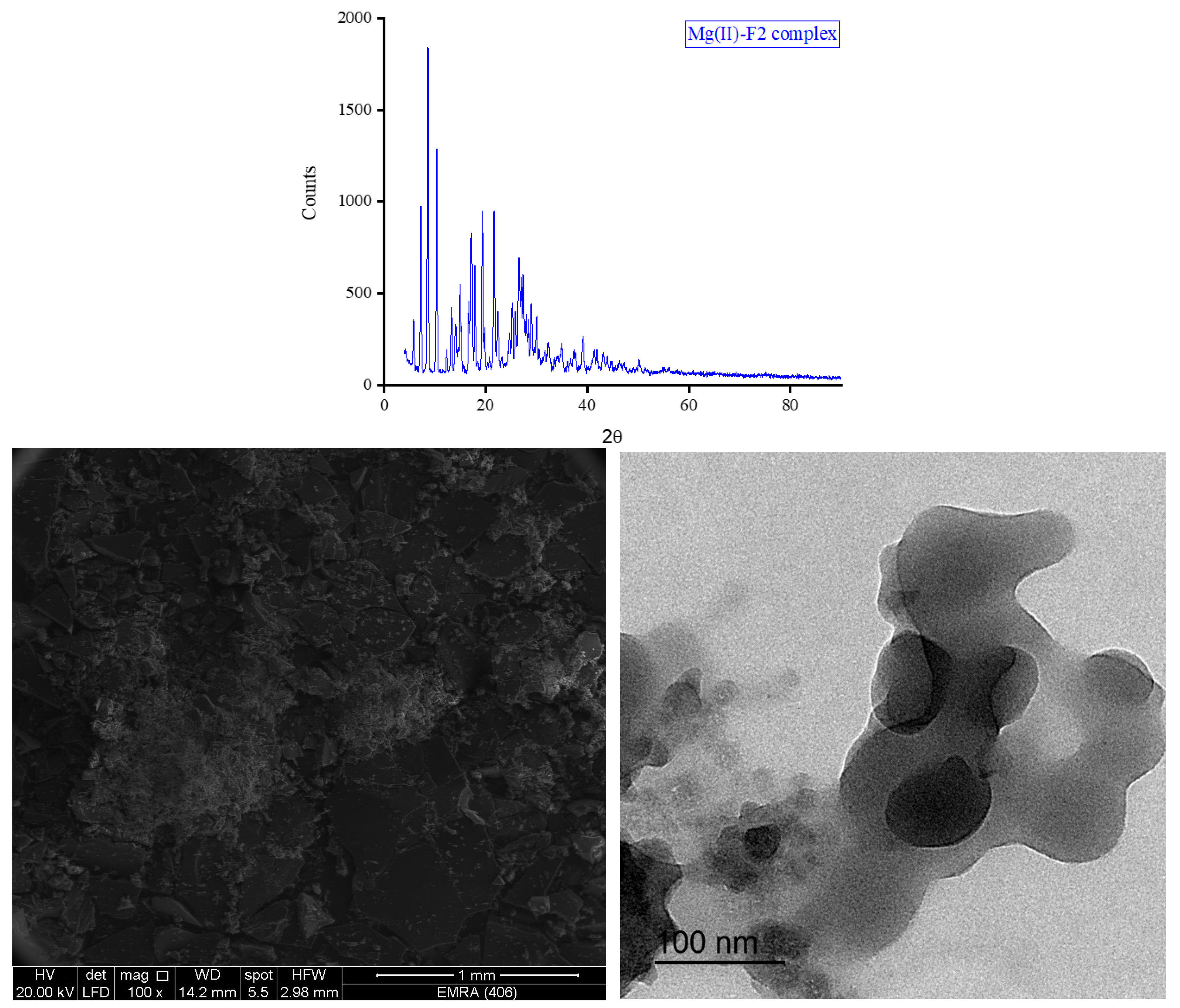
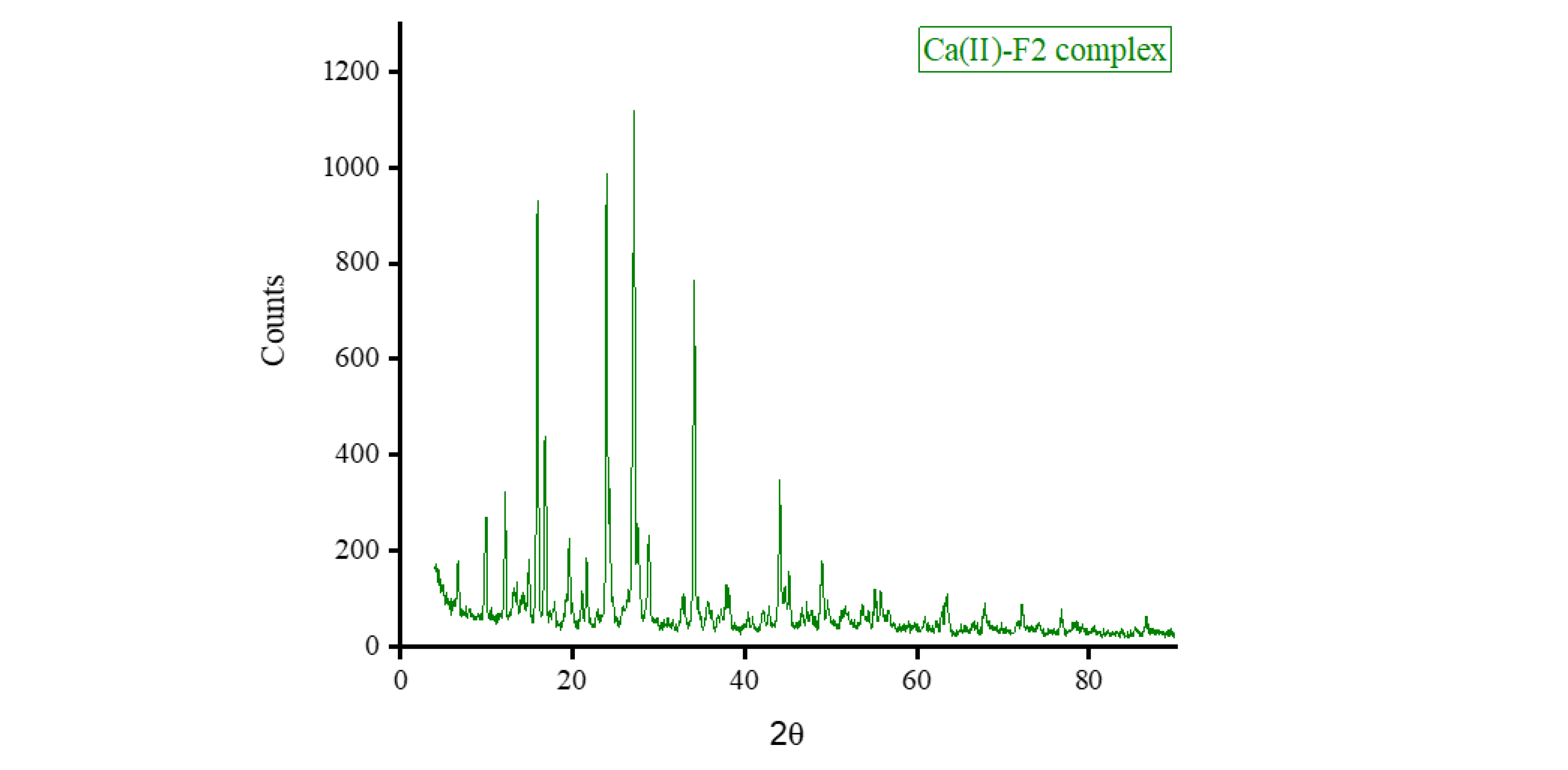

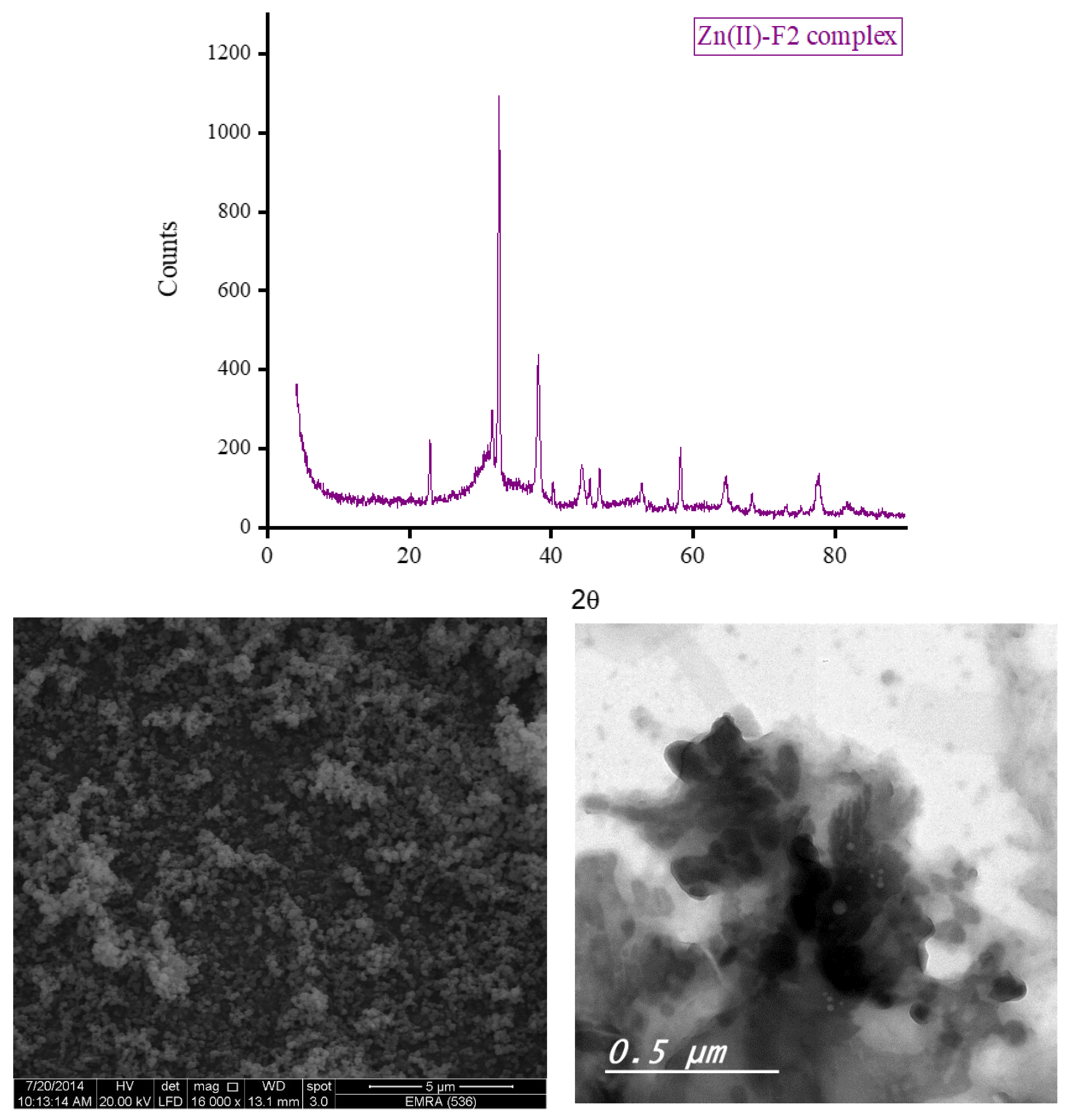
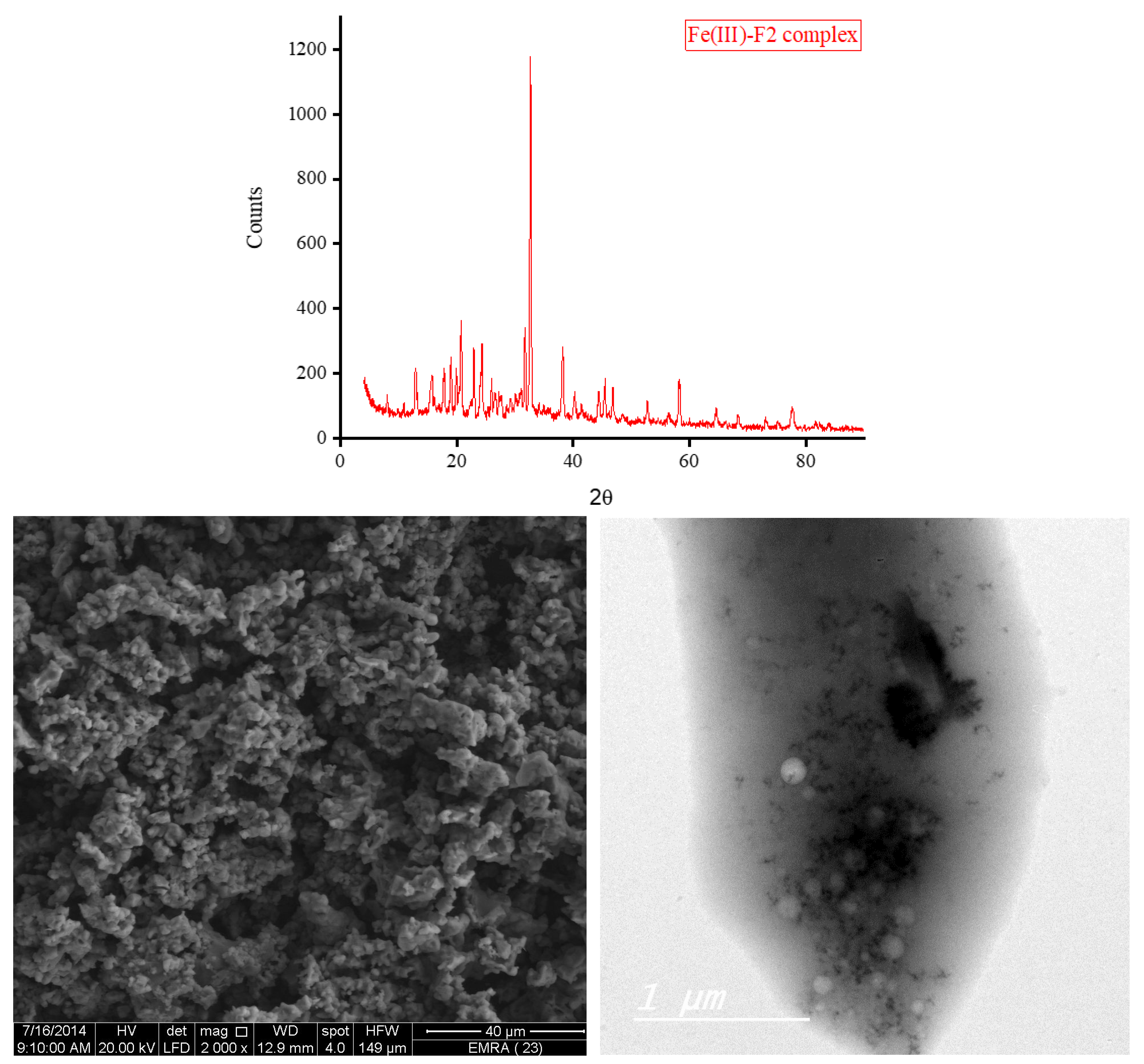
3.6. Biological Screening
3.6.1. Antibacterial Activity
3.6.2. Antifungal Activity
4. Conclusions
Funding
Acknowledgments
References
- A.C. Tella, J.A. A.C. Tella, J.A. Obaleye, M.D. Olawale, J.M.V. Ngororabanga, A.S. Ogunlaja, S.A. Bourned, C.R. Chimie 22 (1) (2019) 3-12.
- G.L. Eichhorn, L.G. G.L. Eichhorn, L.G. Marzilli, Advances in Inorganic Biochemistry Models in Inorganic Chemistry, PTR Prentice-Hall, Inc, New Jersey, 1994.
- M.N. Hughes, The Inorganic Chemistry of Biological Processes, 2nd ed., Wiley, Chichester [England], 1984.
- E. Alessio, Bioinorganic Medicinal Chemistry, Wiley-VCH Verlag GmbH and Co. KGaA, 2011.
- F. Trudu, F. F. Trudu, F. Amato, P. Vaňhara, T. Pivetta, E.M. Peña-Méndez, J. Havel, J. Appl. Biomed. 13 (2) (2015) 79.
- U. Singh, A.M. U. Singh, A.M. Malla, I.A. Bhat, A. Ahmad, M.N. Bukhari, S. Bhat, S. Anayu-tullah, A. A. Hashmi, Microb. Pathog. 93 (2016) 172.
- P.P. Netalkar, S.P. P.P. Netalkar, S.P. Netalkar, V.K. Revankar, Polyhedron 100 (2015) 215.
- M.A. Ragheb, M.A. M.A. Ragheb, M.A. Eldesouki, M.S. Mohamed, Spectrochim. Acta A 138 (2015) 585.
- S.P. Fricker, Dalton transactions, 43 (2007) 4903.
- A. Jurowska, K. A. Jurowska, K. Jurowski, J. Szklarzewicz, B. Buszewski, T. Kalenik, W. Piekoszewski, Cur. Med. Chem., 23(29) (2016) 3322.
- E.K. Efthimiadou, H. Thomadaki, Y. Sanakis, C.P. Raptopoulou, N. Katsaros, A. Scorilas, A. Karaliota, G. Psomas, J. Inorg. Biochem. 101 (2007) 64.
- L.M.M. Vieira, M.V. de Almeida, M.C.S. Lourenço, F.A.F.M. Bezerra, A.P.S. Fontes, Eur. J. Med. Chem., 44 (2009) 4107-4111.
- S.A. Sadeek, W.H. S.A. Sadeek, W.H. El-Shwiniy, J. Mol. Struct., 98 (2010) 130.
- H.F. Abd El-Halim, G.G. H.F. Abd El-Halim, G.G. Mohamed, M.M.I. El-Dessouky, W.H. Mahmoud, Spectrochim. Acta A, 82 (2011) 8.
- W. Qi, J. W. Qi, J. Huang, Z. An, Acta Crystallogr. 64 (2008) m302.
- P. Drevenšek, J. P. Drevenšek, J. Košmrlj, G. Giester, T. Skauge, E. Sletten, K. Sepčić, I. Turel, J. Inorg. Biochem. 100 (2006) 1755.
- Bauer, A.W.; Kirby, W.M.M.; Sherris, J.C.; Turck, M. Antibiotic Susceptibility Testing by a Standardized Single Disk Method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- J.J. Biemer, Antimicrobial susceptibility testing by the Kirby-Bauer disc diffu-sion method, Ann. Clin. Lab. Sci. 3 (1973) 135-140.
- M.C. Serrano, M. Ramírez, D. Morilla, A. Valverde, M. Chávez, A. Espinel-Ingroff, R. Claro, A. Fernández, C. Almeida, E. Martín-Mazuelos, A comparative study of the disc diffusion method with the broth microdilution and Etest methods for voriconazole susceptibility testing of Aspergillus spp., J. An-timicrob. Chemo-ther. 53 (2004) 739-742.
- Allan, J.R.; Baird, N.D.; Kassyk, A.L. Some first row transition metal complexes of nicotinamide and nicotinic acid. J. Therm. Anal. Calorim. 1979, 16, 79–90. [Google Scholar] [CrossRef]
- Öztirk, .F.; Şekerci, M.; Özdemir, E. Preparation of complexes of 1-amino-6,7-O-cyclohexylidene-4-azaheptane with transition metal acetates. Russ. J. Gen. Chem. 2006, 76, 33–36. [Google Scholar] [CrossRef]
- Refat, M.S.; El-Sayed, M.Y.; Hassan, R.F. Study of the chemical structure and the microbial effect of iron(III) metal ions with four consecutive generations of quinolones in a nanometric form for the purpose of increasing the efficacy of antibacterial and antifungal drugs. Appl. Organomet. Chem. 2017, 32, e4195. [Google Scholar] [CrossRef]
- G.B. Deacon, R. Phillips, Relationships between the carbon-oxygen stretching frequencies of carboxylato complexes and the type of carboxylate coordination, Coord. Chem. Rev. 33 (1980) 227-250.
- K. Nakamoto, Infrared Spectra of Inorganic and Coordination Compounds, Wiley Interscience, John Wiley & Sons, New York, NY, USA, 2nd edition, 1970.
- El-Megharbel, S.M.; Hegab, M.S.; Manaaa, E.-S.A.; Al-Humaidi, J.Y.; Refat, M.S. Synthesis and physicochemical characterizations of coordination between palladium(ii) metal ions with floroquinolone drugs as medicinal model against cancer cells: novel metallopharmaceuticals. New J. Chem. 2018, 42, 9709–9719. [Google Scholar] [CrossRef]
- S.M. El-Megharbel, M.A. Hussien, M.S. Refat, In-Situ Copper(II) Complexes of Some Quinolone Drug Ligands Were Discussed for Their Molecular Structures: Syn-thesis in Binary Solvent, Journal of Computational and Theoretical Nanoscience 14 (1) (2017) 561-576.
- M.W. Nassar, K.A.M. Attia, R.A. Said, A. El-Olemy, M.A. Hasan, Second De-rivative Synchronous Spectrofluorimetric Determination of Lomefloxacin Hydrochlo-ride in Presence of Its Decarboxylated Degradation Product INTRODUCTION, Ijppr. Human 11 (1) (2017) 397-418.

| Drug | Molar ratio (metal to ligand) |
Complexes | References |
|---|---|---|---|
| F1 | 1:1 | [UO2F1(H2O)2](NO3)2 [ThF1(H2O)4]Cl4 [ZnF1(H2O)4]Cl2 [CuF1(H2O)4]Cl2·2H2O [NiF1(H2O)4]Cl2·H2O [CoF1(H2O)4]Cl2 [FeF1(H2O)4]Cl3·H2O [MnF1(H2O)4]Cl2 [CrF1(H2O)4]Cl3 |
[13-15] |
| 1:2 | [ZrO(F1)2Cl]Cl·15H2O [Y(F1)2Cl2]Cl·12H2O |
||
| 1:3 | [UO2(F1)3](NO3)2·4H2O [Bi(F1)3(H2O)2] |
||
| F2 | 1:1 | - | [13, 16] |
| 1:2 | [Pt(F2)2] [Zn(F2)2(H2O)]·2H2O |
||
| 1:3 | [Bi(F2)3(H2O)2] |
| Sample | Gram-positive bacteria strains | Gram-negative bacteria strains | |||
|---|---|---|---|---|---|
| B. subtilis | S. pneumoniae | S. aureus | E. coli | P. aeruginosa | |
| DMSO (− control) | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Streptomycin (+ control) | 18.0 | 17.0 | 20.0 | 22.0 | 27.0 |
| Mg(II)-F1 | 6.0 | 1.0 | 2.0 | 5.0 | 4.0 |
| Ca(II)-F1 | 5.0 | 3.0 | 8.0 | 2.0 | 8.0 |
| Zn(II)-F1 | 9.0 | 5.0 | 12.0 | 7.0 | 3.0 |
| Fe(III)-F1 | 16.0 | 19.0 | 21.0 | 19.0 | 24.0 |
| Mg(II)-F2 | 18.0 | 20.0 | 17.0 | 20.0 | 18.0 |
| Ca(II)-F2 | 10.0 | 15.0 | 16.0 | 14.0 | 19.0 |
| Zn(II)-F2 | 8.0 | 11.0 | 18.0 | 20.0 | 24.0 |
| Fe(III)-F2 | 4.0 | 13.0 | 5.0 | 13.0 | 14.0 |
| Sample | Fungal strains | ||
|---|---|---|---|
| A. niger | Penicillium sp. | C. albicans | |
| DMSO (− control) | 0.0 | 0.0 | 0.0 |
| Ketoconazole (+ control) | 18.0 | 21.0 | 21.0 |
| Mg(II)-F1 | 2.0 | 3.0 | 1.0 |
| Ca(II)-F1 | 5.0 | 7.0 | 1.0 |
| Zn(II)-F1 | 5.0 | 9.0 | 7.0 |
| Fe(II)-F1 | 19.0 | 23.0 | 21.0 |
| Mg(II)-F2 | 11.0 | 17.0 | 18.0 |
| Ca(II)-F2 | 9.0 | 11.0 | 15.0 |
| Zn(II)-F2 | 15.0 | 18.0 | 17.0 |
| Fe(II)-F2 | 11.0 | 13.0 | 12.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




