Submitted:
23 May 2023
Posted:
29 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Water-Holding Capacity
2.3. Lipid Oxidation
2.4. Total Volatile Basic Nitrogen (TVB-N)
2.5. Protein Oxidation and Denaturation
2.6. Freeze Substitution Histological Observation
2.7. Comprehensive Two-Dimensional Gas Chromatography Time-of-Flight Mass Spectrometry (GC×GC-TOFMS)
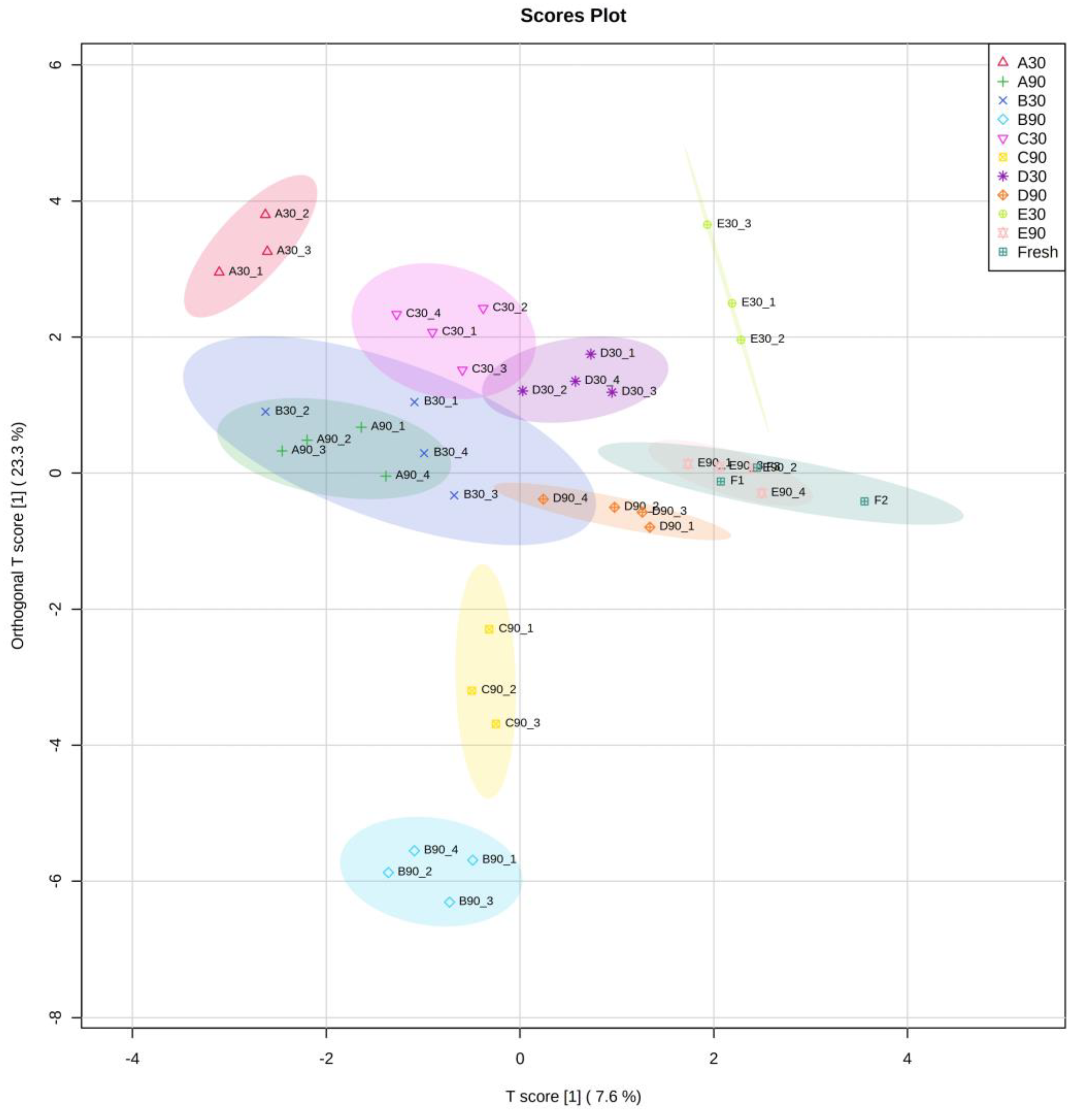
2.8. Data Analysis
3. Results and Discussion
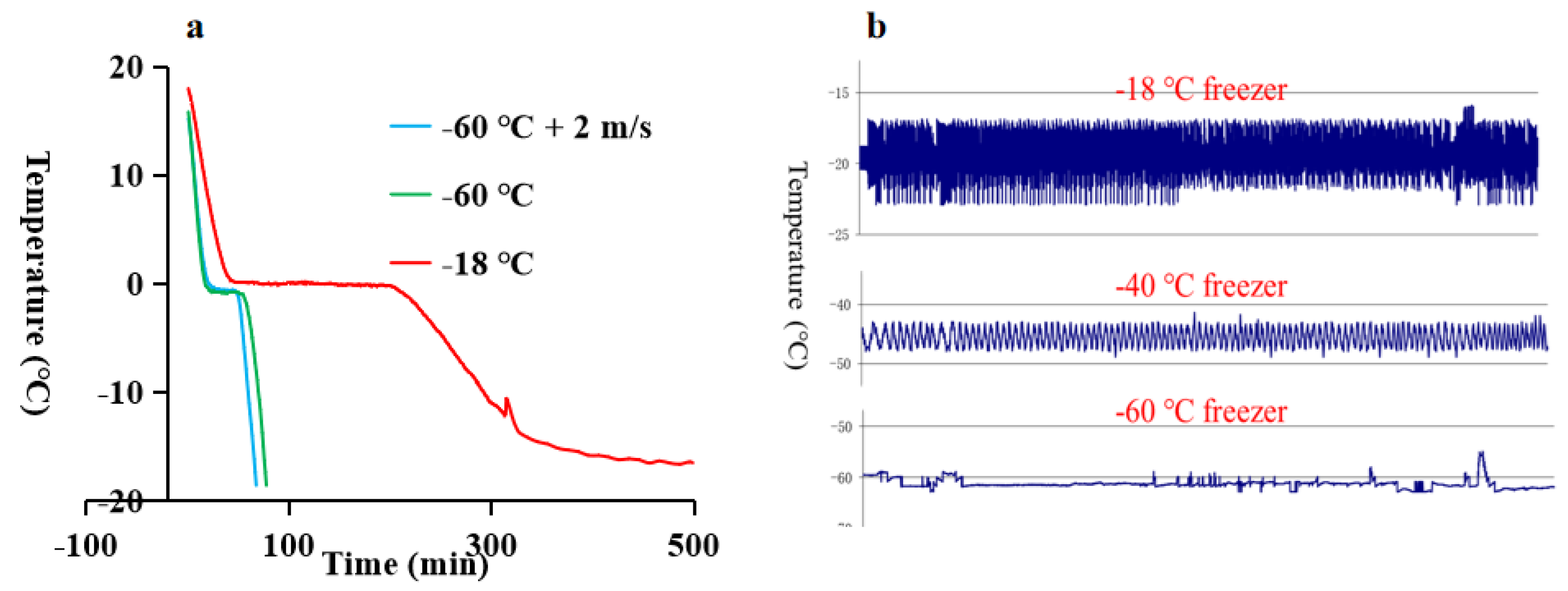
3.1. The Freezing Process and Frozen Storage
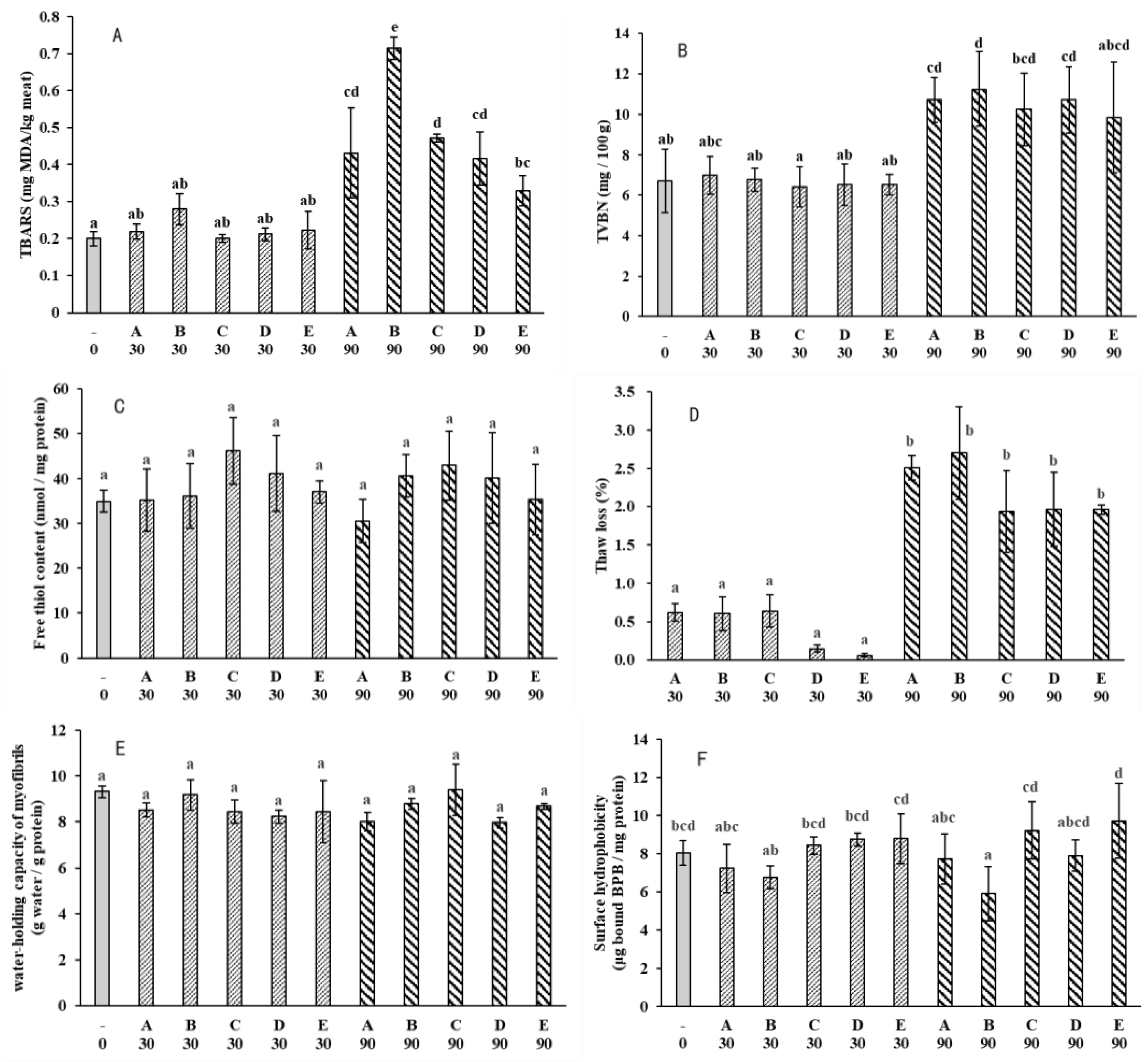
3.2. Water-Holding
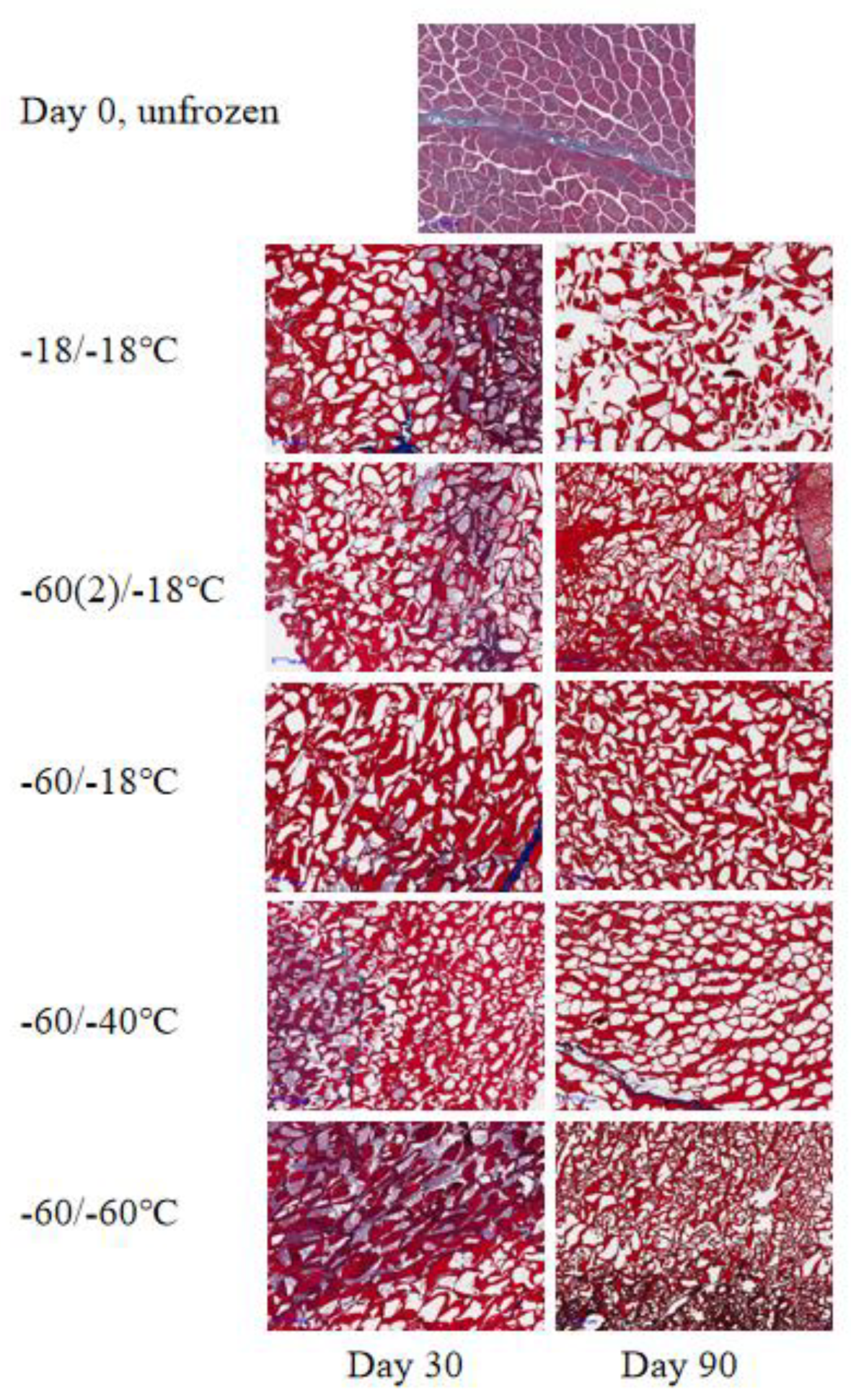
3.3. Microstructure Changes
3.4. Lipid Oxidation
3.5. Protein Changes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fishery Bureau of Ministry of Agriculture of the People’s Republic of China. China Fishery Statistical Yearbook. Beijing, China: China Agriculture Press, 2021.
- Bao, Y., Ertbjerg, P., Estévez, M., Yuan, L., & Gao, R.. Freezing of meat and aquatic food: Underlying mechanisms and implications on protein oxidation[J]. Comprehensive Reviews in Food Science and Food Safety, 2021, 20(6), 5548-5569. [CrossRef]
- Li, X., Qian, S., Song, Y., Guo, Y., Huang, F., Han, D., ... & Blecker, C.. New insights into the mechanism of freeze-induced damage based on ice crystal morphology and exudate proteomics[J]. Food Research International, 2022, 161, 111757. [CrossRef]
- Zhang, Y. , & Ertbjerg, P.. On the origin of thaw loss: Relationship between freezing rate and protein denaturation[M]. Food Chemistry, 2019, 299, 125104. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, N. , & Okazaki, E.. Recent research on factors influencing the quality of frozen seafood[J]. Fisheries Science, 2020, 86, 231–244. [Google Scholar] [CrossRef]
- Tolstorebrov, I. , Eikevik, T. M., & Bantle, M.. Effect of low and ultra-low temperature applications during freezing and frozen storage on quality parameters for fish[J]. International Journal of Refrigeration, 2016, 63, 37–47. [Google Scholar] [CrossRef]
- Hsieh, R. J., & Kinsella, J. E.. Lipoxygenase generation of specific volatile flavor carbonyl compounds in fish tissues[J]. Journal of Agricultural and Food Chemistry, 1989, 37(2), 279-286. [CrossRef]
- Domínguez, R., Pateiro, M., Gagaoua, M., Barba, F. J., Zhang, W., & Lorenzo, J. M.. A comprehensive review on lipid oxidation in meat and meat products[J]. Antioxidants, 2019, 8(10), 429. [CrossRef]
- Dunkel, A. , Steinhaus, M., Kotthoff, M., Nowak, B., Krautwurst, D., Schieberle, P., & Hofmann, T.. Nature’s chemical signatures in human olfaction: a foodborne perspective for future biotechnology[J]. Angewandte Chemie International Edition, 2014, 53, 7124–7143. [Google Scholar] [CrossRef] [PubMed]
- Ratel, J. , & Engel, E.. Determination of benzenic and halogenated volatile organic compounds in animal-derived food products by one-dimensional and comprehensive two-dimensional gas chromatography-mass spectrometry[J]. Journal of Chromatography A, 2009, 1216, 7889–7898. [Google Scholar] [CrossRef]
- Wang, W. , Feng, X., Zhang, D., Li, B., Sun, B., Tian, H., & Liu, Y.. Analysis of volatile compounds in Chinese dry-cured hams by comprehensive two-dimensional gas chromatography with high-resolution time-of-flight mass spectrometry[J]. Meat Science, 2018, 140, 14–25. [Google Scholar] [CrossRef]
- Wang, H. , Zhu, Y., Zhang, J., Wang, X., & Shi, W.. Characteristic volatile compounds in different parts of grass carp by comprehensive two-dimensional gas chromatography/time-of-flight mass spectrometry[J]. International Journal of Food Properties, 2011, 23, 777–796. [Google Scholar] [CrossRef]
- Giri, A. , Khummueng, W., Mercier, F., Kondjoyan, N., Tournayre, P., Meurillon, M.,... Engel, E.. Relevance of two-dimensional gas chromatography and high resolution olfactometry for the parallel determination of heat-induced toxicants and odorants in cooked food[J]. Journal of Chromatography A, 2015, 1388, 217–226. [Google Scholar] [CrossRef]
- Ji, W., Bao, Y., Wang, K., Yin, L. and Zhou, P.. Protein changes in shrimp (Metapenaeus ensis) frozen stored at different temperatures and the relation to water-holding capacity. International Journal of Food Science and Technology, 2021, 56(8), 3924-3937. [CrossRef]
- Bao, Y., & Ertbjerg, P.. Relationship between oxygen concentration, shear force and protein oxidation in modified atmosphere packaged pork[J]. Meat Science, 2015, 110, 174-179. [CrossRef]
- Chelh, I., Gatellier, P., & Santé-Lhoutellier, V. (2006). A simplified procedure for myofibril hydrophobicity determination[J]. Meat Science, 2066, 74(4), 681-683. [CrossRef]
- Bao, Y. , Wang, K., Yang, H., Regenstein, J. M., Ertbjerg, P., & Zhou, P.. Protein degradation of black carp (Mylopharyngodon piceus) muscle during cold storage[J]. Food Chemistry, 2020, 308, 125576. [Google Scholar] [CrossRef]
- Chen, Y. , & Pan, B. S.. Freezing tilapia by airblast and liquid nitrogen – freezing point and freezing rate[J]. International Journal of Food Science and Technology, 1995, 30, 167–173. [Google Scholar] [CrossRef]
- Qu, J. , Zhang, L., Yin, L. a., Liu, J., Sun, Z., & Zhou, P. (2021). Changes in bioactive proteins and serum proteome of human milk under different frozen storage[J]. Food Chemistry, 2021, 352, 129436. [Google Scholar] [CrossRef]
- Huff-Lonergan, E. , & Lonergan, S. M.. Mechanisms of water-holding capacity of meat: The role of postmortem biochemical and structural changes[J]. Meat Science, 2005, 71, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Hamm, R.. Functional properties of the myofibrillar system and their measurements. In P. J. Bechtel (Ed.). Muscle as Food. London, 1986: Academic Press Inc. Food Science and Technology (pp. 135-191).
- Liu, J. , Arner, A., Puolanne, E., & Ertbjerg, P.. On the water-holding of myofibrils: Effect of sarcoplasmic protein denaturation[J]. Meat Science, 2016, 119, 32–40. [Google Scholar] [CrossRef]
- Estévez, M. , Ventanas, S., Heinonen, M., & Puolanne, E.. Protein carbonylation and water-holding capacity of pork subjected to frozen storage: Effect of muscle type, premincing, and packaging[J]. Journal of Agricultural and Food Chemistry, 2011, 59, 5435–5443. [Google Scholar] [CrossRef] [PubMed]
- Offer, G. , & Knight, P.. Structural basis of water-holding in meat. Part 1: General principles and water uptake in meat processing. London, 1988: Elsevier Applied Science. In R. Lawrie (Ed.), Developments in Meat Science, Vol 4 (pp. 63-171).
- Hughes, J. , Oiseth, S., Purslow, P., & Warner, R.. A structural approach to understanding the interactions between color, water-holding capacity and tenderness[J]. Meat Science, 2014, 98, 520–532. [Google Scholar] [CrossRef]
- Cai, L., Wan, J., Li, X., & Li, J.. Effects of different thawing methods on physicochemical properties and structure of largemouth bass (Micropterus salmoides)[J]. Journal of Food Science, 2020, 85(3), 582-591. [CrossRef]
- Leygonie, C., Britz, T. J., & Hoffman, L. C.. Impact of freezing and thawing on the quality of meat[J]. Meat Science, 2012, 91(2), 93-98. [CrossRef]
- Lv, Y., & Xie, J.. Quality of cuttlefish as affected by different thawing methods[J]. International Journal of Food Properties, 2022, 25(1), 33-52. [CrossRef]
- Ngapo, T. M., Babare, I. H., Reynolds, J., & Mawson, R. F.. Freezing rate and frozen storage effects on the ultrastructure of samples of pork[J]. Meat Science, 1999, 53(3), 159-168. [CrossRef]
- Vilgis, T. A. . Soft matter food physics—the physics of food and cooking[J]. Reports on Progress in Physics, 2015, 78, 124602. [Google Scholar] [CrossRef]
- Baron, C. P. , Kjaersgard, I. V. H., Jessen, F., & Jacobsen, C. (2007). Protein and lipid oxidation during frozen storage of rainbow trout (Oncorhynchus mykiss)[J]. Journal of Agricultural and Food Chemistry, 2007, 55, 8118–8125. [Google Scholar] [CrossRef]
- Hou, Q. , Cheng, Y., Kang, D., Zhang, W., & Zhou, G.. Quality changes of pork during frozen storage: comparison of immersion solution freezing and air blast freezing[J]. International Journal of Food Science and Technology, 2020, 55, 109–118. [Google Scholar] [CrossRef]
- Huang, L. , Xiong, Y. L., Kong, B., Huang, X., & Li, J.. Influence of storage temperature and duration on lipid and protein oxidation and flavour changes in frozen pork dumpling filler[J]. Meat Science, 2013, 95, 295–301. [Google Scholar] [CrossRef]
- Ozen, B. O. , & Soyer, A.. Effect of plant extracts on lipid and protein oxidation of mackerel (Scomber scombrus) mince during frozen storage[J]. Journal of Food Science and Technology-Mysore, 2018, 55, 120–127. [Google Scholar] [CrossRef]
- Karlsdottir, M. G. , Sveinsdottir, K., Kristinsson, H. G., Villot, D., Craft, B. D., & Arason, S.. Effects of temperature during frozen storage on lipid deterioration of saithe (Pollachius virens) and hoki (Macruronus novaezelandiae) muscles[J]. Food Chemistry, 2014, 156, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M, Shahidi, F., Zhong, Y., and Eskin, N.A.M.. Lipid Oxidation. In: Biochemistry of Foods, Third Edition, Ed. Eskin, 2013, N.A.M., Elsevier, pp. 419-478.
- Leduc, F., Krzewinski, F., Le Fur, B., N’Guessan, A., Malle, P., Kol, O., & Duflos, G.. Differentiation of fresh and frozen/thawed fish, European sea bass (Dicentrarchus labrax), gilthead seabream (Sparus aurata), cod (Gadus morhua) and salmon (Salmo salar), using volatile compounds by SPME/GC/MS[J]. Journal of the Science of Food and Agriculture, 2012, 92(12), 2560-2568. [CrossRef]
- Zhang, Z., Li, G., Luo, L., & Chen, G.. Study on seafood volatile profile characteristics during storage and its potential use for freshness evaluation by headspace solid phase microextraction coupled with gas chromatography–mass spectrometry[J]. Analytica Chimica Acta, 2010, 659(1-2), 151-158. [CrossRef]
- Pan, N. , Dong, C., Du, X., Kong, B., Sun, J., & Xia, X.. Effect of freeze-thaw cycles on the quality of quick-frozen pork patty with different fat content by consumer assessment and instrument-based detection[J]. Meat Science, 2021, 172, 108313. [Google Scholar] [CrossRef] [PubMed]
- Utrera, M. , Morcuende, D., Ganhao, R., & Estevez, M.. Role of Phenolics Extracting from Rosa canina L. on Meat Protein Oxidation During Frozen Storage and Beef Patties Processing[J]. Food and Bioprocess Technology, 2015, 8, 854–864. [Google Scholar] [CrossRef]
- Zheng, Y, Qiu, Z, Wang, X. . Protein oxidation and tandem mass tag-based proteomic analysis in the dorsal muscle of farmed obscure pufferfish subjected to multiple freeze–thaw cycles. Journal of Food Processing and Preservation, 2020, 44, e14721. [Google Scholar] [CrossRef]
- Qian, S., Li, X., Wang, H., Mehmood, W., Zhang, C., & Blecker, C.. Effects of frozen storage temperature and duration on changes in physicochemical properties of beef myofibrillar protein[J]. Journal of Food Quality, 2021, 8836749. [CrossRef]
| Indicator | time t | freezing rate FR | temperature ST | t*FR | t*ST |
| TVBN | *** | NS | NS | NS | NS |
| TBARS | *** | *** | *** | ** | *** |
| Water-holding of myofibrils | NS | NS | * | * | NS |
| Thaw loss | *** | NS | NS | NS | NS |
| Thiols | NS | ** | NS | NS | NS |
| Surface hydrophobicity | NS | *** | *** | NS | NS |
| Compounds | Freezing rate | Storage temperature | Storage time |
| Hexanoic acid | NS | NS | ** |
| Heptanoic acid | NS | NS | * |
| Pentadecanal | * | NS | NS |
| Octanoic acid | NS | NS | NS |
| 2-Propenal, 3-phenyl- | NS | NS | NS |
| Nonanoic acid | NS | NS | ** |
| n-Decanoic acid | NS | NS | NS |
| Heptanal | *** | *** | NS |
| 2-Hexenal | *** | *** | NS |
| 2-Pentanone | *** | *** | *** |
| 1-Pentanol | *** | *** | *** |
| 1-Heptadecanol | NS | *** | *** |
| 2-Butanone, 3-methyl- | NS | *** | ** |
| 2-Butanone, 3-hydroxy- | NS | ** | * |
| Octanal | NS | NS | NS |
| 2,3-Octanedione | * | * | NS |
| 5-Hepten-2-one, 6-methyl- | NS | NS | NS |
| 1-Hexanol | *** | *** | ** |
| 3-Heptanol, 6-methyl- | NS | NS | NS |
| 2-Nonanone | NS | NS | * |
| Nonanal | NS | NS | NS |
| 2-Octenal | NS | NS | NS |
| 1-Octen-3-ol | * | * | NS |
| 1-Heptanol | *** | *** | ** |
| Decanal | NS | ** | * |
| 2,4-Heptadienal | *** | *** | NS |
| (5Z)-Octa-1,5-dien-3-ol | *** | *** | ** |
| 4-Hepten-1-ol | *** | *** | NS |
| 1-Hexanol, 2-ethyl- | NS | NS | * |
| 2-Nonenal | NS | NS | NS |
| 1-Octanol | NS | NS | * |
| 2,7-Octadien-1-ol | *** | *** | NS |
| Undecanal | *** | *** | NS |
| 3-Undecanone | *** | NS | ** |
| 2-Undecanone | NS | * | NS |
| 2-Octen-1-ol | ** | NS | NS |
| 2-Undecanone | NS | * | NS |
| 1-Nonanol | NS | * | * |
| 11-Dodecenol | *** | *** | NS |
| 2(5H)-furanone | NS | NS | * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




