Submitted:
23 May 2023
Posted:
24 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. chemicals
2.2. Donated Blood
2.3. Particle Preparation
2.4. Particle Characterization
2.4.1. Concentration Determination
2.4.2. Size, Zeta Potential and Conductivity
2.4.3. Hemoglobin Content
2.4.4. Percentage of Functional Hemoglobin
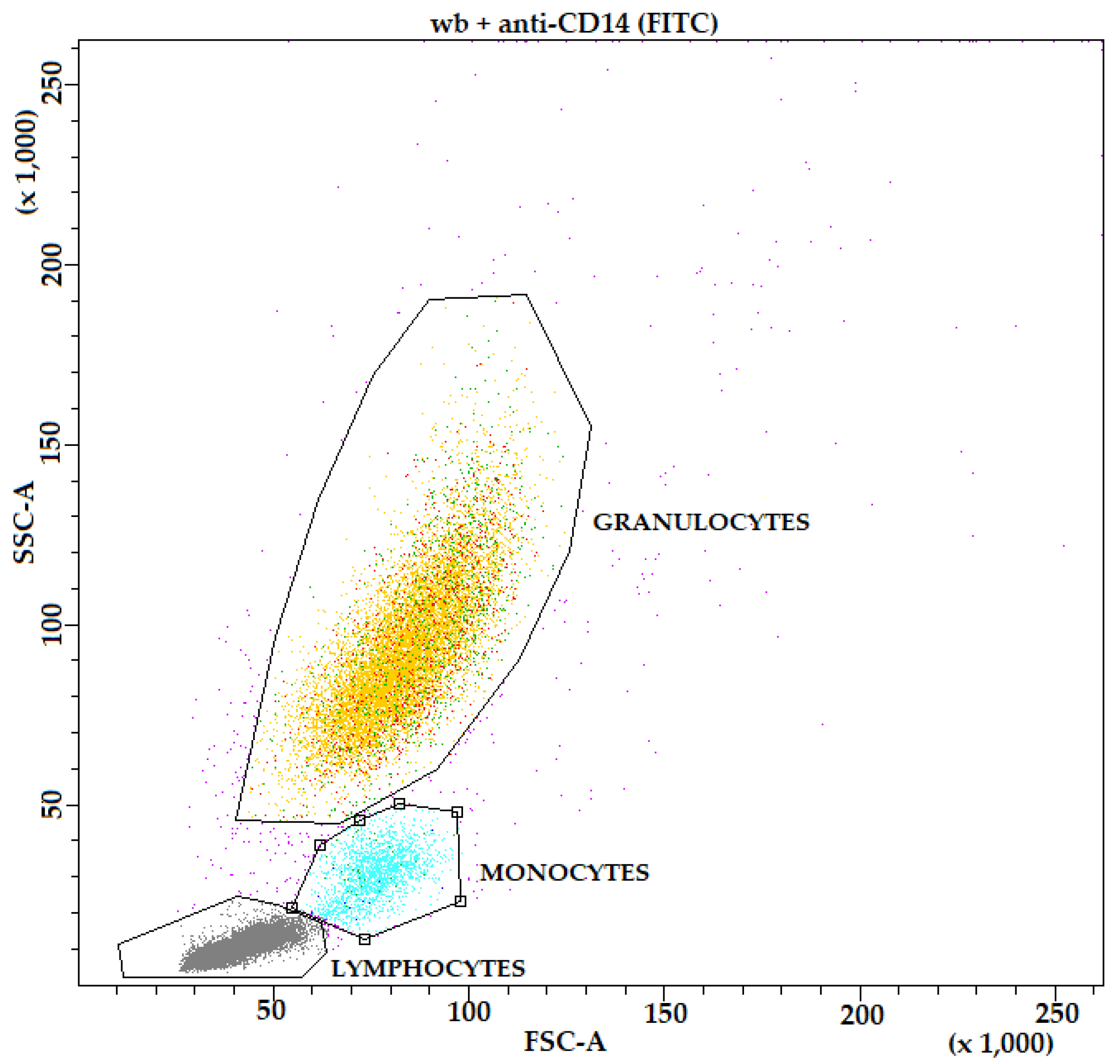
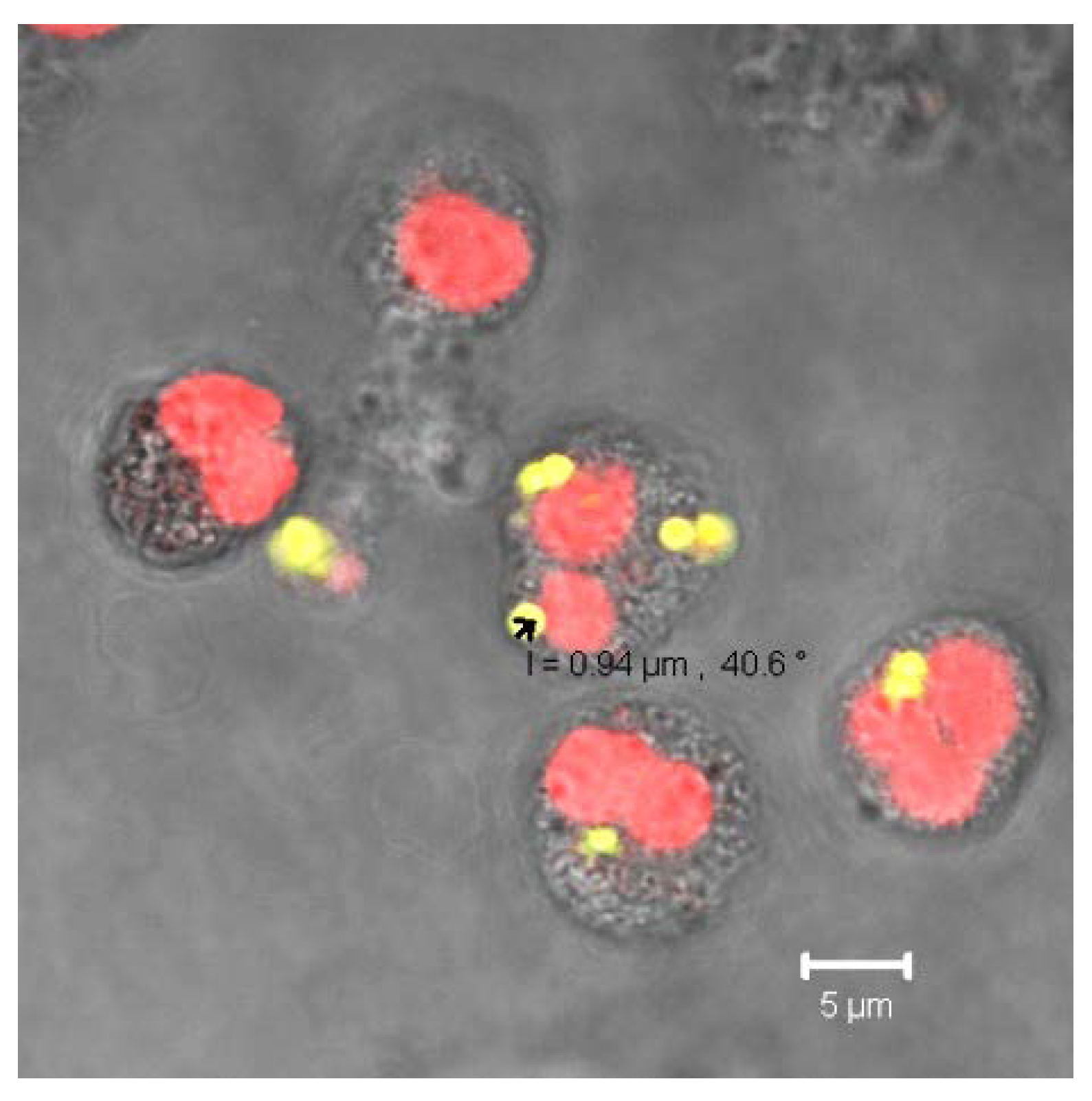
2.5. Indirect Phagocytosis Test
| Sample * | Incubation with | Purpose |
|---|---|---|
| HbMP | - | HbMP autofluorescence |
| DAPI-HbMP | DAPI | DAPI-impact |
| PI-HbMP | PI | PI-impact |
| wb | - | wb/background-noise |
| incub-wb | - | Impact of incubation-time |
| DAPI-wb | DAPI | DAPI-impact |
| PI-wb | PI | PI-impact |
| anti-CD14-wb | Anti-CD14, FITC | Identify monocyte population |
| anti-CD163-wb | Anti-CD163, APC | Identify monocyte population |
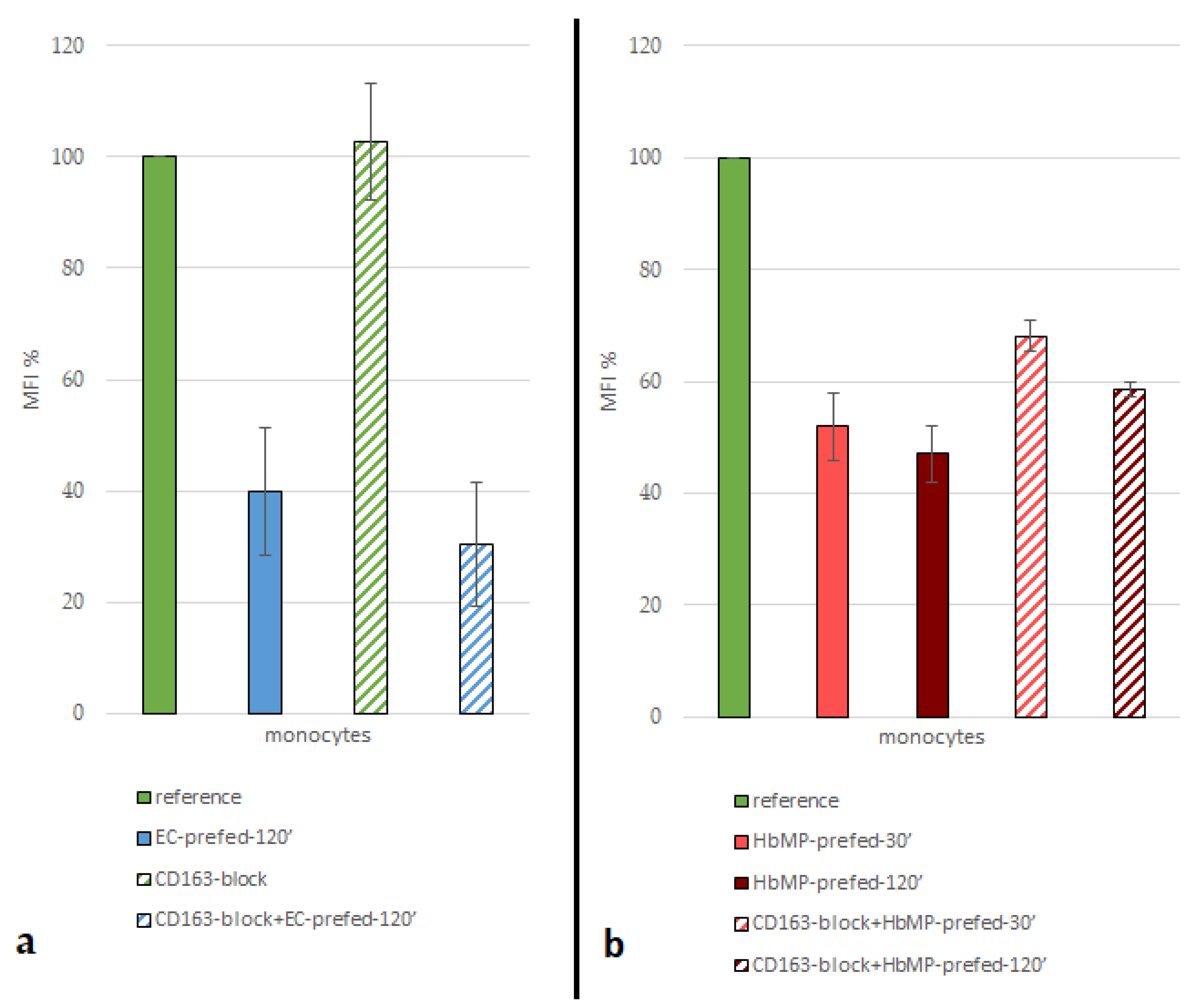
| Sample 1 | AB-use 2 | pre-fed with | Purpose | |
|---|---|---|---|---|
| reference | no | no | Max. MFI | |
| EC-prefed-120’ 3 | no | E. coli, untagged | Extent of E. coli-phagocytosis | |
| CD163-block | yes | no | AB-impact on FITC-E. coli phagocytosis | |
| CD163-block+EC-prefed-120’ | yes | E. coli, untagged | AB-impact on untagged E. coli phagocytosis | |
| HbMP-prefed-30’ | no | HbMP | Unhindered uptake of HbMP, 30 min. | |
| HbMP-prefed-120’ | no | HbMP | Unhindered uptake of HbMP, 120 min. | |
| CD163-block+HbMP-prefed-30’ | yes | HbMP | uptake of HbMP with blocked CD163, 30 min. | |
| CD163-block+HbMP-prefed-120’ | yes | HbMP | uptake of HbMP with blocked CD163, 120 min. |
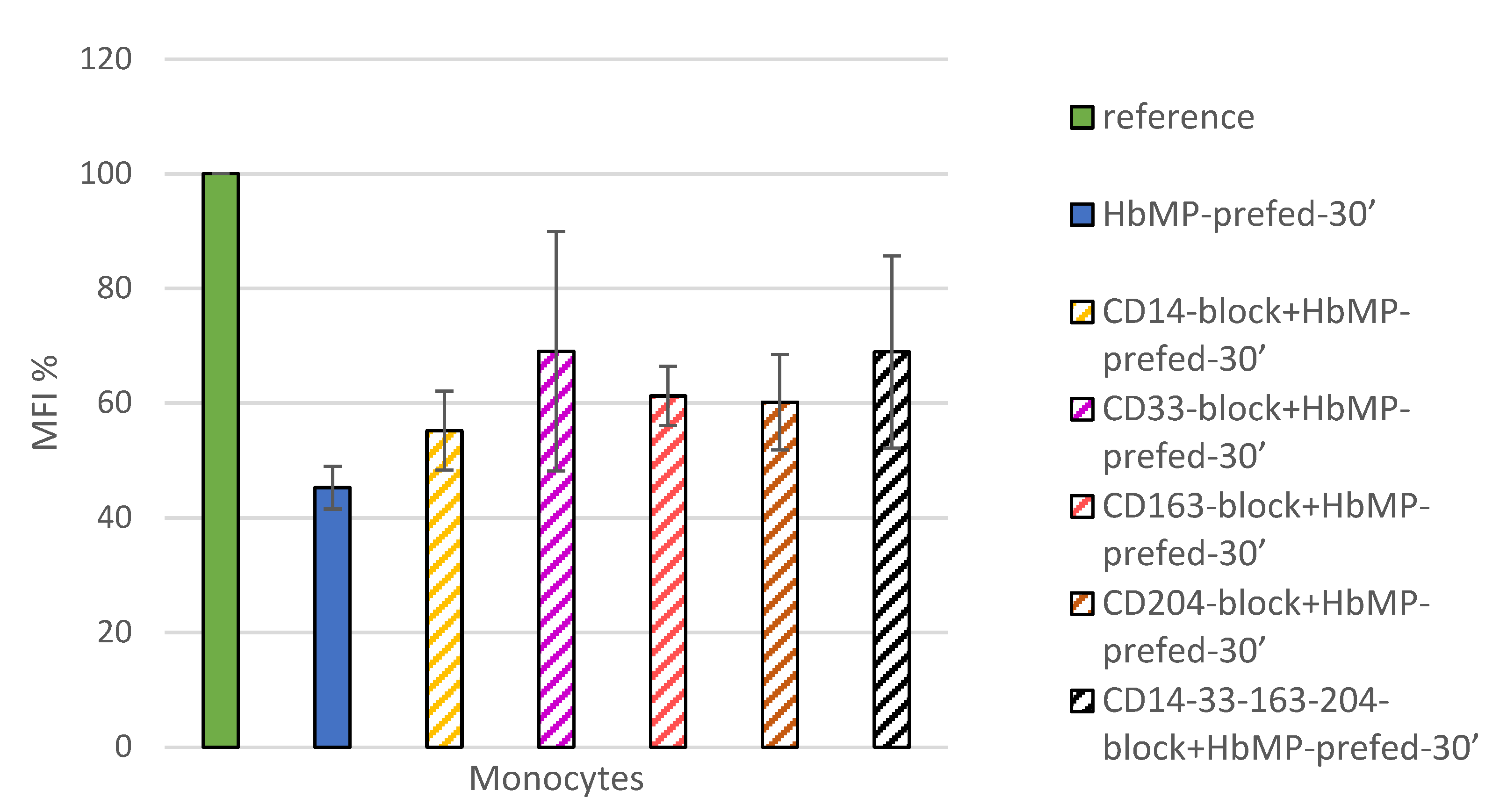
| Sample 1 | AB-use 2 | pre-fed with | Purpose | |
|---|---|---|---|---|
| reference | no | no | Max. MFI | |
| HbMP-prefed-30’ | no | HbMP | Unhindered uptake of HbMP, 30 min. | |
| CD14-block | yes | no | AB-impact on FITC-E. coli phagocytosis | |
| CD33-block | yes | no | AB-impact on FITC-E. coli phagocytosis | |
| CD204-block | yes | no | AB-impact on FITC-E. coli phagocytosis | |
| CD14,33,163,204-block | yes | no | AB-impact on FITC-E. coli phagocytosis | |
| CD14-block+HbMP-prefed-30’ | yes | HbMP | uptake of HbMP with blocked CD14, 30 min. | |
| CD33-block+HbMP-prefed-30’ | yes | HbMP | uptake of HbMP with blocked CD33, 30 min. | |
| CD204-block+HbMP-prefed-30’ | yes | HbMP | uptake of HbMP with blocked CD204, 30 min. | |
| CD14, 33, 163, 204-block+HbMP-prefed-30’ | yes | HbMP | uptake of HbMP with blockedCD14, 33, 163, 204, 30 min. |
3. Results
3.1. Particle Characterization
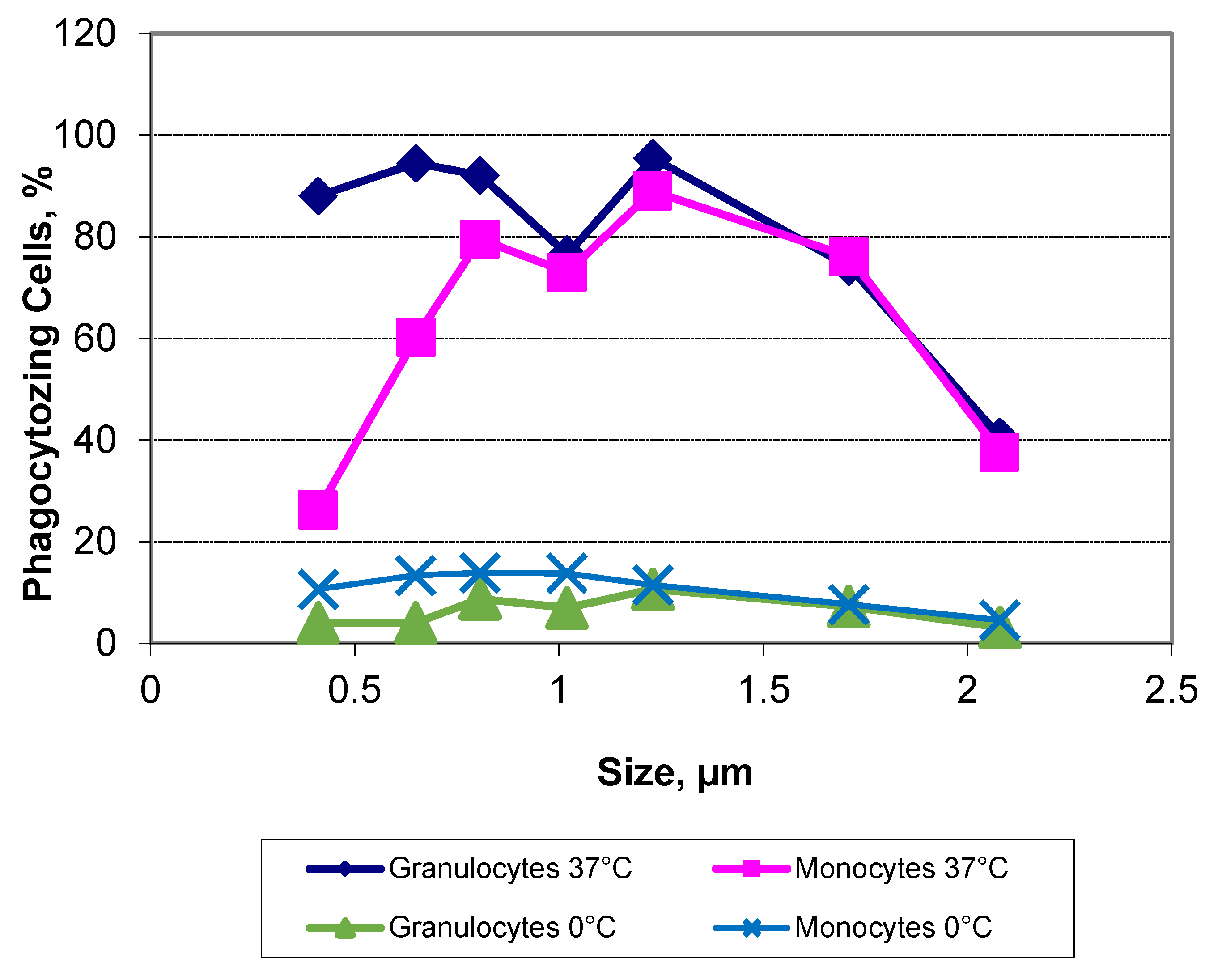
3.2. Exclusion of Confounders, Indirect Phagocytosis-Test
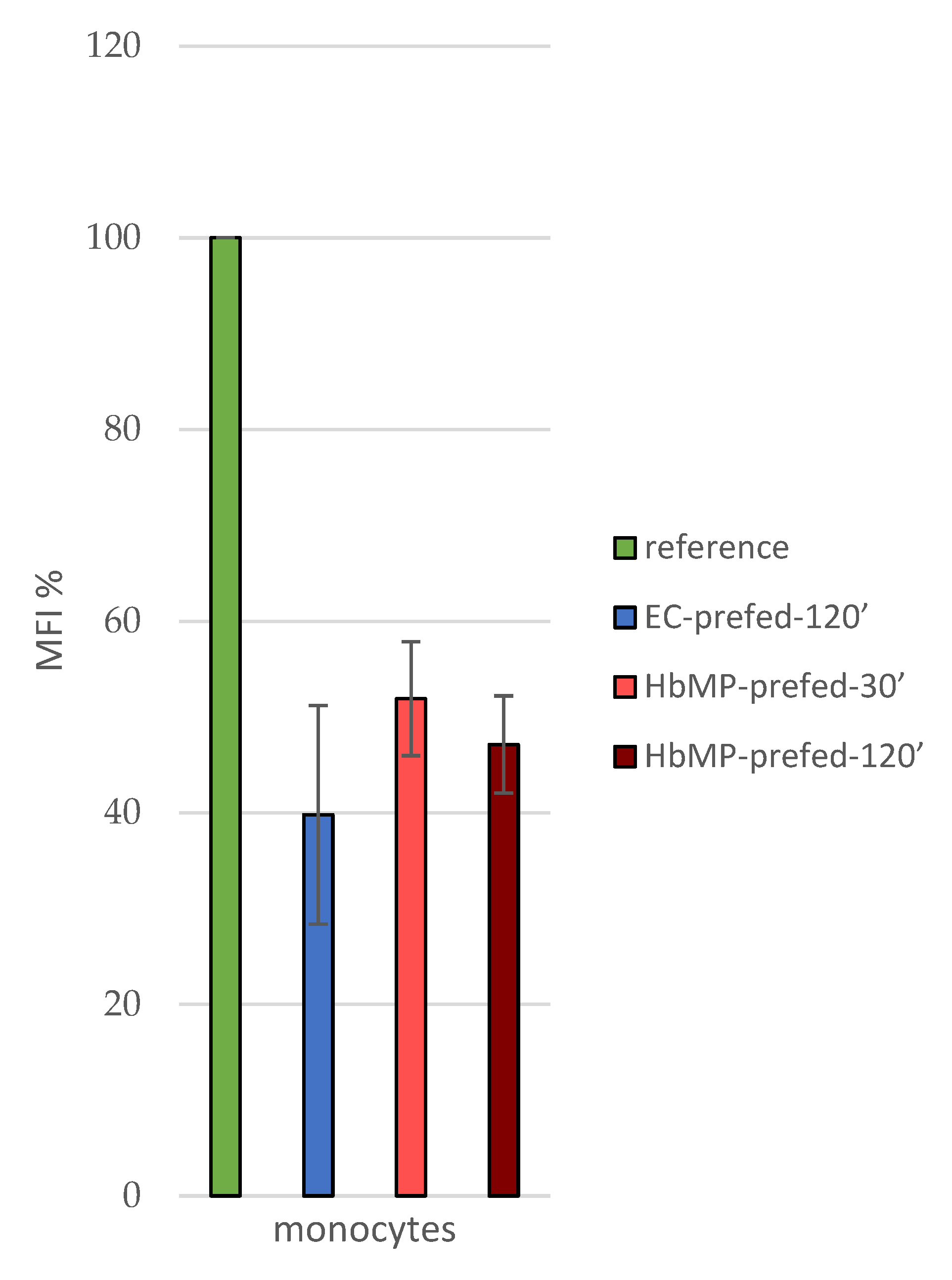
3.3. Indirect Phagocytosis-Test No 1
3.4. Indirect Phagocytosis-Test No 2
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Yoshida, T.; Prudent, M.; D'Alessandro, A. Red blood cell storage lesion: causes and potential clinical consequences. Blood Transfus 2019, 17, 27–52. [Google Scholar] [CrossRef] [PubMed]
- Aoki, Y. The risk of severe bacterial infection in non-transfusion-dependent thalassemia. Pediatr Res 2021. [Google Scholar] [CrossRef]
- Yao, C.Y.; Chien, J.H.; Chuang, H.Y.; Ho, T.F. Associated Factors With Acute Transfusion Reaction From Hospital Online Reporting Events: A Retrospective Cohort Study. J Patient Saf 2020, 16, e303–e309. [Google Scholar] [CrossRef] [PubMed]
- Passwater, M. Antibody Formation in Transfusion Therapy. J Infus Nurs 2018, 41, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Estep, T.N. Pharmacokinetics and mechanisms of plasma removal of hemoglobin-based oxygen carriers. Artif Cells Nanomed Biotechnol 2015, 43, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; Guinn, N.R.; Lowery, D.R.; Shore-Lesserson, L.; Shander, A. Blood Substitutes and Oxygen Therapeutics: A Review. Anesth Analg 2021, 132, 119–129. [Google Scholar] [CrossRef]
- Wieder, T.; Lang, F. Erythrozyten. In Hämostaseologie; Springer: Berlin Heidelberg, 2010; pp. 113–119. [Google Scholar]
- Alayash, A.I. Oxygen therapeutics: can we tame haemoglobin? Nat Rev Drug Discov 2004, 3, 152–159. [Google Scholar] [CrossRef]
- Minneci, P.C.; Deans, K.J.; Zhi, H.; Yuen, P.S.; Star, R.A.; Banks, S.M.; Schechter, A.N.; Natanson, C.; Gladwin, M.T.; Solomon, S.B. Hemolysis-associated endothelial dysfunction mediated by accelerated NO inactivation by decompartmentalized oxyhemoglobin. J Clin Invest 2005, 115, 3409–3417. [Google Scholar] [CrossRef]
- Sulkowski, J.P.; Cooper, J.N.; Pearson, E.G.; Connelly, J.T.; Rintoul, N.; Kilbaugh, T.J.; Deans, K.J.; Minneci, P.C. Hemolysis-Associated Nitric Oxide Dysregulation during Extracorporeal Membrane Oxygenation. J Extra Corpor Technol 2014, 46, 217–223. [Google Scholar] [CrossRef]
- Alayash, A.I. Setbacks in blood substitutes research and development: a biochemical perspective. Clin Lab Med 2010, 30, 381–389. [Google Scholar] [CrossRef]
- Alayash, A.I. Mechanisms of Toxicity and Modulation of Hemoglobin-based Oxygen Carriers. Shock 2019, 52, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Bunn, H.F.; Jandl, J.H. The renal handling of hemoglobin. Trans Assoc Am Physicians 1968, 81, 147–152. [Google Scholar] [PubMed]
- Schaer, D.J.; Alayash, A.I.; Buehler, P.W. Gating the radical hemoglobin to macrophages: the anti-inflammatory role of CD163, a scavenger receptor. Antioxid Redox Signal 2007, 9, 991–999. [Google Scholar] [CrossRef]
- Rentsendorj, O.; Zhang, X.; Williams, M.C.; Buehler, P.W.; D'Agnillo, F. Transcriptional Suppression of Renal Antioxidant Enzyme Systems in Guinea Pigs Exposed to Polymerized Cell-Free Hemoglobin. Toxics 2016, 4. [Google Scholar] [CrossRef]
- Buehler, P.W.; Vallelian, F.; Mikolajczyk, M.G.; Schoedon, G.; Schweizer, T.; Alayash, A.I.; Schaer, D.J. Structural stabilization in tetrameric or polymeric hemoglobin determines its interaction with endogenous antioxidant scavenger pathways. Antioxid Redox Signal 2008, 10, 1449–1462. [Google Scholar] [CrossRef]
- Bleeker, W.K.; Berbers, G.A.; den Boer, P.J.; Agterberg, J.; Rigter, G.; Bakker, J.C. Effect of polymerization on clearance and degradation of free hemoglobin. Biomater Artif Cells Immobilization Biotechnol 1992, 20, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; Akha, A.S.; Holtby, R.J. Crosslinked, polymerized, and PEG-conjugated hemoglobin-based oxygen carriers: clinical safety and efficacy of recent and current products. Curr Drug Discov Technol 2012, 9, 158–165. [Google Scholar] [CrossRef]
- Prapan, A.; Suwannasom, N.; Kloypan, C.; Chaiwaree, S.; Steffen, A.; Xiong, Y.; Kao, I.; Pruß, A.; Georgieva, R.; Bäumler, H. Surface Modification of Hemoglobin-Based Oxygen Carriers Reduces Recognition by Haptoglobin, Immunoglobulin, and Hemoglobin Antibodies. Coatings 2019, 9, 454. [Google Scholar] [CrossRef]
- Awasthi, V.; Yee, S.H.; Jerabek, P.; Goins, B.; Phillips, W.T. Cerebral oxygen delivery by liposome-encapsulated hemoglobin: a positron-emission tomographic evaluation in a rat model of hemorrhagic shock. J Appl Physiol (1985) 2007, 103, 28–38. [Google Scholar] [CrossRef]
- Rameez, S.; Guzman, N.; Banerjee, U.; Fontes, J.; Paulaitis, M.E.; Palmer, A.F.; Patel, R.P.; Honavar, J. Encapsulation of hemoglobin inside liposomes surface conjugated with poly(ethylene glycol) attenuates their reactions with gaseous ligands and regulates nitric oxide dependent vasodilation. Biotechnol Prog 2012, 28, 636–645. [Google Scholar] [CrossRef]
- Kloypan, C.; Suwannasom, N.; Chaiwaree, S.; Prapan, A.; Smuda, K.; Baisaeng, N.; Pruß, A.; Georgieva, R.; Bäumler, H. In-vitro haemocompatibility of dextran-protein submicron particles. Artificial Cells, Nanomedicine, and Biotechnology 2019, 47, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Yamasaki, K.; Maruyama, T.; Otagiri, M. Comparison of the Pharmacokinetic Properties of Hemoglobin-Based Oxygen Carriers. Journal of Functional Biomaterials 2017, 8, 11. [Google Scholar] [CrossRef]
- Xiong, Y.; Steffen, A.; Andreas, K.; Müller, S.; Sternberg, N.; Georgieva, R.; Bäumler, H. Hemoglobin-Based Oxygen Carrier Microparticles: Synthesis, Properties, and In Vitro and In Vivo Investigations. Biomacromolecules 2012, 13, 3292–3300. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.C.; Liu, L.; Ship, N.; Kluger, R.H.; Pang, K.S. Role of haptoglobin on the uptake of native and beta-chain [trimesoyl-(Lys82)beta-(Lys82)beta] cross-linked human hemoglobins in isolated perfused rat livers. Drug Metab Dispos 2008, 36, 937–945. [Google Scholar] [CrossRef]
- Gburek, J.; Konopska, B.; Juszczynska, K.; Piwowar, A.; Dziegiel, P.; Borska, S.; Tolosano, E.; Golab, K. Hemoglobin - a novel ligand of hepatocyte ectopic F1-ATPase. J Physiol Pharmacol 2015, 66, 823–830. [Google Scholar]
- Schaer, D.J.; Schaer, C.A.; Buehler, P.W.; Boykins, R.A.; Schoedon, G.; Alayash, A.I.; Schaffner, A. CD163 is the macrophage scavenger receptor for native and chemically modified hemoglobins in the absence of haptoglobin. Blood 2006, 107, 373–380. [Google Scholar] [CrossRef]
- Ascenzi, P.; Bocedi, A.; Visca, P.; Altruda, F.; Tolosano, E.; Beringhelli, T.; Fasano, M. Hemoglobin and heme scavenging. IUBMB Life (International Union of Biochemistry and Molecular Biology: Life) 2005, 57, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Drieghe, S.; Stove, V.; Decruyenaere, J.; Delanghe, J. Interpretation of hemolysis tests following administration of a second-generation hemoglobin-based oxygen carrier. Acta Clin Belg 2013, 68, 282–286. [Google Scholar] [CrossRef]
- Baek, J.H.; Zhou, Y.; Harris, D.R.; Schaer, D.J.; Palmer, A.F.; Buehler, P.W. Down Selection of Polymerized Bovine Hemoglobins for Use as Oxygen Releasing Therapeutics in a Guinea Pig Model. Toxicological Sciences 2012, 127, 567–581. [Google Scholar] [CrossRef]
- Baumler, H.; Xiong, Y.; Liu, Z.Z.; Patzak, A.; Georgieva, R. Novel hemoglobin particles--promising new-generation hemoglobin-based oxygen carriers. Artif Organs 2014, 38, 708–714. [Google Scholar] [CrossRef]
- Kao, I.; Xiong, Y.; Steffen, A.; Smuda, K.; Zhao, L.; Georgieva, R.; Pruss, A.; Baumler, H. Preclinical In Vitro Safety Investigations of Submicron Sized Hemoglobin Based Oxygen Carrier HbMP-700. Artif Organs 2018, 42, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Kloypan, C.; Prapan, A.; Suwannasom, N.; Chaiwaree, S.; Kaewprayoon, W.; Steffen, A.; Xiong, Y.; Baisaeng, N.; Georgieva, R.; Baumler, H. Improved oxygen storage capacity of haemoglobin submicron particles by one-pot formulation. Artif Cells Nanomed Biotechnol 2018, 46, S964–S972. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Liu, Z.Z.; Georgieva, R.; Smuda, K.; Steffen, A.; Sendeski, M.; Voigt, A.; Patzak, A.; Bäumler, H. Nonvasoconstrictive Hemoglobin Particles as Oxygen Carriers. ACS Nano 2013, 7, 7454–7461. [Google Scholar] [CrossRef] [PubMed]
- LPT Report, No. 34476: Pharmacokinetic study of HbMP-700 by single 2-hr intravenous infusion to CD®rats (unpublished). 2017.
- Kelley, J.L.; Ozment, T.R.; Li, C.; Schweitzer, J.B.; Williams, D.L. Scavenger Receptor-A (CD204): A Two-Edged Sword in Health and Disease. Critical Reviews in Immunology 2014, 34, 241–261. [Google Scholar] [CrossRef] [PubMed]
- Smuda, K.; Gienger, J.; Honicke, P.; Neukammer, J. Function of Hemoglobin-Based Oxygen Carriers: Determination of Methemoglobin Content by Spectral Extinction Measurements. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Etzerodt, A.; Kjolby, M.; Nielsen, M.J.; Maniecki, M.; Svendsen, P.; Moestrup, S.K. Plasma clearance of hemoglobin and haptoglobin in mice and effect of CD163 gene targeting disruption. Antioxid Redox Signal 2013, 18, 2254–2263. [Google Scholar] [CrossRef]
- Gressner, A.M.; Gressner, O.A. Hämopexin. In Springer Reference Medizin; Springer: Berlin Heidelberg, 2019; pp. 1060–1061. [Google Scholar]
- Yan, Y.; Gause, K.T.; Kamphuis, M.M.J.; Ang, C.-S.; O’Brien-Simpson, N.M.; Lenzo, J.C.; Reynolds, E.C.; Nice, E.C.; Caruso, F. Differential Roles of the Protein Corona in the Cellular Uptake of Nanoporous Polymer Particles by Monocyte and Macrophage Cell Lines. ACS Nano 2013, 7, 10960–10970. [Google Scholar] [CrossRef]
- Report KF3042301AJ2 (2014) Untersuchungen zur Funktion phagozytierender Zellen unter Verwendungfunktionalisierter Polymerpartikel. 2014.
- Champion, J.A.; Walker, A.; Mitragotri, S. Role of Particle Size in Phagocytosis of Polymeric Microspheres. Pharmaceutical Research 2008, 25, 1815–1821. [Google Scholar] [CrossRef]
- Montel, L.; Pinon, L.; Fattaccioli, J. A Multiparametric and High-Throughput Assay to Quantify the Influence of Target Size on Phagocytosis. Biophysical Journal 2019, 117, 408–419. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Simberg, D.; Papini, E.; Farhangrazi, Z.S. Complement activation by drug carriers and particulate pharmaceuticals: Principles, challenges and opportunities. Adv Drug Deliv Rev 2020, 157, 83–95. [Google Scholar] [CrossRef]
- Raschke, T.M. Water structure and interactions with protein surfaces. Current Opinion in Structural Biology 2006, 16, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, S.M. Cancer nanomedicine and the complement system activation paradigm: Anaphylaxis and tumour growth. Journal of Controlled Release 2014, 190, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Investigation of protein adsorptiononnanocarriers for intravenous drugtargeting. Inaugural Dissertation, Freie Universität Berlin, refubium.fu-berlin.de, 2013. [Google Scholar]
- Lück, M.; Schröder, W.; Paulke, B.-R.; Blunk, T.; Müller, R.H. Complement activation by model drug carriers for intravenous application: determination by two-dimensional electrophoresis. Biomaterials 1999, 20, 2063–2068. [Google Scholar] [CrossRef] [PubMed]
- Goldfischer, S.; Novikoff, A.B.; Albala, A.; Biempica, L. Hemoglobin uptake by rat hepatocytes and its breakdown within lysosomes. Journal of Cell Biology 1970, 44, 513–529. [Google Scholar] [CrossRef]
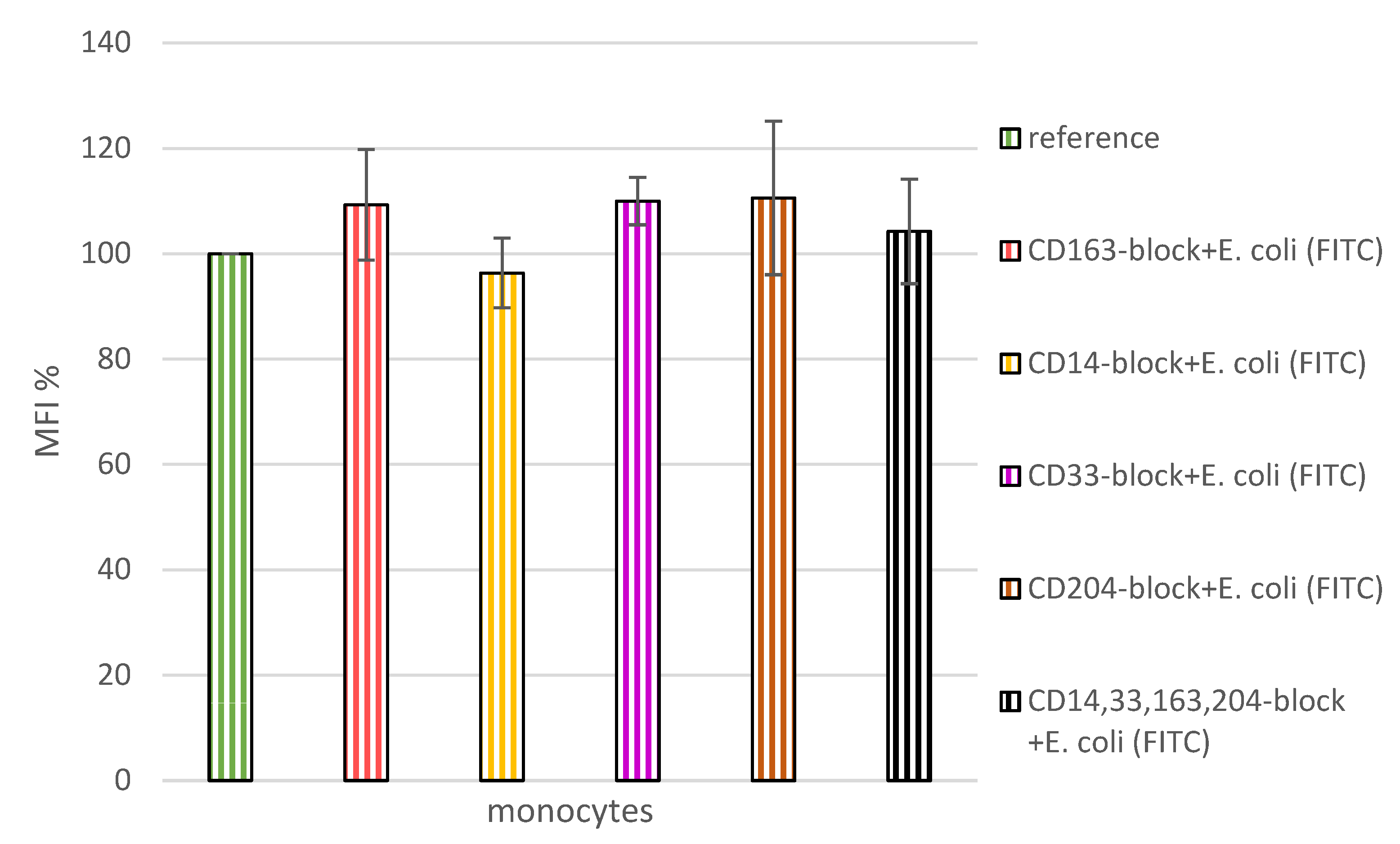
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




