Submitted:
22 May 2023
Posted:
23 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Analytical Methods
2.3. Box Benhken Design
3. Results
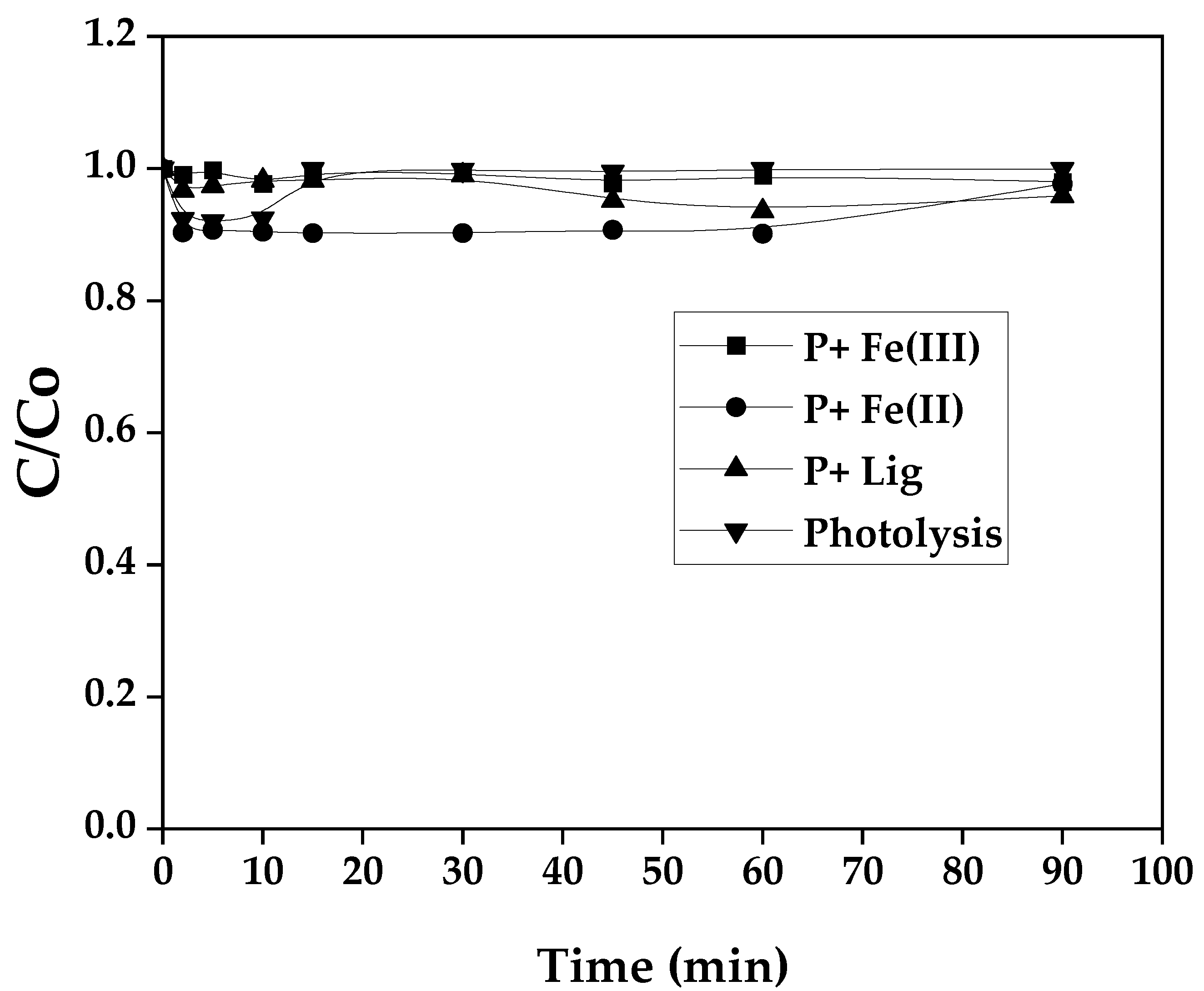
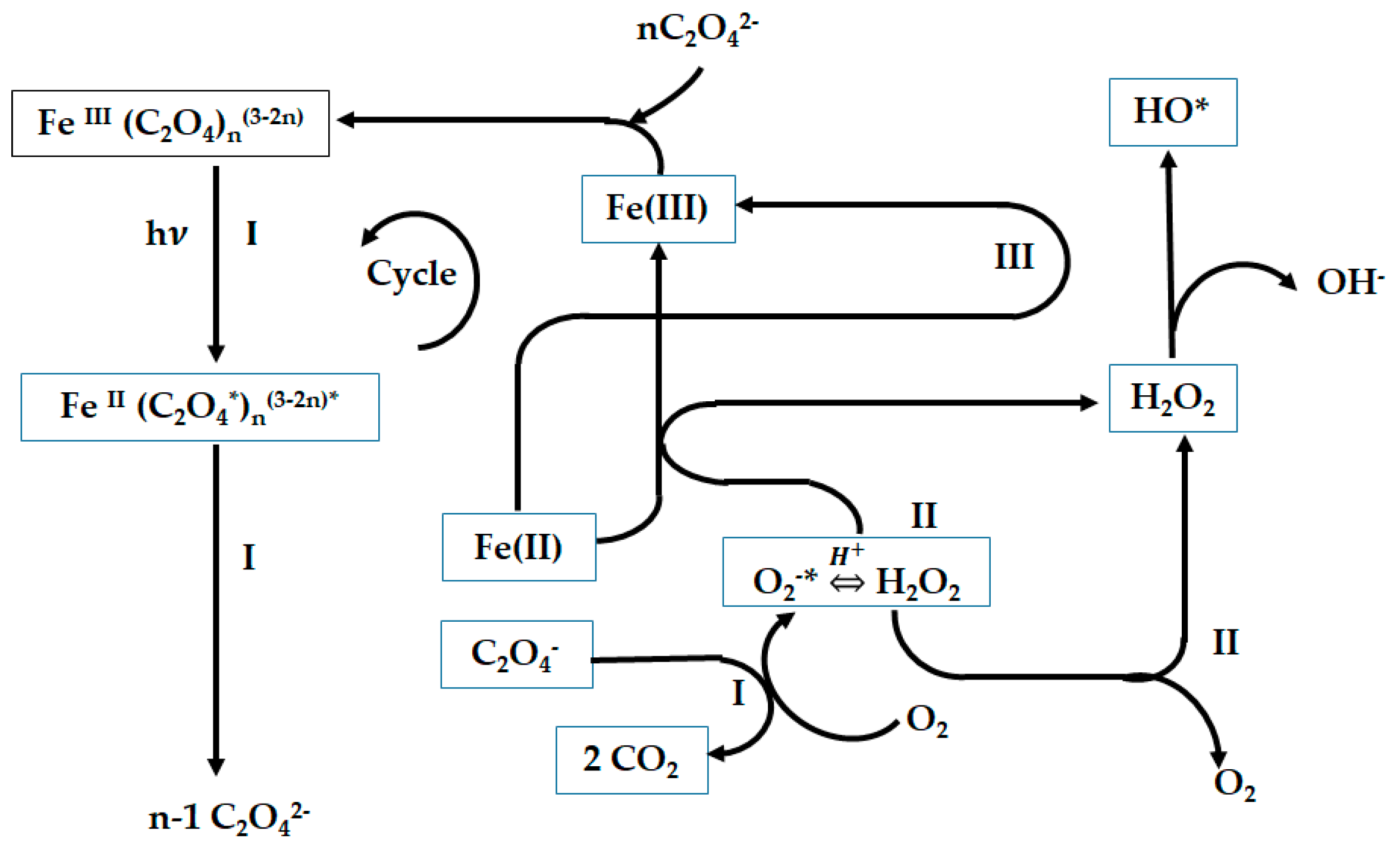
3.1. Homogeneous Photo-Fenton System
3.2. Photofenton Evalution of Possible Reactions
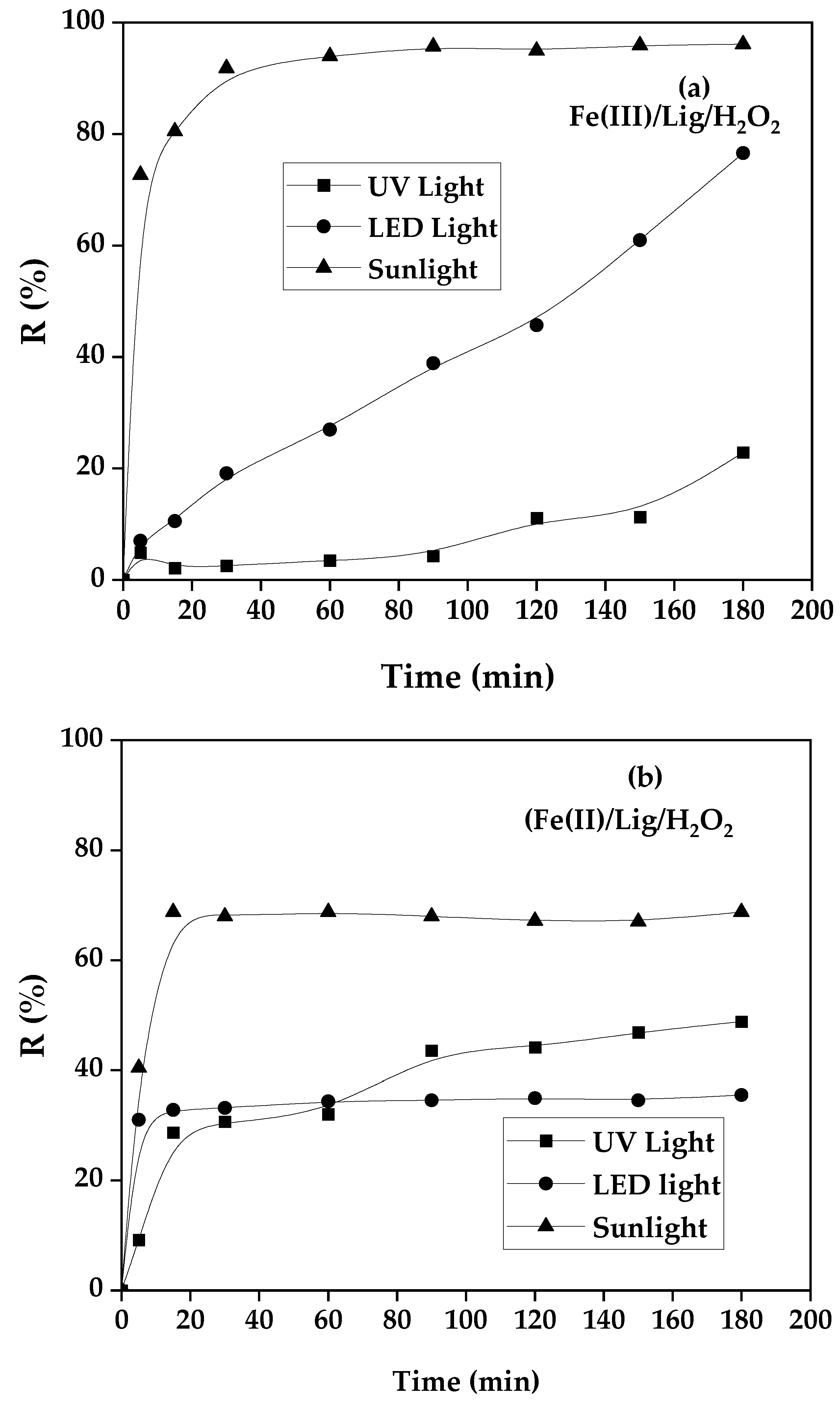
3.3. Influence of Light Sources Nature
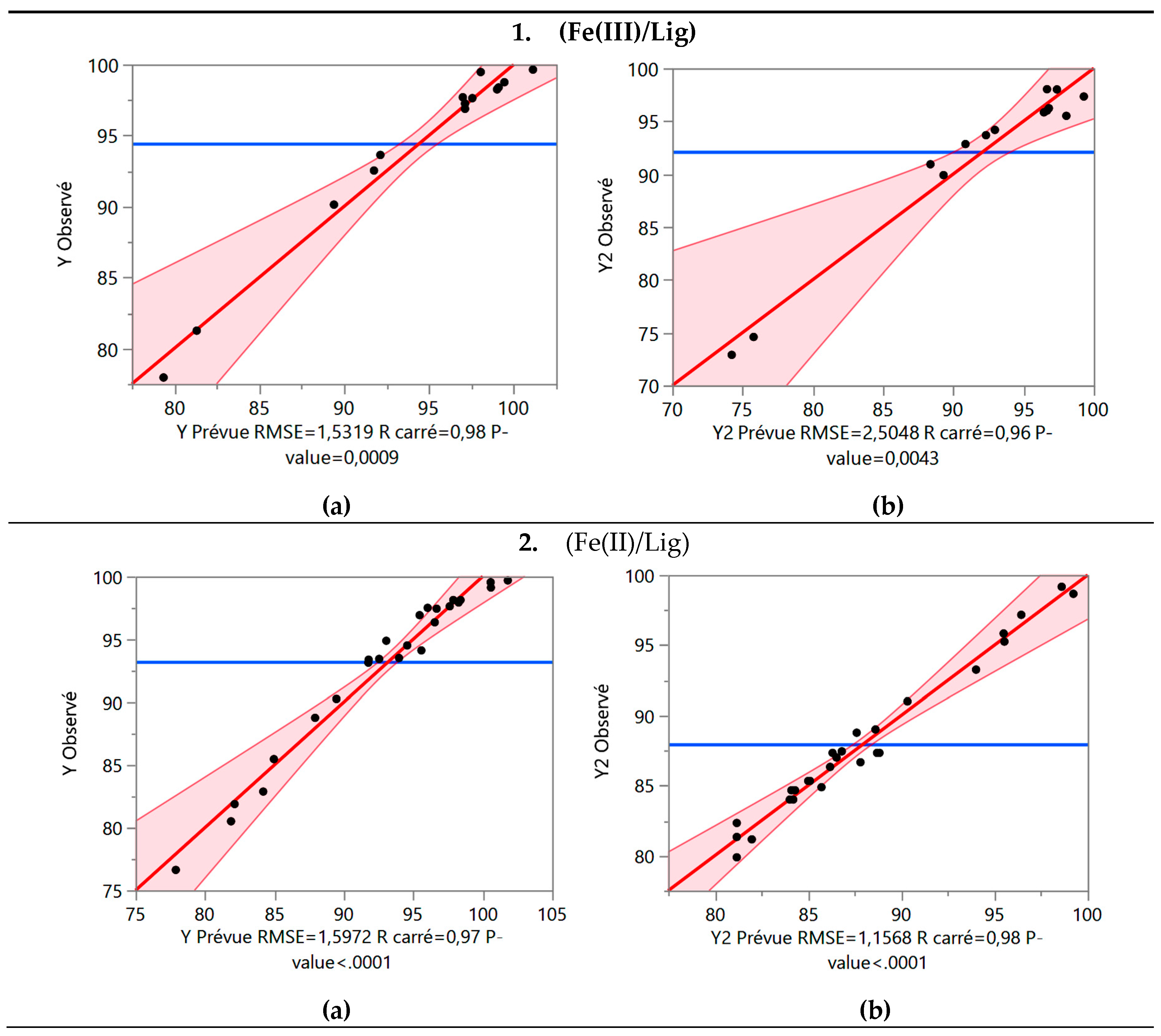
3.4. The BBD Analysis
- 1.
- (Fe(III)/Lig)
- 2.
- (Fe(II)/Lig) :
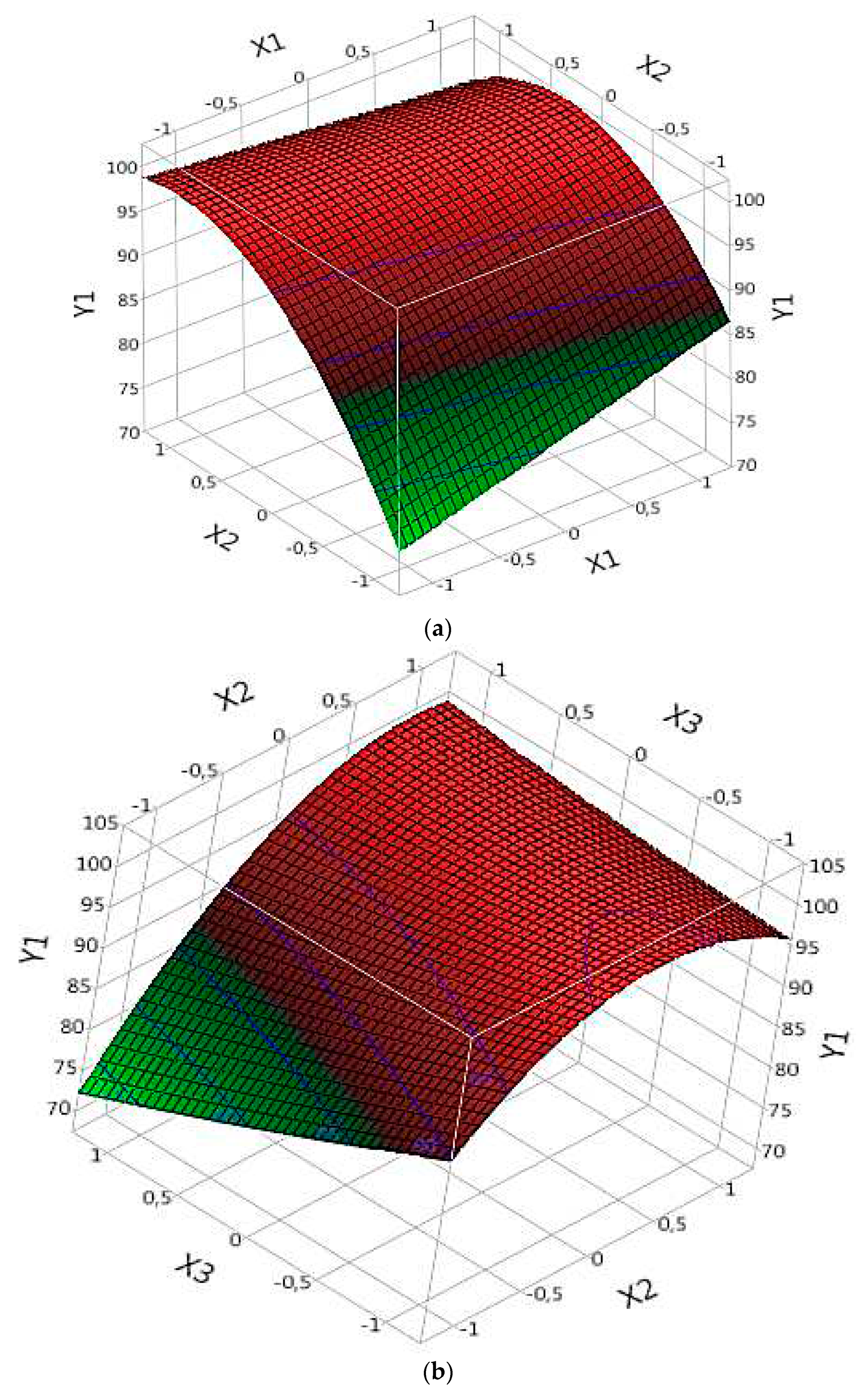
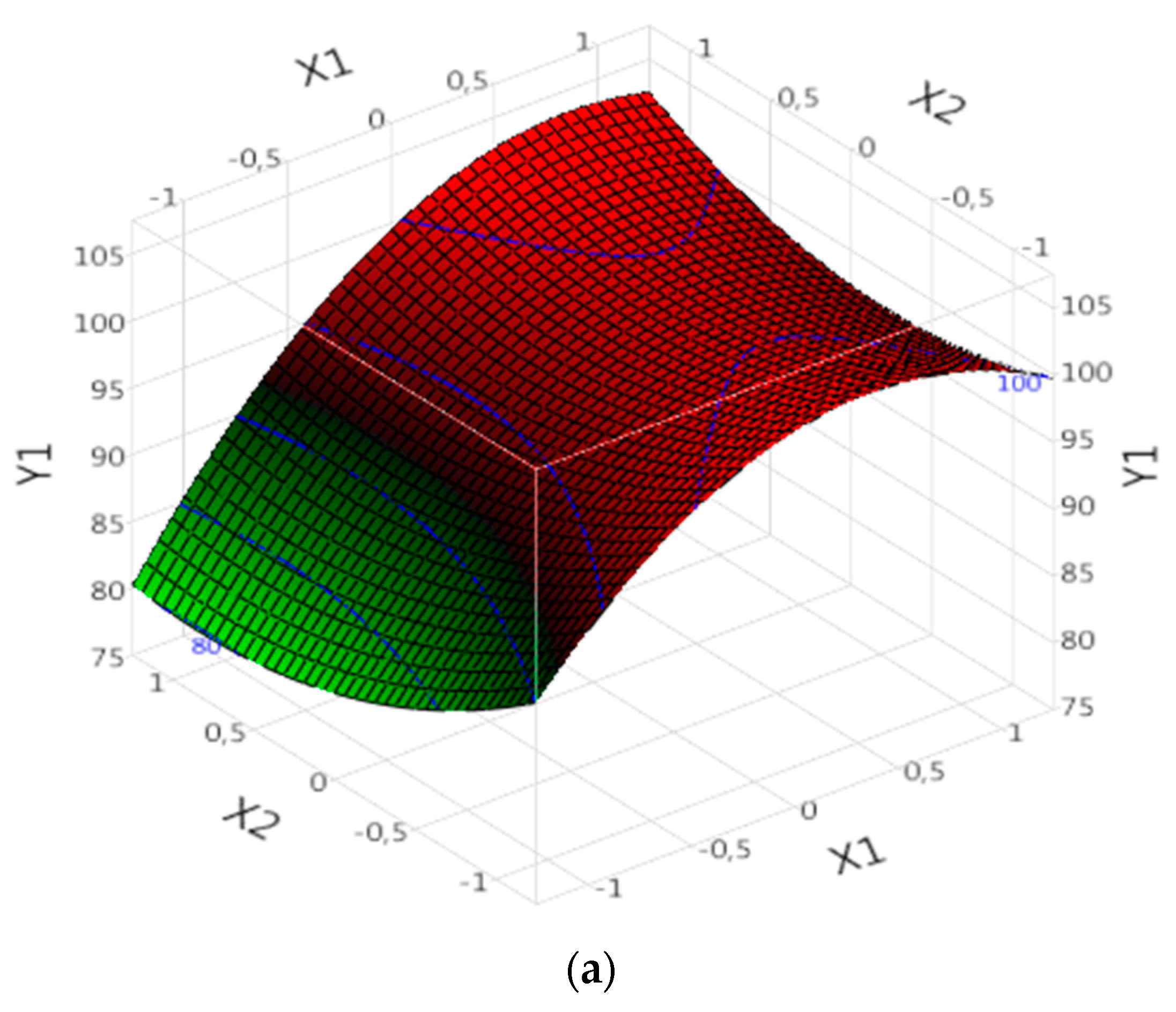
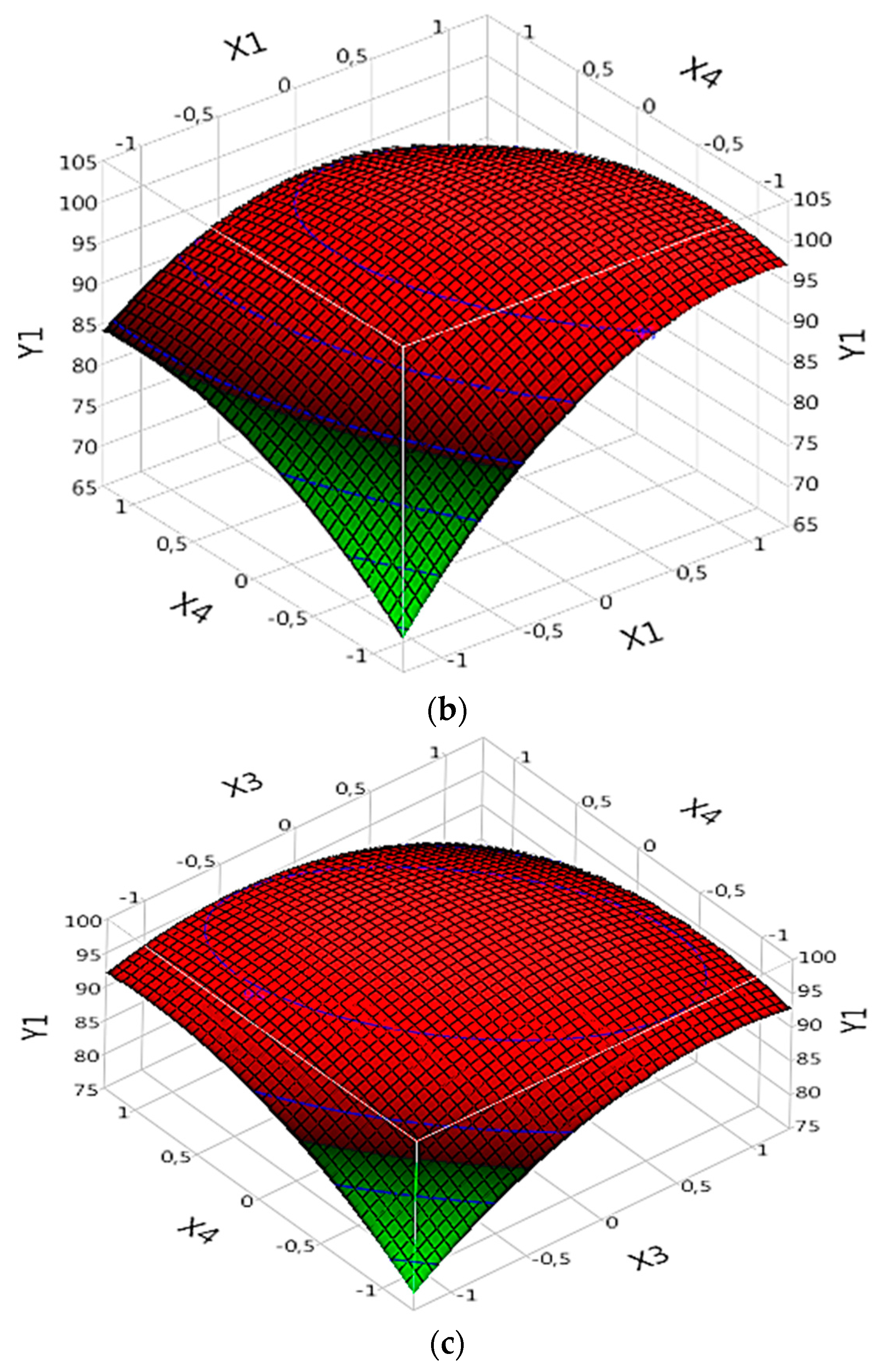
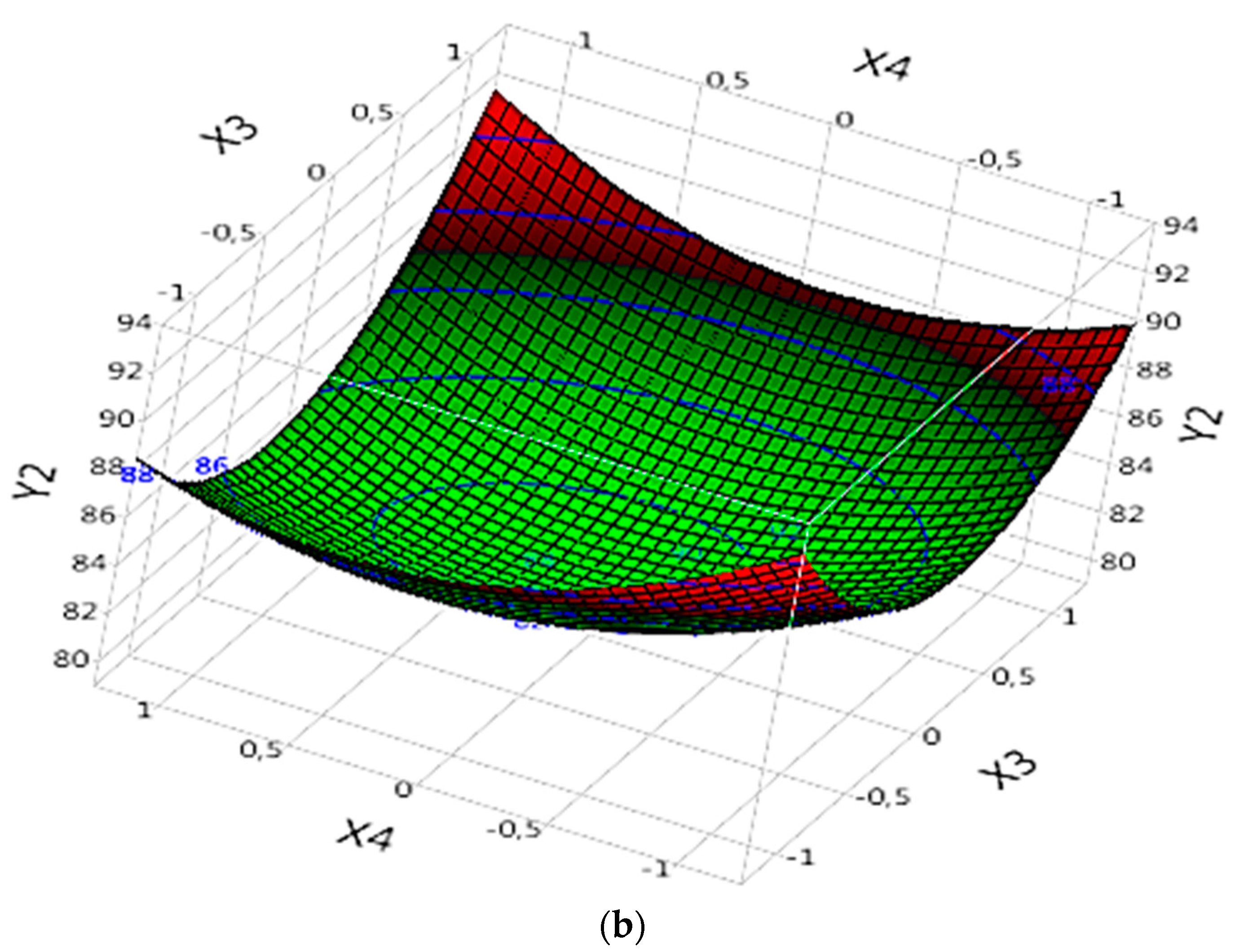
3.5. Response Surface Analysis
3.5.1. Effect of Parameters on R%:
3.5.1.1. Complex (Fe(III)/Lig)
3.5.1.2. Complex (Fe(II)/Lig)
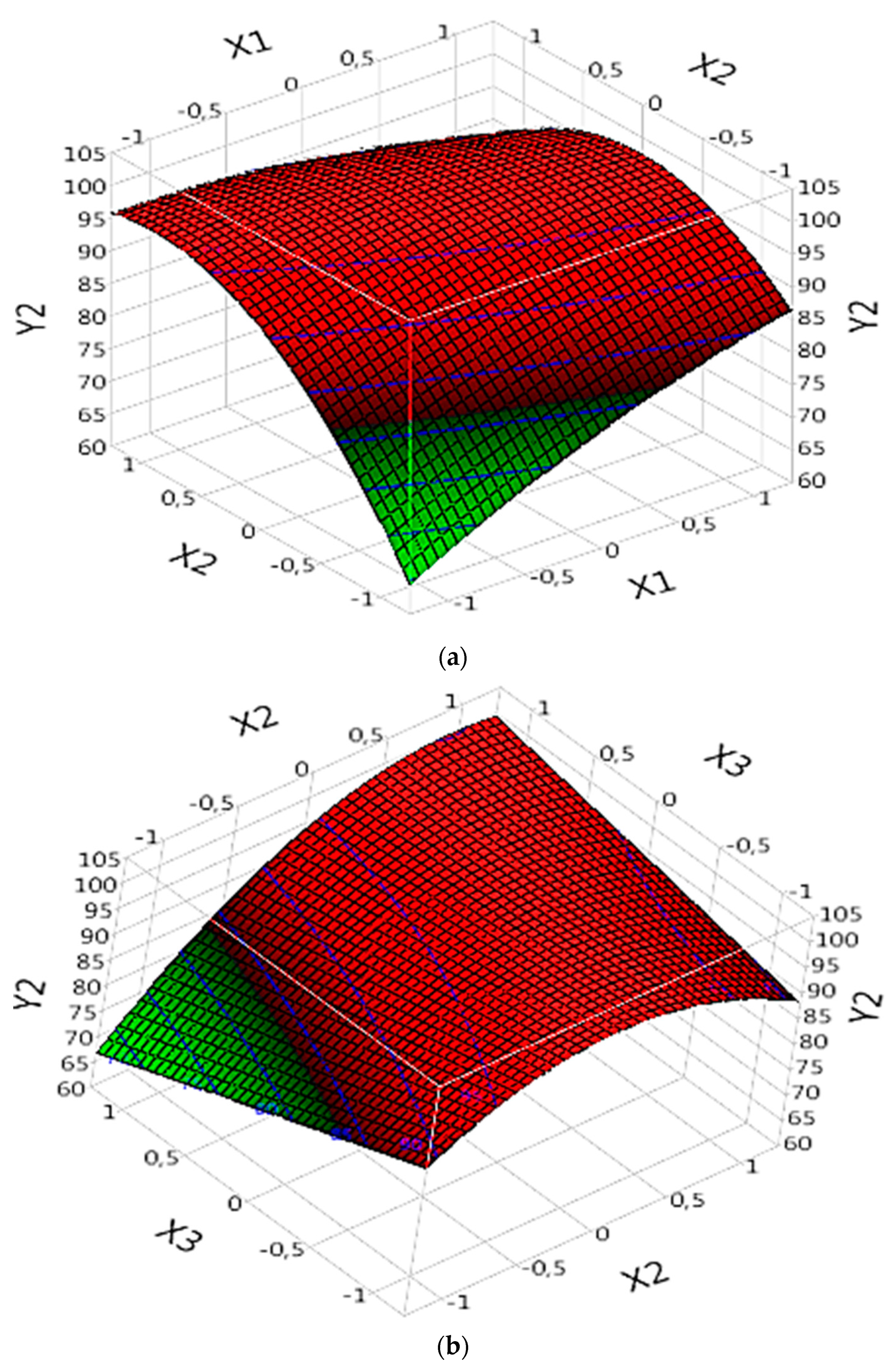
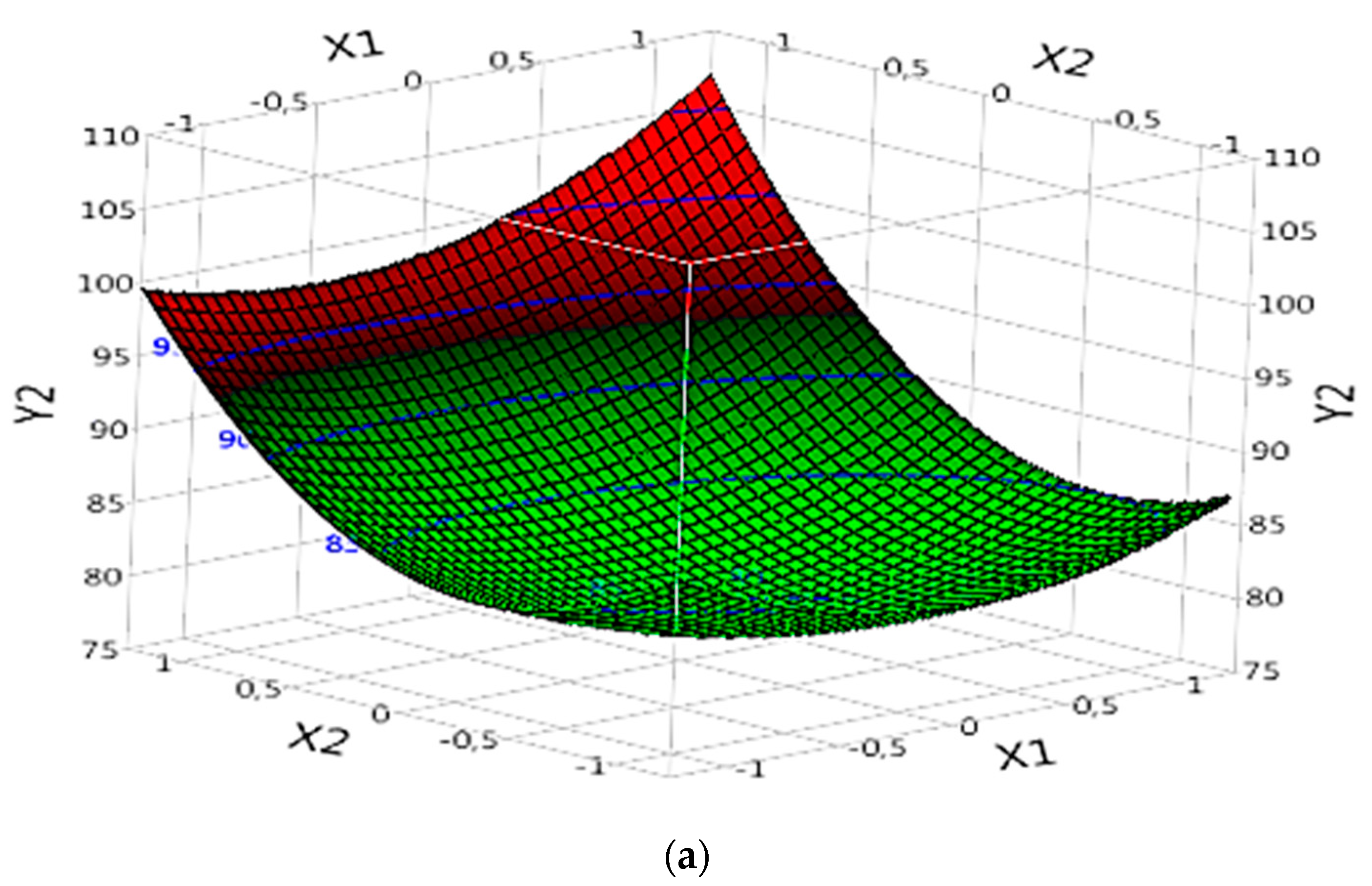
3.5.2. Effect of Parameters on DOC%
3.5.2.1. Complex (Fe(III)/Lig)
3.5.2.2. Complex (Fe(II)/Lig)
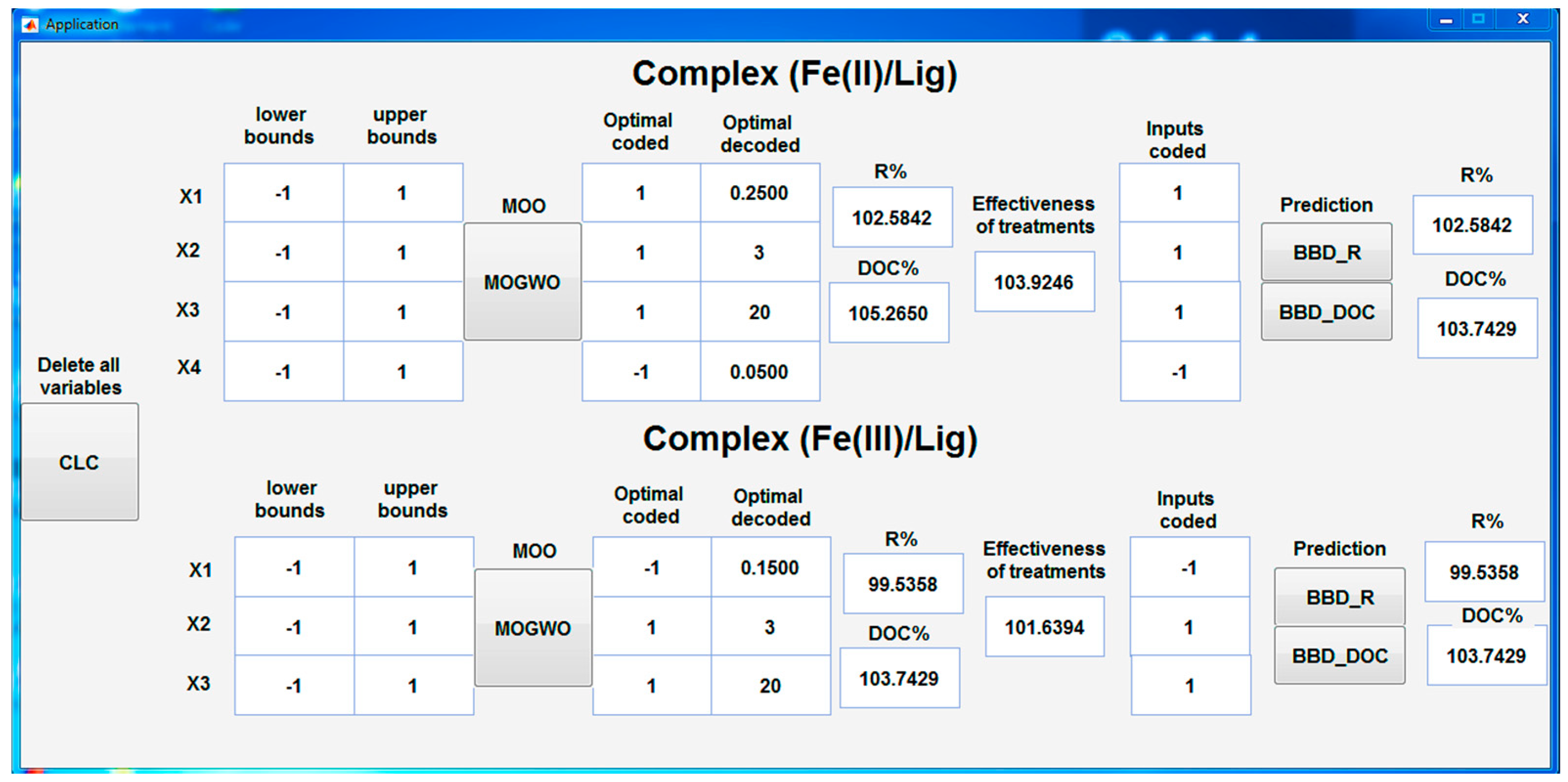
3.6. Multi-Objective Optimization and Validation
| R (%) | DOC(%) | R (%)+DCO (%)/2 | |
|---|---|---|---|
| (Fe(III)/Lig) | |||
| |||
| Experimental | 98.73 | 99.87 | 99.30 |
| Predicted response | 99.53 | 103.74 | 101.63 |
| Error | 0.8 | 3.87 | 2.33 |
| (Fe(II)/Lig) | |||
| |||
| Experimental | 99.63 | 99.92 | 99.77 |
| Predicted response | 102.58 | 105.26 | 103.92 |
| Error | 2.95 | 5.34 | 4.14 |
3.7. Interface for Optimization and Prediction
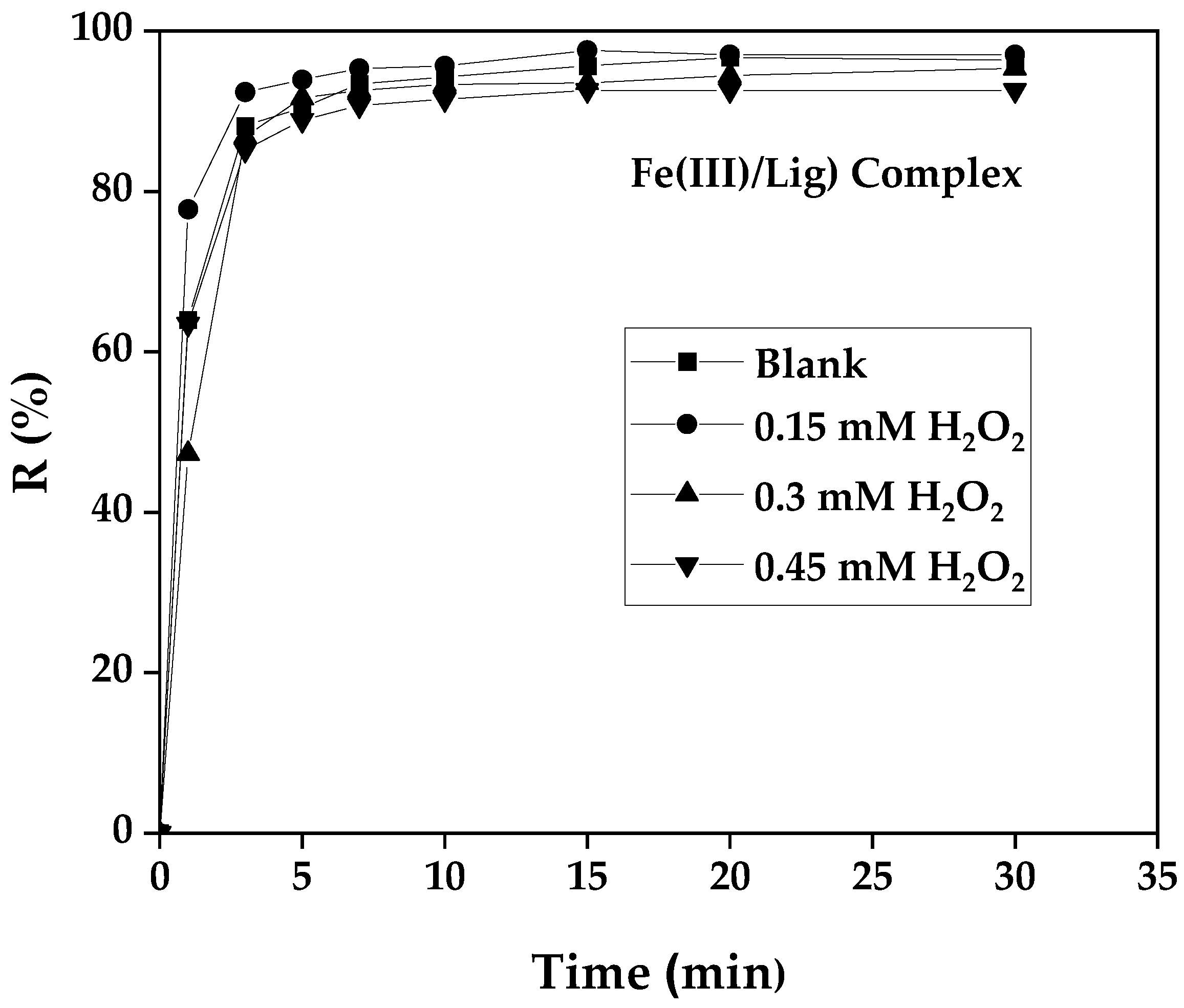
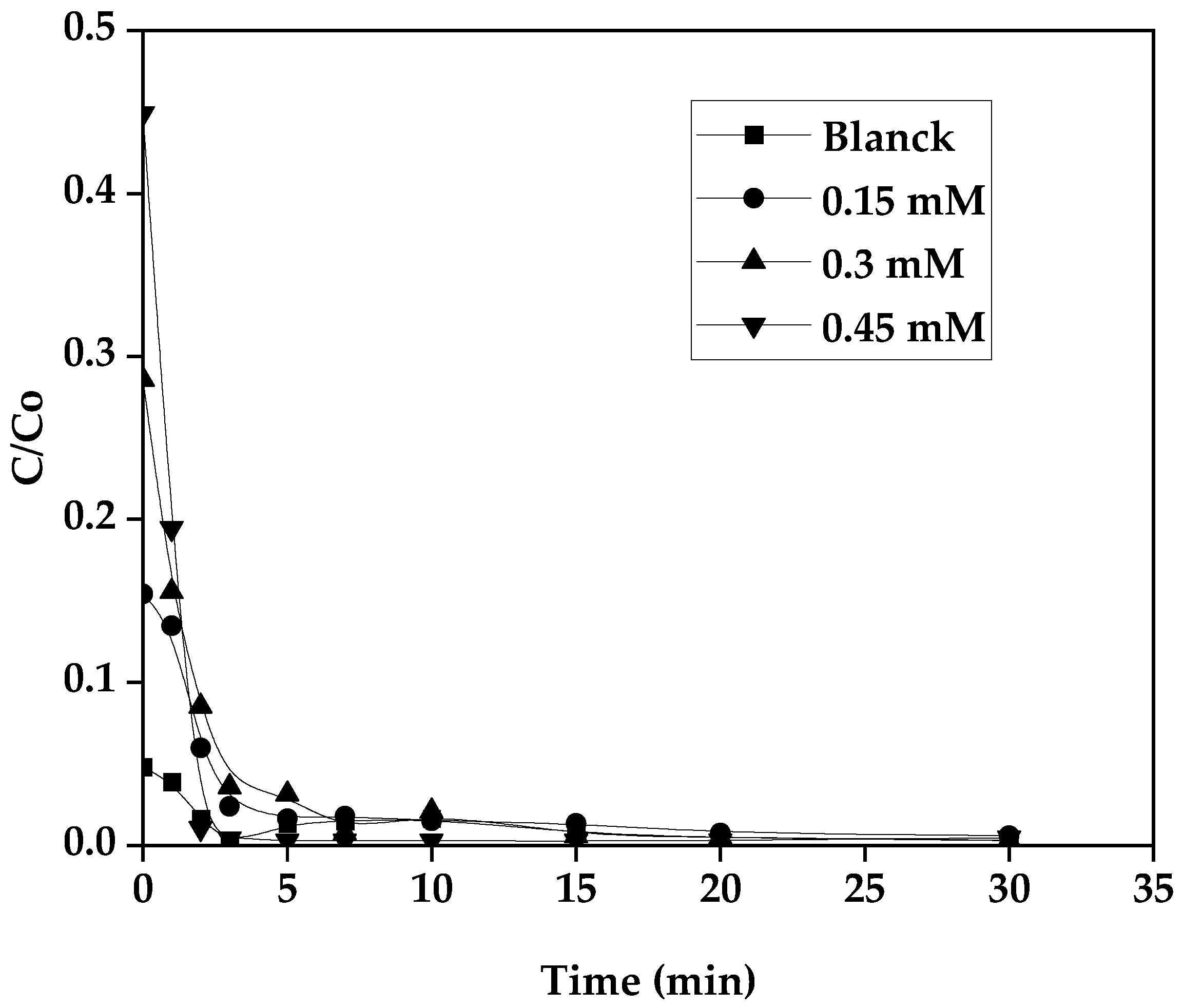
3.8. Influence of H2O2 Concentration
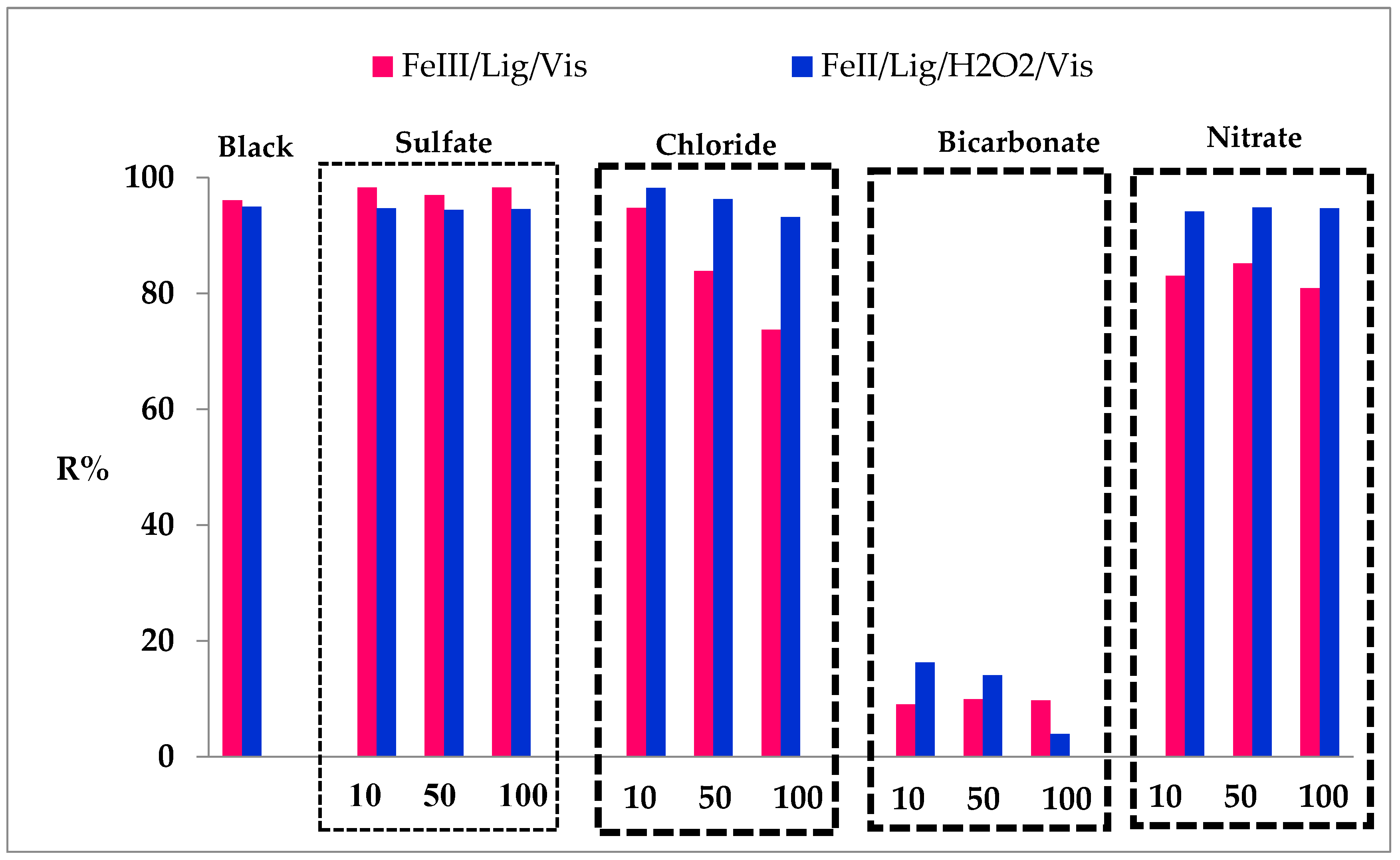
3.9. Effect of Ions for the Two Processes Involved (Fe(III)/Lig) and (Fe(II)/Lig)
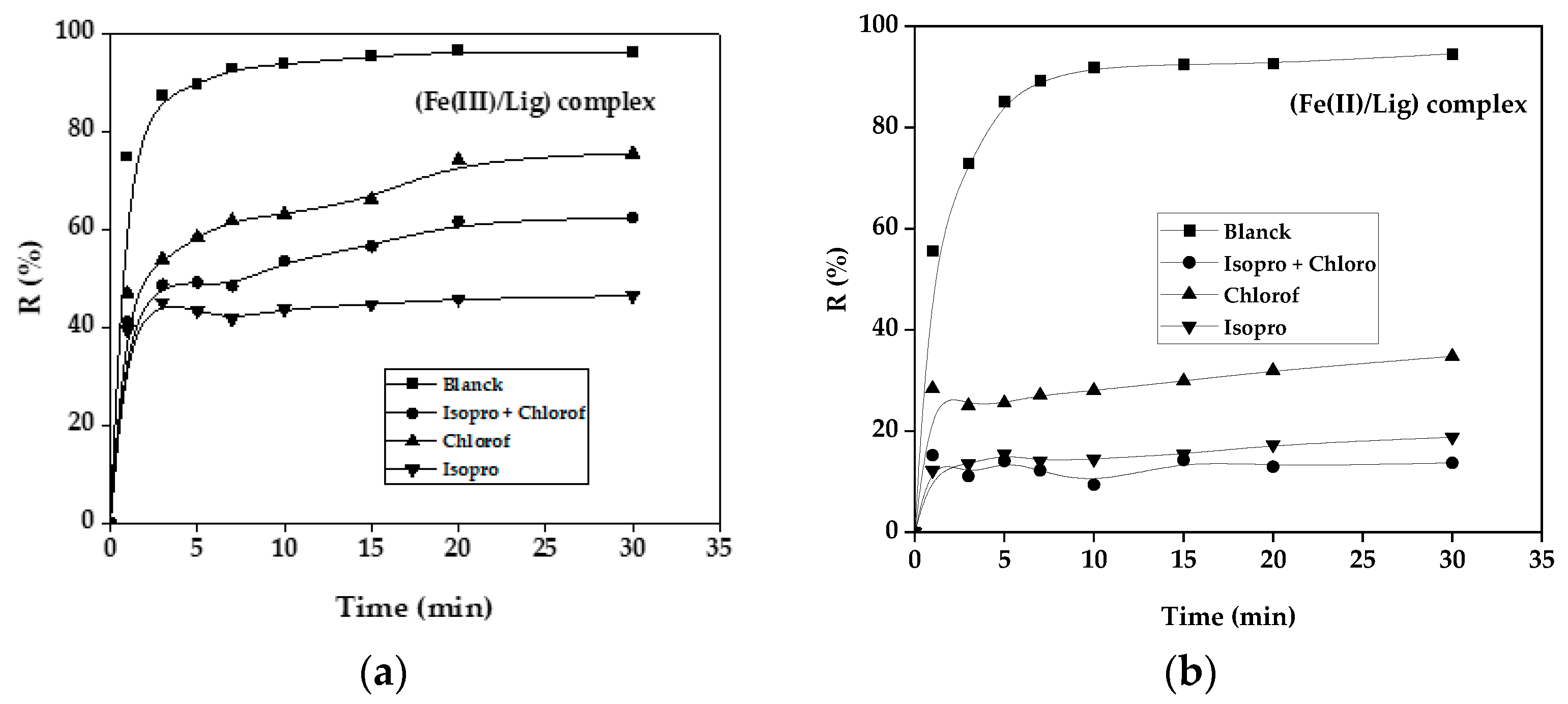
3.10. Effect of Scavengers
3.11. Kinetic Study of the Degradation of GS Dye with Complex (Fe(III)/Lig) and (Fe(II)/Lig) System
4. Conclusions
References
- Compton, M.; Willis, S.; Rezaie, B.; Humes, K. Food Processing Industry Energy and Water Consumption in the Pacific Northwest. Innovative Food Science & Emerging Technologies 2018, 47, 371–383. https://doi.org/10.1016/j.ifset.2018.04.001. [CrossRef]
- Obaideen, K.; Shehata, N.; Sayed, E.T.; Abdelkareem, M.A.; Mahmoud, M.S.; Olabi, A.G. The Role of Wastewater Treatment in Achieving Sustainable Development Goals (SDGs) and Sustainability Guideline. Energy Nexus 2022, 7, 100112. https://doi.org/10.1016/j.nexus.2022.100112. [CrossRef]
- Pham, V.H.T.; Kim, J.; Chang, S.; Bang, D. Investigating Bio-Inspired Degradation of Toxic Dyes Using Potential Multi-Enzyme Producing Extremophiles. Microorganisms 2023, 11, 1273. https://doi.org/10.3390/microorganisms11051273. [CrossRef]
- Koul, B.; Bhat, N.; Abubakar, M.; Mishra, M.; Arukha, A.P.; Yadav, D. Application of Natural Coagulants in Water Treatment: A Sustainable Alternative to Chemicals. Water 2022, 14, 3751. https://doi.org/10.3390/w14223751. [CrossRef]
- Clarizia, L.; Russo, D.; Di Somma, I.; Marotta, R.; Andreozzi, R. Homogeneous Photo-Fenton Processes at near Neutral PH: A Review. Applied Catalysis B: Environmental 2017, 209, 358–371. https://doi.org/10.1016/j.apcatb.2017.03.011. [CrossRef]
- Benramdane, I.K.; Nasrallah, N.; Amrane, A.; Kebir, M.; Trari, M.; Fourcade, F.; Assadi, A.A.; Maachi, R. Optimization of the Artificial Neuronal Network for the Degradation and Mineralization of Amoxicillin Photoinduced by the Complex Ferrioxalate with a Gradual and Progressive Approach of the Ligand. Journal of Photochemistry and Photobiology A: Chemistry 2021, 406, 112982. https://doi.org/10.1016/j.jphotochem.2020.112982. [CrossRef]
- Zhang, Y.-J.; Chen, J.-J.; Huang, G.-X.; Li, W.-W.; Yu, H.-Q.; Elimelech, M. Distinguishing Homogeneous Advanced Oxidation Processes in Bulk Water from Heterogeneous Surface Reactions in Organic Oxidation. Proc. Natl. Acad. Sci. U.S.A. 2023, 120, e2302407120. https://doi.org/10.1073/pnas.2302407120. [CrossRef]
- Xu, L.; Qi, L.; Han, Y.; Lu, W.; Han, J.; Qiao, W.; Mei, X.; Pan, Y.; Song, K.; Ling, C.; et al. Improvement of Fe2+/Peroxymonosulfate Oxidation of Organic Pollutants by Promoting Fe2+ Regeneration with Visible Light Driven g-C3N4 Photocatalysis. Chemical Engineering Journal 2022, 430, 132828. https://doi.org/10.1016/j.cej.2021.132828. [CrossRef]
- Garg, A.; Kaur, G.; Sangal, V.K.; Bajpai, P.K.; Upadhyay, S. Optimization Methodology Based on Neural Networks and Box-Behnken Design Applied to Photocatalysis of Acid Red 114 Dye. Environmental Engineering Research 2019, 25, 753–762. https://doi.org/10.4491/eer.2019.246. [CrossRef]
- Dissanayake, M.; Liyanage, N.; Herath, C.; Rathnayake, S.; Fernando, E.Y. Mineralization of Persistent Azo Dye Pollutants by a Microaerophilic Tropical Lake Sediment Mixed Bacterial Consortium. Environmental Advances 2021, 3, 100038. https://doi.org/10.1016/j.envadv.2021.100038. [CrossRef]
- Bouchelkia, N.; Tahraoui, H.; Amrane, A.; Belkacemi, H.; Bollinger, J.-C.; Bouzaza, A.; Zoukel, A.; Zhang, J.; Mouni, L. Jujube Stones Based Highly Efficient Activated Carbon for Methylene Blue Adsorption: Kinetics and Isotherms Modeling, Thermodynamics and Mechanism Study, Optimization via Response Surface Methodology and Machine Learning Approaches. Process Safety and Environmental Protection 2022.
- Tahraoui, H.; Belhadj, A.-E.; Hamitouche, A.; Bouhedda, M.; Amrane, A. Predicting the Concentration of Sulfate (SO42-) in Drinking Water Using Artificial Neural Networks: A Case Study: Médéa-Algeria. DESALINATION AND WATER TREATMENT 2021, 217, 181–194. https://doi.org/10.5004/dwt.2021.26813. [CrossRef]
- Tahraoui, H.; Amrane, A.; Belhadj, A.-E.; Zhang, J. Modeling the Organic Matter of Water Using the Decision Tree Coupled with Bootstrap Aggregated and Least-Squares Boosting. Environmental Technology & Innovation 2022, 27, 102419. https://doi.org/10.1016/j.eti.2022.102419. [CrossRef]
- Tahraoui, H.; Belhadj, A.E.; Hamitouche, A.E. Prediction of the Bicarbonate Amount in Drinking Water in the Region of Médéa Using Artificial Neural Network Modelling. Kemija u industriji 2020, 69, 595–602. https://doi.org/10.15255/KUI.2020.002. [CrossRef]
- Bousselma, A.; Abdessemed, D.; Tahraoui, H.; Amrane, A. Artificial Intelligence and Mathematical Modelling of the Drying Kinetics of Pre-Treated Whole Apricots. Kemija u industriji 2021. https://doi.org/10.15255/KUI.2020.079. [CrossRef]
- Yahoum, M.M.; Toumi, S.; Hentabli, S.; Tahraoui, H.; Lefnaoui, S.; Hadjsadok, A.; Amrane, A.; Kebir, M.; Moula, N.; Assadi, A.A. Experimental Analysis and Neural Network Modeling of the Rheological Behavior of Xanthan Gum and Its Derivatives. Materials 2023, 16, 2565. [CrossRef]
- Zamouche, M.; Chermat, M.; Kermiche, Z.; Tahraoui, H.; Kebir, M.; Bollinger, J.-C.; Amrane, A.; Mouni, L. Predictive Model Based on K-Nearest Neighbor Coupled with the Gray Wolf Optimizer Algorithm (KNN_GWO) for Estimating the Amount of Phenol Adsorption on Powdered Activated Carbon. Water 2023, 15, 493. [CrossRef]
- Zamouche, M.; Tahraoui, H.; Laggoun, Z.; Mechati, S.; Chemchmi, R.; Kanjal, M.I.; Amrane, A.; Hadadi, A.; Mouni, L. Optimization and Prediction of Stability of Emulsified Liquid Membrane (ELM): Artificial Neural Network. Processes 2023, 11, 364. [CrossRef]
- Tahraoui, H.; Belhadj, A.-E.; Amrane, A.; Houssein, E.H. Predicting the Concentration of Sulfate Using Machine Learning Methods. Earth Science Informatics 2022, 15, 1023–1044. https://doi.org/10.1007/s12145-022-00785-9. [CrossRef]
- Hadadi, A.; Imessaoudene, A.; Bollinger, J.-C.; Bouzaza, A.; Amrane, A.; Tahraoui, H.; Mouni, L. Aleppo Pine Seeds (Pinus Halepensis Mill.) as a Promising Novel Green Coagulant for the Removal of Congo Red Dye: Optimization via Machine Learning Algorithm. Journal of Environmental Management 2023, 331, 117286. [CrossRef]
- Tahraoui, H.; Belhadj, A.-E.; Triki, Z.; Boudella, N.R.; Seder, S.; Amrane, A.; Zhang, J.; Moula, N.; Tifoura, A.; Ferhat, R.; et al. Mixed Coagulant-Flocculant Optimization for Pharmaceutical Effluent Pretreatment Using Response Surface Methodology and Gaussian Process Regression. Process Safety and Environmental Protection 2022, S0957582022010102. https://doi.org/10.1016/j.psep.2022.11.045. [CrossRef]
- Nedjhioui, M.; Nasrallah, N.; Kebir, M.; Tahraoui, H.; Bouallouche, R.; Assadi, A.A.; Amrane, A.; Jaouadi, B.; Zhang, J.; Mouni, L. Designing an Efficient Surfactant–Polymer–Oil–Electrolyte System: A Multi-Objective Optimization Study. Processes 2023, 11, 1314. [CrossRef]
- Tahraoui, H.; Belhadj, A.E.; Moula, N.; Bouranene, S.; Amrane, A. Optimisation and Prediction of the Coagulant Dose for the Elimination of Organic Micropollutants Based on Turbidity. Kemija u industriji 2021. https://doi.org/10.15255/KUI.2021.001. [CrossRef]
- Imessaoudene, A.; Cheikh, S.; Hadadi, A.; Hamri, N.; Bollinger, J.-C.; Amrane, A.; Tahraoui, H.; Manseri, A.; Mouni, L. Adsorption Performance of Zeolite for the Removal of Congo Red Dye: Factorial Design Experiments, Kinetic, and Equilibrium Studies. Separations 2023, 10, 57. https://doi.org/10.3390/separations10010057. [CrossRef]
- Luna, A.J.; Chiavone-Filho, O.; Machulek, A.; De Moraes, J.E.F.; Nascimento, C.A.O. Photo-Fenton Oxidation of Phenol and Organochlorides (2,4-DCP and 2,4-D) in Aqueous Alkaline Medium with High Chloride Concentration. Journal of Environmental Management 2012, 111, 10–17. https://doi.org/10.1016/j.jenvman.2012.06.014. [CrossRef]
- Hussain, S.; Aneggi, E.; Goi, D. Catalytic Activity of Metals in Heterogeneous Fenton-like Oxidation of Wastewater Contaminants: A Review. Environ Chem Lett 2021, 19, 2405–2424. https://doi.org/10.1007/s10311-021-01185-z. [CrossRef]
- Girón-Navarro, R.; Martínez-Miranda, V.; Teutli-Sequeira, E.A.; Linares-Hernández, I.; Martínez-Cienfuegos, I.G.; Sánchez-Pozos, M.; Santoyo-Tepole, F. A Solar PhotoFenton Process with Calcium Peroxide from Eggshell and Ferrioxalate Complexes for the Degradation of the Commercial Herbicide 2,4-D in Water. Journal of Photochemistry and Photobiology A: Chemistry 2023, 438, 114550. https://doi.org/10.1016/j.jphotochem.2023.114550. [CrossRef]
- Lefnaoui, S.; Moulai-Mostefa, N. Investigation and Optimization of Formulation Factors of a Hydrogel Network Based on Kappa Carrageenan–Pregelatinized Starch Blend Using an Experimental Design. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2014, 458, 117–125.
- Srivastav, A.; Rawat, T.; Singh, J.; Nawaz, S. Multiobjective Grey Wolf Optimization Based Allocation of SVC in Power System. In Proceedings of the 2022 2nd International Conference on Innovative Sustainable Computational Technologies (CISCT); IEEE, 2022; pp. 1–5. [CrossRef]
- Tripathi, V.K.; Singh, M. An Efficient Metrics Based Self-adaptive Design Model by Multiobjective Gray Wolf Optimization with Extreme Learning Machine for Autonomic Computing System Application. Concurrency and Computation: Practice and Experience 2022, 34, e6609. [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical Review of Rate Constants for Reactions of Hydrated Electrons, Hydrogen Atoms and Hydroxyl Radicals (⋅OH/⋅O − in Aqueous Solution. Journal of Physical and Chemical Reference Data 1988, 17, 513–886. https://doi.org/10.1063/1.555805. [CrossRef]
- Wang, J.L.; Xu, L.J. Advanced Oxidation Processes for Wastewater Treatment: Formation of Hydroxyl Radical and Application. Critical Reviews in Environmental Science and Technology 2012, 42, 251–325. https://doi.org/10.1080/10643389.2010.507698. [CrossRef]
- Machulek, A.; Moraes, J.E.F.; Okano, L.T.; Silvérioc, C.A.; Quina, F.H. Photolysis of Ferric Ions in the Presence of Sulfate or Chloride Ions: Implications for the Photo-Fenton Process. Photochem Photobiol Sci 2009, 8, 985–991. https://doi.org/10.1039/b900553f. [CrossRef]
- Liao, C.-H.; Kang, S.-F.; Wu, F.-A. Hydroxyl Radical Scavenging Role of Chloride and Bicarbonate Ions in the H2O2/UV Process. Chemosphere 2001, 44, 1193–1200. https://doi.org/10.1016/S0045-6535(00)00278-2. [CrossRef]
- Devi, L.G.; Munikrishnappa, C.; Nagaraj, B.; Rajashekhar, K.E. Effect of Chloride and Sulfate Ions on the Advanced Photo Fenton and Modified Photo Fenton Degradation Process of Alizarin Red S. Journal of Molecular Catalysis A: Chemical 2013, 374–375, 125–131. https://doi.org/10.1016/j.molcata.2013.03.023. [CrossRef]
- Khan, J.; Tariq, M.; Muhammad, M.; H. Mehmood, M.; Ullah, I.; Raziq, A.; Akbar, F.; Saqib, M.; Rahim, A.; Niaz, A. Kinetic and Thermodynamic Study of Oxidative Degradation of Acid Yellow 17 Dye by Fenton-like Process: Effect of HCO3−, CO32−, Cl− and SO42− on Dye Degradation. Bull. Chem. Soc. Eth. 2019, 33, 243. https://doi.org/10.4314/bcse.v33i2.5. [CrossRef]
- Cheng, G.; Wan, J.; Li, Q.; Sun, L.; Zhang, Y.; Li, Z.; Dang, C.; Fu, J. Degradation of Reactive Brilliant Red X-3B by Photo-Fenton-like Process: Effects of Water Chemistry Factors and Degradation Mechanism. Water 2022, 14, 380. https://doi.org/10.3390/w14030380. [CrossRef]
- Vavilapalli, D.S.; Behara, S.; Peri, R.G.; Thomas, T.; Muthuraaman, B.; Rao, M.S.R.; Singh, S. Enhanced Photo-Fenton and Photoelectrochemical Activities in Nitrogen Doped Brownmillerite KBiFe2O5. Sci Rep 2022, 12, 5111. https://doi.org/10.1038/s41598-022-08966-8. [CrossRef]
- Hu, L.; Wang, P.; Xiong, S.; Chen, S.; Yin, X.; Wang, L.; Wang, H. The Attractive Efficiency Contributed by the In-Situ Reactivation of Ferrous Oxalate in Heterogeneous Fenton Process. Applied Surface Science 2019, 467–468, 185–192. https://doi.org/10.1016/j.apsusc.2018.10.151. [CrossRef]
- Ramírez-Carranza, D.R.; Macedo-Miranda, G.; González-Blanco, G.; Mireya-Martínez, S.; González-Juárez, J.C.; Beristain-Cardoso, R. Effect of Fe (II) Concentration on Metronidazole Degradation by Fenton Process: Performance and Kinetic Study. MRS Advances 2020, 5, 3265–3272. https://doi.org/10.1557/adv.2020.406. [CrossRef]
| (Fe(III)/Lig) | (Fe(II)/Lig) | |||||
|---|---|---|---|---|---|---|
| Level | -1 | 0 | 1 | -1 | 0 | 1 |
| X1 : Fe3+ (mM) | 0.15 | 0.2 | 0.25 | 0.15 | 0.2 | 0.25 |
| X2 : R (Fe2+/Oxy) | 1 | 2 | 3 | 1 | 2 | 3 |
| X3 : P (mg/L) | 10 | 15 | 20 | 10 | 15 | 20 |
| X4 : H2O2 (mM) | 0.1 | 0.1 | 0.1 | 0.05 | 0.1 | 0.15 |
| N | Fe(III) mM | R | P (mg/L) | R% | Predicted R% |
DCO% | Predicted DCO% |
|---|---|---|---|---|---|---|---|
| 1 | -1 | 0 | 1 | 93.63 | 91.765 | 90.92 | 90.8675 |
| 2 | -1 | 0 | -1 | 98.75 | 99.44375 | 95.83 | 96.4525 |
| 3 | 0 | 1 | -1 | 99.47 | 101.12875 | 94.17 | 98.0475 |
| 4 | 0 | 1 | 1 | 98.25 | 98.0525 | 97.33 | 92.9625 |
| 5 | 0 | 0 | 0 | 97.25 | 81.32125 | 96.00 | 74.23 |
| 6 | 1 | 0 | -1 | 99.64 | 89.40625 | 95.50 | 89.305 |
| 7 | 0 | 0 | 0 | 97.25 | 97.126666667 | 96.00 | 96.673333333 |
| 8 | -1 | -1 | 0 | 81.25 | 97.126666667 | 72.89 | 96.673333333 |
| 9 | 0 | -1 | -1 | 92.53 | 97.126666667 | 92.83 | 96.673333333 |
| 10 | 1 | 0 | 1 | 97.69 | 99.10375 | 98.00 | 96.805 |
| 11 | 0 | 0 | 0 | 96.88 | 97.55875 | 98.02 | 92.33 |
| 12 | 1 | -1 | 0 | 90.13 | 79.3675 | 89.89 | 75.7875 |
| 13 | -1 | 1 | 0 | 98.38 | 92.14125 | 96.22 | 88.3725 |
| 14 | 0 | -1 | 1 | 77.95 | 96.99625 | 74.58 | 97.3775 |
| 15 | 1 | 1 | 0 | 97.63 | 99.015 | 93.67 | 99.2925 |
| N | Fe(III) mM | R | P (mg/L) | H2O2 | R% | Predicted R% |
DCO% | Predicted DCO% |
|---|---|---|---|---|---|---|---|---|
| 1 | -1 | -1 | 0 | 0 | 93.3684211 | 91.783589792 | 84.66 | 84.30125 |
| 2 | -1 | 0 | -1 | 0 | 81.875 | 82.1340886 | 87.33 | 86.316666667 |
| 3 | -1 | 0 | 0 | -1 | 76.625 | 77.897187208 | 85.33 | 85.102083333 |
| 4 | -1 | 0 | 0 | 1 | 88.75 | 87.926861425 | 85.33 | 85.012083333 |
| 5 | -1 | 0 | 1 | 0 | 80.5 | 81.876075783 | 86.66 | 87.815 |
| 6 | -1 | 1 | 0 | 0 | 85.4601542 | 84.960772492 | 93.25 | 94.012916667 |
| 7 | 0 | -1 | -1 | 0 | 96.3461538 | 96.546645842 | 84 | 84.022083333 |
| 8 | 0 | -1 | 0 | -1 | 97.5 | 96.03454425 | 84 | 84.205 |
| 9 | 0 | -1 | 0 | 1 | 99.1153846 | 100.59058042 | 81.16 | 81.985 |
| 10 | 0 | -1 | 1 | 0 | 97.9230769 | 98.264594575 | 86.33 | 86.185416667 |
| 11 | 0 | 0 | -1 | -1 | 82.875 | 84.193519208 | 87.33 | 88.702916667 |
| 12 | 0 | 0 | -1 | 1 | 93.5 | 93.973193425 | 84.88 | 85.722916667 |
| 13 | 0 | 0 | 0 | 0 | 97.625 | 97.625 | 81.33 | 81.18 |
| 14 | 0 | 0 | 0 | 0 | 97.625 | 97.625 | 79.88 | 81.18 |
| 15 | 0 | 0 | 0 | 0 | 97.625 | 97.625 | 82.33 | 81.18 |
| 16 | 0 | 0 | 1 | -1 | 94.5 | 94.560506392 | 87.44 | 86.81125 |
| 17 | 0 | 0 | 1 | 1 | 90.25 | 89.465180608 | 88.77 | 87.61125 |
| 18 | 0 | 1 | -1 | 0 | 93.4293059 | 92.540380442 | 99.16 | 98.60375 |
| 19 | 0 | 1 | 0 | -1 | 96.9151671 | 95.45367925 | 95.83 | 95.491666667 |
| 20 | 0 | 1 | 0 | 1 | 94.1028278 | 95.581991517 | 95.25 | 95.531666667 |
| 21 | 0 | 1 | 1 | 0 | 97.4293059 | 96.681406075 | 97.16 | 96.437083333 |
| 22 | 1 | -1 | 0 | 0 | 99.5384615 | 100.57154303 | 84.66 | 84.11125 |
| 23 | 1 | 0 | -1 | 0 | 93.125 | 91.762632183 | 91 | 90.331666667 |
| 24 | 1 | 0 | 0 | -1 | 98.125 | 98.400730792 | 89 | 88.617083333 |
| 25 | 1 | 0 | 0 | 1 | 94.875 | 93.055405008 | 87 | 86.527083333 |
| 26 | 1 | 0 | 1 | 0 | 98.125 | 97.879619367 | 87.33 | 88.83 |
| 27 | 1 | 1 | 0 | 0 | 99.6863753 | 101.80490643 | 98.66 | 99.232916667 |
| Complex |
|
||||||||
| Response | a . R% | b. DCO% | |||||||
| i | Term | βi | Std Error | t Ratio | Prob > |t| | βi | Std Error | t Ratio | Prob > |t| |
| 0 | Intercept | 97.126667 | 0.884437 | 109.82 | <.0001* | 96.673333 | 1.44613 | 66.85 | <.0001* |
| 1 | X1 | 1.635 | 0.541605 | 3.02 | 0.0295* | 2.65 | 0.88557 | 2.99 | 0.0304* |
| 2 | X2 | 6.48375 | 0.541605 | 11.97 | <.0001* | 6.4 | 0.88557 | 7.23 | 0.0008* |
| 3 | X3 | -2.85875 | 0.541605 | -5.28 | 0.0032* | -2.1875 | 0.88557 | -2.47 | 0.0565 |
| 4 | X1*X2 | -2.4075 | 0.765945 | -3.14 | 0.0256* | -4.8875 | 1.252385 | -3.90 | 0.0114* |
| 5 | X1*X3 | 0.7925 | 0.765945 | 1.03 | 0.3482 | 1.8525 | 1.252385 | 1.48 | 0.1992 |
| 6 | X2*X3 | 3.34 | 0.765945 | 4.36 | 0.0073* | 5.3525 | 1.252385 | 4.27 | 0.0079* |
| 7 | X1*X1 | 0.0491667 | 0.797221 | 0.06 | 0.9532 | -1.585417 | 1.303524 | -1.22 | 0.2782 |
| 8 | X2*X2 | -5.328333 | 0.797221 | -6.68 | 0.0011* | -6.920417 | 1.303524 | -5.31 | 0.0032* |
| 9 | X3*X3 | 0.2516667 | 0.797221 | 0.32 | 0.7650 | -0.025417 | 1.303524 | -0.02 | 0.9852 |
| Complex |
|
||||||||
| Response | c. R% | d. DCO% | |||||||
| i | Term | βi | Std Error | t Ratio | Prob > |t| | βi | Std Error | t Ratio | Prob > |t| |
| 0 | Intercept | 97.625 | 0.922142 | 105.87 | <.0001* | 81.18 | 0.6679 | 121.55 | <.0001* |
| 1 | X1 | 6.4080218 | 0.461071 | 13.90 | <.0001* | 1.2575 | 0.33395 | 3.77 | 0.0027* |
| 2 | X2 | -1.397363 | 0.461071 | -3.03 | 0.0105* | 6.2083333 | 0.33395 | 18.59 | <.0001* |
| 3 | X3 | 1.4647436 | 0.461071 | 3.18 | 0.0080* | -0.000833 | 0.33395 | -0.00 | 0.9980 |
| 4 | X4 | 1.1710871 | 0.461071 | 2.54 | 0.0259* | -0.545 | 0.33395 | -1.63 | 0.1286 |
| 5 | X1*X2 | 2.0140452 | 0.798599 | 2.52 | 0.0268* | 1.3525 | 0.578418 | 2.34 | 0.0375* |
| 6 | X1*X3 | 1.59375 | 0.798599 | 2.00 | 0.0692 | -0.75 | 0.578418 | -1.30 | 0.2191 |
| 7 | X2*X3 | 0.6057692 | 0.798599 | 0.76 | 0.4628 | -1.0825 | 0.578418 | -1.87 | 0.0858 |
| 8 | X1*X4 | -3.84375 | 0.798599 | -4.81 | 0.0004* | -0.5 | 0.578418 | -0.86 | 0.4043 |
| 9 | X2*X4 | -1.106931 | 0.798599 | -1.39 | 0.1909 | 0.565 | 0.578418 | 0.98 | 0.3479 |
| 10 | X3*X4 | -3.71875 | 0.798599 | -4.66 | 0.0006* | 0.945 | 0.578418 | 1.63 | 0.1283 |
| 11 | X1*X1 | -5.219975 | 0.691607 | -7.55 | <.0001* | 3.1229167 | 0.500925 | 6.23 | <.0001* |
| 12 | X2*X2 | 2.3751778 | 0.691607 | 3.43 | 0.0049* | 6.1116667 | 0.500925 | 12.20 | <.0001* |
| 13 | X3*X3 | -3.991921 | 0.691607 | -5.77 | <.0001* | 4.0204167 | 0.500925 | 8.03 | <.0001* |
| 14 | X4*X4 | -3.084979 | 0.691607 | -4.46 | 0.0008* | 2.0116667 | 0.500925 | 4.02 | 0.0017* |
| Responses | Final Equation in Terms of Code of Independent Variables | P | F | R2 | RMSE |
|---|---|---|---|---|---|
| (Fe(III)/Lig) | |||||
| R% | 0.0116* | 8.0415 | 0.98 | 1.5319 | |
| DCO% | 0.01272 | 7.0211 | 0.96 | 2.5848 | |
| (Fe(II)/Lig) | |||||
| R% | 0.001* | 3.06125 | 0.97 | 1.5972 | |
| DCO% | 0.0491* | 0.8583 | 0.98 | 1.1568 | |
| Complex Fe(III)/Lig | Complex Fe(II)/Lig | |||
| 1st order | 2nd order | 1st order | 2nd order | |
| R2 | 0.6854 | 0.9684 | 0.5306 | 0.972 |
| Rate kinetic K |
0.4894 | 0.1447 | 0.1686 | 0.126 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




