Submitted:
20 May 2023
Posted:
22 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
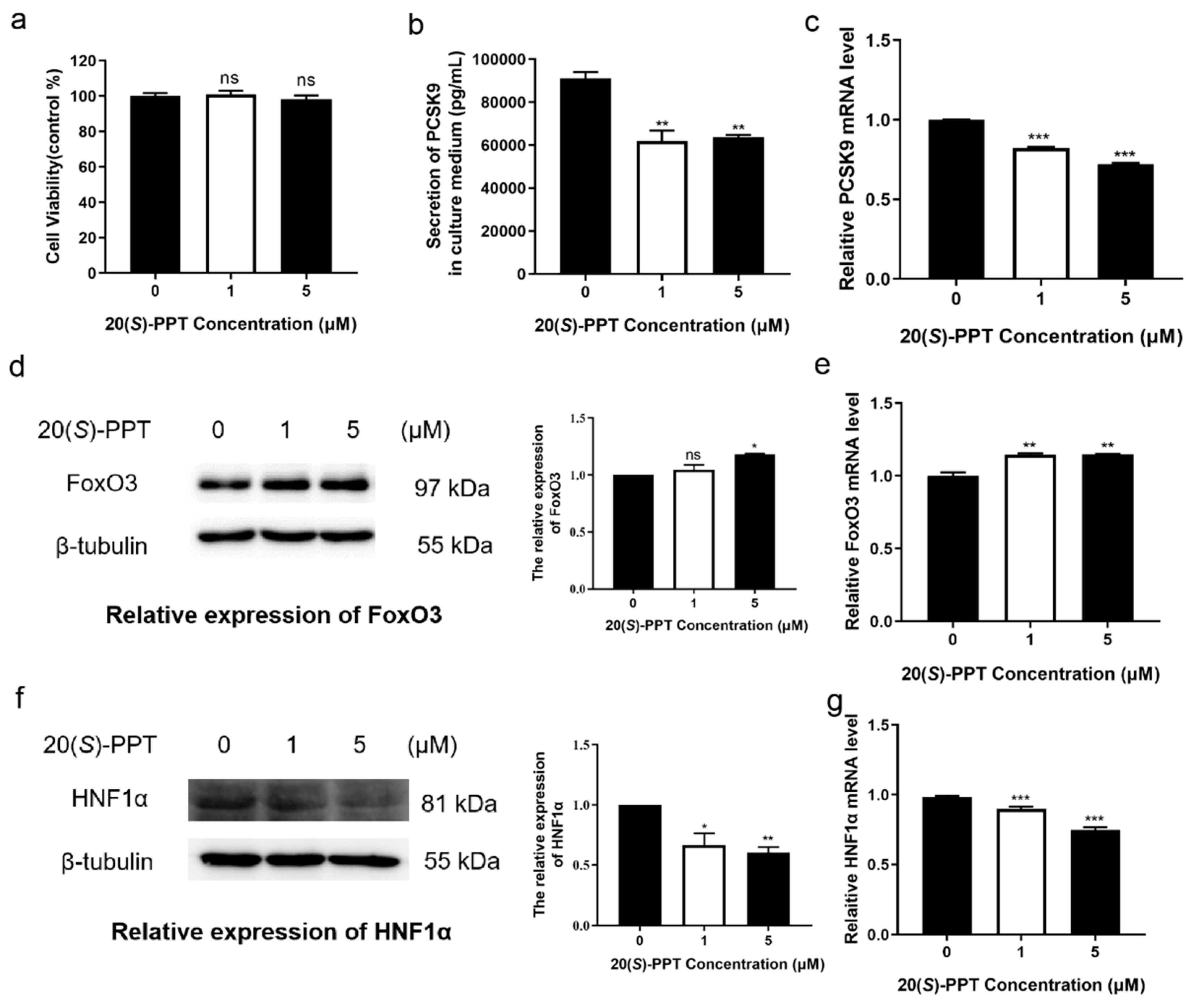
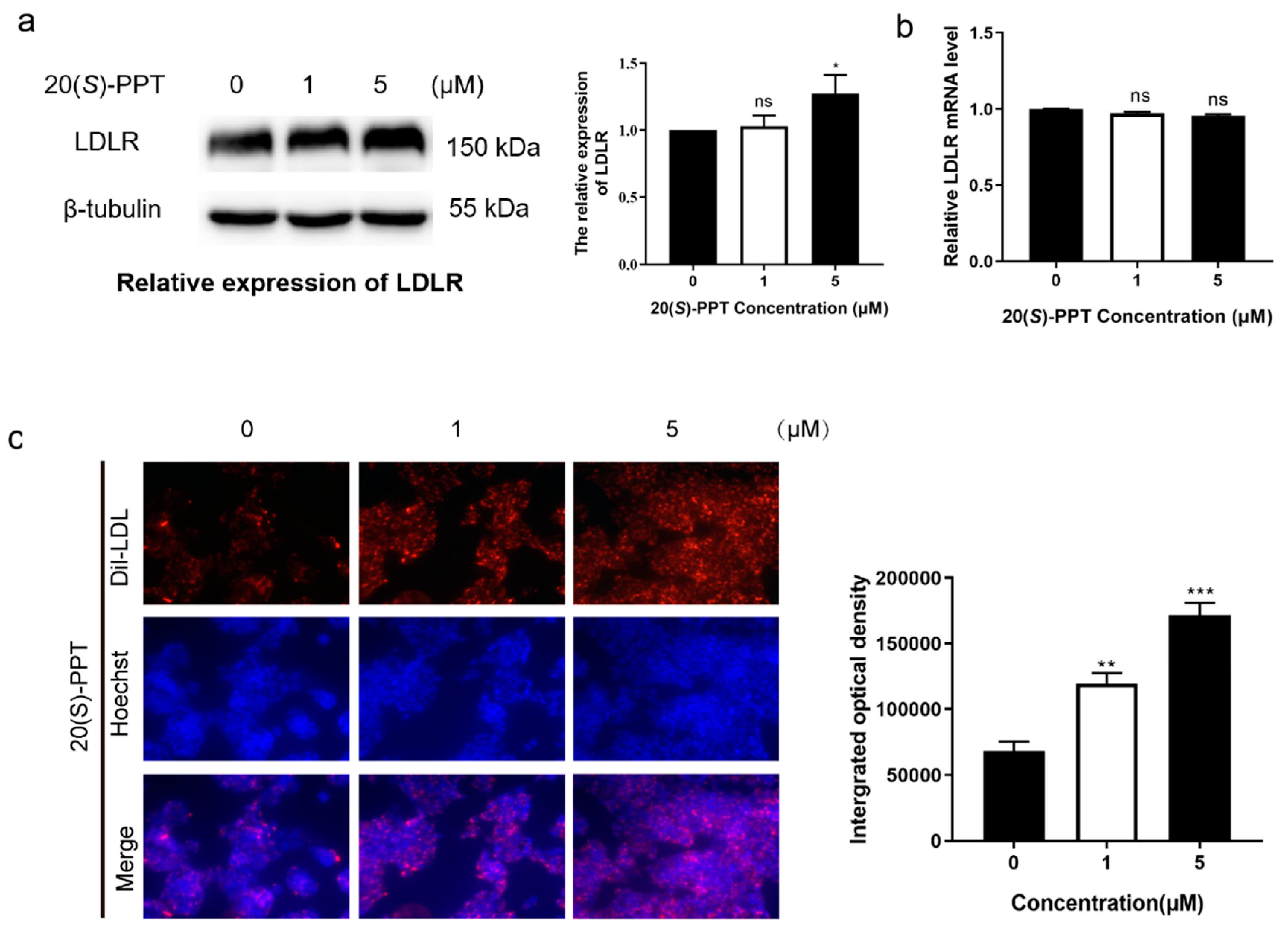
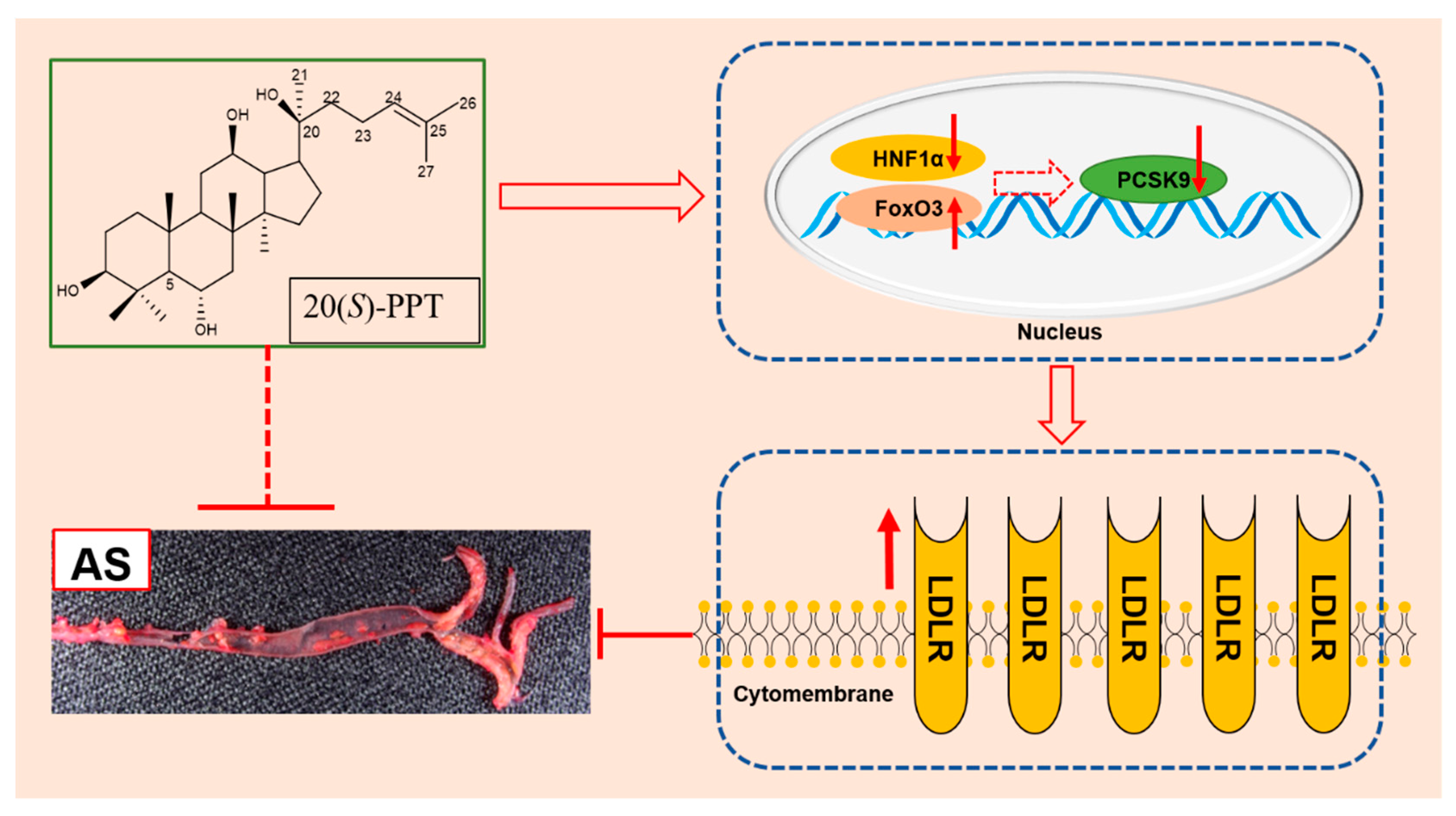
2.1. The transcription and secretion of PCSK9 can be inhibited by 20(S)-PPT via regulating the expression of FoxO3 and HNF1α in HepG2 cells
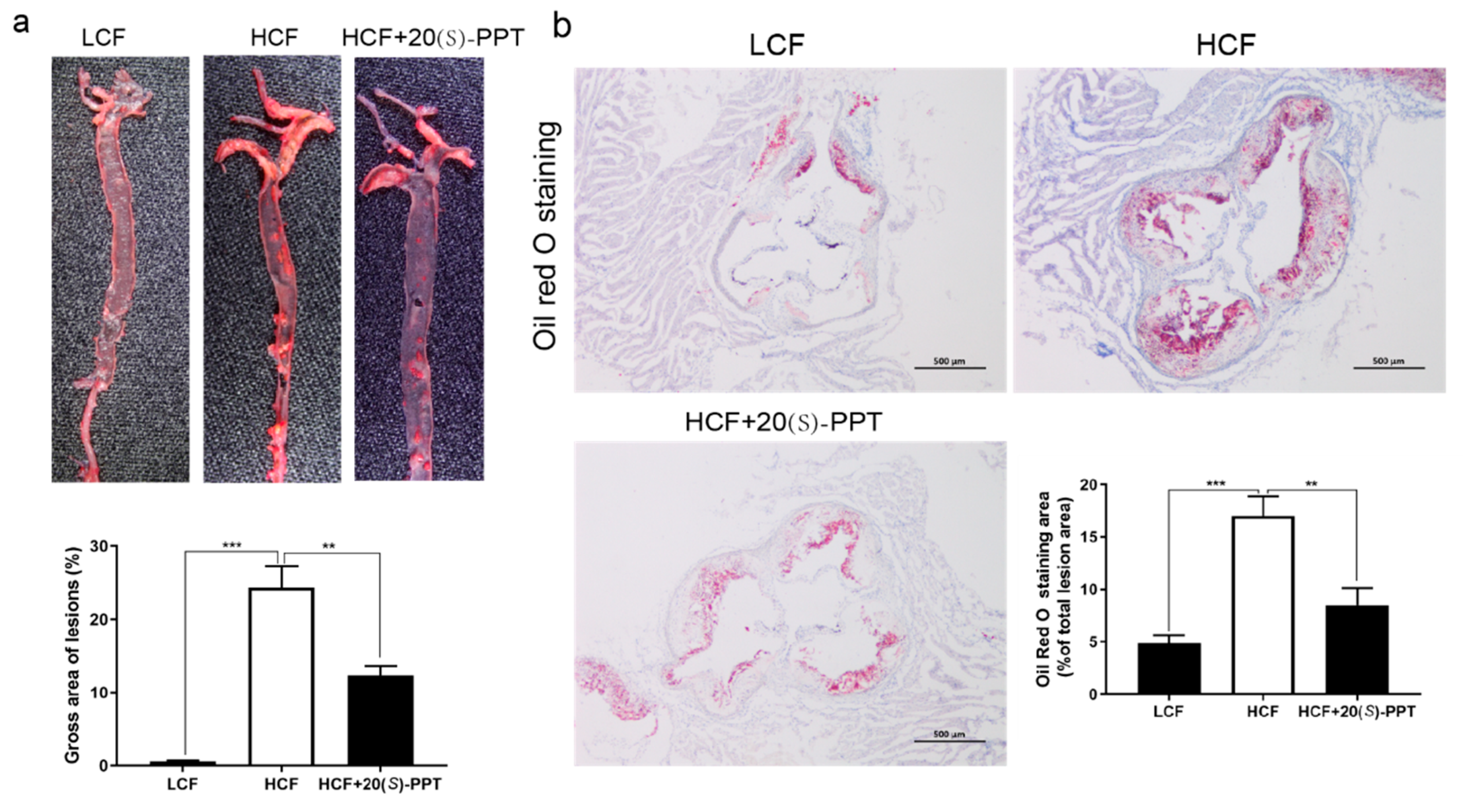
2.3. AS in ApoE-/- mice can be alleviated by 20(S)-PPT
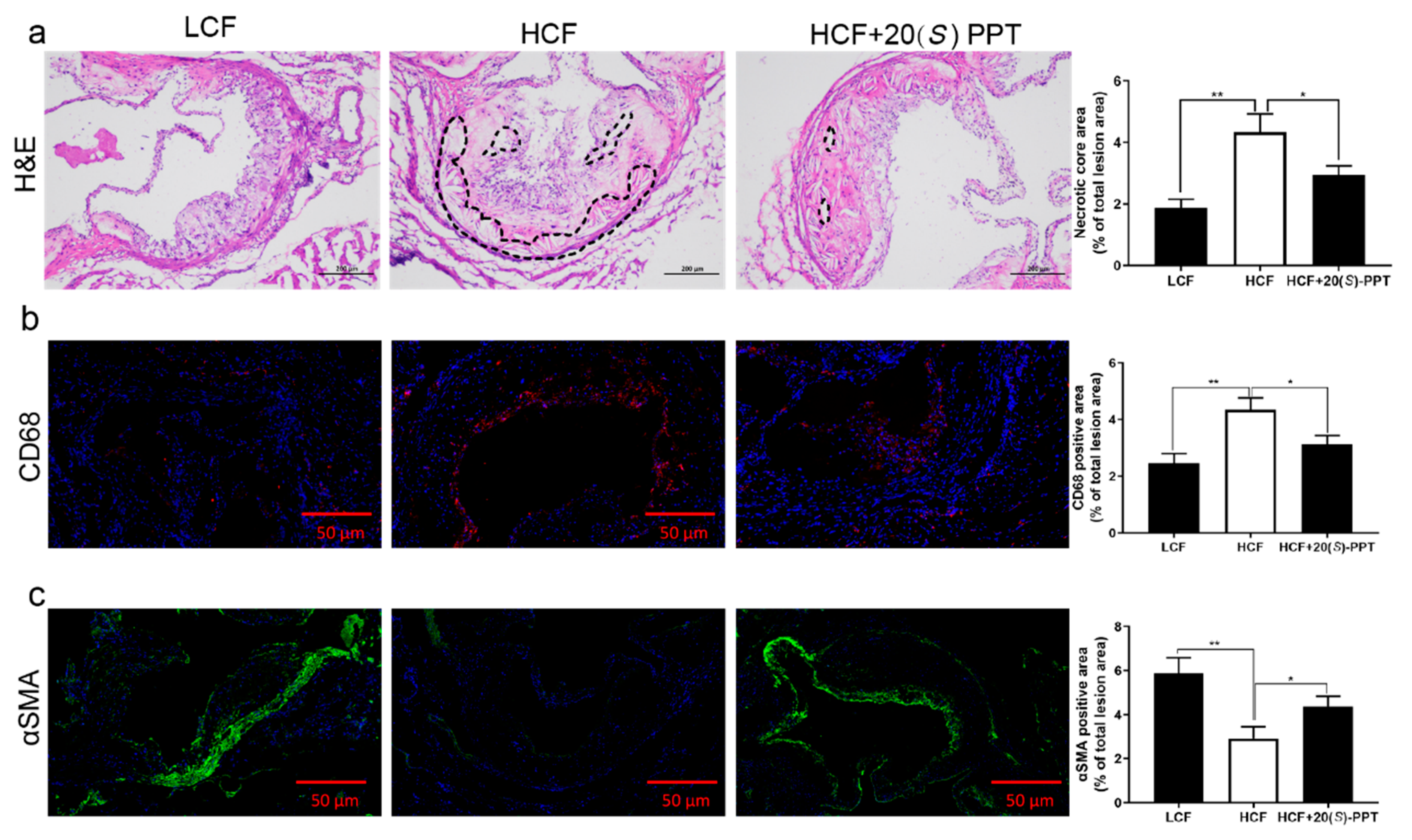
2.4. Treatment with 20(S)-PPT reduced the vulnerability of plaque formation in aortic roots and increased plaque stability
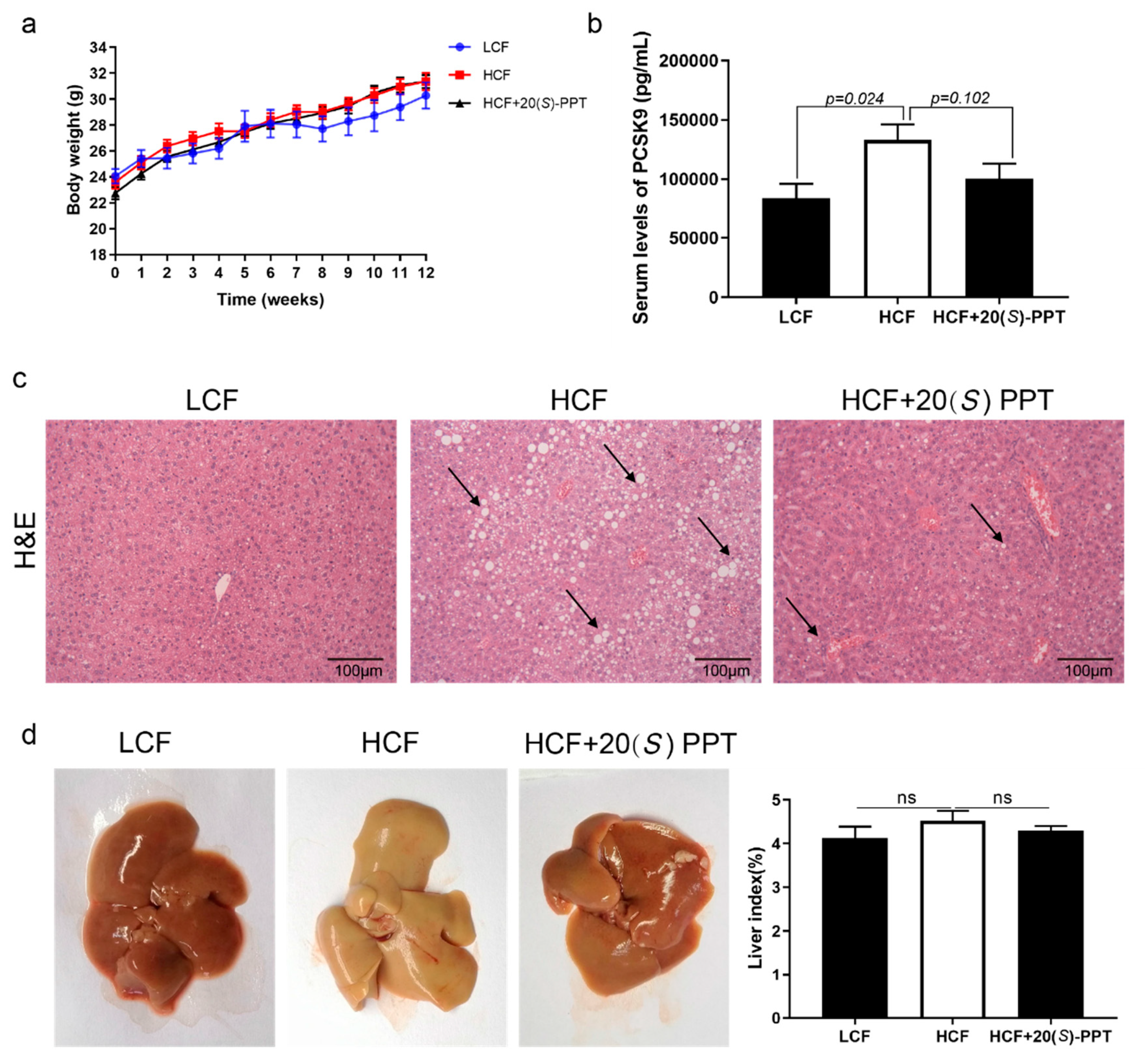
2.5. Effect of 20(S)-PPT on the hepatic lesions of ApoE-/- mice
3. Discussion
4. Materials and methods
4.1. Reagents
4.2. Western blotting
4.3. Cell culture
4.4. PCSK9 ELISA
4.5. Dil-LDL absorption test
4.6. RT-PCR
4.7. Animal experiments
4.8. Measurement of atherosclerotic lesions
4.9. Immunostaining assay
4.10. Hematoxylin and eosin (H&E) staining
4.11. Statistical analyses
Supplementary Materials
Funding
Conflicts of interest
References
- Xu, X.; Song, Z.; Mao, B.; Xu, G. Apolipoprotein A1-Related Proteins and Reverse Cholesterol Transport in Antiatherosclerosis Therapy: Recent Progress and Future Perspectives. Cardiovasc. Ther. 2022, 2022, 4610834. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.X.; Zhang, M.; Long, S.Y.; Tuo, Q.H.; Tian, Y.; Chen, J.X.; Zhang, C.P.; Liao, D.F. Cholesterol in LDL receptor recycling and degradation. Clin. Chim. Acta Int. J. Clin. Chem. 2020, 500, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Bittner, V.A.; Giugliano, R.P.; Brinton, E.A.; Guyton, J.R. PCSK9 inhibitors for prevention of atherosclerotic cardiovascular disease. J. Clin. Lipidol. 2018, 12, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Song, K.H.; Kim, Y.H.; Im, A.R.; Kim, Y.H. Black Raspberry Extract Enhances LDL Uptake in HepG2 Cells by Suppressing PCSK9 Expression to Upregulate LDLR Expression. J. Med. Food 2018, 21, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Yan, B.; Gui, Y.; Tang, Z.; Tai, S.; Zhou, S.; Zheng, X.L. Physiology and role of PCSK9 in vascular disease: Potential impact of localized PCSK9 in vascular wall. J. Cell. Physiol. 2021, 236, 2333–2351. [Google Scholar] [CrossRef] [PubMed]
- Sobati, S.; Shakouri, A.; Edalati, M.; Mohammadnejad, D.; Parvan, R.; Masoumi, J.; Abdolalizadeh, J. PCSK9: A Key Target for the Treatment of Cardiovascular Disease (CVD). Adv. Pharm. Bull. 2020, 10, 502–511. [Google Scholar] [CrossRef]
- Pokrywka, G.S. PCSK9 inhibitors: A non-statin cholesterol-lowering treatment option. Postgrad. Med. 2018, 130, 287–298. [Google Scholar] [CrossRef]
- Raal, F.J.; Stein, E.A.; Dufour, R.; Turner, T.; Civeira, F.; Burgess, L.; Langslet, G.; Scott, R.; Olsson, A.G.; Sullivan, D.; et al. PCSK9 inhibition with evolocumab (AMG 145) in heterozygous familial hypercholesterolaemia (RUTHERFORD-2): A randomised, double-blind, placebo-controlled trial. Lancet 2015, 385, 331–340. [Google Scholar] [CrossRef]
- McDonagh, M.; Peterson, K.; Holzhammer, B.; Fazio, S. A Systematic Review of PCSK9 Inhibitors Alirocumab and Evolocumab. J. Manag. Care Spec. Pharm. 2016, 22, 641–653. [Google Scholar] [CrossRef]
- Guedeney, P.; Sorrentino, S.; Giustino, G.; Chapelle, C.; Laporte, S.; Claessen, B.E.; Ollier, E.; Camaj, A.; Kalkman, D.N.; Vogel, B.; et al. Indirect comparison of the efficacy and safety of alirocumab and evolocumab: A systematic review and network meta-analysis. Eur. Heart Journal. Cardiovasc. Pharmacother. 2021, 7, 225–235. [Google Scholar] [CrossRef]
- Zhao, Z.; Tuakli-Wosornu, Y.; Lagace, T.A.; Kinch, L.; Grishin, N.V.; Horton, J.D.; Cohen, J.C.; Hobbs, H.H. Molecular characterization of loss-of-function mutations in PCSK9 and identification of a compound heterozygote. Am. J. Hum. Genet. 2006, 79, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Liu, Z.; Hu, X.; Liu, X.; Gui, L.; Cai, Z.; Dai, C. Protective Effect of Panax Notoginseng Saponins on Apolipoprotein-E-deficient Atherosclerosis-prone Mice. Curr. Pharm. Des. 2022, 28, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, H.; Udayama, M.; Dokan, R.; Kasai, R.; Yamasaki, K.; Tanaka, O.J.N.M. Identification and quantification of 20(S)-protopanaxatriol and its 20S,24S-epoxide in human urine with the treatment of alkaline cleavage after the ingestion of Sanchi Ginseng, the roots of Panax notoginseng. 2002, 56, 34–39. [Google Scholar]
- Sun, H.; Yang, Z.; Ye, Y. Structure and biological activity of protopanaxatriol-type saponins from the roots of Panax notoginseng. Int. Immunopharmacol. 2006, 6, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, S.; Qu, B.; Chi, H.; Quan, Y.; Wu, X. Efficient separation determination of protopanaxatriol ginsenosides Rg1, Re, Rf, Rh1, Rg2 by HPLC. J. Pharm. Biomed. Anal. 2019, 170, 48–53. [Google Scholar] [CrossRef]
- Fan, J.S.; Liu, D.N.; Huang, G.; Xu, Z.Z.; Jia, Y.; Zhang, H.G.; Li, X.H.; He, F.T. Panax notoginseng saponins attenuate atherosclerosis via reciprocal regulation of lipid metabolism and inflammation by inducing liver X receptor alpha expression. J. Ethnopharmacol. 2012, 142, 732–738. [Google Scholar] [CrossRef]
- Yuan, Z.; Liao, Y.; Tian, G.; Li, H.; Jia, Y.; Zhang, H.; Tan, Z.; Li, X.; Deng, W.; Liu, K.; et al. Panax notoginseng saponins inhibit Zymosan A induced atherosclerosis by suppressing integrin expression, FAK activation and NF-κB translocation. J. Ethnopharmacol. 2011, 138, 150–155. [Google Scholar] [CrossRef]
- Jia, Y.; Li, Z.Y.; Zhang, H.G.; Li, H.B.; Liu, Y.; Li, X.H. Panax notoginseng saponins decrease cholesterol ester via up-regulating ATP-binding cassette transporter A1 in foam cells. J. Ethnopharmacol. 2010, 132, 297–302. [Google Scholar] [CrossRef]
- Quan, H.Y.; Yuan, H.D.; Jung, M.S.; Ko, S.K.; Park, Y.G.; Chung, S.H. Ginsenoside Re lowers blood glucose and lipid levels via activation of AMP-activated protein kinase in HepG2 cells and high-fat diet fed mice. Int. J. Mol. Med. 2012, 29, 73–80. [Google Scholar] [CrossRef]
- Oh, G.S.; Yoon, J.; Lee, G.G.; Oh, W.K.; Kim, S.W. 20(S)-protopanaxatriol inhibits liver X receptor α-mediated expression of lipogenic genes in hepatocytes. J. Pharmacol. Sci. 2015, 128, 71–77. [Google Scholar] [CrossRef]
- Yang, R.; Yin, D.; Yang, D.; Liu, X.; Zhou, Q.; Pan, Y.; Li, J.; Li, S. Xinnaokang improves cecal microbiota and lipid metabolism to target atherosclerosis. Lett. Appl. Microbiol. 2021, 73, 779–792. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Y.S.; Huang, Y.P.; Jiang, C.H.; Gao, M.; Zheng, X.; Yin, Z.Q.; Zhang, J. Gypenoside LVI improves hepatic LDL uptake by decreasing PCSK9 and upregulating LDLR expression. Phytomedicine Int. J. Phytother. Phytopharm. 2021, 91, 153688. [Google Scholar] [CrossRef] [PubMed]
- Bays, H.E.; Jones, P.H.; Orringer, C.E.; Brown, W.V.; Jacobson, T.A. National Lipid Association Annual Summary of Clinical Lipidology 2016. J. Clin. Lipidol. 2016, 10, S1–S43. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.J.; Jin, J.L.; Guo, L.N.; Sun, J.; Wu, N.Q.; Guo, Y.L.; Liu, G.; Dong, Q.; Li, J.J. Beneficial impact of epigallocatechingallate on LDL-C through PCSK9/LDLR pathway by blocking HNF1α and activating FoxO3a. J. Transl. Med. 2020, 18, 195. [Google Scholar] [CrossRef] [PubMed]
- Tangirala, R.K.; Rubin, E.M.; Palinski, W. Quantitation of atherosclerosis in murine models: Correlation between lesions in the aortic origin and in the entire aorta, and differences in the extent of lesions between sexes in LDL receptor-deficient and apolipoprotein E-deficient mice. J. Lipid Res. 1995, 36, 2320–2328. [Google Scholar] [CrossRef] [PubMed]
- Daugherty, A.; Tall, A.R.; Daemen, M.; Falk, E.; Fisher, E.A.; García-Cardeña, G.; Lusis, A.J.; Owens, A.P., 3rd; Rosenfeld, M.E.; Virmani, R. Recommendation on Design, Execution, and Reporting of Animal Atherosclerosis Studies: A Scientific Statement From the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e131–e157. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Sniffen, S.; McGill Percy, K.C.; Pallaval, V.B.; Chidipi, B. Gut Dysbiosis and Immune System in Atherosclerotic Cardiovascular Disease (ACVD). Microorganisms 2022, 10, 108. [Google Scholar] [CrossRef]
- Lambert, G.; Sjouke, B.; Choque, B.; Kastelein, J.J.; Hovingh, G.K. The PCSK9 decade. J. Lipid Res. 2012, 53, 2515–2524. [Google Scholar] [CrossRef]
- Nishikido, T.; Ray, K.K. Non-antibody Approaches to Proprotein Convertase Subtilisin Kexin 9 Inhibition: siRNA, Antisense Oligonucleotides, Adnectins, Vaccination, and New Attempts at Small-Molecule Inhibitors Based on New Discoveries. Front. Cardiovasc. Med. 2018, 5, 199. [Google Scholar] [CrossRef]
- Giunzioni, I.; Tavori, H. New developments in atherosclerosis: Clinical potential of PCSK9 inhibition. Vasc. Health Risk Manag. 2015, 11, 493–501. [Google Scholar] [CrossRef]
- Zhang, H.; Min, L.; Lu, X.; Hong, F.; Yang, S. New Progress in Ginseng Anti-Aging Effects on Blood and Cardiovascular System.
- Chen, H.C.; Chen, P.Y.; Wu, M.J.; Tai, M.H.; Yen, J.H. Tanshinone IIA Modulates Low Density Lipoprotein Uptake via Down-Regulation of PCSK9 Gene Expression in HepG2 Cells. PLoS ONE 2016, 11, e0162414. [Google Scholar] [CrossRef] [PubMed]
- Mbikay, M.; Sirois, F.; Simoes, S.; Mayne, J.; Chrétien, M. Quercetin-3-glucoside increases low-density lipoprotein receptor (LDLR) expression, attenuates proprotein convertase subtilisin/kexin 9 (PCSK9) secretion, and stimulates LDL uptake by Huh7 human hepatocytes in culture. FEBS Open Bio 2014, 4, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Wu, M.; Li, H.; Kraemer, F.B.; Adeli, K.; Seidah, N.G.; Park, S.W.; Liu, J. Strong induction of PCSK9 gene expression through HNF1alpha and SREBP2: Mechanism for the resistance to LDL-cholesterol lowering effect of statins in dyslipidemic hamsters. J. Lipid Res. 2010, 51, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Shende, V.R.; Wu, M.; Singh, A.B.; Dong, B.; Kan, C.F.; Liu, J. Reduction of circulating PCSK9 and LDL-C levels by liver-specific knockdown of HNF1α in normolipidemic mice. J. Lipid Res. 2015, 56, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Dong, B.; Park, S.W.; Lee, H.S.; Chen, W.; Liu, J. Hepatocyte nuclear factor 1alpha plays a critical role in PCSK9 gene transcription and regulation by the natural hypocholesterolemic compound berberine. J. Biol. Chem. 2009, 284, 28885–28895. [Google Scholar] [CrossRef] [PubMed]
- Tai, M.H.; Chen, P.K.; Chen, P.Y.; Wu, M.J.; Ho, C.T.; Yen, J.H. Curcumin enhances cell-surface LDLR level and promotes LDL uptake through downregulation of PCSK9 gene expression in HepG2 cells. Mol. Nutr. Food Res. 2014, 58, 2133–2145. [Google Scholar] [CrossRef]
- Ben Djoudi Ouadda, A.; Gauthier, M.S.; Susan-Resiga, D.; Girard, E.; Essalmani, R.; Black, M.; Marcinkiewicz, J.; Forget, D.; Hamelin, J.; Evagelidis, A.; et al. Ser-Phosphorylation of PCSK9 (Proprotein Convertase Subtilisin-Kexin 9) by Fam20C (Family With Sequence Similarity 20, Member C) Kinase Enhances Its Ability to Degrade the LDLR (Low-Density Lipoprotein Receptor). Arterioscler. Thromb. Vasc. Biol. 2019, 39, 1996–2013. [Google Scholar] [CrossRef]
- Tagliabracci, V.S.; Wiley, S.E.; Guo, X.; Kinch, L.N.; Durrant, E.; Wen, J.; Xiao, J.; Cui, J.; Nguyen, K.B.; Engel, J.L.; et al. A Single Kinase Generates the Majority of the Secreted Phosphoproteome. Cell 2015, 161, 1619–1632. [Google Scholar] [CrossRef]
- Andrés-Manzano, M.J.; Andrés, V.; Dorado, B. Oil Red O and Hematoxylin and Eosin Staining for Quantification of Atherosclerosis Burden in Mouse Aorta and Aortic Root. Methods Mol. Biol. 2015, 1339, 85–99. [Google Scholar] [CrossRef]
- Gough, P.J.; Gordon, S.; Greaves, D.R. The use of human CD68 transcriptional regulatory sequences to direct high-level expression of class A scavenger receptor in macrophages in vitro and in vivo. Immunology 2001, 103, 351–361. [Google Scholar] [CrossRef]
| Nonstandard Abbreviations and Acronyms | |
|---|---|
| CVDs | cardiovascular diseases |
| LDL-C | Low-density lipoprotein cholesterol |
| HDL-C | high-density lipoprotein cholesterol |
| RT-PCR | real time-polymerase chain reaction |
| ELISA | Enzyme-linked Immunosorbent Assay |
| PCSK9 | Proprotein Convertase Subtilisin/Kexin Type 9 |
| 20(S)-PPT | 20(S)-protopanaxatriol |
| LDLR | Low-density lipoprotein receptor |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| αSMA | αSmooth Muscle Actin |
| FoxO3 | Forkhead box O3 |
| HCF | high-cholesterol diet |
| LCF | low-cholesterol diet |
| ERK | extracellular regulated protein kinases |
| FBS | fetal bovine serum |
| BCA | Bicinchoninic acid |
| ECL | Enhanced chemiluminescence |
| PMSF | phenylmethanesulfonyl fluoride |
| Gene | Forward primer (5′-3′) | Reverse primer (5′-3′) |
|---|---|---|
| ldlr | GAACCCATCAAAGAGTGCG | TCTTCCTGACCTCGTGCC |
| pcsk9 | CCAAGCCTCTTCTTACTTCACC | GCATCGTTCTGCCATCACT |
| FoxO3 | GCAAACCTGCCCGTCAT | TCAAAGTTAAAATCCAACCCAT |
| Hnf1α | ACGACGATGGGGAAGACTTC | GACTTGACCATCTTCGCCAC |
| β-actin | CCCTGGCACCCAGCAC | GCCGATCCACACGGAGTAC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




