Submitted:
16 May 2023
Posted:
18 May 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
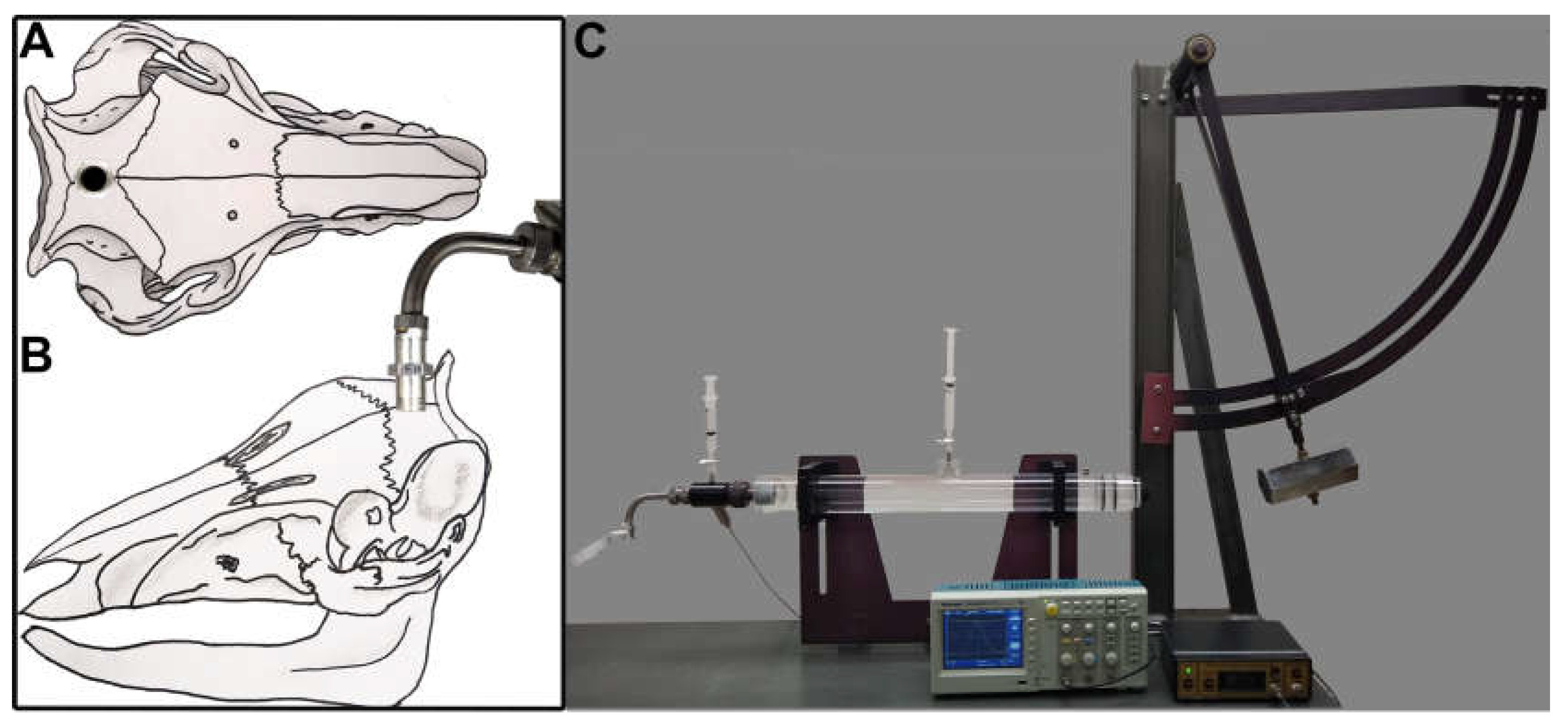
2.2. Surgical preparation and injury induction
2.3. Physiology
2.4. Tissue processing
2.5. Immunohistochemistry
2.5.1. Previous labeling of thalamic and corpus callosal tissue for axonal injury and microglial activity index assessments. All immunohistological labeling and analyses that was used to determine injury model scalability was done on freshly generated tissue during the conduct of the studies published previously Lafrenaye et al., 2015[54]. Mean thalamic data for injured and sham groups was previously published in Lafrenaye et al., 2015[54], however, corpus callosum data was not reported. For immunohistological labeling, a random well (1–6) was selected using a random number generator and six pieces of tissue containing the thalamic or twelve sections containing the corpus callosum were taken for immunolabeling. Tissue from all animals was processed concomitantly to reduce variability between animals.
2.5.2. Labeling of stored thalamic tissue for immunohistochemistry efficacy assessments.
2.6. Image analysis
2.6.1. Quantitative image analysis of diffuse axonal injury
2.6.2. Quantitative image analysis of microglial activity index
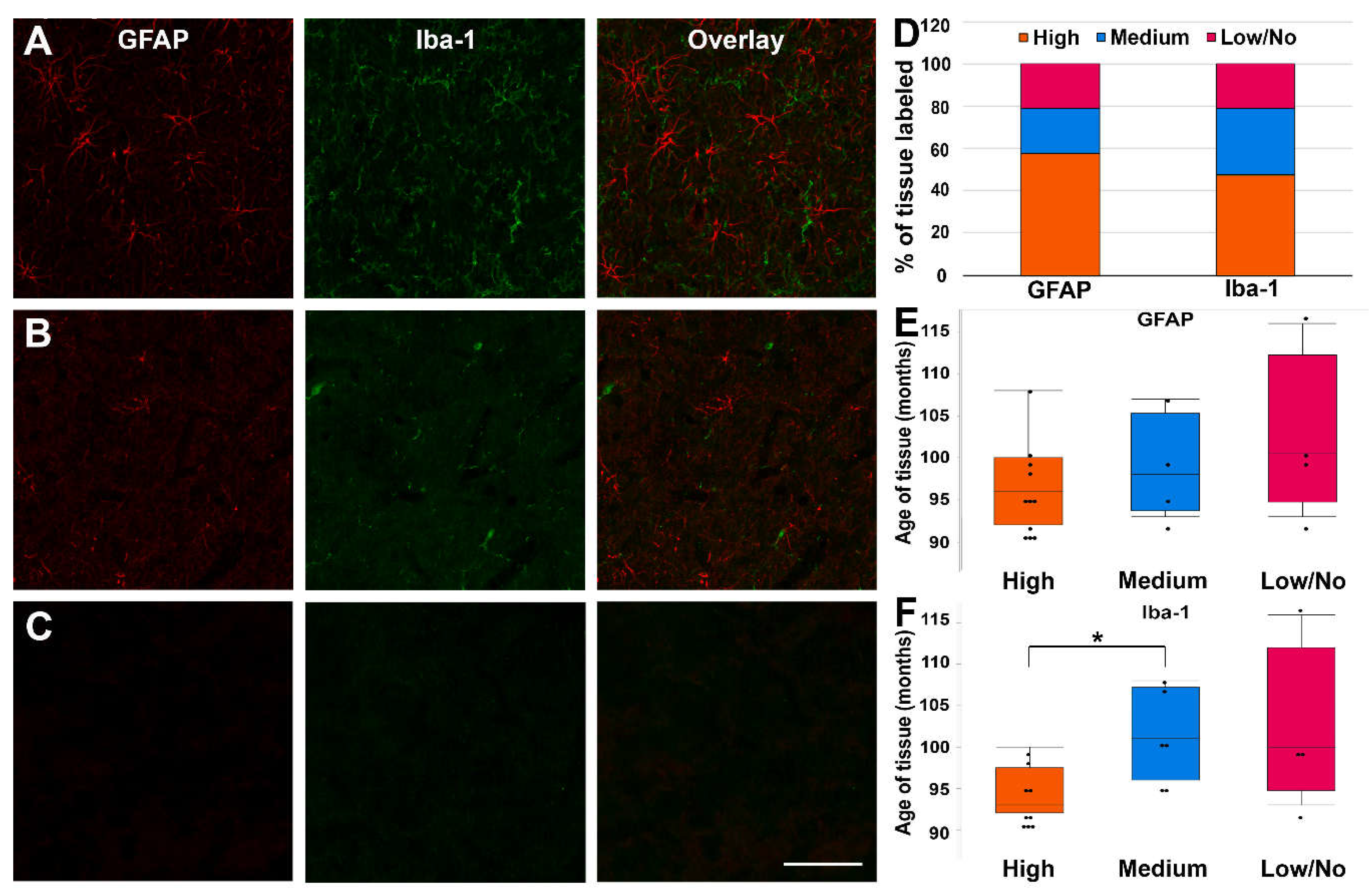
2.6.3. Qualitative analysis of glial labeling in old pig tissue
2.7. Statistical analysis
3. Results
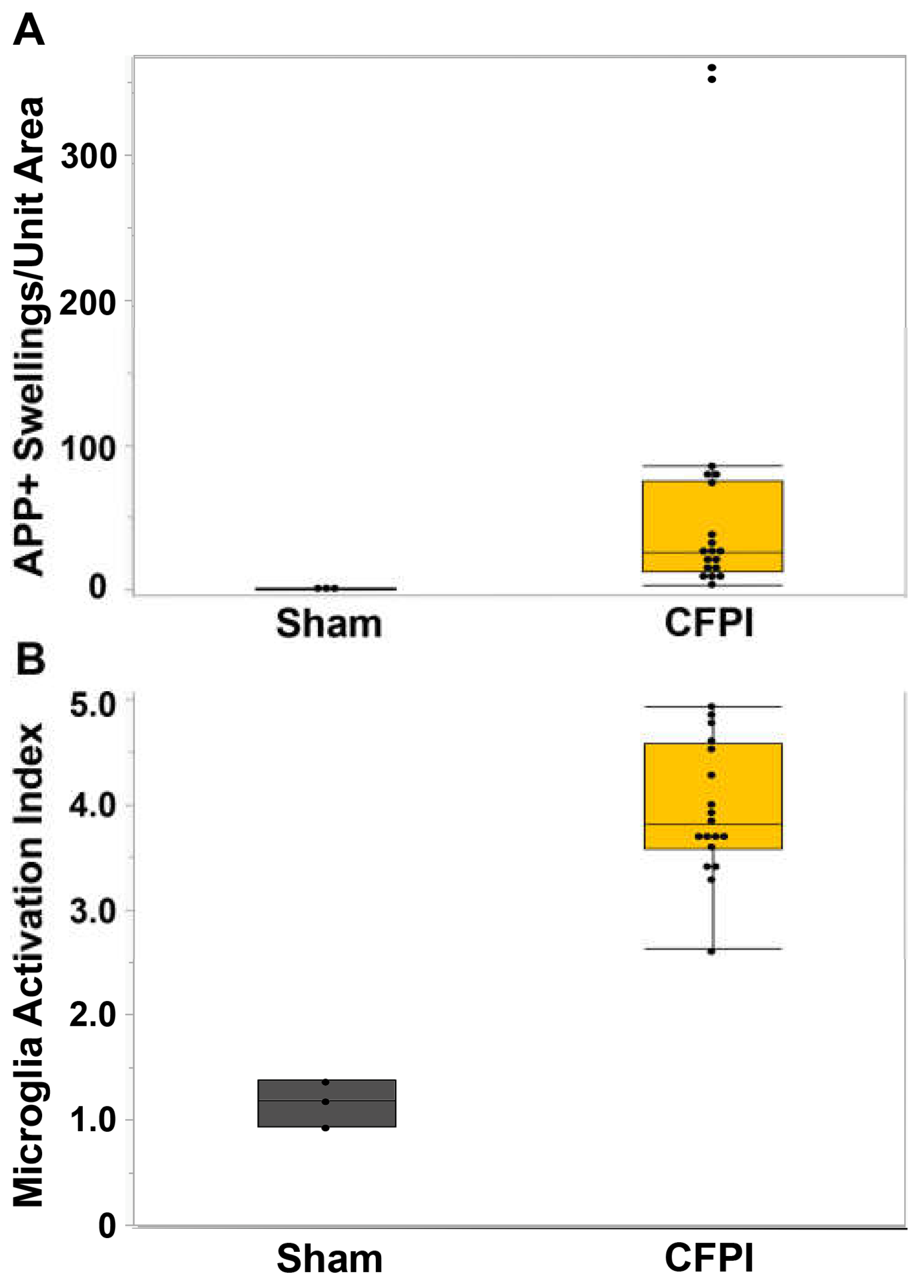
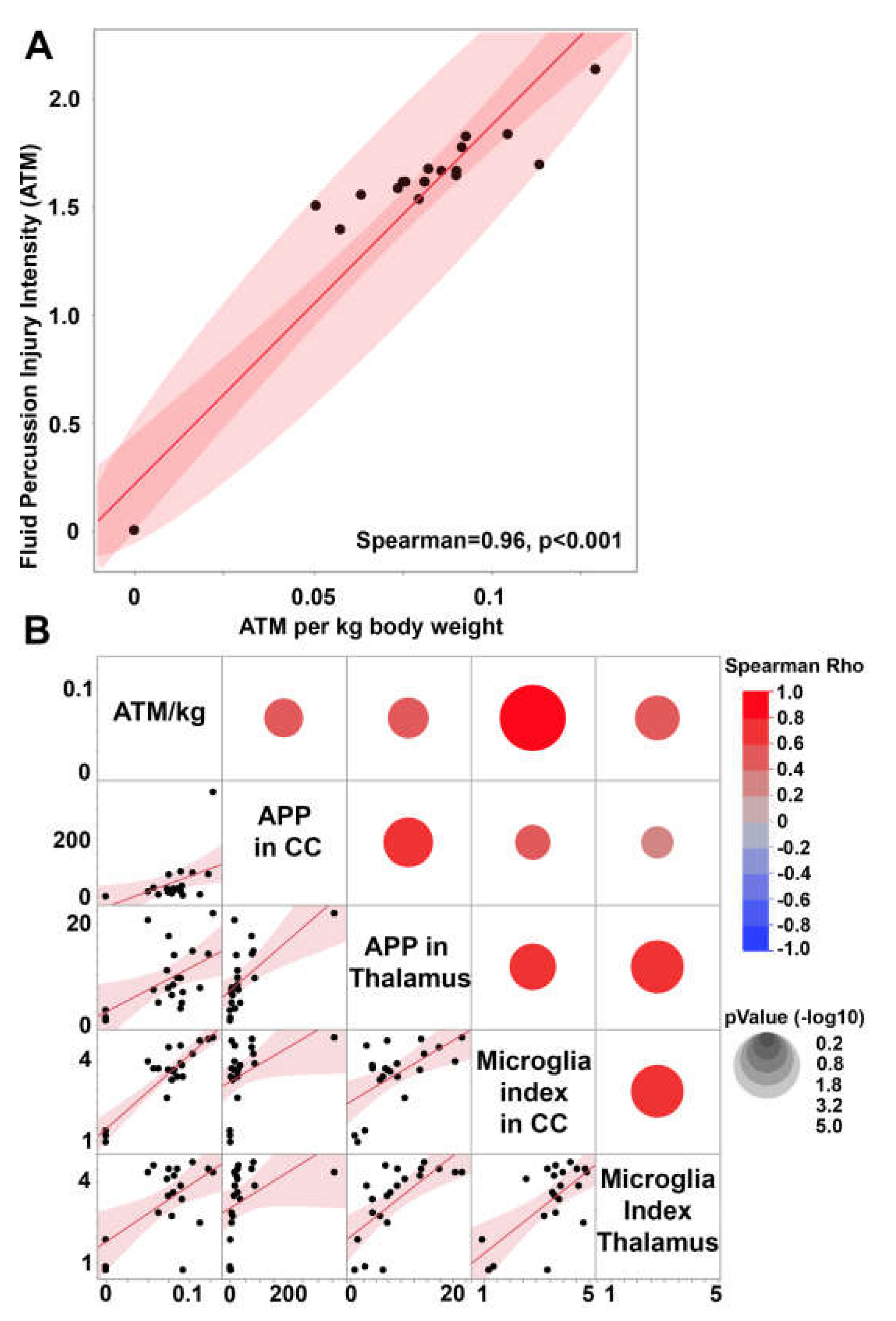
3.1. Central fluid percussion injury induces significant diffuse axonal injury in the thalamus and the corpus callosum that correlate with microglial activation.
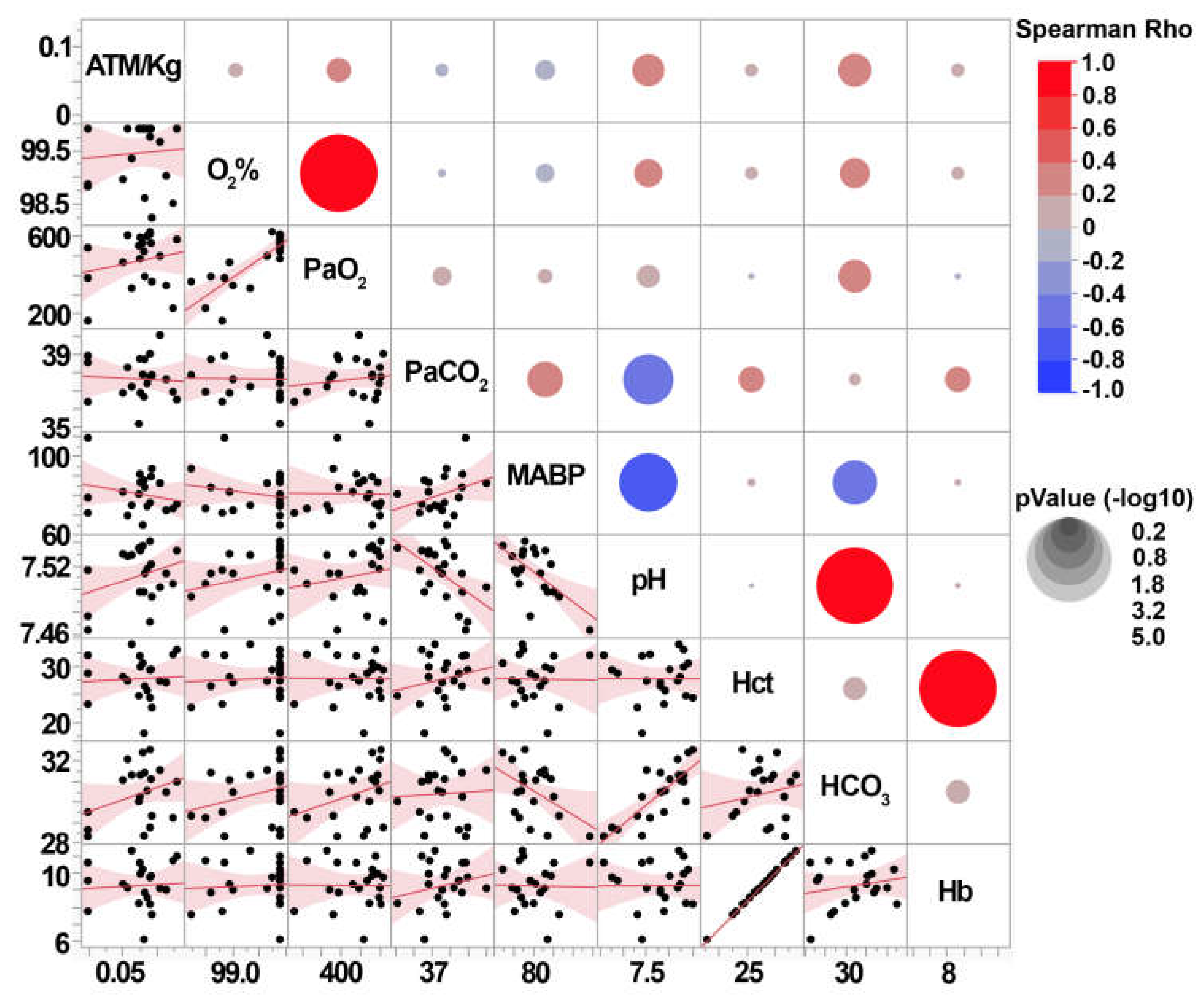
3.2. Central fluid percussion injury generates scalable diffuse pathology that does not significantly impact systemic physiology.
3.3. Fixed pig tissue maintains the capacity for immunohistochemical labeling for years after refrigerated storage.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- James, S.L.; Theadom, A.; Ellenbogen, R.G.; Bannick, M.S.; Montjoy-Venning, W.; Lucchesi, L.R.; Abbasi, N.; Abdulkader, R.; Abraha, H.N.; Adsuar, J.C.; et al. Global, Regional, and National Burden of Traumatic Brain Injury and Spinal Cord Injury, 1990–2016: A Systematic Analysis for the Global Burden of Disease Study 2016. The Lancet Neurology 2019, 18, 56–87. [Google Scholar] [CrossRef]
- Ortiz-Prado, E.; Mascialino, G.; Paz, C.; Rodriguez-Lorenzana, A.; Gómez-Barreno, L.; Simbaña-Rivera, K.; Diaz, A.M.; Coral-Almeida, M.; Espinosa, P.S. A Nationwide Study of Incidence and Mortality Due to Traumatic Brain Injury in Ecuador (2004-2016). Neuroepidemiology 2019, 1–12. [Google Scholar] [CrossRef]
- Brau, R.H.; Acevedo-Salas, Y.; Giovannetti, K. Epidemiological Trends of Traumatic Brain and Spinal Cord Injury in Puerto Rico from November 10th, 2006, through May 24th, 2011. Puerto Rico Health Sciences Journal 2018, 37, 67–77. [Google Scholar]
- Faul, M.; Coronado, V. Chapter 1 - Epidemiology of Traumatic Brain Injury. In Handbook of Clinical Neurology; Traumatic Brain Injury, Part I; Grafman, J., Salazar, A.M., Eds.; Elsevier, 2015; Vol. 127, pp. 3–13. [Google Scholar]
- Maas, A.I.; Roozenbeek, B.; Manley, G.T. Clinical Trials in Traumatic Brain Injury: Past Experience and Current Developments. Neurotherapeutics 2009, 7, 115–126. [Google Scholar] [CrossRef]
- Taylor, C.A.; Bell, J.M.; Breiding, M.J.; Xu, L. Traumatic Brain Injury–Related Emergency Department Visits, Hospitalizations, and Deaths — United States, 2007 and 2013. MMWR. Surveillance Summaries 2017, 66, 1–16. [Google Scholar] [CrossRef]
- Gardner, R.C.; Yaffe, K. Epidemiology of Mild Traumatic Brain Injury and Neurodegenerative Disease. Molecular and Cellular Neuroscience 2015, 66, 75–80. [Google Scholar] [CrossRef]
- Coughlin, J.M.; Wang, Y.; Munro, C.A.; Ma, S.; Yue, C.; Chen, S.; Airan, R.; Kim, P.K.; Adams, A.V.; Garcia, C.; et al. Neuroinflammation and Brain Atrophy in Former NFL Players: An in Vivo Multimodal Imaging Pilot Study. Neurobiology of Disease 2015, 74, 58–65. [Google Scholar] [CrossRef]
- Grossman, E.J.; Inglese, M. The Role of Thalamic Damage in Mild Traumatic Brain Injury. Journal of Neurotrauma 2016, 33, 163–167. [Google Scholar] [CrossRef]
- Little, D.M.; Kraus, M.F.; Joseph, J.; Geary, E.K.; Susmaras, T.; Zhou, X.J.; Pliskin, N.; Gorelick, P.B. Thalamic Integrity Underlies Executive Dysfunction in Traumatic Brain Injury. Neurology 2010, 74, 558–564. [Google Scholar] [CrossRef]
- Gennarelli, T.A.; Thibault, L.E.; Adams, J.H.; Graham, D.I.; Thompson, C.J.; Marcincin, R.P. Diffuse Axonal Injury and Traumatic Coma in the Primate. Annals of neurology 1982, 12, 564–574. [Google Scholar] [CrossRef]
- Povlishock, J.T. Pathobiology of Traumatically Induced Axonal Injury in Animals and Man. Ann Emerg Med 1993, 22, 980–986. [Google Scholar] [CrossRef] [PubMed]
- Sherriff, F.E.; Bridges, L.R.; Sivaloganathan, S. Early Detection of Axonal Injury after Human Head Trauma Using Immunocytochemistry for Beta-Amyloid Precursor Protein. Acta neuropathologica 1994, 87, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, S.M.; Nash, M.J.; Sweeting, C.J.; Graham, D.I.; Roberts, G.W. β-Amyloid Precursor Protein (ΒAPP) as a Marker for Axonal Injury after Head Injury. Neuroscience Letters 1993, 160, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Meaney, D.F. Axonal Damage in Traumatic Brain Injury. The Neuroscientist 2000, 6, 483–495. [Google Scholar] [CrossRef]
- Scheid, R.; Walther, K.; Guthke, T.; Preul, C.; von Cramon, D.Y. Cognitive Sequelae of Diffuse Axonal Injury. Archives of neurology 2006, 63, 418–424. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, X.; Xu, L.; Zhang, R.; Li, Z.; Yue, X.; Qiao, D. Traumatic Axonal Injury: Neuropathological Features, Postmortem Diagnostic Methods, and Strategies. Forensic Sci Med Pathol 2022, 18, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Mckee, A.C.; Daneshvar, D.H. The Neuropathology of Traumatic Brain Injury. In Handbook of Clinical Neurology; Elsevier B.V., 2015; Vol. 127, pp. 45–66.
- Velázquez, A.; Ortega, M.; Rojas, S.; González-Oliván, F.J.; Rodríguez-Baeza, A. Widespread Microglial Activation in Patients Deceased from Traumatic Brain Injury. Brain Injury 2015, 29, 1126–1133. [Google Scholar] [CrossRef]
- Coughlin, J.M.; Wang, Y.; Munro, C.A.; Ma, S.; Yue, C.; Chen, S.; Airan, R.; Kim, P.K.; Adams, A. V.; Garcia, C.; et al. Neuroinflammation and Brain Atrophy in Former NFL Players: An in Vivo Multimodal Imaging Pilot Study. Neurobiology of Disease 2015, 74, 58–65. [Google Scholar] [CrossRef]
- Ramlackhansingh, A.F.; Brooks, D.J.; Greenwood, R.J.; Bose, S.K.; Turkheimer, F.E.; Kinnunen, K.M.; Gentleman, S.; Heckemann, R. a; Gunanayagam, K.; Gelosa, G.; et al. Inflammation after Trauma: Microglial Activation and Traumatic Brain Injury. Annals of neurology 2011, 70, 374–383. [Google Scholar] [CrossRef]
- Zhou, Y.; Lui, Y.W.; Zuo, X.-N.; Milham, M.P.; Reaume, J.; Grossman, R.I.; Ge, Y. Characterization of Thalamocortical Association Using Amplitude and Connectivity of FMRI in Mild Traumatic Brain Injury. J Magn Reson Imaging 2014, 39, 1558–1568. [Google Scholar] [CrossRef]
- Morganti-Kossmann, M.C.; Satgunaseelan, L.; Bye, N.; Kossmann, T. Modulation of Immune Response by Head Injury. Injury 2007, 38, 1392–1400. [Google Scholar] [CrossRef]
- Nizamutdinov, D.; Shapiro, L.A. Overview of Traumatic Brain Injury: An Immunological Context. Brain Sciences 2017, 7. [Google Scholar] [CrossRef]
- Kelley, B.J.; Lifshitz, J.; Povlishock, J.T. Neuroinflammatory Responses after Experimental Diffuse Traumatic Brain Injury. J Neuropathol Exp Neurol 2007, 66, 989–1001. [Google Scholar] [CrossRef]
- Das, M.; Mohapatra, S.; Mohapatra, S.S. New Perspectives on Central and Peripheral Immune Responses to Acute Traumatic Brain Injury. Journal of Neuroinflammation 2012, 9, 236. [Google Scholar] [CrossRef]
- Velázquez, A.; Ortega, M.; Rojas, S.; González-Oliván, F.J.; Rodríguez-Baeza, A. Widespread Microglial Activation in Patients Deceased from Traumatic Brain Injury. Brain Injury 2015, 29, 1126–1133. [Google Scholar] [CrossRef]
- Ramlackhansingh, A.F.; Brooks, D.J.; Greenwood, R.J.; Bose, S.K.; Turkheimer, F.E.; Kinnunen, K.M.; Gentleman, S.; Heckemann, R. a; Gunanayagam, K.; Gelosa, G.; et al. Inflammation after Trauma: Microglial Activation and Traumatic Brain Injury. Annals of neurology 2011, 70, 374–383. [Google Scholar] [CrossRef]
- Mannix, R.C.; Whalen, M.J. Traumatic Brain Injury, Microglia, and Beta Amyloid. International journal of Alzheimer’s disease 2012, 2012, 608732. [Google Scholar] [CrossRef]
- Neumann, H.; Kotter, M.R.; Franklin, R.J.M. Debris Clearance by Microglia: An Essential Link between Degeneration and Regeneration. Brain : a journal of neurology 2009, 132, 288–295. [Google Scholar] [CrossRef]
- Smith, C. Review: The Long-Term Consequences of Microglial Activation Following Acute Traumatic Brain Injury. Neuropathology and Applied Neurobiology 2013, 39, 35–44. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Perry, V.H. Microglial Physiology: Unique Stimuli, Specialized Responses. Annual review of immunology 2009, 27, 119–145. [Google Scholar] [CrossRef]
- Karve, I.P.; Taylor, J.M.; Crack, P.J. The Contribution of Astrocytes and Microglia to Traumatic Brain Injury. British Journal of Pharmacology 2015. [Google Scholar] [CrossRef]
- Tay, T.L.; Hagemeyer, N.; Prinz, M. The Force Awakens: Insights into the Origin and Formation of Microglia. Current Opinion in Neurobiology 2016, 39, 30–37. [Google Scholar] [CrossRef]
- Kigerl, K. a; Gensel, J.C.; Ankeny, D.P.; Alexander, J.K.; Donnelly, D.J.; Popovich, P.G. Identification of Two Distinct Macrophage Subsets with Divergent Effects Causing Either Neurotoxicity or Regeneration in the Injured Mouse Spinal Cord. The Journal of neuroscience : the official journal of the Society for Neuroscience 2009, 29, 13435–13444. [Google Scholar] [CrossRef]
- Loane, D.J.; Kumar, A. Microglia in the TBI Brain: The Good, the Bad, and the Dysregulated. Experimental Neurology 2016, 275, 316–327. [Google Scholar] [CrossRef]
- Chen, Y.; Swanson, R.A. Astrocytes and Brain Injury. Journal of Cerebral Blood Flow and Metabolism 2003, 23, 137–149. [Google Scholar] [CrossRef]
- Lafrenaye, A.D.; Simard, J.M. Bursting at the Seams: Molecular Mechanisms Mediating Astrocyte Swelling. International journal of molecular sciences 2019. [Google Scholar] [CrossRef]
- Laird, M.D.; Vender, J.R.; Dhandapani, K.M. Opposing Roles for Reactive Astrocytes Following Traumatic Brain Injury. NeuroSignals 2008, 16, 154–164. [Google Scholar] [CrossRef]
- Mondello, S.; Muller, U.; Jeromin, A.; Streeter, J.; Hayes, R.L.; Wang, K.K. Blood-Based Diagnostics of Traumatic Brain Injuries. Expert review of molecular diagnostics 2011, 11, 65–78. [Google Scholar] [CrossRef]
- Czeiter, E.; Mondello, S.; Kovacs, N.; Sandor, J.; Gabrielli, A.; Schmid, K.; Tortella, F.; Wang, K.K.W.; Hayes, R.L.; Barzo, P.; et al. Brain Injury Biomarkers May Improve the Predictive Power of the IMPACT Outcome Calculator. Journal of neurotrauma 2012, 29, 1770–1778. [Google Scholar] [CrossRef]
- Papa, L. Potential Blood-Based Biomarkers for Concussion. Sports Med Arthrosc 24, 115. [CrossRef]
- Schumacher, M.; Denier, C.; Oudinet, J.P.; Adams, D.; Guennoun, R. Progesterone Neuroprotection: The Background of Clinical Trial Failure. Journal of Steroid Biochemistry and Molecular Biology 2016, 160, 53–66. [Google Scholar] [CrossRef]
- Dai, J.X.; Ma, Y.B.; Le, N.Y.; Cao, J.; Wang, Y. Large Animal Models of Traumatic Brain Injury. International Journal of Neuroscience 2018, 128, 243–254. [Google Scholar] [CrossRef]
- Kochanek, P.M.; Jackson, T.C.; Jha, R.M.; Clark, R.S.B.; Okonkwo, D.O.; Bayır, H.; Poloyac, S.M.; Wagner, A.K.; Empey, P.E.; Conley, Y.P.; et al. Paths to Successful Translation of New Therapies for Severe Traumatic Brain Injury in the Golden Age of Traumatic Brain Injury Research: A Pittsburgh Vision. Journal of Neurotrauma 2019, neu.2018.6203. [Google Scholar] [CrossRef] [PubMed]
- Seok, J.; Warren, H.S.; Cuenca, A.G.; Mindrinos, M.N.; Baker, H.V.; Xu, W.; Richards, D.R.; McDonald-Smith, G.P.; Gao, H.; Hennessy, L.; et al. Genomic Responses in Mouse Models Poorly Mimic Human Inflammatory Diseases. Proceedings of the National Academy of Sciences of the United States of America 2013, 110, 3507–3512. [Google Scholar] [CrossRef] [PubMed]
- Sauleau, P.; Lapouble, E.; Val-Laillet, D.; Malbert, C.H. The Pig Model in Brain Imaging and Neurosurgery. Animal 2009, 3, 1138–1151. [Google Scholar] [CrossRef]
- Wernersson, R.; Schierup, M.H.; Jørgensen, F.G.; Gorodkin, J.; Panitz, F.; Staerfeldt, H.-H.; Christensen, O.F.; Mailund, T.; Hornshøj, H.; Klein, A.; et al. Pigs in Sequence Space: A 0.66X Coverage Pig Genome Survey Based on Shotgun Sequencing. BMC genomics 2005, 6, 70. [Google Scholar] [CrossRef]
- Fairbairn, L.; Kapetanovic, R.; Sester, D.P.; Hume, D. a The Mononuclear Phagocyte System of the Pig as a Model for Understanding Human Innate Immunity and Disease. Journal of leukocyte biology 2011, 89, 855–871. [Google Scholar] [CrossRef]
- Lind, N.M.; Moustgaard, A.; Jelsing, J.; Vajta, G.; Cumming, P.; Hansen, A.K. The Use of Pigs in Neuroscience: Modeling Brain Disorders. Neuroscience and Biobehavioral Reviews 2007, 31, 728–751. [Google Scholar] [CrossRef]
- Marklund, N.; Hillered, L. Animal Modelling of Traumatic Brain Injury in Preclinical Drug Development: Where Do We Go from Here? British Journal of Pharmacology 2011, 164, 1207–1229. [Google Scholar] [CrossRef]
- Statler, K.D.; Jenkins, L.W.; Dixon, C.E.; Clark, R.S.; Marion, D.W.; Kochanek, P.M. The Simple Model versus the Super Model: Translating Experimental Traumatic Brain Injury Research to the Bedside. J Neurotrauma 2001, 18, 1195–1206. [Google Scholar] [CrossRef]
- Gorse, K.M.; Lafrenaye, A.D. The Importance of Inter-Species Variation in Traumatic Brain Injury-Induced Alterations of Microglial-Axonal Interactions. Frontiers in Neurology 2018, 9. [Google Scholar] [CrossRef]
- Lafrenaye, A.D.; Todani, M.; Walker, S.A.; Povlishock, J.T. Microglia Processes Associate with Diffusely Injured Axons Following Mild Traumatic Brain Injury in the Micro Pig. Journal of Neuroinflammation 2015, 12, 186. [Google Scholar] [CrossRef] [PubMed]
- Lafrenaye, A.D.; Mondello, S.; Wang, K.K.; Yang, Z.; Povlishock, J.T.; Gorse, K.; Walker, S.; Hayes, R.L.; Kochanek, P.M. Circulating GFAP and Iba-1 Levels Are Associated with Pathophysiological Sequelae in the Thalamus in a Pig Model of Mild TBI. Scientific Reports 2020, 10, 13369. [Google Scholar] [CrossRef] [PubMed]
- Lafrenaye, A.; Mondello, S.; Povlishock, J.; Gorse, K.; Walker, S.; Hayes, R.; Wang, K.; Kochanek, P.M. Operation Brain Trauma Therapy: An Exploratory Study of Levetiracetam Treatment Following Mild Traumatic Brain Injury in the Micro Pig. Frontiers in Neurology 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- National Research Council GUIDE LABORATORY ANIMALS FOR THE CARE AND USE OF Eighth Edition Committee for the Update of the Guide for the Care and Use of Laboratory Animals Institute for Laboratory Animal Research Division on Earth and Life Studies; 2011; ISBN 978-0-309-15400-0.
- Bramlett, H.M.; Kraydieh, S.; Green, E.J.; Dietrich, W.D. Temporal and Regional Patterns of Axonal Damage Following Traumatic Brain Injury: A Beta-Amyloid Precursor Protein Immunocytochemical Study in Rats. Journal of Neuropathology and Experimental Neurology 1997, 56, 1132–1141. [Google Scholar] [CrossRef]
- Taetzsch, T.; Levesque, S.; McGraw, C.; Brookins, S.; Luqa, R.; Bonini, M.G.; Mason, R.P.; Oh, U.; Block, M.L. Redox Regulation of NF-ΚB P50 and M1 Polarization in Microglia. Glia 2015, 63, 423–440. [Google Scholar] [CrossRef] [PubMed]
- Byrnes, K.R.; Loane, D.J.; Stoica, B. a; Zhang, J.; Faden, A.I. Delayed MGluR5 Activation Limits Neuroinflammation and Neurodegeneration after Traumatic Brain Injury. Journal of Neuroinflammation 2012, 9, 43. [Google Scholar] [CrossRef]
- Haynes, S.E.; Hollopeter, G.; Yang, G.; Kurpius, D.; Dailey, M.E.; Gan, W.-B.; Julius, D. The P2Y12 Receptor Regulates Microglial Activation by Extracellular Nucleotides. Nature neuroscience 2006, 9, 1512–1519. [Google Scholar] [CrossRef]
- van Eijck, M.M.; Schoonman, G.G.; van der Naalt, J.; de Vries, J.; Roks, G. Diffuse Axonal Injury after Traumatic Brain Injury Is a Prognostic Factor for Functional Outcome: A Systematic Review and Meta-Analysis. Brain Injury 2018, 32, 395–402. [Google Scholar] [CrossRef]
- Davceva, N.; Sivevski, A.; Basheska, N. Traumatic Axonal Injury, a Clinical-Pathological Correlation. Journal of Forensic and Legal Medicine 2017, 48, 35–40. [Google Scholar] [CrossRef]
- Vieira, R. de C.A.; Paiva, W.S.; de Oliveira, D.V.; Teixeira, M.J.; de Andrade, A.F.; de Sousa, R.M.C. Diffuse Axonal Injury: Epidemiology, Outcome and Associated Risk Factors. Front Neurol 2016, 7, 178. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.S.N.; Jolly, A.; Zimmerman, K.; Bourke, N.J.; Scott, G.; Cole, J.H.; Schott, J.M.; Sharp, D.J. Diffuse Axonal Injury Predicts Neurodegeneration after Moderate–Severe Traumatic Brain Injury. Brain 2020, 143, 3685–3698. [Google Scholar] [CrossRef]
- Mesfin, F.B.; Gupta, N.; Hays Shapshak, A.; Taylor, R.S. Diffuse Axonal Injury. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2023. [Google Scholar]
- Hoffe, B.; Holahan, M.R. The Use of Pigs as a Translational Model for Studying Neurodegenerative Diseases. Frontiers in Physiology 2019, 10, 838. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Sinha, S.; Suri, V.; Agarwal, D.; Bisht, A.; Garg, K.; Gupta, D.; Kakkar, A.; Kale, S.; Lalwani, S.; et al. Histological Changes in Thalamus in Short Term Survivors Following Traumatic Brain Injury: An Autopsy Study. Neurology India 2013, 61, 599. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Parker, D.; Whyte, J.; Hart, T.; Pluta, J.; Ingalhalikar, M.; Coslett, H.B.; Verma, R. Disrupted Structural Connectome Is Associated with Both Psychometric and Real-World Neuropsychological Impairment in Diffuse Traumatic Brain Injury. Journal of the International Neuropsychological Society 2014, 20, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Banks, S.D.; Coronado, R.A.; Clemons, L.R.; Abraham, C.M.; Pruthi, S.; Conrad, B.N.; Morgan, V.L.; Guillamondegui, O.D.; Archer, K.R. Thalamic Functional Connectivity in Mild Traumatic Brain Injury: Longitudinal Associations With Patient-Reported Outcomes and Neuropsychological Tests. Archives of Physical Medicine and Rehabilitation 2016, 97, 1254–1261. [Google Scholar] [CrossRef]
- Adams, J.; Doyle, D.; Ford, I.; Gennarelli, T.; Graham, D.; McLellan, D. Diffuse Axonal Injury in Head Injury: Definition, Diagnosis and Grading. Histopathology 1989, 15, 49–59. [Google Scholar] [CrossRef]
- Prus, R.; Pokotylo, P.; Logash, M.; Zvir, T. Morphological Particularities and Morphometry of Rats’ Kidneys under the Effect of Experimental Mild Traumatic Brain Injury. Folia Morphologica 2021, 80, 310–316. [Google Scholar] [CrossRef]
- Tekin Neijmann, Ş.; Kural, A.; Sever, N.; Dogan, H.; Sarıkaya, S. Evaluation of Renal Function in Rats with Moderate and Mild Brain Trauma. Ulus Travma Acil Cerrahi Derg 2022, 28, 1–7. [Google Scholar] [CrossRef]
- Ruan, F.; Chen, J.; Yang, J.; Wang, G. MILD TRAUMATIC BRAIN INJURY ATTENUATES PNEUMONIA-INDUCED LUNG INJURY BY MODULATIONS OF ALVEOLAR MACROPHAGE BACTERICIDAL ACTIVITY AND M1 POLARIZATION. Shock 2022, 58, 400. [Google Scholar] [CrossRef]
- Vermeij, J.-D.; Aslami, H.; Fluiter, K.; Roelofs, J.J.; van den Bergh, W.M.; Juffermans, N.P.; Schultz, M.J.; Van der Sluijs, K.; van de Beek, D.; van Westerloo, D.J. Traumatic Brain Injury in Rats Induces Lung Injury and Systemic Immune Suppression. Journal of Neurotrauma 2013, 30, 2073–2079. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Jung, H.; Youn, D.H.; Kim, T.Y.; Han, S.W.; Kim, B.J.; Lee, J.J.; Jeon, J.P. Mild Traumatic Brain Injury and Subsequent Acute Pulmonary Inflammatory Response. J Korean Neurosurg Soc 2022, 65, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhao, W.-G.; Zhang, W.-F. Acute Kidney Injury in Patients with Severe Traumatic Brain Injury: Implementation of the Acute Kidney Injury Network Stage System. Neurocrit Care 2011, 14, 377–381. [Google Scholar] [CrossRef]
- Holland, M.C.; Mackersie, R.C.; Morabito, D.; Campbell, A.R.; Kivett, V.A.; Patel, R.; Erickson, V.R.; Pittet, J.-F. The Development of Acute Lung Injury Is Associated with Worse Neurologic Outcome in Patients with Severe Traumatic Brain Injury. Journal of Trauma and Acute Care Surgery 2003, 55, 106. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; Imai, Y.; Ohsawa, K.; Nakajima, K.; Fukuuchi, Y.; Kohsaka, S. Microglia-Specific Localisation of a Novel Calcium Binding Protein, Iba1. Molecular Brain Research 1998, 57, 1–9. [Google Scholar] [CrossRef]
- Rodriguez-Callejas, J.D.; Fuchs, E.; Perez-Cruz, C. Evidence of Tau Hyperphosphorylation and Dystrophic Microglia in the Common Marmoset. Frontiers in Aging Neuroscience 2016, 8. [Google Scholar] [CrossRef]
- Ahn, J.-H.; Choi, J.-H.; Park, J.-H.; Yan, B.-C.; Kim, I.-H.; Lee, J.-C.; Lee, D.-H.; Kim, J.-S.; Shin, H.-C.; Won, M.-H. Comparison of Alpha-Synuclein Immunoreactivity in the Spinal Cord between the Adult and Aged Beagle Dog. Lab Anim Res 2012, 28, 165–170. [Google Scholar] [CrossRef]
- Gaigé, S.; Bonnet, M.S.; Tardivel, C.; Pinton, P.; Trouslard, J.; Jean, A.; Guzylack, L.; Troadec, J.-D.; Dallaporta, M. C-Fos Immunoreactivity in the Pig Brain Following Deoxynivalenol Intoxication: Focus on NUCB2/Nesfatin-1 Expressing Neurons. NeuroToxicology 2013, 34, 135–149. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



