Submitted:
22 April 2023
Posted:
23 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
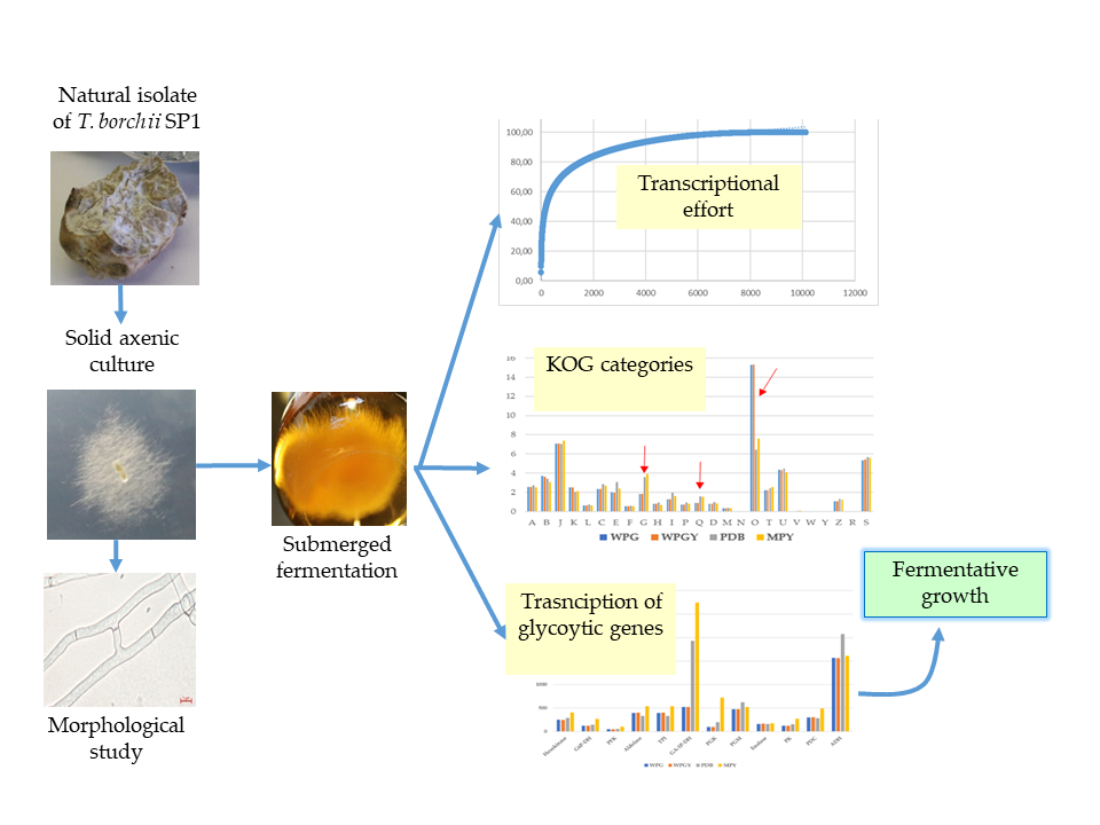
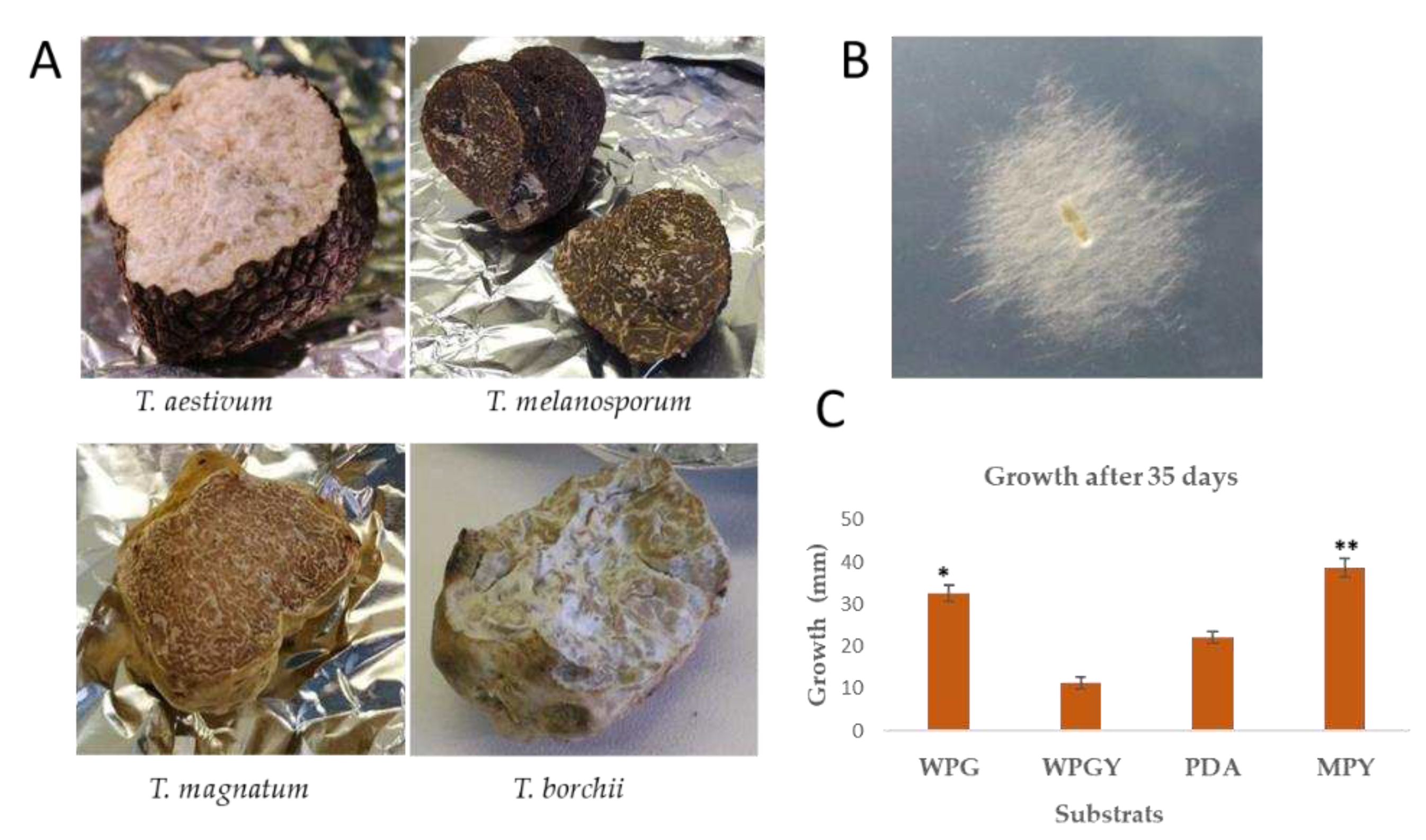
2.1. Culture conditions, growth rate, and biomass production.
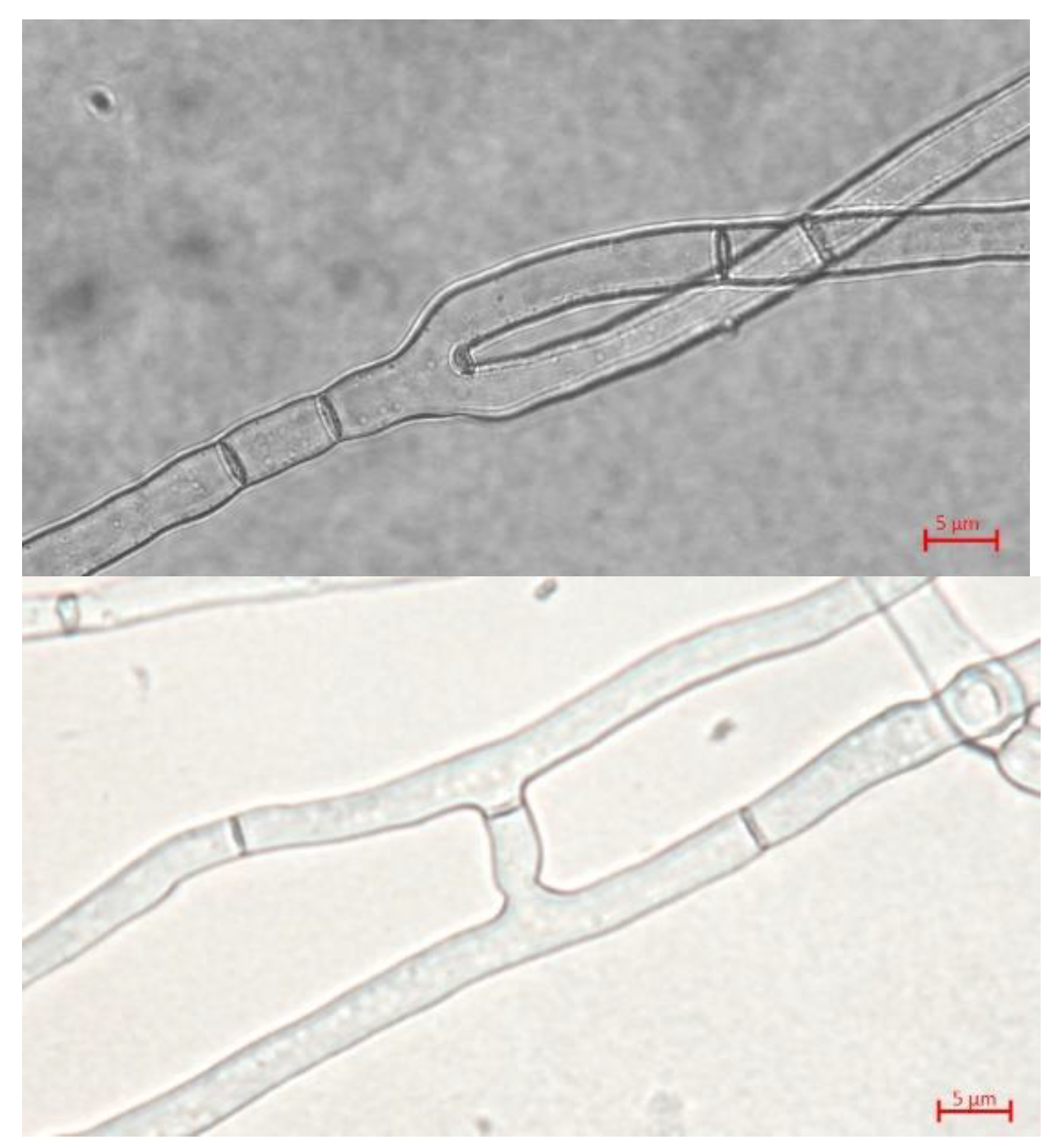
2.2. Hyphal morphology
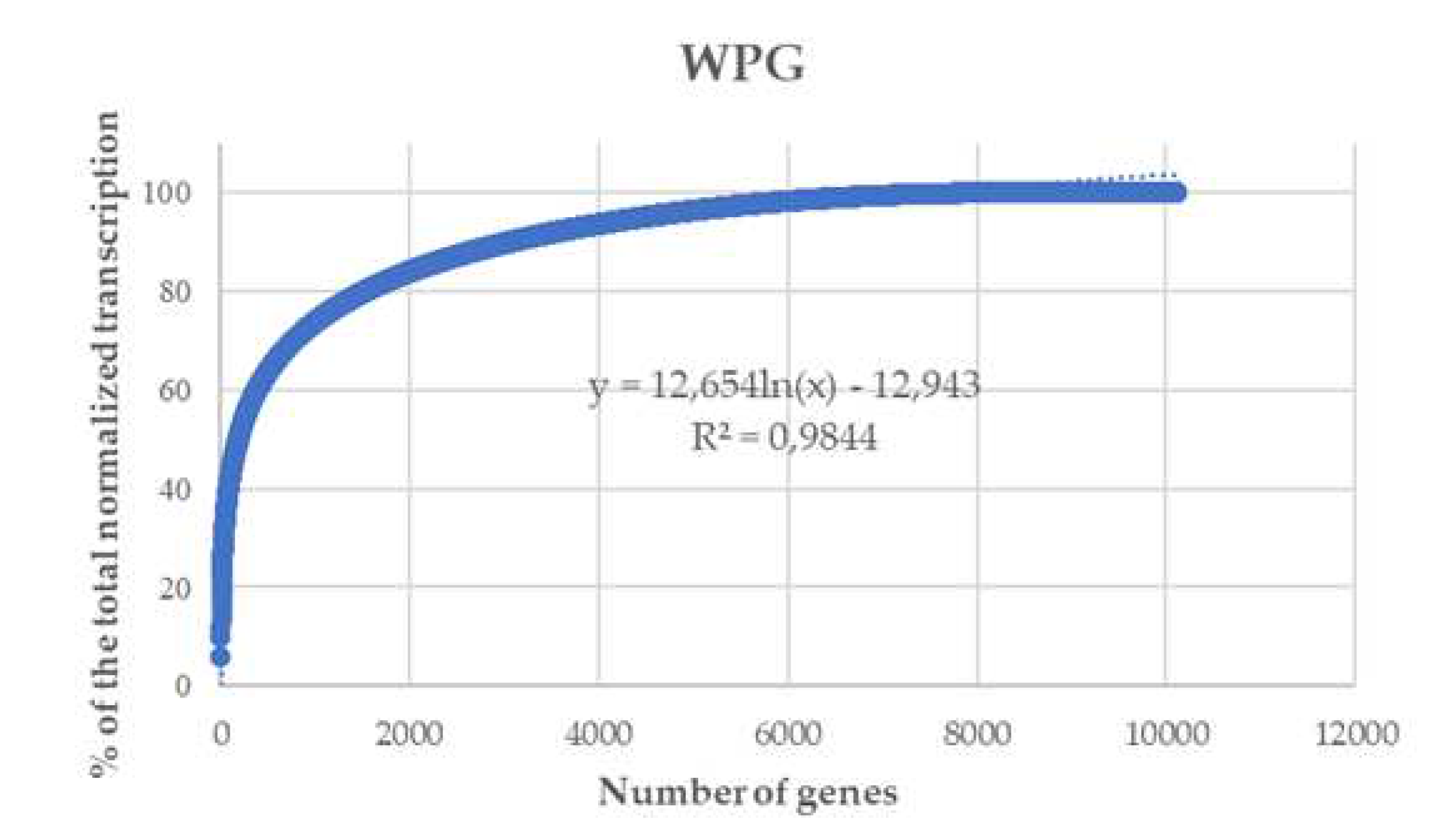
2.3. RNA-Seq and transcriptome analysis
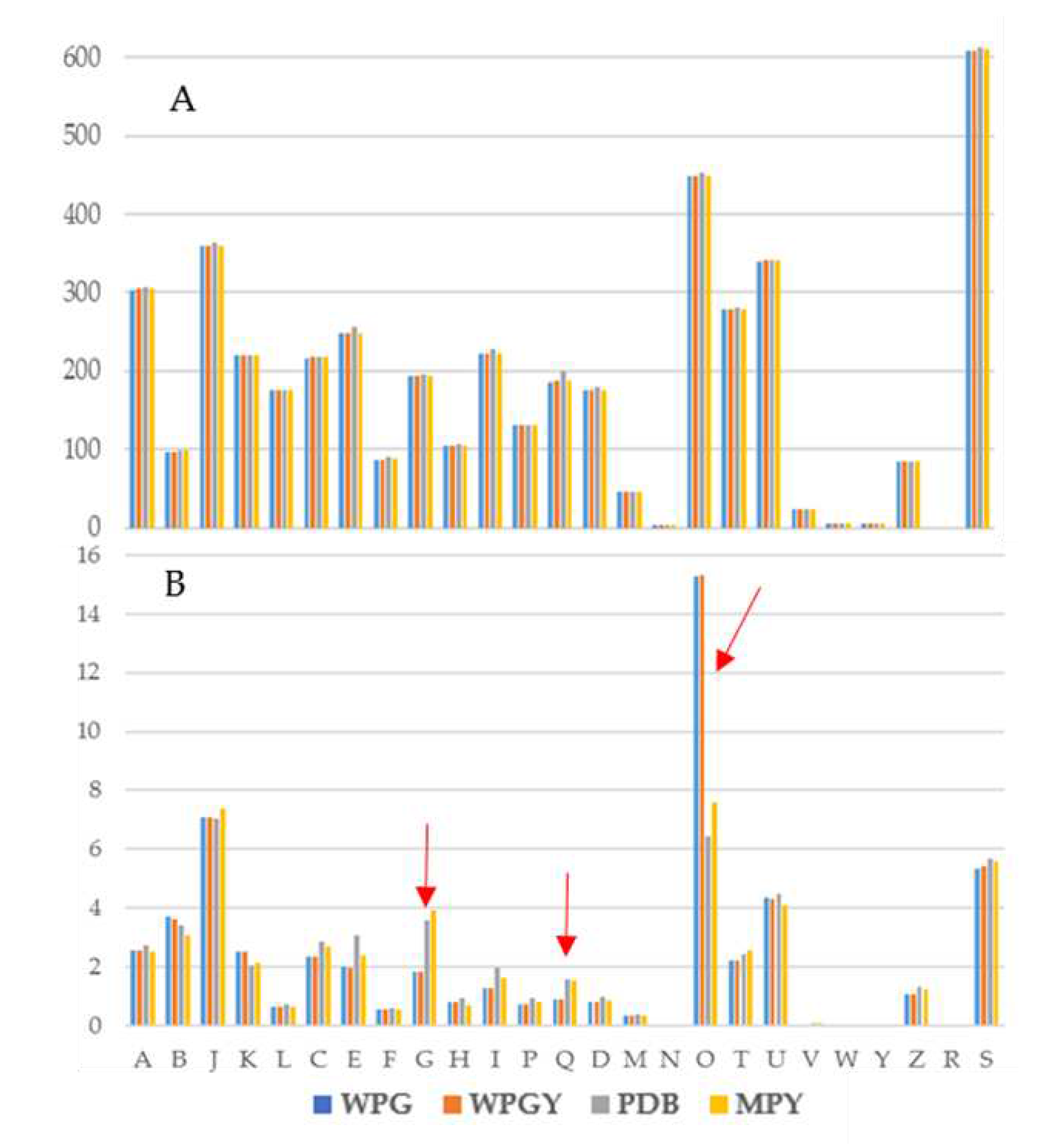
2.4. Comparison of the transcriptomes from the different culture media
2.5. Pathways of central metabolism
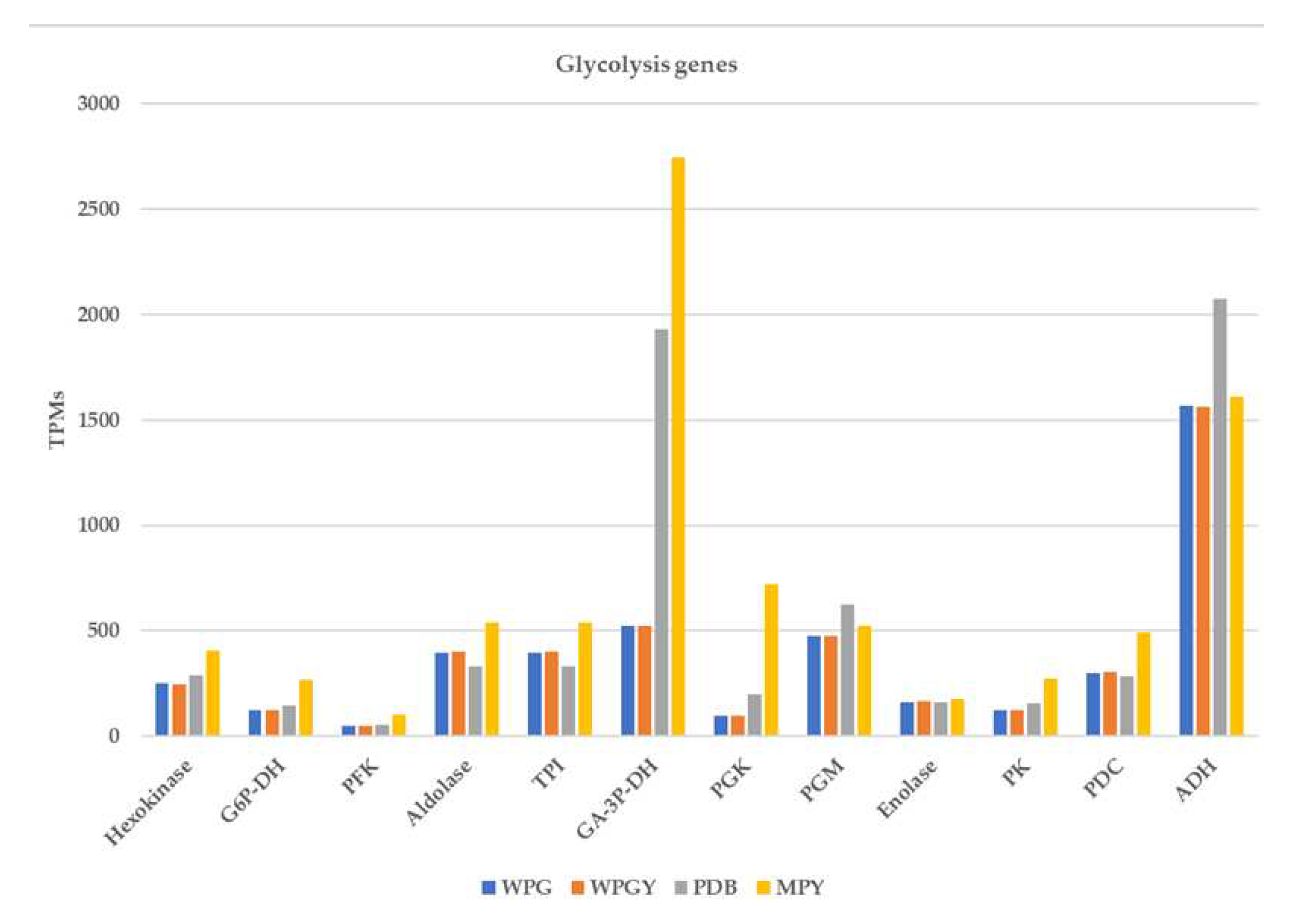
2.5.1. Central metabolism glycolysis genes
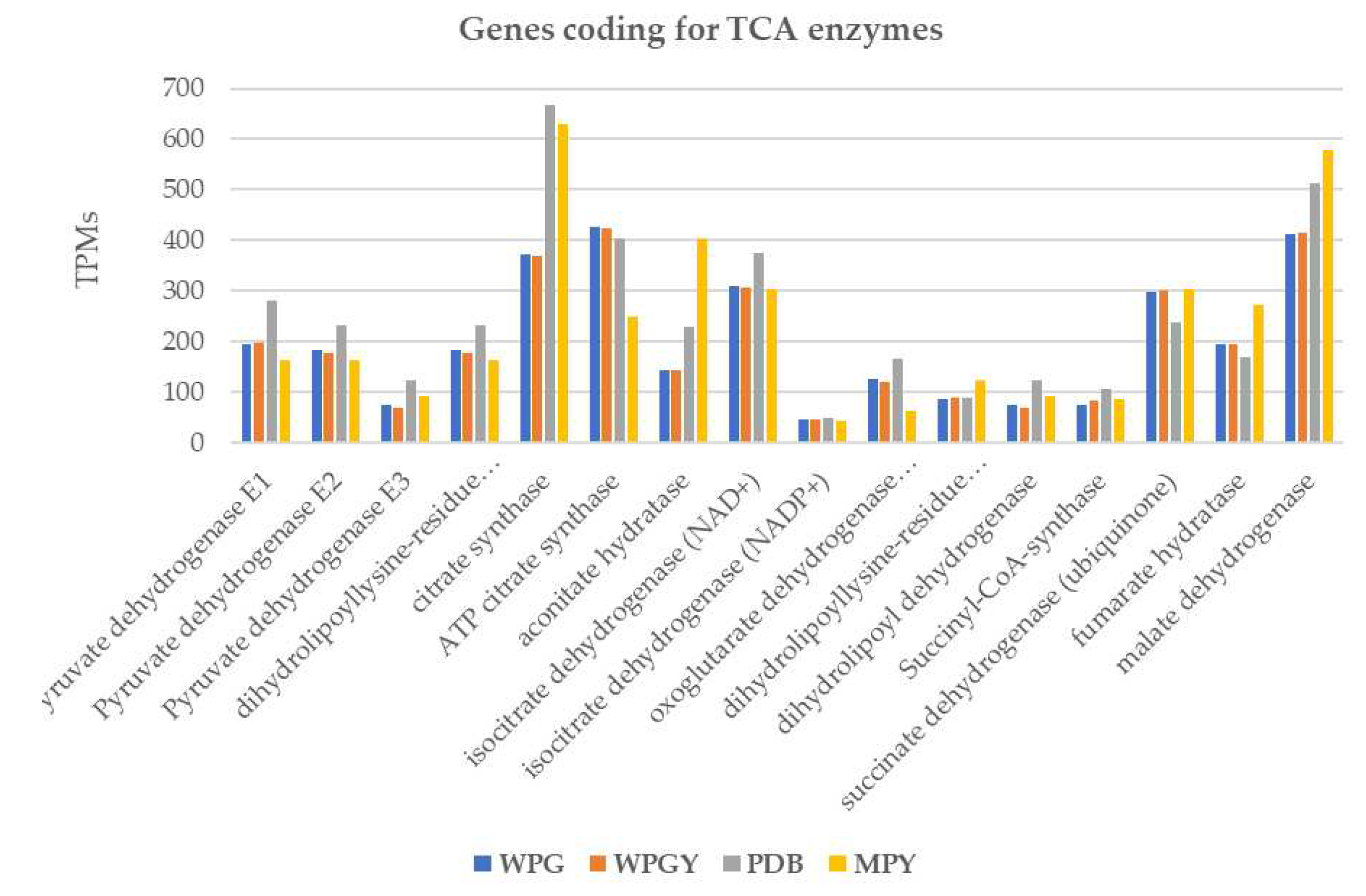
2.5.2. TCA and glyoxylate cycle
2.5.3. Respiratory chain
2.6. Genes involved in secondary metabolism pathways – Clusters
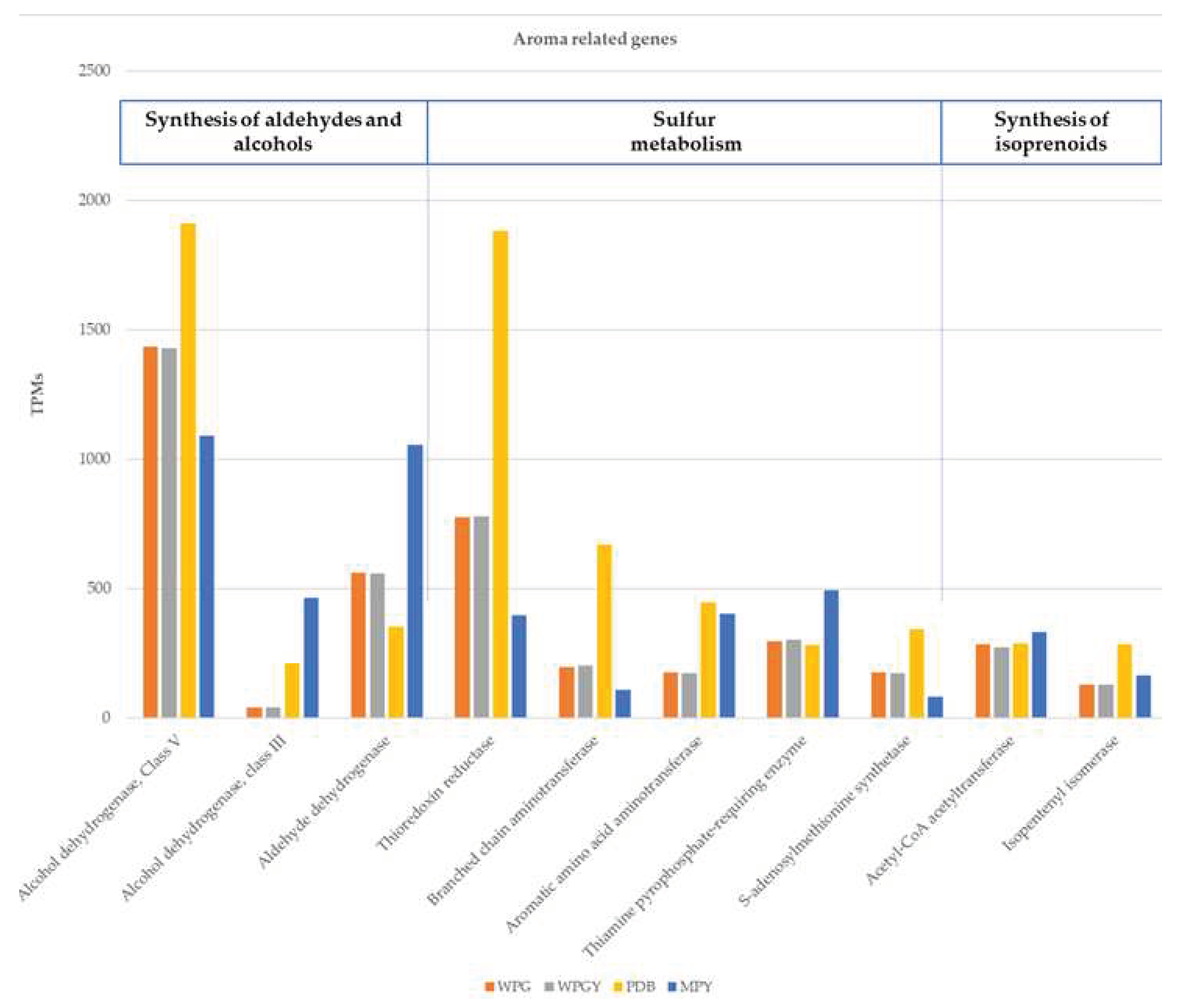
2.7. Genes related to the synthesis of volatile compounds
3. Discussion
3.1. SP1 is a new mycelial isolate of Tuber borchii.
3.2. RNA-Seq and transcriptome analysis
3.3. Comparison of the transcriptomes from the different culture media
3.4. Pathways of central metabolism
3.5. Genes involved in secondary metabolism – Clusters
3.6. Genes related to the synthesis of volatile compounds
4. Materials and Methods
4.1. Strain and culture conditions
4.2. Growth rate
4.3. Biomass
4.4. Molecular characterization
4.5. Morphology
4.6. RNA seq
4.7. Transcriptome data analysis
4.8. Analysis of metabolic pathways
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
References
- Mustafa, A.M.; Angeloni, S.; Nzekoue, F.K.; Abouelenein, D.; Sagratini, G.; Caprioli, G.; Torregiani, E. An Overview on Truffle Aroma and Main Volatile Compounds. Molecules 2020, 25, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Mello, A.; Murat, C.; Bonfante, P. Truffles: Much More than a Prized and Local Fungal Delicacy. FEMS Microbiol Lett 2006, 260, 1–8. [Google Scholar] [CrossRef]
- Zambonelli, A., IM, MC True Truffle (Tuber Spp.) in the World ; 2016; Vol. 47; 333-349. [CrossRef]
- Splivallo, R.; Cullere, L. The Smell of Truffles : From Aroma Biosynthesis to Product Quality. 2016, 393–407. [CrossRef]
- Bonito, G.; Trappe, J.M.; Rawlinson, P.; Vilgalys, R. Improved Resolution of Major Clades within Tuber and Taxonomy of Species within the Tuber Gibbosum Complex. Mycologia 2010, 102, 1042–1057. [Google Scholar] [CrossRef] [PubMed]
- Hall, I.R.; Brown, G.T.; Zambonelli, A. Taming the Truffle. The History Lore and Science of the Ultimate Mushroom; 1st ed.; Portland, OR, USA, 2007, 304.
- Lacourt, I.; Abba, S.; Bonfante, P.; Martin, F. Isolation and Characterization of Differentially Expressed Genes in the Mycelium and Fruit Body of Tuber Borchii. 2002, 68, 4574–4582. 68. [CrossRef]
- Marathe, SJ; Hamzi, W. ; Bashein, A.M.; Deska, J.; Seppänen-Laakso, T.; Singhal, R.S.; Shamekh, S. Anti-Angiogenic and Anti-Inflammatory Activity of the Summer Truffle (Tuber Aestivum Vittad.) Extracts and a Correlation with the Chemical Constituents Identified Therein. Food Research International 2020, 137. [Google Scholar] [CrossRef]
- Zhao, W.; Wang, X.H.; Li, HM; Wang, S. H.; Chen, T.; Yuan, Z.P.; Tang, Y.J. Isolation and Characterization of Polysaccharides with the Antitumor Activity from Tuber Fruiting Bodies and Fermentation System. Appl Microbiol Biotechnol 2014, 98, 1991–2002. [Google Scholar] [CrossRef] [PubMed]
- Li, YY; Wang, G. ; Li, HM; Zhong, J.J.; Tang, Y.J. Volatile Organic Compounds from a Tuber Melanosporum Fermentation System. Food Chem 2012, 135, 2628–2637. [Google Scholar] [CrossRef] [PubMed]
- Vita, F.; Taiti, C.; Pompeiano, A.; Bazihizina, N.; Lucarotti, V.; Mancuso, S.; Alpi, A. Volatile Organic Compounds in Truffle (Tuber Magnatum Pico): Comparison of Samples from Different Regions of Italy and from Different Seasons. Sci Rep 2015, 5, 12629. [Google Scholar] [CrossRef]
- Shah, N.; Marathe, SJ; Croce, D. ; Ciardi, M.; Longo, V.; Juilus, A.; Shamekh, S. An Investigation of the Antioxidant Potential and Bioaccumulated Minerals in Tuber Borchii and Tuber Maculatum Mycelia Obtained by Submerged Fermentation. Arch Microbiol 2022, 204, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.J.; Liu, R.S.; Li, H.M. Current Progress on Truffle Submerged Fermentation: A Promising Alternative to Its Fruiting Bodies. Appl Microbiol Biotechnol 2015, 99, 2041–2053. [Google Scholar] [CrossRef]
- Zambonelli, A.; Iotti, M.; Hall, I. Current Status of Truffle Cultivation: Recent Results and Future Perspectives. Micol Ital 2015, 44, 31–40. [Google Scholar] [CrossRef]
- Lacourt, I.; Duplessis, S.; Abbà, S.; Bonfante, P.; Martin, F. Isolation and Characterization of Differentially Expressed Genes in the Mycelium and Fruit Body of Tuber Borchii. Appl Environ Microbiol 2002, 68, 4574–4582. [Google Scholar] [CrossRef]
- Splivallo, R.; Bossi, S.; Maffei, M.; Bonfante, P. Discrimination of Truffle Fruiting Body versus Mycelial Aromas by Stir Bar Sorptive Extraction. Phytochemistry 2007, 68, 2584–2598. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, H.; Tang, Y. Comparison of Sterol Composition between Tuber Fermentation Mycelia and Natural Fruiting Bodies. Food Chem 2012, 132, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Tirillini, B.; Verdelli, G.; Paolocci, F.; Ciccioli, P.; Frattoni, M. The Volatile Organic Compounds from the Mycelium of Tuber Borchii Vitt. Phytochemistry 2000, 55, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Murat, C.; Payen, T.; Noel, B.; Kuo, A.; Morin, E.; Chen, J.; Kohler, A.; Krizsán, K.; Balestrini, R.; Da Silva, C.; et al. Pezizomycetes Genomes Reveal the Molecular Basis of Ectomycorrhizal Truffle Lifestyle. Nat Ecol Evol 2018, 2, 1956–1965. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Marcone, M.F. The Biochemistry and Biological Properties of the World’s Most Expensive Underground Edible Mushroom: Truffles. Food Research International 2011, 44, 2567–2581. [Google Scholar] [CrossRef]
- Martin, F.; Kohler, A.; Murat, C.; Balestrini, R.; Coutinho, P.M.; Jaillon, O.; Montanini, B.; Morin, E.; Noel, B.; Percudani, R.; et al. Périgord Black Truffle Genome Uncovers Evolutionary Origins and Mechanisms of Symbiosis. Nature 2010, 464, 1033–1038. [Google Scholar] [CrossRef]
- Moreno, G.; Manjón, J.L. A New Tuber without Spore Ornamentation. Tuber Melosporum Comb. Bol. Soc. Micol. Madrid 2012, 36, 191–196. [Google Scholar]
- Murat, C.; Martin, F. Truffle Genomics: Investigating an Early Diverging Lineage of Pezizomycotina. In True truffle (Tuber spp.) in the world.; Zambonelli A, Iotti M, M.C., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 137–149. [Google Scholar]
- Murat, C.; Kuo, A.; Barry, K.W.; Clum, A.; Dockter, R.B.; Fauchery, L.; Iotti, M.; Kohler, A.; Labutti, K.; Lindquist, E.A.; et al. Draft Genome Sequence of Tuber Borchii Vittad., a Whitish Edible Truffle. American Society for Microbiology 2018, 6, 1–2. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, Z.; Xu, L.; Shi, W.; Liu, X.; Wang, F. Increasing Production of Truffle Polysaccharides in the Solid-State Fermentation of Tuber Melanosporum by Diosgenin Based on Orthogonal Matrix and Nonlinear Regression Analysis. Food Sci Technol Res 2020, 26, 487–494. [Google Scholar] [CrossRef]
- Tang, Y.J.; Wang, G.; Li, YY; Zhong, J. J. Fermentation Condition Outweighed Truffle Species in Affecting Volatile Organic Compounds Analyzed by Chromatographic Fingerprint System. Anal Chim Acta 2009, 647, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Fontana, A. Atti Del Congresso Internazionale Sul Tartufo. In Proceedings of the MICELI DI FUNGHI IPOGEI IN COLTURA PURA; 1967; pp. 127–133. [Google Scholar]
- Iotti, M.; Amicucci, A.; Stocchi, V.; Zambonelli, A. Morphological and Molecular Characterization of Mycelia of Some Tuber Species in Pure Culture. New Phytologist 2002, 155, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Vahdatzadeh, M.; Deveau, A.; Splivallo, R. The Role of the Microbiome of Truffles in Aroma Formation: A Meta-Analysis Approach. Appl Environ Microbiol 2015, 81, 6946–6952. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, E.; Gioacchini, A.M.; Zambonelli, A.; Bertini, L.; Stocchi, V. Determination of Microbial Volatile Organic Compounds from Staphylococcus Pasteuri against Tuber Borchii Using Solid-Phase Microextraction and Gas Chromatography / Ion Trap Mass Spectrometry. RAPID COMMUNICATIONS IN MASS SPECTROMETRY 2015, 19, 3411–3415. [Google Scholar] [CrossRef] [PubMed]
- Perlińska-Lenart, U.; Piłsyk, S.; Gryz, E.; Turło, J.; Hilszczańska, D.; Kruszewska, J.S. Identification of Bacteria and Fungi Inhabiting Fruiting Bodies of Burgundy Truffle (Tuber aestivum Vittad.). Arch Microbiol 2020, 202, 2727–2738. [Google Scholar] [CrossRef]
- Leonardi, P.; Murat, C.; Puliga, F.; Iotti, M.; Zambonelli, A. Ascoma Genotyping and Mating Type Analyses of Mycorrhizas and Soil Mycelia of Tuber Borchii in a Truffle Orchard Established by Mycelial Inoculated Plants. Environ Microbiol 2020, 22, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Murat, C.; Rubini, A.; Riccioni, C.; De la Varga, H.; Akroume, E.; Belfiori, B.; Guaragno, M.; Le Tacon, F.; Robin, C.; Halkett, F.; et al. Fine-Scale Spatial Genetic Structure of the Black Truffle (Tuber Melanosporum) Investigated with Neutral Microsatellites and Functional Mating Type Genes. New Phytologist 2013, 199, 176–187. [Google Scholar] [CrossRef]
- Moore, D. Fungal Morphogenesis. Developmental and Cell Biology Series.; Cambridge, UK.; Cambridge University Press, 1998.
- Graziosi, S.; Hall, I.R.; Zambonelli, A. The Mysteries of the White Truffle: Its Biology, Ecology and Cultivation. Encyclopedia 2022, 2, 1959–1971. [Google Scholar] [CrossRef]
- Amicucci, A.; Zambonelli, A.; Iotti, M.; Polidori, E.; Menotta, M.; Saltarelli, R.; Potenza, L.; Stocchi, V. Morphological and Molecular Modifications Induced by Different Carbohydrate Sources in Tuber Borchii. J Mol Microbiol Biotechnol 2010, 18, 120–128. [Google Scholar] [CrossRef]
- Zampieri, E.; Balestrini, R.; Kohler, A.; Abbà, S.; Martin, F.; Bonfante, P. The Perigord Black Truffle Responds to Cold Temperature with an Extensive Reprogramming of Its Transcriptional Activity. Fungal Genetics and Biology 2011, 48, 585–591. [Google Scholar] [CrossRef]
- Ling, Y.Y.; Ling, Z.L.; Zhao, R.L. Construction of a Heat-Resistant Strain of Lentinus Edodes by Fungal Hsp20 Protein Overexpression and Genetic Transformation. Front Microbiol 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- De Maio, A.; Vazquez, D. Extracellular Heat Shock Proteins: A New Location, a New Function. Shock 2013, 40, 239–246. [Google Scholar] [CrossRef]
- Kumar, S.; Sanjeev, C.; Sanjeev, C.; Mohd, S.; Shah, A. Sustainable Management of Potato Pests and Diseases. In; Chandan Maharana, Vinod Kumar Padala, A.B.H., M. Nikhil Raj, Amit Paschapur, Chaitra Bhat, A.K.S., Subbanna, and A.R.N.S., Eds.; 2022, ISBN 9789811676949.
- Keller, N.P. Fungal Secondary Metabolism: Regulation, Function and Drug Discovery. Nat Rev Microbiol 2019, 17, 167–180. [Google Scholar] [CrossRef]
- Pusztahelyi, T.; Holb, I.J.; Pócsi, I. Secondary Metabolites in Fungus-Plant Interactions. Front Plant Sci 2015, 6, 1–23. [Google Scholar] [CrossRef]
- Tang, Y.J.; Zhu, L.L. ; Li, DS; Mi, ZY; Li, HM Significance of Inoculation Density and Carbon Source on the Mycelial Growth and Tuber Polysaccharides Production by Submerged Fermentation of Chinese Truffle Tuber Sinense. Process Biochemistry 2008, 43, 576–586. [Google Scholar] [CrossRef]
- Simon Andrews FastQC. A Quality Control Tool for High Throughput Sequence Data. A Quality Control Tool for High Throughput Sequence Data. 2015.
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast Universal RNA-Seq Aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Liao, Y.; Smyth, G.K.; Shi, W. FeatureCounts: An Efficient General Purpose Program for Assigning Sequence Reads to Genomic Features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef]
- Cantalapiedra, C.P.; Hern̗andez-Plaza, A.; Letunic, I.; Bork, P.; Huerta-Cepas, J. EggNOG-Mapper v2: Functional Annotation, Orthology Assignments, and Domain Prediction at the Metagenomic Scale. Mol Biol Evol 2021, 38, 5825–5829. [Google Scholar] [CrossRef]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, L. Near-Optimal Probabilistic RNA-Seq Quantification. Nat Biotechnol 2016, 34, 525–527. [Google Scholar] [CrossRef] [PubMed]

| Culture medium | Fresh weight (mg) | Dry weight (mg) |
|---|---|---|
| WPG | 535±103 | 41±7 |
| PDB | 470±89 | 38±1 |
| MPY | 1600±270 | 127±22 |
| Sample | Total reads | RPKM | Number of genes expressed (x103) | % of annotated genes | Genes KOG |
|---|---|---|---|---|---|
| WPG | 116,376,579 | 858,673.42 | 10.1 | 82.2 | 4,554 |
| WPGY | 125,457,058 | 848,604.73 | 10.1 | 82.2 | 4,654 |
| MPY | 47,969,567 | 803,655.02 | 10.2 | 82.9 | 4,568 |
| PDB | 157,335,190 | 803,793.44 | 10.6 | 86.2 | 4,620 |
| Genes in the reference genome | 12.3 | 100 | 5,686 | ||
| Sample | 10% | 25% | 50% | 75% |
|---|---|---|---|---|
| WPG | 3/1 | 18/9 | 185/108 | 1084/669 |
| WPGY | 3/1 | 19/9 | 186/108 | 1091/669 |
| PDB | 7/2 | 44/21 | 315/171 | 1392/847 |
| MPY | 8/2 | 43/18 | 271/144 | 1250/752 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




