Submitted:
12 April 2023
Posted:
14 April 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
iPSC cell lines and maintenance
Cortical neuron differentiation and treatments
Western blots
Immunofluorescence staining
Quantification and statistical analysis
3. Results
3.1. Results
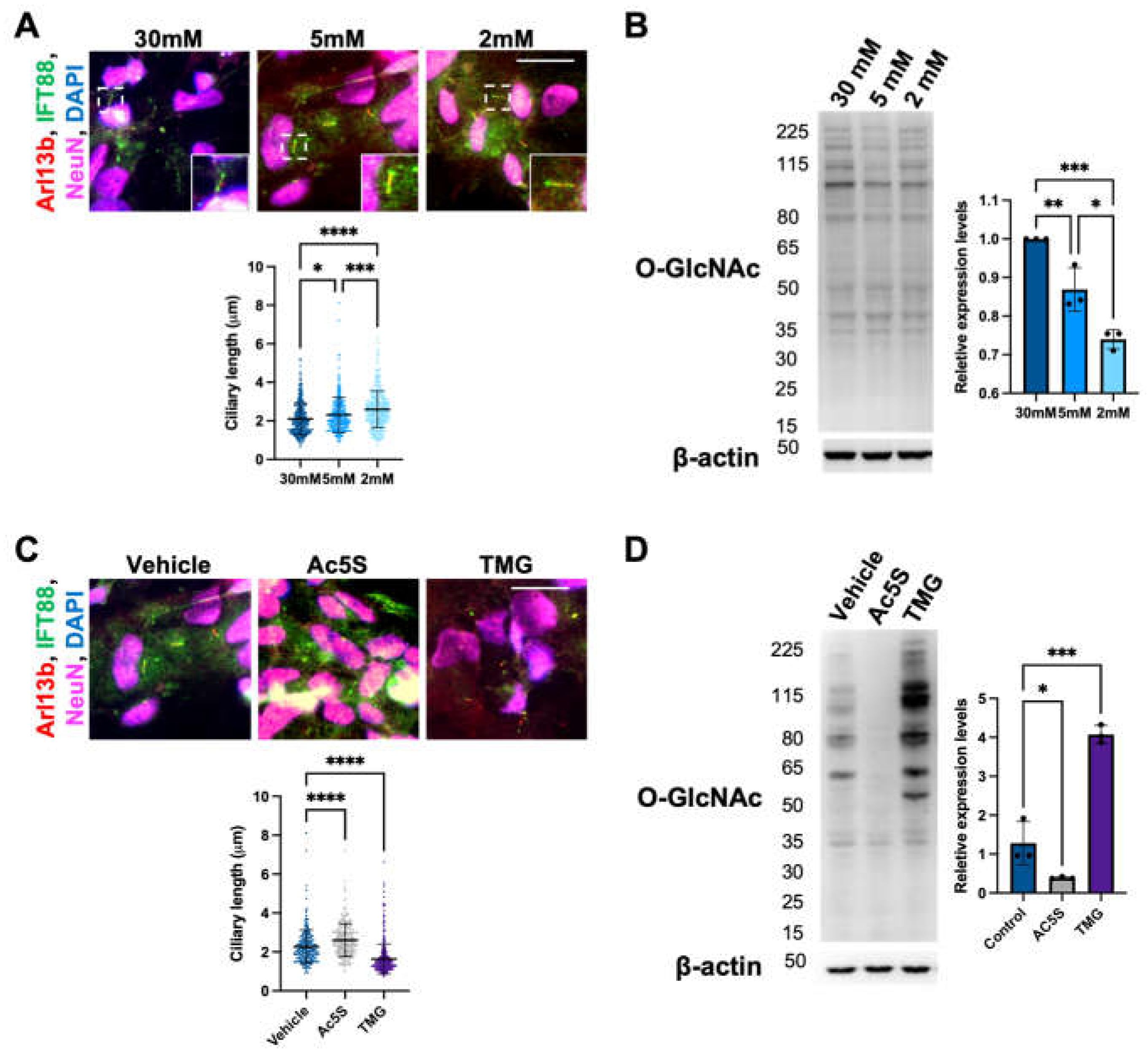
3.1.1. Cilia length in cortical neurons is negatively correlated with O-GlcNAc levels
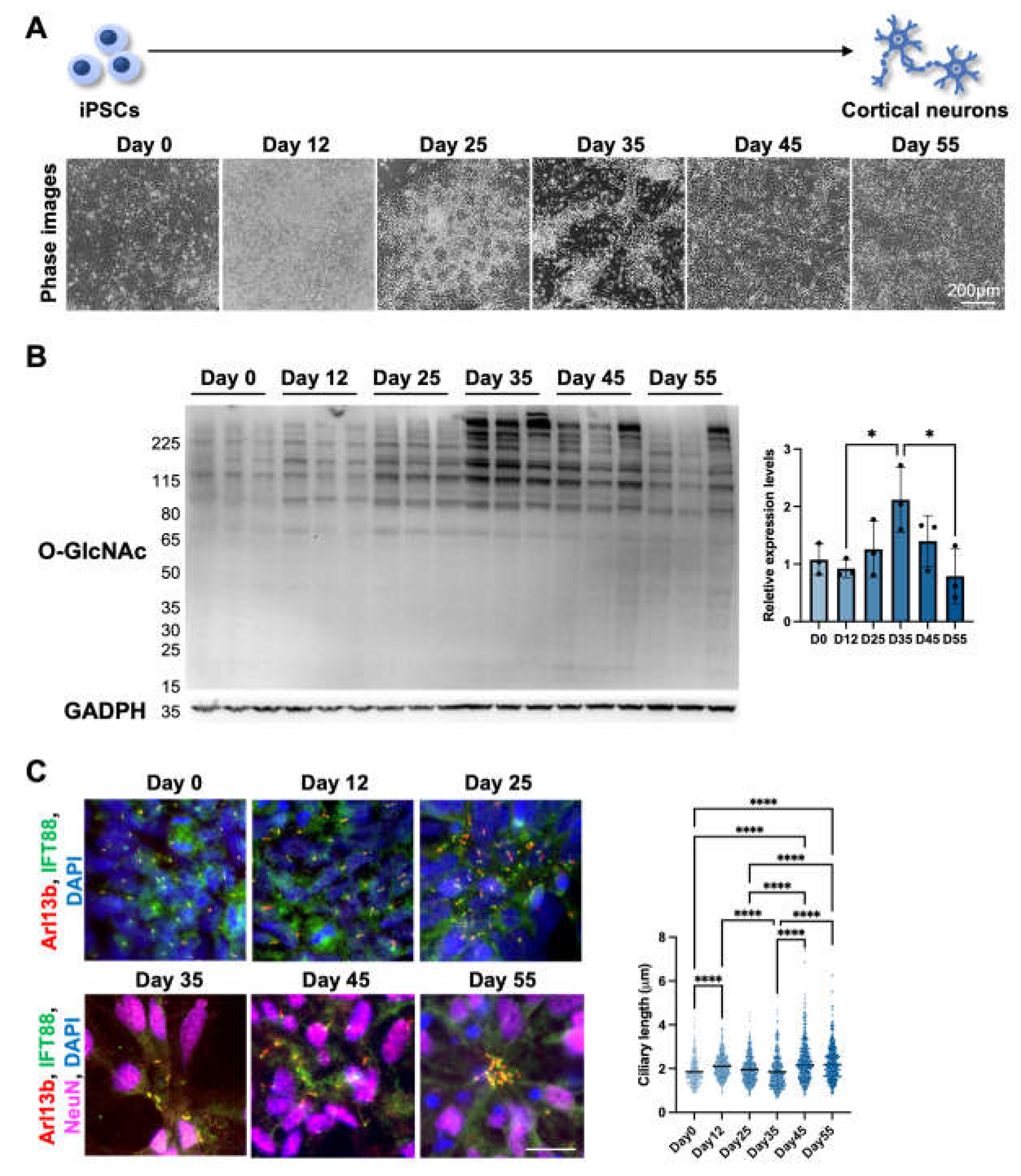
3.1.2. Cilia formation is partially regulated by O-GlcNAc levels during cortical neuron differentiation
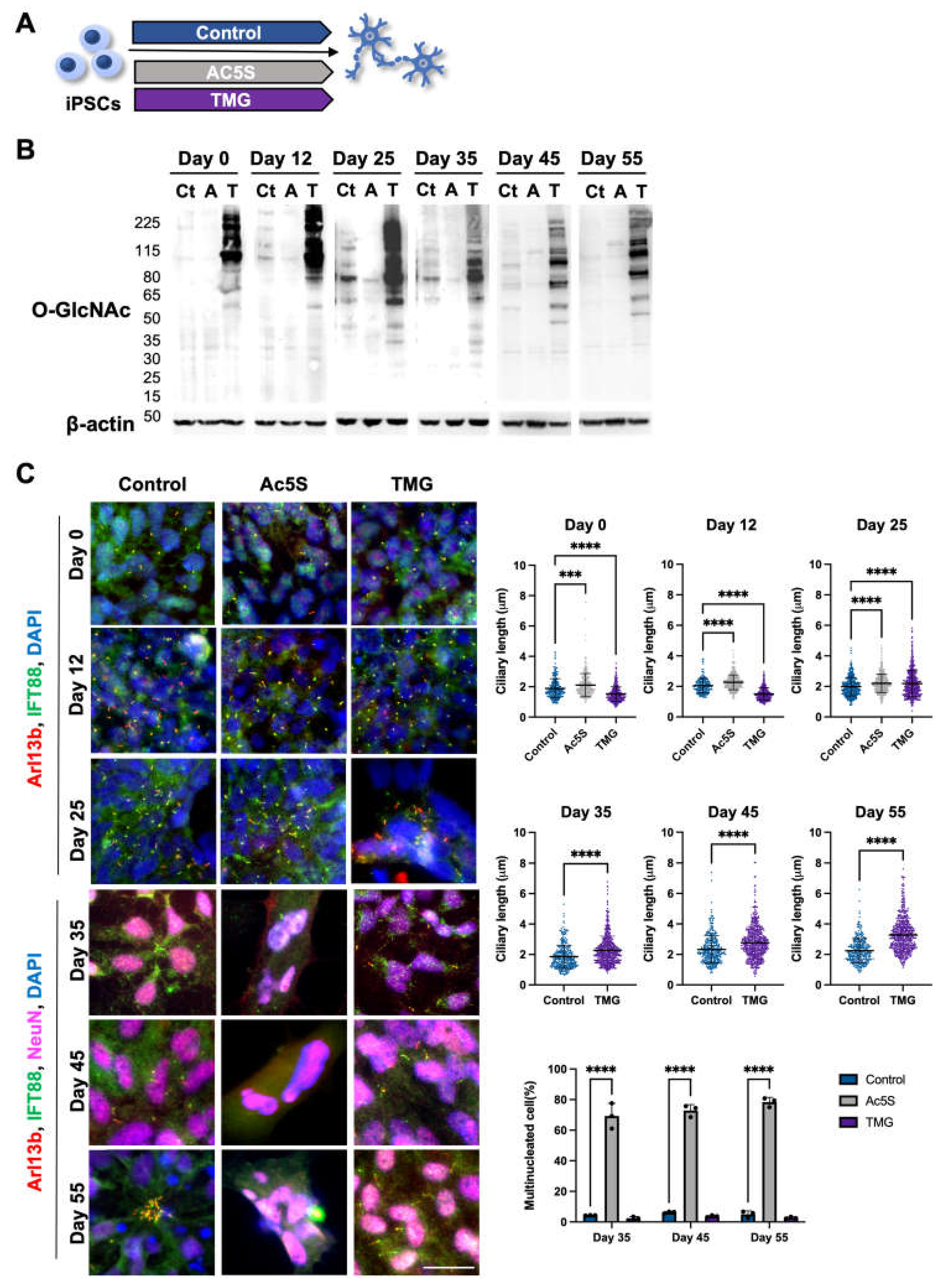
3.1.3. Long-term perturbation of OGN causes ciliary defect during cortical neuron development
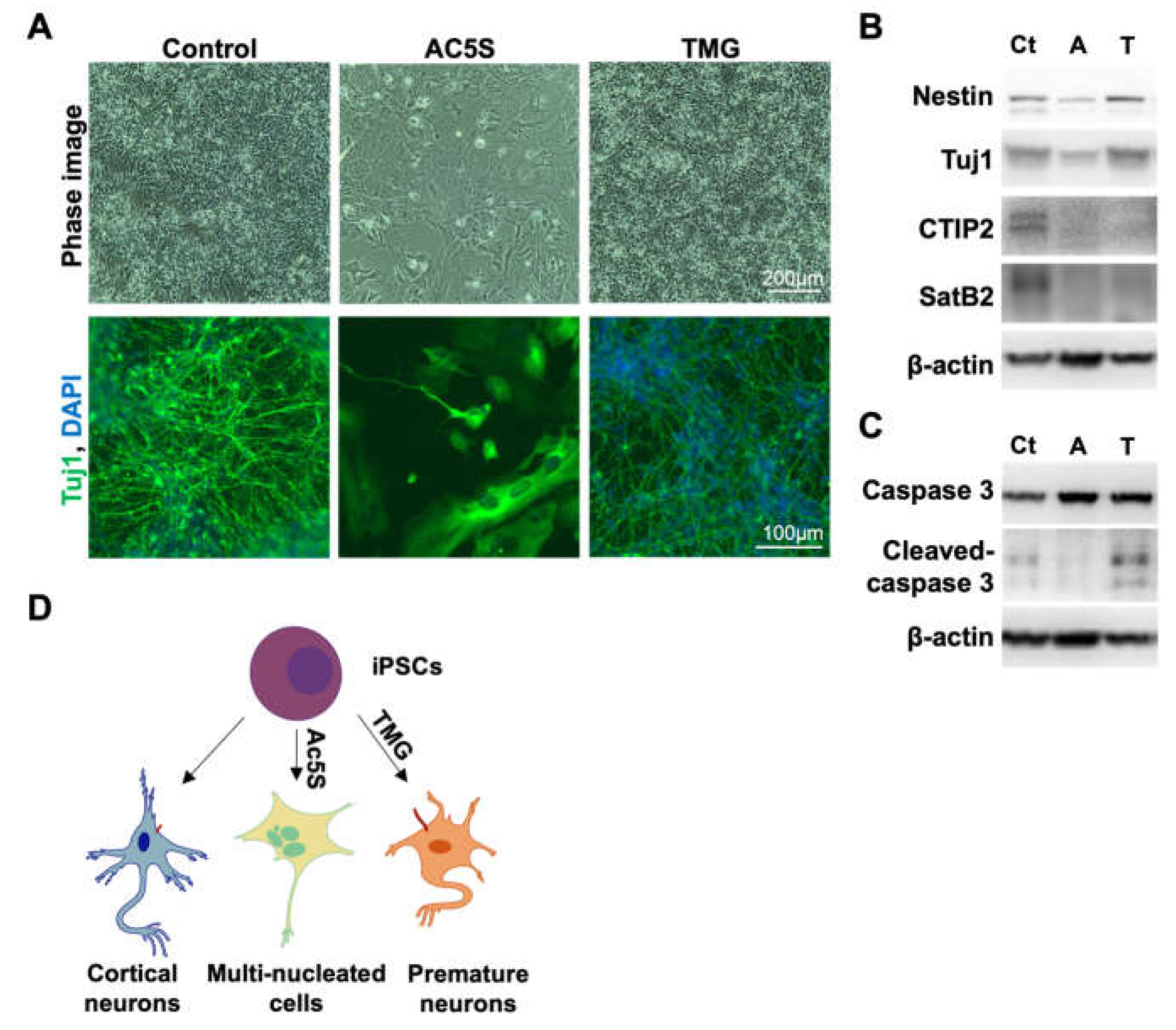
3.1.4. Altered OGN and subsequent cilia formation interferes with cortical neuronal differentiation
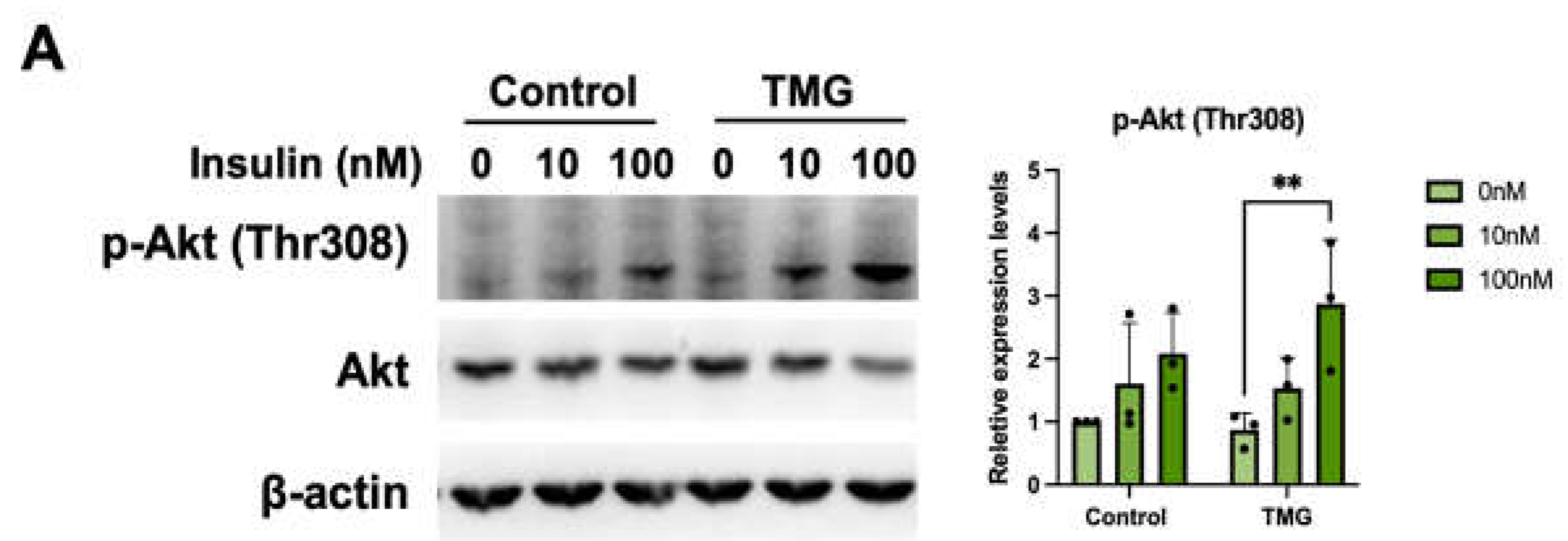
3.1.5. Increased primary cilia length leads to higher insulin sensitivity of TMG-treated neurons
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pazour, G.J. and G.B. Witman, The vertebrate primary cilium is a sensory organelle. Current Opinion in Cell Biology, 2003. 15(1): p. 105-110. [CrossRef]
- Goto, H., A. Inoko, and M. Inagaki, Cell cycle progression by the repression of primary cilia formation in proliferating cells. Cell Mol Life Sci, 2013. 70(20): p. 3893-905. [CrossRef]
- Mirvis, M., T. Stearns, and W. James Nelson, Cilium structure, assembly, and disassembly regulated by the cytoskeleton. Biochem J, 2018. 475(14): p. 2329-2353. [CrossRef]
- Plotnikova, O.V., E.A. Golemis, and E.N. Pugacheva, Cell cycle-dependent ciliogenesis and cancer. Cancer Res, 2008. 68(7): p. 2058-61. [CrossRef]
- Inoko, A., et al., Trichoplein and Aurora A block aberrant primary cilia assembly in proliferating cells. J Cell Biol, 2012. 197(3): p. 391-405. [CrossRef]
- Besschetnova, T.Y., et al., Identification of signaling pathways regulating primary cilium length and flow-mediated adaptation. Curr Biol, 2010. 20(2): p. 182-7. [CrossRef]
- Resnick, A. and U. Hopfer, Force-response considerations in ciliary mechanosensation. Biophys J, 2007. 93(4): p. 1380-90. [CrossRef]
- Sun, S., et al., Three-dimensional architecture of epithelial primary cilia. Proc Natl Acad Sci U S A, 2019. 116(19): p. 9370-9379. [CrossRef]
- Wang, L., et al., Alterations in renal cilium length during transient complete ureteral obstruction in the mouse. J Anat, 2008. 213(2): p. 79-85. [CrossRef]
- Verghese, E., et al., Renal primary cilia lengthen after acute tubular necrosis. J Am Soc Nephrol, 2009. 20(10): p. 2147-53. [CrossRef]
- Collin, G.B., et al., Mutations in ALMS1 cause obesity, type 2 diabetes and neurosensory degeneration in Alstrom syndrome. Nat Genet, 2002. 31(1): p. 74-8.
- Romani, M., A. Micalizzi, and E.M. Valente, Joubert syndrome: congenital cerebellar ataxia with the molar tooth. The Lancet Neurology, 2013. 12(9): p. 894-905. [CrossRef]
- Hernandez-Hernandez, V., et al., Bardet-Biedl syndrome proteins control the cilia length through regulation of actin polymerization. Hum Mol Genet, 2013. 22(19): p. 3858-68. [CrossRef]
- May-Simera, H.L., et al., Primary Cilium-Mediated Retinal Pigment Epithelium Maturation Is Disrupted in Ciliopathy Patient Cells. Cell Rep, 2018. 22(1): p. 189-205. [CrossRef]
- Shi, H., et al., Mutations in OSBPL2 cause hearing loss associated with primary cilia defects via sonic hedgehog signaling. JCI Insight, 2022. 7(4). [CrossRef]
- Luo, N., et al., Primary cilia signaling mediates intraocular pressure sensation. Proc Natl Acad Sci U S A, 2014. 111(35): p. 12871-6. [CrossRef]
- McEwen, D.P., et al., Hypomorphic CEP290/NPHP6 mutations result in anosmia caused by the selective loss of G proteins in cilia of olfactory sensory neurons. Proc Natl Acad Sci U S A, 2007. 104(40): p. 15917-22.
- Butler, M.T. and J.B. Wallingford, Planar cell polarity in development and disease. Nat Rev Mol Cell Biol, 2017. 18(6): p. 375-388. [CrossRef]
- Ehnert, S., et al., TGF-beta1 impairs mechanosensation of human osteoblasts via HDAC6-mediated shortening and distortion of primary cilia. J Mol Med (Berl), 2017. 95(6): p. 653-663.
- Zullo, A., et al., Kidney-specific inactivation of Ofd1 leads to renal cystic disease associated with upregulation of the mTOR pathway. Hum Mol Genet, 2010. 19(14): p. 2792-803. [CrossRef]
- Grisanti, L., et al., Primary cilia maintain corneal epithelial homeostasis by regulation of the Notch signaling pathway. Development, 2016. 143(12): p. 2160-71. [CrossRef]
- Kim, M., et al., The MST1/2-SAV1 complex of the Hippo pathway promotes ciliogenesis. Nat Commun, 2014. 5: p. 5370.
- Town, T., et al., The stumpy gene is required for mammalian ciliogenesis. Proc Natl Acad Sci U S A, 2008. 105(8): p. 2853-8. [CrossRef]
- Breunig, J.J., et al., Primary cilia regulate hippocampal neurogenesis by mediating sonic hedgehog signaling. Proc Natl Acad Sci U S A, 2008. 105(35): p. 13127-32. [CrossRef]
- Baudoin, J.P., et al., Tangentially migrating neurons assemble a primary cilium that promotes their reorientation to the cortical plate. Neuron, 2012. 76(6): p. 1108-22. [CrossRef]
- Bae, J.E., et al., Primary cilia mediate mitochondrial stress responses to promote dopamine neuron survival in a Parkinson's disease model. Cell Death Dis, 2019. 10(12): p. 952. [CrossRef]
- Li, A., et al., Ciliary transition zone activation of phosphorylated Tctex-1 controls ciliary resorption, S-phase entry and fate of neural progenitors. Nat Cell Biol, 2011. 13(4): p. 402-11. [CrossRef]
- Lee, C.H., et al., Primary cilia mediate early life programming of adiposity through lysosomal regulation in the developing mouse hypothalamus. Nat Commun, 2020. 11(1): p. 5772. [CrossRef]
- Choi, H., et al., Nucleus pulposus primary cilia alter their length in response to changes in extracellular osmolarity but do not control TonEBP-mediated osmoregulation. Sci Rep, 2019. 9(1): p. 15469.
- Matsumoto, M., et al., Dynamic Changes in Ultrastructure of the Primary Cilium in Migrating Neuroblasts in the Postnatal Brain. J Neurosci, 2019. 39(50): p. 9967-9988. [CrossRef]
- Stilling, S., et al., PIP2 determines length and stability of primary cilia by balancing membrane turnovers. Commun Biol, 2022. 5(1): p. 93. [CrossRef]
- Sachamitr, P., et al., PRMT5 inhibition disrupts splicing and stemness in glioblastoma. Nat Commun, 2021. 12(1): p. 979. [CrossRef]
- Tian, J.L. and H. Qin, OGN Regulates Primary Ciliary Length by Promoting Microtubule Disassembly. iScience, 2019. 12: p. 379-391.
- Yang, Y., et al., Mixed-lineage leukemia protein 2 suppresses ciliary assembly by the modulation of actin dynamics and vesicle transport. Cell Discov, 2019. 5: p. 33. [CrossRef]
- Yang, Y., et al., CYLD mediates ciliogenesis in multiple organs by deubiquitinating Cep70 and inactivating HDAC6. Cell Res, 2014. 24(11): p. 1342-53. [CrossRef]
- Ozcan, S., S.S. Andrali, and J.E. Cantrell, Modulation of transcription factor function by O-GlcNAc modification. Biochim Biophys Acta, 2010. 1799(5-6): p. 353-64.
- Li, C., et al., Hypertonic stress modulates eNOS function through O-GlcNAc modification at Thr-866. Sci Rep, 2021. 11(1): p. 11272.
- Wang, S., et al., Extensive crosstalk between OGN and phosphorylation regulates Akt signaling. PLoS One, 2012. 7(5): p. e37427.
- Yi, W., et al., Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science, 2012. 337(6097): p. 975-80. [CrossRef]
- Hart, G.W., Three Decades of Research on OGN - A Major Nutrient Sensor That Regulates Signaling, Transcription and Cellular Metabolism. Front Endocrinol (Lausanne), 2014. 5: p. 183.
- Swamy, M., et al., Glucose and glutamine fuel protein OGN to control T cell self-renewal and malignancy. Nat Immunol, 2016. 17(6): p. 712-20.
- Guo, H. Guo, H., et al., O-Linked N-Acetylglucosamine (O-GlcNAc) Expression Levels Epigenetically Regulate Colon Cancer Tumorigenesis by Affecting the Cancer Stem Cell Compartment via Modulating Expression of Transcriptional Factor MYBL1. J Biol Chem, 2017. 292(10): p. 4123-4137.
- Gelinas, R., et al., AMPK activation counteracts cardiac hypertrophy by reducing OGN. Nat Commun, 2018. 9(1): p. 374.
- Umapathi, P., et al., Excessive OGN Causes Heart Failure and Sudden Death. Circulation, 2021. 143(17): p. 1687-1703.
- Ferrer, C.M., et al., OGN regulates cancer metabolism and survival stress signaling via regulation of the HIF-1 pathway. Mol Cell, 2014. 54(5): p. 820-31.
- de Queiroz, R.M., et al., Changes in O-Linked N-Acetylglucosamine (O-GlcNAc) Homeostasis Activate the p53 Pathway in Ovarian Cancer Cells. J Biol Chem, 2016. 291(36): p. 18897-914.
- Peterson, S.B. and G.W. Hart, New insights: A role for OGN in diabetic complications. Crit Rev Biochem Mol Biol, 2016. 51(3): p. 150-61.
- Park, J., et al., OGN ameliorates the pathological manifestations of Alzheimer's disease by inhibiting necroptosis. Sci Adv, 2021. 7(3).
- Liu, F., et al., OGN regulates phosphorylation of tau: a mechanism involved in Alzheimer's disease. Proc Natl Acad Sci U S A, 2004. 101(29): p. 10804-9.
- Constable, S., et al., O-GlcNAc transferase regulates transcriptional activity of human Oct4. Glycobiology, 2017. 27(10): p. 927-937.
- Akinbiyi, E.O., et al., Blocked O-GlcNAc cycling alters mitochondrial morphology, function, and mass. Sci Rep, 2021. 11(1): p. 22106.
- Park, S.J., et al., Increased OGN of Drp1 by amyloid-beta promotes mitochondrial fission and dysfunction in neuronal cells. Mol Brain, 2021. 14(1): p. 6.
- Shin, H., et al., OGN of the Tumor Suppressor FOXO3 Triggers Aberrant Cancer Cell Growth. Cancer Res, 2018. 78(5): p. 1214-1224.
- Yu, F., et al., Ciliary defects caused by dysregulation of O-GlcNAc modification are associated with diabetic complications. Cell Res, 2019. 29(2): p. 171-173.
- Zachara, N.E. and G.W. Hart, Cell signaling, the essential role of O-GlcNAc! Biochim Biophys Acta, 2006. 1761(5-6): p. 599-617.
- Gabriel, E., et al., CPAP promotes timely cilium disassembly to maintain neural progenitor pool. EMBO J, 2016. 35(8): p. 803-19. [CrossRef]
- Ma, R., et al., Primary cilia and ciliary signaling pathways in aging and age-related brain disorders. Neurobiol Dis, 2022. 163: p. 105607. [CrossRef]
- Ishii, S., et al., Primary cilia safeguard cortical neurons in neonatal mouse forebrain from environmental stress-induced dendritic degeneration. Proc Natl Acad Sci U S A, 2021. 118(1). [CrossRef]
- Guemez-Gamboa, A., N.G. Coufal, and J.G. Gleeson, Primary cilia in the developing and mature brain. Neuron, 2014. 82(3): p. 511-21. [CrossRef]
- Tian, J.L. and F.I. Gomeshtapeh, Potential Roles of OGN in Primary Cilia- Mediated Energy Metabolism. Biomolecules, 2020. 10(11).
- Han, Y.M., et al., Leptin-promoted cilia assembly is critical for normal energy balance. J Clin Invest, 2014. 124(5): p. 2193-7. [CrossRef]
- Kirk, S.L., et al., Maternal obesity induced by diet in rats permanently influences central processes regulating food intake in offspring. PLoS One, 2009. 4(6): p. e5870. [CrossRef]
- Rocha, C. and P. Prinos, Post-transcriptional and Post-translational Modifications of Primary Cilia: How to Fine Tune Your Neuronal Antenna. Front Cell Neurosci, 2022. 16: p. 809917. [CrossRef]
- Wang, G., et al., Regulation of neural progenitor cell motility by ceramide and potential implications for mouse brain development. J Neurochem, 2008. 106(2): p. 718-33. [CrossRef]
- He, Q., et al., Primary cilia in stem cells and neural progenitors are regulated by neutral sphingomyelinase 2 and ceramide. Mol Biol Cell, 2014. 25(11): p. 1715-29. [CrossRef]
- Thomas, S., et al., Cilia in hereditary cerebral anomalies. Biol Cell, 2019. 111(9): p. 217-231. [CrossRef]
- Gerdes, J.M., E.E. Davis, and N. Katsanis, The vertebrate primary cilium in development, homeostasis, and disease. Cell, 2009. 137(1): p. 32-45. [CrossRef]
- Li, Z., et al., Checkpoint kinase 1-induced phosphorylation of O-linked beta-N-acetylglucosamine transferase regulates the intermediate filament network during cytokinesis. J Biol Chem, 2017. 292(48): p. 19548-19555.
- Si-Tayeb, K., et al., Generation of human induced pluripotent stem cells by simple transient transfection of plasmid DNA encoding reprogramming factors. BMC Dev Biol, 2010. 10: p. 81. [CrossRef]
- Wu, H.F. and N. Zeltner, Efficient Differentiation of Postganglionic Sympathetic Neurons using Human Pluripotent Stem Cells under Feeder-free and Chemically Defined Culture Conditions. J Vis Exp, 2020(159).
- Shi, Y., et al., Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses. Nat Neurosci, 2012. 15(3): p. 477-86, S1. [CrossRef]
- Shi, Y., P. Kirwan, and F.J. Livesey, Directed differentiation of human pluripotent stem cells to cerebral cortex neurons and neural networks. Nat Protoc, 2012. 7(10): p. 1836-46. [CrossRef]
- King, C.R., et al., Fbxo41 Promotes Disassembly of Neuronal Primary Cilia. Sci Rep, 2019. 9(1): p. 8179. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



