Submitted:
15 March 2023
Posted:
15 March 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
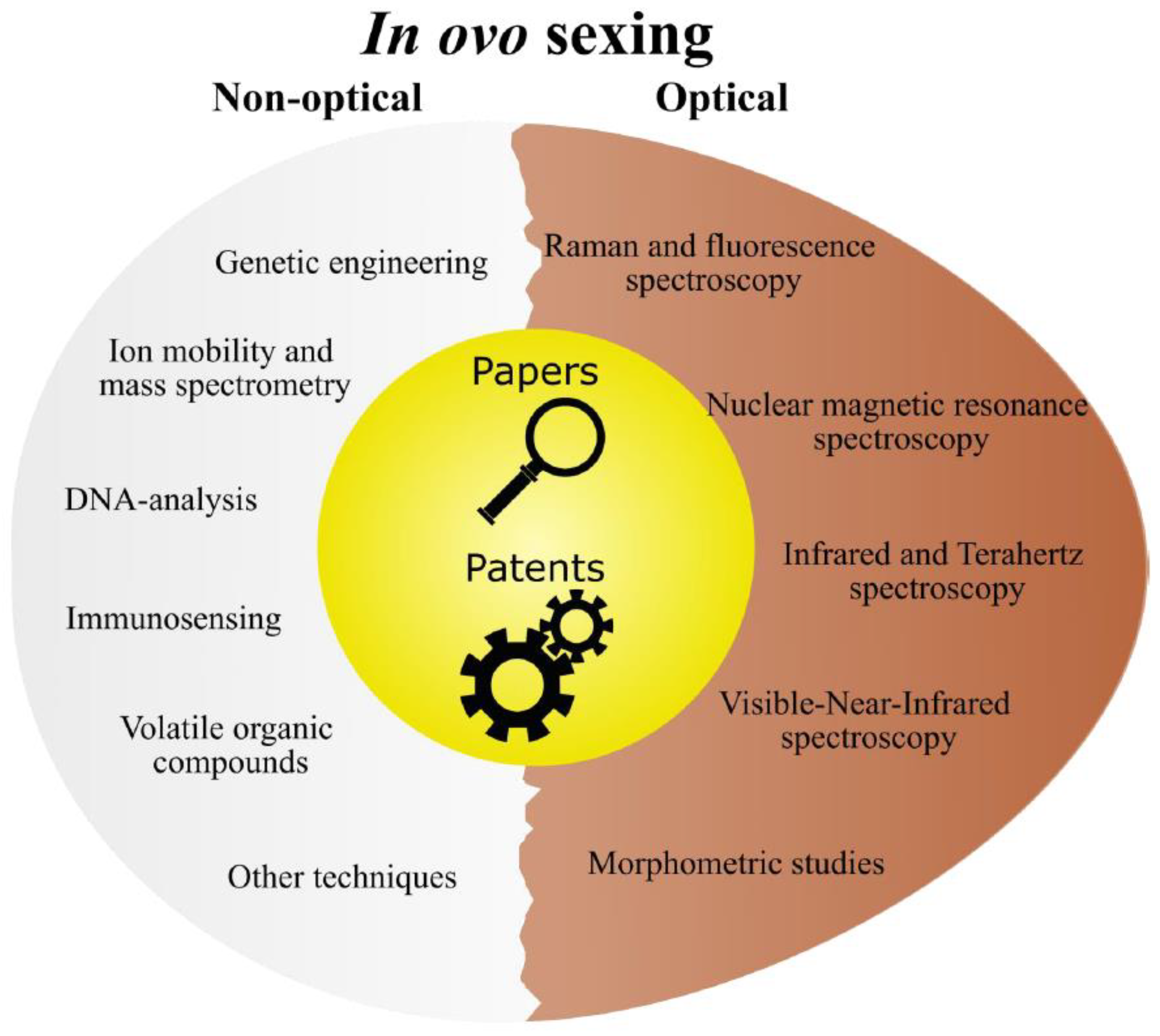
2. Non-optical in ovo sexing methods
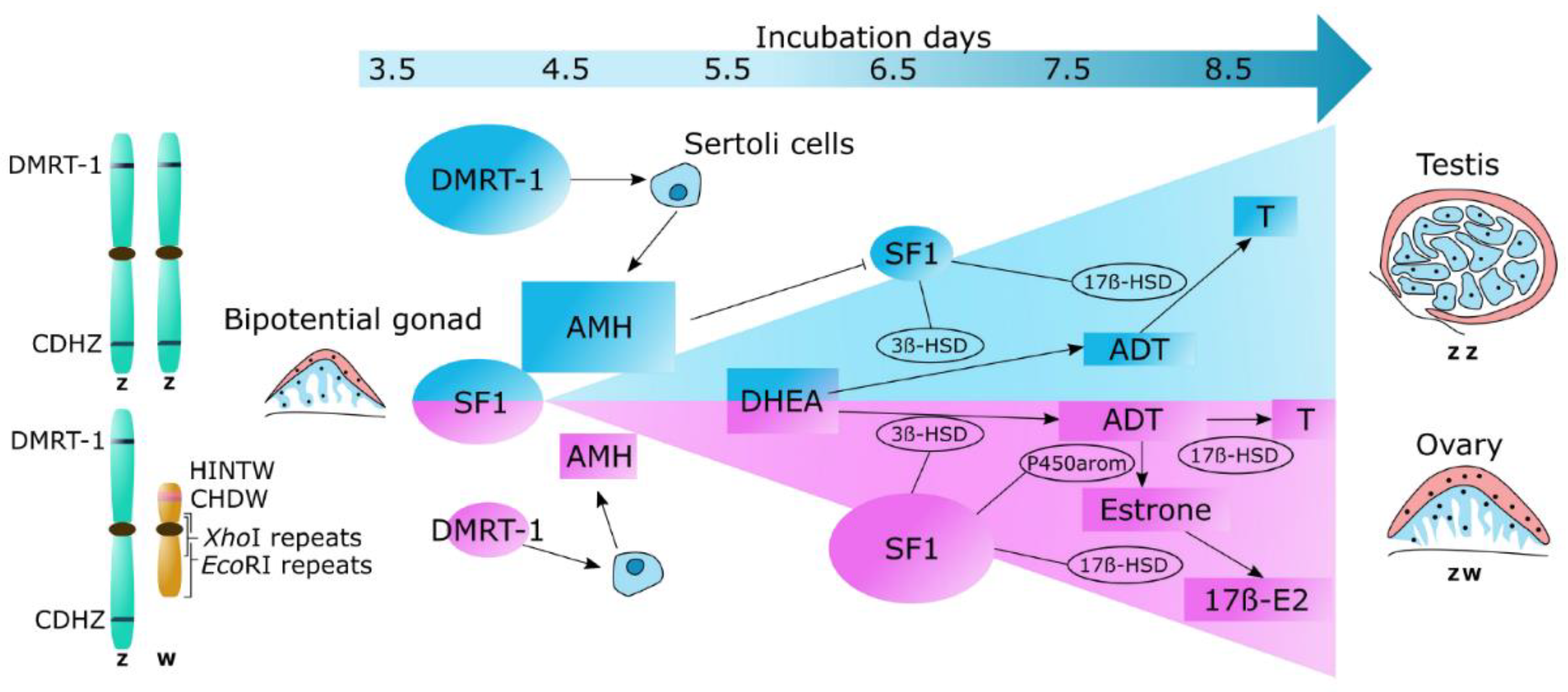
2.1. DNA analysis
2.2. Immunosensing
| Hormone | Sample | Detection (days) | Remarks | Reference |
| Papers | ||||
| Luteinizing hormone, T, E2, P4 | Blood | 17 and 20 | Detection with RIA Progesterone and estradiol with the bigger differences between males and females |
Tanabe et al. [23] |
| E1, E2-17β, E2-17αsulphate, E1, E2-17β, E2-17αglucuronide | AF | 8 to 17 | Detection with RIA | Gill et al. [24] |
| T, A4 | Yolk | Differences were not sufficient for sex determination | Müller et al. [31] | |
| E2 | AF | 17 | Detection with RIA Significant differences |
Phelps et al. [11] |
| E2-17ss | AF | 17 | Use of a yeast transactivation assay Significant differences |
Tran et al. [28] |
| T, E2, A4, P4, DHT | Yolk | Differences were not sufficient for sex determination | Aslam et al. [32] | |
| E1S, E2, T | AF | 9 and 10 | Detection with ELISA Significant differences for E1S on day 9 and for E2 on day 10 |
Weissmann et al. [30] |
| E2, T, E1, A4, DHT | Serum | 8 to 16 | Detection with ELISA Significant differences with T, DHT, and E1 |
Wang et al. [33] |
| Patents | ||||
| E2, E2-17ss, estriol, E1, T, DHT | AF | 11 to 19 | Phelps [25] | |
| E2, E2-17ss, estriol, E1, T, DHT | AF | 11 to 19 | Tyczkowski et al. [26] | |
| E2 | AF | 17 | Butt and Tran [27] | |
| E1S | AF | 9 and 10 | Einspanier [29] | |
2.3. Ion mobility- and mass spectrometry
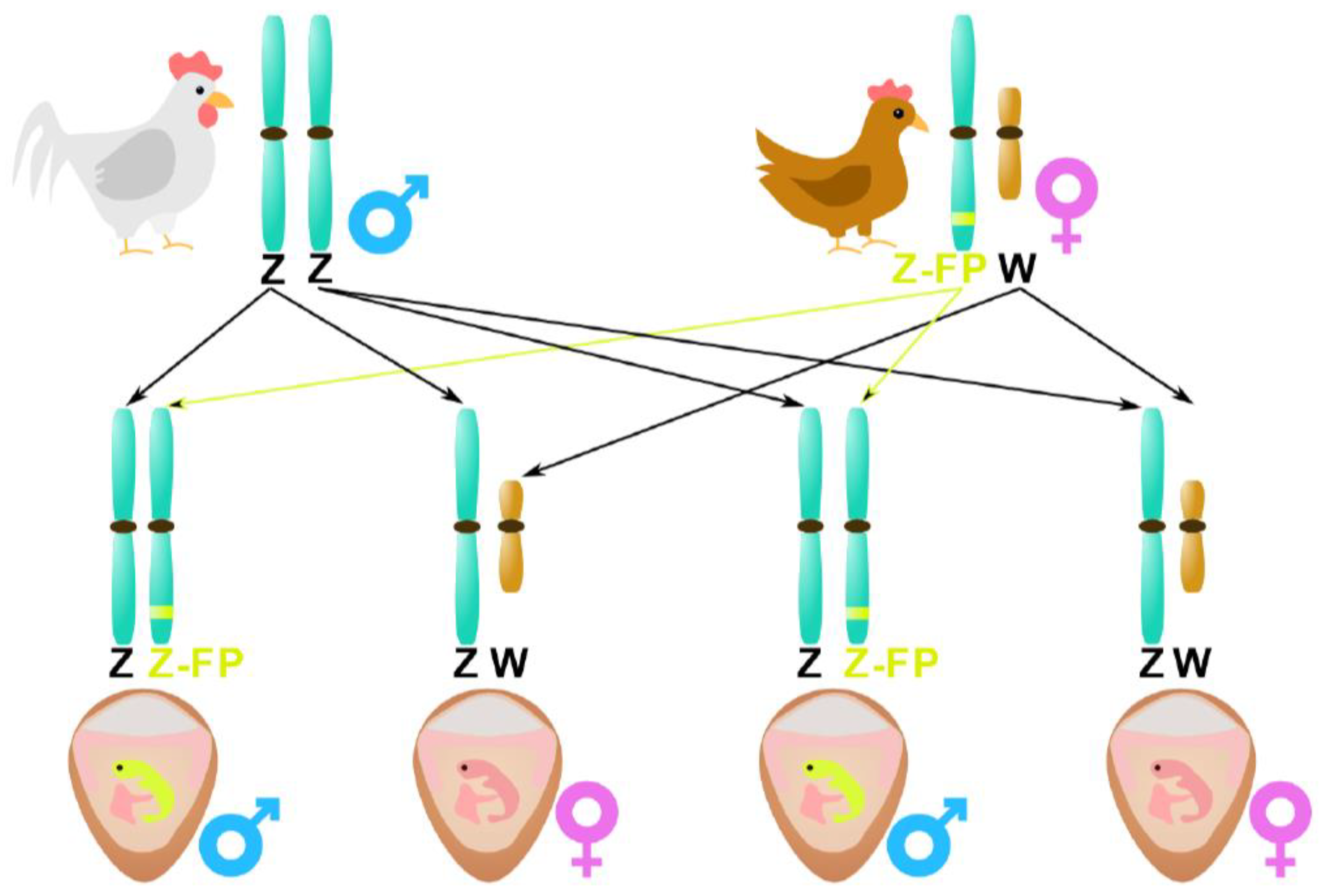
2.4. Genetic engineering
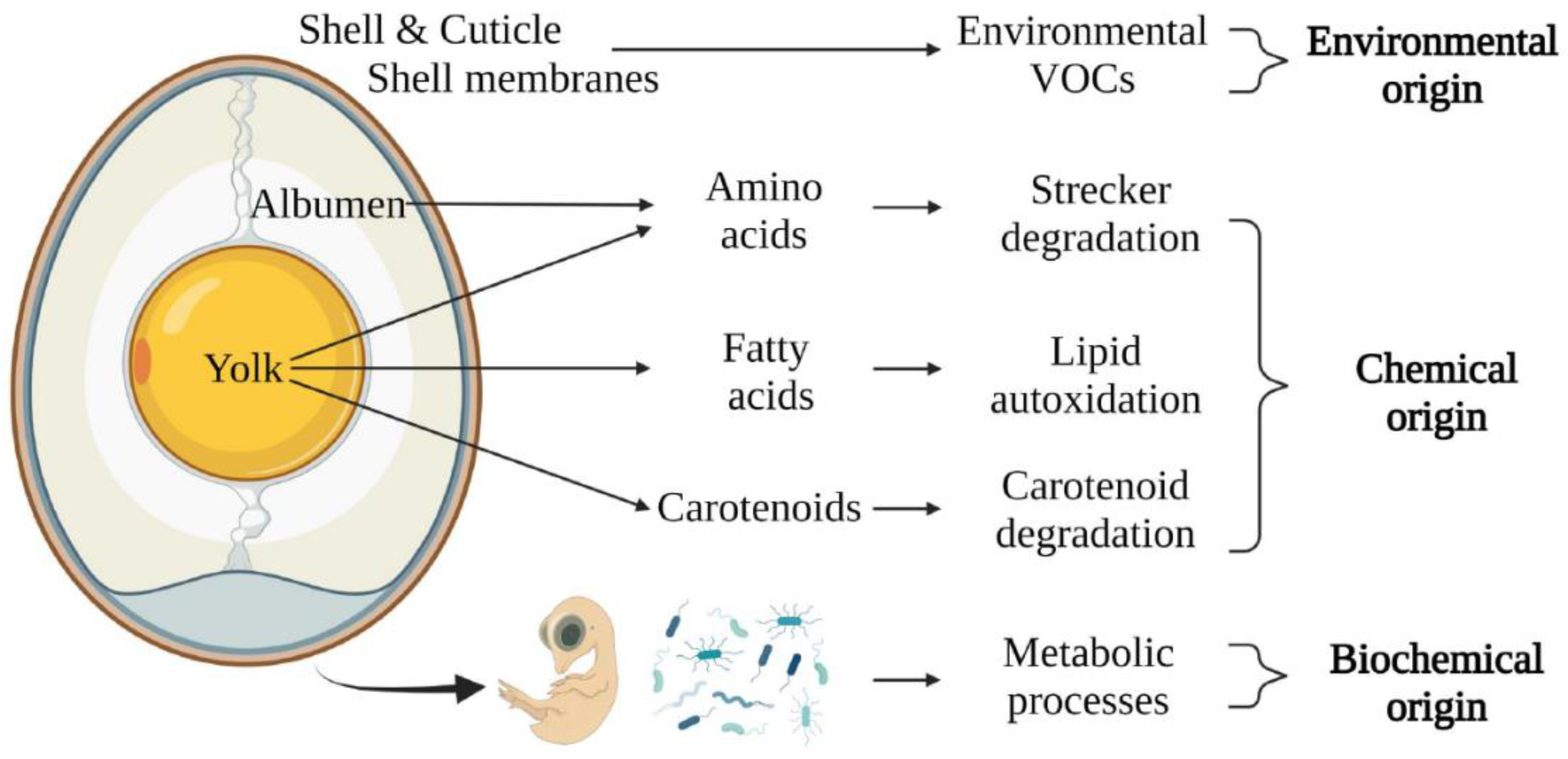
2.5. Volatile Organic Compounds
2.6. Other techniques
| Category | Target |
Incubation day Accuracy Throughput |
Technology (patents)/ research gaps (papers) |
Advantages (+)/ Drawbacks (-) |
References |
| DNA analysis | gDNA (e.g., CHD, XhoI, SWIM, HINTW) from different samples (e.g., AF, blood, feathers, brain) | Days 8 to 13 100 % Slow (slower in case of need for DNA extraction), PCR can take more than 60 minutes |
Technology > Research Lack of papers using AF Several possible female-specific sequences are not reported in the literature |
+ High accuracy Early in incubation (> day 2.5) - Lengthy processes and expensive Can be highly invasive if not using AF |
Patents ⊗ Griffiths [14], ⊗ Clinton [19], ∅ Dalton et al. [59], ∅ Arie and Danielli [60], ∅ Molina and Espeut [61] Papers Li et al. [15], Haunshi et al. [17], Clinton et al. [18], He et al. [20] |
| Immunosensing | Sexual hormones (e.g., E2 or estrone-3-sulfate) in AF or blood | Minimum day 8 Reported as 99 % on day 9 Relatively slow using ELISA/RIA, which can take up to 90 minutes. |
Technology ≅ Research Well defined in both papers and patents. |
+ Applicable in AF - Limited to after day 8 of incubation. Lower accuracies than DNA analysis Expensive and lengthy testing. |
Patents ∅ Tyczkowski et al. [26], ⊕ Butt and Tran [27], ∅ Einspanier [29], ⊕ Phelps [25] Papers Phelps et al. [11], Tanabe et al. [23], Weissmann et al. [30,62], Müller et al. [31], Aslam et al. [32], Wang et al. [33] |
| IMS and MS | Metabolites in AF | Days 9 to 10 90 % to higher than 95 % Relatively fast |
Technology > Research Only defined by two different groups of inventors in the patent literature No work has been published in the scientific literature |
+ Quick sampling and relatively fast analysis - No scientific literature on physiological background |
Patents ∅ Daum and Atkinson [37], ∅ Bruins and Stutterheim [38,63], ∅ Stutterheim [64] Papers - |
| Genetic engineering | gDNA | Days 0 to 2.5 100 % High if using labels |
Technology > Research Only one short communication in papers, mainly present in patents. |
+ High accuracies at an early incubation stage. - Not accepted in several countries (see text) Invasive injection of a labeled molecule (antibody/DNA sequence) |
Patents ∅ Decuypere and Fey [34], ∅ Offen [41], ⊗ Beisswanger [42] Papers Doran et al. [40] |
| VOC analysis | VOCs released from the egg | Day 0 or during incubation Accuracy not defined Seconds to minutes |
Technology > Research Patents use real-time analysis systems such as THz spectroscopy, SIFT- or PTR-MS Research papers use conventional SPME-GC-MS analysis and more research is required for generating robust sexing models |
+ Non-invasive Potential for early-stage application - Contamination risk of egg odor by environment or other eggs |
Patents ∅ Rivers [45], ∅ Gabbai [48], ∅ Knepper et al. [65], ⊕ Tongli et al. [66] Papers Webster et al. [43], Costanzo et al. [44], Xiang et al. [49] |
| Other | 1) Electrical and magnetic polarity of the egg 2) Dynamic electromagnetic spectrum 3) Sex-specific compounds close to the eggshell |
Day 0 Accuracies unknown No throughputs defined |
Technology > Research Only defined in different patents and no scientific literature |
+ Before incubation - No scientific evidence |
Patents ⊗ Young [51], ⊗ Williams [52,53], ⊗ Chadfield [55], ⊗ Pacala et al. [56], ⊕ Visser [57], ∅ Rastoutsau et al. [58] Papers - |
| Abbreviations: d, day; gDNA, genomic DNA; AF, allantoic fluid; E2, estradiol; ELISA, enzyme-linked immunosorbent assay; RIA, radioimmunoassay; THz, terahertz; SPME-GC-MS, solid phase micro extraction–gas chromatography-mass spectrometry; SIFT-MS, selected ion flow tube-mass spectrometry; PTR-MS, proton transfer reaction-mass spectrometry; IMS and MS, ion mobility and mass spectrometry; ⊕, granted; ∅, pending; ⊗, ceased/rejected/discontinued. | |||||
3. Optical in ovo sexing methods
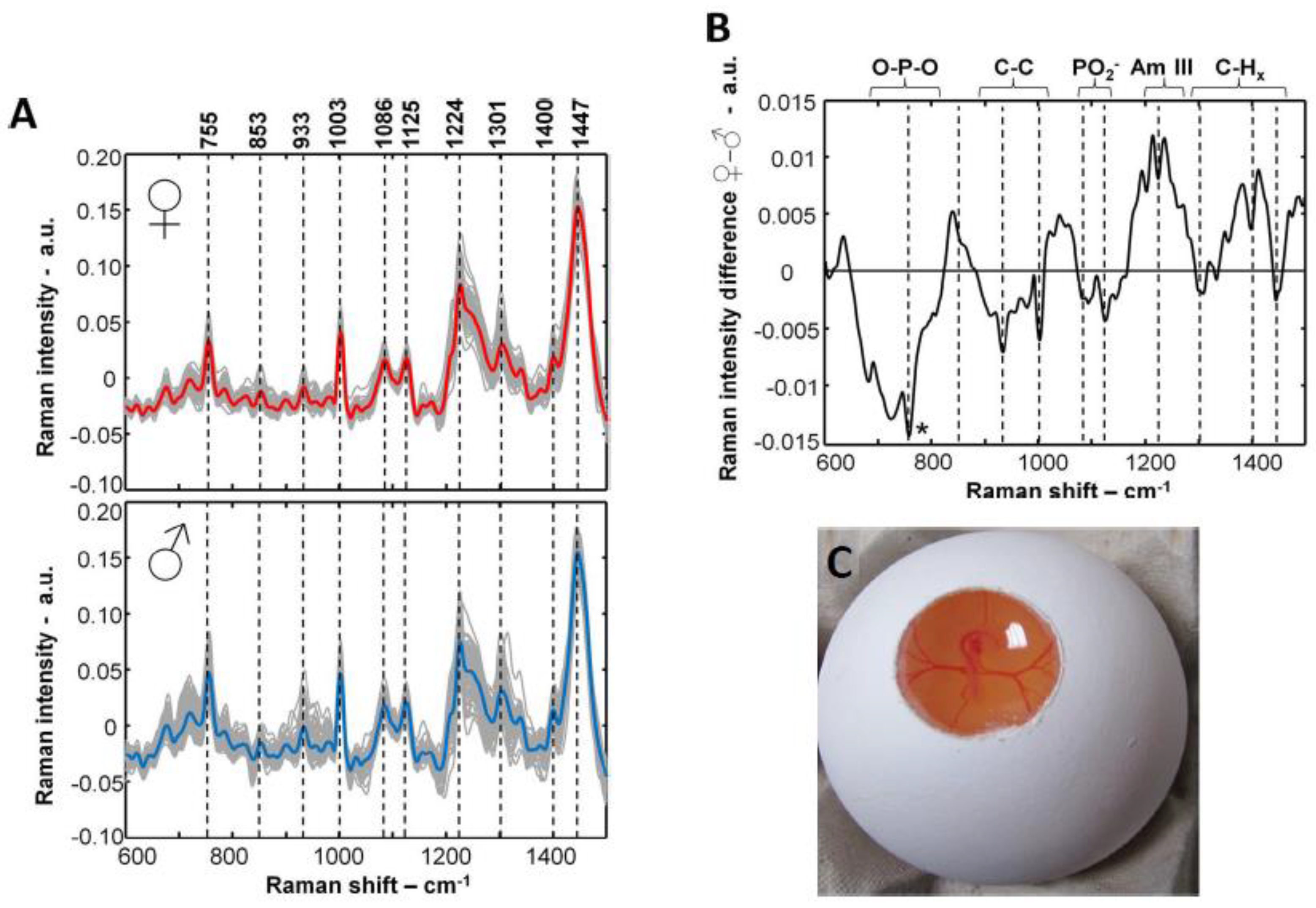
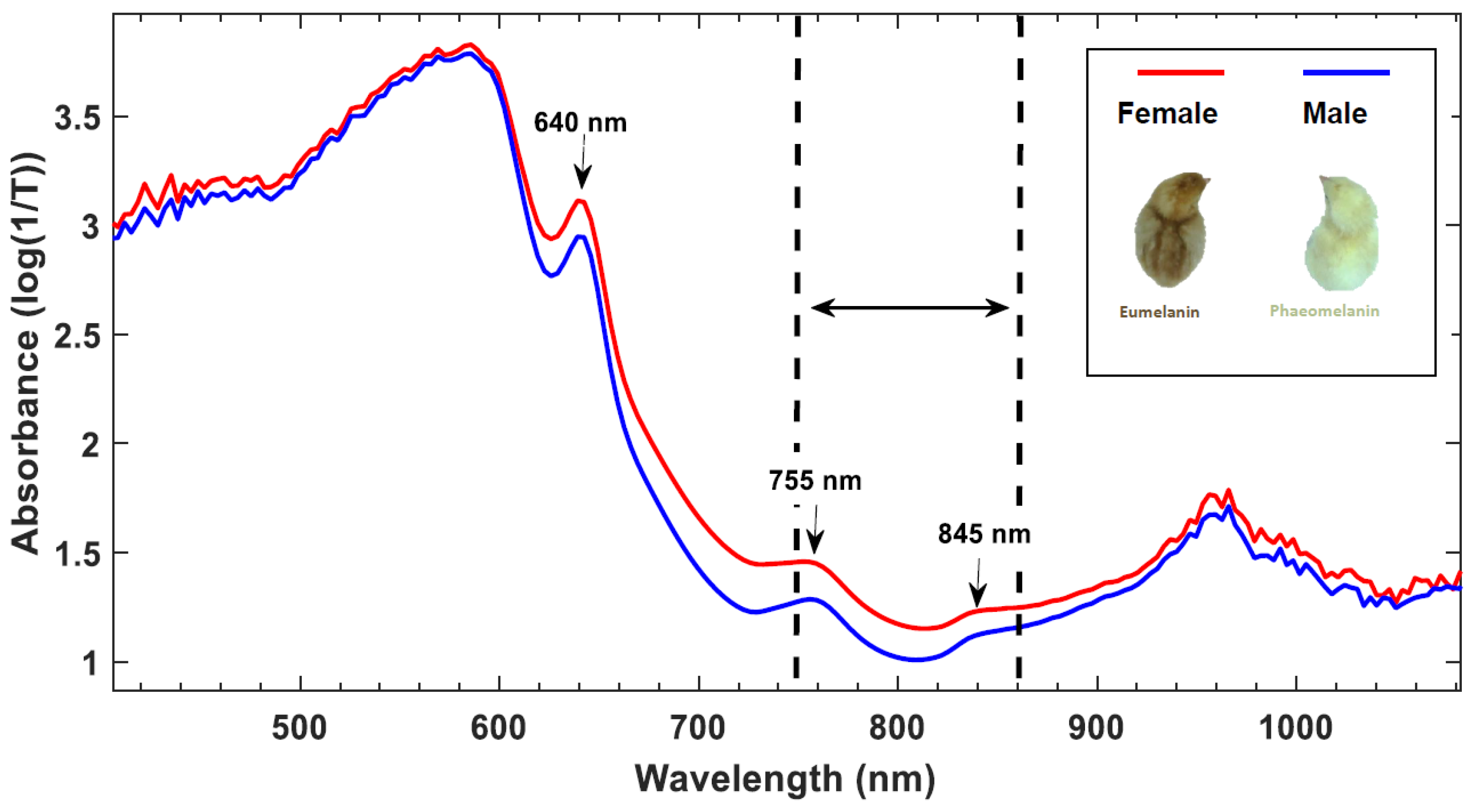
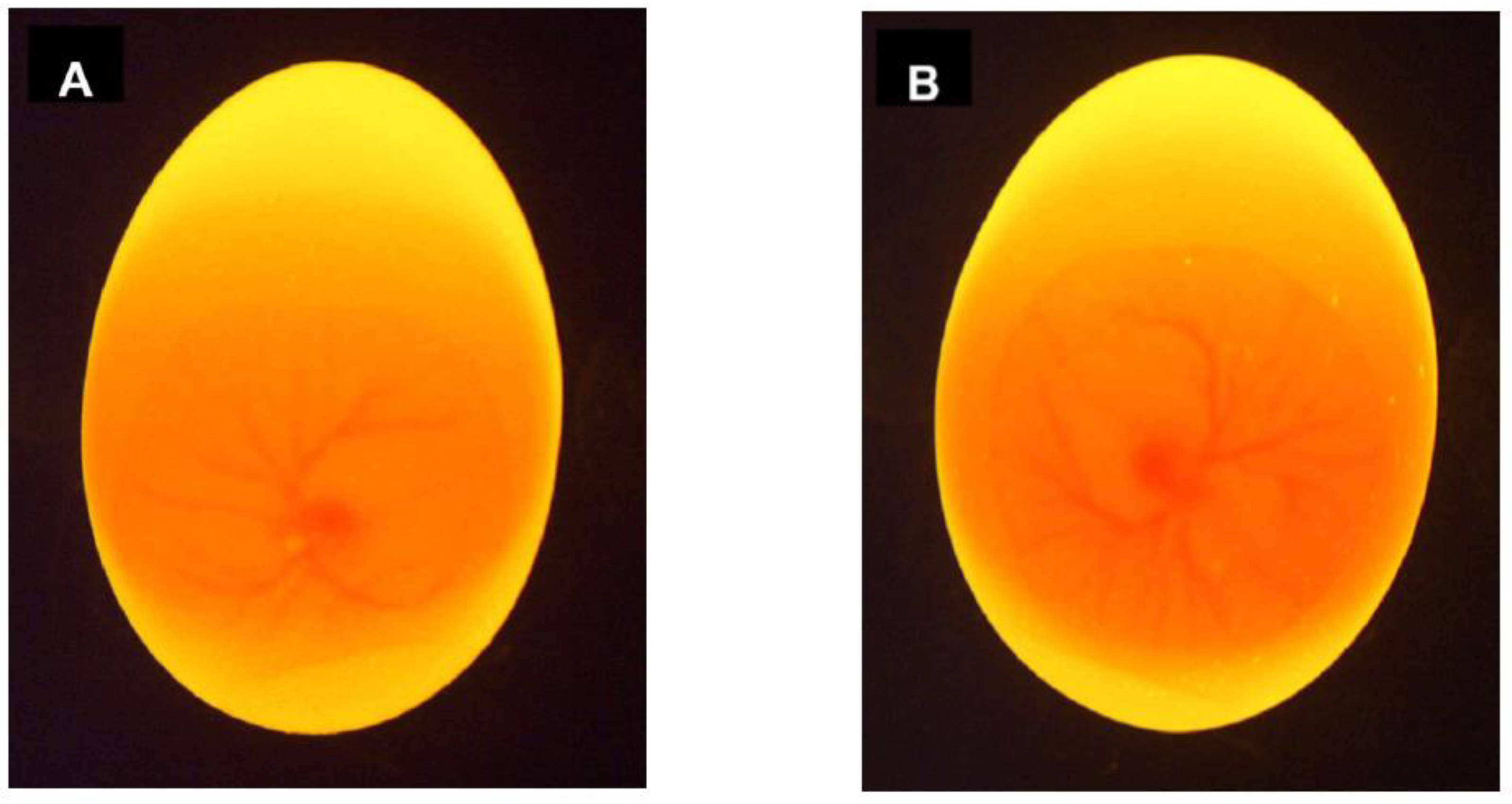
3.1. Raman and fluorescence spectroscopy
3.2. Infrared and Terahertz spectroscopy
3.3. Visible-Near-Infrared spectroscopy
3.4. Nuclear Magnetic Resonance spectroscopy
3.5. Morphometric studies
| Category | Target |
Incubation day Accuracy Throughput |
Technology (patents)/ research gaps (papers) |
Advantages (+)/ Drawbacks (-) |
References |
| Raman and fluorescence spectroscopy | Blood, eggshell, germinal disk, or other embryonic structures. | Day 3.5 91 % accuracy on blood Day 6 92.3 % on embryo and blood Low to high throughput depending on the invasiveness |
Technology ≅ Research The applicability on blood vessels is well reported in academic literature and is protected by patents Technology > Research A scientific gap exists in the analysis of the eggshell, germinal disk, or other embryonic structures since these are only reported in patent literature |
+ Early applicable in incubation No liquid or tissue sampling required - Intensive sampling procedure and relatively invasive process |
Patents ⊗ Baron et al. [71], ⊕ Steiner et al. [72], ⊕ Galli et al. [77], ⊕ Schortgen [78], ⊕ Opitz et al. [79], ⊕ Popp et al. [81], ⊕ Schart [133], ∅ Herzog and Hurlin [134], ∅ Steiner et al. [135], ⊕ Hurlin et al. [136] Papers Galli et al. [69,74,75], Gokdag et al. [137] |
| IR and THz spectroscopy | Germinal disk | Day 0 Accuracy not defined Average throughput |
Technology ≅ Research From the same group of authors, both patents and papers are reported for the IR approach Technology > Research Only a patent is reported for the non-invasive THz approach |
+ Applicable in an early stage - IR approach infringes hatchability |
Patents ∅ Steiner et al. [85], ∅ May et al. [86] Papers Steiner et al. [83,84] |
| VIS-NIR spectroscopy | Heartbeat, blood hemoglobin, movement, feather color, yolk ratio, or growth (opacity) |
Days 0 to 14 depending on the technology High accuracies on color sexing 99 % Blood sexing or other spectral features range from 80 to 90 % High throughput |
Technology ≅ Research Color sexing in the egg is commercially available and is described in both patent and paper literature Blood detection through the eggshell is described in paper and patent literature Technology > Research Other VIS-NIR spectroscopy on specific egg regions on specific days is only described in patents and lacks scientific background |
+ Has the potential for non-invasive implementation Equipment is affordable Early application potential - Lack of physiological background for sex determining spectral features described in patents |
Patents ⊕ McKay [91], ∅ Preusse [96], ⊕ Fujitani [95,101], ⊗ Rams and Toellner [100], ∅ Hebrank [97], ∅ Wang et al. [98], ∅ Colvin [102], ⊕ Ngadi et al. [104,106], ⊗ Zhao et al. [105,113], ⊕ Rozenboim and Ben Dor [107], ⊗ Pan et al. [108], ∅ Green [109], ⊕ Zhu [110], ∅ Fischer and Meissner [111,112] Papers Gohler et al. [90], Corion et al. [92], Khaliduzzaman et al. [93], Rahman et al. [94], Li et al. [103], Pan et al. [138] |
| NMR spectroscopy | Gonads, hormones, or metabolites | Days 4 to 11 for hormones and metabolites, accuracy not defined and relatively fast Day18 gonads, accuracy not defined and relatively slow |
Technology > Research Almost only patent literature has been reported A single research paper reports the difficulty of detecting gonads |
+ Non-invasive Early incubation - No prediction accuracies are reported or known Gonad sexing relatively late |
Patents ∅ Bruins and Stutterheim [63], ⊕ Reynells and Flegal [116], ∅ Haase et al. [118], ∅ Hergenroder et al. [119], ⊕ Sewiolo and Ziroff [120] Papers Davenel et al. [117] |
| Morphometric studies | Eggshell morphology or blood morphology | Day 0 for eggshell, ~93 to 100 % Relatively fast (using imaging technique) Day 4 for blood 89.74 % Relatively high throughput expected, yet equilibrium time needed in a horizontal position |
Technology > Research Numerous patents are filed and a few papers One paper is using the formulas from a patent Not all papers did find sexual differences Using the shape index, there was a trend for female eggs to be more spherical, although the overlap was big with male eggs Blood vessel morphology sexing is described both in patent and paper literature, and it might require more research to verify the robustness |
+ Non-invasive Before or early incubation - Not enough scientific evidence that proves this technique works robustly nor an explanation of why there are sexual differences in the egg shape Blood vessel imaging requires a 2 minutes equilibration time in a horizontal position |
Patents Taniguchi ⊕ [121], ∅ [122], ⊕ [123], ∅ [124], ∅ [125], ⊕ [126], ⊕ Xiaohui et al. [129], ⊗ Li et al. [130], ⊗ Li [131], ⊕ Chen et al. [132], ∅ [139] Papers Yilmaz-Dikmen et al. [127], Rutkowska et al. [140] |
| Abbreviations: d, day; IR, infrared; THz, terahertz; VIS-NIR, visible-near-infrared; ⊕, granted; ∅, pending; ⊗, ceased/rejected/discontinued. | |||||
4. Trends in in ovo sexing techniques
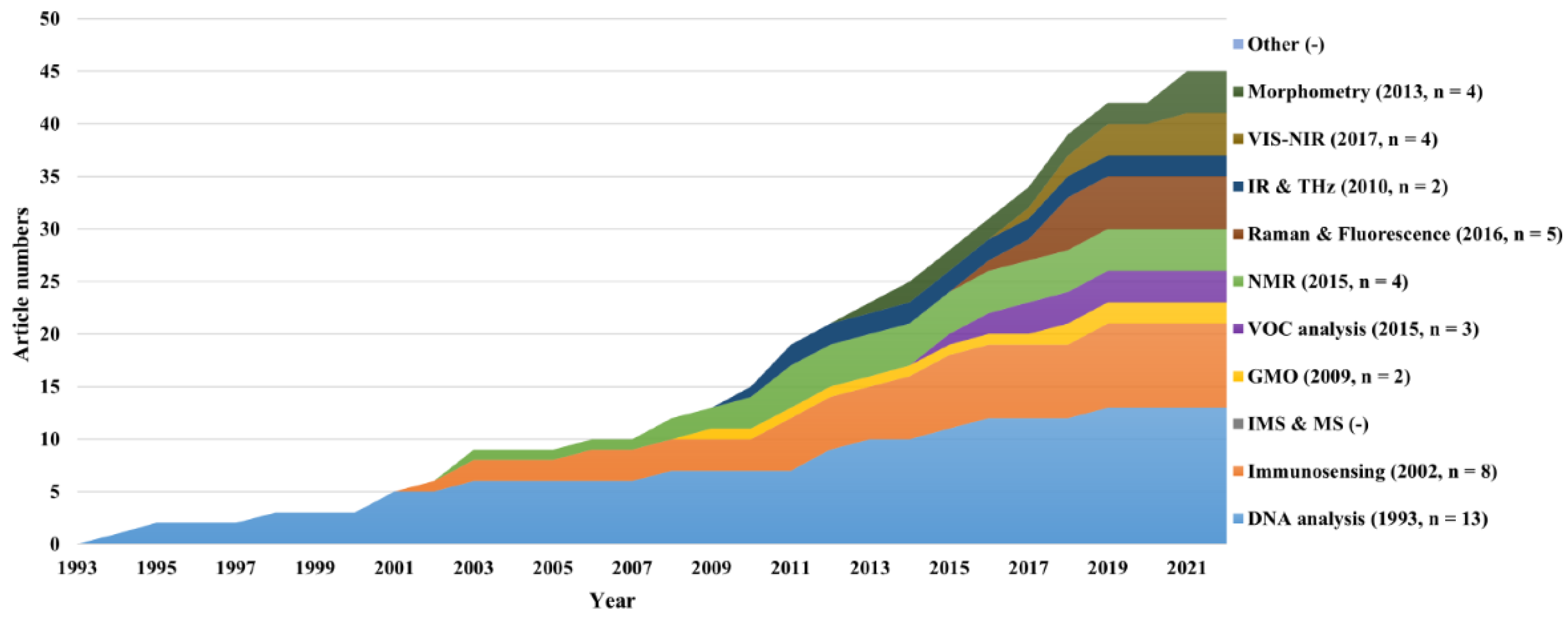
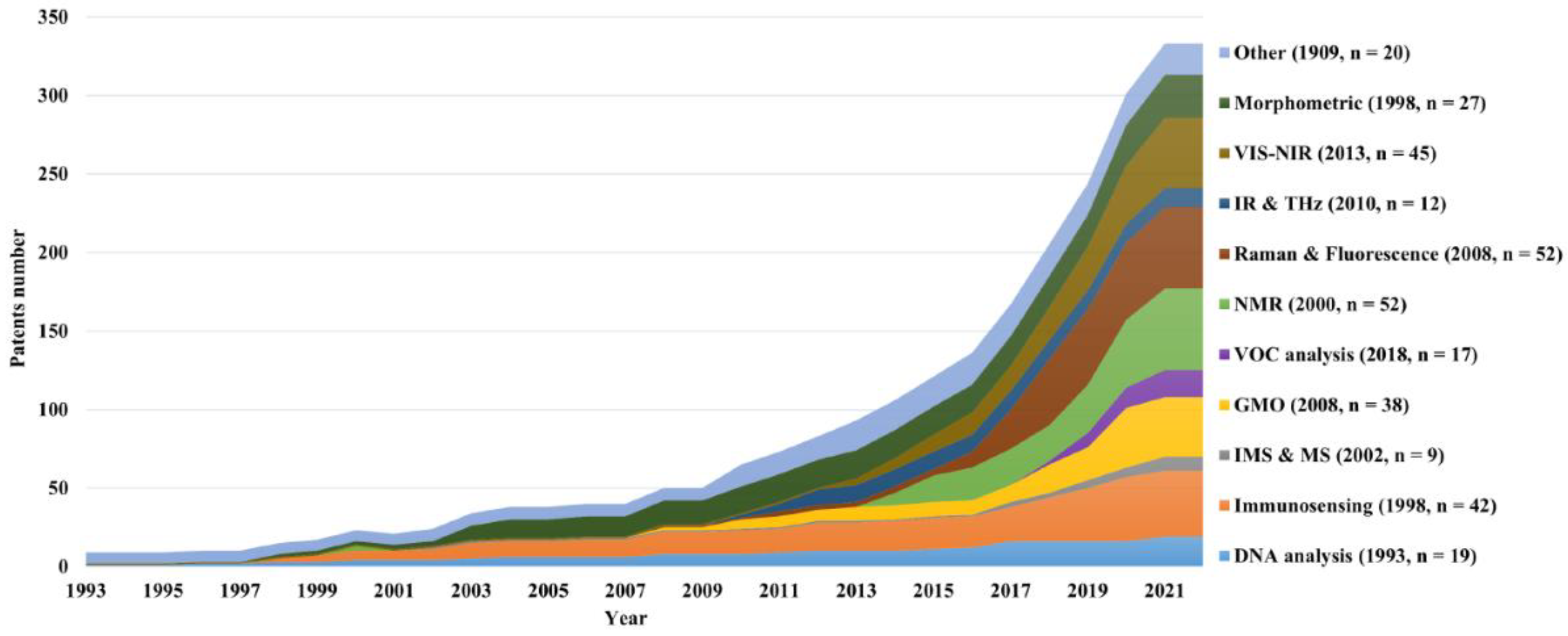
4.1. Publication trends of papers and patents over time
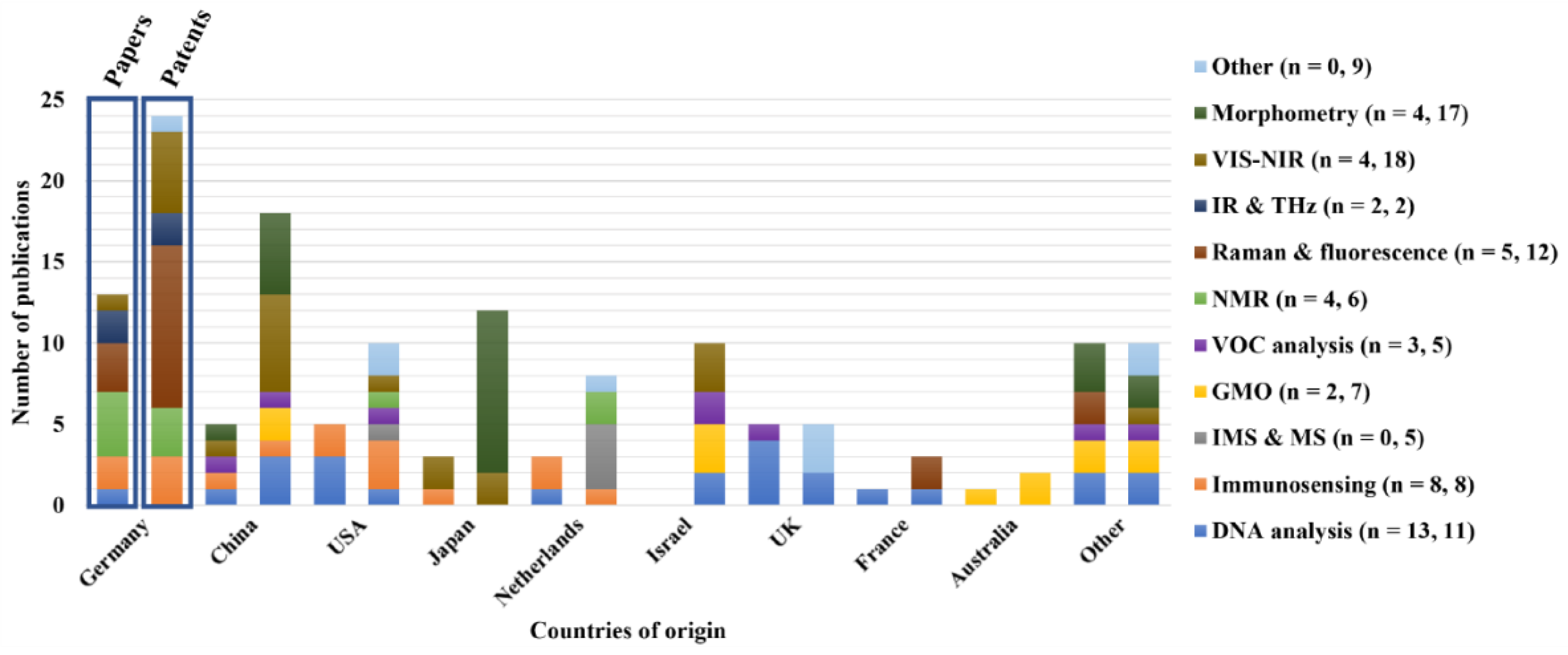
4.2. Geographical distribution of techniques
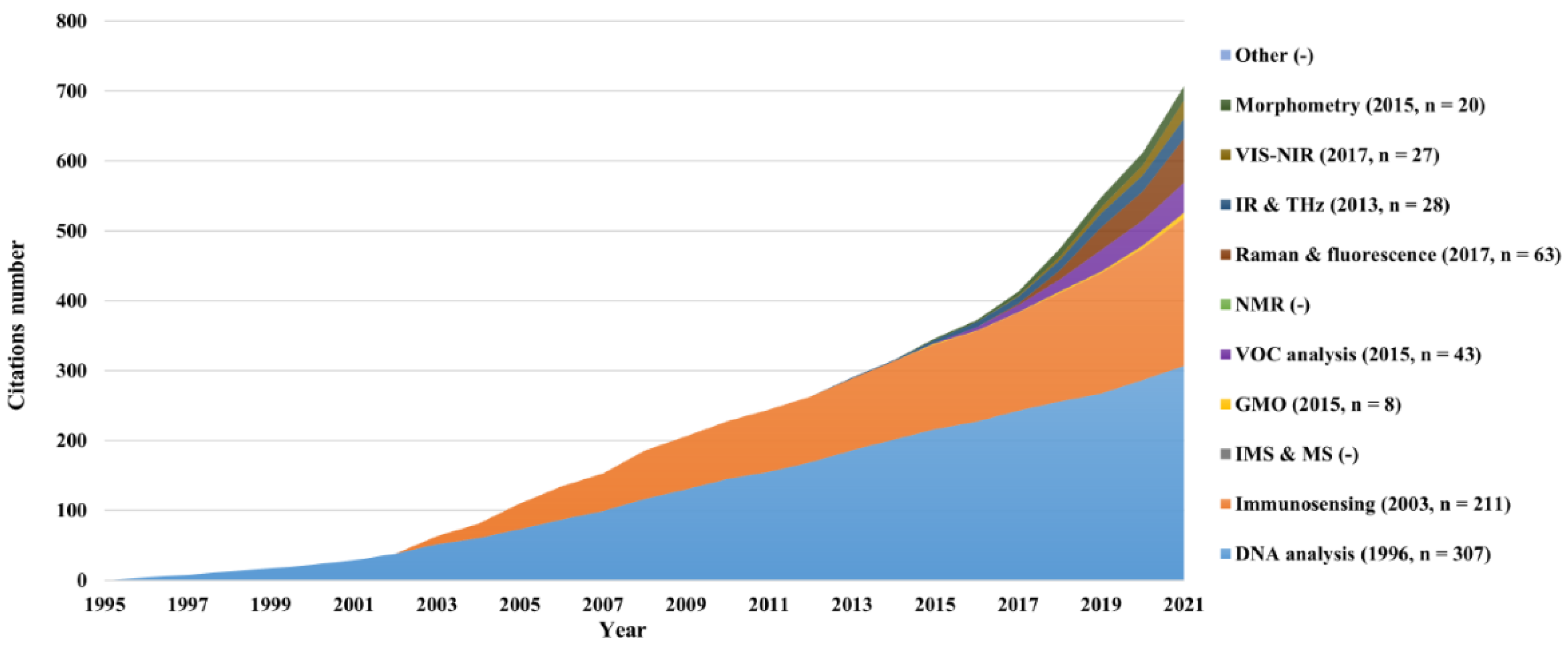
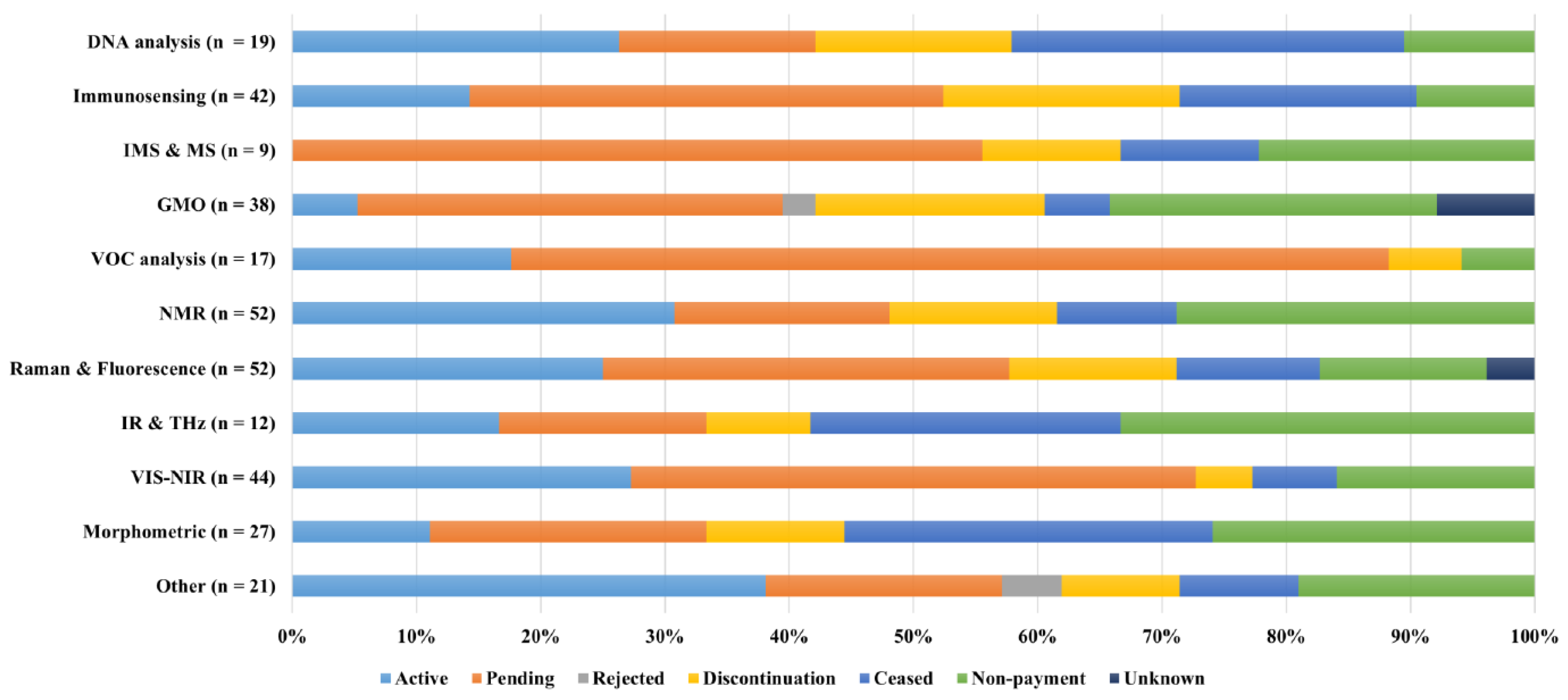
4.3. Papers citations and legal status patents
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Authors’ information (optional)
Abbreviations
| IMS | ion mobility spectrometry |
| MS | mass spectrometry |
| GMO | genetic modified organisms |
| VOC | volatile organic compounds |
| PCR | polymerase chain reaction |
| DMRT-1 | doublesex and mab-3 related transcription factor 1 |
| CHD | chromodomain helicase DNA |
| WPKCI | W-protein kinase C iota |
| gDNA | genomic DNA |
| AF | allantoic fluid |
| FRET | fluorescence resonance energy transfer |
| qPCR | quantitative PCR |
| ELISA | enzyme-linked immunosorbent assay |
| RIA | radioimmunoassay |
| DHT | dihydrotestosterone |
| A4 | androstenedione |
| E1 | estrone |
| E2 | estradiol |
| E1S | estrone sulfate |
| P4 | progesterone |
| T | testosterone |
| PGCs | primordial germ cells |
| GC-MS | gas chromatography-mass spectrometer |
| PTR-MS | proton transfer reaction-mass spectrometry |
| SIFT-MS | selected ion flow tube-mass spectrometry |
| THz | Terahertz |
| SPME-GS-MS | solid phase micro extraction-gas chromatography-mass spectrometry |
| VIS-NIR | visible-near-infrared |
| NMR | nuclear magnetic resonance |
| MRI | magnetic resonance imaging |
| IR | infrared |
References
- Bruijnis MRN, Blok V, Stassen EN, Gremmen HGJ. Moral “lock-in” in responsible innovation: The ethical and social aspects of killing day-old chicks and its alternatives. J Agric Environ Ethics. 2015;28(5):939–60. [CrossRef]
- Reithmayer C, Mußhoff O, Danne M. Alternatives to culling male chicks – the consumer perspective. Br Food J. 2020;122(3):753–65. [CrossRef]
- Gautron J, Réhault-Godbert S, Van de Braak TGH, Dunn IC. Review: What are the challenges facing the table egg industry in the next decades and what can be done to address them? Animal [Internet]. 2021;(xxxx):100282. Available from. [CrossRef]
- Reithmayer C, Mußhoff O. Consumer preferences for alternatives to chick culling in Germany. Poult Sci. 2019;98(10):4539–48. [CrossRef]
- Der Bundesregierung. Entwurf eines gesetzes zur änderung des tierschutzgesetzes – verbot des kükentötens. Der Bundesregierung; 2019 p. 176.
- Française, R. Journal Officiel - Lois et Décrets [Internet]. Paris; 2022 p. 56–7. Available from: https://www.legifrance.gouv.fr/jorf/jo/2022/02/06/0031.
- Consiglio DEL, Sono E. REGOLAMENTO (CE) N. 1099/2009 DEL CONSIGLIO del 24 settembre 2009 relativo alla protezione degli animali durante l’abbattimento [Internet]. Brussels: Unione europea; 2009 p. 1–30. Available from: https://eur-lex.europa.eu/legal-content/IT/TXT/?uri=celex%3A32009R1099.
- Busse M, Kernecker ML, Zscheischler J, Zoll F, Siebert R. Ethical Concerns in Poultry Production: A German Consumer Survey About Dual Purpose Chickens. J Agric Environ Ethics [Internet]. 2019;32(5–6):905–25. Available from. [CrossRef]
- Rosenbruch, M. The sensitivity of chicken embryos in incubated eggs. ALTEX. 1997;14(3):111–3.
- Mellor DJ, Diesch TJ. Birth and hatching: Key events in the onset of awareness in the lamb and chick. N Z Vet J [Internet]. 2007;55(2):51–60. Available from. [CrossRef]
- Phelps P, Bhutada A, Bryan S, Chalker A, Ferrell B, Neuman S, et al. Automated identification of male layer chicks prior to hatch. Worlds Poult Sci J. 2003;59(1):33–8.
- Krautwald-Junghanns ME, Cramer K, Fischer B, Förster A, Galli R, Kremer F, et al. Current approaches to avoid the culling of day-old male chicks in the layer industry, with special reference to spectroscopic methods. Poult Sci. 2018;97(3):749–57. [CrossRef]
- Sinclair A, Smith C. Sex-determination and methods of specifying same [Internet]. 2010. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2010088742A1.
- Griffiths, R. Avian CHD genes and their use in methods for sex identification in birds [Internet]. Vol. 1. WO9639505A1, 1996. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO9639505A1.
- Li H, Hu Y, Song C, Ji G, Liu H, Xu W, et al. A new primer for sex identification of ducks and a minimally invasive technique for sampling of allantoic fluid to detect sex during bird embryo development. Sex Dev. 2015;9(3):173–81. [CrossRef]
- Petitte JN, Kegelmever AE. Rapid sex determination of chick embryos using the polymerase chain reaction1. Anim Biotechnol. 1995;6(2):119–30.
- Haunshi S, Pattanayak A, Bandyopadhaya S, Saxena SC, Bujarbaruah KM. A simple and quick DNA extraction procedure for rapid diagnosis of sex of chicken and chicken embryos. J Poult Sci. 2008;45:75–81. [CrossRef]
- Clinton M, Nandi S, Zhao D, Olson S, Peterson P, Burdon T, et al. Real-Time Sexing of Chicken Embryos and Compatibility with in ovo Protocols. Sex Dev. 2016;10(4):210–6. [CrossRef]
- Clinton, M. Avian sex determination method [Internet]. 2004. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2004016812A1.
- He L, Martins P, Huguenin J, Van TNN, Manso T, Galindo T, et al. Simple, sensitive and robust chicken specific sexing assays, compliant with large scale analysis. PLoS One. 2019;14(3):1–17. [CrossRef]
- Smith CA, Roeszler KN, Hudson QJ, Sinclair AH. Avian sex determination: What, when and where? Cytogenet Genome Res. 2007;117(1–4):165–73.
- Smith CA, Sinclair AH. Sex determination: Insights from the chicken. BioEssays. 2004;26(2):120–32. [CrossRef]
- Tanabe Y, Nakamura T, Fujioka K, Doi O. Production and secretion of sex steroid hormones by the testes, the ovary, and the adrenal glands of embryonic and young chickens (Gallus domesticus). Gen Comp Endocrinol. 1979;39(1):26–33. [CrossRef]
- Gill D, V. , Robertson HA, Betz TW. In vivo estrogen synthesis by the developing chicken (Gallus gallus) embryo. Gen Comp Endocrinol. 1983;49(2):176–86.
- Phelps, P. Method of sorting birds in ovo [Internet]. 1998. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO9814781A1.
- Tyczkowski J, Mahato D, Chalker A. Competitive particle immunoassay methods utilizing fluorescence microscopy [Internet]. 2006. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2006124456A3.
- Butt T, Tran HT. Compositions and methods for gender sorting [Internet]. 2002. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCA2445162A1.
- Tran HT, Ferrell W, Butt TR. An estrogen sensor for poultry sex sorting. J Anim Sci. 2010;88(4):1358–64. [CrossRef]
- Einspanier, A. Method for the in-ovo sex identification of chicks [Internet]. 2017. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2017109133A1.
- Weissmann A, Reitemeier S, Hahn A, Gottschalk J, Einspanier A. Sexing domestic chicken before hatch: A new method for in ovo gender identification. Theriogenology [Internet]. 2013;80(3):199–205. Available from. [CrossRef]
- Müller W, Eising CM, Dijkstra C, Groothuis TGG. Sex differences in yolk hormones depend on maternal social status in leghorn chickens (Gallus gallus domesticus). Proc R Soc B Biol Sci. 2002;269(1506):2249–55. [CrossRef]
- Aslam MA, Hulst M, Hoving-Bolink RAH, Smits MA, de Vries B, Weites I, et al. Yolk concentrations of hormones and glucose and egg weight and egg dimensions in unincubated chicken eggs, in relation to egg sex and hen body weight. Gen Comp Endocrinol [Internet]. 2013;187:15–22. Available from. [CrossRef]
- Wang Y, Jin G, Ma M, Xiang X. Sex differences in serum steroid hormone levels during embryonic development in hen eggs. Poult Sci. 2019;98(11):6053–62. [CrossRef]
- Decuypere E, Fey F. Method for avian sex determination. 2011;1(19):33. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2014099265A1.
- Güneş E, Movassaghi H, Unsal F, Güneş NT. GMO Policies and Practices: A Global Overview with Special Focus on Turkey. In: Policy Issues in Genetically Modified Crops: A Global Perspective. 2020. p. 29–56.
- Arce L, Gallegos J, Garrido-Delgado R, Medina LM, Sielemann S, Wortelmann T. Ion mobility spectrometry a versatile analytical tool for metabolomics applications in food science. Curr Metabolomics. 2014;2(4):264–71. [CrossRef]
- Daum KA, Atkinson DA. Gender determination of avian embryo [Internet]. US63365339B1, 2002. Available from: https://worldwide.espacenet.com/patent/search/family/026795695/publication/US6365339B1?q=pn%3DUS6365339B1.
- Bruins W, Stutterheim W. Method and system for the non-destructive in ovo determination of fowl gender [Internet]. WO2017204636A2, 2017. Available from: https://worldwide.espacenet.com/patent/search/family/059366469/publication/WO2017204636A2?q=pn%3DWO2017204636A2&queryLang=en%3Ade%3Afr.
- Bruetschy, C. The EU regulatory framework on genetically modified organisms (GMOs). Transgenic Res [Internet]. 2019;28(s2):169–74. Available from. [CrossRef]
- Doran TJ, Morris KR, Wise TG, O’Neil TE, Cooper CA, Jenkins KA, et al. Sex selection in layer chickens. Anim Prod Sci. 2018;58(3):476–80. [CrossRef]
- Offen, D. Methods for gender determination of avian embryos in unhatched eggs and means thereof [Internet]. 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2017094015A1.
- Beisswanger, R. Method for suppressing the male sex in birds, specifically in laying poultry, by inserting a lethal DNA sequence into sex chromosomes that is active only in male embryos Roland 2002.pdf [Internet]. 2002. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DDE10248361A1.
- Webster B, Hayes W, Pike TW. Avian egg odour encodes information on embryo sex, fertility and development. PLoS One. 2015;10(1). [CrossRef]
- Costanzo A, Panseri S, Giorgi A, Romano A, Caprioli M, Saino N. The odour of sex: Sex-related differences in volatile compound composition among barn swallow eggs carrying embryos of either sex. PLoS One. 2016;11(11):1–17.
- Rivers, AR. System and method for determining the sex and viability of poultry eggs prior to hatching [Internet]. US20210181174A1, 2021. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2021181174A1.
- Knepper P, O’Hayer M, Hoopes J. System and method for in ovo sexing of avian embryos [Internet]. WO2018023105A1, 2018. p. 33. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2018023105A1.
- Tongli S, Lin X, Xiaohai Y. The invention discloses a terahertz poultry embryo sex detection device [Internet]. CN208891459U, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN208891459U.
- Gabbai, E. A system and method for non-invasively determining egg properties [Internet]. WO2019021275A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2019021275A1.
- Xiang X, Hu G, Jin Y, Jin G, Ma M. Nondestructive characterization gender of chicken eggs by odor using SPME/GC-MS coupled with chemometrics. Poult Sci. 2022;101(3):101619. [CrossRef]
- Corion M, Ketelaere B De, Hertog M, Lammertyn J. Profiling the emission of volatile organic compounds from chicken hatching eggs in the first half of incubation. In: IFRG eMeeting 2021. [Online]: IFRG; 2021.
- Young, RD. Electrical and magnetic polarity and sex indicator [Internet]. GB176672A, 1922. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DGB176672A.
- Williams, Henry A. Improvements in and connected with appliances adapted to inidcate the sex of any living thing and for other analogous purposes [Internet]. 1920. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DGB150655A.
- Williams, HA. A new apparatus to indicate the sex of birds or animals and to tell whether an egg is fertile and what sex it will hatch [Internet]. GB190900909A, 1909. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DGB190900909A.
- Pacala N, Bencsik I, Bencsik I, Corin Ni, Stanculet JJ, Anna BA. Apparatus and method for determining the genetic sex [Internet]. 2007. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DRO121316B1.
- Chadfield, C. Improvements in or relating to methods of and means for determining the presence of living organisms and or the sex of living organisms [Internet]. GB462237A, 1935. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DGB462237A.
- Pacala N, Bencsik I, Bencsik I, Corin Ni, Stanculet JJ, Anna BA. Apparatus and method for determining the genetic sex [Internet]. RO121316B1, 2007. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DRO121316B1.
- Visser CFT. Method for preparing an invasive test of an egg and for determining a gender of an embryo in an egg [Internet]. US2012231487A1, 2012. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DEP2473030B1.
- Rastoutsau U, Lukyanau A, Vinogradov S, Kosolobov A, Galitsky N, Gromov R. A method of identifying the state of eggs in the process of incubation and a system for implementing the same [Internet]. WO2018042225A1, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2018042225A1.
- Dalton M, Daly J, Mccabe M, Walsh P. Genomic analysis device and methods of use [Internet]. 2016. Available from: https://worldwide.espacenet.com/patent/search/family/053887156/publication/WO2016020838A1?q=pn%3DWO2016020838A1.
- Arie A, Danielli A. Method and system for detecting a target within a population of molecules [Internet]. US20190137504A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2019137504A1.
- Molina F, Espeut J. Method for determining a specific characteristic of an embryo in an unhatched egg [Internet]. Vol. 1. EP3599465A1, 2020. p. 1–34. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DEP3599465A1.
- Weissmann A, Förster A, Gottschalk J, Reitemeier S, Krautwald-Junghanns ME, Preisinger R, et al. In ovo-gender identification in laying hen hybrids: effects on hatching and production performance. Eur Poult Sci. 2014;78. [CrossRef]
- Bruins W, Stutterheim W. Gender, viability and/or developmental stage determination of avian embryos in ovo [Internet]. WO2014021715A3, 2014. Available from: https://worldwide.espacenet.com/patent/search/family/050028615/publication/WO2014021715A2?q=WO 2014%2F021715.
- Stutterheim WM, Van Bommel L, Bruins WS, Dijksterhuis E, Van De Wel J, Leemker M. Egg characteristic determining device [Internet]. WO2021145771A1, 2021. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DNL2016818B1.
- Knepper P, O’Hayer M, Hoopes J. System and method for in ovo sexing of avian embryos [Internet]. WO2018023105A1, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2018023105A1.
- Shi T, Xu L, Yu X. The invention discloses a terahertz poultry embryo sex detection device [Internet]. CN208891459U, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN208891459U.
- Kingslake R, Thompson BJ. Optics. In: Encyclopedia Britannica [Internet]. 2020. Available from: https://www.britannica.
- Atkins P, de Paula J, Keeler J. Physical chemistry. 11th ed. Biophysical Chemistry. Oxford University Press; 2018.
- Galli R, Preusse G, Schnabel C, Bartels T, Cramer K, Krautwald-Junghanns ME, et al. Sexing of chicken eggs by fluorescence and Raman spectroscopy through the shell membrane. PLoS One [Internet]. 2018;13(2). Available from. [CrossRef]
- Harz M, Krause M, Bartels T, Cramer K, Rösch P, Popp J. Minimal invasive gender determination of birds by means of UV-resonance Raman spectroscopy. Anal Chem. 2008;80(4):1080–6. [CrossRef]
- Baron S, Catherinot L, Sarr Y, Schortgen M, Thouand G, Assaf AA. Procede non invasif de determination de la fertilite et ou du sexe d un oeuf [Internet]. FR3075965A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DFR3075965A1.
- Steiner G, Preusse G, Galli R, Koch E. Method and device for optical in ovo sex determination of fertilized and incubated birds’ eggs [Internet]. US20190383782A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2019383782A1.
- Galli R, Koch E, Preusse G, Schnabel C, Bartels T, Krautwald-Junghanns M-E, et al. Contactless in ovo sex determination of chicken eggs. Curr Dir Biomed Eng [Internet]. 2017;3(2):131–4. Available from: http://www.degruyter.com/view/j/cdbme.2017.3.issue-2/cdbme-2017-0027/cdbme-2017-0027. [CrossRef]
- Galli R, Preusse G, Uckermann O, Bartels T, Krautwald-Junghanns ME, Koch E, et al. In ovo sexing of chicken eggs by fluorescence spectroscopy. Anal Bioanal Chem [Internet]. 2017;409(5):1185–94. Available from. [CrossRef]
- Galli R, Preusse G, Uckermann O, Bartels T, Krautwald-Junghanns M-E, Koch E, et al. In ovo sexing of domestic chicken eggs by raman spectroscopy. Anal Chem. 2016;88:8657–63. [CrossRef]
- Steiner G, Preusse G, Koch E, Galli R, Schnabel C, Preusse J. Method for classifying spectra of objects having complex information content [Internet]. CA3047337A1, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCA3047337A1.
- Galli R, Preusse G, Koch E, Steiner G, Krautwald-Junghanns M-EE, Bartels T. Method and device for the Raman spectroscopic, in ovo sex determination of fertilised and incubated birds’ eggs [Internet]. AU2015283366B2, 2016. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DAU2015283366B2.
- Schortgen, M. Non-invasive device for determining the fertility and/or the sex of an egg, and corresponding method [Internet]. US20170160202A1, 2017. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS10060854B2.
- Opitz J, Fischer B, Morgenstern P, Schreiber J, Gerich C. Methods for determining the sex of bird’s eggs [Internet]. US008364247B2, 2013. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS8364247B2.
- Dörksen H, Krahl J, Staufenbiel J. Device and method for in-ovo determination of the sex of a fertilised bird egg [Internet]. WO2021144420A1, 2021. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2021144420A1.
- Popp J, Roesch P, Bartels T, Krause M, Harz M. Bird sex determination involves testing DNA relevant cell material of bird with light, and measuring molecule oscillations, where spectrum of molecule oscillations resulting from light is detected, and is compared [Internet]. DE102007013107A1, 2008. p. 1–5. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DDE102007013107A1.
- Tiersch, TR. Identification of sex in chickens by flow cytometry. Worlds Poult Sci J. 2003;59(1):25–32.
- Steiner G, Bartels T, Stelling A, Krautwald-Junghanns M-E, Fuhrmann H, Sablinskas V, et al. Gender determination of fertilized unincubated chicken eggs by infrared spectroscopic imaging. Anal Bioanal Chem [Internet]. 2011 Jul 9 [cited 2017 Oct 17];400(9):2775–82. Available from: http://link.springer.com/10.1007/s00216-011-4941-3. [CrossRef]
- Steiner G, Bartels T, Krautwald-Junghanns M-E, Koch E. Bird sexing by Fourier transform infrared spectroscopy. In: Mahadevan-Jansen A, Petrich W, editors. Biomedical Vibrational Spectroscopy IV: Advances in Research and Industry [Internet]. 2010. p. 75600D. Available from: https://proceedings.spiedigitallibrary.org/proceeding.aspx?doi=10.1117/12.841627.
- Steiner G, Koch E, Krautwald-Junghanns M-E, Bartels T. Method and device for determining the sex of fertilized, non-incubated bird eggs [Internet]. US8624190B2, 2014. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS8624190B2.
- May T, Weber K, Popp J. Method and arrangement for non-invasive non-destructive identification of molecule-specific and or biological properties of an internal structure of a biological examination item through an optically impenetrable barrier [Internet]. WO2014086335A1, 2014. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2014086335A1.
- Zhang W, Brown ER, Rahman M, Norton ML. Observation of terahertz absorption signatures in microliter DNA solutions. Appl Phys Lett. 2013;102(2):2–6. [CrossRef]
- De Ketelaere B, Bamelis F, Kemps B, Decuypere E, De Baerdemaeker J. Non-destructive measurements of the egg quality. Worlds Poult Sci J. 2004;60(3):289–302. [CrossRef]
- Amigo JM, Babamoradi H, Elcoroaristizabal S. Hyperspectral image analysis. A tutorial. Anal Chim Acta. 2015;896:34–51.
- Göhler D, Fischer B, Meissner S. In-ovo sexing of 14-day-old chicken embryos by pattern analysis in hyperspectral images (VIS/NIR spectra): A non-destructive method for layer lines with gender-specific down feather color. Poult Sci [Internet]. 2016;96(1):1–4. Available from: https://academic.oup.com/ps/article-lookup/doi/10.3382/ps/pew282. [CrossRef]
- McKay, JC. Spectrophotometric analysis of embryonic chick feather color [Internet]. US20140069336A1, 2014. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2014069336A1.
- Corion M, Keresztes J, De Ketelaere B, Saeys W. In ovo sexing of eggs from brown breeds with a gender-specific color using visible-near-infrared spectroscopy: effect of incubation day and measurement configuration. Poult Sci [Internet]. 2022;101(5):101782. Available from. [CrossRef]
- Khaliduzzaman A, Shinichi F, Ayuko K, Tetsuhito S, Yuichi O, Naoshi K. Non-invasive broiler chick embryo sexing based on opacity value of incubated eggs. Comput Electron Agric [Internet]. 2019;158(March):30–5. Available from. [CrossRef]
- Rahman A, Syduzzaman M, Khaliduzzaman A, Fujitani S, Kashimori A, Suzuki T, et al. Nondestructive sex-specific monitoring of early embryonic development rate in white layer chicken eggs using visible light transmission. Br Poult Sci [Internet]. 2020;61:209–16. Available from. [CrossRef]
- Fujitani, S. Nondestructive inspection device of hatching egg, and hatching egg inspection program used for the same [Internet]. JP2017227471A, 2017. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DJP2017227471A.
- Preusse G, Steiner G, Galli R, Koch E, Schnabel C. Method for in-ovo fertilisation determination and gender determination on a closed egg [Internet]. 2020. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2020400640A1.
- Hebrank, JH. Methods and apparatus for detecting the presence of eggs in an eggs in an egg flat [Internet]. WO2006078499A2, 2006. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2006078499A2.
- Wang M, Luo J, Wu L, Han D, Zeng Y. Device and method for gender identification of egg embryo based on heart rate measurement [Internet]. CN109431484A, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN109431484A.
- Wu L, Luo J, Wang M, Han D, Zeng Y. Egg embryo sex determination device based on heart rate measurement [Internet]. CN209611122U, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN209611122U.
- Rams P, Toellner A. Improvements in and relating to the determination of the sex of eggs of fowls [Internet]. GB457543A, 1935. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DGB457543A.
- Fujitani, S. Sexual orientation selection device of hatching egg before incubation [Internet]. JP2019017336A, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DJP2019017336A.
- Colvin, A. Devices and methods for determining analytes [Internet]. WO2018154389A1, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2018154389A1.
- Li Q-X, Wang QH, Ma M, Xiao S-J, Shi H. Non-destructive detection of male and female information of early duck embryos based on visible/near infrared spectroscopy and deep learning. Spectrosc Spectr Anal. 2021;41(6):2021.
- Ngadi M, Liu L, Zheng C. Systems, devices, and methods for detecting fertility and gender of unhatched eggs [Internet]. WO2016131124A1, 2016. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2016131124A1.
- Zhao X, Song Z, Guan Y, Tan F, Shang T, Di G, et al. Gender identification method for chick embryo in near-infrared hatching egg at earlier stage incubation [Internet]. CN103472008A, 2013. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN103472008A.
- Ngadi M, Liu L, Zheng C. Systems, devices, and methods for detecting fertility and gender of unhatched eggs [Internet]. US20200302604A1, 2020. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2020302604A1.
- Rozenboim I, Ben Dor E. Hyperspectral Identification of Egg Fertility and Gender [Internet]. US20130044210A1, 2013. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2013044210A1.
- Pan L, Hu P, Tu K, Zhang W, Sun Y, Li Z. Gender determination method for chicken hatching egg incubation early embryo based on hyperspectral image [Internet]. CN104316473A, 2015. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN104316473A.
- Green, Y. Method and system for spectral determination of egg gender and fertility [Internet]. WO2018235070A1, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2018235070A1.
- Zhu Z, Hong Q, Wu L, Wang Q, Ma M. Early-stage chick embryo gender identification method [Internet]. CN109142248A, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN109142248A.
- Fischer B, Meissner S. Device for examining hatching eggs [Internet]. WO2019174661A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2019174661A1.
- Fischer B, Meissner S. Apparatus for inspecting bird eggs [Internet]. JP2021099334A, 2021. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DJP2021099334A.
- Zhao X, Cai L, Song Z, Guan Y, Tan F, Tian F, et al. Near-infrared gender recognition device for chicken embryo in hatching egg during early stage of incubation [Internet]. CN103461218A, 2013. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN103461218A.
- Corion M, Keresztes J, De Ketelaere B, Saeys W. Innovative, non-invasive, fast and highly accurate visible- near-infrared in ovo sexing method on brown chicken eggs. In: 26th World’s Poultry Congress. [Online]: World’s Poultry Congress; 2020.
- Nageswara Rao, BD. Nuclear magnetic resonance: An early history. Resonance. 2015;20(11):969–85.
- Reynells RD, Flegal CJ. Method and apparatus for avian pre-hatch sex determination [Internet]. US6029080A, 2000. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS6029080A.
- Davenel A, Eliat PA, Quellec S, Nys Y. Attempts for early gender determination of chick embryos in ovo using Magnetic Resonance Imaging. XXII Eur Symp Qual Poult Meat XVI Eur Symp Qual Eggs Egg Prod. 2015;2–5.
- Haase A, Schusser BM, Molina-Romero M, Goméz PA, Aigner M, Huber S, et al. Automated noninvasive determining the sex of an embryo and the fertility of a brid’s egg [Internet]. WO2019092265A1, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2019092265A1.
- Hergenröder R, Lambert J, Telfah A. Method for determining the sex of bird eggs in ovo [Internet]. WO2020126253A1, 2020. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DWO2020126253A1.
- Sewiolo B, Ziroff A. Method for determining the sex of an embryo in an egg [Internet]. US20160057977A1, 2016. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2016057977A1.
- Taniguchi, R. Method for sexing fowl egg and apparatus therefor [Internet]. JP2001238559A, 2001. Available from: https://worldwide.espacenet.com/patent/search/family/026582006/publication/JP2001238559A?q=pn%3DJP2001238559A.
- Taniguchi, R. Method and apparatus for determining the sex of a fertilized egg [Internet]. US20040040515A1, 2004. Available from: https://worldwide.espacenet.com/patent/search/family/031977095/publication/US2004040515A1?q=pn%3DUS2004040515A1.
- Taniguchi, R. Method and apparatus for determining the sex of a fertilized egg [Internet]. US007167579B2, 2007. Available from: https://worldwide.espacenet.com/patent/search/family/027800473/publication/US7167579B2?q=US7167579B2.
- Taniguchi, R. Steric strain measuring method of egg [Internet]. JP2010038897A, 2010. Available from: https://worldwide.espacenet.com/patent/search/family/042011587/publication/JP2010038897A?q=pn%3DJP2010038897A.
- Taniguchi, R. Method for identifying sex of fertilized egg based on outline thereof [Internet]. JP2011142866A, 2011. p. 1–4. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DJP2011142866A.
- Taniguchi, R. Sex identification device for fertilized eggs, sex identification method for fertilized eggs, and program [Internet]. EP3878274A1, 2021. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DEP3878274A1.
- Yilmaz-Dikmen B, Dikmen S. A morphometric method of sexing white layer eggs. Rev Bras Ciência Avícola [Internet]. 2013;15(3):203–10. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-635X2013000300006&lng=en&nrm=iso&tlng=en.
- Zhu ZH, Ye ZF, Tang Y. Nondestructive identification for gender of chicken eggs based on GA-BPNN with double hidden layers. J Appl Poult Res. 2021;30(4). [CrossRef]
- Xiaohui D, Ziqiaojing Y, Zhengting L. Method forlossless determination of genders of poultry embryo eggs [Internet]. CN107041325A, 2017. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN107041325B.
- Li Z, Li R, Wang T. Method and monitoring device for early discriminating embryonic male and female of birds [Internet]. CN1189616A, 1998.Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN1189616A.
- Li, Z. Method and apparatus for judging sex of domestic chicken in embryo egg early incubation period [Internet]. CN1742569A, 2006. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN1742569A.
- Chen L, Zhang C, Xue X, Fan S, Huang W, Li J, et al. Sex detection system and method for chicken embryonated eggs [Internet]. CN110771533A, 2020. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DCN110771533A.
- Schart, C. Method and device for adjusting the laser focus of an excitation laser for vessels through which blood flows for optical measurements - Schart 2017.pdf [Internet]. EP3244197A1, 2017. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DEP3244197A1.
- Herzog S, Hurlin J. Method and system for determining the sex in-ovo or determining the sex of living birds [Internet]. WO2018115280A1, 2018. Available from: https://worldwide.espacenet.com/patent/search? 2018.
- Steiner G, Preusse G, Koch E, Galli R, Schnabel C. Producing a hole in a bird egg for determining the sex of the bird egg [Internet]. 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS2019339244A1.
- Hurlin J, Meissner S, Fischer B. Method and apparatus for creating an opening in the calcified shell [Internet]. US10458967B2, 2019. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DUS10458967B2.
- Gokdag U, Cinar E. Application of genetic algorithm for egg gender determination. In 26th IEEE Signal Processing and Communications Applications Conference (SIU); 2018 [cited 2022 May 3]. Available from: https://www.webofscience.com/wos/woscc/full-record/WOS:000511448500190.
- Pan L, Zhang W, Yu M, Sun Y, Gu X, Ma L, et al. Gender determination of early chicken hatching eggs embryos by hyperspectral imaging. Nongye Gongcheng Xuebao/Transactions Chinese Soc Agric Eng. 2016;32(1):181–6.
- Taniguchi, R. Sex determination method of sperm egg and device thereof [Internet]. JP2018186779A, 2018. Available from: https://worldwide.espacenet.com/patent/search?q=pn%3DJP2018186779A.
- Rutkowska J, Dubiec A, Nakagawa S. All eggs are made equal: Meta-analysis of egg sexual size dimorphism in birds. J Evol Biol. 2014;27(1):153–60. [CrossRef]
- United Nations Statistics Division. UNData - A world of information [Internet]. 2022. Available from: https://data.un.org/Data.aspx?q=Number+of+R&d=FAO&f=itemCode%3A1067.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




