Submitted:
10 March 2023
Posted:
13 March 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Synthesis
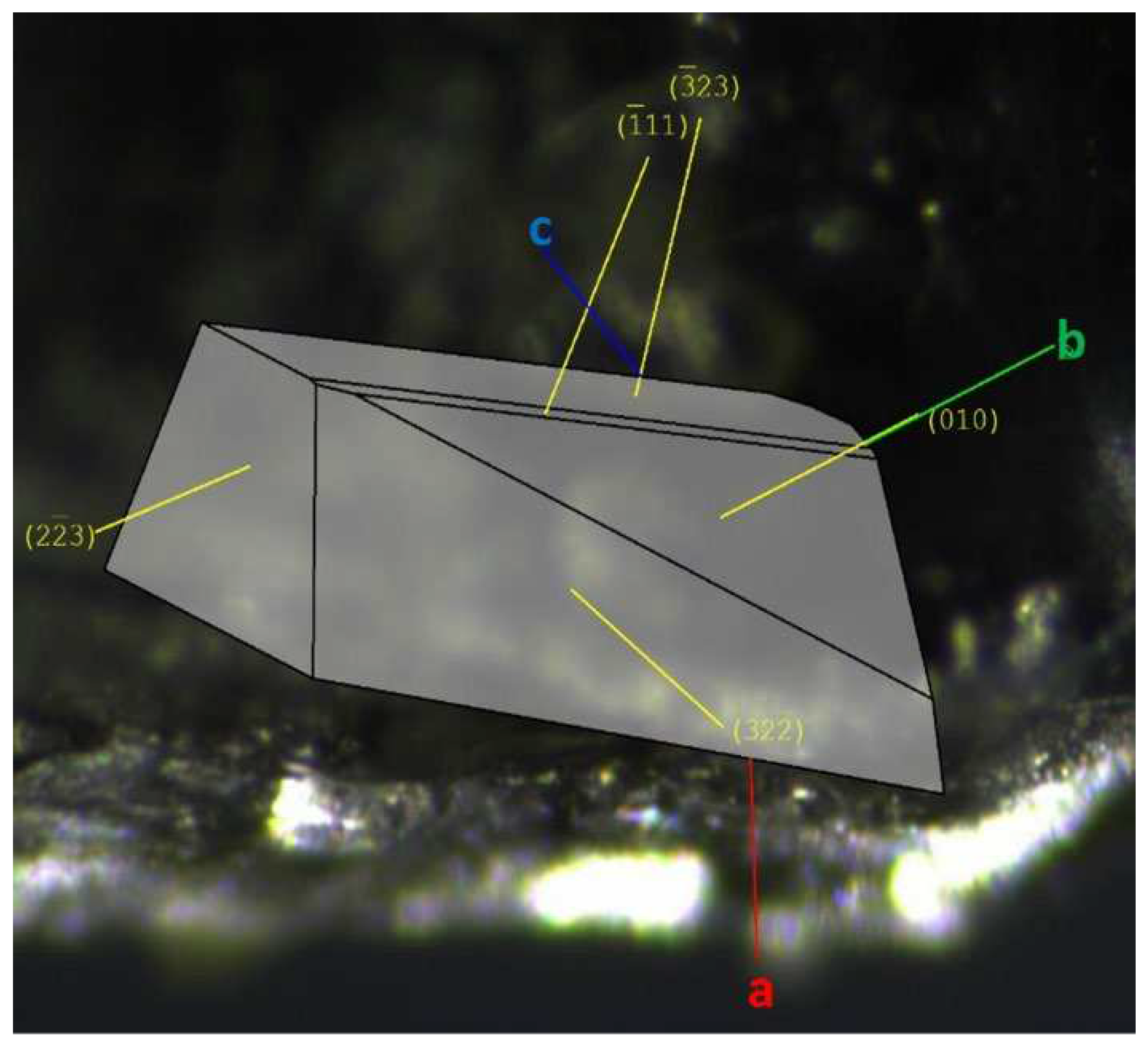
2.2. XRay Crystallography experimental conditions description
2.3. Dielectric spectroscopy
2.4. Pyroelectric Measurements
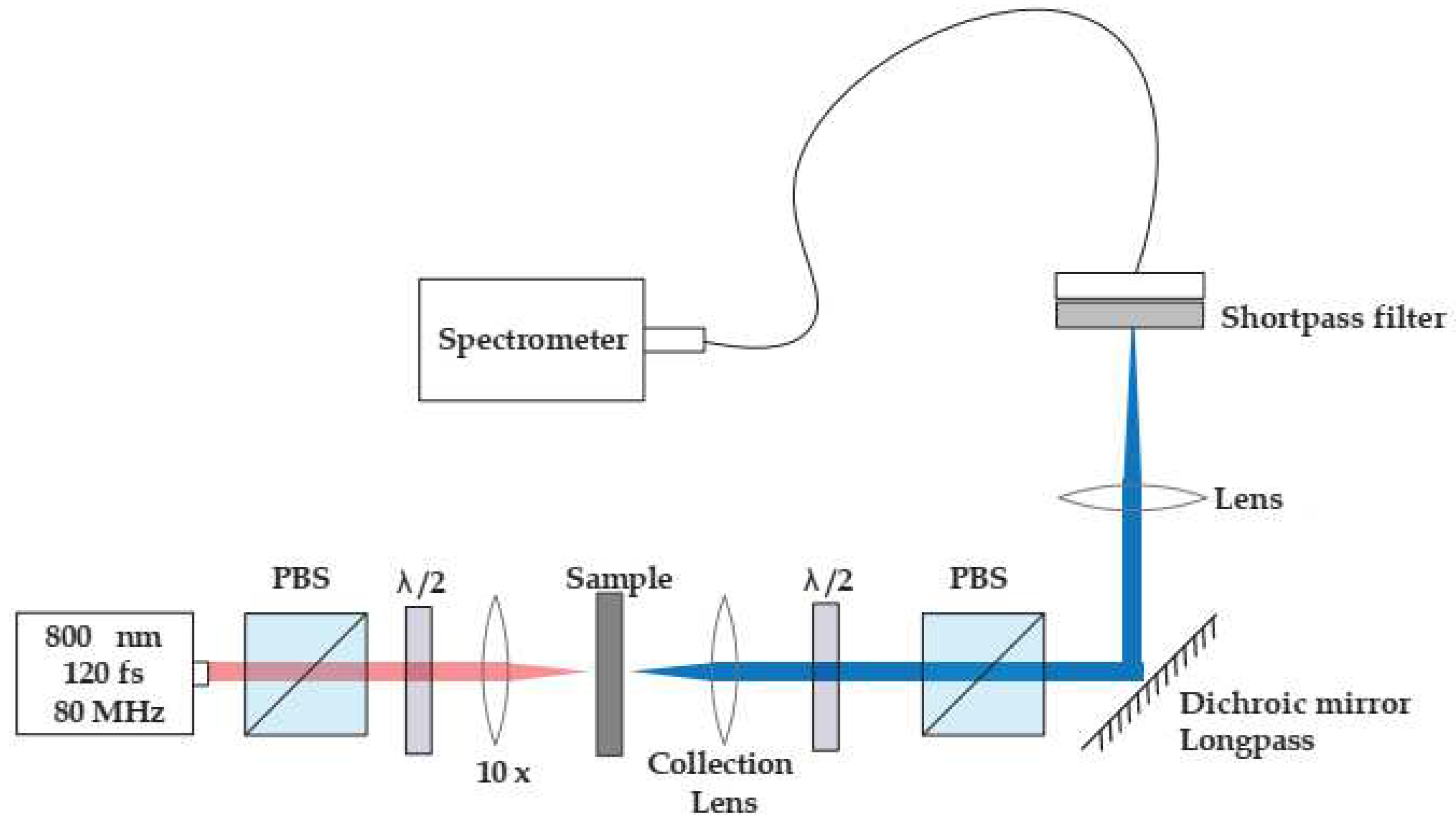
2.5. Second Harmonic Generation
3. Results and Discussion
3.1. Synthesis
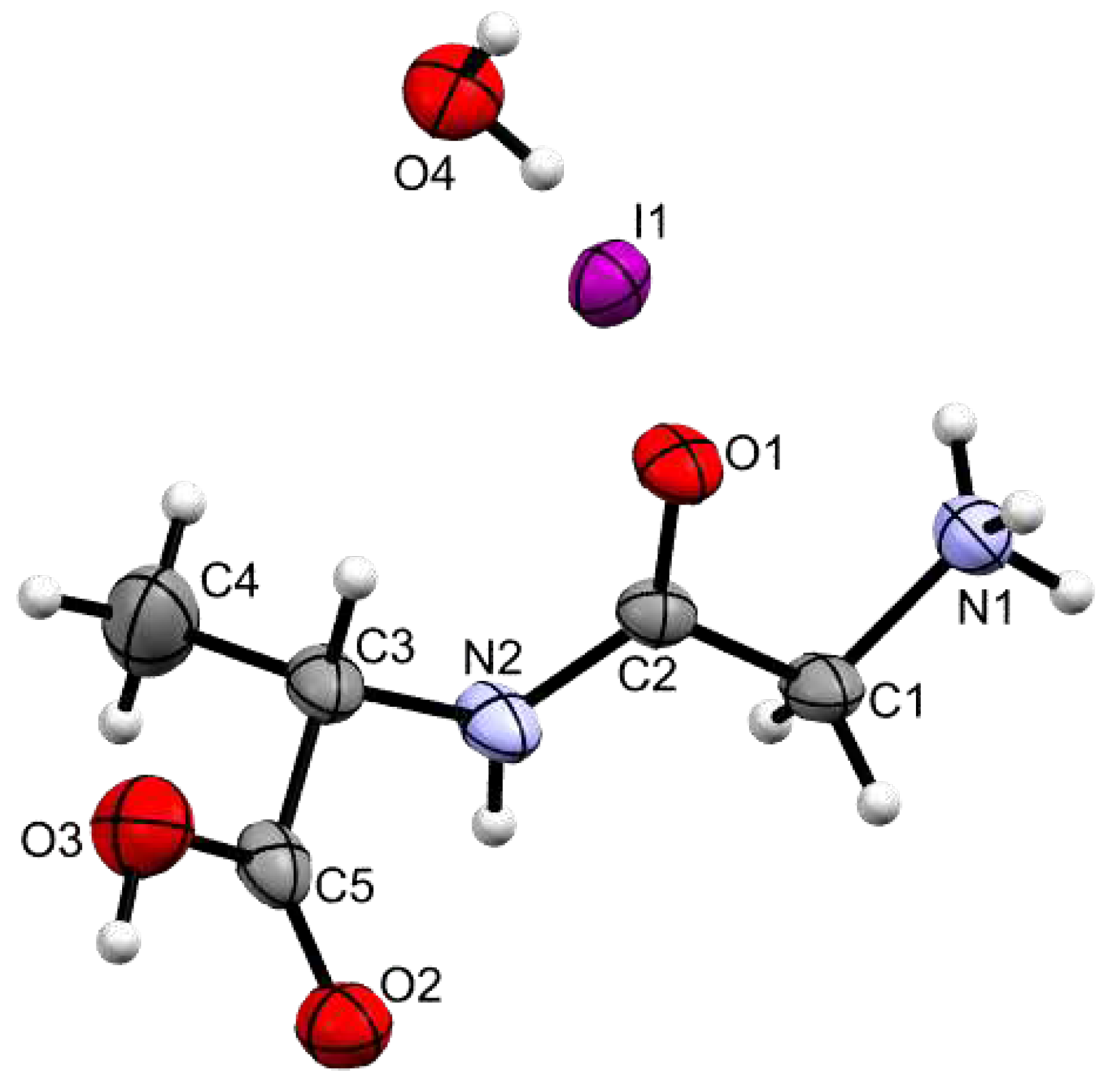
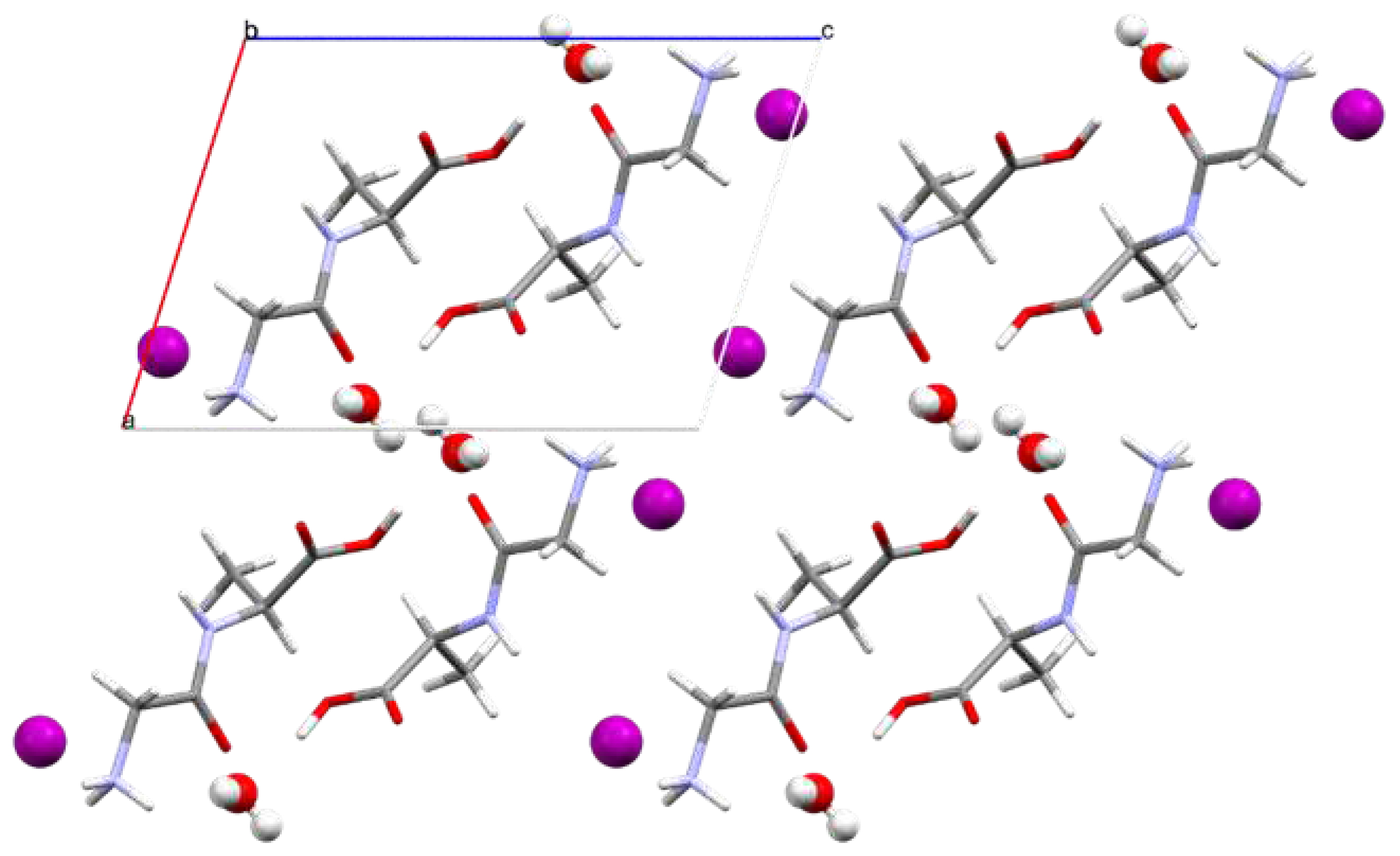
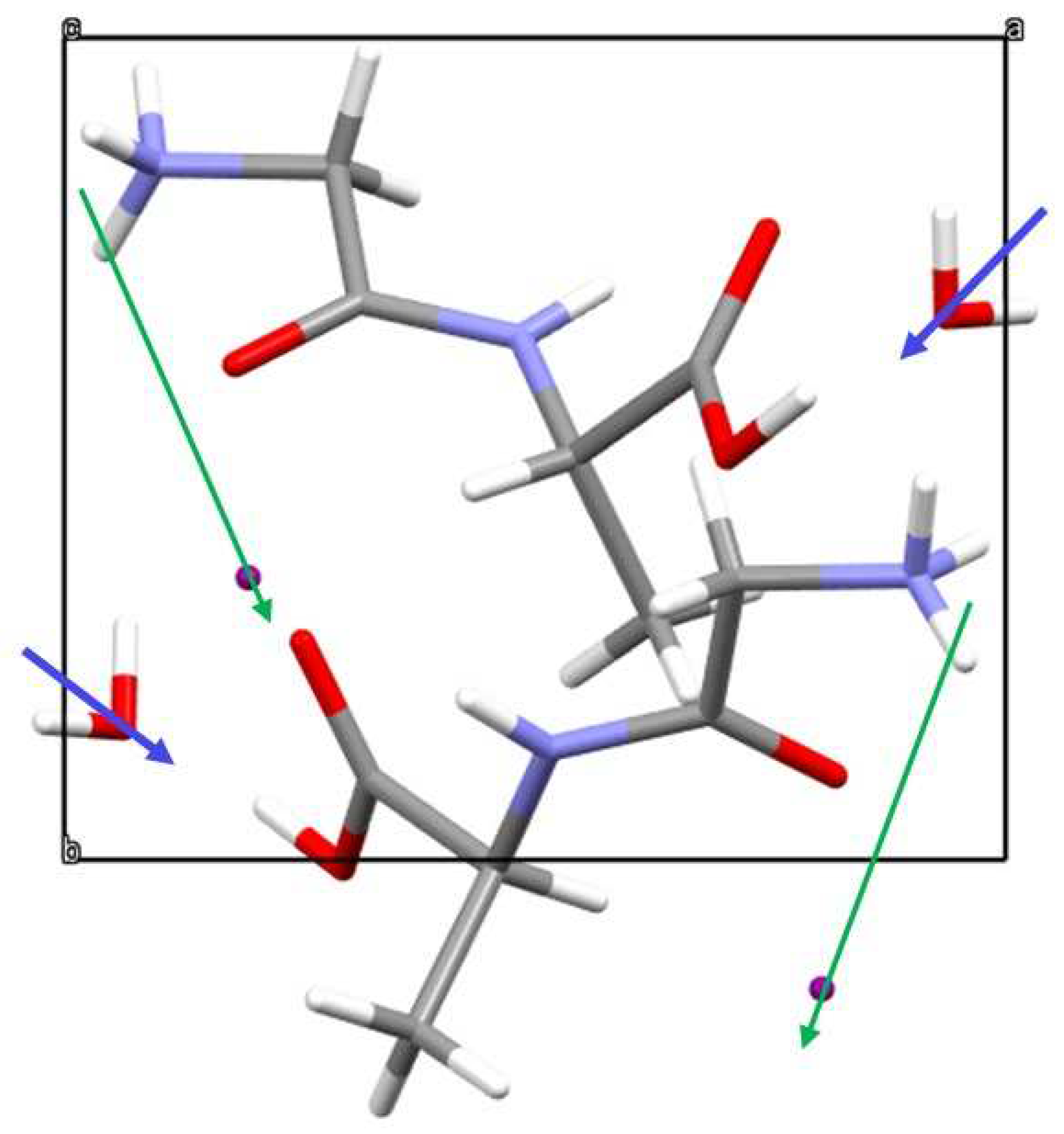
3.2. Crystal Structure
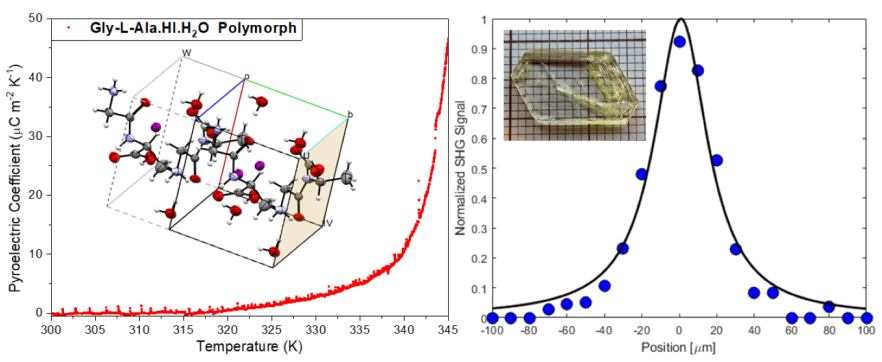
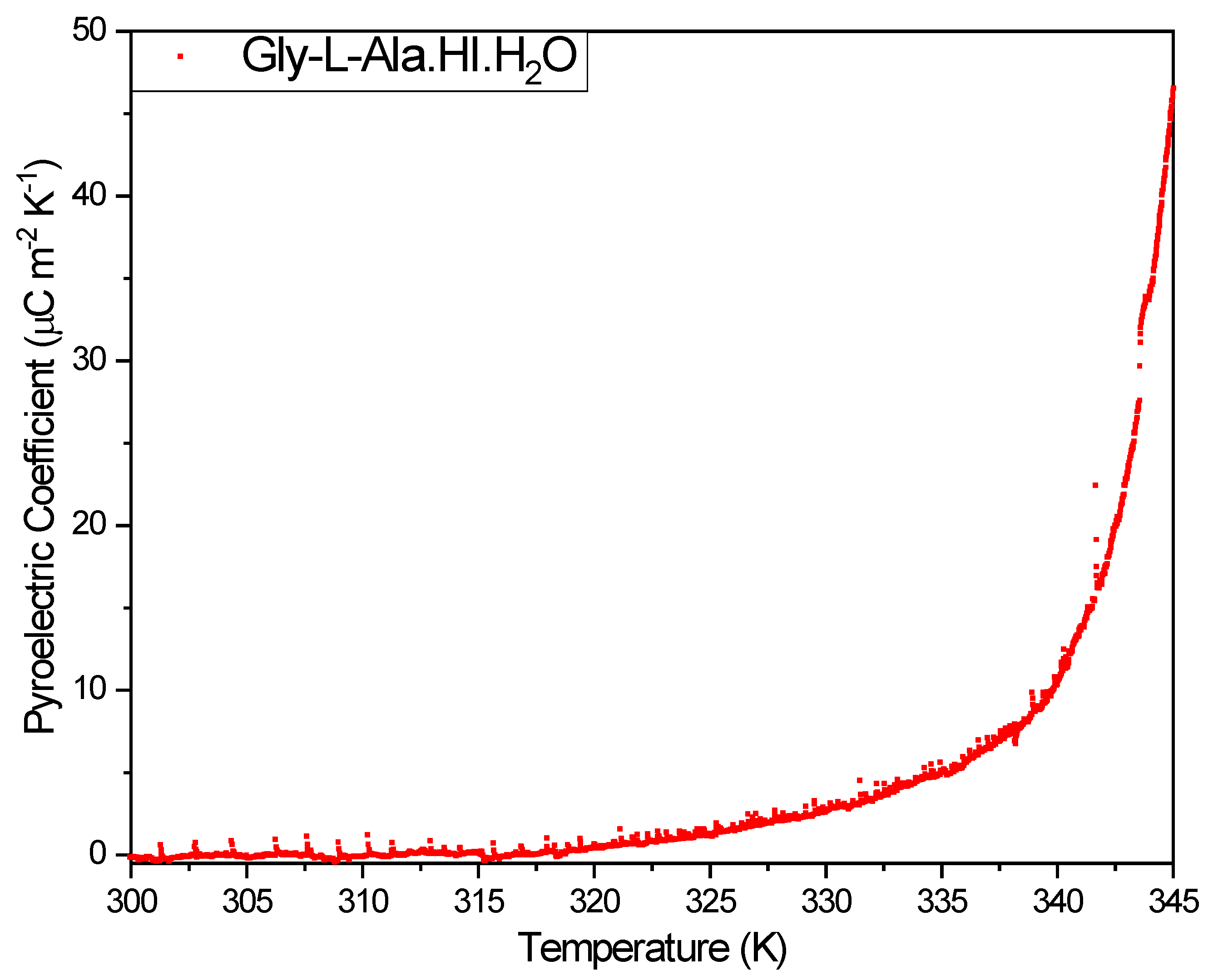
3.3. Pyroelectric properties
3.4. Thermal Properties
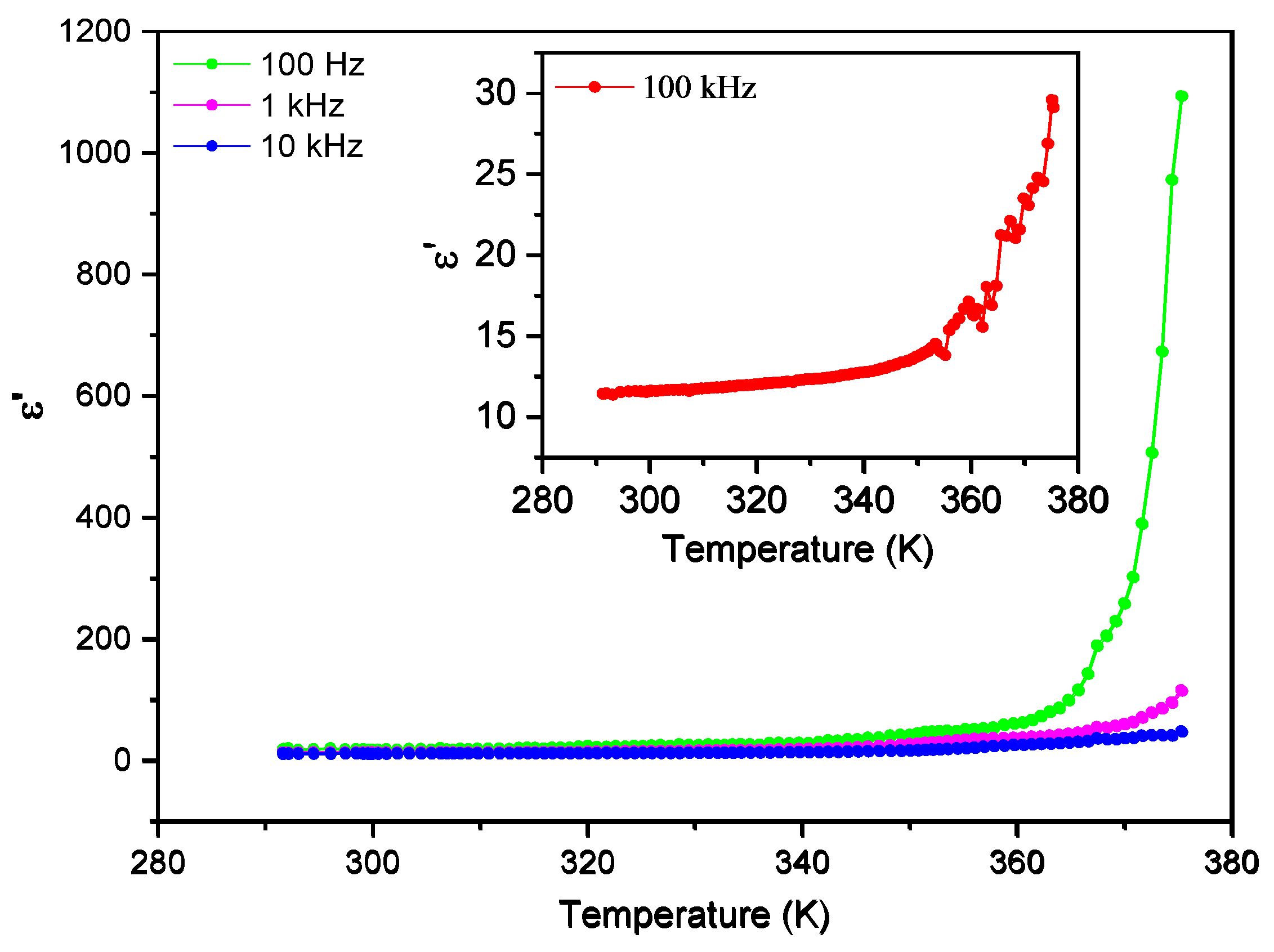
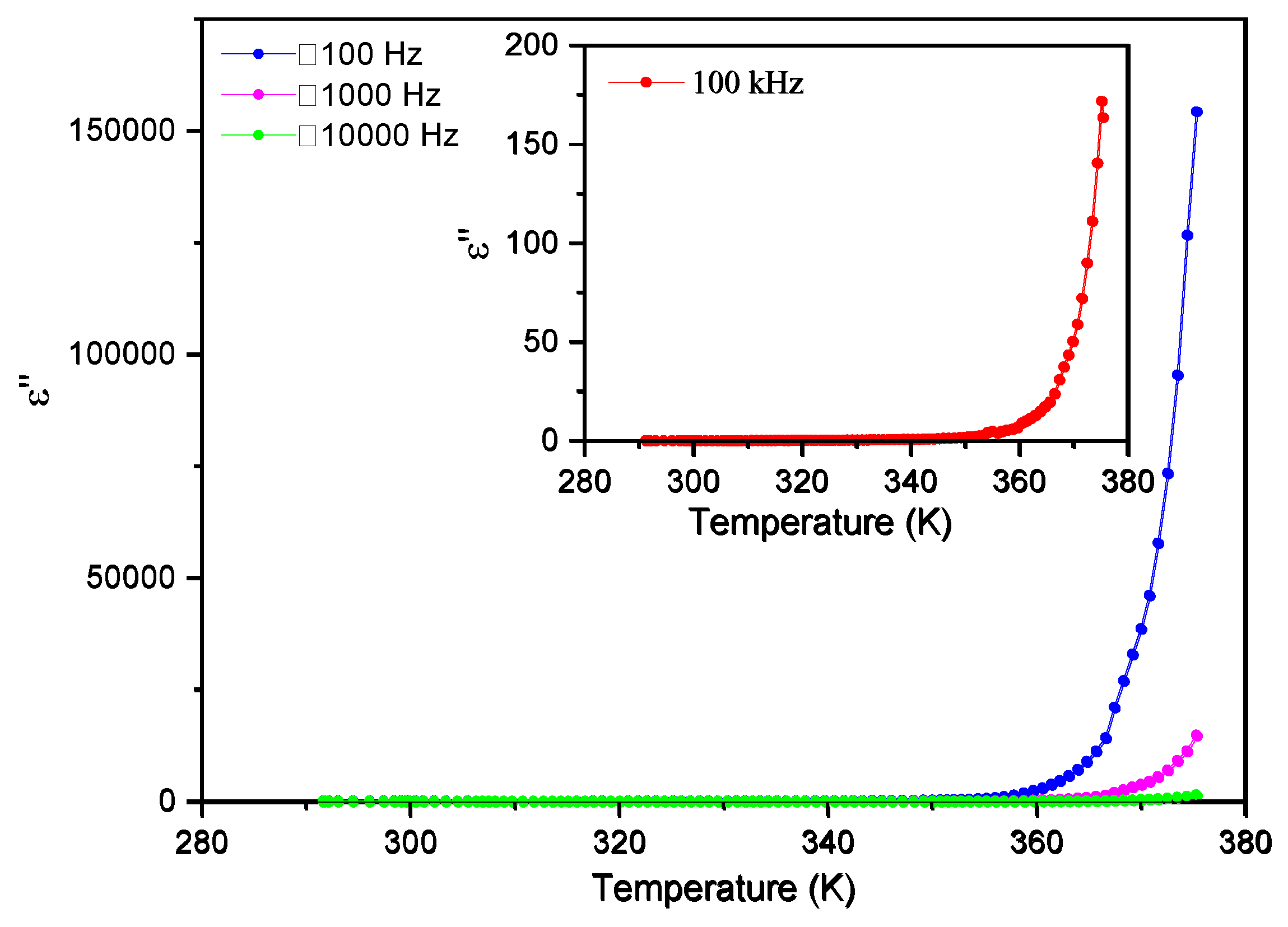
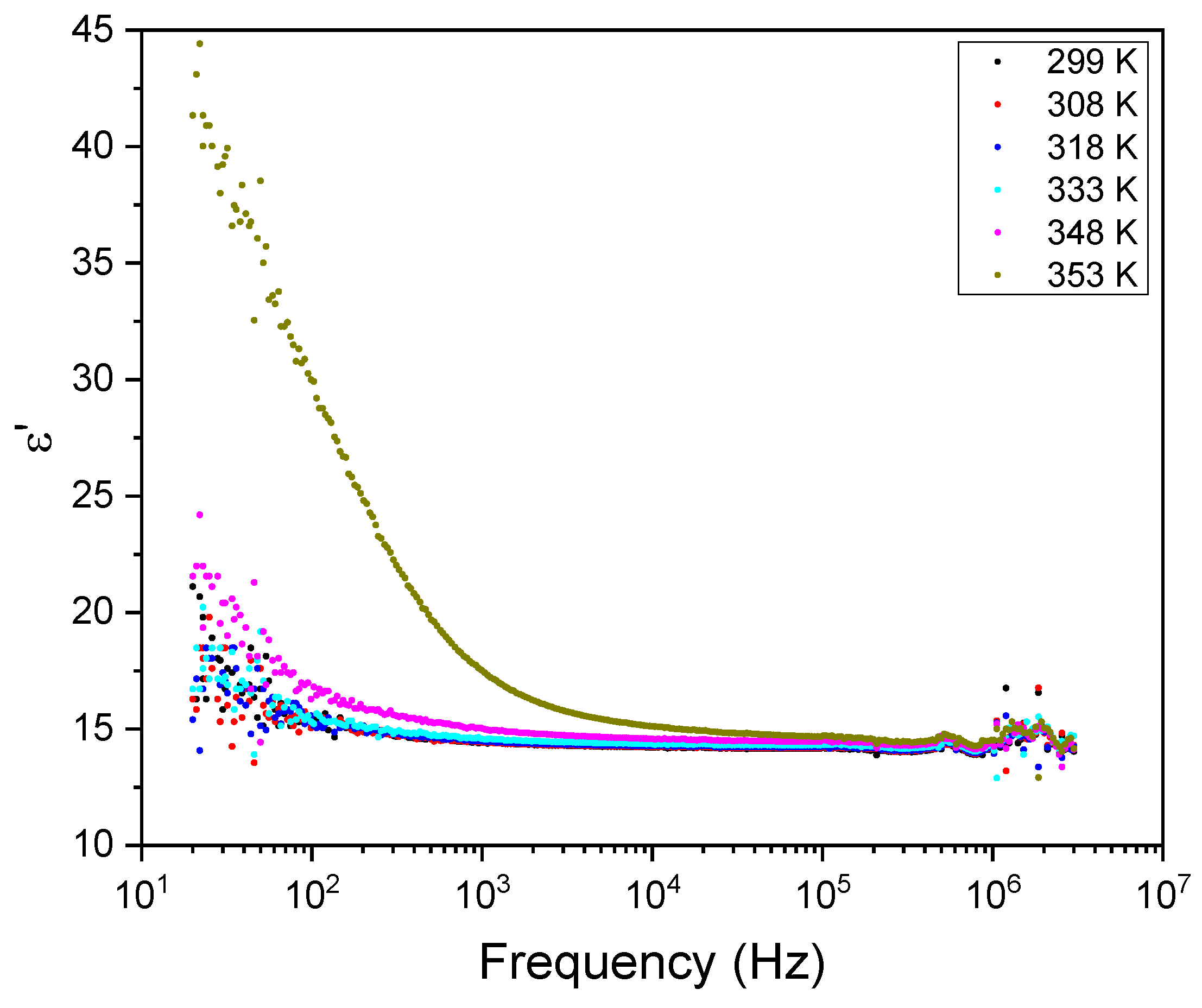
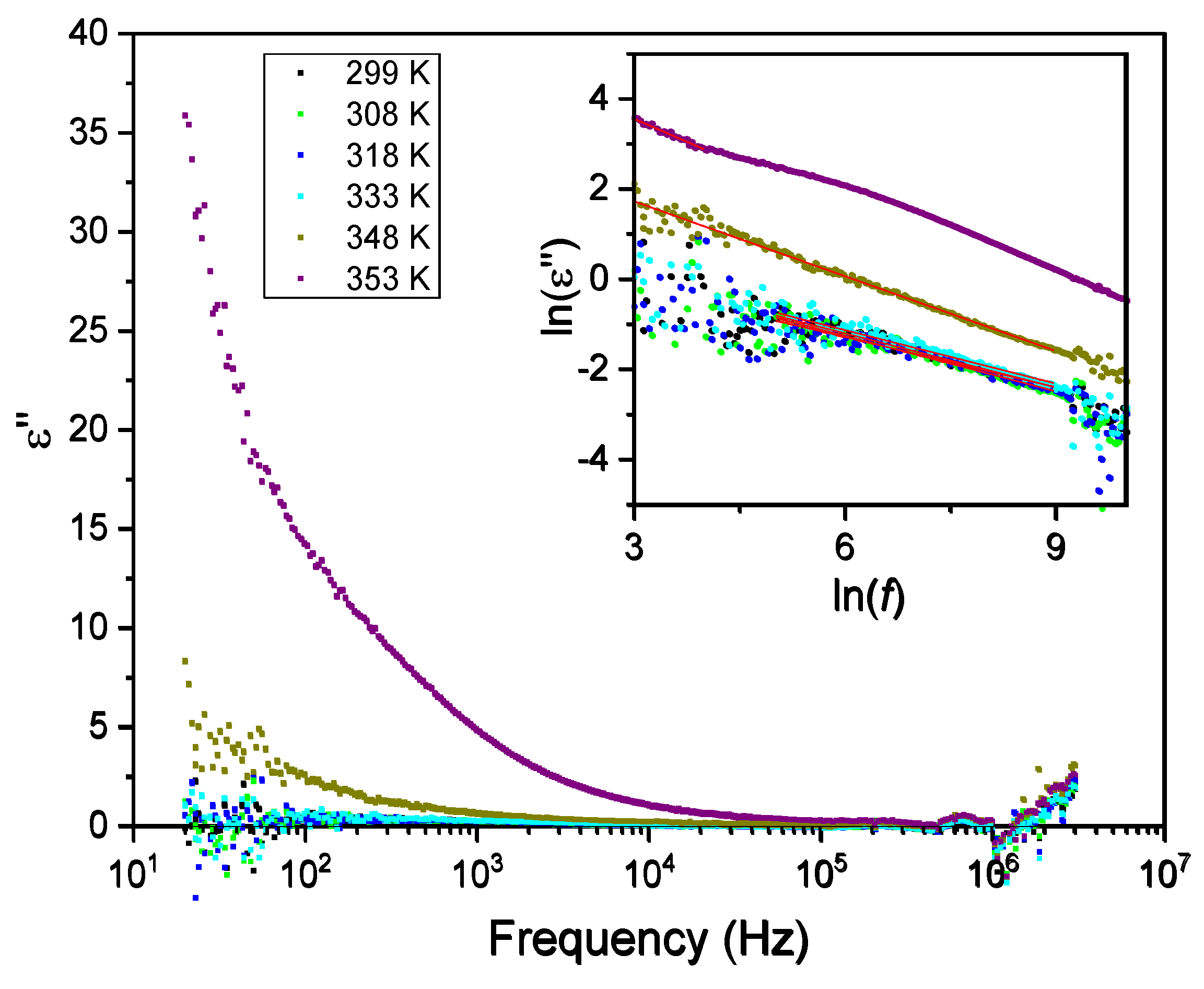
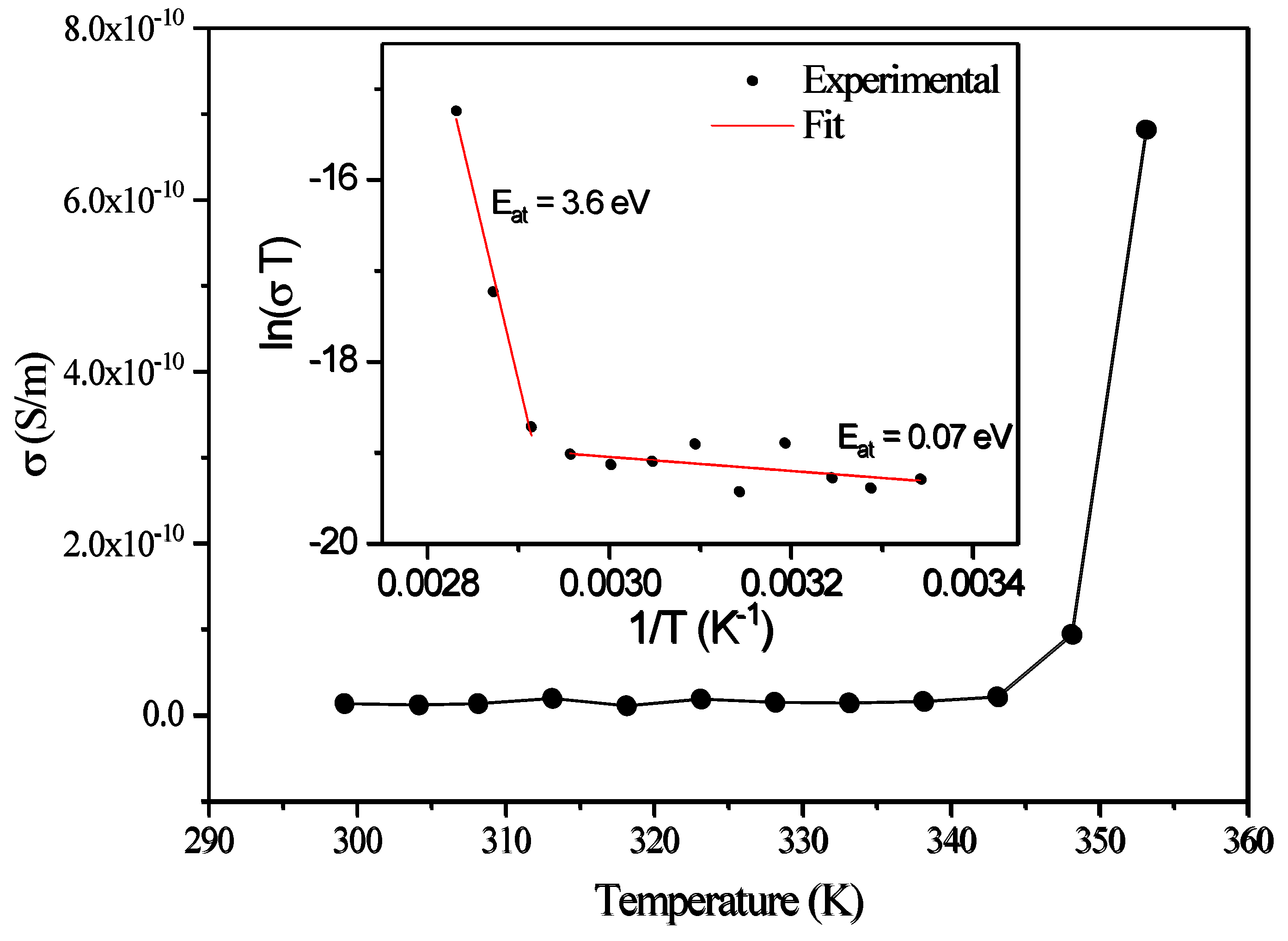
3.5. Dielectric Spectroscopy
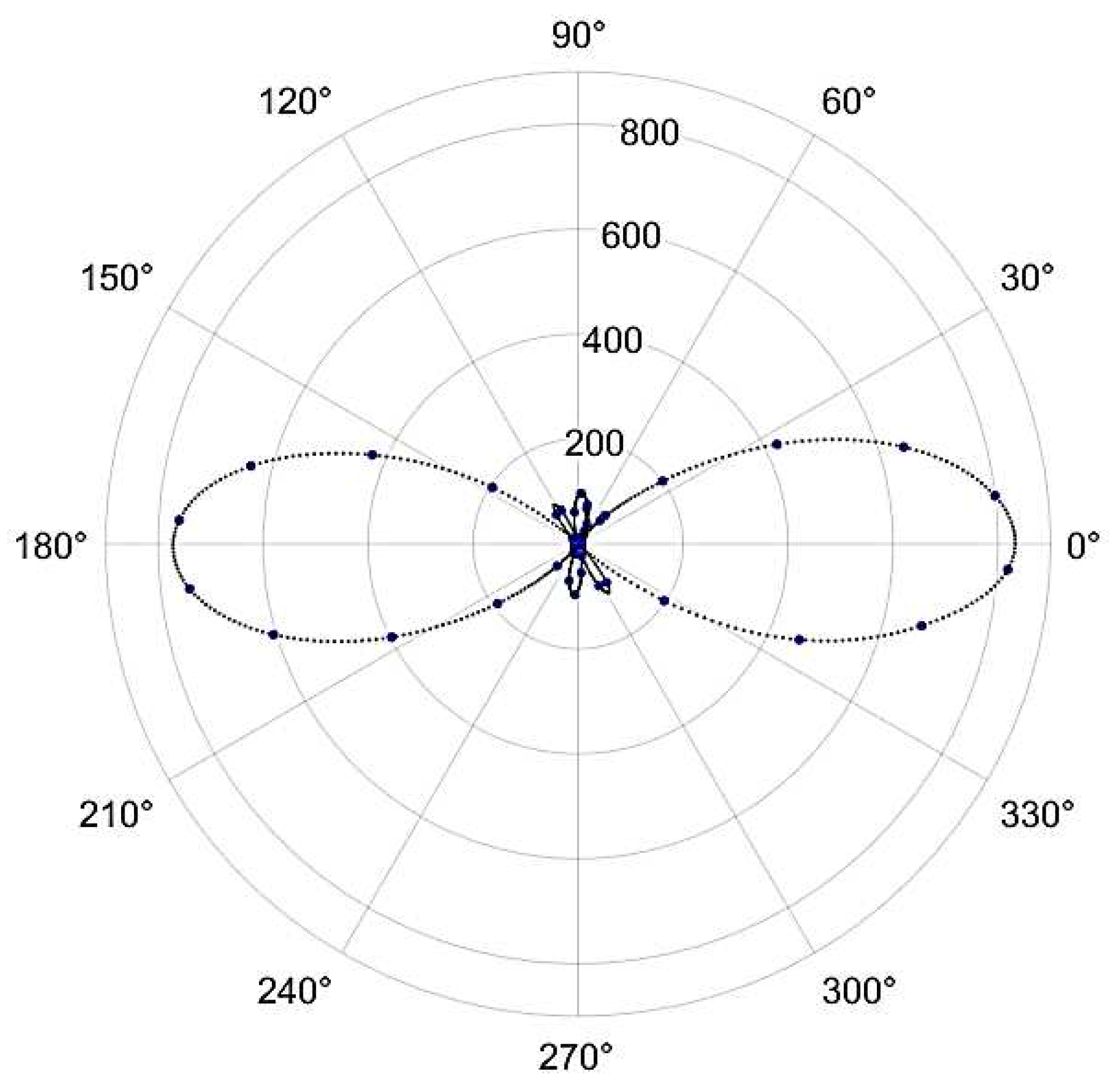
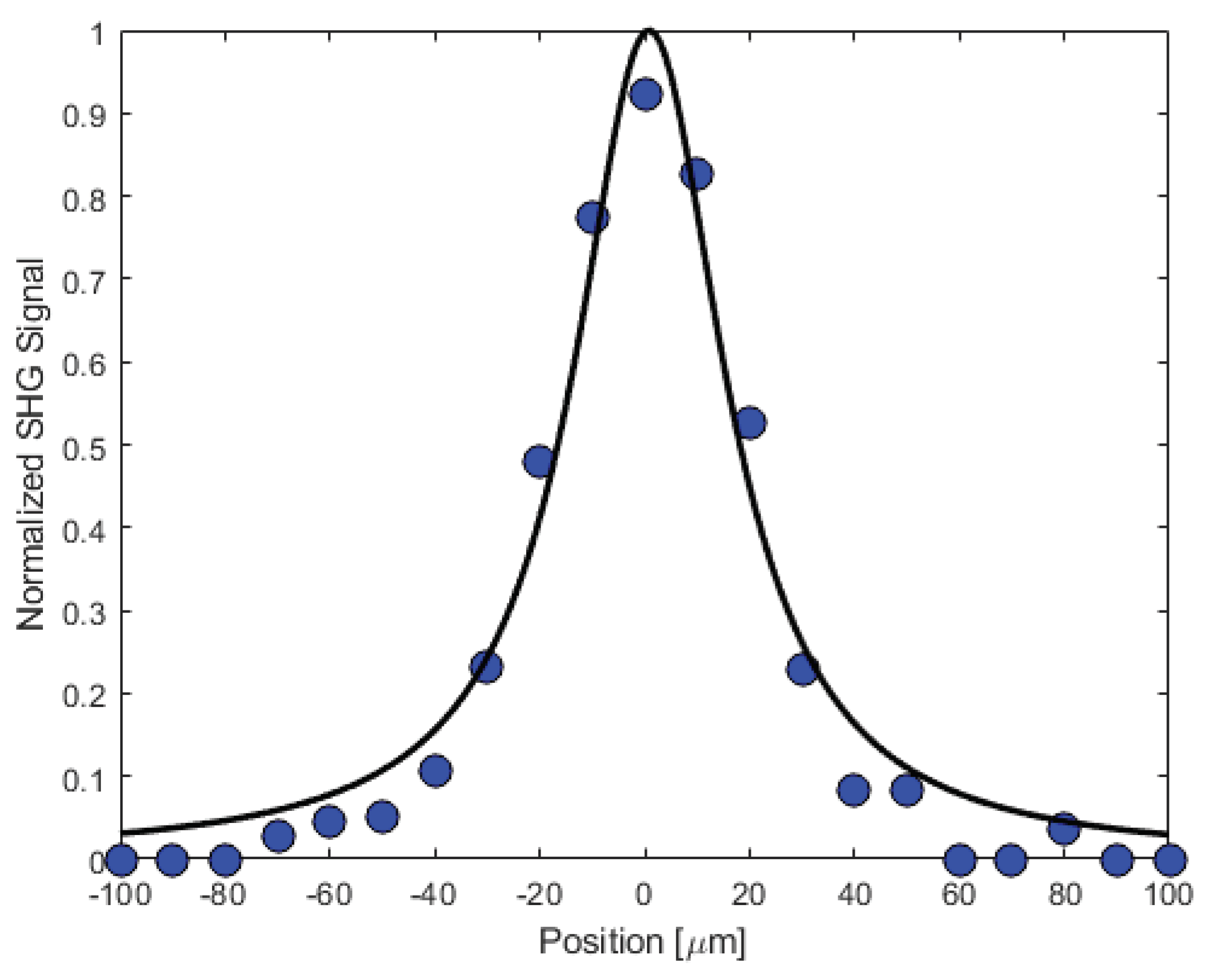
3.6. Second Harmonic Response
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kehrer, A.; Dou, S.-q.; Weiss, A. 79,81Br−127I−NQR, and Crystal Structure of Glycyl-l-alanine Hydrobromide Monohydrate and Hydroiodide Monohydrate. Z. Naturforsch. A 1989, 44, 659–668. [Google Scholar] [CrossRef]
- Kehrer, A.; Weiss, A. The pyroelectric coefficient of glycyl-l-alanine hydrobromide monohydrate and glycyl-alanine hydrolodide monohydrate. Ferroelectrics 1990, 106, 405–410. [Google Scholar] [CrossRef]
- Tranter, T.C. Crystal Structure of Glycyl-L-Alanine Hydrobromide. Nature 1954, 173, 221–222. [Google Scholar] [CrossRef]
- Tranter, T.C. Crystal Structure of Glycyl-L-Alanine Hydrochloride. Nature 1956, 177, 37–38. [Google Scholar] [CrossRef]
- Prasad, C. Bioactive cyclic dipeptides. Peptides 1995, 16, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Mendham, A.P.; Dines, T.J.; Snowden, M.J.; Chowdhry, B.Z.; Withnall, R. Vibrational spectroscopy and DFT calculations of di-amino acid cyclic peptides. Part I: cyclo(Gly-Gly), cyclo(L-Ala-L-Ala) and cyclo(L-Ala-Gly) in the solid state and in aqueous solution. J. Raman Spectrosc. 2009, 40, 1478–1497. [Google Scholar] [CrossRef]
- Rabone, J.; Yue, Y.F.; Chong, S.Y.; Stylianou, K.C.; Bacsa, J.; Bradshaw, D.; Darling, G.R.; Berry, N.G.; Khimyak, Y.Z.; Ganin, A.Y.; et al. An Adaptable Peptide-Based Porous Material. Science 2010, 329, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Barone, G.; Puliti, R. Correlation Between Phase Transition Thermodynamics and Crystal Features of Solid Small Peptides. J. Therm. Anal. Calorim. 1999, 57, 119–132. [Google Scholar] [CrossRef]
- Capelli, S.C.; Burgi, H.-B.; Dittrich, B.; Grabowsky, S.; Jayatilaka, D. Hirshfeld atom refinement. IUCrJ 2014, 1, 361–379. [Google Scholar] [CrossRef]
- Huang, F.; Nau, W.M. A Conformational Flexibility Scale for Amino Acids in Peptides. Angew. Chem. Int. Ed. 2003, 42, 2269–2272. [Google Scholar] [CrossRef]
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of silver and molybdenum microfocus X-ray sources for single-crystal structure determination. J. Appl. Crystallogr. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G. Crystal structure refinement with SHELXL. Acta Crystallogr. Sec. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Farrugia, L. WinGX and ORTEP for Windows: an update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: from visualization to analysis, design and prediction. J. Appl. Crystallogr. 2020, 53, 226–235. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. Sec. B 2016, 72, 171–179. [Google Scholar] [CrossRef]
- Isakov, D.V.; Ferreira, F.P.; Barbosa, J.; Ribeiro, J.L.; de Matos Gomes, E.; Belsley, M.S. Piezoelectric, nonliner optical, electro-optical, and pyroelectric properties of cesium hydrogen malate hydrate crystal. Appl. Phys. Lett. 2007, 90, 073505. [Google Scholar] [CrossRef]
- Fleck, S.; Weiss, A. Dielectric and pyroelectric properties of lithium hydrogen dimalate, LiH3(C4H4O5)2. Z. Naturforsch. A 1986, 41, 1289–1296. [Google Scholar] [CrossRef]
- Bhalla, A.S.; Cook, W.R.; Liu, S.T. Numerical data and functional relationships in science and technology; Nelson, D.F., Ed.; Springer: Berlin, 1993; Volume Vol. 29b, pp. 121–351.
- Mishuk, E.; Weissbuch, I.; Lahav, M.; Lubomirsky, I. Pyroelectricity in Nonpolar Directions in Crystals: Enantiomeric Disorder and Surface Wetting in Racemic α-Amino-Acids. Cryst. Growth. Des. 2014, 14, 3839–3848. [Google Scholar] [CrossRef]
- Do, H.T.; Chua, Y.Z.; Habicht, J.; Klinksiek, M.; Hallermann, M.; Zaitsau, D.; Schick, C.; Held, C. Melting properties of peptides and their solubility in water. Part 1: dipeptides based on glycine or alanine. Rsc. Adv. 2019, 9, 32722–32734. [Google Scholar] [CrossRef]
- Kremer, F.; Schönhals, A. Broadband dielectric spectroscopy; Springer Science & Business Media: 2002.
- Nuernberg, R.B. Numerical comparison of usual Arrhenius-type equations for modeling ionic transport in solids. Ionics 2020, 26, 2405–2412. [Google Scholar] [CrossRef]
- Franchini, C.; Reticcioli, M.; Setvin, M.; Diebold, U. Polarons in materials. Nat. Rev. Mater. 2021, 6, 560–586. [Google Scholar] [CrossRef]
- Tulip, P.R.; Bates, S.P. First principles determination of structural, electronic and lattice dynamical properties of a model dipeptide molecular crystal. Mol. Phys. 2009, 107, 2201–2212. [Google Scholar] [CrossRef]
- Tzankov, P.; Petrov, V. Effective second-order nonlinearity in acentric optical crystals with low symmetry. Appl. Optics 2005, 44, 6971–6985. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Weiner, A.M. Efficiency of short-pulse type-I second-harmonic generation with simultaneous spatial walk-off, temporal walk-off, and pump depletion. Ieee J. Quantum Elect. 2003, 39, 1600–1618. [Google Scholar] [CrossRef]
- D'Errico, J. Fminspleas available at MATLAB Central File Exchange. (accessed on Retrieved March 7).

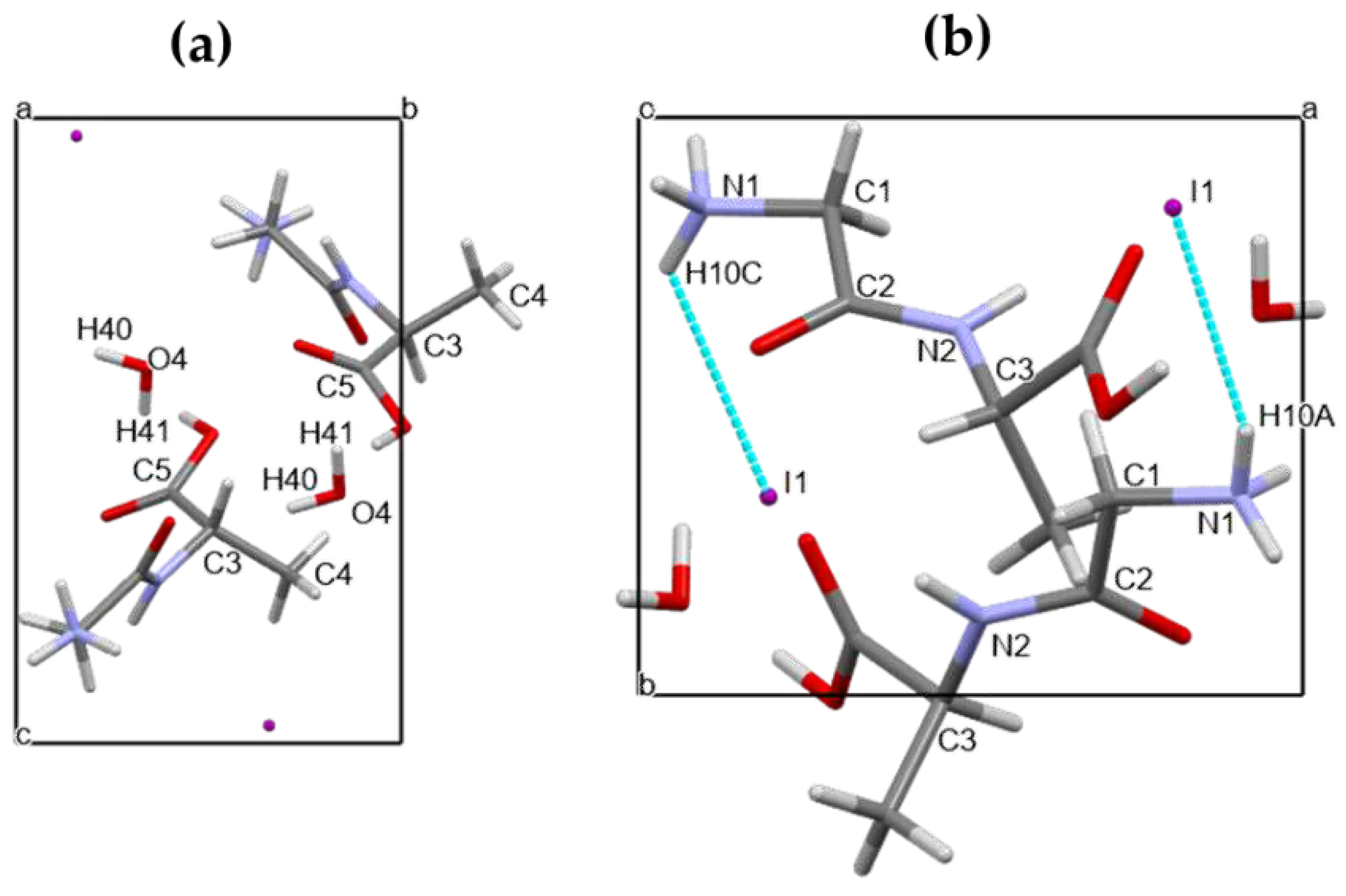
| D-H...A | d(H...A) | d(D...A) | <(DHA) | Symmetry operation |
| C(1)-H(1B)...I(1) | 3.26 | 3.953(3) | 129.5 | -x+1,y-1/2,-z+2 |
| N(1)-H(10B)...O(2) | 2.30(6) | 2.902(6) | 128(6) | x-1,y,z |
| N(1)-H(10B)...O(4) | 2.47(6) | 3.110(8) | 133(6) | x,y-1,z |
| N(1)-H(10A)...I(1) | 2.77(4) | 3.570(9) | 152(6) | x,y-1,z |
| N(1)-H(10C)...I(1) | 3.13(11) | 3.642(4) | 119(10) | -x,y-1/2,-z+2 |
| N(1)-H(10C)...I(1) | 2.96(11) | 3.598(9) | 130(11) | |
| N(2)-H(20)...I(1) | 2.76(10) | 3.637(5) | 169(7) | -x+1,y-1/2,-z+2 |
| O(3)-H(30)...O(4) | 1.77(10) | 2.662(6) | 167(9) | -x+1,y-1/2,-z+1 |
| O(4)-H(41)...O(1) | 2.026 | 2.824(4) | 161.29(15) | -x,y+1/2,-z+1 |
| O(4)-H(40)...O(1) | 2.233 | 2.982(6) | 152.86 |
| Material | ε´ | 𝑝 (µC/m2K) | Ref. | ||
| Gly-L-AlaHI.H2O (Poly2) | 11.5 | 45 (345 K) | This work | ||
| Gly-L-AlaHI.H2O (Poly1) | __ | 15.5 (357 K) | [2] | ||
| CsH(C4H4O5)∙H2O | 11.2 | 2.5 (245 K) | [16] | ||
| LiH3(C4H4O5)2 | 8.1 | 6.1 | [17] | ||
| (NH2CH2COOH)3H2SO4 | 25 | 350 | [18] | ||
| DL-Alanine | __ | 5.5 | [19] | ||
| KH2PO4 | 45 | 300 (120 K) | [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




