Submitted:
24 February 2023
Posted:
27 February 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and reagents
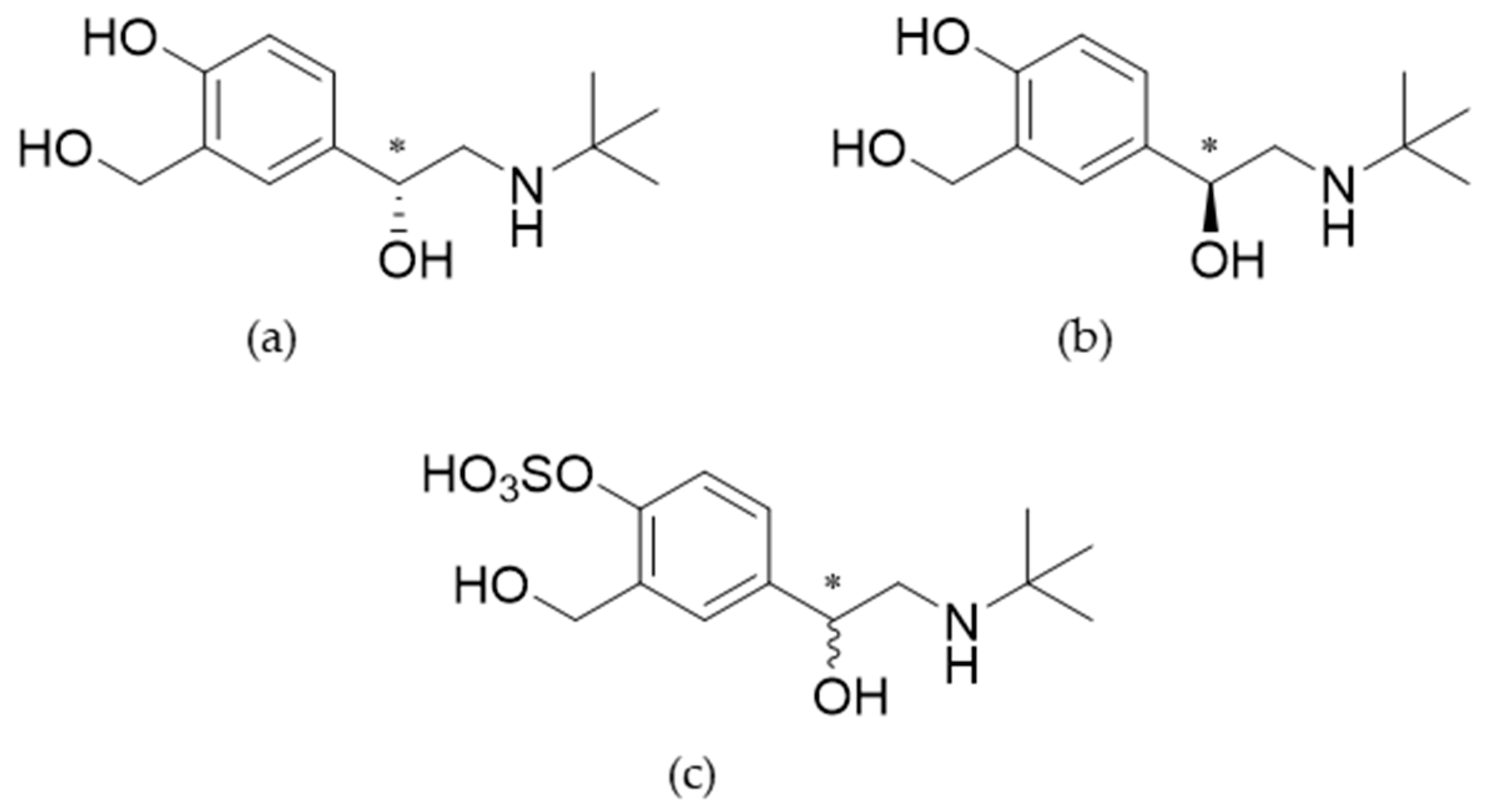
2.2. Synthesis of salbutamol-4’-O-sulfate as reference
2.3. Characterization of salbutamol-4’-O-sulfate
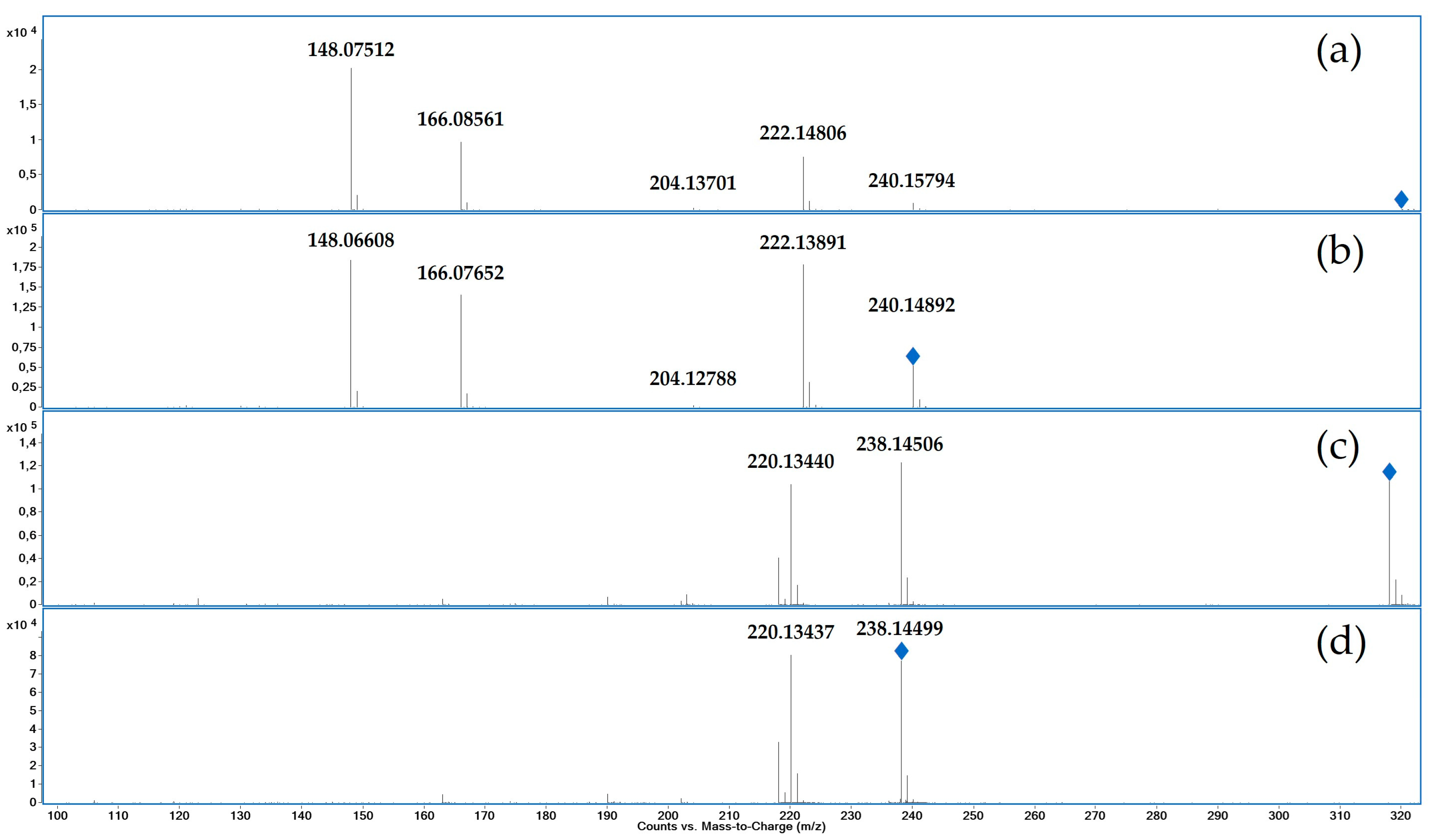
2.3.1. UHPLC-QTOF-MS
2.3.2. Nuclear Magnetic Resonance
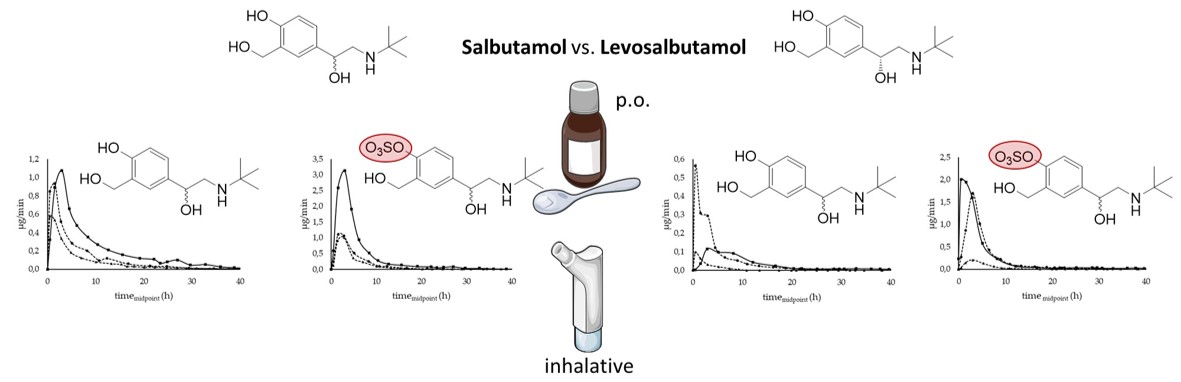
2.4. Study design
2.5. Sample preparation
2.6. Instruments and chromatographic conditions
2.7. Data analysis
| Time (min) | A1 (%) | B2 (%) |
| 0.00 | 95 | 5 |
| 1.00 | 95 | 5 |
| 5.00 | 60 | 40 |
| 6.00 | 5 | 95 |
| 7.90 | 5 | 95 |
| 8.00 | 95 | 5 |
| Ion source parameters | ||||
| Gas temperature | 170 °C | |||
| Gas flow | 17 l/min | |||
| Nebulizer | 10 psi | |||
| Sheath gas temperature | 400 °C | |||
| Sheath gas flow | 12 l/min | |||
| Capillary voltage | 4000 V | |||
| Nozzle voltage | 500 V | |||
| MRM parameters | ||||
| Precursor ion [m/z] | Product ion [m/z] | Collision energy | ||
| Salbutamol |
[M+H]+ = 240.0 |
222.1 166.1 148.1 121.1 91.0 77.1 |
8 12 16 25 48 56 |
|
| Salbutamol-4’-O-sulfate | [M+H]+ = 320.0 |
240.0 222.0 166.0 148.0 77.0 |
4 16 16 32 80 |
|
| [M-H]- = 318.0 |
238.0 |
25 |
||
| Salbutamol-d9 | [M+H]+ = 249.2 | 231.1 166.1 148.1 121.1 |
8 12 16 25 |
|
| Salbutamol glucuronide | [M+H]+ = 416.0 |
298.0 240.0 224.0 222.0 148.0 |
12 18 29 20 20 |
|
|
[M-H]- = 414.0 |
396.0 220.0 146.0 |
18 25 25 |
||
3. Results
3.1. Characterization of reference by UHPLC-QTOF-MS and NMR
3.2. Administration of salbutamol through dry powder inhaler and metered dose inhaler
3.3. Pharmacokinetic evaluation of urine data
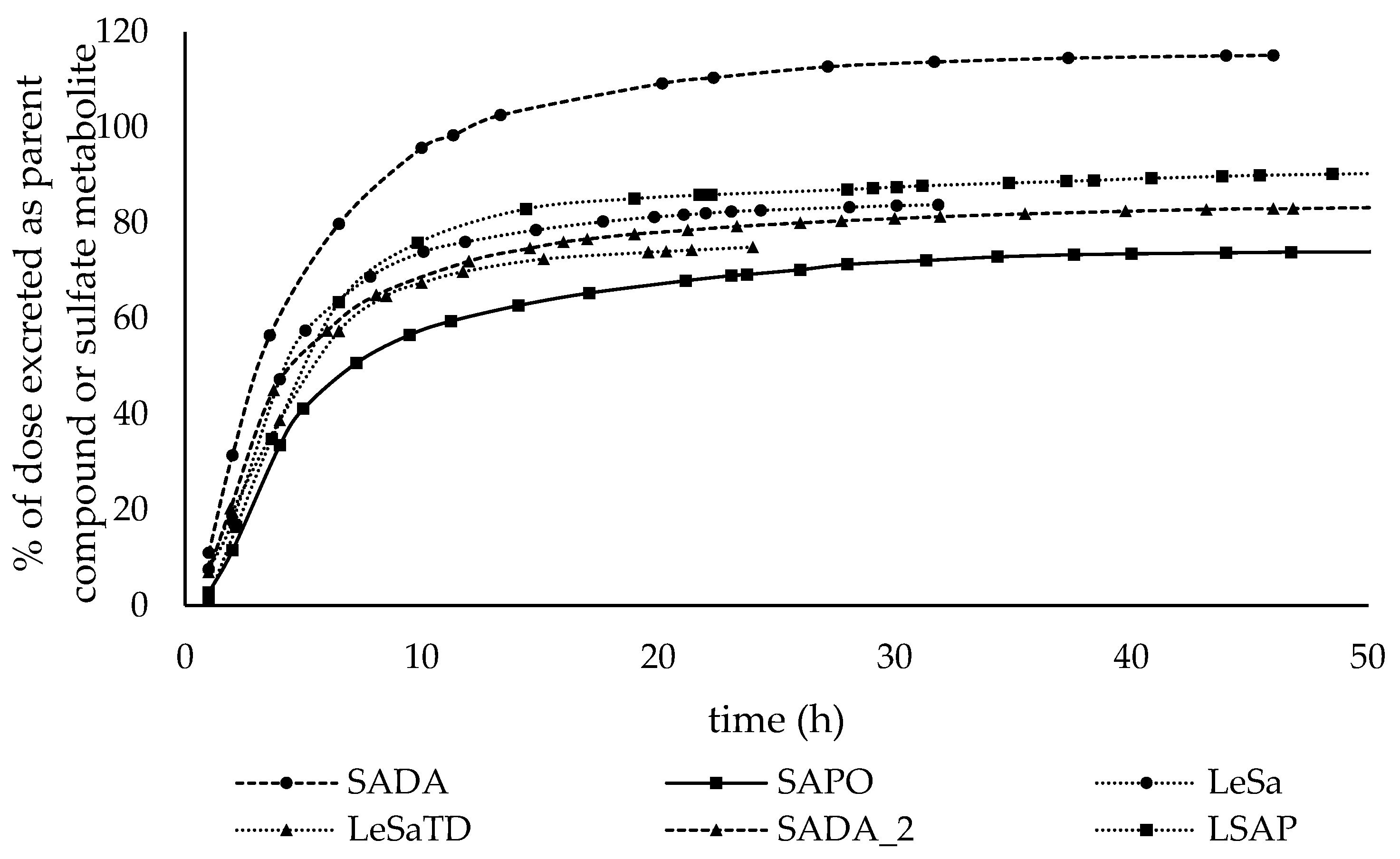
3.3.1. Cumulative excretion of salbutamol and salbutamol-4’-O-sulfate
3.3.2. Urinary excretion rates
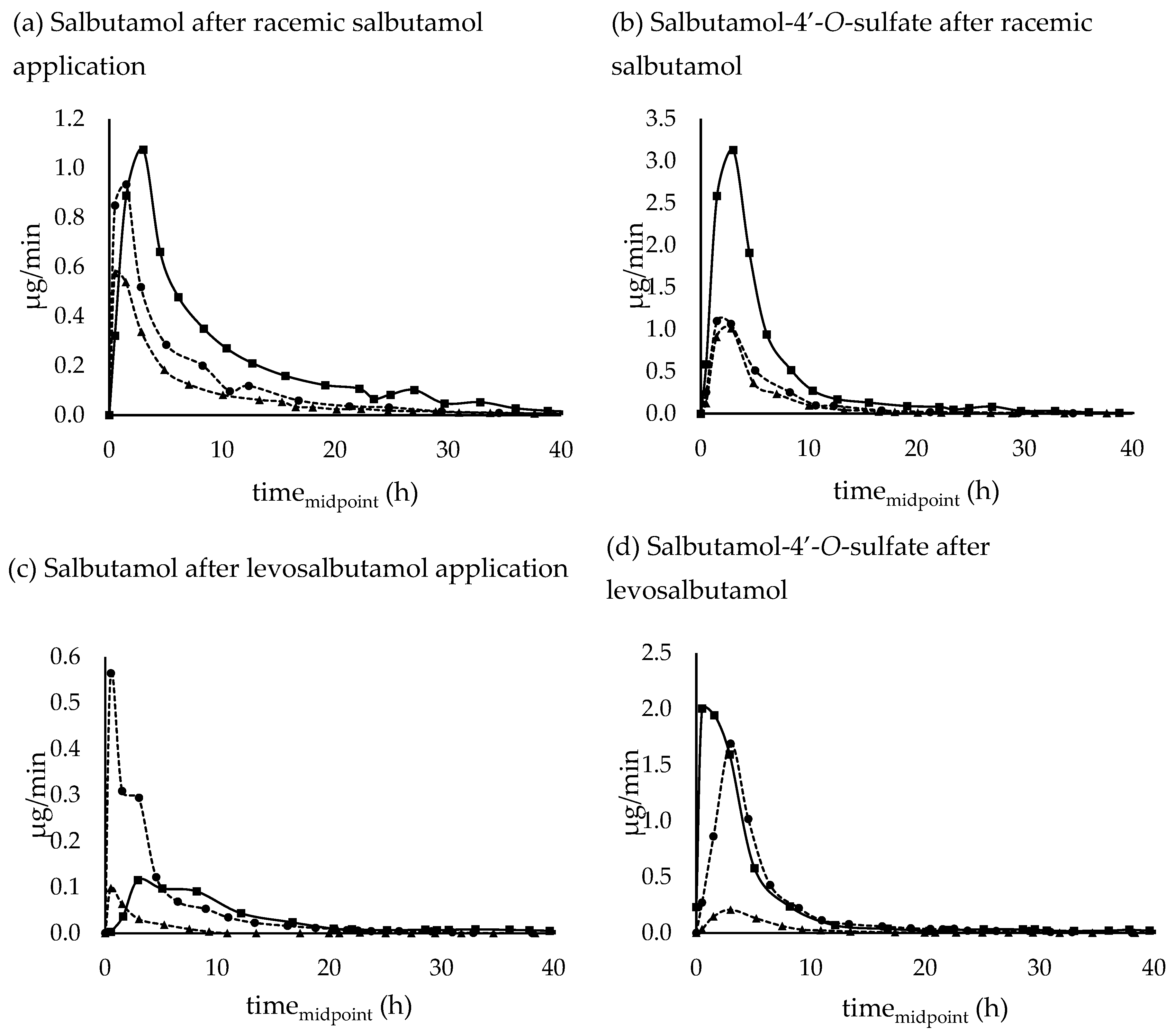
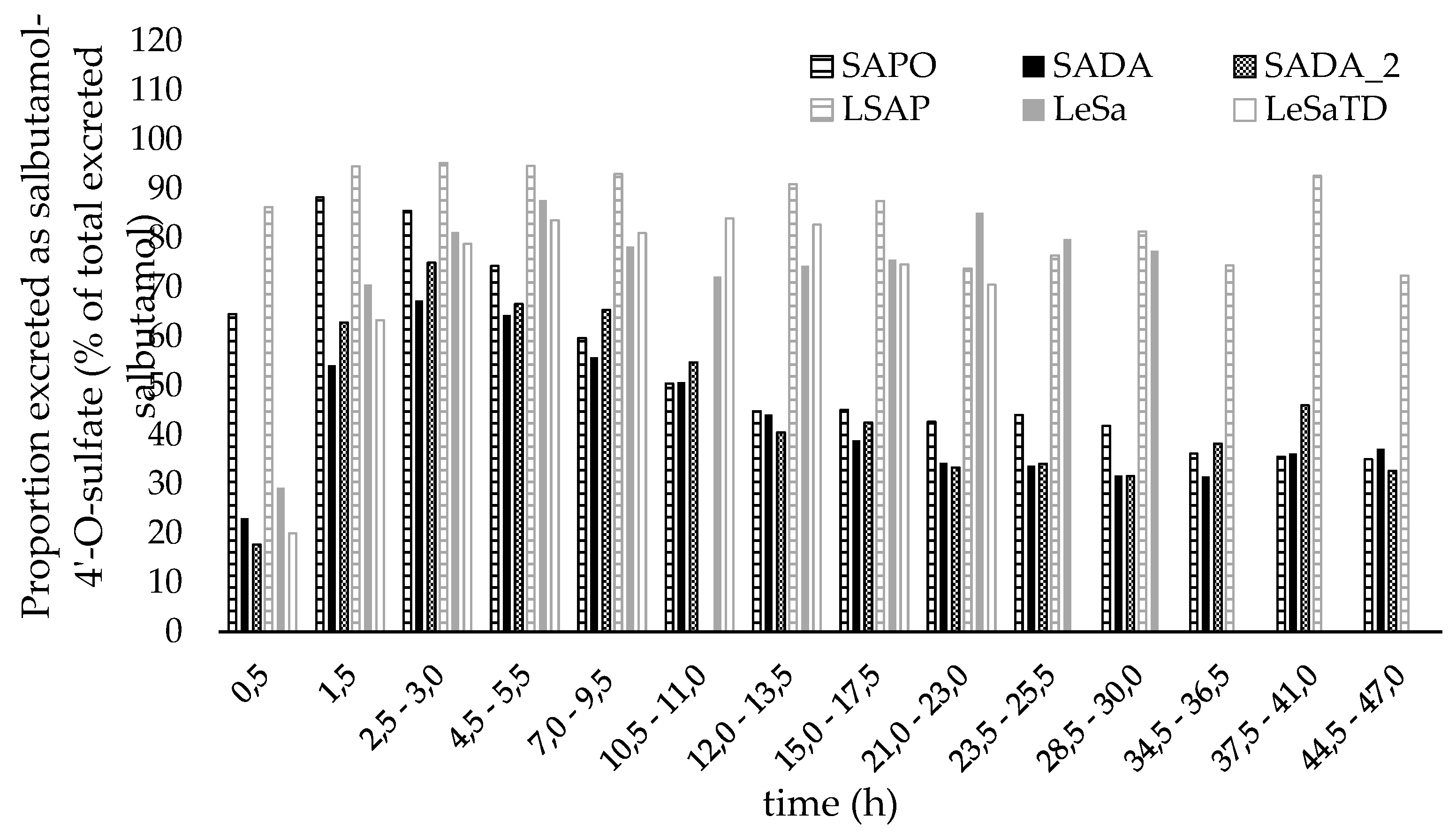
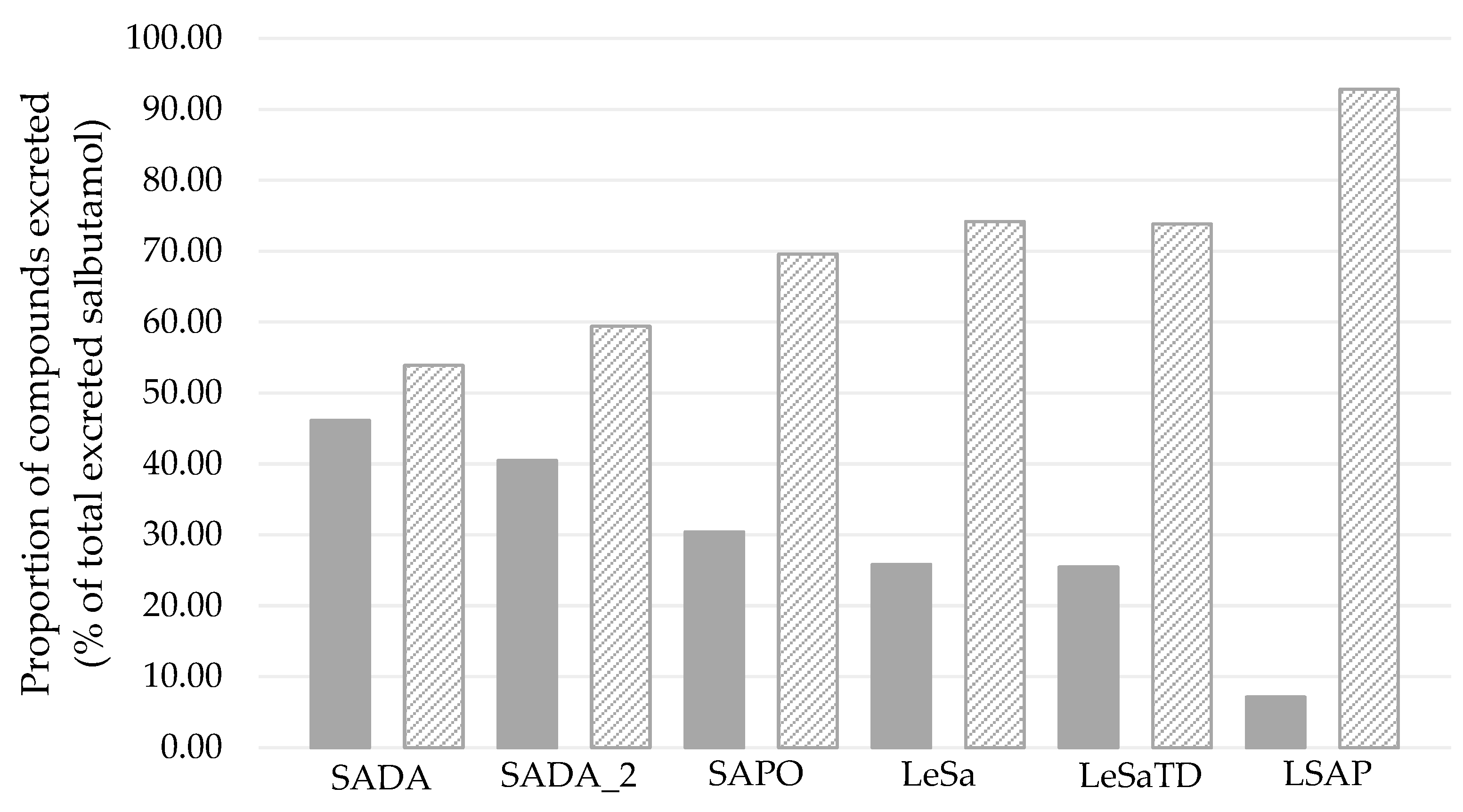
3.3.2. Salbutamol-4’-O-sulfate in relation to unchanged salbutamol
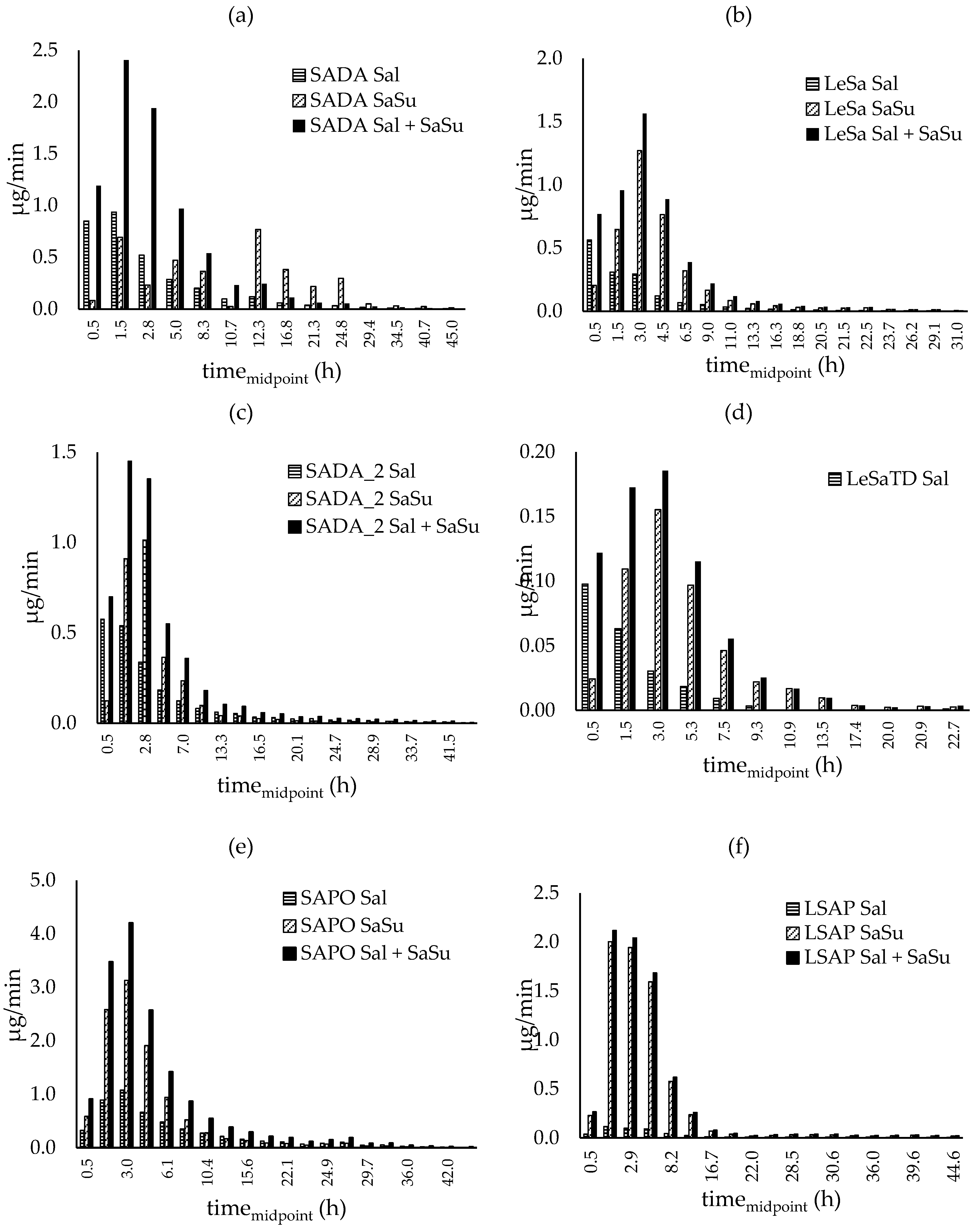
3.3.4. Elimination constant and elimination half-life
4. Discussion
4.1. Biosynthesis and characterization of salbutamol-4’-O-sulfate
4.2. Administration route and pharmakokinetic of salbutamol and its sulfo metabolite
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boulton, D.W.; Fawcett, J.P. The pharmacokinetics of levosalbutamol: what are the clinical implications? Clinical pharmacokinetics 2001, 40, 23-40. [CrossRef]
- Ko, K.; Kurogi, K.; Davidson, G.; Liu, M.Y.; Sakakibara, Y.; Suiko, M.; Liu, M.C. Sulfation of ractopamine and salbutamol by the human cytosolic sulfotransferases. J Biochem 2012, 152, 275-283. [CrossRef]
- Jacobson, G.A.; Raidal, S.; Robson, K.; Narkowicz, C.K.; Nichols, D.S.; Haydn Walters, E. Bronchopulmonary pharmacokinetics of (R)-salbutamol and (S)-salbutamol enantiomers in pulmonary epithelial lining fluid and lung tissue of horses. British journal of clinical pharmacology 2017, 83, 1436-1445. [CrossRef]
- Sun, Y.; Harps, L.C.; Bureik, M.; Parr, M.K. Human Sulfotransferase Assays With PAPS Production in situ. Frontiers in Molecular Biosciences 2022, 9. [CrossRef]
- Teubner, W. Charakterisierung von Sulfotransferasen im Gastrointestinaltrakt von Mensch und Ratte und Aktivierung von Promutagenen in V79- Zellen, die eine intestinale Form (1B1) des Menschen und der Ratte exprimieren. 2001.
- Gamage, N.; Barnett, A.; Hempel, N.; Duggleby, R.G.; Windmill, K.F.; Martin, J.L.; McManus, M.E. Human sulfotransferases and their role in chemical metabolism. Toxicol Sci 2006, 90, 5-22. [CrossRef]
- Riches, Z.; Stanley, E.L.; Bloomer, J.C.; Coughtrie, M.W.H. Quantitative Evaluation of the Expression and Activity of Five Major Sulfotransferases (SULTs) in Human Tissues: The SULT “Pie”. Drug Metabolism and Disposition 2009, 37, 2255. [CrossRef]
- Walle, T.; Eaton, E.A.; Walle, U.K.; Pesola, G.R. Stereoselective metabolism ofRS-albuterol in humans. Clinical Reviews in Allergy & Immunology 1996, 14, 101-113. [CrossRef]
- Boulton, D.W.; Fawcett, J.P. Pharmacokinetics and pharmacodynamics of single oral doses of albuterol and its enantiomers in humans. Clin Pharmacol Ther 1997, 62, 138-144. [CrossRef]
- Mareck, U.; Guddat, S.; Schwenke, A.; Beuck, S.; Geyer, H.; Flenker, U.; Elers, J.; Backer, V.; Thevis, M.; Schänzer, W. Determination of salbutamol and salbutamol glucuronide in human urine by means of liquid chromatography-tandem mass spectrometry. Drug Test Anal 2011, 3, 820-827. [CrossRef]
- Boulton, D.W.; Fawcett, J.P. Enantioselective disposition of salbutamol in man following oral and intravenous administration. British journal of clinical pharmacology 1996, 41, 35-40. [CrossRef]
- Ward, J.K.; Dow, J.; Dallow, N.; Eynott, P.; Milleri, S.; Ventresca, G.P. Enantiomeric disposition of inhaled, intravenous and oral racemic-salbutamol in man--no evidence of enantioselective lung metabolism. British journal of clinical pharmacology 2000, 49, 15-22. [CrossRef]
- Melchor, R.; Biddiscombe, M.F.; Mak, V.H.; Short, M.D.; Spiro, S.G. Lung deposition patterns of directly labelled salbutamol in normal subjects and in patients with reversible airflow obstruction. Thorax 1993, 48, 506-511. [CrossRef]
- Nakpheng, T.; Songkarak, S.; Suwandecha, T.; Sritharadol, R.; Chunhachaichana, C.; Srichana, T. Evidences for salbutamol metabolism by respiratory and liver cell lines. Drug metabolism and pharmacokinetics 2017, 32, 127-134. [CrossRef]
- Morgan, D.J.; Paull, J.D.; Richmond, B.H.; Wilson-Evered, E.; Ziccone, S.P. Pharmacokinetics of intravenous and oral salbutamol and its sulphate conjugate. Br J Clin Pharmacol 1986, 22, 587-593. [CrossRef]
- Sun, Y.; Machalz, D.; Wolber, G.; Parr, M.K.; Bureik, M. Functional Expression of All Human Sulfotransferases in Fission Yeast, Assay Development, and Structural Models for Isoforms SULT4A1 and SULT6B1. Biomolecules 2020, 10, 1517. [CrossRef]
- Orlovius, A.-K.L. Sulfokonjugierte Sypathomimetika in der Dopinganalytik: Synthese, Charakterisierung und Analyse.; Rheinische Friedrich-Wilhelms-Universität Bonn: https://hdl.handle.net/20.500.11811/6086, 2014.
- Purchartová, K.; Valentová, K.; Pelantová, H.; Marhol, P.; Cvačka, J.; Havlíček, L.; Křenková, A.; Vavříková, E.; Biedermann, D.; Chambers, C.S.; et al. Prokaryotic and Eukaryotic Aryl Sulfotransferases: Sulfation of Quercetin and Its Derivatives. ChemCatChem 2015, 7, 3152-3162. [CrossRef]
- Horst, M.; Hartog, A.; Morabet, R.; Marais, A.; Kircz, M.; Wever, R. Enzymatic Sulfation of Phenolic Hydroxy Groups of Various Plant Metabolites by an Arylsulfotransferase. European Journal of Organic Chemistry 2015, 2015. [CrossRef]
- Ragan, M.A. Phenol sulfate esters: ultraviolet, infrared, 1H and 13C nuclear magnetic resonance spectroscopic investigation. Canadian Journal of Chemistry 1978, 56, 2681-2685. [CrossRef]
- Nishikawa, M.; Masuyama, Y.; Nunome, M.; Yasuda, K.; Sakaki, T.; Ikushiro, S. Whole-cell-dependent biosynthesis of sulfo-conjugate using human sulfotransferase expressing budding yeast. Applied Microbiology and Biotechnology 2018, 102, 723-732. [CrossRef]
| Salbutamola) | Salbutamol-4’-O-sulfatea) |
Chemical shift differences Δδ =δOsulfate –δOH |
||||
| position | 1H | 13C | 1H | 13C | 1H | 13C |
| 1 | 4.72, ddb), 10.0, 2.8 Hz | 69.71 | 4.77, ddb), 10.2, 2.6 Hz | 69.69 | +0.05 | –0.02 |
| 2 | 2.74/2.83, AB db) 11.8, 10.0, 2.8 Hz | 49.14 | 2.69/2.81c) | 49.67 | –0.05/–0.02 | +0.53 |
| 4 | 53.89 | 54.68d) | +0.79 | |||
| 5 | 1.20, s | 26.12 | 1.23, s | 25.91 | +0.03 | –0.21 |
| 1’ | 132.93 | 137.46 | +4.53 | |||
| 2’ | 7.31, d, 2.3 Hz | 125.88 | 7.43, d, 2.1 Hz | 124.86 | +0.12 | –1.02 |
| 3’ | 128.10 | 134.57 | +6.47 | |||
| 3’-CH2 | 4.47, s | 58.24 | 4.55, s | 58.30 | +0.08 | +0.06 |
| 4’ | 153.43 | 149.59 | –3.84 | |||
| 5’ | 6.73, d, 8.3 Hz | 114.16 | 7.27. d, 8.3 Hz | 121.46 | +0.54 | +7.30 |
| 6’ | 7.07, dd, 8.3, 2.3 Hz | 124.96 | 7.20, dd 8.3, 2.1 Hz | 121.50 | +0.13 | –3.46 |
| DPI1 | MDI2 | MDI2_2 | |
| Percentage of dose recovered in urine3 | 80 % | 115 % | 83 % |
| Proportion of parent compound4 | 42 % | 46 % | 41 % |
| Proportion of salbutamol-4’-O-sulfate4 | 58 % | 54 % | 59 % |
| salbutamol | salbutamol-4’-O-sulfate | |
| tmax urine | ||
| Oral racemate (SAPO – 2 mg) | 3 h | 3 h |
| Inhaled aerosol racemate (SADA – 600 µg) | 1.5 h | 1.5 h |
| Oral levosalbutamol (LSAP – 1 mg) | 3 h | 0.5 h |
| Inhaled levosalbutamol (LeSa – 630 µg) Inhaled levosalbutamol (LeSaTD – 90 µg) |
0.5 h 0.5 h |
3 h 3 h |
| total urinary excretion | ||
| Oral racemate (SAPO – 2 mg)1 | 449 µg | 1030 µg |
| Inhaled aerosol racemate (SADA – 600 µg)1 | 318 µg | 372 µg |
| Oral levosalbutamol (LSAP – 1 mg)1 | 65 µg | 847 µg |
| Inhaled levosalbutamol (LeSa – 630 µg)1 Inhaled levosalbutamol (LeSaTD – 90 µg)1 |
129 µg 19 µg |
371 µg 55 µg |
| t½ (urine) | salbutamol | salbutamol-4’-O-sulfate |
| Oral racemate (SAPO – 2 mg) | 5.6 h | 4.7 h |
| Inhaled aerosol racemate (SADA – 600 µg) | 5.0 h | 4.4 h |
| Inhaled aerosol racemate (SADA_2 – 600 µg) | 6.9 h | 6.5 h |
| Oral levosalbutamol (LSAP – 1 mg) | 8.9 h | 6.9 h |
| Inhaled levosalbutamol (LeSa – 630 µg) Inhaled levosalbutamol (LeSaTD – 90 µg) |
3.8 h 3.3 h |
3.7 h 4.2 h |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




