Submitted:
24 December 2022
Posted:
03 January 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Generation of conditional NEDD9 knock IN (Floxed-STOP-NEDD9) transgenic strain and locus insertion.
2.2. Generation of FVB/J-MMTV-Cre-NEDD9+/+ mice for mammary gland specific upregulation of NEDD9.
2.3. Generation of FVB/J-MMTV-Cre-NEDD9+/+ mice for mammary gland-specific upregulation of NEDD9.
2.4. PCR screening strategy for the genotyping of the homozygous NEDD9fx/fx knock-in mice.
2.5. PCR screening strategy for the genotyping of the induced NEDD9 knock-in line (with removed transcription stop cassette).
2.6. PCR screening strategy for the genotyping of the homozygous NEDD9/Cre/Erbb2 mice.
2.7. Generation of Mouse Embryonic Fibroblasts with NEDD9 knock-in.
2.8. Quantification of Branching Density in Mammary Gland Whole-Mounts.
2.9. Cell Culture, Plasmids, and other Reagents.
2.10. Lentivirus constructs, Cell Infection and Transfection Reagents.
2.11. Lentivirus constructs, Cell Infection and Transfection Reagents.
2.12. Tumor Micro Array and Patient Data.
2.13. Immunohistochemistry and Scoring Procedures.
2.14. Fluorescent immunohistochemistry and Hematoxylin & Eosin (H&E) staining.
2.15. Histopathology Evaluation and Grading.
2.16. Western blotting
2.17. MTT-based Proliferation and Viability Assay.
2.18. Drug Treatment and Viability Assay.
2.19. Acini Formation, Imaging and Quantification Procedures.
2.20. Kaplan–Meier Analysis.
2.21. Statistical analysis.
3. Results
3.1. NEDD9 expression correlates with disease progression, outcomes, and HER2+ pathological score.

3.2. Generation of conditional NEDD9 knock-in (KI) transgenic mouse model.

3.3. Production and analysis of mammary gland-specific expression of NEDD9.
3.4. NEDD9 overexpression alters mammary gland architecture by increasing mammary gland budding and branching morphogenesis and cooperates with HER2.
3.5. Cooperation between NEDD9 and HER2 promotes the early initiation of tumorigenesis.
3.6. NEDD9 overexpression is associated with mammary intra-epithelia neoplasia.
3.7. NEDD9 overexpression causes hyperproliferation of luminal cells.
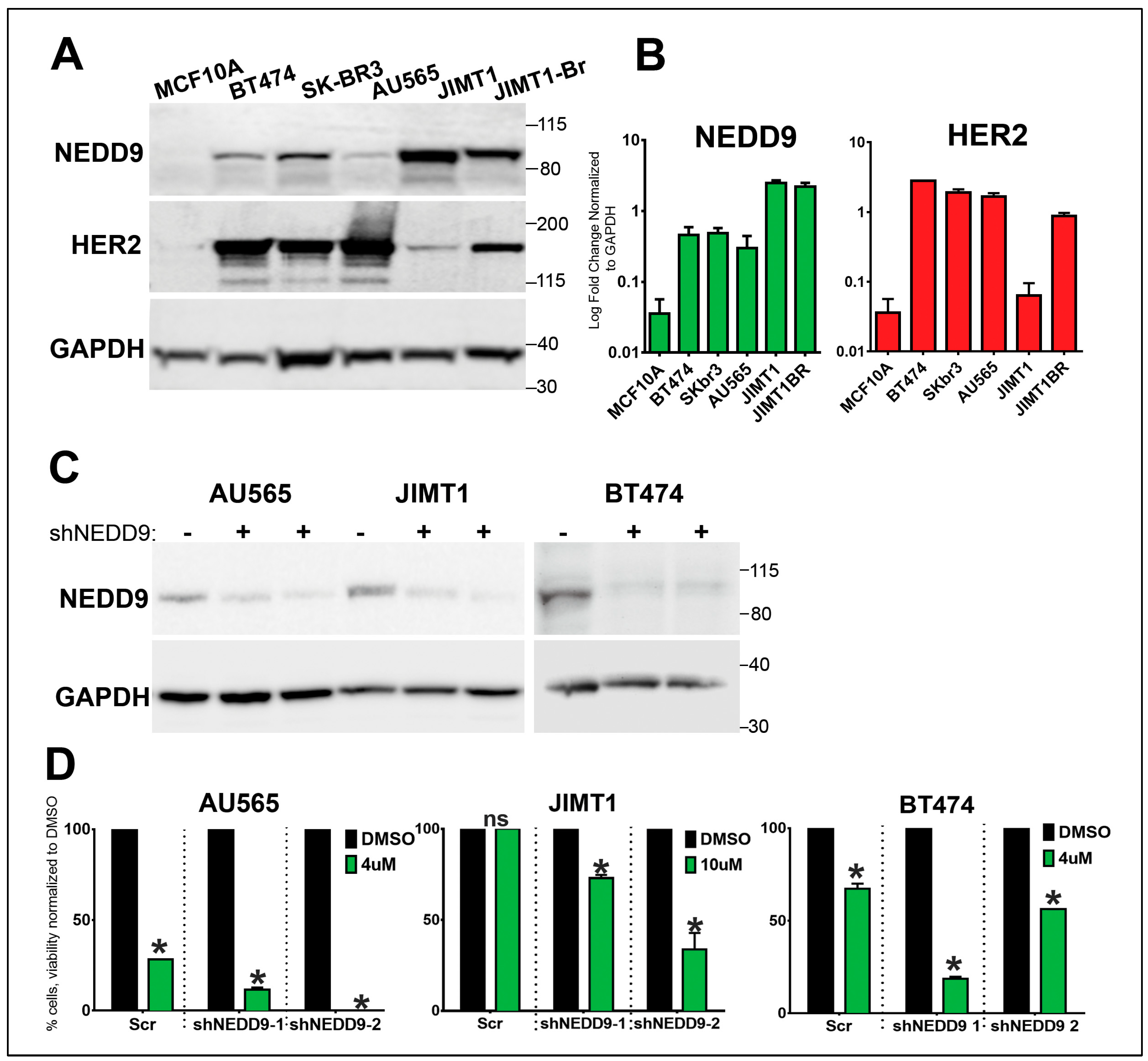
3.8. NEDD9 is overexpressed in human HER2+ breast cancer cell lines.
3.9. NEDD9 overexpression in normal mammary epithelial cells causes increased proliferation.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cancer Facts & Figures 2022. 2022, American Cancer Society: Atlanta.
- Giaquinto, A.N., et al., Breast Cancer Statistics, 2022. Ca-a Cancer Journal for Clinicians, 2022. 72(6): p. 524-541. [CrossRef]
- Loibl, S. and L. Gianni, HER2-positive breast cancer. Lancet, 2017. 389(10087): p. 2415-2429. [CrossRef]
- Arteaga, C.L., et al., Treatment of HER2-positive breast cancer: current status and future perspectives. Nature Reviews. Clinical Oncology, 2012. 9(1): p. 16-32. [CrossRef]
- Muthuswamy, S.K., Trastuzumab resistance: all roads lead to SRC. Nature Medicine, 2011. 17: p. 416. [CrossRef]
- Peiro, G., et al., Src, a potential target for overcoming trastuzumab resistance in HER2-positive breast carcinoma. (1532-1827 (Electronic)). [CrossRef]
- Belsches-Jablonski, A.P., et al., Src family kinases and HER2 interactions in human breast cancer cell growth and survival. Oncogene, 2001. 20: p. 1465. [CrossRef]
- Shagisultanova, E., et al., Preclinical and clinical studies of the NEDD9 scaffold protein in cancer and other diseases. Gene, 2015. 567(1): p. 1-11. [CrossRef]
- Ahn, J., V. Sanz-Moreno, and C.J. Marshall, The metastasis gene NEDD9 product acts through integrin β3 and Src to promote mesenchymal motility and inhibit amoeboid motility. Journal of Cell Science, 2012. 125(7): p. 1814. [CrossRef]
- Muller, W.J., et al., Synergistic interaction of the Neu proto-oncogene product and transforming growth factor alpha in the mammary epithelium of transgenic mice. Molecular and Cellular Biology, 1996. 16(10): p. 5726-5736. [CrossRef]
- Hsu, J.L. and M.-C. Hung, The role of HER2, EGFR, and other receptor tyrosine kinases in breast cancer. Cancer metastasis reviews, 2016. 35(4): p. 575-588. [CrossRef]
- Sima, N., et al., The Overexpression of Scaffolding Protein NEDD9 Promotes Migration and Invasion in Cervical Cancer via Tyrosine Phosphorylated FAK and SRC. PLOS ONE, 2013. 8(9): p. e74594. [CrossRef]
- Izumchenko, E., et al., NEDD9 Promotes Oncogenic Signaling in Mammary Tumor Development. Cancer Research, 2009. 69(18): p. 7198-7206. [CrossRef]
- Pugacheva, E.N., et al., HEF1-dependent Aurora A activation induces disassembly of the primary cilium. Cell, 2007. 129(7): p. 1351-1363. [CrossRef]
- Cabodi, S., et al., p130Cas as a New Regulator of Mammary Epithelial Cell Proliferation, Survival, and HER2-Neu Oncogene–Dependent Breast Tumorigenesis. Cancer Research, 2006. 66(9): p. 4672-4680. [CrossRef]
- Little, J.L., et al., A requirement for Nedd9 in luminal progenitor cells prior to mammary tumorigenesis in MMTV-HER2/ErbB2 mice. Oncogene, 2013. 33: p. 411. [CrossRef]
- Dai, J., et al., Downregulation of NEDD9 by apigenin suppresses migration, invasion, and metastasis of colorectal cancer cells. (1096-0333 (Electronic)). [CrossRef]
- Li, Y., et al., HEF1, a novel target of Wnt signaling, promotes colonic cell migration and cancer progression. Oncogene, 2011. 30: p. 2633. [CrossRef]
- Speranza, M.C., et al., NEDD9, a novel target of miR-145, increases the invasiveness of glioblastoma. Oncotarget, 2012. 3(7): p. 723-34. [CrossRef]
- Wang, H., et al., NEDD9 overexpression is associated with the progression of and an unfavorable prognosis in epithelial ovarian cancer. Human Pathology, 2014. 45(2): p. 401-408. [CrossRef]
- Li, G., et al., Conditional loss of PTEN leads to precocious development and neoplasia in the mammary gland. Development, 2002. 129(17): p. 4159-4170. [CrossRef]
- Zhong, J., et al., NEDD9 stabilizes focal adhesions, increases binding to the extra-cellular matrix and differentially effects 2D versus 3D cell migration. PloS one, 2012. 7(4): p. e35058-e35058. [CrossRef]
- Jozefczuk, J., K. Drews, and J. Adjaye, Preparation of Mouse Embryonic Fibroblast Cells Suitable for Culturing Human Embryonic and Induced Pluripotent Stem Cells. Jove-Journal of Visualized Experiments, 2012(64). [CrossRef]
- Ice, R.J., et al., NEDD9 Depletion Destabilizes Aurora A Kinase and Heightens the Efficacy of Aurora A Inhibitors: Implications for Treatment of Metastatic Solid Tumors. Cancer Research, 2013. 73(10): p. 3168-3180. [CrossRef]
- Stanko, J.P. and S.E. Fenton, Quantifying Branching Density in Rat Mammary Gland Whole-mounts Using the Sholl Analysis Method. J Vis Exp, 2017(125). [CrossRef]
- Stanko, J.P. and S.E. Fenton, Quantifying Branching Density in Rat Mammary Gland Whole-mounts Using the Sholl Analysis Method. Journal of visualized experiments : JoVE, 2017(125): p. 55789. [CrossRef]
- Lyle, L.T., et al., Alterations in Pericyte Subpopulations Are Associated with Elevated Blood–Tumor Barrier Permeability in Experimental Brain Metastasis of Breast Cancer. Clinical Cancer Research, 2016. 22(21): p. 5287-5299. [CrossRef]
- Zhou, X. and Y.M. Agazie, Molecular mechanism for SHP2 in promoting HER2-induced signaling and transformation. The Journal of biological chemistry, 2009. 284(18): p. 12226-12234. [CrossRef]
- Addison, J.B., et al., Functional Hierarchy and Cooperation of EMT Master Transcription Factors in Breast Cancer Metastasis. Molecular Cancer Research, 2021. 19(5): p. 784-798. [CrossRef]
- Tiscornia, G., O. Singer, and I.M. Verma, Production and purification of lentiviral vectors. Nat Protoc, 2006. 1(1): p. 241-5. [CrossRef]
- Whately, K.M., et al., Nuclear Aurora-A kinase-induced hypoxia signaling drives early dissemination and metastasis in breast cancer: implications for detection of metastatic tumors. Oncogene, 2021. 40(37): p. 5651-5664. [CrossRef]
- Lánczky, A. and B. Győrffy, Web-Based Survival Analysis Tool Tailored for Medical Research (KMplot): Development and Implementation. J Med Internet Res, 2021. 23(7): p. e27633. [CrossRef]
- Pugacheva, E.N. and E.A. Golemis, The focal adhesion scaffolding protein HEF1 regulates activation of the Aurora-A and Nek2 kinases at the centrosome. Nature Cell Biology, 2005. 7(10): p. 937-U18. [CrossRef]
- Whately, K.M., et al., Nuclear Aurora-A kinase-induced hypoxia signaling drives early dissemination and metastasis in breast cancer: implications for detection of metastatic tumors. Oncogene, 2021. 40(37): p. 5651-5664. [CrossRef]
- Brulet, P., et al., Monoclonal antibodies against trophectoderm-specific markers during mouse blastocyst formation. Proc Natl Acad Sci U S A, 1980. 77(7): p. 4113-7. [CrossRef]
- Wu, M., et al., Gli Transcription Factors Mediate the Oncogenic Transformation of Prostate Basal Cells Induced by a Kras-Androgen Receptor Axis. J Biol Chem, 2016. 291(49): p. 25749-25760. [CrossRef]
- Cardiff, R.D., et al., The mammary pathology of genetically engineered mice: the consensus report and recommendations from the Annapolis meeting. Oncogene, 2000. 19(8): p. 968-988. [CrossRef]
- Cardiff, R.D., D. Moghanaki, and R.A. Jensen, Genetically Engineered Mouse Models of Mammary Intraepithelial Neoplasia. Journal of Mammary Gland Biology and Neoplasia, 2000. 5(4): p. 421-437. [CrossRef]
- Jones, B.C., et al., Dual Targeting of Mesenchymal and Amoeboid Motility Hinders Metastatic Behavior. Mol Cancer Res, 2017. 15(6): p. 670-682. [CrossRef]
- Muthuswamy, S.K., et al., ErbB2, but not ErbB1, reinitiates proliferation and induces luminal repopulation in epithelial acini. Nature Cell Biology, 2001. 3(9): p. 785-792. [CrossRef]
- Györffy, B., et al., An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Research and Treatment, 2010. 123(3): p. 725-731. [CrossRef]
- Mihály, Z. and B. Győrffy Improving Pathological Assessment of Breast Cancer by Employing Array-Based Transcriptome Analysis. Microarrays, 2013. 2, 228-242. [CrossRef]
- Guy, C.T., et al., Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proc Natl Acad Sci U S A, 1992. 89(22): p. 10578-82. [CrossRef]
- Andrechek, E.R., et al., Amplification of the neu/erbB-2 oncogene in a mouse model of mammary tumorigenesis. Proceedings of the National Academy of Sciences of the United States of America, 2000. 97(7): p. 3444-3449. [CrossRef]
- Williams, J.M. and C.W. Daniel, Mammary ductal elongation: differentiation of myoepithelium and basal lamina during branching morphogenesis. Dev Biol, 1983. 97(2): p. 274-90. [CrossRef]
- Macias, H. and L. Hinck, Mammary gland development. Wiley Interdiscip Rev Dev Biol, 2012. 1(4): p. 533-57. [CrossRef]
- O'Brien, N.A., et al., Activated phosphoinositide 3-kinase/AKT signaling confers Resistance to trastuzumab but not lapatinib. Mol Cancer Ther, 2010. 9(6): p. 1489-502. [CrossRef]
- Pugacheva, E.N. and E.A. Golemis, The focal adhesion scaffolding protein HEF1 regulates activation of the Aurora-A and Nek2 kinases at the centrosome. Nature Cell Biology, 2005. 7(10): p. 937-946. [CrossRef]
- Fragomeni, S.M., A. Sciallis, and J.S. Jeruss, Molecular Subtypes and Local-Regional Control of Breast Cancer. Surgical oncology clinics of North America, 2018. 27(1): p. 95-120. [CrossRef]
- Berger, M.B., J.M. Mendrola, and M.A. Lemmon, ErbB3/HER3 does not homodimerize upon neuregulin binding at the cell surface. FEBS Letters, 2004. 569(1-3): p. 332-336. [CrossRef]
- Bazley, L.A. and W.J. Gullick, The epidermal growth factor receptor family. Endocrine-Related Cancer Endocr Relat Cancer, 2005. 12(Supplement_1): p. S17-S27. [CrossRef]
- Beigbeder, A., F.J.M. Chartier, and N. Bisson, MPZL1 forms a signalling complex with GRB2 adaptor and PTPN11 phosphatase in HER2-positive breast cancer cells. Scientific Reports, 2017. 7(1): p. 11514. [CrossRef]
- Little, J.L., et al., A requirement for Nedd9 in luminal progenitor cells prior to mammary tumorigenesis in MMTV-HER2/ErbB2 mice. Oncogene, 2014. 33(4): p. 411-20. [CrossRef]
- Jin, Y., et al., NEDD9 promotes lung cancer metastasis through epithelial–mesenchymal transition. International Journal of Cancer, 2014. 134(10): p. 2294-2304. [CrossRef]
- Speranza, M.C., et al., NEDD9, a novel target of miR-145, increases the invasiveness of glioblastoma. Oncotarget; Vol 3, No 7: July 2012, 2012. [CrossRef]
- Li, Y., et al., HEF1, a novel target of Wnt signaling, promotes colonic cell migration and cancer progression. Oncogene, 2011. 30(23): p. 2633-43. [CrossRef]
- Iida, J., et al., Role for chondroitin sulfate glycosaminoglycan in NEDD9-mediated breast cancer cell growth. Experimental Cell Research, 2015. 330(2): p. 358-370. [CrossRef]
- Guy, C.T., et al., Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proceedings of the National Academy of Sciences, 1992. 89(22): p. 10578-10582. [CrossRef]
- Zelazny, E., et al., Cooperating Oncogenic Events in Murine Mammary Tumorigenesis: Assessment of ErbB2, Mutant p53, and Mouse Mammary Tumor Virus. Experimental and Molecular Pathology, 2001. 70(3): p. 183-193. [CrossRef]
- Williams, J.M. and C.W. Daniel, Mammary ductal elongation: Differentiation of myoepithelium and basal lamina during branching morphogenesis. Developmental Biology, 1983. 97(2): p. 274-290. [CrossRef]
- Tortora, G., Mechanisms of Resistance to HER2 Target Therapy. JNCI Monographs, 2011. 2011(43): p. 95-98. [CrossRef]
- Mihály, Z., et al., A meta-analysis of gene expression-based biomarkers predicting outcome after tamoxifen treatment in breast cancer. Breast Cancer Research and Treatment, 2013. 140(2): p. 219-232. [CrossRef]
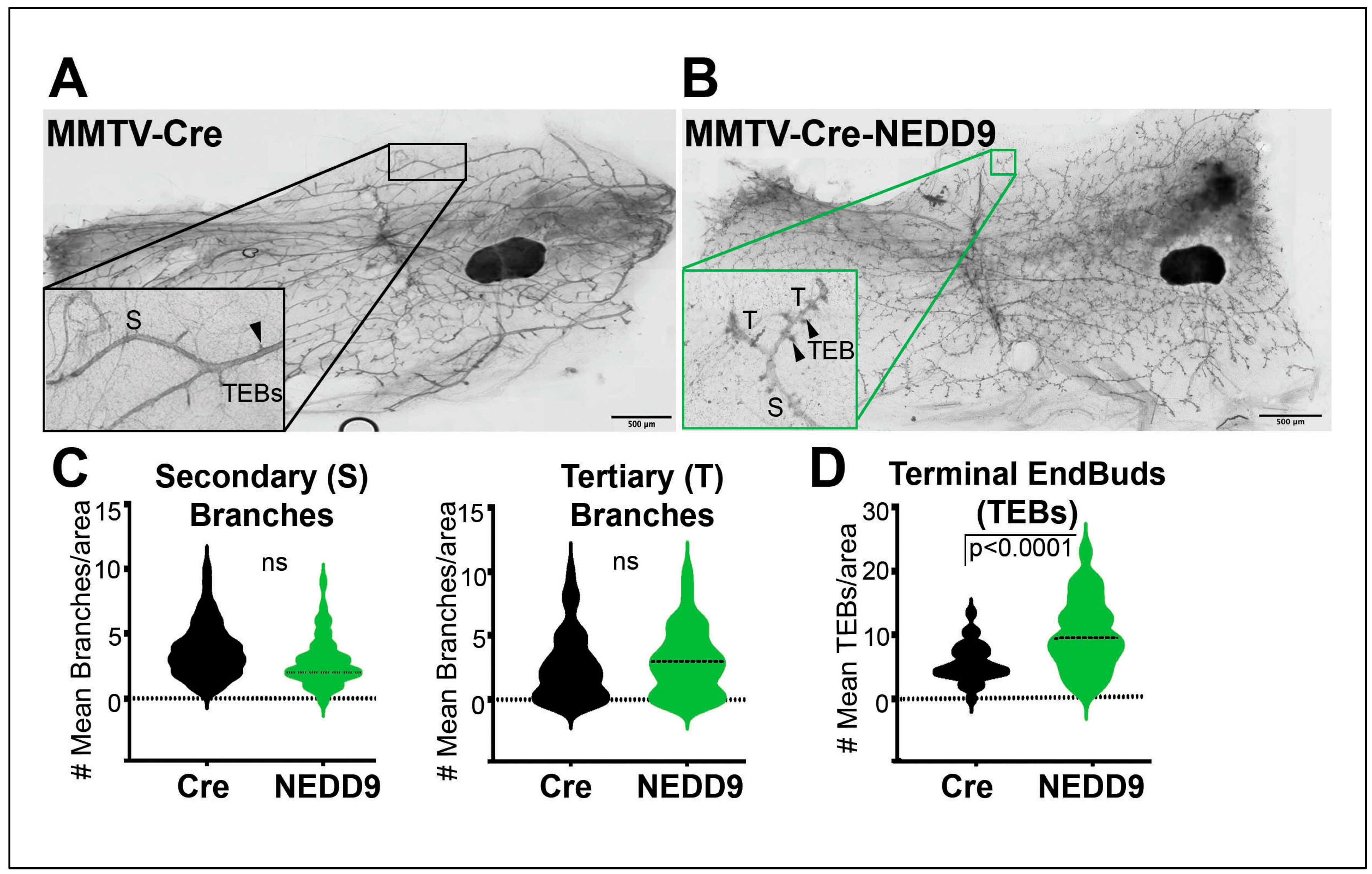
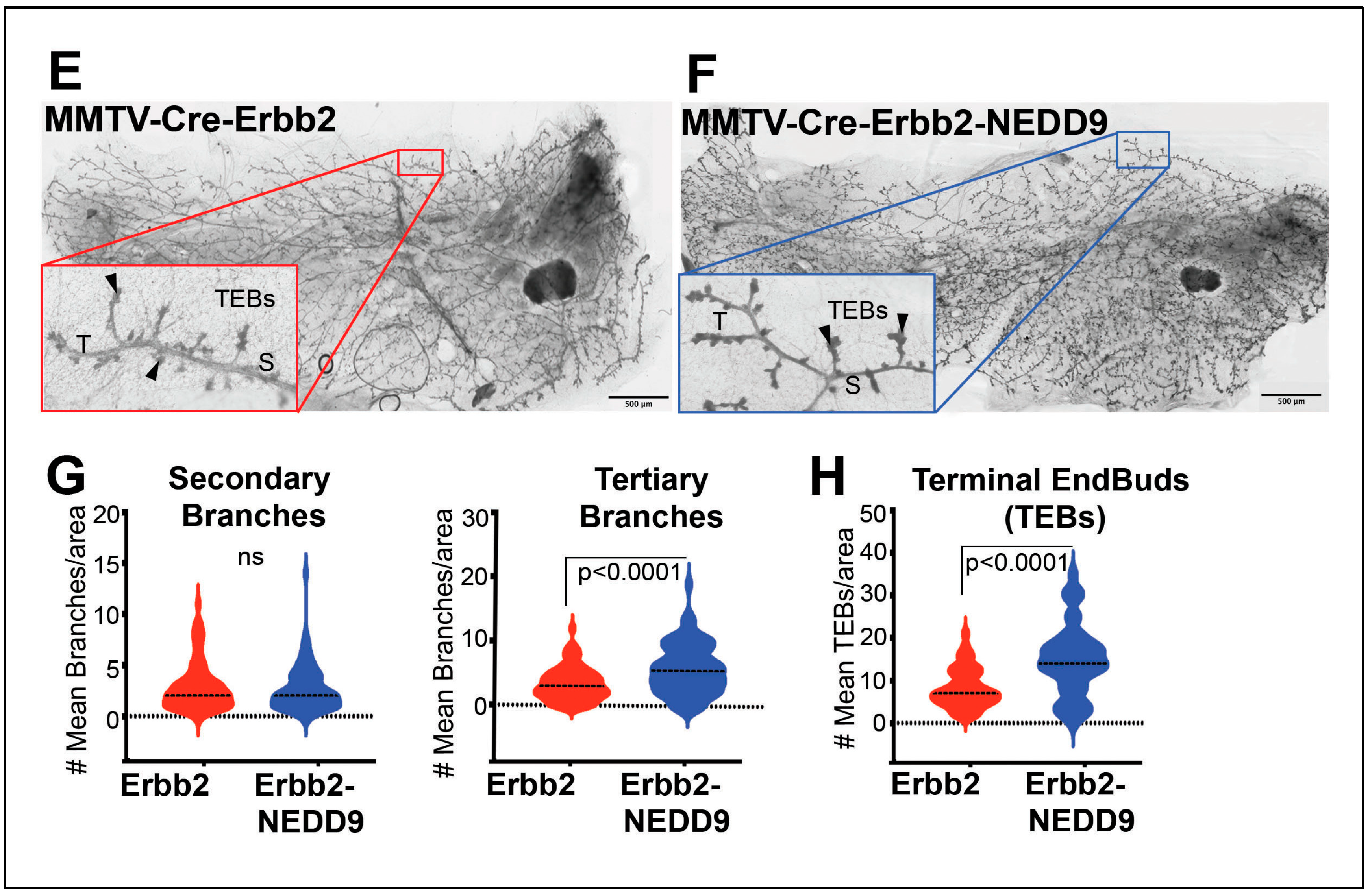
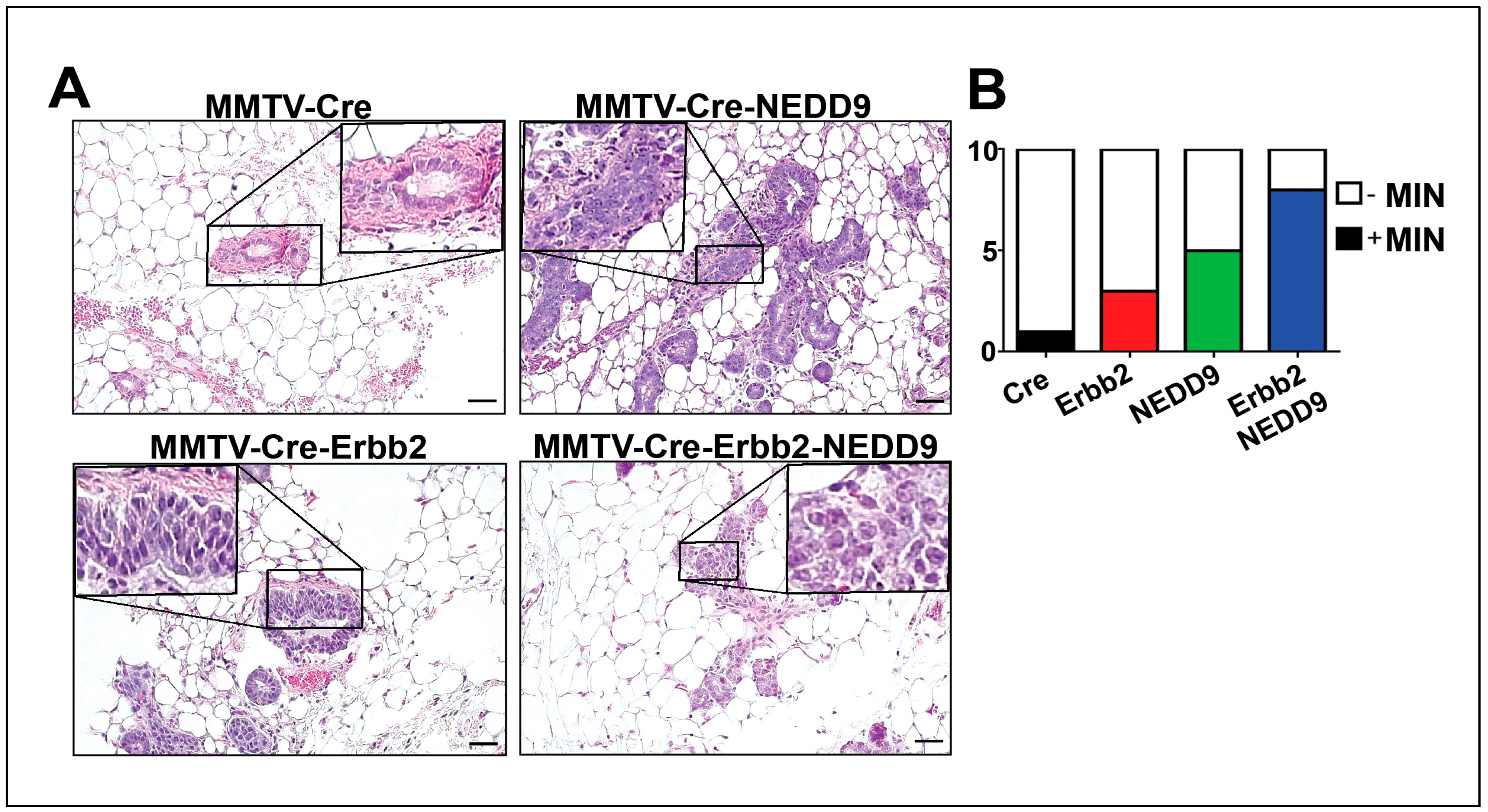


| Genotype | MIN+, N (%) | MIN-, N(%) |
|---|---|---|
| MMTV-Cre | 1 (10%) | 9 (90%) |
| MMTV-Cre-Erbb2 | 3 (30%) | 7 (70%) |
| MMTV-Cre-NEDD9 | 5 (50%) | 5 (50%) |
| MMTV-Cre-Erbb2-NEDD9 | 8 (80%) | 2 (20%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




