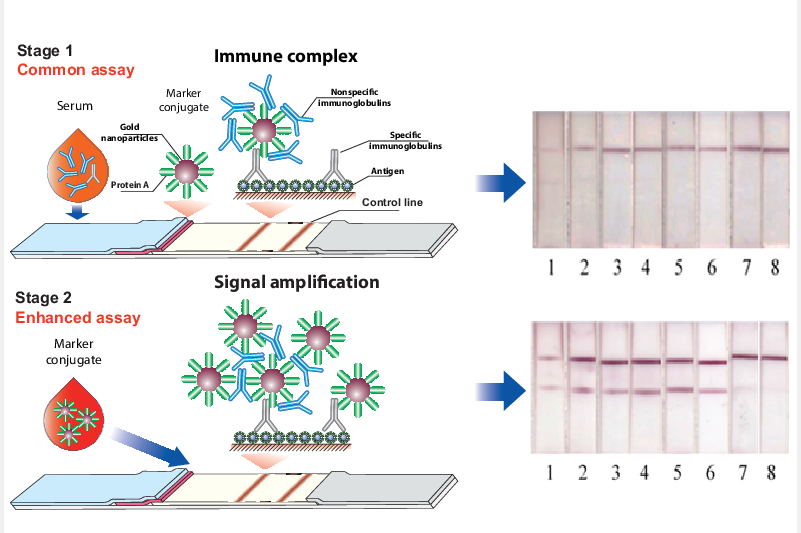
Nowadays, the presence of pathogen-specific antibodies in the blood is widely controlled by a serodiagnostic technique based on the lateral flow immunoassay (LFIA). However, its common one-stage format with an antigen immobilized in the binding zone of a test strip and a nanodis-persed label conjugated with immunoglobulin-binding proteins is associated with risks of very low analytical signals. It is caused by the presence of non-specific immunoglobulins in very large excess to the target antibodies in the tested samples thus decreasing their binding with the detected labels. In this study, the first stage of the immunochromatographic serodiagnosis was carried out in its traditional format using a conjugate of gold nanoparticles with staphylococcal immunoglobulin-binding protein A and an antigen immobilized on a working membrane. At the second stage, a labeled immunoglobulin-binding protein was added, which enhanced the coloration of the bound immune complexes. The use of two separated steps, binding of specific antibodies, and further coloration of the formed complexes allowed a significant reducing the influence of non-specific immunoglobulins on the assay results. The proposed approach was ap-plied for the serodiagnosis using a recombinant RBD protein of SARS-CoV-2. As a result, an in-crease in the intensity of test zone coloration by more than two orders of magnitude was demonstrated, which enabled to significantly reduce false-negative results. When testing a panel of 16 positive and 8 negative serum samples, the diagnostic sensitivity of the LFIA was 62.5% for the common format and 100% for the enhanced format; the diagnostic specificity of both variants was 100%.