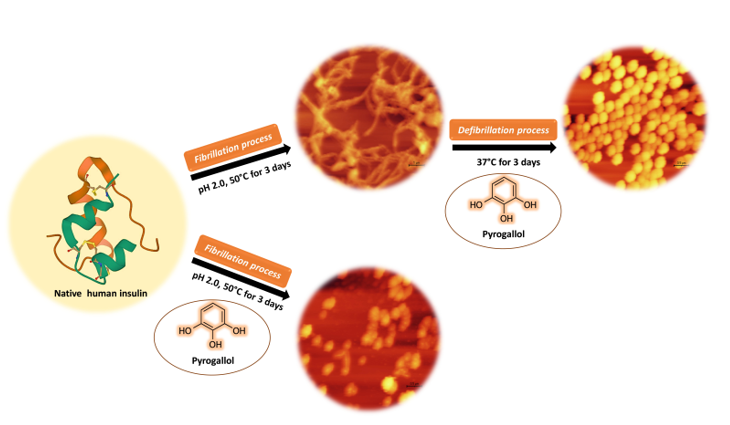
One of the major problems caused by repeated subcutaneous insulin injections in diabetic patients is insulin amyloidosis. Understanding the molecular mechanism of amyloid fibril formation of insulin and finding effective compounds to its inhibit or eliminate is very important and extensive research has been done on it. In this study, the anti-amyloidogenic and destabilizing effects of the pyrogallol, as a phenolic compound, on human insulin protein were investigated by CR absorbance, ThT and ANS fluorescence, FTIR spectroscopy and atomic force microscopy. According to the obtained results, the formation of amyloid fibrils at pH 2.0 and 50 °C was confirmed by CR, ThT, ANS, FTIR assays. Microscopic images also showed the twisted and long structures of amyloid fibrils. Simultaneous incubation of the protein with pyrogallol at different concentrations reduced the intensities of CR, ThT and ANS in a dose-dependent manner and no trace of fibrillar structures was observed in the microscopic images. FTIR spectroscopy also showed that the position of amide I band in the spectrum of samples containing pyrogallol was shifted. Based on the findings in this study, it can be concluded that pyrogallol can be effective in preventing and suppressing human insulin amyloid fibrils.