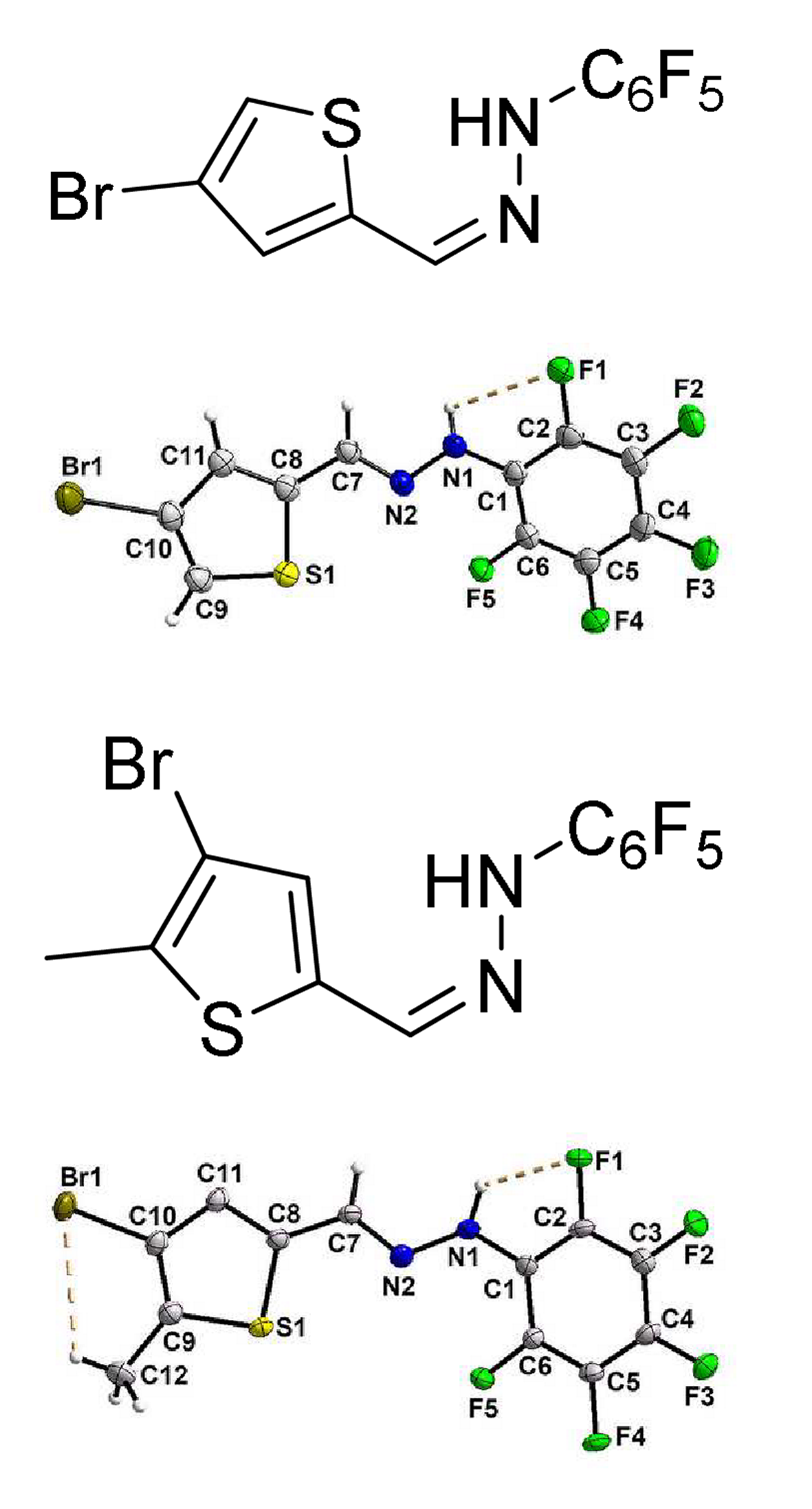
The crystals, C11H4BrF5N2S, (I), 1-((4-bromothiophen-2-yl)methylene)-2-(perfluorophenyl)hydrazine and C12H6BrF5N2S, (II), 1-((4-bromo-5-methylthiophen-2-yl)methylene)-2-(perfluorophenyl)hydrazine are molecules with two rings and hydrazone part like a centre of the molecule. The compounds have been synthesized and characterized by elemental, spectroscopic (1H-NMR) analysis. The crystal structures of the solid phase were determined by single crystal X-ray diffraction method. They crystallize in the monoclinic space group with Z = 4 and Z = 2 molecules per unit-cell. The compound (I) crystallizes as a racemate in the centrosymmetric space group and the compound (II) crystallizes as a non-racemate in the non-centrosymmetric space group. The “absolute configuration and conformation for bond values” were derived from the anomalous dispersion (ad) for (II). The crystal structures are revealed diverse non-covalent interactions such as intra- and interhydrogen bonding, π-ring···π-ring, C-H···π-ring and they were investigated. The expected stereochemistry of hydrazones atoms C7, N2 and N1 were confirmed for (I) and (II). The hole molecule of the (I), and (II) possesses “a boat conformation” like a 6-membered ring. The results of the single crystal studies are reproduced with the help of Hirshfeld surface study and Gaussian software.