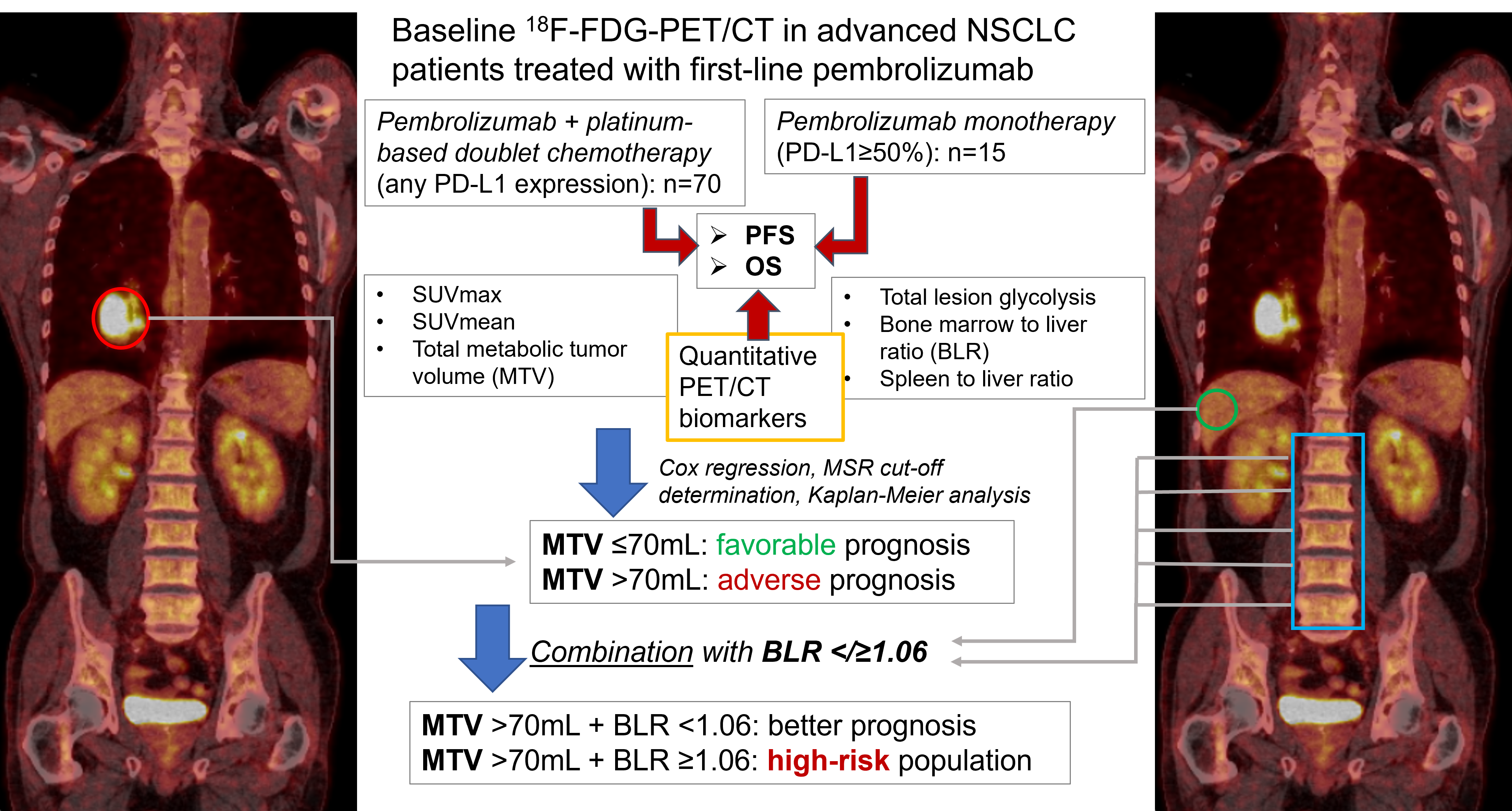
Quantitative biomarkers derived from positron-emission tomography/computed tomography (PET/CT) have been suggested as prognostic variables in immune-checkpoint inhibitor (ICI) treated non-small cell lung cancer (NSCLC). As such data for first-line ICI therapy and especially for chemotherapy-ICI combinations are still scarce, we retrospectively evaluated baseline 18F-FDG-PET/CT of 85 consecutive patients receiving first-line pembrolizumab with chemotherapy (n=70) or as monotherapy (n=15). Maximum and mean standardized uptake value, metabolic tumor volume (MTV), total lesion glycolysis and bone marrow-/ spleen to liver ratio (BLR/SLR) were calculated. Kaplan-Meier analyses and Cox-regression models were used to assess progression-free/overall survival (PFS/OS) and their determinant variables. Multivariate selection for PFS/OS revealed MTV as most relevant PET/CT biomarker (p<0.001). Median PFS/OS were significantly longer in patients with MTV≤70mL versus >70mL (PFS: 10 months (M; 95% confidence interval 4-16) vs. 4M (3-5), p=0.001; OS: not reached vs. 10M (5-15), p=0.004). Disease control rate was 81% vs. 53% for MTV≤/>70mL (p=0.007). BLR ≤1.06 versus >1.06 was associated with better outcomes (PFS: 8M (4-13) vs. 4M (3-6), p=0.034; OS: 19M (12-/) vs. 6M (4-12), p=0.005). In patients with MTV>70mL, concomitant BLR≤1.06 indicated a better prognosis. Higher MTV is associated with inferior PFS/OS in first-line ICI treated NSCLC, with BLR allowing additional risk stratification.