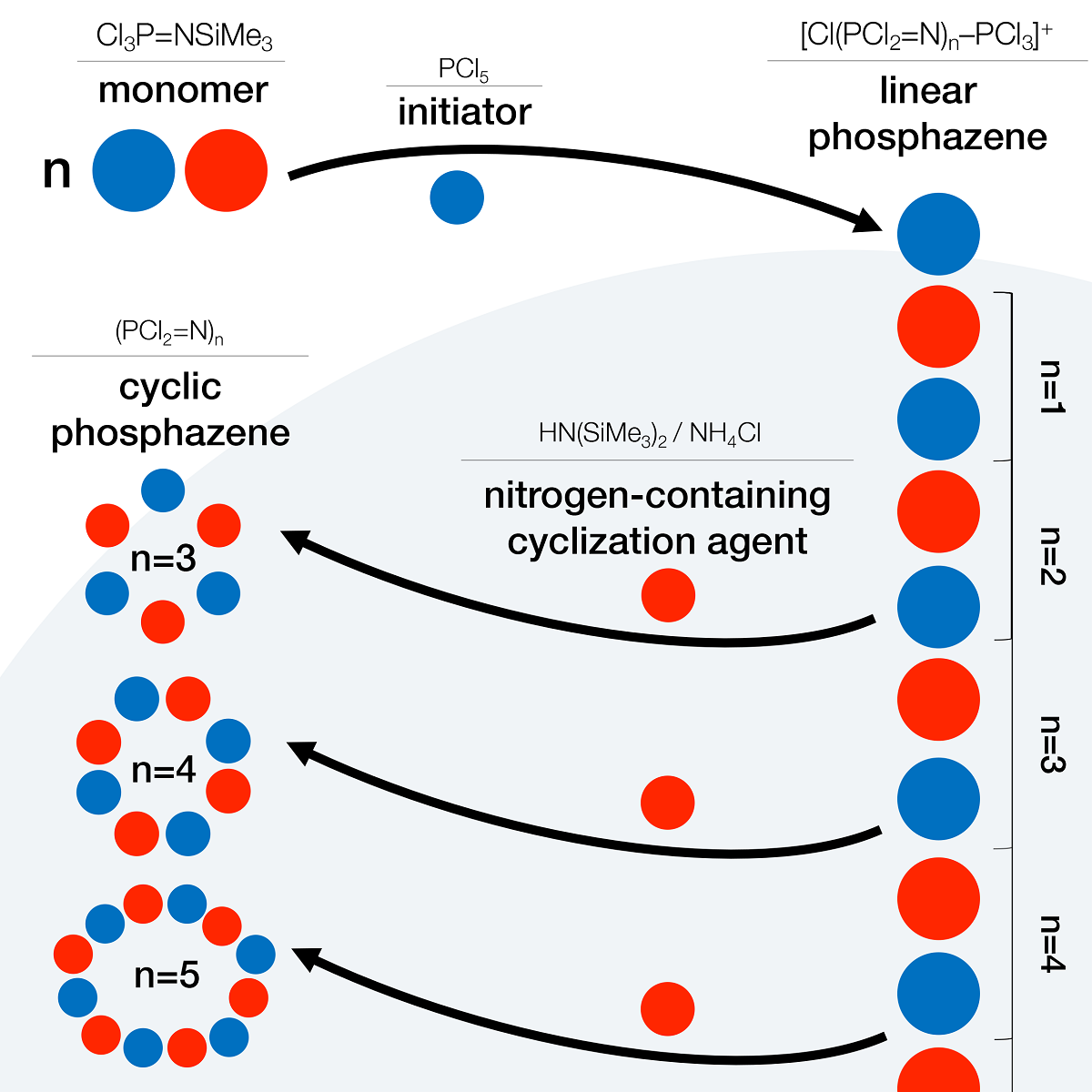
Despite a significant number of investigations in the field of phosphazene chemistry, the mechanism of this class cyclic compounds formation is still poorly studied. At the same time, a thorough understanding of this process is necessary both for the direct production of phosphazene rings with a given size, and for the controlled cyclization reaction when it is secondary and undesirable. Here we have synthesized a series of short linear phosphazene oligomers with the general formula Cl[PCl2=N]n–PCl3+PCl6– and studied their tendency to form cyclic structures under the influence of elevated temperature or in the presence of nitrogen-containing agents, such as hexamethyldisilazane (HMDS) or ammonium chloride. It was established that linear oligophosphazenes are inert when heated in the absence of the mentioned cyclization agents, and the formation of cyclic products occurs only when these agents are involved in the process. It is for the first time shown the ability to obtain the desired size phosphazene cycle from corresponding linear chain. Known obstacles like side interaction with the PCl6– counterion and a tendency of longer chains to undergo crosslinking elongation instead of cyclization are still relevant and ways to overcome them are being discussed.