Submitted:
24 December 2020
Posted:
25 December 2020
You are already at the latest version
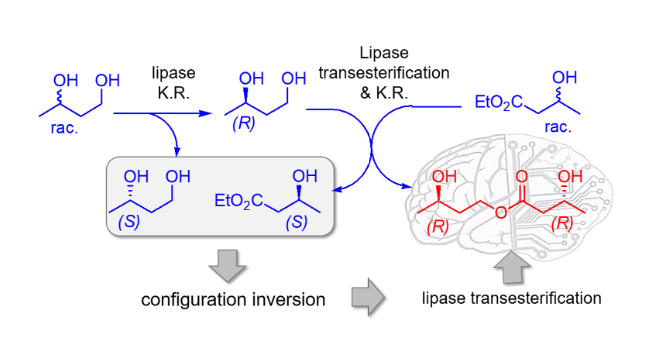
Abstract
Subscription
Notify me about updates to this article or when a peer-reviewed version is published.
This version is not peer-reviewed.
Submitted:
24 December 2020
Posted:
25 December 2020
You are already at the latest version

© 2026 MDPI (Basel, Switzerland) unless otherwise stated