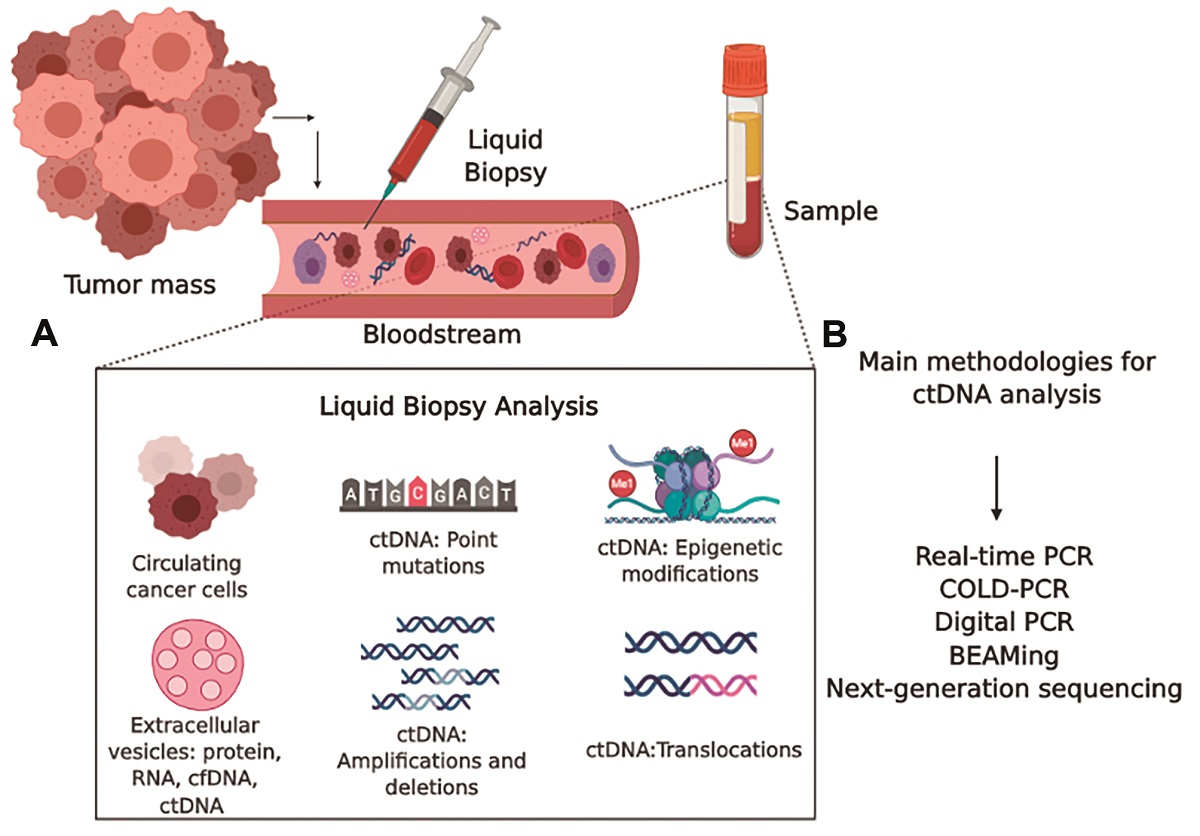
Circulating tumor DNA (ctDNA) in fluids has gained attention because ctDNA seems to identify tumor-specific abnormalities, which could be used for diagnosis, follow-up of treatment, and prognosis: the so-called liquid biopsy. Liquid biopsy is a minimally invasive approach and presents the sum of ctDNA from primary and secondary tumor sites. It has been possible not only to quantify the amount of ctDNA but also to identify (epi)genetic changes. Specific mutations in genes have been identified in the plasma of patients with several types of cancer, which highlights ctDNA as a possible cancer biomarker. However, achieving detectable concentrations of ctDNA in body fluids is not an easy task. ctDNA fragments present a short half-life, and there are no cut-off values to discriminate high and low ctDNA concentrations. Here, we discuss the use of ctDNA as a cancer biomarker, the main methodologies, the inherent difficulties, and the clinical predictive value of ctDNA.