Submitted:
21 March 2023
Posted:
21 March 2023
Read the latest preprint version here
Abstract
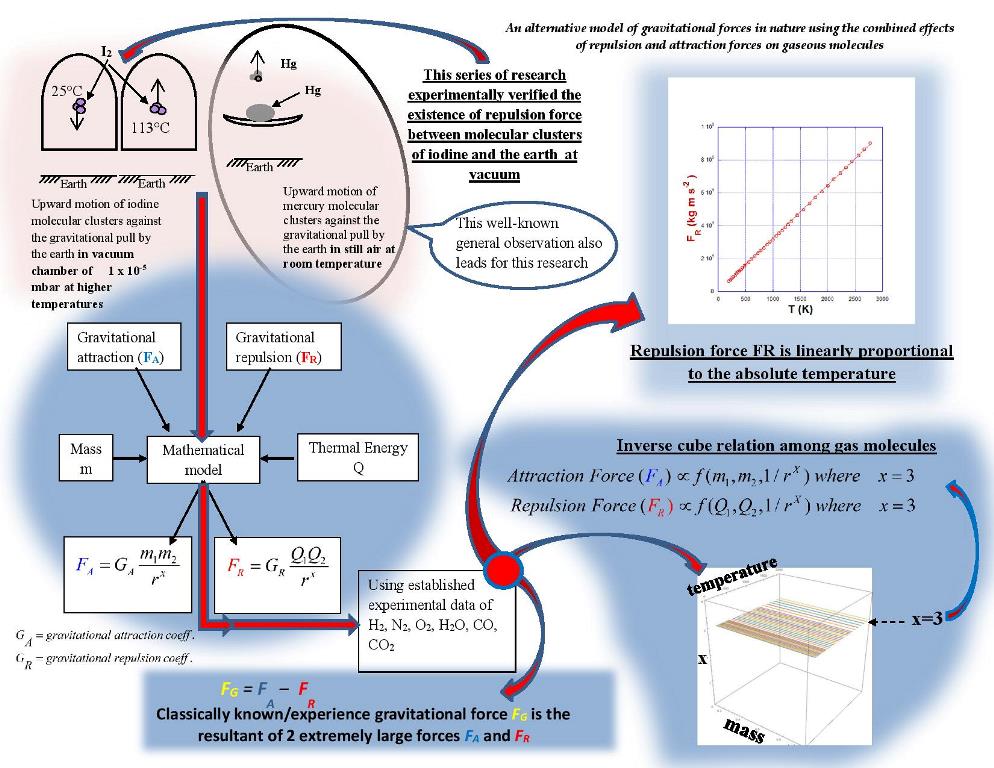
Keywords:
1. Introduction
- Newtonian and Einsteinian gravity concepts, highlighting foundations of our present understanding
- Early notions of the gravitational repulsion force and its recent revelations
- The challenges of a major assumption in the kinetic theory of ideal gas, used in the derivation of the ideal gas equation
1.1. Brief note on Newtonian and Einsteinian Gravity Concepts:
1.2. Early Notions of Gravitational Repulsion Force:
1.3. Recent Revelation of the Gravitational Repulsion Force:
1.4. Challenges of a Major Assumption in the Derivation of the Kinetic Theory of Ideal Gas:
The intermolecular force in gaseous state is zero and as such molecules exhibit no force among themselves
1.5. Behavior of Real gas – the Van Der Waals Equation
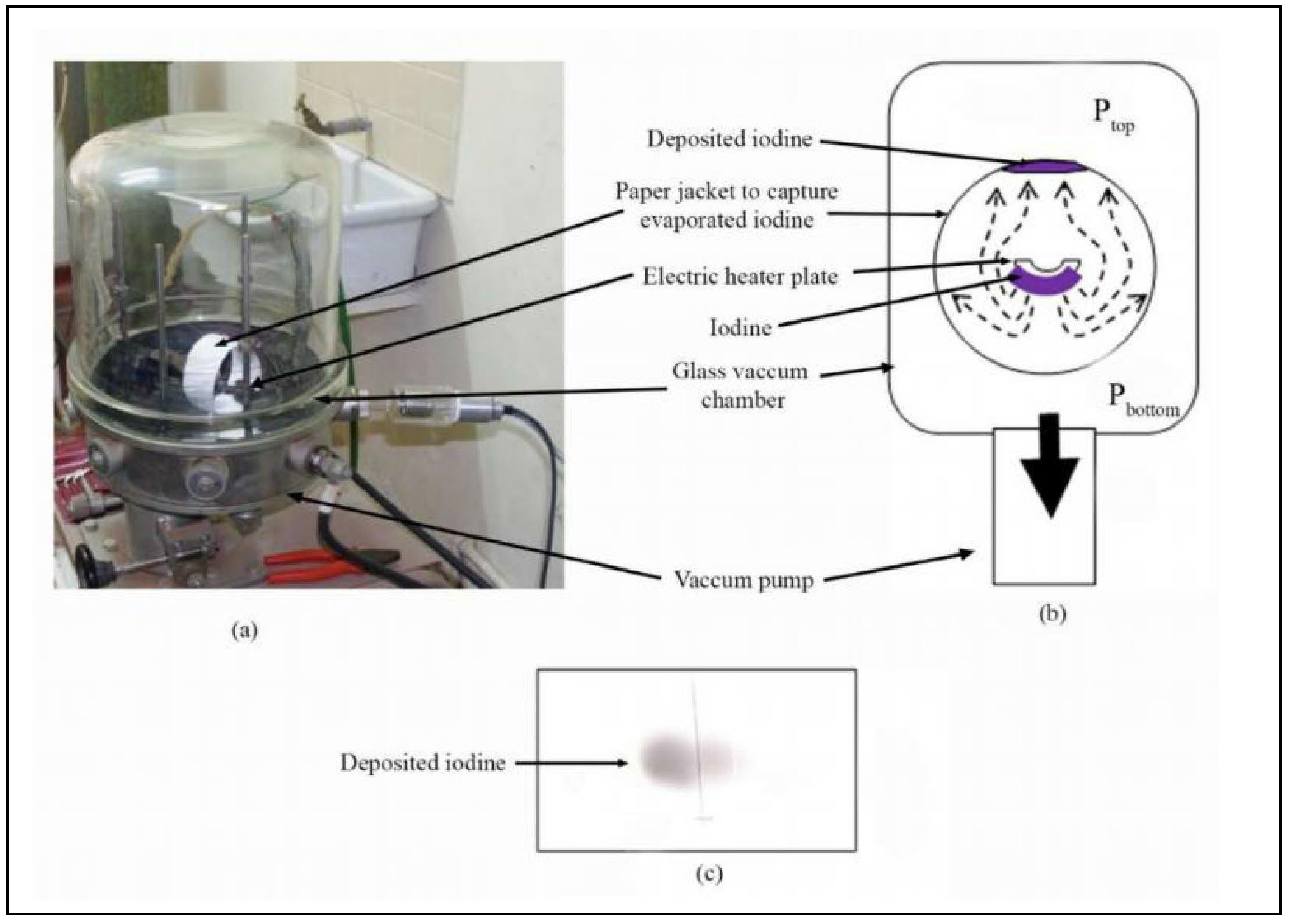
2. Groundbreaking Experiment on Gravitational Repulsion:
- (a)
- Vacuum deposition chamber
- (b)
- A layer of iodine was gradually heat evaporated (ejected in the downward direction) inside the vacuum chamber. The electrical heater plate itself covers the iodine particles from moving directly in the upward direction. The iodine source was surrounded with a paper jacket in order to capture the deposition geometry of iodine. The paper was placed 50 mm radially away from the iodine source. Pressure in the chamber was ~ 1x10 –5 mbar, average mean free path is greater than 6.6 m and air density was approximately 12.6 ng m –3. Pressure at the top () of the chamber was higher than the bottom (); >
- (c)
- Photograph of deposited iodine on inner top part of the paper.
2.1. Takeaways from the Above Iodine Experiment:
- 1)
- There is a repulsive force acting on iodine particles, in the direction opposite to the Earth’s gravitational force.
- 2)
- The repulsive force is dependent on the thermal energy of the particle (as observed by the temperature T, which is a manifestation of the thermal energy Q).
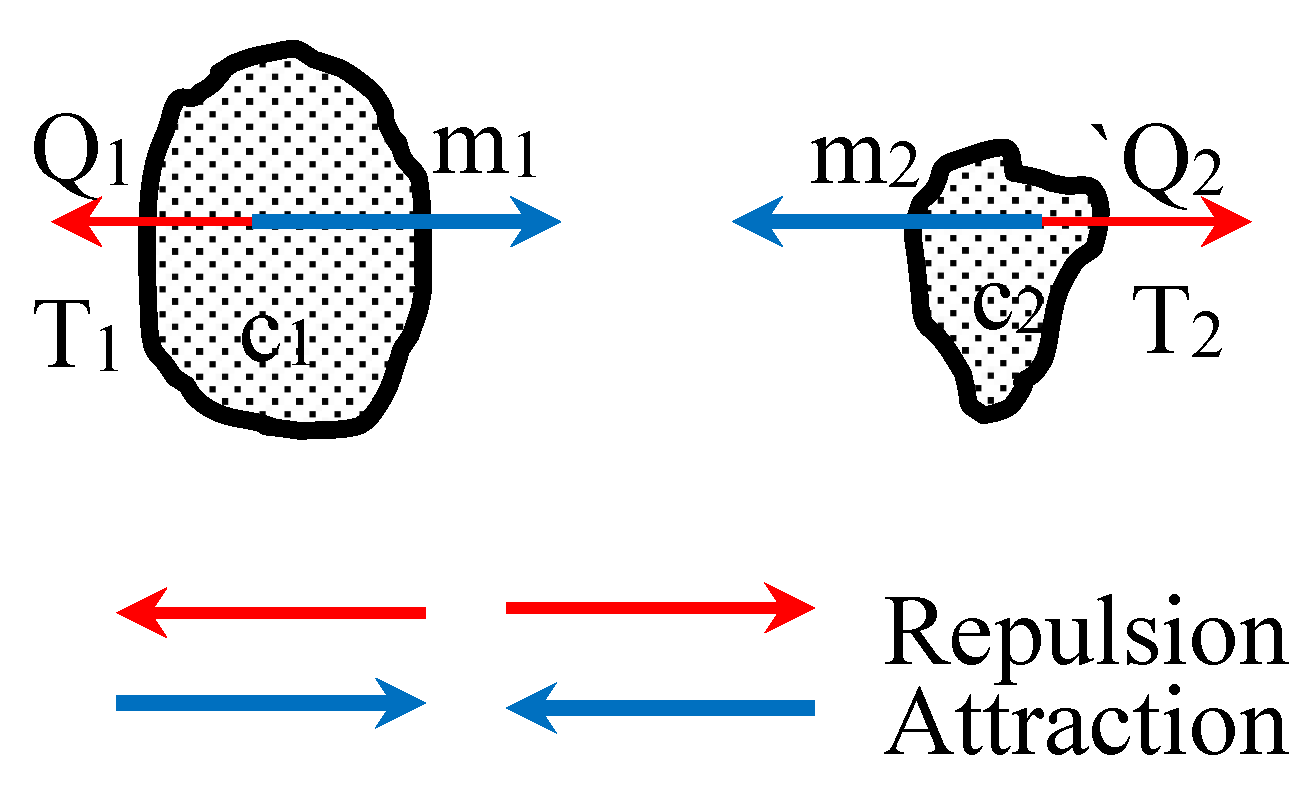
2.2. Forces Between Two Entities of Matter:
- 1)
- Based on the conventional gravitational law:
- 2)
- Experimental findings presented in author’s publication [2] show that:
- 1)
- The model could be applied to explain certain natural phenomena observed in the atmosphere, some of which have been studied in the previous experiments in this research program [1].
- 2)
- Forces at the micro level (between gas molecules) may be generalized to the macro level to explain the behavior of the universe.
3. Alternative Mathematical Model for the Gravitational Repulsion and Attraction Forces, Yielding Behavior of Gases:
3.1. Introduction to the Analysis:
- a)
- A mathematical model for the gravitational repulsion force
- b)
- A revised mathematical model for the gravitational attraction force
- c)
- Orders of magnitude of both gravitational repulsion coefficient and gravitational attraction coefficient
- d)
- Orders of magnitude of both gravitational repulsion and gravitational attraction forces
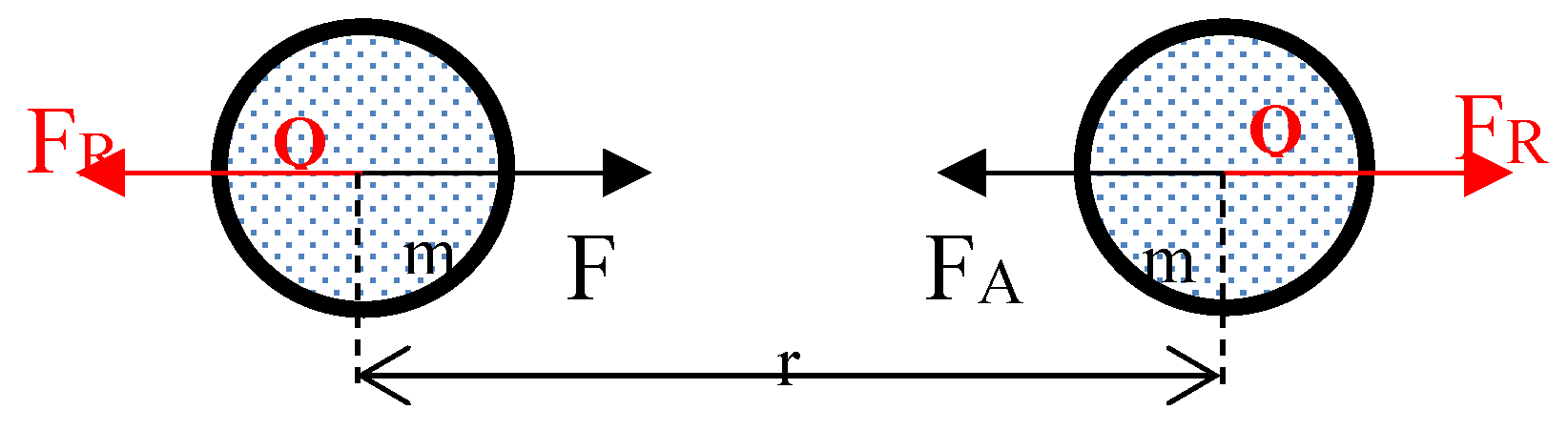
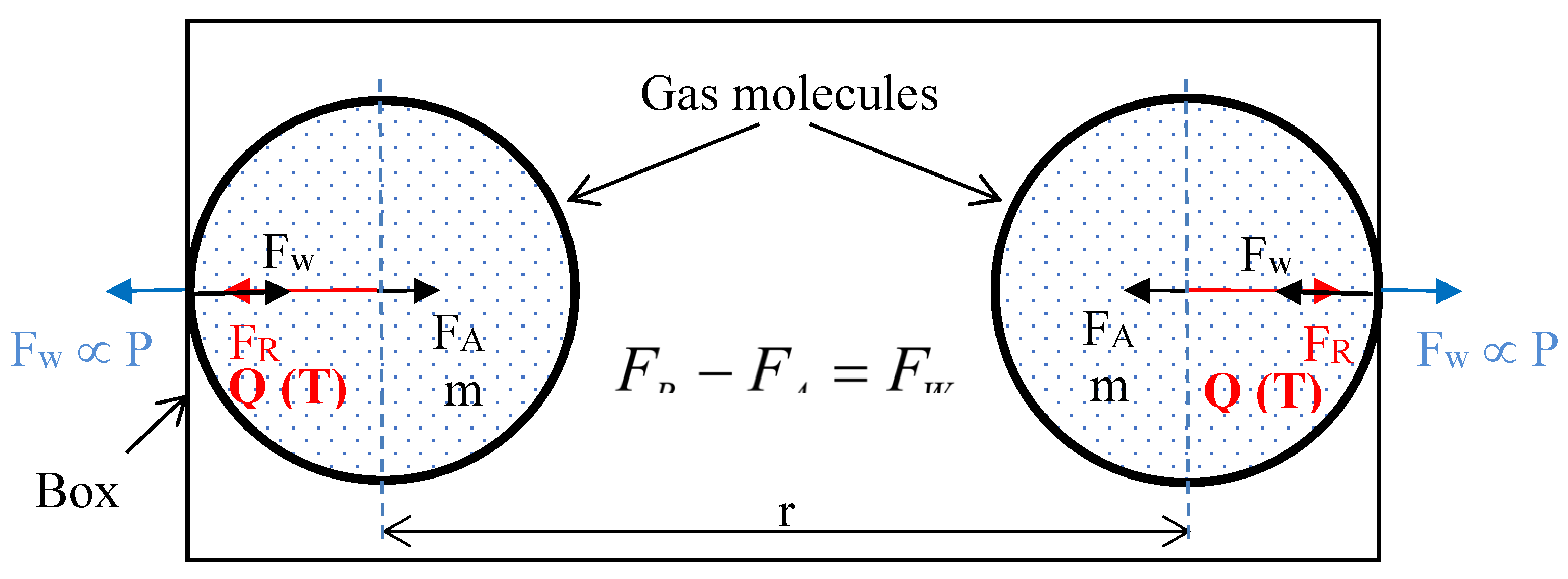
3.2. Forces Between Two Gas Molecules:
3.3. Mathematical Model for Gravitational Repulsion and Attraction Coefficient:
- 1
- The value of x
- 2
- The value of y
- 3
-
The orders of magnitude of the relevant coefficients:
- a.
- GR – Gravitational Repulsion Coefficient
- b.
- GA – Gravitational Attraction Coefficient
- 4
-
The orders of magnitude of the forces:
- a.
- FR – Gravitational Repulsion Force
- b.
- FA – Gravitational Attraction Force
3.4. Building Relationships to Derive x, y, GR, GA, FR and FA:
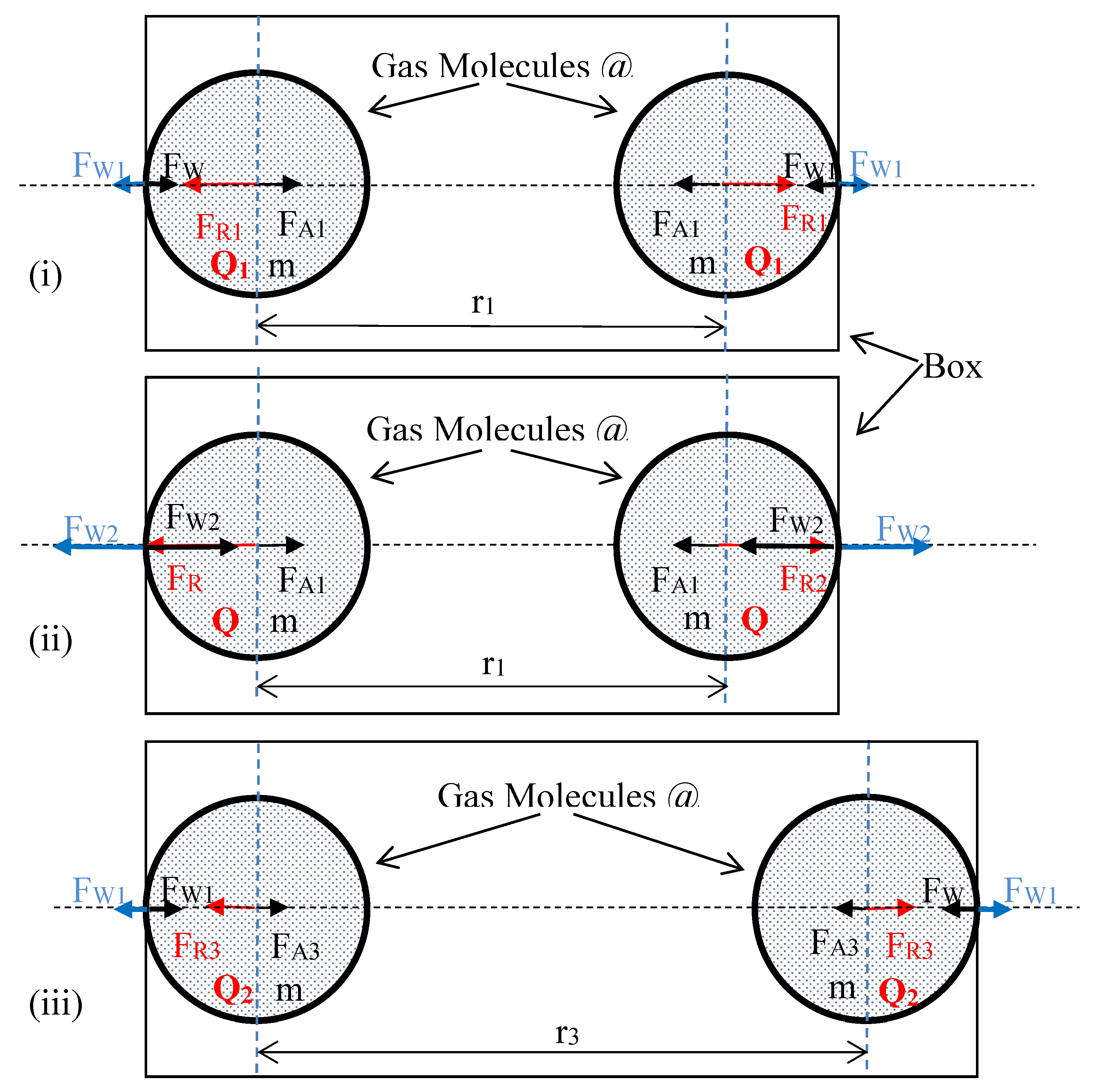
- (i)
- Thermal energy of each molecule is Q1 (corresponding temperature is T1) and the distance between them is r1. The molecules apply the force FW1 on the wall.
- (ii)
- Thermal energy of each molecule is Q2, Q2 > Q1 (corresponding temperature is T2, T2 > T1). Both molecules are at the temperature T2, and the distance r1 between them is kept unchanged, hence, applying the force FW2, FW2> FW1 on the wall.
- (iii)
- Thermal energy of each molecule is Q2 (corresponding temperature is T2). The distance between the two molecules is r3, in order that, the resulting force applying on the wall is equal to FW1.
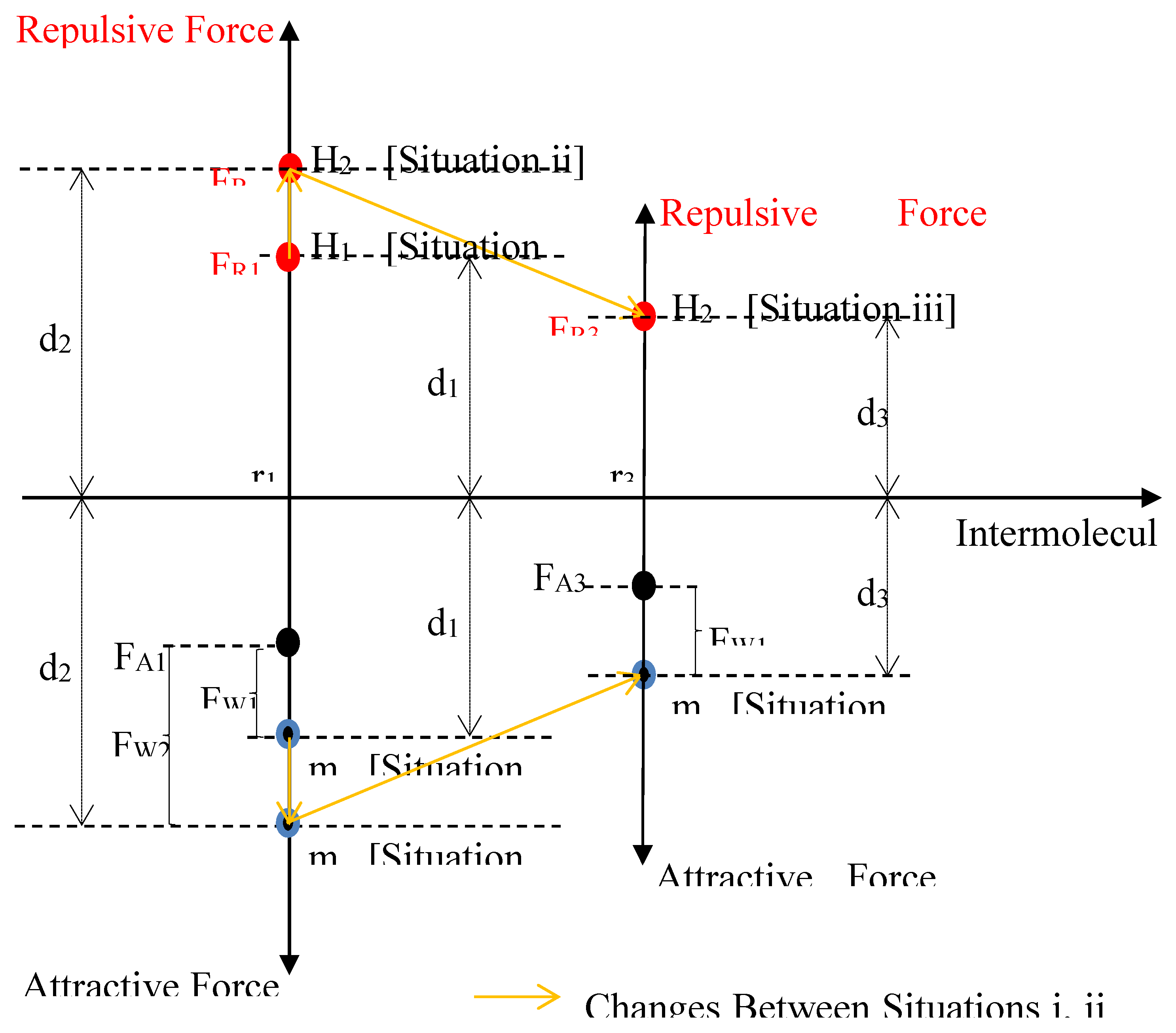
- (i)
- The two molecules at temperature T1 and distance r1 apart. Force on the wall is FW1. Sum of forces FA1 and FW1 is equal to FR1 (indicated as the distance d1).
- (ii)
- The two molecules at temperature T2 and distance r1 apart. Force on the wall is FW2. Sum of forces FA1 and FW2 is equal to FR2 (indicated as the distance d2).
- (iii)
- The two molecules at temperature T2 and distance r3 apart. Force on the wall is FW1. Sum of forces FA3 and FW1 is equal to FR3 (indicated as the distance d3).
3.5. Translating Intermolecular Force to Pressure:
3.6. Solving for Parameters x, GR, GA, FR and FA:
4. Calculation of x, y, GR, GA, FR and FA Based on Thermodynamic Properties of Gas:
4.1. Determining Values of P and r as T Varies:
4.2. Observations of Characteristic Behaviors of x, GR, GA, FR and FA:
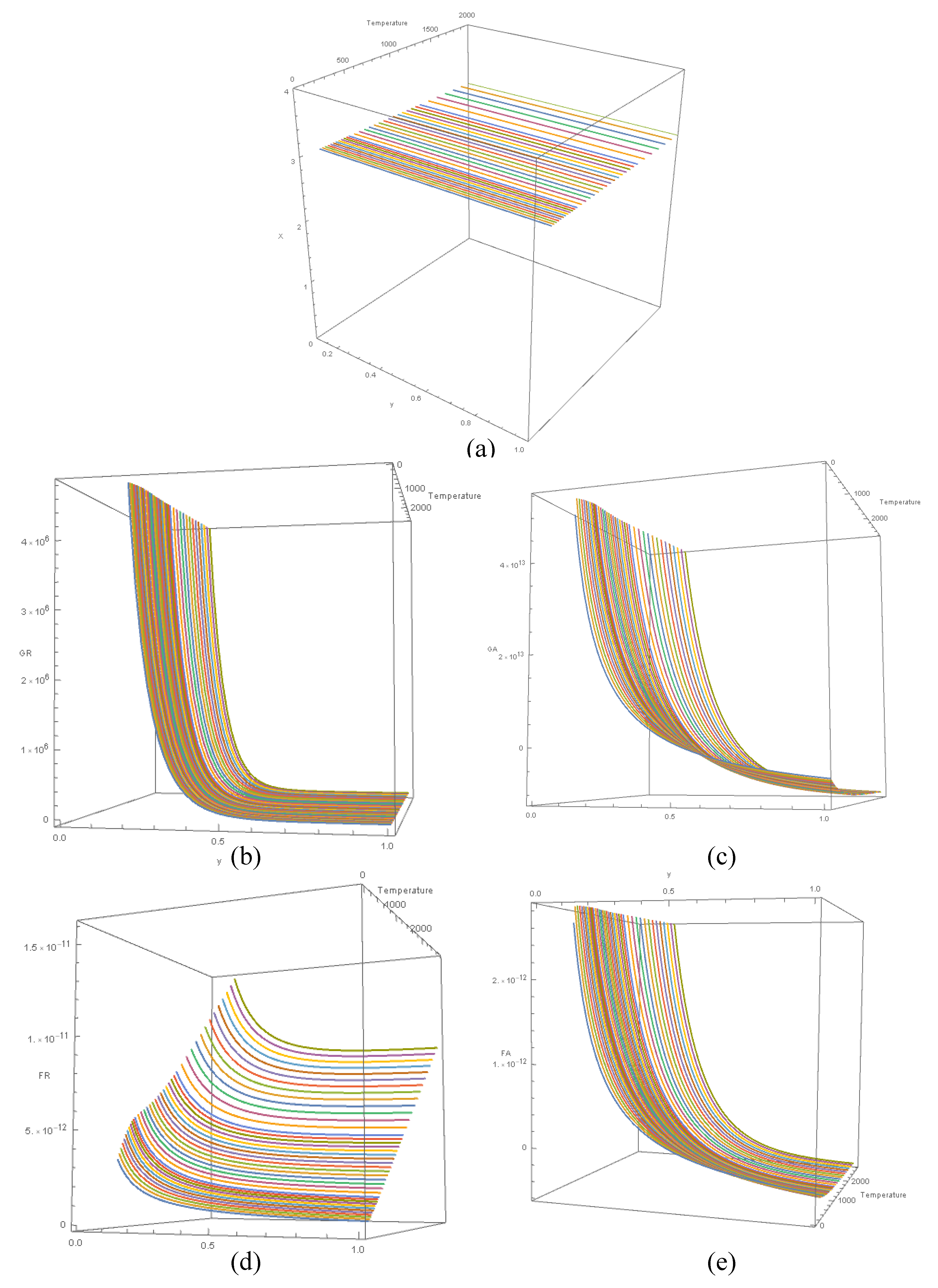
- (a)
- Variation of x relative to T and y. Value of x remains constant at 3 regardless of T and y.
- (b)
-
Variation of GR relative to T and y. GR remains positive over the entire range of T and y. For any given value T:
- i.
- When y < 0.5, as y decreases, the rate of change of GR is very high.
- ii.
- When y > 0.5, as y increases, GR is much stable, compared when y values are low.
- (c)
-
Variation of GA relative to T and y. For any given value T:
- i.
- When y < 0.5, GA is positive, and the rate of change of GA vs. y is high.
- ii.
- When y > 0.5, GA is negative, and the rate of change of GA vs. y is comparatively low.
- (d)
-
Variation of FR relative to T and y. FR is linearly proportionate to T at any given value of y. Gradient of the linear relationship FR vs. T is greater when y < 0.5 compared to when y > 0.5. Further, in the linear relationship FR vs. T:
- i.
- When y < 0.5, intercept > 0
- ii.
- When y ≈ 0.5, intercept ≈ 0
- iii.
- When y > 0.5, intercept < 0
- (e)
-
Variation of FA relative to T and y.
- i.
- When y < 0.5, FA is positive, and the rate of change of FA vs. y is high.
- ii.
- When y > 0.5, FA is negative, and the rate of change of FA vs. y is comparatively low.
- iii.
- Hence, when y ≈ 0.5, FA changes polarity.
- -
- When y < 0.5, the rate of change of both GR and GA vs. y is high as y decreases.
- -
- When y > 0.5, both GR and GA are comparatively stable as y increases.
- -
- For any given value y, variation of GA vs. T appears significant; comparatively more than GR. In classical models of gravitational forces on matter, the dependency of GA on temperature has not been known; Equation 1 is not dependent on temperature. However, the above analysis for gaseous form of matter shows otherwise. Therefore, FA also depends on T (Equation 24).
- FR is linearly proportionate to T at any given value of y (Figure 7(d)); thus, vindicating the mathematical model Equation 5. Significantly, this result confirms the findings presented in a previous paper [2] of the series of publications emanating from this research program (see Supplementary Figure S1). It states therein, [2] p148:
4.3. The Significance x = 3.0, Inverse Proportionality to the Cube of the Distance:
4.4. The Significance of y = 0.5:
4.5. Units of GR, GA, FR and FA:
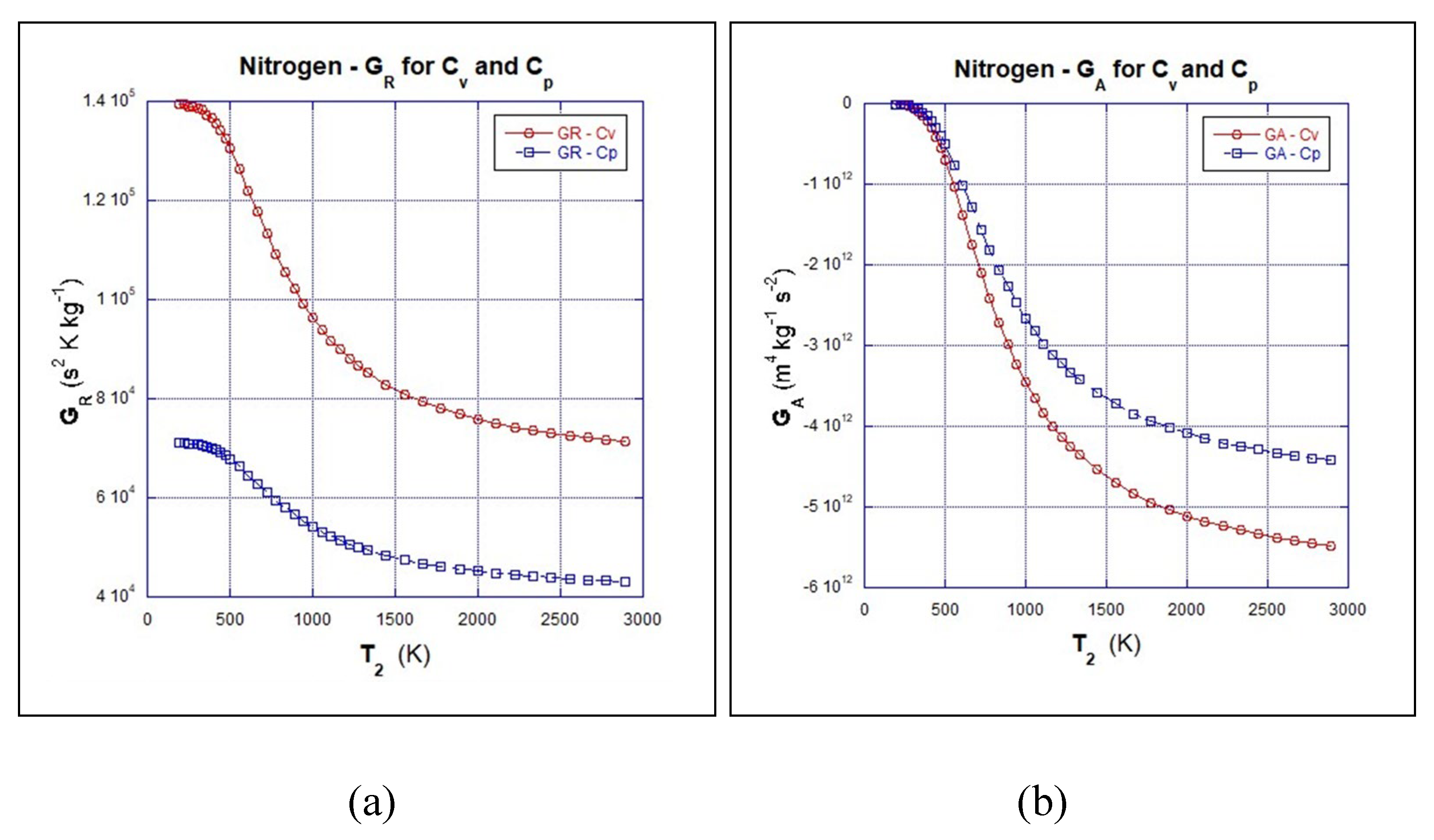
4.6. Orders of the Magnitude of GR and GA:
- (a)
- Results of GR from calculations using cv and cp
- (b)
- Results of GA from calculations using cv and cp
4.7. Orders of the Magnitude of FR and FA:
4.8. Negative Gravitational Attraction Forces Among Gas Molecules:
4.9. The Gist of the Model and the Outcomes:
5. Discussion
- extrapolation of graphs FR vs. T crosses (0,0)
- negative FA tends to become positive as T approaches 0 K
- positive value of FR increases
- negative value of FA increases
In addition to attractive and repulsive forces of water-droplets of a cloud with earth, there exist attractive and repulsive forces among water droplets within the cloud. These forces acting inside the cloud explain the accumulative (flocking together) nature of the cloud which has not been explained by the classical theories. The equilibrium of these two forces will confine the droplets to a certain area as a floccule. The repulsiveness does not allow shrinking and finally collapsing the cloud while the attractive force keeps the droplets together without dispersion. [1] p4
- Heavy gas molecules (such as CFC) in the upper atmosphere
- Brownian Motion
- Condensation/evaporation/sublimation
- Expansion/contraction of gas/liquid/solid
- and more
6. Conclusions
Supplementary Materials
Availability of data and material
Code availability
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piyadasa, C.K.G. Advances of High Energy Physics, Special issue: Dark Matter and Dark Energy in General Relativity and Modified Theories of Gravity 2020, 1 (2020). [CrossRef]
- Piyadasa, C.K.G. Physics Essays 32, 10 (2019). [CrossRef]
- J. H. Keenan and J. Kaye, Gas Tables: Thermodynamic Properties of Air, Products of Combustion and Component Gases, Compressible Flow Function Including Those of Ascher H. Shapiro and Gilbert M. Edelman (J. Wiley, 1948).
- Piyadasa, C.K.G. Canadian Journal of Pure and Applied Sciences 5, 1715 (2011).
- Piyadasa, C.K.G. Canadian Journal of Pure and Applied Sciences 6, 1991 (2012).
- M. Villata, EPL (Europhysics Letters) 94, 20001 (2011). [CrossRef]
- Wanas, M. The Other Side of Gravity and Geometry: Antigravity and Anticurvature. Advances in High Energy Physics 2012, 2012, 752613. [Google Scholar] [CrossRef]
- G. R. Fowles, Introduction to Modern Optics (Dover Publications, New York, 1975).
- V. K. Oikonomou and N. Karagiannakis, Journal of Gravity 2014, 625836 (2014). [CrossRef]
- Newton, A. Motte, and J. Machin, The Mathematical Principles of Natural Philosophy (B. Motte, 1729), v. 1.
- Einstein and R., W. Lawson, Relativity: The Special and the General Theory (Crown Trade Paperbacks, 1961).
- Park, R.S.; Folkner, W.M.; Konopliv, A.S.; Williams, J.G.; Smith, D.E.; Zuber, M.T. Precession of Mercury’s Perihelion from Ranging to the MESSENGER Spacecraft. Astron. J. 2017, 153, 121. [Google Scholar] [CrossRef]
- M. Maggiore, Gravitational Waves: Volume 1: Theory and Experiments (OUP Oxford, 2008).
- P. Schneider, J. Ehlers, and E. E. Falco, Gravitational Lenses (Springer, 1999).
- W. Cowen-Breen, Effects of Gravitational Time Dilation in the Apparent Accelerated Expansion of the Universe (CreateSpace Independent Publishing Platform, 2018). [CrossRef]
- P. J. E. Peebles and B. Ratra, Reviews of Modern Physics 75, 559 (2003). [CrossRef]
- R. R. Caldwell, Physics Letters B 545, 23 (2002). [CrossRef]
- S. Weinberg, Reviews of Modern Physics 61, 1 (1989). [CrossRef]
- R. J. Adler, B. Casey, and O. C. Jacob, American Journal of Physics 63, 620 (1995). [CrossRef]
- S. Hossenfelder, Experimental Search for Quantum Gravity (Springer International Publishing, 2017), p.^pp. 109-120.
- S. Carroll, S. M. Carroll, and Addison-Wesley, Spacetime and Geometry: An Introduction to General Relativity (Addison Wesley, 2004).
- P. Davies, C. SANDY, P. C. W. Davies, and C. BARBISAN, Forces of Nature (Cambridge University Press, 1986).
- N. Gorkavyi and A. Vasilkov, Monthly Notices of the Royal Astronomical Society 461, 2929 (2016). [CrossRef]
- G. C. MCVITTIE, General Relativity and Cosmology, Vol. IV. (Chapman and Hall, London, 1956), 07/28 edn., Vol. 4, The Journal of the Royal Aeronautical Society, 551.
- V. Sahni and A. Krasiński, General Relativity and Gravitation 40, 1557 (2008). [CrossRef]
- C. H. McGruder, Physical Review D 25, 3191 (1982). [CrossRef]
- Z. Göttingen, Nachrichten von der Gesellschaft der Wissenschaften, Mathematisch-Physikalische Klasse (Weidmannsche Buchhandlung, Berlin, 1917).
- V. L. Fitch, C. C. A. P. University, D. R. Marlow, P. University, M. A. E. Dementi, and M. A. E. Dementi, Critical Problems in Physics: Proceedings of a Conference Celebrating the 250th Anniversary of Princeton University, Princeton, New Jersey, October 31, November 1, November 2, 1996 (Princeton University Press, 1997).
- M. Janssen and C. Lehner, The Cambridge Companion to Einstein (Cambridge University Press, 2014), v. 1.
- M. D. Jones, Psience: Easyread Comfort Edition (CREATESPACE PUB, 2009), p.^pp. 432.
- S. Perlmutter et al., The Astrophysical Journal 517, 565 (1999).
- G. Riess et al., The Astronomical Journal 116, 1009 (1998). [CrossRef]
- Chicone and B. Mashhoon, Journal of Mathematical Physics 53, 042501 (2012). [CrossRef]
- J. S. Wang and F. Y. Wang, A&A 564, A137 (2014). [CrossRef]
- K. Bamba, S. i. Nojiri, S. D. Odintsov, and D. Sáez-Gómez, Physical Review D 90, 124061 (2014). [CrossRef]
- P. J. Steinhardt, Philosophical Transactions: Mathematical, Physical and Engineering Sciences 361, 2497 (2003). [CrossRef]
- R. E. Sonntag, C. Borgnakke, and G. J. Van Wylen, Fundamentals of Thermodynamics (Wiley, 2003).
- R. M. Haberle, in Encyclopedia of Atmospheric Sciences (Second Edition), edited by G. R. North, J. Pyle, and F. Zhang (Academic Press, Oxford, 2015), pp. 168.
- S. K. Atreya, P. R. Mahaffy, H. B. Niemann, M. H. Wong, and T. C. Owen, Planetary and Space Science 51, 105 (2003). [CrossRef]
- B. Cambel, D. P. Duclos, and T. P. Anderson, Real Gases (Academic Press, 1963), v. 2.
- E. F. Obert, Concepts of Thermodynamics (McGraw-Hill, 1960).
- I. Burshtein, Introduction to Thermodynamics and Kinetic Theory of Matter (Wiley, 2005).
- T. Vlugt, J. V. Eerden, M. Dijkstra, B. Smit, and D. Frenkel, 2009).
- K. Singh, in Engineered Nanoparticles, edited by A. K. Singh (Academic Press, Boston, 2016), pp. 19.
- K. G. Zloshchastiev, International Journal of Modern Physics B 33, 1950184 (2019). [CrossRef]
- B. G. Sidharth, International Journal of Modern Physics A 13, 2599 (1998). [CrossRef]
- S. Perlmutter et al., Nature 391, 51 (1998). [CrossRef]
- J. A. S. Lima, Brazilian Journal of Physics 34, 194 (2004). [CrossRef]
- Ø. Elgarøy and Ø. Grøn, Entropy 15, 3620 (2013). [CrossRef]
- S. Saha, A. Mondal, and C. Corda, International Journal of Theoretical Physics 57, 1417 (2018). [CrossRef]
- W. Kaiserl. Akademie der Wissenschaften in, 32 v. (vol. 65 (1872).
- B. G. Golovkin, World Scientific News 94, 8 (2018).
- J. L. Gay-Lussac, Annal. Chim. 43 137 (1802).
- S. Chandrasekhar, Reviews of Modern Physics 15, 1 (1943). [CrossRef]
- C. A. de Coulomb, Théorie des machines simples (Librairie scientifique et technique Albert Blanchard, 2002).
- C. V. Boys, Proceedings (Royal Institution of Great Britain, London, 1896), Vol. 14, p.^pp. 355.
- V. N. Popok, in Polymer-Based Multifunctional Nanocomposites and Their Applications, edited by K. Song, C. Liu, and J. Z. Guo (Elsevier, 2019), pp. 35.
- Real Gases—The Effects of Size and Intermolecular Forces. https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_A_Molecular_Approach_(Tro)/05%3A_Gases/5.10%3A_Real_Gases-_The_Effects_of_Size_and_Intermolecular_Forces.
- A. Michaud, International Journal of Engineering Research and Development 7, 16 (2013).
- L. Iorio, Journal of Cosmology and Astroparticle Physics 2010, 018 (2010). [CrossRef]
- Dark Matter May Explain the Puzzling Change in Earth-Sun Distance, (Ref:arxiv.org/abs/1001.1697:). https://www.technologyreview.com/2010/01/14/206662/dark-matter-may-explain-the-puzzling-change-in-earth-sun-distance/.
- B. P. Abbott et al., Nature 551, 85 (2017).
| Interaction or Force | Relative Strength |
Range (m) | Existing Models |
|---|---|---|---|
| Weak (Subatomic) | 1025 | 10 –18 | Electroweak Theory |
| Strong (Subatomic) | 1038 | 10 –15 | Quantum Chromodynamics |
| Electromagnetic | 1036 | ꝏ | Quantum Electrodynamics |
| Gravitational | 1 | ꝏ | General Relativity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




